Submitted:
18 September 2024
Posted:
19 September 2024
Read the latest preprint version here
Abstract
Keywords:
Introduction
The Sympathetic Nervous System and Noradrenergic Neurons
ME3 Subtype: Norepinephrine Deficiency
ME1 & ME2 Subtype: Reduced Norepinephrine Transporters and Beta-2 Adrenergic Receptor Down-Regulation
Overtraining Syndrome
Negative Feedback Mechanism of Norepinephrine Regulation
Insulin’s Role in Regulating Norepinephrine Transporters
Distinguishing ME1 and ME2 Subtypes Based on Insulin Dynamics
ME1: Does It Have a Prodrome Phase?
The Effect of Metformin on Long COVID Risk: Implications for the ME2 Subtype
Postural Orthostatic Tachycardia Syndrome (POTS)
Microclots in ME/CFS and Long COVID
Glucocorticoid Receptor Resistance
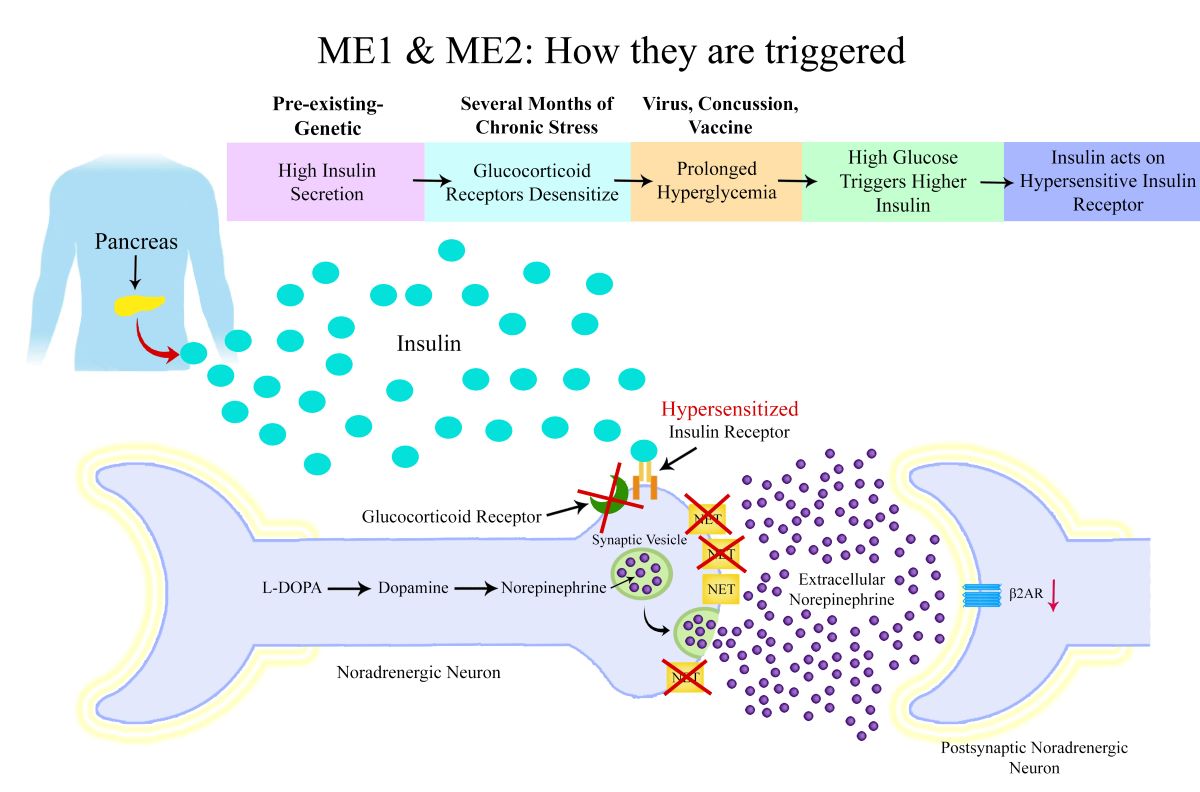
The Sequence of Events Leading to ME
- Glucocorticoid Receptor Resistance or Low Cortisol: Glucocorticoids, such as cortisol, regulate insulin sensitivity, particularly in neurons. Due to GR resistance or low cortisol levels, there is a reduction in GR activity. This leads to increased insulin receptor sensitivity in neurons, as cortisol normally acts to promote insulin resistance in the body. The reduced GR function or low cortisol levels amplify insulin’s effect on neurons, making them more susceptible to hyperinsulinemia.
- Hyperglycaemia Trigger: The second major step in the sequence is the occurrence of a triggering event—such as a viral infection, vaccination, concussion, or physical injury—that induces a period of hyperglycemia. This period of high blood sugar levels, combined with the genetically predisposed insulin dysregulation, leads to elevated insulin secretion from the pancreas.
- Sympathetic Nervous System Over-Activation: In individuals with genetic dysregulation of insulin secretion and increased insulin receptor sensitivity on noradrenergic neurons, the hyperinsulinemia results in over-activation of the SNS. The insulin receptors on noradrenergic neurons, now hypersensitive due to the reduced GR function, become over-stimulated by the excess insulin. This triggers down-regulation of NETS and reduced reuptake of norepinephrine, contributing to a state of sympathetic overdrive.
- Downstream Effects of Norepinephrine Dysregulation: Over time, the NET down-regulation and chronic SNS over-activation leads to impaired norepinephrine reuptake, norepinephrine depletion in neurons and eventual down-regulation of adrenergic receptors.
Autonomic Dysfunction Across ME Subtypes
ME2: Hypovolemic Postural Orthostatic Tachycardia Syndrome (POTS)
ME3: Orthostatic Intolerance (OI) from Norepinephrine Deficiency
ME1: Initial Orthostatic Intolerance (IOI)
The Effect of Testosterone on Symptom Profile and Illness Risk
Is There a Cholinergic ME Subtype of Noradrenergic Dysfunction?
Potential Links to Endometriosis and Polycystic Ovary Syndrome
Endometriosis
Polycystic Ovary Syndrome (PCOS)
Dopamine Deficiency in ME
Evidence of Dopamine Deficiency in ME
Mechanisms of Dopamine Deficiency in ME
Mast Cell Activation
Norepinephrine Dysregulation in the Brain and Its Impact on Astrocyte Function
Norepinephrine Dysregulation of Astrocytes and Glucose Uptake
Impact of High Lactate Levels on Sleep
Lactate as a Preferential Fuel
Sex Differences in Astrocytic Response to Norepinephrine Dysregulation
Norepinephrine, Astrocytes and the Glymphatic System
Norepinephrine, Astrocytes and Neurotransmitter Regulation
Astrocytic Regulation of Cerebral Blood Flow
Ehlers-Danlos Syndrome and Compensatory POTS: Potential Overlap with Myalgic Encephalomyelitis
Categorising and Naming Subtypes
Potential Treatments
Pharmacological Interventions
Non-Pharmacological Interventions
Testing for Subtypes
Conclusion
Author Contributions
Data Availability Statement
Conflicts of Interest
References
- Arron HE, Marsh BD, Kell DB, Khan MA, Jaeger BR, Pretorius E. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: The Biology of a Neglected Disease. Front Immunol. 2024;15:1386607. DOI: 10.3389/fimmu.2024.1386607.
- Słomko J, Estévez-López F, Kujawski S, Zawadka-Kunikowska M, Tafil-Klawe M, Klawe JJ, et al. Autonomic Phenotypes in Chronic Fatigue Syndrome (CFS) Are Associated with Illness Severity: A Cluster Analysis. J. Clin. Med. 2020, 9(8), 2531. DOI: 10.3390/jcm9082531.
- Morris G, Anderson G, Maes M. Hypothalamic-Pituitary-Adrenal Hypofunction in Myalgic Encephalomyelitis (ME)/Chronic Fatigue Syndrome (CFS) as a Consequence of Activated Immune-Inflammatory and Oxidative and Nitrosative Pathways. Mol Neurobiol. 2016; 2017 Nov;54(9):6806-6819. DOI: 10.1007/s12035-016-0170-2.
- Walitt B, Singh K, LaMunion SR, Hallett M, Jacobson S, Chen K, et al. Deep Phenotyping of Post-Infectious Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Nat Commun. 2023; 15(1):907. DOI: 10.1038/s41467-024-45107-3.
- Hoel F, Hoel A, Pettersen IK, Rekeland IG, Risa K, Alme K, et al. A Map of Metabolic Phenotypes in Patients with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. JCI Insight. 2021;6(16):e149217. DOI: 10.1172/jci.insight.149217.
- Das S, Taylor K, Kozubek J, Sardell J, Gardner S. Genetic Risk Factors for ME/CFS Identified Using Combinatorial Analysis. J Transl Med. 2022;20(1):598. DOI: 10.1186/s12967-022-03815-8.
- Wyller VB, Vitelli V, Sulheim D, Fagermoen E, Winger A, Godang K, et al. Altered Neuroendocrine Control and Association to Clinical Symptoms in Adolescent Chronic Fatigue Syndrome: A Cross-Sectional Study. J Transl Med. 2016;14(1):121. DOI: 10.1186/s12967-016-0873-1.
- Nguyen CC, Kumar S, Zucknick M, Kristensen VN, Gjerstad J, Nilsen H, Wyller VB. Associations Between Clinical Symptoms, Plasma Norepinephrine, and Deregulated Immune Gene Networks in Subgroups of Adolescents with Chronic Fatigue Syndrome. Brain Behav Immun. 2019;76:82-96. DOI: 10.1016/j.bbi.2018.11.008.
- Kavelaars A, Kuis W, Knook LME, Sinnema G, Heijnen CJ. Disturbed Neuroendocrine-Immune Interactions in Chronic Fatigue Syndrome. J Clin Endocrinol Metab. 2000;85(2):692-696. DOI: 10.1210/jcem.85.2.6379.
- Fry AC, Schilling BK, Weiss LW, Chiu LZF. β2-Adrenergic Receptor Downregulation and Performance Decrements During High-Intensity Resistance Exercise Overtraining. J Appl Physiol. 2006;101(6):1664-72. DOI: 10.1152/japplphysiol.01599.2005.
- Goto T, Kikuchi S, Mori K, Nakayama T, Fukuta H, Seo Y, et al. Cardiac β-Adrenergic Receptor Downregulation, Evaluated by Cardiac PET, in Chronotropic Incompetence. J Nucl Med. 2021;62(7): 996–998. DOI: 10.2967/jnumed.120.253419.
- Robertson SD, Matthies HJG, Owens WA, Sathananthan V, Christianson NSB, Kennedy JP, et al. Insulin Reveals Akt Signaling as a Novel Regulator of Norepinephrine Transporter Trafficking and Norepinephrine Homeostasis. J Neurosci. 2010;30(34):11305-16. DOI: 10.1523/JNEUROSCI.0126-10.2010.
- Taylor K, Pearson M, Das S, Sardell J, Chocian K, Gardner S. Genetic risk factors for severe and fatigue dominant long COVID and commonalities with ME/CFS identified by combinatorial analysis. J Transl Med. 2023;21(1):775. 2023. DOI: 10.1186/s12967-023-04588-4.
- Cen J, Sargsyan E, Bergsten P. Fatty acids stimulate insulin secretion from human pancreatic islets at fasting glucose concentrations via mitochondria-dependent and -independent mechanisms. Nutrition & Metabolism. 2016;13(59) DOI: 10.1186/s12986-016-0119-5.
- Yuan T, Song S, Zhao T, Duo Y, Wang S, Gao J, Liu S, Dong Y, Li R, Fu Y, Zhao W. Patterns of Insulin Secretion During First-Phase Insulin Secretion in Normal Chinese Adults. Front Endocrinol. 2021;12:738427. DOI: 10.3389/fendo.2021.738427.
- Giunta S, Giordani C, De Luca M, Olivier F. Long-COVID-19 autonomic dysfunction: An integrated view in the framework of inflammaging. Mech Ageing Dev. 2024;218:111915. DOI: 10.1016/j.mad.2024.111915.
- Bramante CT, Buse JB, Liebovitz DM, Nicklas JM, Puskarich MA, Cohen K, et al. Outpatient treatment of COVID-19 and incidence of post-COVID-19 condition over 10 months (COVID-OUT): a multicentre, randomised, quadruple-blind, parallel-group, phase 3 trial. Lancet Infect Dis. 2023;23(10):1119-1129. DOI: 10.1016/S1473-3099(23)00299-2.
- Montefusco L, Ben Nasr M, D’Addio F, Loretelli C, Rossi A, Pastore I, et al. Acute and long-term disruption of glycometabolic control after SARS-CoV-2 infection. Nat Metab. 2021;3(6):774-785. DOI: 10.1038/s42255-021-00407-6.
- Barreto EA, Cruz AS, Veras FP, Leiria LO, et al. COVID-19-related hyperglycemia is associated with infection of hepatocytes and stimulation of gluconeogenesis. Proc Natl Acad Sci U S A. 2023;120(21): e2217119120. DOI: https://doi.org/10.1073/pnas.2217119120.
- Nunes JM, Kruger A, Proal A, Kell DB, Pretorius E. The Occurrence of Hyperactivated Platelets and Fibrinaloid Microclots in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). Pharmaceuticals. 2022;15(8):931. DOI: 10.3390/ph15080931.
- Pretorius E, Vlok M, Venter C, Bezuidenhout JA, Laubscher GJ, Steenkamp J, Kell DB. Persistent clotting protein pathology in Long COVID/Post-Acute Sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin. Cardiovasc Diabetol. 2021;20(1):172. DOI: 10.1186/s12933-021-01359-7.
- Pretorius E, Venter C, Laubscher GJ, Lourens PJ, Steenkamp J, Kell DB. Prevalence of readily detected amyloid blood clots in ‘unclotted’ Type 2 Diabetes Mellitus and COVID-19 plasma: a preliminary report. Cardiovasc Diabetol. 2020;19(1):193. DOI: 10.1186/s12933-020-01165-7.
- Sun Z, Fan Y, Zha Q, Zhu MY. Corticosterone up-regulates expression and function of norepinephrine transporter in SK-N-BE(2)C cells. J Neurochem. 2010;113(1): 105–116. DOI: 10.1111/j.1471-4159.2010.06587.x.
- Fedorowski A, Fanciulli A, Raj SR, Sheldon R, Shibao CA, Sutton R. Cardiovascular autonomic dysfunction in post-COVID-19 syndrome: a major health-care burden. Nat Rev Cardiol. 2024;21(6):379-395. DOI: 10.1038/s41569-023-00962-3.
- Sheikh NA, Phillips AA, Ranada S, Lloyd M, Kogut K, Bourne KM, Jorge JG, Lei LY, Sheldon RS, Exner DV, Runte M, Raj SR. Mitigating Initial Orthostatic Hypotension: Mechanistic Roles of Muscle Contraction Versus Sympathetic Activation. Hypertension. 2022;79(3):638-647. DOI: 10.1161/HYPERTENSIONAHA.121.18580.
- Carbajal-García A, Reyes-García J, Casas-Hernández MF, Flores-Soto E, Díaz-Hernández V, Solís-Chagoyán H, Sommer B, Montaño LM. Testosterone augments β2 adrenergic receptor genomic transcription increasing salbutamol relaxation in airway smooth muscle. Mol. Cell. Endocrinol. 2020;510:110801 DOI: 10.1016/j.mce.2020.110801.
- Scardapane L, Cardinali DP. Effect of estradiol and testosterone on catechol-O-methyl transferase activity of rat superior cervical ganglion, pineal gland, anterior hypophysis and hypothalamus. J Neural Transm. 1977;40(1):81-6. DOI: 10.1007/BF01250282.
- Boneva RS, Lin JM, Wieser F, Nater UM, Ditzen B, Taylor RN, Unger ER. Endometriosis as a comorbid condition in chronic fatigue syndrome (CFS): secondary analysis of data from a CFS case-control study. Front. Pediatr. 2019; 7: 195. DOI: 10.3389/fped.2019.00195.
- Mathias JR, Franklin RR. Neural dysfunction of the gastrointestinal tract associated with endometriosis: a disease of insulin sensitivity. Fertil Steril. 2002;77(25) DOI: 10.1016/S0015-0282(01)03088-6.
- Crosby LD, Kalanidhi S, Bonilla A, Subramanian A, Ballon JS, Bonilla H. Off-label use of Aripiprazole shows promise as a treatment for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): a retrospective study of 101 patients treated with a low dose of Aripiprazole. J Transl Med. 2021;19(1):50. DOI: 10.1186/s12967-021-02721-9.
- Sharpe M, Clements A, Hawton K, Young AH, Sargent P, Cowen PJ. Increased prolactin response to buspirone in chronic fatigue syndrome. J. Affect. Disord. 1996;41(1):71-76. DOI: https://doi.org/10.1016/0165-0327(96)00075-4.
- Figlewicz DP, Szot P, Chavez M, Woods SC, Veith RC. Intraventricular insulin increases dopamine transporter mRNA in rat VTA/substantia nigra. Brain Res. 1994;644(2):331-4. DOI: 10.1016/0006-8993(94)91698-5.
- Theoharides TC, Singh LK, Boucher W, Pang X, Letourneau R, Webster E, Chrousos G. Corticotropin-releasing hormone induces skin mast cell degranulation and increased vascular permeability: A possible explanation for its proinflammatory effects. Endocrinology. 1998;139(1):403-13. DOI: 10.1210/endo.139.1.5660.
- Fink, K., Velebit, J., Vardjan, N., Zorec, R., & Kreft, M. (2021). Noradrenaline-induced l-lactate production requires d -glucose entry and transit through the glycogen shunt in single-cultured rat astrocytes. Journal of Neuroscience Research, 99(4), 1084–1098. https://doi.org/10.1002/jnr.24783.
- Lee JS, Sato W, Son CG. Brain-regional characteristics and neuroinflammation in ME/CFS patients from neuroimaging: A systematic review and meta-analysis. Autoimmun. Rev. 2024;23:2. DOI: https://doi.org/10.1016/j.autrev.2023.103484.
- Litvin M, Clark AL, Fisher SJ. Recurrent hypoglycemia: boosting the brain’s metabolic flexibility. J Clin Invest. 2013;123(5): 1922–1924. DOI: 10.1172/JCI69796.
- Ibrahim MMH, Bheemanapally K, Briski KP. Norepinephrine regulation of adrenergic receptor expression, 5′ AMP-activated protein kinase activity, and glycogen metabolism and mass in male versus female hypothalamic primary astrocyte cultures. ASN Neuro. 2020;12:17. DOI: 10.1177/1759091420974134.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



