Submitted:
13 October 2024
Posted:
14 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
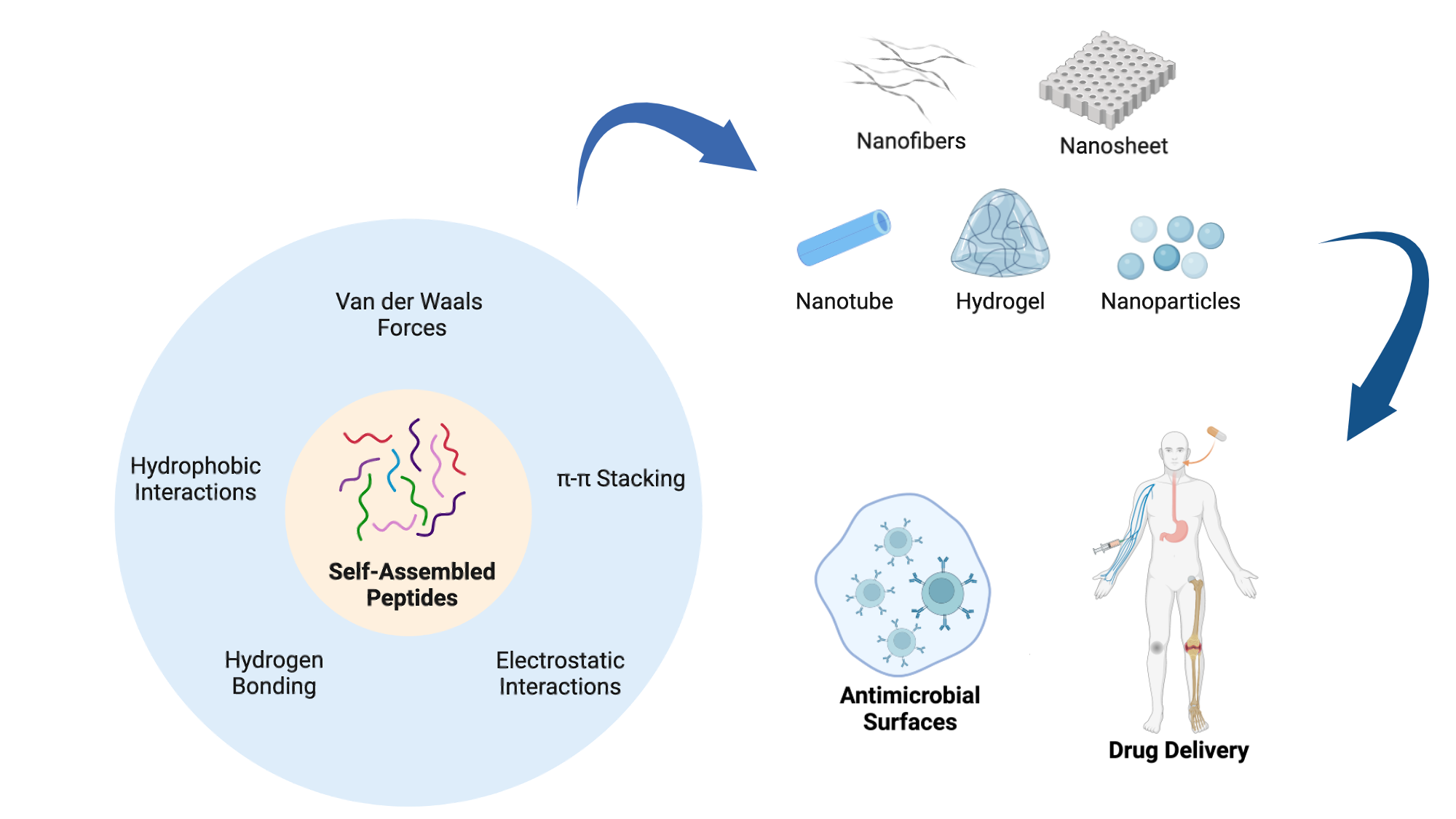
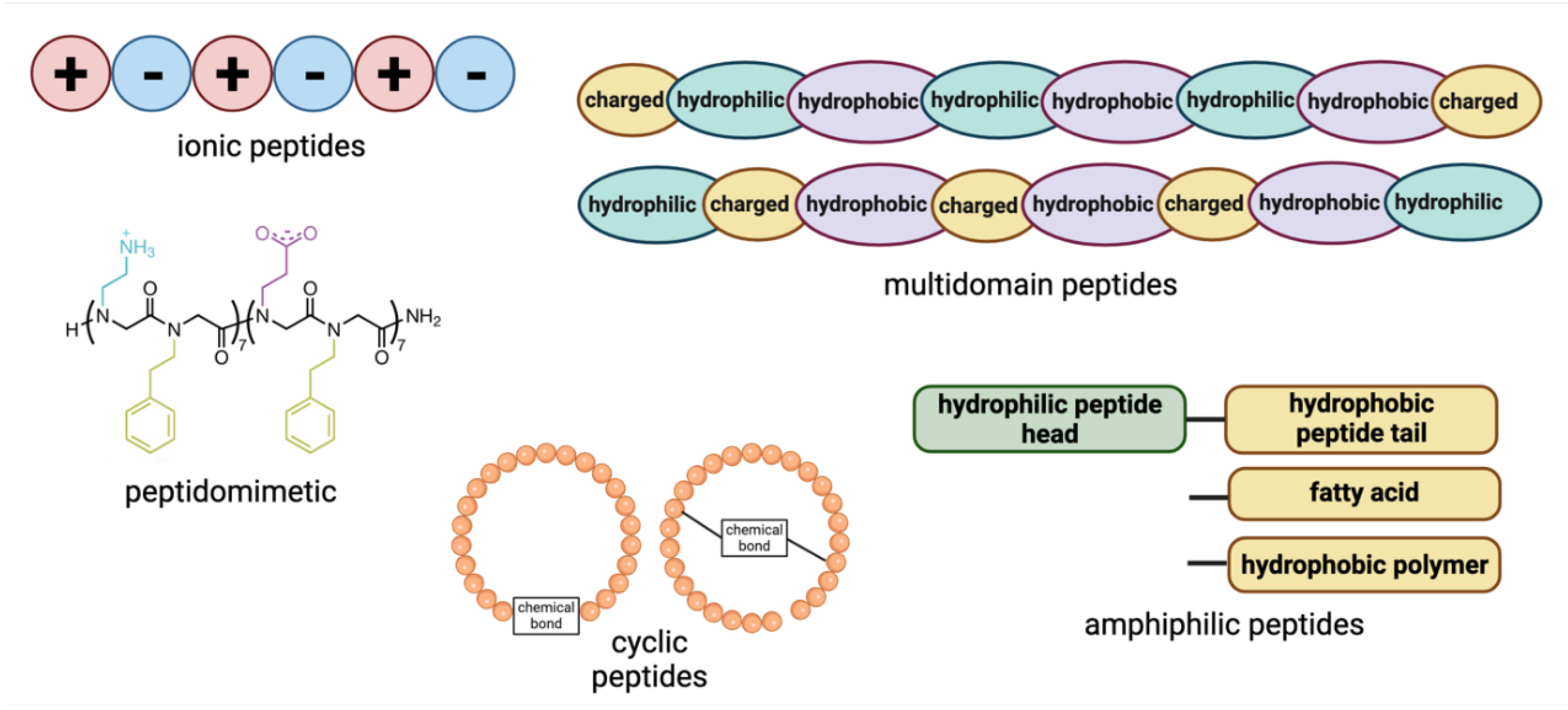
2. Classification of Self-Assembled Peptides
2.1. Ionic Peptides
2.2. Amphiphilic Peptides
2.3. Peptidomimetics
2.4. Cyclic Peptides
2.5. Multidomain Peptides
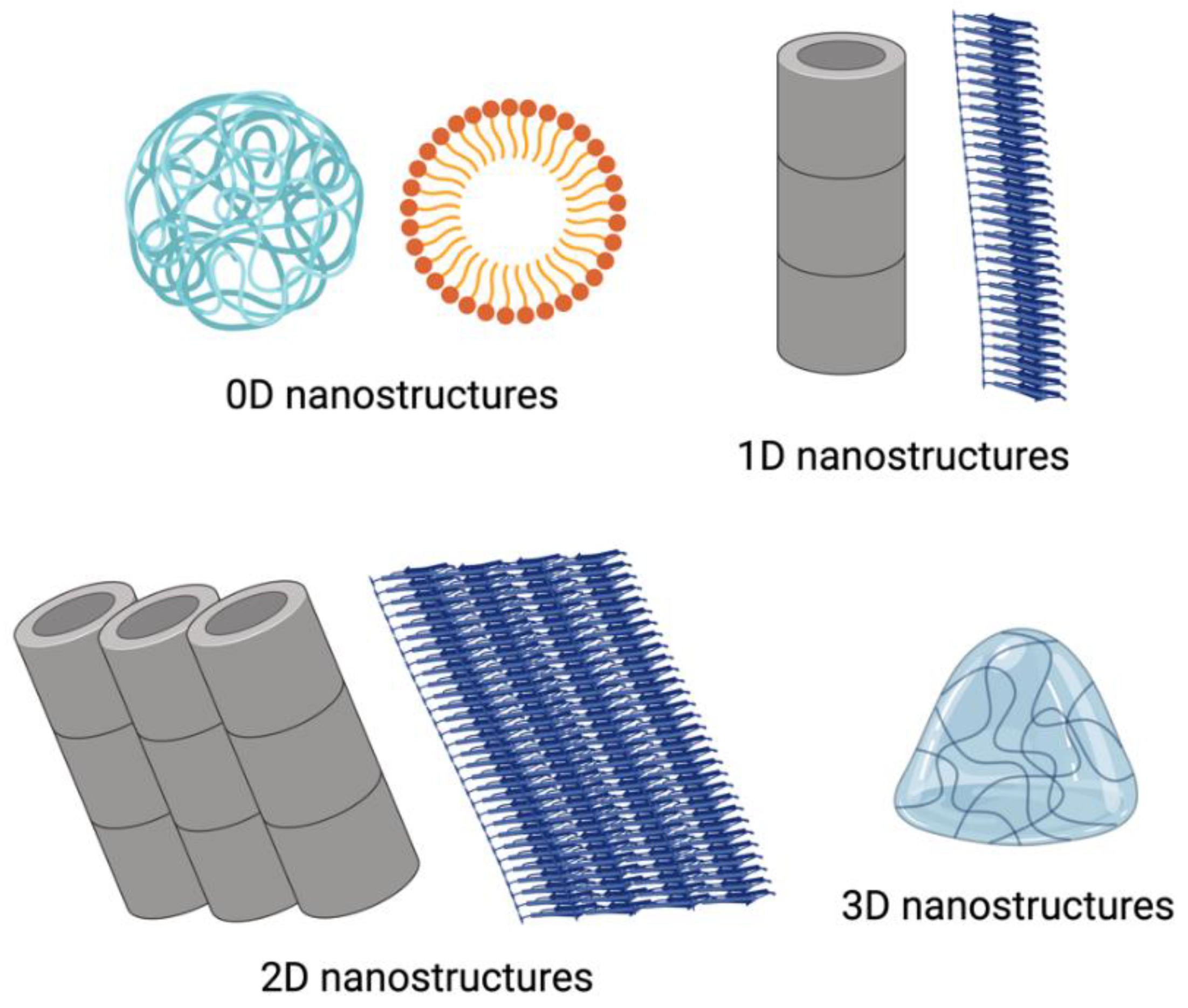
3. Different Types of Self-Assembled Nanostructures
4. Hybrid Materials
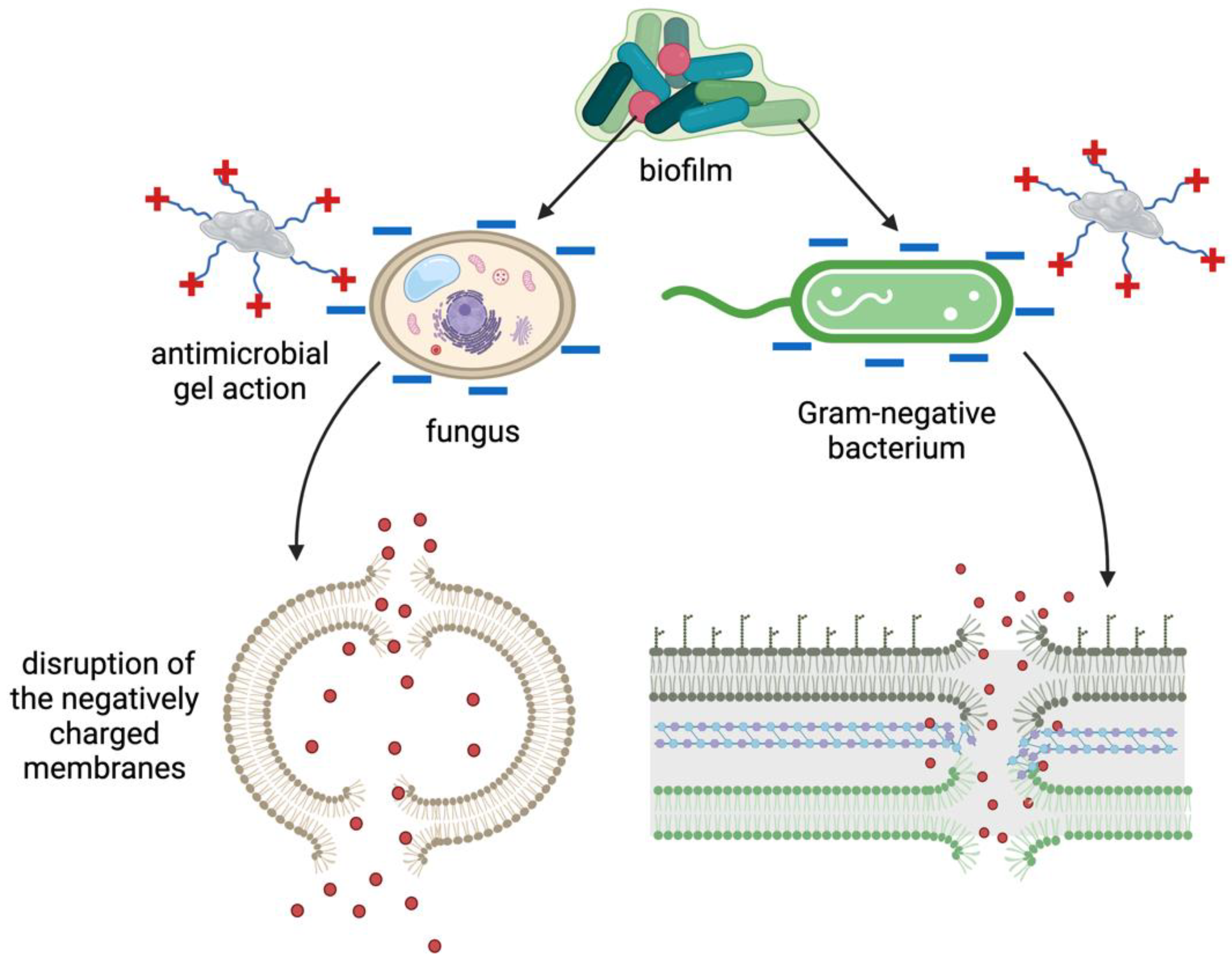
5. Application of Peptide Materials to Prevent Biofilm-Associated Infections
5.1. Peptide-Modified Polymers as Promising Antimicrobial Materials
5.2. Antimicrobial Peptide-Based Hydrogels
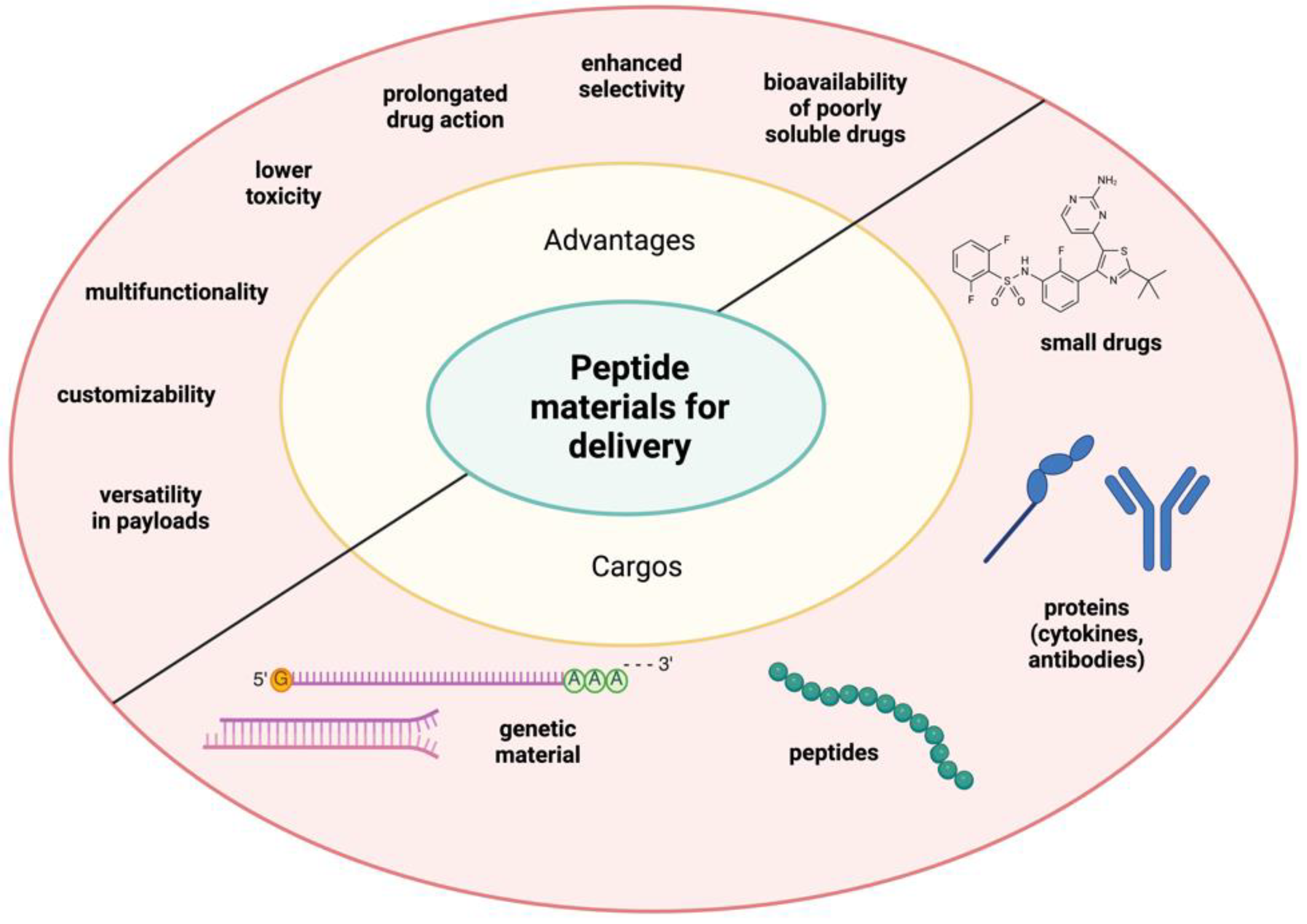
6. Application of Peptide Materials for Drug Delivery
7. Self-Assembling Peptides as Matrix Mimetics in Pharmaceutical Market and Clinical Trials
8. Sheets of Peptidomimetics
9. Conclusions and Future Vision
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Frokjaer, S.; Otzen, D.E. Protein drug stability: a formulation challenge. Nat. Rev. Drug Discov. 2005, 4, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Schulz, M.D.; Wagener, K.B. Precision Polymers through ADMET Polymerization. Macromol. Chem. Phys. 2014, 215, 1936–1945. [Google Scholar] [CrossRef]
- Perrier, S. 50th Anniversary Perspective: RAFT Polymerization A User Guide. Macromolecules 2017, 50, 7433–7447. [Google Scholar] [CrossRef]
- Wang, Z.; Luo, H.; Wang, H.; Xiao, M.; Jia, H.; Ren, C.; Liu, J. Peptide-Based Supramolecular Therapeutics for Fighting Major Diseases. Adv. Funct. Mater. 2024, 34. [Google Scholar] [CrossRef]
- Yang, J.; An, H.-W.; Wang, H. Self-Assembled Peptide Drug Delivery Systems. ACS Appl. Bio Mater. 2020, 4, 24–46. [Google Scholar] [CrossRef]
- Ghadiri, M.R.; Granja, J.R.; Milligan, R.A.; McRee, D.E.; Khazanovich, N. Self-assembling organic nanotubes based on a cyclic peptide architecture. Nature 1993, 366, 324–327. [Google Scholar] [CrossRef]
- Zhang, S.; Lockshin, C.; Cook, R.; Rich, A. Unusually stable β-sheet formation in an ionic self-complementary oligopeptide. Biopolymers: Original Research on Biomolecules 1994, 34, 663–672. [Google Scholar] [CrossRef]
- Wang, T.-T.; Xia, Y.-Y.; Gao, J.-Q.; Xu, D.-H.; Han, M. Recent Progress in the Design and Medical Application of In Situ Self-Assembled Polypeptide Materials. Pharmaceutics 2021, 13, 753. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Gao, L.; Zhang, X.; Wang, P.; Liu, Y.; Feng, J.; Zhang, C.; Zhao, C.; Zhang, S. “PP-type” self-assembling peptides with superior rheological properties. Nanoscale Advances 2021, 3, 6056–6062. [Google Scholar] [CrossRef]
- Yang, H.; Fung, S.-Y.; Pritzker, M.; Chen, P. Ionic-Complementary Peptide Matrix for Enzyme Immobilization and Biomolecular Sensing. Langmuir 2009, 25, 7773–7777. [Google Scholar] [CrossRef]
- Wan, Z.; Lu, S.; Zhao, D.; Ding, Y.; Chen, P. Arginine-rich ionic complementary peptides as potential drug carriers: Impact of peptide sequence on size, shape and cell specificity. Nanomedicine: Nanotechnology, Biol. Med. 2016, 12, 1479–1488. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Ji, W.; Palmer, L.C.; Weber, B.; Barz, M.; Stupp, S.I. Programmable Assembly of Peptide Amphiphile via Noncovalent-to-Covalent Bond Conversion. J. Am. Chem. Soc. 2017, 139, 8995–9000. [Google Scholar] [CrossRef] [PubMed]
- Epps III, T.H.; O'Reilly, R.K. Block copolymers: controlling nanostructure to generate functional materials–synthesis, characterization, and engineering. Chemical Science 2016, 7, 1674–1689. [Google Scholar] [CrossRef] [PubMed]
- von Maltzahn, G.; Vauthey, S.; Santoso, S.; Zhang, S. Positively Charged Surfactant-like Peptides Self-assemble into Nanostructures. Langmuir 2003, 19, 4332–4337. [Google Scholar] [CrossRef]
- Vauthey, S.; Santoso, S.; Gong, H.; Watson, N.; Zhang, S. Molecular self-assembly of surfactant-like peptides to form nanotubes and nanovesicles. Proc. Natl. Acad. Sci. 2002, 99, 5355–5360. [Google Scholar] [CrossRef]
- Webber, M.J.; Appel, E.A.; Meijer, E.; Langer, R. Supramolecular biomaterials. Nat. Mater. 2016, 15, 13–26. [Google Scholar] [CrossRef]
- Mazza, M.; Notman, R.; Anwar, J.; Rodger, A.; Hicks, M.; Parkinson, G.; McCarthy, D.; Daviter, T.; Moger, J.; Garrett, N.; et al. Nanofiber-Based Delivery of Therapeutic Peptides to the Brain. ACS Nano 2013, 7, 1016–1026. [Google Scholar] [CrossRef]
- Hutchinson, J.A.; Hamley, I.W.; Torras, J.; Alemán, C.; Seitsonen, J.; Ruokolainen, J. Self-Assembly of Lipopeptides Containing Short Peptide Fragments Derived from the Gastrointestinal Hormone PYY3–36: From Micelles to Amyloid Fibrils. J. Phys. Chem. B 2019, 123, 614–621. [Google Scholar] [CrossRef]
- Li, Z.; Lin, Z. Self-Assembly of Bolaamphiphiles into 2D Nanosheets via Synergistic and Meticulous Tailoring of Multiple Noncovalent Interactions. ACS Nano 2021, 15, 3152–3160. [Google Scholar] [CrossRef]
- Nagai, A.; Nagai, Y.; Qu, H.; Zhang, S. Dynamic Behaviors of Lipid-Like Self-Assembling Peptide A6D and A6K Nanotubes. J. Nanosci. Nanotechnol. 2007, 7, 2246–2252. [Google Scholar] [CrossRef]
- Mondal, P.; Dey, J.; Roy, S.; Dasgupta, S.B. Self-Assembly, In Vitro Gene Transfection, and Antimicrobial Activity of Biodegradable Cationic Bolaamphiphiles. Langmuir 2023, 39, 10021–10032. [Google Scholar] [CrossRef] [PubMed]
- Mannige, R.V.; Haxton, T.K.; Proulx, C.; Robertson, E.J.; Battigelli, A.; Butterfoss, G.L.; Zuckermann, R.N.; Whitelam, S. Peptoid nanosheets exhibit a new secondary-structure motif. Nature 2015, 526, 415–420. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Q.; Wong, C.T.T.; Li, X. Chemoselective Peptide Cyclization and Bicyclization Directly on Unprotected Peptides. J. Am. Chem. Soc. 2019, 141, 12274–12279. [Google Scholar] [CrossRef] [PubMed]
- Shirazi, A.N.; Mozaffari, S.; Sherpa, R.T.; Tiwari, R.; Parang, K. Efficient Intracellular Delivery of Cell-Impermeable Cargo Molecules by Peptides Containing Tryptophan and Histidine. Molecules 2018, 23, 1536. [Google Scholar] [CrossRef]
- Galler, K.M.; Aulisa, L.; Regan, K.R.; D’Souza, R.N.; Hartgerink, J.D. Self-Assembling Multidomain Peptide Hydrogels: Designed Susceptibility to Enzymatic Cleavage Allows Enhanced Cell Migration and Spreading. J. Am. Chem. Soc. 2010, 132, 3217–3223. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.N.; Hartgerink, J.D. Self-Assembling Multidomain Peptide Nanofibers for Delivery of Bioactive Molecules and Tissue Regeneration. Accounts Chem. Res. 2017, 50, 714–722. [Google Scholar] [CrossRef]
- Lopez-Silva, T.L.; Leach, D.G.; Li, I.-C.; Wang, X.; Hartgerink, J.D. Self-Assembling Multidomain Peptides: Design and Characterization of Neutral Peptide-Based Materials with pH and Ionic Strength Independent Self-Assembly. ACS Biomater. Sci. Eng. 2018, 5, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Wickremasinghe, N.C.; Kumar, V.A.; Shi, S.; Hartgerink, J.D. Controlled Angiogenesis in Peptide Nanofiber Composite Hydrogels. ACS Biomater. Sci. Eng. 2015, 1, 845–854. [Google Scholar] [CrossRef]
- Yang, X.; Ma, L.; Lu, K.; Zhao, D. Mechanism of Peptide Self-assembly and Its Study in Biomedicine. Protein J. 2024, 43, 464–476. [Google Scholar] [CrossRef]
- La Manna, S.; Di Natale, C.; Onesto, V.; Marasco, D. Self-assembling peptides: From design to biomedical applications. International Journal of Molecular Sciences 2021, 22, 12662. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Hu, T.; Liang, R.; Wei, M. Application of Zero-Dimensional Nanomaterials in Biosensing. Front. Chem. 2020, 8, 320. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, X.; Wan, K.; Zhou, N.; Wei, G.; Su, Z. Supramolecular peptide nano-assemblies for cancer diagnosis and therapy: from molecular design to material synthesis and function-specific applications. J. Nanobiotechnology 2021, 19, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhu, J.; Fang, T.; Li, M.; Chen, G.; Chen, Q. Supramolecular biomaterials for enhanced cancer immunotherapy. J. Mater. Chem. B 2022, 10, 7183–7193. [Google Scholar] [CrossRef]
- Tarvirdipour, S.; Huang, X.; Mihali, V.; Schoenenberger, C.-A.; Palivan, C.G. Peptide-Based Nanoassemblies in Gene Therapy and Diagnosis: Paving the Way for Clinical Application. Molecules 2020, 25, 3482. [Google Scholar] [CrossRef] [PubMed]
- Gatto, E.; Toniolo, C.; Venanzi, M. Peptide Self-Assembled Nanostructures: From Models to Therapeutic Peptides. Nanomaterials 2022, 12, 466. [Google Scholar] [CrossRef]
- Brahmachari, S.; Arnon, Z.A.; Frydman-Marom, A.; Gazit, E.; Adler-Abramovich, L. Diphenylalanine as a Reductionist Model for the Mechanistic Characterization of β-Amyloid Modulators. ACS Nano 2017, 11, 5960–5969. [Google Scholar] [CrossRef]
- Min, K.; Lee, S.; Lee, E.; Lee, Y.; Yi, H.; Kim, D. Facile Nondestructive Assembly of Tyrosine-Rich Peptide Nanofibers as a Biological Glue for Multicomponent-Based Nanoelectrode Applications. Adv. Funct. Mater. 2018, 28. [Google Scholar] [CrossRef]
- Cormier, A.R.; Pang, X.; Zimmerman, M.I.; Zhou, H.-X.; Paravastu, A.K. Molecular Structure of RADA16-I Designer Self-Assembling Peptide Nanofibers. ACS Nano 2013, 7, 7562–7572. [Google Scholar] [CrossRef]
- Merg, A.D.; Touponse, G.; van Genderen, E.; Zuo, X.; Bazrafshan, A.; Blum, T.; Hughes, S.; Salaita, K.; Abrahams, J.P.; Conticello, V.P. 2D Crystal Engineering of Nanosheets Assembled from Helical Peptide Building Blocks. Angew. Chem. Int. Ed. 2019, 58, 13507–13512. [Google Scholar] [CrossRef]
- Dai, B.; Li, D.; Xi, W.; Luo, F.; Zhang, X.; Zou, M.; Cao, M.; Hu, J.; Wang, W.; Wei, G.; et al. Tunable assembly of amyloid-forming peptides into nanosheets as a retrovirus carrier. Proc. Natl. Acad. Sci. 2015, 112, 2996–3001. [Google Scholar] [CrossRef]
- Lee, J.; Choe, I.R.; Kim, N.-K.; Kim, W.-J.; Jang, H.-S.; Lee, Y.-S.; Nam, K.T. Water-Floating Giant Nanosheets from Helical Peptide Pentamers. ACS Nano 2016, 10, 8263–8270. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.B.P.; Gomes, V.; Ferreira, P.M.T.; Martins, J.A.; Jervis, P.J. Peptide-Based Supramolecular Hydrogels as Drug Delivery Agents: Recent Advances. Gels 2022, 8, 706. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, Z.; Han, H.; Xu, Z.; Li, S.; Zhu, Y.; Chen, Y.; Ge, L.; Zhang, Y. Short and simple peptide-based pH-sensitive hydrogel for antitumor drug delivery. Chinese Chemical Letters 2022, 33, 1936–1940. [Google Scholar] [CrossRef]
- Taheri-Ledari, R.; Maleki, A. Antimicrobial therapeutic enhancement of levofloxacin via conjugation to a cell-penetrating peptide: An efficient sonochemical catalytic process. J. Pept. Sci. 2020, 26, e3277. [Google Scholar] [CrossRef]
- Rakesh, K.P.; Suhas, R.; Gowda, D.C. Anti-inflammatory and Antioxidant Peptide-Conjugates: Modulation of Activity by Charged and Hydrophobic Residues. Int. J. Pept. Res. Ther. 2017, 25, 227–234. [Google Scholar] [CrossRef]
- Hermanson, G.T. Bioconjugate Techniques; Academic press: 2013.
- Alves, P.M.; Barrias, C.C.; Gomes, P.; Martins, M.C.L. How can biomaterial-conjugated antimicrobial peptides fight bacteria and be protected from degradation? Acta Biomaterialia 2024, 181, 98–116. [Google Scholar] [CrossRef] [PubMed]
- Fisher, S.A.; Baker, A.E.; Shoichet, M.S. Designing Peptide and Protein Modified Hydrogels: Selecting the Optimal Conjugation Strategy. J. Am. Chem. Soc. 2017, 139, 7416–7427. [Google Scholar] [CrossRef]
- Donlan, R.M. Biofilms and Device-Associated Infections. Emerg. Infect. Dis. 2001, 7, 277–281. [Google Scholar] [CrossRef]
- Mah, T.-F. Biofilm-Specific Antibiotic Resistance. Futur. Microbiol. 2012, 7, 1061–1072. [Google Scholar] [CrossRef]
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. Journal of the Chinese Medical Association 2018, 81, 7–11. [Google Scholar] [CrossRef]
- Caliari, S.R.; Burdick, J.A. A practical guide to hydrogels for cell culture. Nat. Methods 2016, 13, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Galdiero, E.; Lombardi, L.; Falanga, A.; Libralato, G.; Guida, M.; Carotenuto, R. Biofilms: Novel Strategies Based on Antimicrobial Peptides. Pharmaceutics 2019, 11, 322. [Google Scholar] [CrossRef]
- Lombardi, L.; Shi, Y.; Falanga, A.; Galdiero, E.; de Alteriis, E.; Franci, G.; Chourpa, I.; Azevedo, H.S.; Galdiero, S. Enhancing the Potency of Antimicrobial Peptides through Molecular Engineering and Self-Assembly. Biomacromolecules 2019, 20, 1362–1374. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, L.; Stellato, M.I.; Oliva, R.; Falanga, A.; Galdiero, M.; Petraccone, L.; D’errico, G.; De Santis, A.; Galdiero, S.; Del Vecchio, P. Antimicrobial peptides at work: interaction of myxinidin and its mutant WMR with lipid bilayers mimicking the P. aeruginosa and E. coli membranes. Sci. Rep. 2017, 7, srep44425. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Wu, T.; Wang, W.; Li, B.; Wang, M.; Chen, L.; Xia, H.; Zhang, T. Biofunctions of antimicrobial peptide-conjugated alginate/hyaluronic acid/collagen wound dressings promote wound healing of a mixed-bacteria-infected wound. Int. J. Biol. Macromol. 2019, 140, 330–342. [Google Scholar] [CrossRef]
- Rezaei, N.; Hamidabadi, H.G.; Khosravimelal, S.; Zahiri, M.; Ahovan, Z.A.; Bojnordi, M.N.; Eftekhari, B.S.; Hashemi, A.; Ganji, F.; Darabi, S.; et al. Antimicrobial peptides-loaded smart chitosan hydrogel: Release behavior and antibacterial potential against antibiotic resistant clinical isolates. Int. J. Biol. Macromol. 2020, 164, 855–862. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Jin, Y.; Wang, X.; Yao, S.; Li, Y.; Wu, Q.; Ma, G.; Cui, F.; Liu, H. An antimicrobial peptide-loaded gelatin/chitosan nanofibrous membrane fabricated by sequential layer-by-layer electrospinning and electrospraying techniques. Nanomaterials 2018, 8, 327. [Google Scholar] [CrossRef]
- Hozumi, K.; Nomizu, M. Mixed Peptide-Conjugated Chitosan Matrices as Multi-Receptor Targeted Cell-Adhesive Scaffolds. Int. J. Mol. Sci. 2018, 19, 2713. [Google Scholar] [CrossRef]
- Wang, C.; Hong, T.; Cui, P.; Wang, J.; Xia, J. Antimicrobial peptides towards clinical application: Delivery and formulation. Adv. Drug Deliv. Rev. 2021, 175, 113818. [Google Scholar] [CrossRef]
- Sahariah, P.; Sorensen, K.K.; Hjalmarsdottir, M.A.; Sigurjonsson, O.E.; Jensen, K.J.; Masson, M.; Thygesen, M.B. Antimicrobial peptide shows enhanced activity and reduced toxicity upon grafting to chitosan polymers. Chem. Commun. 2015, 51, 11611–11614. [Google Scholar] [CrossRef]
- Barbosa, M.; Costa, F.; Monteiro, C.; Duarte, F.; Martins, M.C.L.; Gomes, P. Antimicrobial coatings prepared from Dhvar-5-click-grafted chitosan powders. Acta Biomater. 2018, 84, 242–256. [Google Scholar] [CrossRef] [PubMed]
- Alves, P.M.; Pereira, R.F.; Costa, B.; Tassi, N.; Teixeira, C.; Leiro, V.; Monteiro, C.; Gomes, P.; Costa, F.; Martins, M.C.L. Thiol–Norbornene Photoclick Chemistry for Grafting Antimicrobial Peptides onto Chitosan to Create Antibacterial Biomaterials. ACS Appl. Polym. Mater. 2022, 4, 5012–5026. [Google Scholar] [CrossRef]
- Hilpert, K.; Elliott, M.; Jenssen, H.; Kindrachuk, J.; Fjell, C.D.; Körner, J.; Winkler, D.F.; Weaver, L.L.; Henklein, P.; Ulrich, A.S.; et al. Screening and Characterization of Surface-Tethered Cationic Peptides for Antimicrobial Activity. Chem. Biol. 2009, 16, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Cui, Z.; Luo, Q.; Bannon, M.S.; Gray, V.P.; Bloom, T.G.; Clore, M.F.; Hughes, M.A.; Crawford, M.A.; Letteri, R.A. Molecular engineering of antimicrobial peptide (AMP)–polymer conjugates. Biomater. Sci. 2021, 9, 5069–5091. [Google Scholar] [CrossRef]
- Lozeau, L.D.; Grosha, J.; Kole, D.; Prifti, F.; Dominko, T.; Camesano, T.A.; Rolle, M.W. Collagen tethering of synthetic human antimicrobial peptides cathelicidin LL37 and its effects on antimicrobial activity and cytotoxicity. Acta Biomater. 2017, 52, 9–20. [Google Scholar] [CrossRef]
- Galdiero, E.; Falanga, A.; Siciliano, A.; Maselli, V.; Guida, M.; Carotenuto, R.; Tussellino, M.; Lombardi, L.; Benvenuto, G.; Galdiero, S. Daphnia magna and Xenopus laevis as in vivo models to probe toxicity and uptake of quantum dots functionalized with gH625. Int. J. Nanomed. 2017, ume 12, 2717–2731. [Google Scholar] [CrossRef]
- Galdiero, E.; Siciliano, A.; Maselli, V.; Gesuele, R.; Guida, M.; Fulgione, D.; Galdiero, S.; Lombardi, L.; Falanga, A. An integrated study on antimicrobial activity and ecotoxicity of quantum dots and quantum dots coated with the antimicrobial peptide indolicidin. Int. J. Nanomed. 2016, ume 11, 4199–4211. [Google Scholar] [CrossRef]
- Shkodenko, L.; Kassirov, I.; Koshel, E. Metal Oxide Nanoparticles Against Bacterial Biofilms: Perspectives and Limitations. Microorganisms 2020, 8, 1545. [Google Scholar] [CrossRef]
- Fonseca, D.R.; Moura, A.; Leiro, V.; Silva-Carvalho, R.; Estevinho, B.N.; Seabra, C.L.; Henriques, P.C.; Lucena, M.; Teixeira, C.; Gomes, P.; et al. Grafting MSI-78A onto chitosan microspheres enhances its antimicrobial activity. Acta Biomater. 2021, 137, 186–198. [Google Scholar] [CrossRef]
- Azoulay, Z.; Aibinder, P.; Gancz, A.; Moran-Gilad, J.; Navon-Venezia, S.; Rapaport, H. Assembly of cationic and amphiphilic β-sheet FKF tripeptide confers antibacterial activity. Acta Biomater. 2021, 125, 231–241. [Google Scholar] [CrossRef]
- Guo, Z.; Wang, Y.; Tan, T.; Ji, Y.; Hu, J.; Zhang, Y. Antimicrobial D-peptide hydrogels. ACS Biomaterials Science & Engineering 2021, 7, 1703–1712. [Google Scholar]
- Cao, F.; Mei, L.; Zhu, G.; Song, M.; Zhang, X. An injectable molecular hydrogel assembled by antimicrobial peptide PAF26 for antimicrobial application. RSC Adv. 2019, 9, 30803–30808. [Google Scholar] [CrossRef]
- Adak, A.; Ghosh, S.; Gupta, V.; Ghosh, S. Biocompatible lipopeptide-based antibacterial hydrogel. Biomacromolecules 2019, 20, 1889–1898. [Google Scholar] [CrossRef]
- Hartgerink, J.D.; Beniash, E.; Stupp, S.I. Self-Assembly and Mineralization of Peptide-Amphiphile Nanofibers. Science 2001, 294, 1684–1688. [Google Scholar] [CrossRef] [PubMed]
- Klok, H.-A. Peptide/protein− synthetic polymer conjugates: quo vadis. Macromolecules 2009, 42, 7990–8000. [Google Scholar] [CrossRef]
- Artar, M.; Souren, E.R.J.; Terashima, T.; Meijer, E.W.; Palmans, A.R.A. Single Chain Polymeric Nanoparticles as Selective Hydrophobic Reaction Spaces in Water. ACS Macro Lett. 2015, 4, 1099–1103. [Google Scholar] [CrossRef]
- Kumar, V.B.; Ozguney, B.; Vlachou, A.; Chen, Y.; Gazit, E.; Tamamis, P. Peptide Self-Assembled Nanocarriers for Cancer Drug Delivery. J. Phys. Chem. B 2023, 127, 1857–1871. [Google Scholar] [CrossRef]
- Nhàn, N.T.T.; Yamada, T.; Yamada, K.H. Peptide-Based Agents for Cancer Treatment: Current Applications and Future Directions. Int. J. Mol. Sci. 2023, 24, 12931. [Google Scholar] [CrossRef]
- Furman, O.; Zaporozhets, A.; Tobi, D.; Bazylevich, A.; Firer, M.A.; Patsenker, L.; Gellerman, G.; Lubin, B.C.R. Novel Cyclic Peptides for Targeting EGFR and EGRvIII Mutation for Drug Delivery. Pharmaceutics 2022, 14, 1505. [Google Scholar] [CrossRef]
- Nasab, D.M.; Taheri, A.; Athari, S.S. Evaluation Anti-inflammatory Effect of Conjugated Gold Nanoparticles with Cortistatin Peptide as Drug Delivery to Asthmatic Lung Tissue. Int. J. Pept. Res. Ther. 2023, 29, 1–8. [Google Scholar] [CrossRef]
- Chauhan, V.S.; Alam, S.; Panda, J.J. Novel dipeptide nanoparticles for effective curcumin delivery. Int. J. Nanomed. 2012, 7, 4207–4222. [Google Scholar] [CrossRef]
- van Hell, A.J.; Fretz, M.M.; Crommelin, D.J.; Hennink, W.E.; Mastrobattista, E. Peptide nanocarriers for intracellular delivery of photosensitizers. J. Control. Release 2010, 141, 347–353. [Google Scholar] [CrossRef]
- Liang, X.; Shi, B.; Wang, K.; Fan, M.; Jiao, D.; Ao, J.; Song, N.; Wang, C.; Gu, J.; Li, Z. Development of self-assembling peptide nanovesicle with bilayers for enhanced EGFR-targeted drug and gene delivery. Biomaterials 2016, 82, 194–207. [Google Scholar] [CrossRef] [PubMed]
- Tian, R.; Wang, H.; Niu, R.; Ding, D. Drug delivery with nanospherical supramolecular cell penetrating peptide–taxol conjugates containing a high drug loading. J. Colloid Interface Sci. 2015, 453, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Zheng, R.; Yang, J.; Mamuti, M.; Hou, D.Y.; An, H.W.; Zhao, Y.; Wang, H. Controllable Self-Assembly of Peptide-Cyanine Conjugates In Vivo as Fine-Tunable Theranostics. Angewandte Chemie 2021, 133, 7888–7898. [Google Scholar] [CrossRef]
- Barve, A.; Jain, A.; Liu, H.; Zhao, Z.; Cheng, K. Enzyme-responsive polymeric micelles of cabazitaxel for prostate cancer targeted therapy. Acta Biomater. 2020, 113, 501–511. [Google Scholar] [CrossRef]
- Wang, Y.; Geng, Q.; Zhang, Y.; Adler-Abramovich, L.; Fan, X.; Mei, D.; Gazit, E.; Tao, K. Fmoc-diphenylalanine gelating nanoarchitectonics: A simplistic peptide self-assembly to meet complex applications. J. Colloid Interface Sci. 2023, 636, 113–133. [Google Scholar] [CrossRef]
- Xu, F.; Liu, J.; Tian, J.; Gao, L.; Cheng, X.; Pan, Y.; Sun, Z.; Li, X. Supramolecular Self-Assemblies with Nanoscale RGD Clusters Promote Cell Growth and Intracellular Drug Delivery. ACS Appl. Mater. Interfaces 2016, 8, 29906–29914. [Google Scholar] [CrossRef]
- Bellotto, O.; D’andrea, P.; Marchesan, S. Nanotubes and water-channels from self-assembling dipeptides. J. Mater. Chem. B 2023, 11, 5378–5389. [Google Scholar] [CrossRef]
- Arul, A.; Rana, P.; Das, K.; Pan, I.; Mandal, D.; Stewart, A.; Maity, B.; Ghosh, S.; Das, P. Fabrication of self-assembled nanostructures for intracellular drug delivery from diphenylalanine analogues with rigid or flexible chemical linkers. Nanoscale Adv. 2021, 3, 6176–6190. [Google Scholar] [CrossRef]
- Chibh, S.; Aggarwal, N.; Mallick, Z.; Sengupta, D.; Sachdeva, P.K.; Bera, C.; Yadav, N.; Chauhan, V.S.; Mandal, D.; Panda, J.J. Bio-piezoelectric phenylalanine-αβ-dehydrophenylalanine nanotubes as potential modalities for combinatorial electrochemotherapy in glioma cells. Biomater. Sci. 2023, 11, 3469–3485. [Google Scholar] [CrossRef] [PubMed]
- Kushner, A.M.; Guan, Z. Modular Design in Natural and Biomimetic Soft Materials. Angew. Chem. Int. Ed. 2011, 50, 9026–9057. [Google Scholar] [CrossRef]
- Seow, W.Y.; Hauser, C.A. Short to ultrashort peptide hydrogels for biomedical uses. 17. [CrossRef]
- Das, R.; Gayakvad, B.; Shinde, S.D.; Rani, J.; Jain, A.; Sahu, B. Ultrashort Peptides—A Glimpse into the Structural Modifications and Their Applications as Biomaterials. ACS Appl. Bio Mater. 2020, 3, 5474–5499. [Google Scholar] [CrossRef]
- Gomes-Porras, M.; Cárdenas-Salas, J.; Álvarez-Escolá, C. Somatostatin Analogs in Clinical Practice: A Review. Int. J. Mol. Sci. 2020, 21, 1682. [Google Scholar] [CrossRef] [PubMed]
- berg, K.; Lamberts, S.W. Somatostatin analogues in acromegaly and gastroenteropancreatic neuroendocrine tumours: past, present and future. Endocrine-Related Cancer 2016, 23, R551–R566. [Google Scholar]
- Amato, G.; Mazziotti, G.; Rotondi, M.; Iorio, S.; Doga, M.; Sorvillo, F.; Manganella, G.; Di Salle, F.; Giustina, A.; Carella, C. Long-term effects of lanreotide SR and octreotide LAR® on tumour shrinkage and GH hypersecretion in patients with previously untreated acromegaly. Clin. Endocrinol. 2002, 56, 65–71. [Google Scholar] [CrossRef]
- Grimaldi, M.; Santoro, A.; Buonocore, M.; Crivaro, C.; Funicello, N.; Saponetti, M.S.; Ripoli, C.; Rodriquez, M.; De Pasquale, S.; Bobba, F.; et al. A New Approach to Supramolecular Structure Determination in Pharmaceutical Preparation of Self-Assembling Peptides: A Case Study of Lanreotide Autogel. Pharmaceutics 2022, 14, 681. [Google Scholar] [CrossRef] [PubMed]
- Pouget, E.; Fay, N.; Dujardin, E.; Jamin, N.; Berthault, P.; Perrin, L.; Pandit, A.; Rose, T.; Valéry, C.; Thomas, D.; et al. Elucidation of the Self-Assembly Pathway of Lanreotide Octapeptide into β-Sheet Nanotubes: Role of Two Stable Intermediates. J. Am. Chem. Soc. 2010, 132, 4230–4241. [Google Scholar] [CrossRef]
- Liu, X.; Ren, H.; Peng, A.; Cheng, H.; Chen, J.; Xia, X.; Liu, T.; Wang, X. The Effect of RADA16-I and CDNF on Neurogenesis and Neuroprotection in Brain Ischemia-Reperfusion Injury. Int. J. Mol. Sci. 2022, 23, 1436. [Google Scholar] [CrossRef]
- Song, H.; Cai, G.-H.; Liang, J.; Ao, D.-S.; Wang, H.; Yang, Z.-H. Three-dimensional culture and clinical drug responses of a highly metastatic human ovarian cancer HO-8910PM cells in nanofibrous microenvironments of three hydrogel biomaterials. J. Nanobiotechnology 2020, 18, 1–19. [Google Scholar] [CrossRef]
- Wang, R.; Wang, Z.; Guo, Y.; Li, H.; Chen, Z. Design of a RADA16-based self-assembling peptide nanofiber scaffold for biomedical applications. J. Biomater. Sci. Polym. Ed. 2019, 30, 713–736. [Google Scholar] [CrossRef] [PubMed]
- Gelain, F.; Luo, Z.; Rioult, M.; Zhang, S. Self-assembling peptide scaffolds in the clinic. npj Regen. Med. 2021, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sankar, S.; O’Neill, K.; Bagot D’Arc, M.; Rebeca, F.; Buffier, M.; Aleksi, E.; Fan, M.; Matsuda, N.; Gil, E.S.; Spirio, L. Clinical use of the self-assembling peptide RADA16: a review of current and future trends in biomedicine. Frontiers in Bioengineering and Biotechnology 2021, 9, 679525. [Google Scholar] [CrossRef] [PubMed]
- Serban, M.A. Translational biomaterials—the journey from the bench to the market—think ‘product’. Current Opinion in Biotechnology 2016, 40, 31–34. [Google Scholar] [CrossRef]
- Olivier, G.K.; Cho, A.; Sanii, B.; Connolly, M.D.; Tran, H.; Zuckermann, R.N. Antibody-Mimetic Peptoid Nanosheets for Molecular Recognition. ACS Nano 2013, 7, 9276–9286. [Google Scholar] [CrossRef]
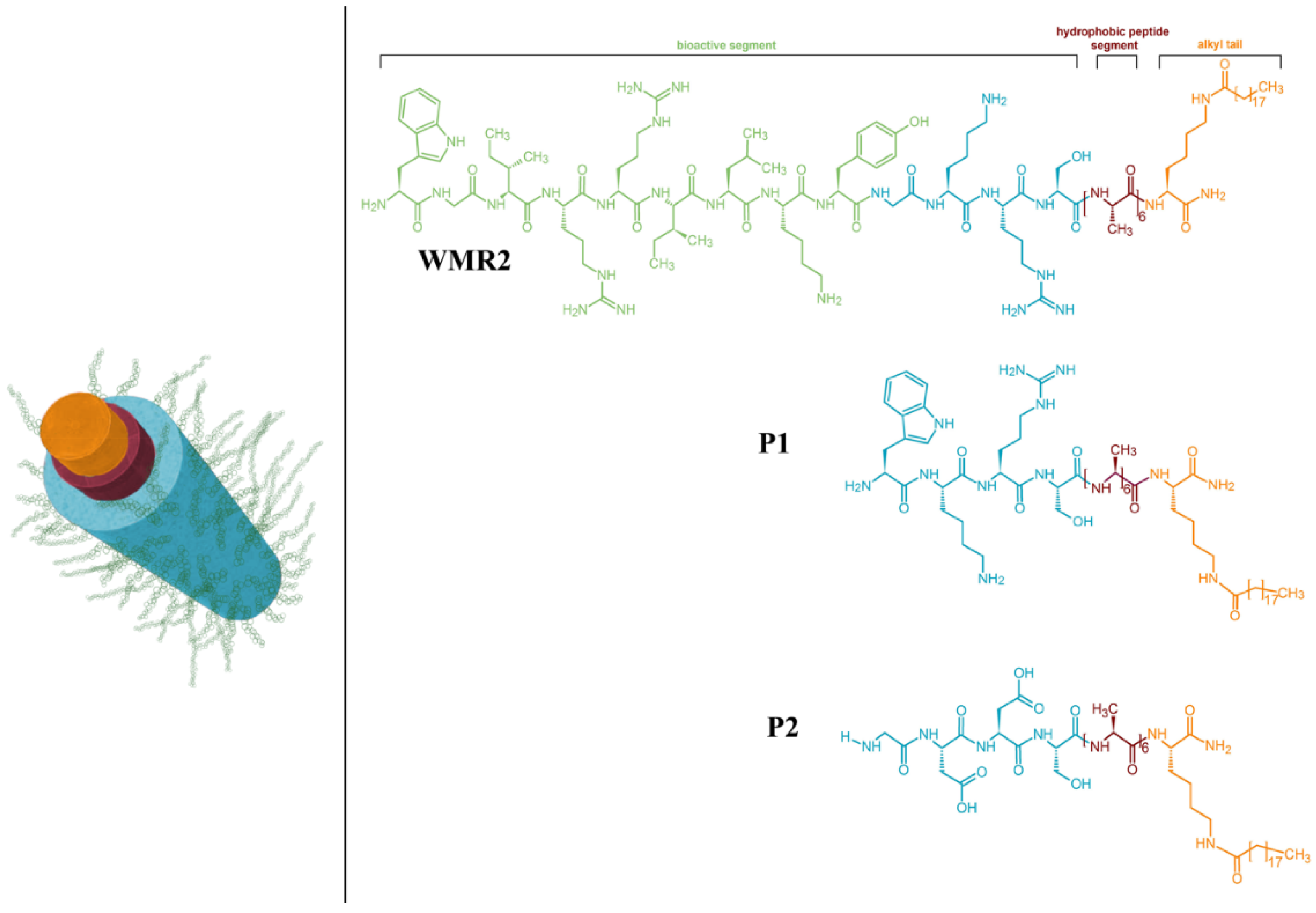
| Peptide Classification | Peptide Sequence | Structure/Formation | Biological Role/Application | Ref. |
|---|---|---|---|---|
| Ionic Peptides | EFK16-II | β-sheet | Glucose biosensing, biosensor design | 10 |
| EAK16-II | β-sheet | Anticancer activity, biomaterial mimicking ECM | 11 | |
| RADA16 | Nanofibers | Tissue engineering, hemostatic agent | 102, 103 | |
| Amphiphilic Peptides | C16IKPEAP/C16IKPEAPG | Micelles and fibrils | Gastrointestinal peptide hormone-related applications | 18 |
| KLVFFAK | Amyloid 2D nanosheets | Antimicrobial hydrogels, biofilm prevention | 27 | |
| Ac-RKKWFW-NH2 (PAF26) | β-sheet hydrogel | Antimicrobial activity against Candida and S. aureus | 60 | |
| NAVSIQKKK | Lipopeptide hydrogel | Antibacterial, wound healing | 61 | |
| Peptidomimetics | KLVFFAK (KKd-11) (D-amino acids) | Hydrogel | Antimicrobial efficacy, biofilm prevention | 59 |
| Fc-FFRGD | Nanostructure and hydrogel | Biomimetic material for drug encapsulation | 76 | |
| Cyclic Peptides | Lanreotide | Hydrogel | Treatment of acromegaly and tumours | 95, 96 |
| P6 and P9 | Short cyclic peptides | Drug delivery to EGFR-targeted cells | 67 | |
| Antimicrobial Peptides | Tet213 | Collagen-integrated | Wound healing, antimicrobial | 43 |
| Piscidin-1 | Chitosan scaffold integration | Porous biomaterial, biodegradation | 44 | |
| LL-37 | Conjugated onto collagen | Antibacterial against various pathogens | 53 | |
| Dipeptides | Diphenylalanine (FF) | Nanotubes | Drug release, stable nanostructures | 23 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




