Submitted:
27 August 2024
Posted:
28 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
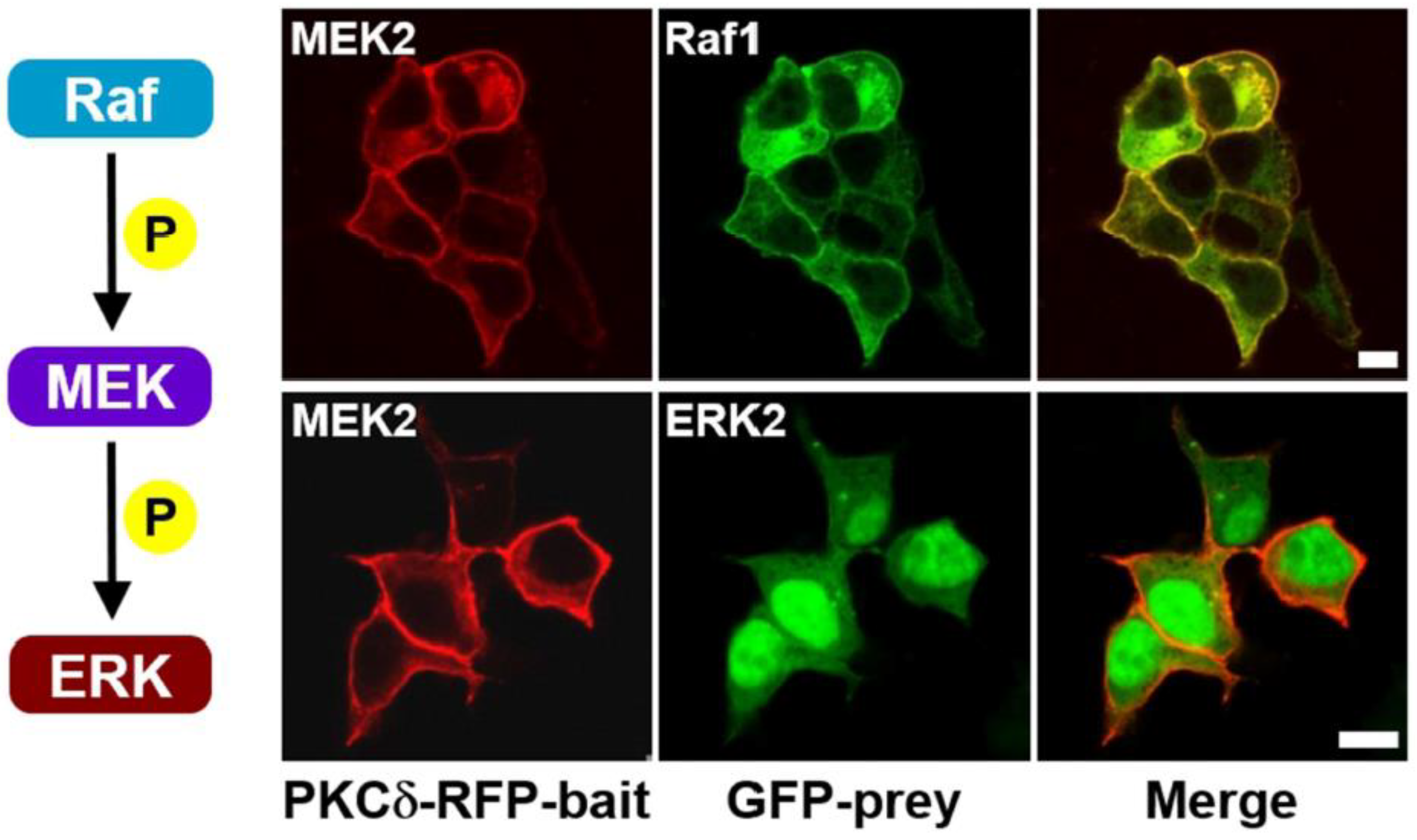
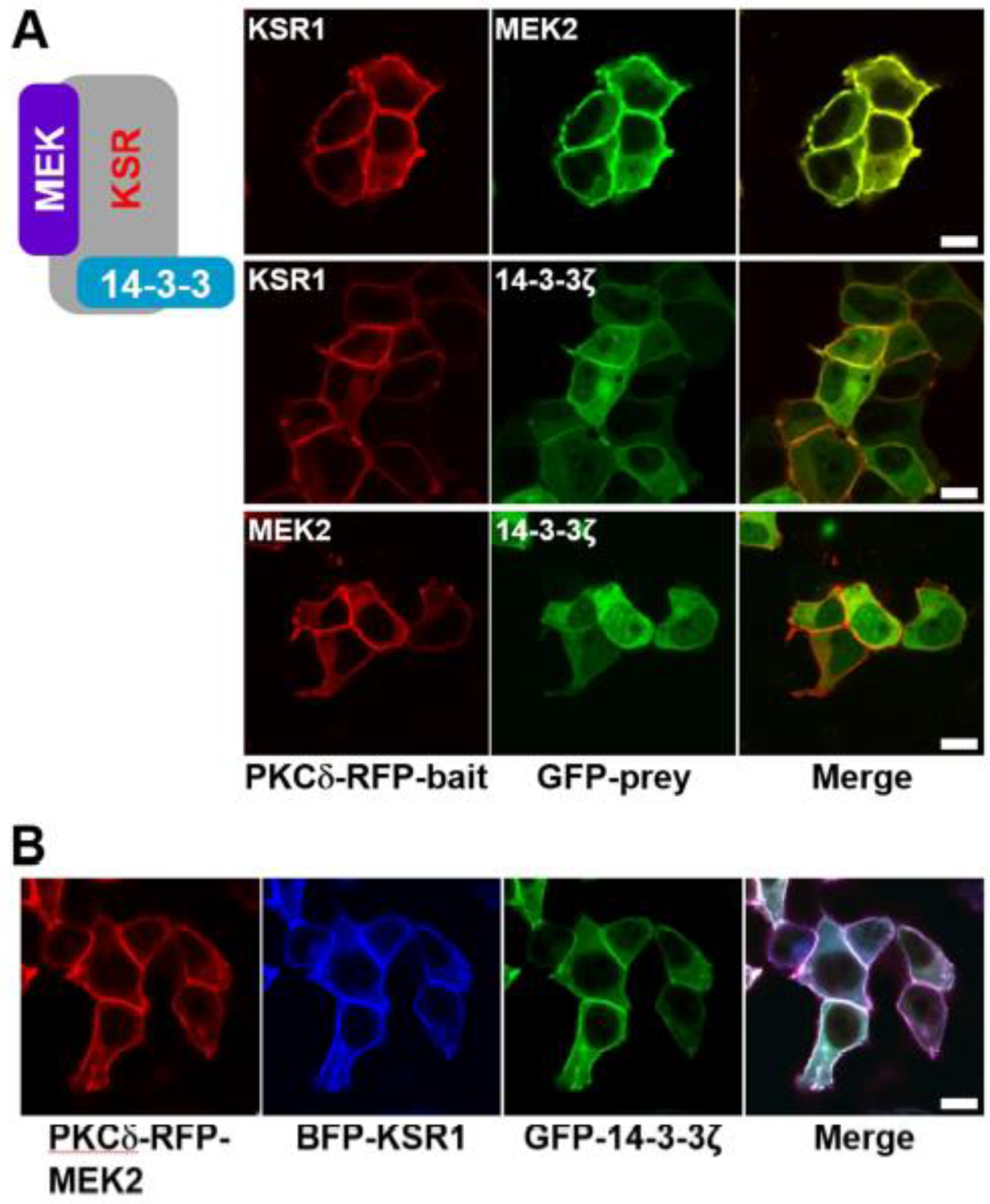
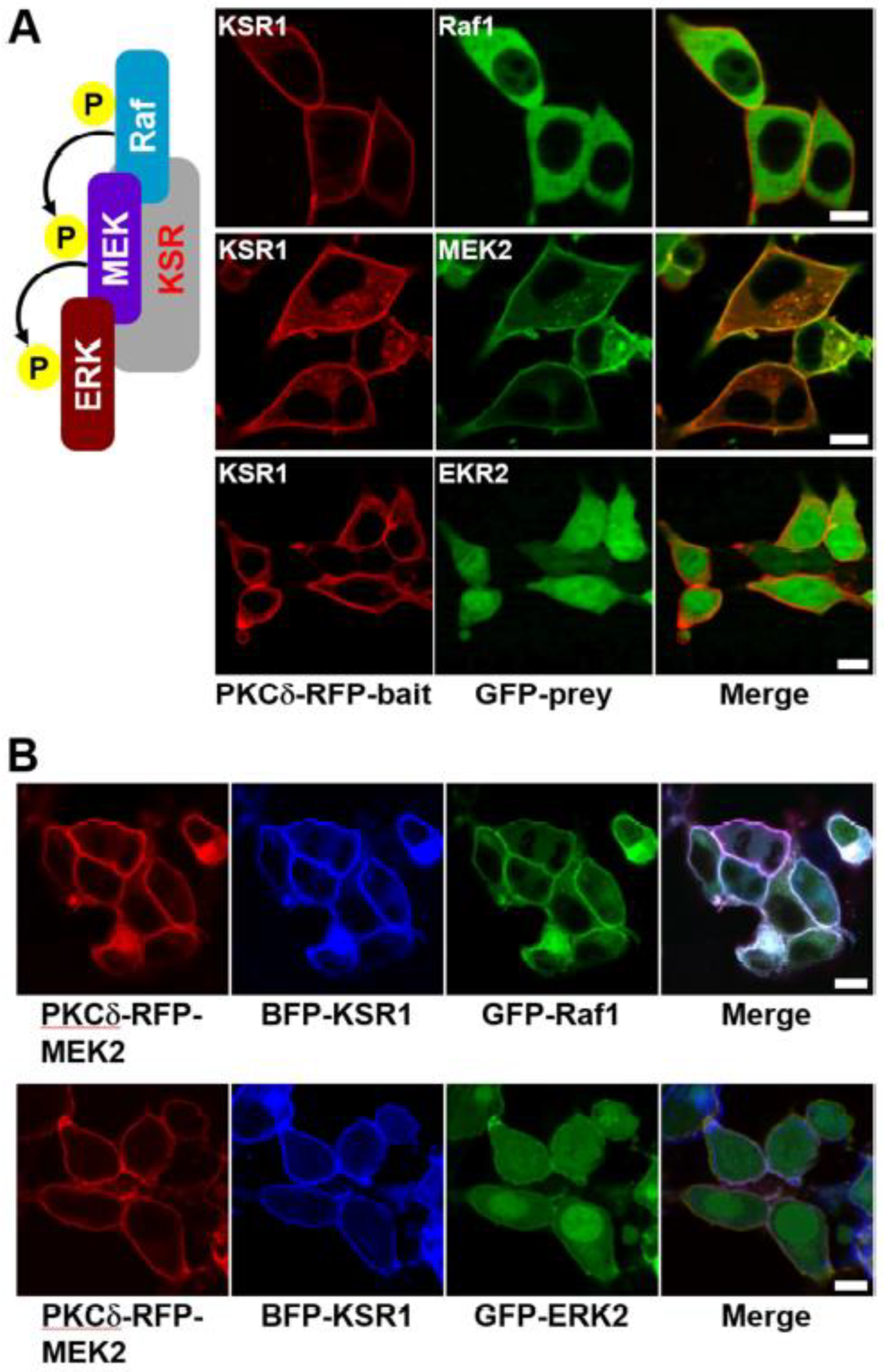
2. Results and Discussion
3. Materials and Methods
Construction of Bait/Prey Expression Vectors
Co-Expression of the Bait/Prey Protein Pair in Cultured Cells
Confocal Imaging
Co-Immunoprecipitation (Co-IP) and GST Pull-Down Assays
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, W.; Liu, H.T. MAPK signal pathways in the regulation of cell proliferation in mammalian cells. Cell Res. 2002, 12, 9–18. [CrossRef]
- Ma, M.; Bordignon P.; Dotto, G.P.; Pelet, S. Visualizing cellular heterogeneity by quantifying the dynamics of MAPK activity in live mammalian cells with synthetic fluorescent biosensors. Heliyon 2020, 6, e05574. [CrossRef]
- Ullah, R.; Yiu, Q.; Snell, A.H.; Wan, L. RAF–MEK–ERK pathway in cancer evolution and treatment. Semin. Cancer Biol. 2022, 85, 123–154. [CrossRef]
- Martin-Vega, A.; Cobb, M.H. Navigation the ERK1/2 MAPK cascade. Biomolecules 2023, 13, 1555.
- McKay, M.M.; Ritt, D.A.; Morrison, D.K. Signaling dynamics of the KSR1 scaffold complex. Proc. Natl. Acad. Sci. U.S.A. 2009, 106, 11022–11027. [CrossRef]
- Meister, M.; Tomasovic, A.; Banninb, A.; Tikkanen, R. Mitogen-activated protein (MAP) kinase scaffolding proteins: a recount. Int. J. Mol. Sci. 2013, 14, 4854–4884. [CrossRef]
- Kolch, W. Coordination ERK/MAPK signaling through scaffolds and inhibitors. Nat. Rev. Mol. Cell Biol. 2005, 6, 827–837. [CrossRef]
- Parvathaneni, S.; Li, Z.; Sacks, D.B. Calmodulin influences MAPK signaling by binding KSR1. J. Biol. Chem. 2021, 296, 100577. [CrossRef]
- Shaul, Y.D.; Seger, R. The MEK/ERK cascade: From signaling specificity to diverse functions. Biochim. Biophys. Acta 2007, 1773, 1213–1226. [CrossRef]
- Liu, Z.; Krstic, A.; Neve, A.; Casalou, C.; Rauch, N.; Wynne, K.; Cassidy, H.; McCann, A.; Kavanagh, E.; McCann, B.; Blanco, A.; Rauch, J.; Kolch, W. Kinase suppressor of RAS 1 (KSR1) maintains the transformed phenotype of BRAFV600E mutant human melanoma cells. Int. J. Mol. Sci. 2023, 24, 11821. [CrossRef]
- Maloney, R.C.; Zhang, M.; Liu, Y.; Jang, H.; Nussinov, R. The mechanism of activation of MEK1 by B–Raf and KSR1. Cell. Mol. Life Sci. 2022, 79, 281. [CrossRef]
- Lyons, A.C.; Mehta, S.; Zhang, J. Fluorescent biosensors illuminate the spatial regulation of cell signaling across scales. Biochem. J. 2023, 480, 1693–1717. [CrossRef]
- Newman, R.H.; Fosbrink, M.D.; Zhang, J. Genetically encodable fluorescent biosensors for tracking signaling dynamics in living cells. Chem. Rev. 2011, 111, 3614–3666. [CrossRef]
- Tomida, T. Visualization of the spatial and temporal dynamics of MAPK signaling using fluorescence imaging techniques. J. Physiol. Sci. 2015, 65, 37–49. [CrossRef]
- Peterson, A.F.; Ingram K, Huang, E.J.; Parksong, J.; McKenney, C.; Bever, G.S.; Regot, S. Systematic analysis of the MAPK signaling network reveals MAP3K-driven control of cell fate. Cell Syst. 2022, 13, 885–894. [CrossRef]
- Weeks, R.; Mehta, S.; Zhang, J. Genetically encodable biosensors for Ras activity. RSC Chem. Biol. 2024, 5, 312–320. [CrossRef]
- Zaver, S.A.; Johnson, C.J.; Berndt, A.; Simpson, C.L. Live imaging with genetically encoded physiological sensors and optogenetic tools. J. Invest. Dermatol. 2023, 143, 353–361. [CrossRef]
- Heydorn, A.; Lundholt, B.K.; Praestegaard, M.; Pagliaro, L. Protein translocation assays: key tools for accessing new biological information with high-throughput microscopy. Methods Enaymol. 2006, 414, 513–530.
- Piljić, A.; Schultz, C. Analysis of protein complex hierarchy in living cells. ACS Chem. Biol. 2008, 3, 749–755. [CrossRef]
- Mentrup, T.; Häsler, R.; Fluhrer, R.; Saftig, P.; Schröder, B. A cell-based assay reveals nuclear translocation of intracellular domains released by SPPL proteases. Traffic 2015, 16, 871–892. [CrossRef]
- Jain, R.; Saini D.K. Imaging cellular signalling: many ‘moving tales’ in MAP kinase odyssey. Curr. Sci. 2013, 105, 1512–1523.
- Lee, K.B.; Hwang, J.M.; Choi, I.S.; Rho, J.; Choi, J.S.; Kim, G.H.; Kim, S.I.; Kim, S.; Lee, Z.W. Direct monitoring of the inhibition of protein-protein interaction in cells by translocation of PKCδ fusion proteins. Angew. Chem. Int. Ed. 2011, 50, 1314–1317.
- Hwang, J.M.; Lee, K.B.; Choi, J.S.; Rho, J.; Kim, G.H.; Kim, S.I.; Kim, S.; Lee, Z.W. Novel technology for protein-protein interaction-based targeted drug discovery. J. Anal. Sci. Technol. 2011, 2, A61–A65. [CrossRef]
- Wang, Q.J.; Bhattacharyya, D.; Garfield, S.; Nacro, K.; Marquez, V.E.; Blumberg, P.M. Differential localization of protein kinase C δ by phorbol esters and related compounds using a fusion protein with green fluorescent protein. J. Biol. Chem. 1999, 274, 37233–37239. [CrossRef]
- Lim, P.S.; Sutton, C.R.; Rao, S. Protein kinase C in the immune system: from signalling to chromatin regulation. Immunology 2015, 146, 508–522. [CrossRef]
- Kawano, T.; Inokuchi, J.; Eto, M.; Murata, M.; Kang, J.H. Protein kinase C (PKC) isozymes as diagnostic and prognostic biomarkers and therapeutic targets for cancer. Cancers 2022, 14, 5425. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



