Submitted:
17 September 2024
Posted:
18 September 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Cell Culture and Treatments
Bioactive Glass
Cytocompatibility Evaluation
Cell Viability
Real Time PCR (qPCR)
| Name | Sequence |
|---|---|
| AKR1C1 | GCCGTGGAGAAGTGTAAAG/CAGACAGGCTTGTACTTGAG |
| ALP | GAGTATGAGAGTGACGAGAAAG/GAAGTGGGAGTGCTTGTATC |
| BSP | CAGAAGAGGAGGAGGAAGAA/CCCAGTGTTGTAGCAGAAAG |
| COL1 | GGATTCCAGTTCGAGTATGG/CAGTGGTAGGTGATGTTCTG |
| DMT1 | GCTGTCTTCCAAGATGTAGAG/GGATGGGTATGAGAGCAAAG |
| FSP1 | CCTGCCCTTCTCTCATCTTA/GTCCTCATAGGCCTGGATAG |
| GPX4 | AGCTCTTCTGGGAAGTAGAC/CCTCCCTGTACCACATCTAT |
| L34 | GTCCCCGAACCCTGGTAATAGA/GGCCCTGCTGACATGTTTCTT |
| NQO1 | GGATGAGACACCACTGTATTT/CTCCTCATCCTGTACCTCTT |
| NRF2 | CCTGCCCTTCTCTCATCTTA/GTCCTCATAGGCCTGGATAG |
| OPN | CCCATCTCAGAAGCAGAATC/TGGCTTTCGTTGGACTTAC |
| PTGS2 | GCCTGGTCTGATGATGTATG/GTATTAGCCTGCTTGTCTGG |
| RUNX2 | GAATGCCTCTGCTGTTATGA/GAAGACGGTTATGGTCAAGG |
| SLC7A11 | CTGGGTTTCTTGTCCCATATAA/GTTGCCCTTTCCCTCTATTC |
| TfR1 | GTGAGGGATCTGAACCAATAC/TGGAAGTAGCACGGAAGA |
Western Blotting Analysis
Detection of Intracellular Fe2+
Immunofluorescence
Statistical Analysis
3. Results
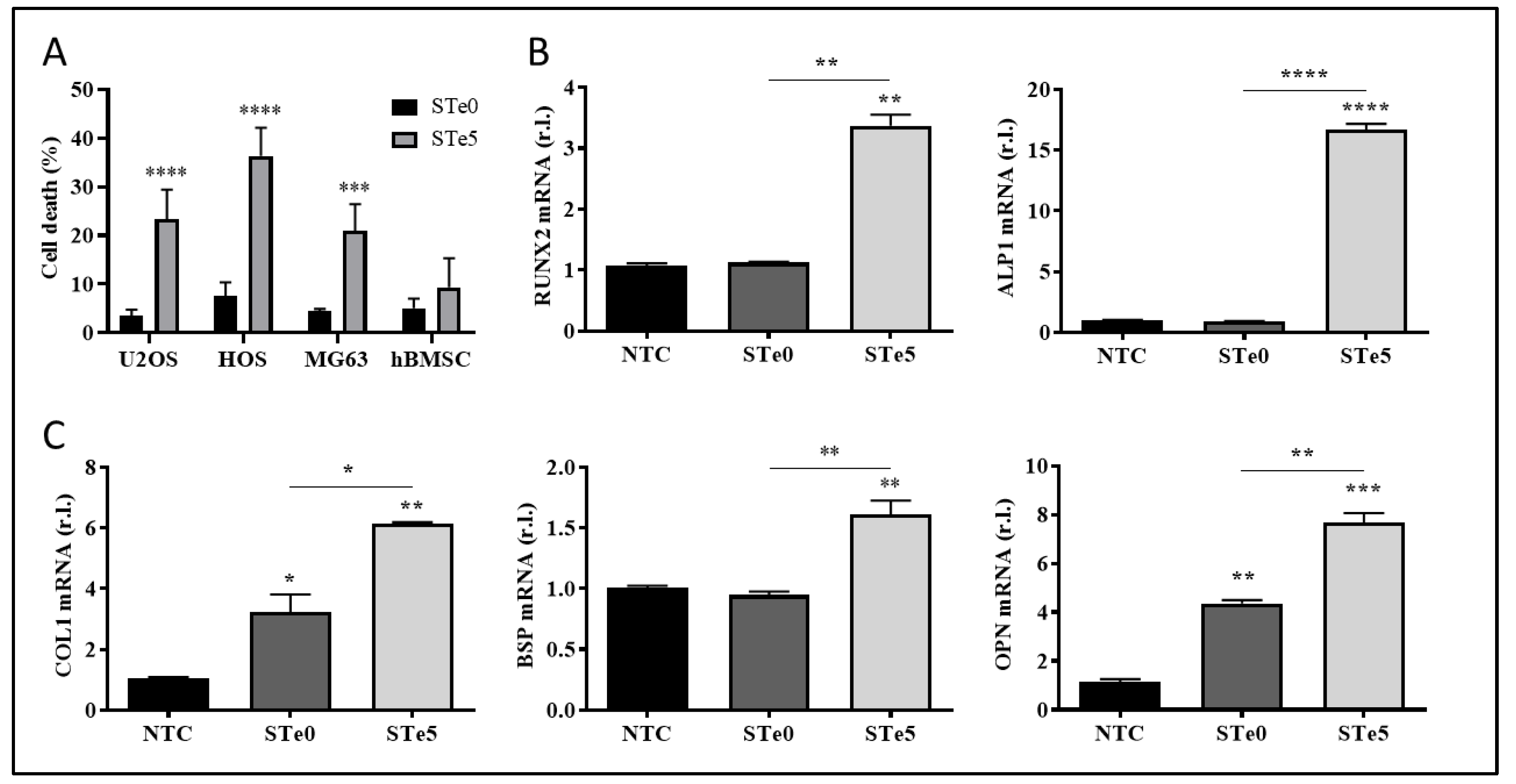
3.1. Selective toxicity of tellurium-doped bioactive glass to osteosarcoma cells and osteoinductive effect on hBMSCs
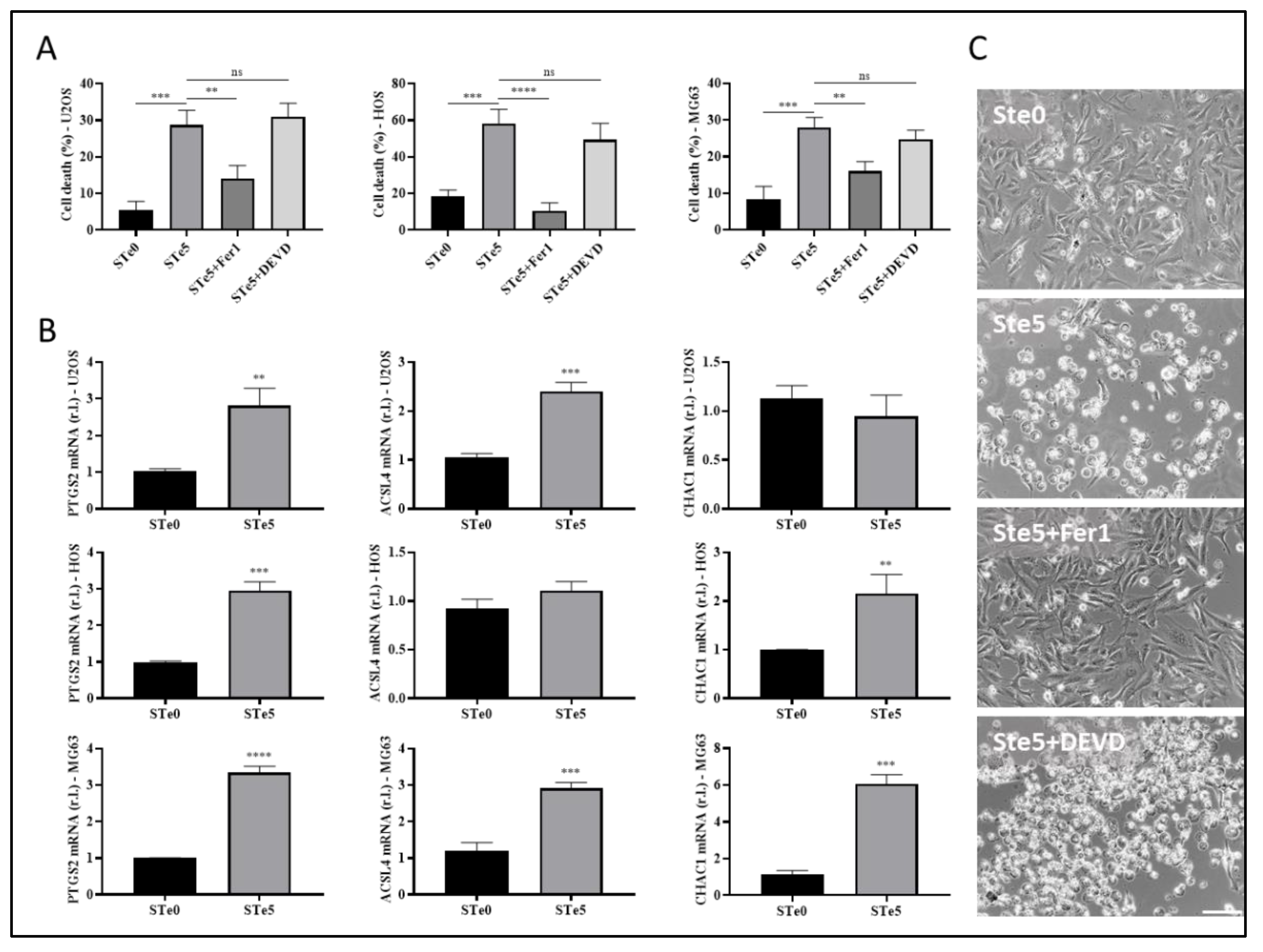
3.2. Tellurium-Doped Bioactive Glass Induces Ferroptosis-Mediated OS Cells Death
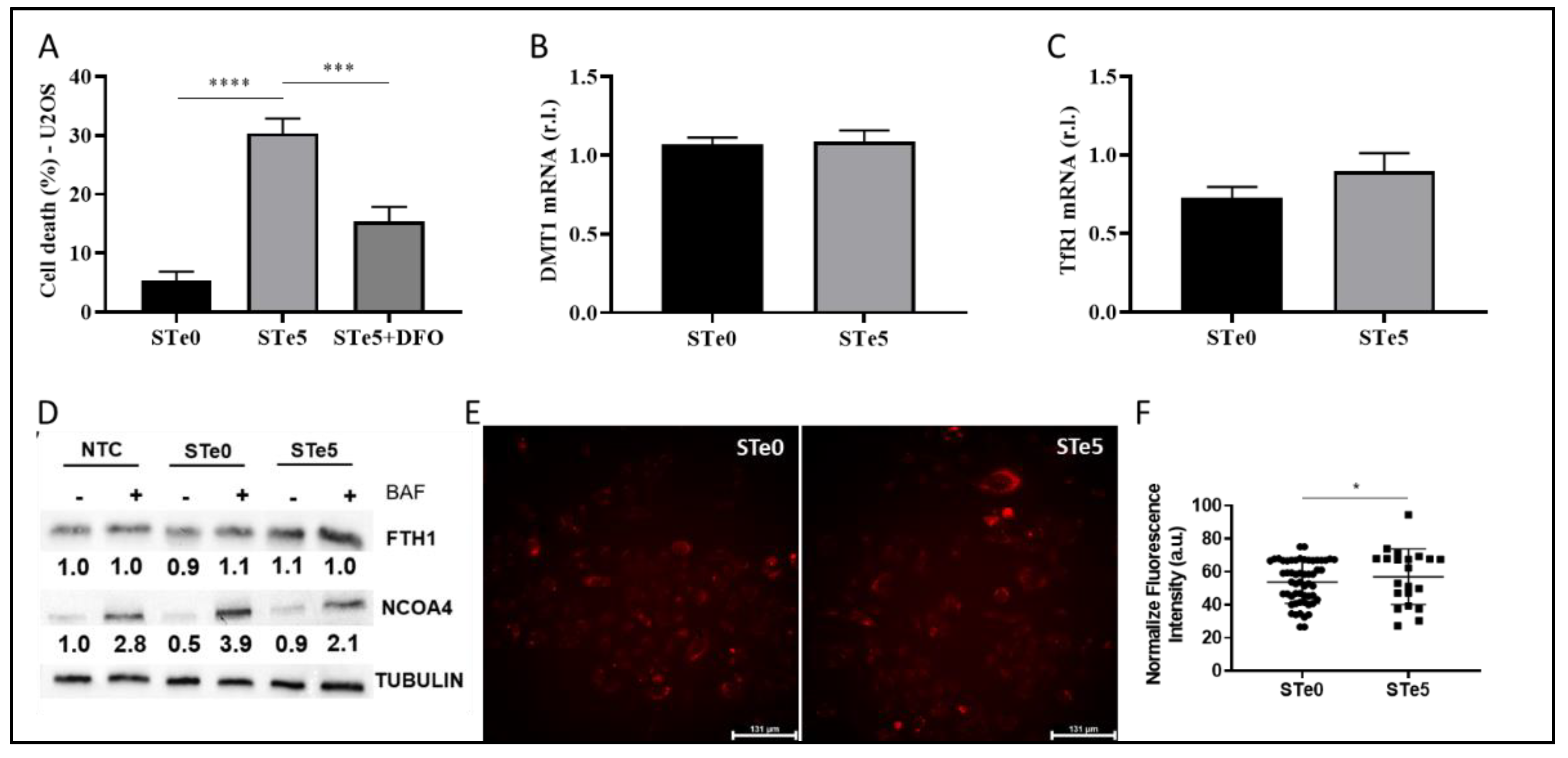
3.3. Role of Iron in Te-BAG-Induced Ferroptosis
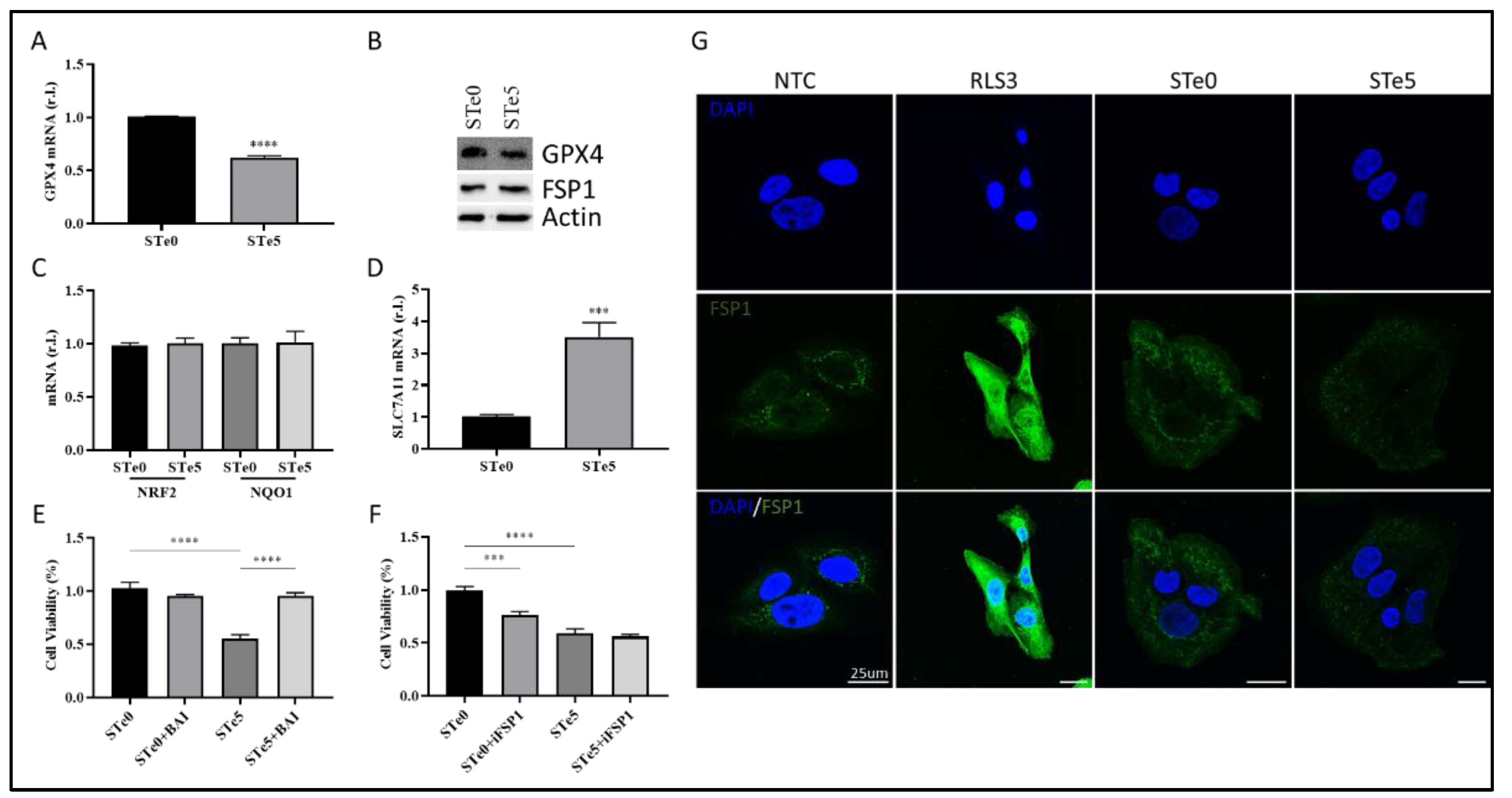
3.4. Te-BAG-Induced Ferroptosis Circumvents the Inhibitory Activity of FSP1
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q. , et al. , Ferroptosis in cancer: From molecular mechanisms to therapeutic strategies. Signal Transduct Target Ther. 2024, 9, 55. [Google Scholar] [PubMed]
- Zheng, Y.; Sun, L.; Guo, J.; Ma, J. The crosstalk between ferroptosis and anti-tumor immunity in the tumor microenvironment: molecular mechanisms and therapeutic controversy. Cancer Commun (Lond) 2023, 43, 1071–1096. [Google Scholar] [CrossRef]
- Corazzari, M.; Collavin, L. Wild-type and mutant p53 in cancer-related ferroptosis. A matter of stress management? Front Genet 2023, 14, 1148192. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, M.; Saverio, V.; Monzani, R.; Ferrari, E.; Piacentini, M.; Corazzari, M. Ferroptosis: a new unexpected chance to treat metastatic melanoma? Cell Cycle 2020, 19, 2411–2425. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Li, H.; Graham, E.T.; Deik, A.A.; Eaton, J.K.; Wang, W.; Sandoval-Gomez, G.; Clish, C.B.; Doench, J.G.; Schreiber, S.L. Cytochrome P450 oxidoreductase contributes to phospholipid peroxidation in ferroptosis. Nat Chem Biol 2020, 16, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Kagan, V.E.; Mao, G.; Qu, F.; Angeli, J.P.; Doll, S.; Croix, C.S.; Dar, H.H.; Liu, B.; Tyurin, V.A.; Ritov, V.B.; et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat Chem Biol 2017, 13, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Ingold, I.; Berndt, C.; Schmitt, S.; Doll, S.; Poschmann, G.; Buday, K.; Roveri, A.; Peng, X.; Porto Freitas, F.; Seibt, T.; et al. Selenium Utilization by GPX4 Is Required to Prevent Hydroperoxide-Induced Ferroptosis. Cell 2018, 172, 409–422. [Google Scholar] [CrossRef] [PubMed]
- Doll, S.; Freitas, F.P.; Shah, R.; Aldrovandi, M.; da Silva, M.C.; Ingold, I.; Goya Grocin, A.; Xavier da Silva, T.N.; Panzilius, E.; Scheel, C.H.; et al. FSP1 is a glutathione-independent ferroptosis suppressor. Nature 2019, 575, 693–698. [Google Scholar] [CrossRef]
- Gagliardi, M.; Cotella, D.; Santoro, C.; Cora, D.; Barlev, N.A.; Piacentini, M.; Corazzari, M. Aldo-keto reductases protect metastatic melanoma from ER stress-independent ferroptosis. Cell Death Dis 2019, 10, 902. [Google Scholar] [CrossRef]
- Dai, E.; Meng, L.; Kang, R.; Wang, X.; Tang, D. ESCRT-III-dependent membrane repair blocks ferroptosis. Biochem Biophys Res Commun 2020, 522, 415–421. [Google Scholar] [CrossRef]
- Gagliardi, M.; Saverio, V.; Rossin, F.; D'Eletto, M.; Corazzari, M. Transglutaminase 2 and Ferroptosis: a new liaison? Cell Death Discov 2023, 9, 88. [Google Scholar] [CrossRef] [PubMed]
- Simpson, E.; Brown, H.L. Understanding osteosarcomas. JAAPA 2018, 31, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhang, H.; Liu, J.; Shang, G. Targeted therapy for osteosarcoma: a review. J Cancer Res Clin Oncol 2023, 149, 6785–6797. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Tan, X.; Jiang, Z.; Wang, H.; Yuan, H. Immune checkpoint inhibitors in osteosarcoma: A hopeful and challenging future. Front Pharmacol 2022, 13, 1031527. [Google Scholar] [CrossRef] [PubMed]
- Panczyszyn, E.; Saverio, V.; Monzani, R.; Gagliardi, M.; Petrovic, J.; Stojkovska, J.; Collavin, L.; Corazzari, M. FSP1 is a predictive biomarker of osteosarcoma cells' susceptibility to ferroptotic cell death and a potential therapeutic target. Cell Death Discov 2024, 10, 87. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Farooq, I.; Iqbal, K. A review of the effect of various ions on the properties and the clinical applications of novel bioactive glasses in medicine and dentistry. Saudi Dent J 2014, 26, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.; Ferreira, F.V.; Lopes, J.H.; Camilli, J.A.; Martin, R.A. Cancer Inhibition and In Vivo Osteointegration and Compatibility of Gallium-Doped Bioactive Glasses for Osteosarcoma Applications. ACS Appl Mater Interfaces 2022, 14, 45156–45166. [Google Scholar] [CrossRef]
- Miola, M.; Massera, J.; Cochis, A.; Kumar, A.; Rimondini, L.; Verne, E. Tellurium: A new active element for innovative multifunctional bioactive glasses. Mater Sci Eng C Mater Biol Appl 2021, 123, 111957. [Google Scholar] [CrossRef]
- Miola, M.; Verne, E. Bioactive and Antibacterial Glass Powders Doped with Copper by Ion-Exchange in Aqueous Solutions. Materials (Basel) 2016, 9. [Google Scholar] [CrossRef]
- Miola, M.; Brovarone, C.V.; Maina, G.; Rossi, F.; Bergandi, L.; Ghigo, D.; Saracino, S.; Maggiora, M.; Canuto, R.A.; Muzio, G.; et al. In vitro study of manganese-doped bioactive glasses for bone regeneration. Mater Sci Eng C Mater Biol Appl 2014, 38, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Verne, E.; Di Nunzio, S.; Bosetti, M.; Appendino, P.; Brovarone, C.V.; Maina, G.; Cannas, M. Surface characterization of silver-doped bioactive glass. Biomaterials 2005, 26, 5111–5119. [Google Scholar] [CrossRef]
- Zhong, C.L.; Qin, B.Y.; Xie, X.Y.; Bai, Y. Antioxidant and Antimicrobial Activity of Tellurium Dioxide Nanoparticles Sols. J Nano Res-Sw 2013, 25, 8–15. [Google Scholar] [CrossRef]
- Kovrlija, I.; Panczyszyn, E.; Demir, O.; Laizane, M.; Corazzari, M.; Locs, J.; Loca, D. Doxorubicin loaded octacalcium phosphate particles as controlled release drug delivery systems: Physico-chemical characterization, in vitro drug release and evaluation of cell death pathway. Int J Pharm 2024, 653, 123932. [Google Scholar] [CrossRef] [PubMed]
- Monzani, R.; Gagliardi, M.; Clemente, N.; Saverio, V.; Panczyszyn, E.; Santoro, C.; Yissachar, N.; Visciglia, A.; Pane, M.; Amoruso, A.; et al. The Gut-Ex-Vivo System (GEVS) Is a Dynamic and Versatile Tool for the Study of DNBS-Induced IBD in BALB/C and C57BL/6 Mice, Highlighting the Protective Role of Probiotics. Biology (Basel) 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Pagliarini, V.; Giglio, P.; Bernardoni, P.; De Zio, D.; Fimia, G.M.; Piacentini, M.; Corazzari, M. Downregulation of E2F1 during ER stress is required to induce apoptosis. J Cell Sci 2015, 128, 1166–1179. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, Y.J.; Qiao, X.Y.; Lin, T.; Wang, Y.C.; Wang, M. Biomaterial-based strategy for bone tumor therapy and bone defect regeneration: An innovative application option. Front Mater 2022, 9. [Google Scholar] [CrossRef]
- Kaou, M.H.; Furko, M.; Balazsi, K.; Balazsi, C. Advanced Bioactive Glasses: The Newest Achievements and Breakthroughs in the Area. Nanomaterials (Basel) 2023, 13. [Google Scholar] [CrossRef]
- Sandoval, J.M.; Leveque, P.; Gallez, B.; Vasquez, C.C.; Buc Calderon, P. Tellurite-induced oxidative stress leads to cell death of murine hepatocarcinoma cells. Biometals 2010, 23, 623–632. [Google Scholar] [CrossRef]
- Rahaman, M.N.; Day, D.E.; Bal, B.S.; Fu, Q.; Jung, S.B.; Bonewald, L.F.; Tomsia, A.P. Bioactive glass in tissue engineering. Acta Biomater 2011, 7, 2355–2373. [Google Scholar] [CrossRef] [PubMed]
- Baniwal, S.K.; Shah, P.K.; Shi, Y.; Haduong, J.H.; Declerck, Y.A.; Gabet, Y.; Frenkel, B. Runx2 promotes both osteoblastogenesis and novel osteoclastogenic signals in ST2 mesenchymal progenitor cells. Osteoporos Int 2012, 23, 1399–1413. [Google Scholar] [CrossRef]
- Wang, X.; Hua, P.; He, C.; Chen, M. Non-apoptotic cell death-based cancer therapy: Molecular mechanism, pharmacological modulators, and nanomedicine. Acta Pharm Sin B 2022, 12, 3567–3593. [Google Scholar] [CrossRef] [PubMed]
- Ji, P.; Yu, L.; Guo, W.C.; Mei, H.J.; Wang, X.J.; Chen, H.; Fang, S.; Yang, J. Doxorubicin Inhibits Proliferation of Osteosarcoma Cells Through Upregulation of the Notch Signaling Pathway. Oncol Res 2014, 22, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Solania, A.; Gonzalez-Paez, G.E.; Wolan, D.W. Selective and Rapid Cell-Permeable Inhibitor of Human Caspase-3. ACS Chem Biol 2019, 14, 2463–2470. [Google Scholar] [CrossRef] [PubMed]
- Dodson, M.; Castro-Portuguez, R.; Zhang, D.D. NRF2 plays a critical role in mitigating lipid peroxidation and ferroptosis. Redox Biol 2019, 23, 101107. [Google Scholar] [CrossRef] [PubMed]
- Hassannia, B.; Vandenabeele, P.; Vanden Berghe, T. Targeting Ferroptosis to Iron Out Cancer. Cancer Cell 2019, 35, 830–849. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Ardehali, H.; Min, J.; Wang, F. The molecular and metabolic landscape of iron and ferroptosis in cardiovascular disease. Nat Rev Cardiol 2023, 20, 7–23. [Google Scholar] [CrossRef] [PubMed]
- Cao, W.; Wang, L.; Xu, H.P. Selenium/tellurium containing polymer materials in nanobiotechnology. Nano Today 2015, 10, 717–736. [Google Scholar] [CrossRef]
- Wu, Y.; Guo, T.; Qiu, Y.; Lin, Y.; Yao, Y.; Lian, W.; Lin, L.; Song, J.; Yang, H. An inorganic prodrug, tellurium nanowires with enhanced ROS generation and GSH depletion for selective cancer therapy. Chem Sci 2019, 10, 7068–7075. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.C.; Song, X.X.; Sun, X.F.; Huang, J.; Zhong, M.Z.; Lotze, M.T.; Zeh, H.J.; Kang, R.; Tang, D.L. Identification of baicalein as a ferroptosis inhibitor by natural product library screening. Biochem Bioph Res Co 2016, 473, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Lovat, P.E.; Di Sano, F.; Corazzari, M.; Fazi, B.; Donnorso, R.P.; Pearson, A.D.; Hall, A.G.; Redfern, C.P.; Piacentini, M. Gangliosides link the acidic sphingomyelinase-mediated induction of ceramide to 12-lipoxygenase-dependent apoptosis of neuroblastoma in response to fenretinide. J Natl Cancer Inst 2004, 96, 1288–1299. [Google Scholar] [CrossRef] [PubMed]
- Creecy, A.; Damrath, J.G.; Wallace, J.M. Control of Bone Matrix Properties by Osteocytes. Front Endocrinol (Lausanne) 2020, 11, 578477. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Peng, Y.; Li, Y.; Kpegah, J.; Chen, S. Multifunctional inorganic biomaterials: New weapons targeting osteosarcoma. Front Mol Biosci 2022, 9, 1105540. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yang, J.; Zhao, N.; Wang, C.; Kamar, S.; Zhou, Y.; He, Z.; Yang, J.; Sun, B.; Shi, X.; et al. Progress in the chemotherapeutic treatment of osteosarcoma. Oncol Lett 2018, 16, 6228–6237. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhao, J.; Feng, H. Targeting iron metabolism in osteosarcoma. Discov Oncol 2023, 14, 31. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, R.; Wang, W.; Wang, C.; Zhang, N.; Shao, X.; He, Q.; Ying, M. Advances in targeted therapy for osteosarcoma based on molecular classification. Pharmacol Res 2021, 169, 105684. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yao, Q.; Peng, Y.; Dong, Z.; Tang, L.; Su, X.; Liu, L.; Chen, C.; Ramalingam, M.; Cheng, L. Identification of Small-Molecule Inhibitors for Osteosarcoma Targeted Therapy: Synchronizing In Silico, In Vitro, and In Vivo Analyses. Front Bioeng Biotechnol 2022, 10, 921107. [Google Scholar] [CrossRef] [PubMed]
- Shiravand, Y.; Khodadadi, F.; Kashani, S.M.A.; Hosseini-Fard, S.R.; Hosseini, S.; Sadeghirad, H.; Ladwa, R.; O'Byrne, K.; Kulasinghe, A. Immune Checkpoint Inhibitors in Cancer Therapy. Curr Oncol 2022, 29, 3044–3060. [Google Scholar] [CrossRef]
- Ritter, J.; Bielack, S.S. Osteosarcoma. Ann Oncol 2010, 21 Suppl 7, vii320–325. [Google Scholar] [CrossRef]
- de Azevedo, J.W.V.; de Medeiros Fernandes, T.A.A.; Fernandes, J.V., Jr.; de Azevedo, J.C.V.; Lanza, D.C.F.; Bezerra, C.M.; Andrade, V.S.; de Araujo, J.M.G.; Fernandes, J.V. Biology and pathogenesis of human osteosarcoma. Oncol Lett 2020, 19, 1099–1116. [Google Scholar] [CrossRef] [PubMed]
| % mol | ||||||
| STe0 | STe5 | |||||
| SiO2 | 48.6 | 43.6 | ||||
| Na2O | 16.7 | 16.7 | ||||
| CaO | 34.2 | 34.2 | ||||
| P2O5 | 0.5 | 0.5 | ||||
| TeO2 | 0.0 | 5.0 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




