Submitted:
26 August 2024
Posted:
28 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Maintenance and Preservation of Strain
2.2. Bioreactor Cultures
2.3. Determination of Growth Kinetics and P3HB Production
2.4. Molecular Weight Determination of P3HB
3. Results and Discussion
3.1. Growth kinetics, OTR Profiles and qO₂ in Bioreactor Cultures at 300 and 500 rpm with OP, OP-PhbP2 ̄ and OP-PhbP3 ̄ Strain
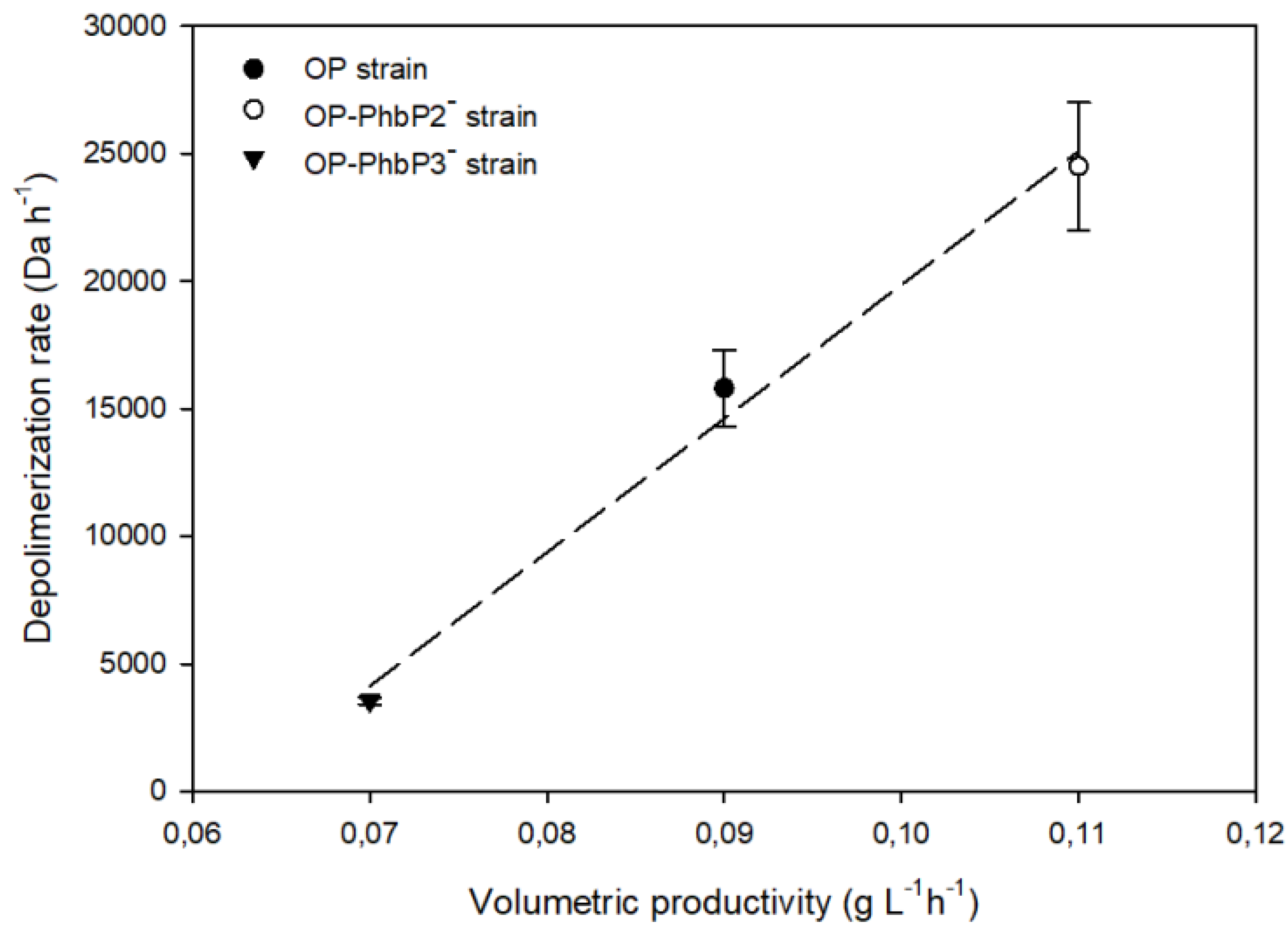
3.2. P3HB Production at 300 and 500 rpm Using the OP and Mutant Strains
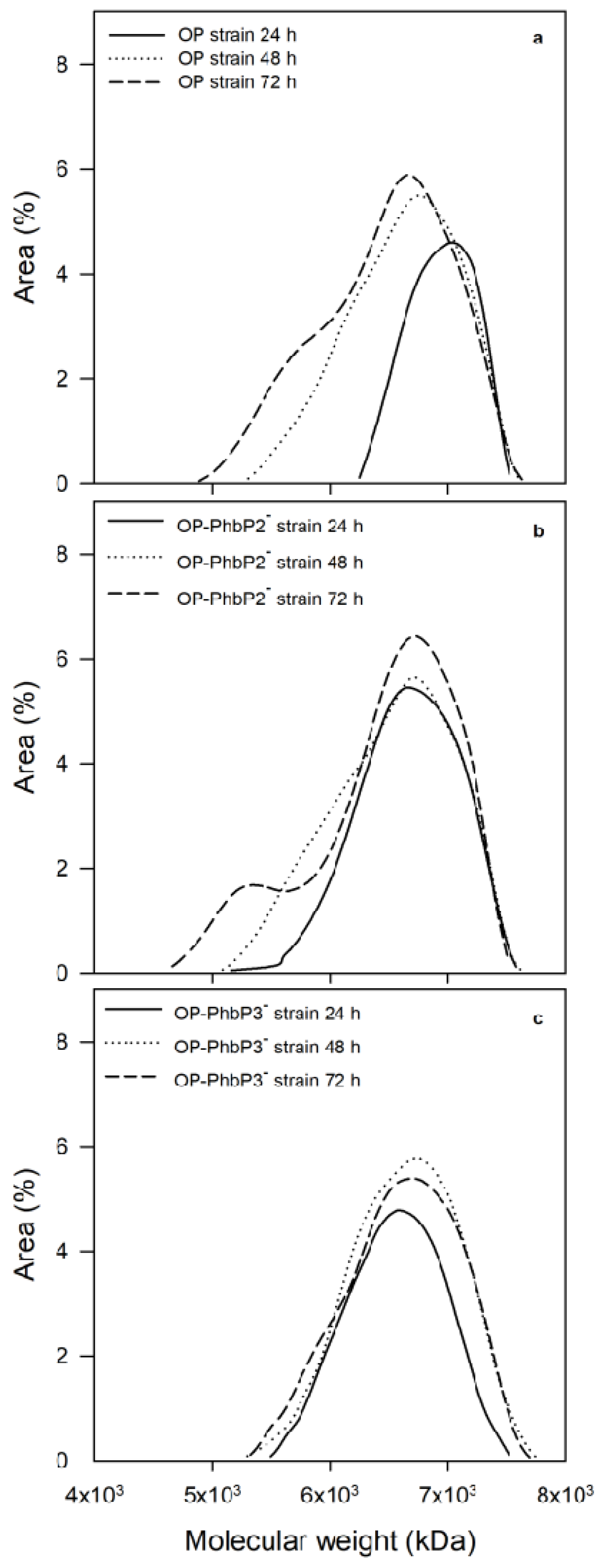
3.3. Molecular Weight Distributions of Polymers Produced by the OP and Mutant Strains
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Philip, S.; Keshavarz, T.; Roy, I. Polyhydroxyalkanoates: biodegradable polymers with a range of applications. J. Chem. Technol. Biotechnol. 2007, 82, 233–247. [Google Scholar] [CrossRef]
- Peña, C.; Castillo, T.; García, A.; Millán, M.; Segura, D. Biotechnological strategies to improve the production of microbial poly-(3-hydroxyburyrate): a review of recent research work. Microbiol Biotechnol. 2014, 7, 278–293. [Google Scholar] [CrossRef] [PubMed]
- Iwata, T. Strong fibers and films of microbial polyesters. Macromol Bios. 2005, 5, 689–701. [Google Scholar] [CrossRef] [PubMed]
- Magagula, S.I.; Mohapi, M.; Sefadi, J.S.; Mochane, M.J. The production and applications of microbial-derived polyhydroxybutyrates. In Microbial Polymers: Applications and Ecological Perspectives. Vaishnav & Choudhary (Eds); Springer Nature. Singapore, 2021, pp. 3-43.
- Andler, R.; Rojas, V.; Pino, V.; Castro, R. I.; Valdés, C.; Kumar, V.; Peña, C.; Díaz-Barrera, A. Efficient production of a polyhydroxyalkanoate by Azotobacter vinelandii OP using apple residues as promising feedstock. Int. J. Biol. Macromol. 2023, 242, 124626. [Google Scholar] [CrossRef] [PubMed]
- Jendrossek, D.; Pfeiffer, D. New insights in the formation of polyhydroxyalkanoate granules (carbonosomes) and novel functions of poly(3-hydroxybutyrate). Environ Microbiol. 2014, 16, 2357–2373. [Google Scholar] [CrossRef] [PubMed]
- Mezzina, M.; Pettinari, J. Phasins, multifaceted polyhydroxyalkanoate granule-associated proteins. Appl. Environ. Microbiol. 2016, 82, 5060–5067. [Google Scholar] [CrossRef]
- Soo-Lee, H.; Lee, H.; Kim, S.; Cho, J.; Suh, M.; Ham, S.; Bhatia, S.; Gurav, R.; Kim, Y.; Lee, E.; Yang, Y. Novel phasins from the arctic Pseudomonas sp. B14-6 enhances the production of polyhydroxybutyrate and increases inhibitor tolerance. Int. J. Biol. Macromol. 2021, 190, 722–729. [Google Scholar]
- Tang, R.; Peng, X.; Weng, C.; Han, Y. The overexpression of phasin and regulator genes promoting the synthesis of polyhydroxybutyrate in Cupriavidus necator H16 under nonstress conditions. Appl. Environ. Microbiol. 2022, 88, e01458–21. [Google Scholar] [CrossRef]
- Handrick, R.; Technow, U.; Reichart, T.; Reinhardt, S.; Sander, T.; Jendrossek, D. The activator of the Rhodospirillum rubrum PHB depolymerase is a polypeptide that is extremely resistant to high temperature (121°C) and other physical or chemical stresses. FEMS Microbiol. Lett. 2004, 230, 265–274. [Google Scholar] [CrossRef]
- Pötter, M.; Steinbüchel, A. Poly(3-hydroxybutyrate) granule-associated proteins: Impacts on poly(3-hydroxybutyrate) synthesis and degradation. Biomacromolecules. 2005, 6, 552–560. [Google Scholar] [CrossRef]
- de Almeida, A.; Nikel, P.I.; Giordano, A.M.; Pettinari, M.J. Effects of granule-associated protein PhaP on glycerol-dependent growth and polymer production in poly (3-hydroxybutyrate)-producing Escherichia coli. Appl. Environ. Microbiol. 2007, 73, 7912–7916. [Google Scholar] [CrossRef] [PubMed]
- Quiroz-Cardoso, R.; Castillo, T.; Galindo, E.; Ruíz Escobedo, J.; Segura, D.; Peña, C. Looking for improved strains of Azotobacter vineladii and favorable culture conditions yielding high molecular weight Poly-3-hydoxybutyrate (P3HB). J. Chem. Technol. Biotechnol. 2024. submitted. [Google Scholar]
- Díaz-Barrera, A.; Andler, R.; Martínez, I.; Peña, C. Poly-3-hydroxybutyrate production by Azotobacter vinelandii strains in batch cultures at different oxygen transfer rates. J. Chem. Technol. Biotechnol. 2016, 91, 1063–1071. [Google Scholar] [CrossRef]
- Díaz-Barrera, A.; Sanchez-Rosales, F.; Padilla-Córdova, C.; Andler, R.; Peña, C. Molecular weight and guluronic/mannuronic ratio of alginate produced by Azotobacter vinelandii at two bioreactor scales under diazotrophic conditions. Bioprocess Biosyst. Eng. 2021, 44, 1275–1287. [Google Scholar] [CrossRef]
- García-Cabrera, R. I.; Valdez-Cruz, N. A.; Blancas-Cabrera, A.; Trujillo-Roldán, M. A. Oxygen transfer rate affect polyhydroxybutyrate production and oxidative stress response in submerged cultures of Rhizobium phaseoli. Biochem. Eng. J. 2020, 162, 107721. [Google Scholar] [CrossRef]
- Gómez-Hernández, E.; Salgado-Lugo, H.; Segura, D.; García, A.; Díaz-Barrera, A.; Peña, C. Production of poly-3-hydroxybutyrate (P3HB) with ultra-high molecular weight (UHMW) by mutant strains of Azotobacter vinelandii under microaerophilic conditions. Appl. Biochem. Biotechnol. 2020, 193, 79–95. [Google Scholar] [CrossRef]
- Ruíz Escobedo, J. Estudio del papel de las proteínas Avin34710 y Avin34720 en el metabolismo de polihidroxibutirato (PHB) en la bacteria Azotobacter vinelandii. M. sC thesis, Universidad Nacional Autónoma de México, México, 2020.
- García, A.; Segura, D.; Espín, G.; Galindo, E.; Castillo, T.; Peña, C. High production of poly-β-hydroxybutyrate (PHB) by an Azotobacter vinelandii mutant altered in PHB regulation using a fed-batch fermentation process. Biochem. Eng. J. 2014, 82, 117–123. [Google Scholar] [CrossRef]
- Castillo, T.; Heinzle, E.; Peifer, S.; Schneider, K.; Peña, C. Oxygen supply strongly influences metabolic fluxes, the production of poly(3-hydroxybutyrate) and alginate, and the degree of acetylation of alginate in Azotobacter vinelandii. Proc. Biochem. 2013, 48, 995–1003. [Google Scholar] [CrossRef]
- García, A.; Ferrer, P.; Albiol, J.; Castillo, T.; Segura, D.; Peña, C. Metabolic flux analysis and the NAD(P)H/NAD(P)(+) ratios in chemostat cultures of Azotobacter vinelandii. Microb. Cell Fact. 2018, 17, 10. [Google Scholar] [CrossRef]
- de Almeida, A.; Catone, M. V.; Rhodius, V. A.; Gross, C. A.; Pettinari, M. J. Unexpected stress-reducing effect of PhaP, a poly(3- hydroxybutyrate) granule-associated protein, in Escherichia coli. Appl. Environ. Microbiol. 2011, 77, 6622–6629. [Google Scholar] [CrossRef]
- Mezzina, M.P.; Wetzler, D.E.; de Almeida, A.; Dinjaski, N.; Prieto, M.A.; Pettinari, M.J. A phasin with extra talents: a polyhydroxyalkanoate granule-associated protein has chaperone activity. Environ. Microbiol. 2015, 17, 1765–1776. [Google Scholar] [CrossRef] [PubMed]
- Bresan, S.; Sznajder, A.; Hauf, W.; Forchhammer, K.; Pfeiffer, D.; Jendrossek, D. Polyhydroxyalkanoate (PHA) granules have no phospholipids. Sci Rep. 2016, 6, 26612. [Google Scholar] [CrossRef]
- Pettinari, J.M.; Chaneton, L.; Vazquez, G.; Steinbuchel, A.; Mendez, B.S. Insertion sequence-like elements associated with putative polyhydroxybutyrate regulatory genes in Azotobacter sp. FA8. Plasmid. 2003, 50, 36–44. [Google Scholar] [CrossRef]
- Millán, M.; Segura, D.; Galindo, E.; Peña, C. Molecular mass of poly-3-hydroxybutyrate (P3HB) produced by Azotobacter vinelandii is determined by the ratio of synthesis and degradation under fixed dissolved oxygen tension. Process Biochem. 2016, 51, 950–958. [Google Scholar] [CrossRef]
- Adaya, L.; Millán, M.; Peña, C.; Jendrossek, D.; Espín, G.; Tinoco-Valencia, R.; Guzmán, J.; Pfeiffer, D.; Segura, D. Inactivation of an intracellular poly-3-hydroxybutyrate depolymerase of Azotobacter vinelandii allows to obtain a polymer of uniform high molecular mass. Appl. Microbiol. Biotechnol. 2018, 102, 2693–2707. [Google Scholar] [CrossRef]
- Millán, M.; Salazar, M.; Segura, D.; Castillo, T.; Díaz-Barrera, A.; Peña, C. Molecular mass of Poly-3-hydroxybutyrate (P3HB) produced by Azotobacter vinelandii is influenced by the polymer content in the inoculum. J. Biotechnol. 2017, 259, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.J.; Lai, W.J.; Zheng, Z.; Wang, H.X.; Chen, G.Q. Effect of over-expression of phasin gene from Aeromonas hydrophila on biosynthesis of copolyesters of 3- hydroxybutyrate and 3-hydroxyhexanoate. FEMS Microbiol. Lett. 2005, 244, 19–25. [Google Scholar] [CrossRef]
- Hiroe, A.; Ushimaru, K.; Tsuge, T. Characterization of polyhydroxyalkanoate (PHA) synthase derived from Delftia acidovorans DS-17 and the influence of PHA production in Escherichia coli. J. Biosci. Bioeng. 2013, 115, 633–638. [Google Scholar] [CrossRef]
- Kusaka, S.; Abe, H.; Lee, S.; Doi, Y. Molecular mass of poly [(R)-3-hydroxybutyric acid] produced in a recombinant Escherichia coli. Appl. Microbiol. Biotechnol. 1997, 47, 140–143. [Google Scholar] [CrossRef]
- Hiroe, A.; Tsuge, K.; Nomura, C. T.; Itaya, M.; Tsuge, T. Rearrangement of gene order in the phaCAB operon leads to effective production of ultrahigh-molecular-weight poly [(R)-3-hydroxybutyrate] in genetically engineered Escherichia coli. Appl. Microbiol. Biotechnol. 2012, 78, 3177–3184. [Google Scholar] [CrossRef]
- Penloglou, G.; Kretza, E.; Chatzidoukas, C.; Parouti, S.; Kiparissides, C. On the control of molecular weight distribution of polyhydroxybutyrate in Azohydromonas lata cultures. Biochem. Eng. J. 2012, 62, 39–47. [Google Scholar] [CrossRef]
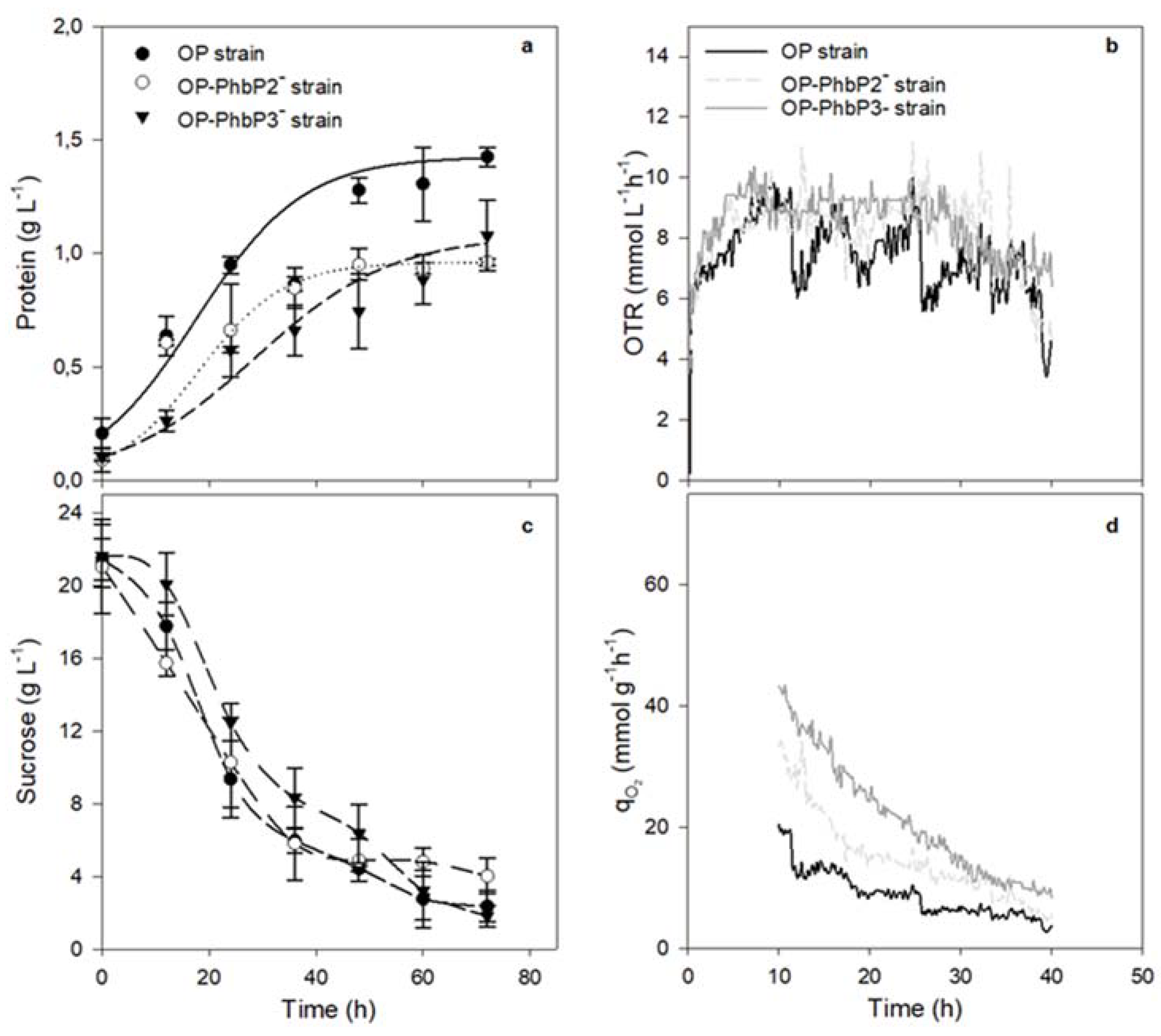
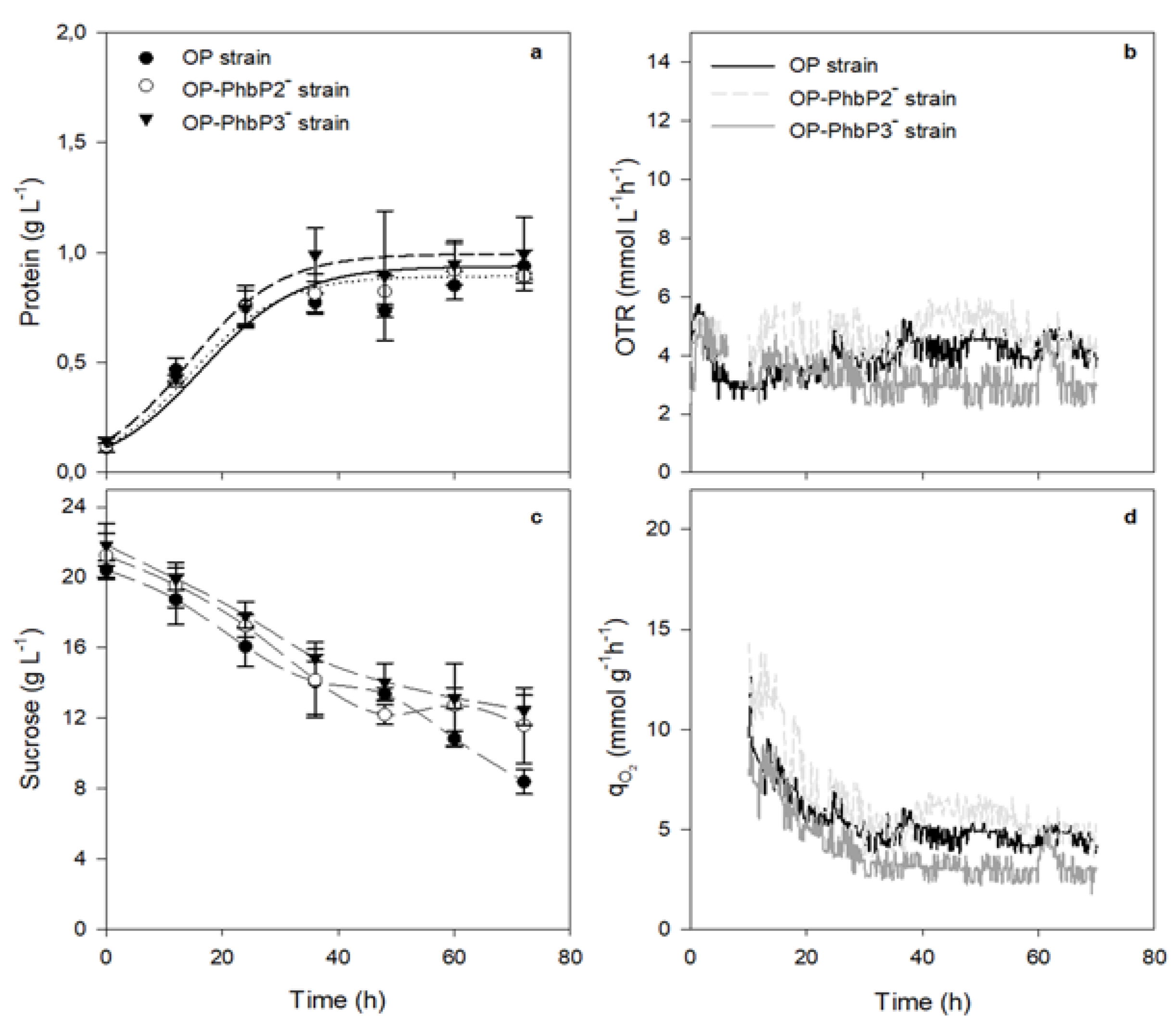
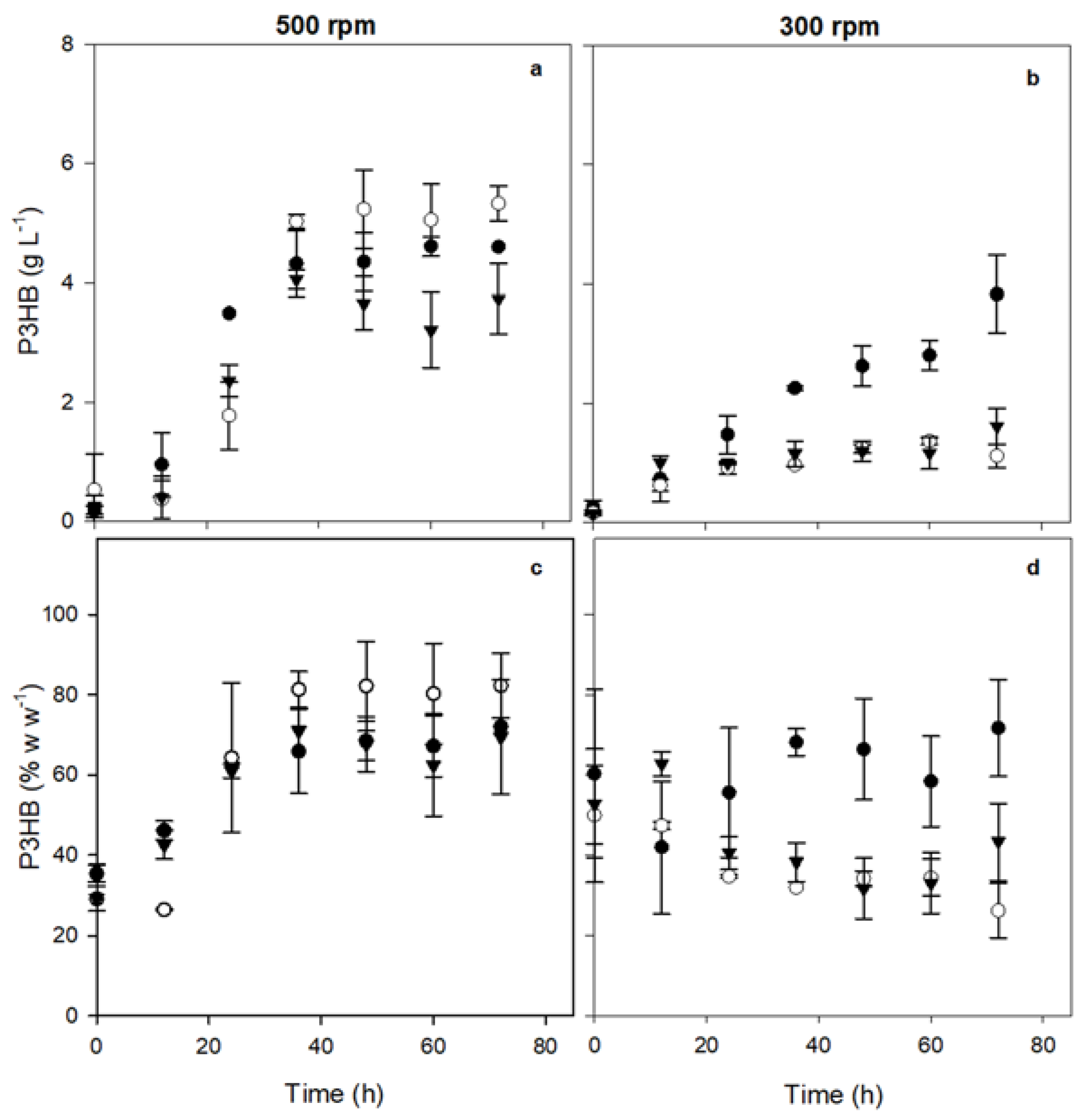
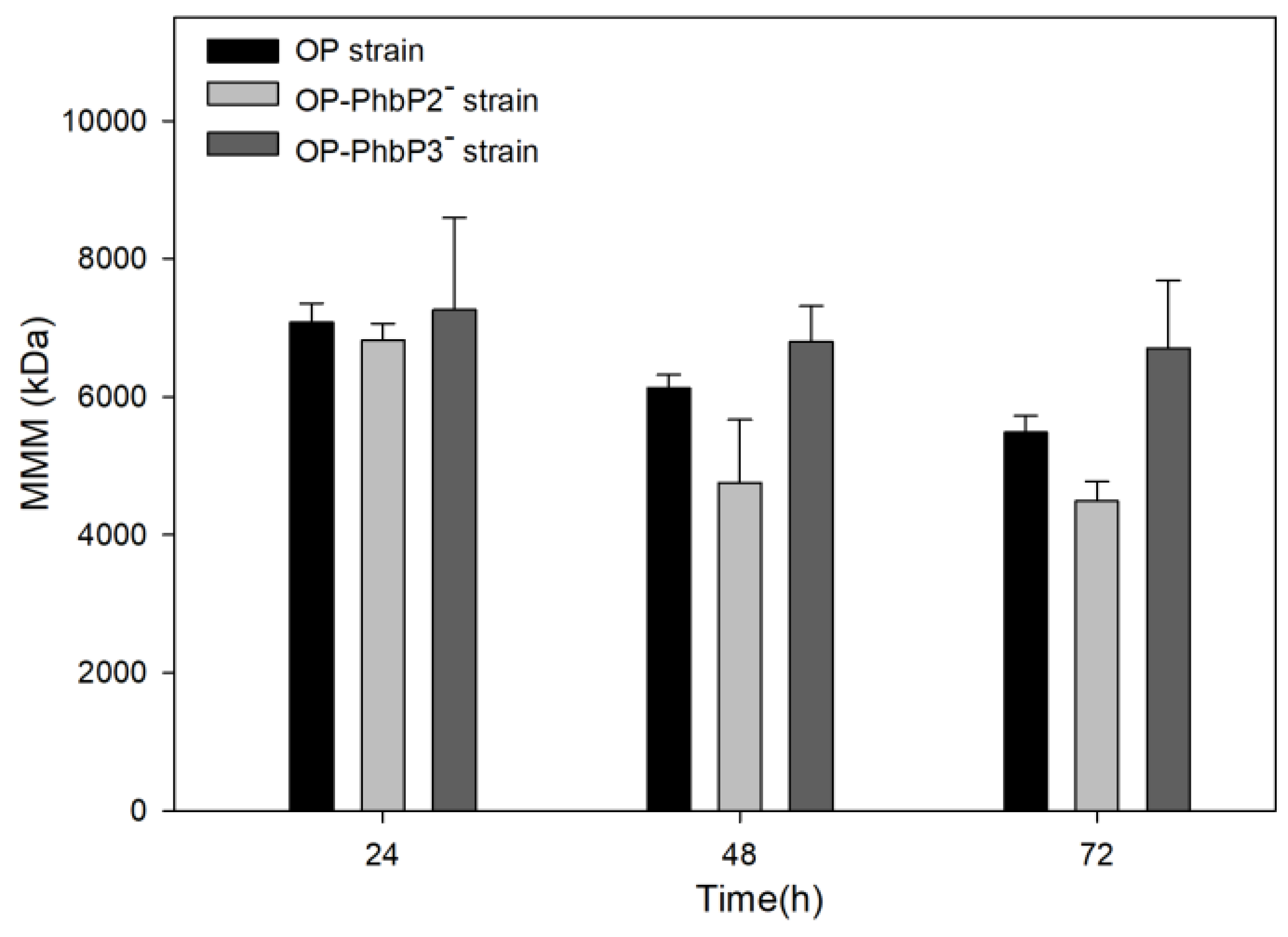
| Strain | Agitation rate (rpm) | OTRₘₐₓ (mmol L⁻¹ h⁻¹) |
qO₂ ᵃ (mmoL g⁻¹ h⁻¹) |
μ (h⁻¹) |
Cellular protein (g L⁻¹) |
|---|---|---|---|---|---|
| OP | 500 | 8.30 ± 0.56 | 9.19 ± 1.01 | 0.08 ± 0.020 | 1.43 ± 0.04 |
| 300 | 3.91 ± 0.90 | 6.88 ± 0.67 | 0.025 ± 0.001 | 0.94 ± 0.07 | |
| OP-PhbP2⁻ | 500 | 8.12 ± 0.24 | 25.5 ± 1.04 | 0.05 ± 0.010 | 0.96 ± 0.02 |
| 300 | 4.94 ± 0.92 | 12.07 ± 3.76 | 0.025 ± 0.001 | 0.89 ± 0.01 | |
| OP-PhbP3⁻ | 500 | 8.94 ± 0.79 | 37.33 ± 1.60 | 0.04 ± 0.010 | 1.08 ± 0.15 |
| 300 | 3.20 ± 1.00 | 6.93 ± 2.31 | 0.027 ± 0.001 | 0.99 ± 0.17 |
| Strain | Agitation rate (rpm) | OTRₘₐₓ (mmol L⁻¹ h⁻¹) |
P3HBₘₐₓ (g L⁻¹) |
% P3HBₘₐₓ (on dry weight) |
|---|---|---|---|---|
| OP | 500 | 8.30 ± 0.56 | 4.60 ± 0.77 | 71.00 ± 1.40 (72 h) |
| 300 | 3.91 ± 0.90 | 3.83 ± 0.65 | 68.99 ± 16.4 (48 h) | |
| OP-PhbP2⁻ | 500 | 8.12 ± 0.24 | 5.33 ± 0.30 | 82.25 ± 8.08 (72 h) |
| 300 | 4.94 ± 0.92 | 1.37 ± 0.06 | 47.25 ± 0.97 (12 h) | |
| OP-PhbP3⁻ | 500 | 8.94 ± 0.79 | 3.74 ± 0.59 | 73.92 ± 4.17 (36 h) |
| 300 | 3.20 ± 1.00 | 1.62 ± 0.29 | 67.14 ± 0.00 (12 h) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




