Submitted:
02 April 2025
Posted:
02 April 2025
Read the latest preprint version here
Abstract
Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) is a debilitating disease characterised by profound fatigue, post-exertional malaise, and multi-systemic dysfunction. This hypothesis proposes that ME/CFS results from noradrenergic neuron dysfunction due to increased neuronal insulin receptor sensitivity, potentially caused by glucocorticoid receptor resistance, high insulin levels, and insulin receptor gene variants. An additional contributing factor is phosphatidylcholine deficiency, which may exacerbate neuronal insulin hypersensitivity and disrupt cellular function. We explore genetic, metabolic, and inflammatory factors that contribute to phosphatidylcholine deficiency and propose a multi-component model of ME/CFS, highlighting the interplay between phosphatidylcholine metabolism, liver dysfunction, neuronal function and inflammatory signaling. Furthermore, we discuss how dysregulated norepinephrine signaling impacts various brain regions and peripheral systems, contributing to the wide-ranging symptomatology of ME/CFS.
Keywords:
Introduction
The Sympathetic Nervous System and Noradrenergic Neurons
Reduced Norepinephrine Transporters and Beta-2 Adrenergic Receptor Down-Regulation
Overtraining Syndrome
Negative Feedback Mechanism of Norepinephrine Regulation
Insulin’s Role in Regulating Norepinephrine Transporters
Glucocorticoid Receptor Resistance
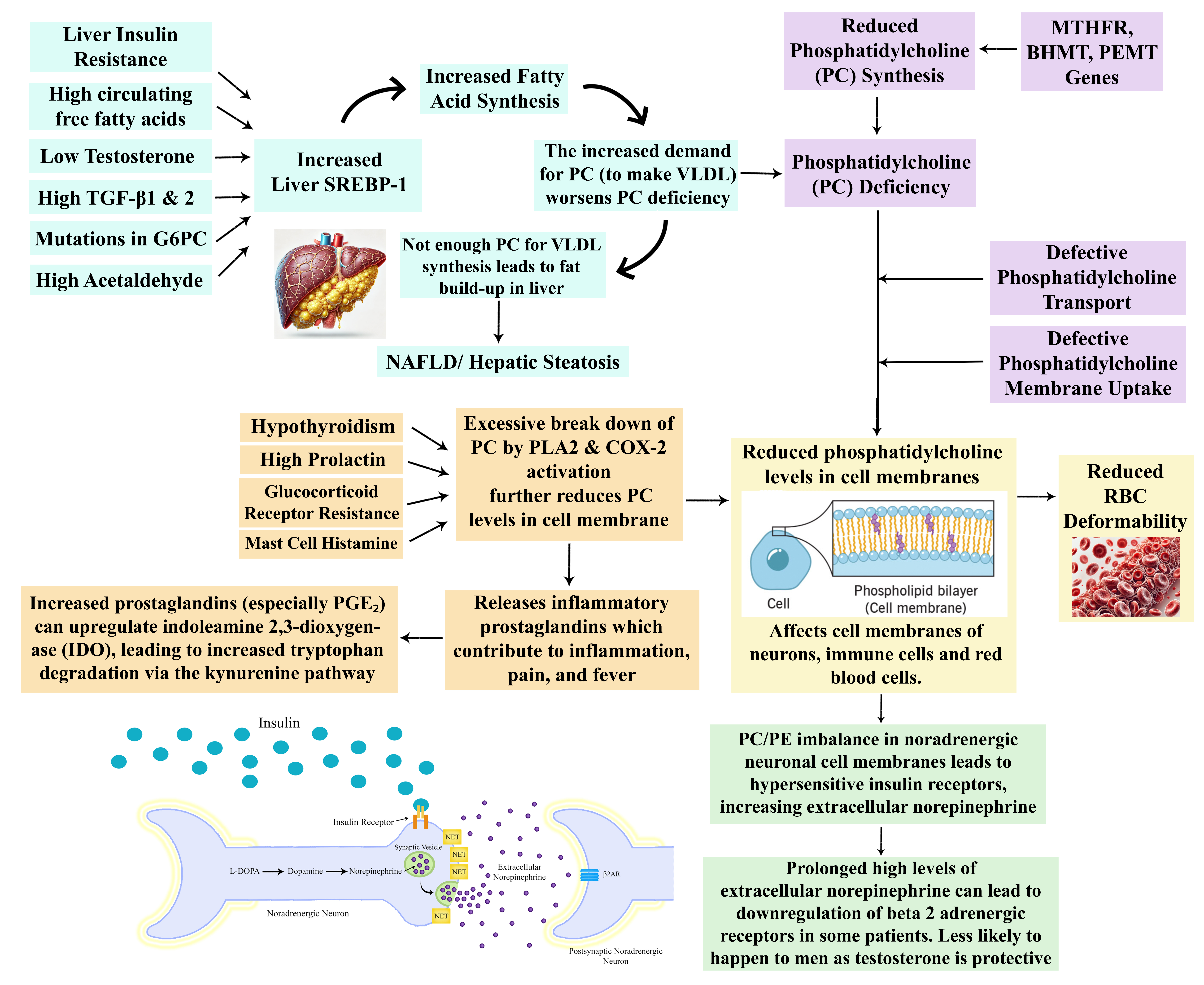
How Cell Membrane Phosphatidylcholine Deficiency Affects Insulin Signaling
Defects in Phosphatidylcholine Transport and Membrane Uptake
Phosphatidylcholine Depletion Due to Phospholipase A2 and COX-2 Activation
Elevated Prostaglandin Levels and Their Pathophysiological Consequences
Mechanisms Driving Phospholipase A2 and COX-2 Activation
Phosphatidylcholine Requirement for VLDL Synthesis and Its Role in Hepatic Lipid Homeostasis
SREBP-1 Activation and the Redistribution of Phosphatidylcholine to VLDL
Factors Contributing to SREBP-1 Activation and Potential Triggers of Phosphatidylcholine Deficiency
- 1.
- Hyperinsulinemia and Hepatic Insulin Resistance
- 2.
- Elevated Circulating Free Fatty Acids (FFAs)
- 3.
- Testosterone Dysregulation
- 4.
- High Acetaldehyde Exposure
- 5.
- TGF-β Signaling and Fibrotic Progression
- 6.
- Iron Overload
Reduced Red Blood Cell Deformability in ME/CFS
Accuracy of the Hemoglobin A1c Test
Is There A Prodrome Phase?
Norepinephrine Dysregulation in the Brain and Its Impact on Astrocyte Function
Norepinephrine Dysregulation of Astrocytes and Glucose Uptake
Impact of High Lactate Levels on Sleep
Lactate as a Preferential Fuel
Sex Differences in Astrocytic Response to Norepinephrine Dysregulation
Norepinephrine, Astrocytes and the Glymphatic System
Norepinephrine, Astrocytes and Neurotransmitter Regulation
Astrocytic Regulation of Cerebral Blood Flow
The Effect of Testosterone on Symptom Profile and Illness Risk
Potential Links to Endometriosis
Mast Cell Activation as Both a Root Cause and a Downstream Effect in ME/CFS
A Component-Based Approach to Understanding and Treating ME/CFS
References
- Arron HE, Marsh BD, Kell DB, Khan MA, Jaeger BR, Pretorius E. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: The Biology of a Neglected Disease. Front Immunol. 2024;15:1386607. [CrossRef]
- Walitt B, Singh K, LaMunion SR, Hallett M, Jacobson S, Chen K, et al. Deep Phenotyping of Post-Infectious Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Nat Commun. 2023; 15(1):907. [CrossRef]
- Nguyen CC, Kumar S, Zucknick M, Kristensen VN, Gjerstad J, Nilsen H, Wyller VB. Associations Between Clinical Symptoms, Plasma Norepinephrine, and Deregulated Immune Gene Networks in Subgroups of Adolescents with Chronic Fatigue Syndrome. Brain Behav Immun. 2019;76:82-96. [CrossRef]
- Kavelaars A, Kuis W, Knook LME, Sinnema G, Heijnen CJ. Disturbed Neuroendocrine-Immune Interactions in Chronic Fatigue Syndrome. J Clin Endocrinol Metab. 2000;85(2):692-696. [CrossRef]
- Fry AC, Schilling BK, Weiss LW, Chiu LZF. β2-Adrenergic Receptor Downregulation and Performance Decrements During High-Intensity Resistance Exercise Overtraining. J Appl Physiol. 2006;101(6):1664-72. [CrossRef]
- Goto T, Kikuchi S, Mori K, Nakayama T, Fukuta H, Seo Y, et al. Cardiac β-Adrenergic Receptor Downregulation, Evaluated by Cardiac PET, in Chronotropic Incompetence. J Nucl Med. 2021;62(7): 996–998. [CrossRef]
- Robertson SD, Matthies HJG, Owens WA, Sathananthan V, Christianson NSB, Kennedy JP, et al. Insulin Reveals Akt Signaling as a Novel Regulator of Norepinephrine Transporter Trafficking and Norepinephrine Homeostasis. J Neurosci. 2010;30(34):11305-16. [CrossRef]
- Das S, Taylor K, Kozubek J, Sardell J, Gardner S. Genetic Risk Factors for ME/CFS Identified Using Combinatorial Analysis. J Transl Med. 2022;20(1):598. [CrossRef]
- Taylor K, Pearson M, Das S, Sardell J, Chocian K, Gardner S. Genetic risk factors for severe and fatigue dominant long COVID and commonalities with ME/CFS identified by combinatorial analysis. J Transl Med. 2023;21(1):775. 2023. [CrossRef]
- Figlewicz DP, Szot P, Chavez M, Woods SC, Veith RC. Intraventricular insulin increases dopamine transporter mRNA in rat VTA/substantia nigra. Brain Res. 1994;644(2):331-4. [CrossRef]
- Sun Z, Fan Y, Zha Q, Zhu MY. Corticosterone up-regulates expression and function of norepinephrine transporter in SK-N-BE(2)C cells. J Neurochem. 2010;113(1): 105–116. [CrossRef]
- Saha AK, Schmidt BR, Wilhelmy J, Nguyen V, Abugherir A, Lechuga TJ, et al. Red blood cell deformability is diminished in patients with Chronic Fatigue Syndrome. Clin Hemorheol Microcirc. 2019;71(1):113–6. [CrossRef]
- da Silva ALG, Vieira LP, Dias LS, Prestes CV, Back GD, Goulart CL, et al. Impact of long COVID on heart rate variability at rest and during deep breathing maneuver. Sci Rep. 2023;13:22695. [CrossRef]
- Lee JS, Sato W, Son CG. Brain-regional characteristics and neuroinflammation in ME/CFS patients from neuroimaging: A systematic review and meta-analysis. Autoimmun. Rev. 2024;23:2. [CrossRef]
- Ibrahim MMH, Bheemanapally K, Briski KP. Norepinephrine regulation of adrenergic receptor expression, 5′ AMP-activated protein kinase activity, and glycogen metabolism and mass in male versus female hypothalamic primary astrocyte cultures. ASN Neuro. 2020;12:17. [CrossRef]
- Carbajal-García A, Reyes-García J, Casas-Hernández MF, Flores-Soto E, Díaz-Hernández V, Solís-Chagoyán H, Sommer B, Montaño LM. Testosterone augments β2 adrenergic receptor genomic transcription increasing salbutamol relaxation in airway smooth muscle. Mol. Cell. Endocrinol. 2020;510:110801. [CrossRef]
- Scardapane L, Cardinali DP. Effect of estradiol and testosterone on catechol-O-methyl transferase activity of rat superior cervical ganglion, pineal gland, anterior hypophysis and hypothalamus. J Neural Transm. 1977;40(1):81-6. [CrossRef]
- Boneva RS, Lin JM, Wieser F, Nater UM, Ditzen B, Taylor RN, Unger ER. Endometriosis as a comorbid condition in chronic fatigue syndrome (CFS): secondary analysis of data from a CFS case-control study. Front. Pediatr. 2019; 7: 195. [CrossRef]
- Mathias JR, Franklin RR. Neural dysfunction of the gastrointestinal tract associated with endometriosis: a disease of insulin sensitivity. Fertil Steril. 2002;77(25). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



