Submitted:
26 August 2024
Posted:
27 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Primary Analysis
2.2. Sensitivity Analysis
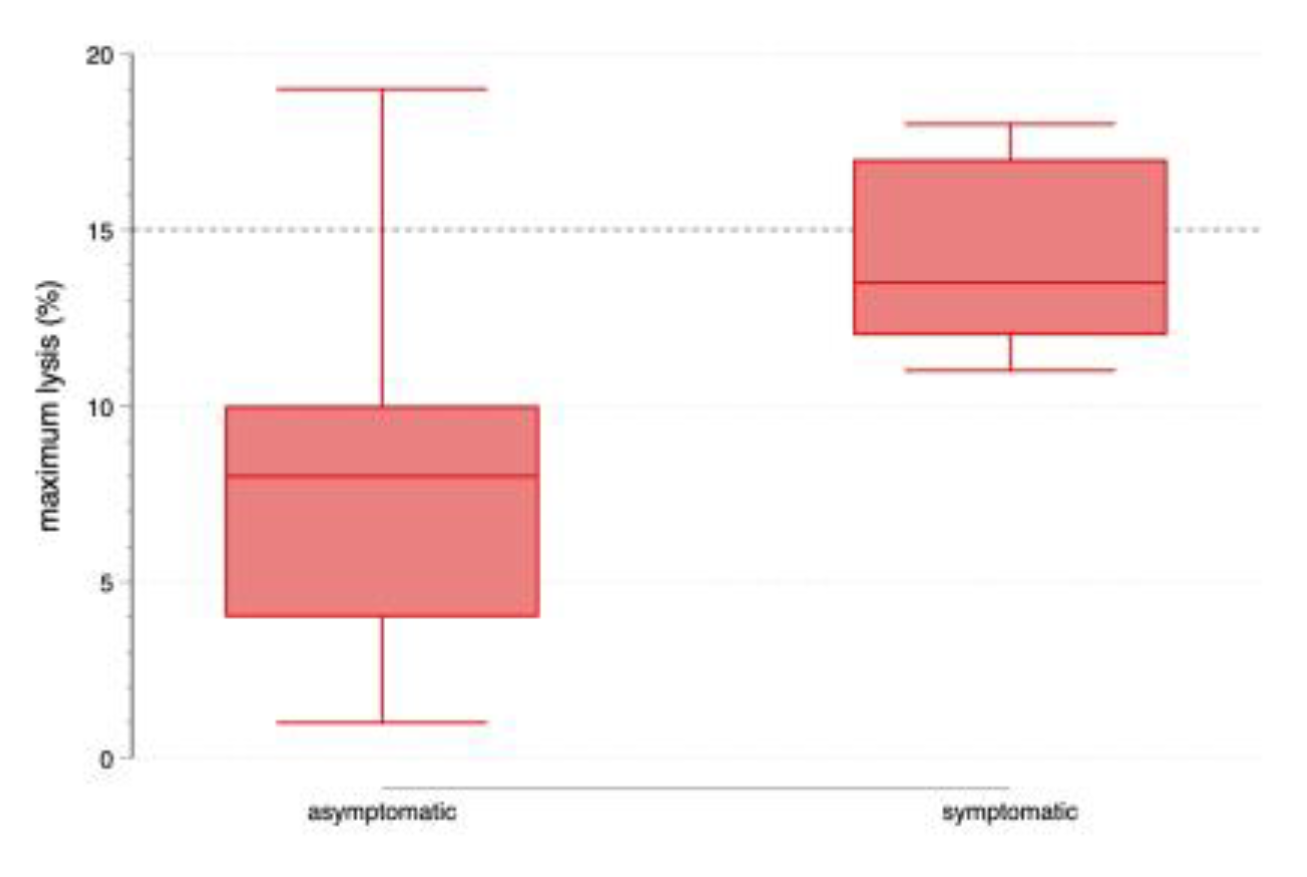
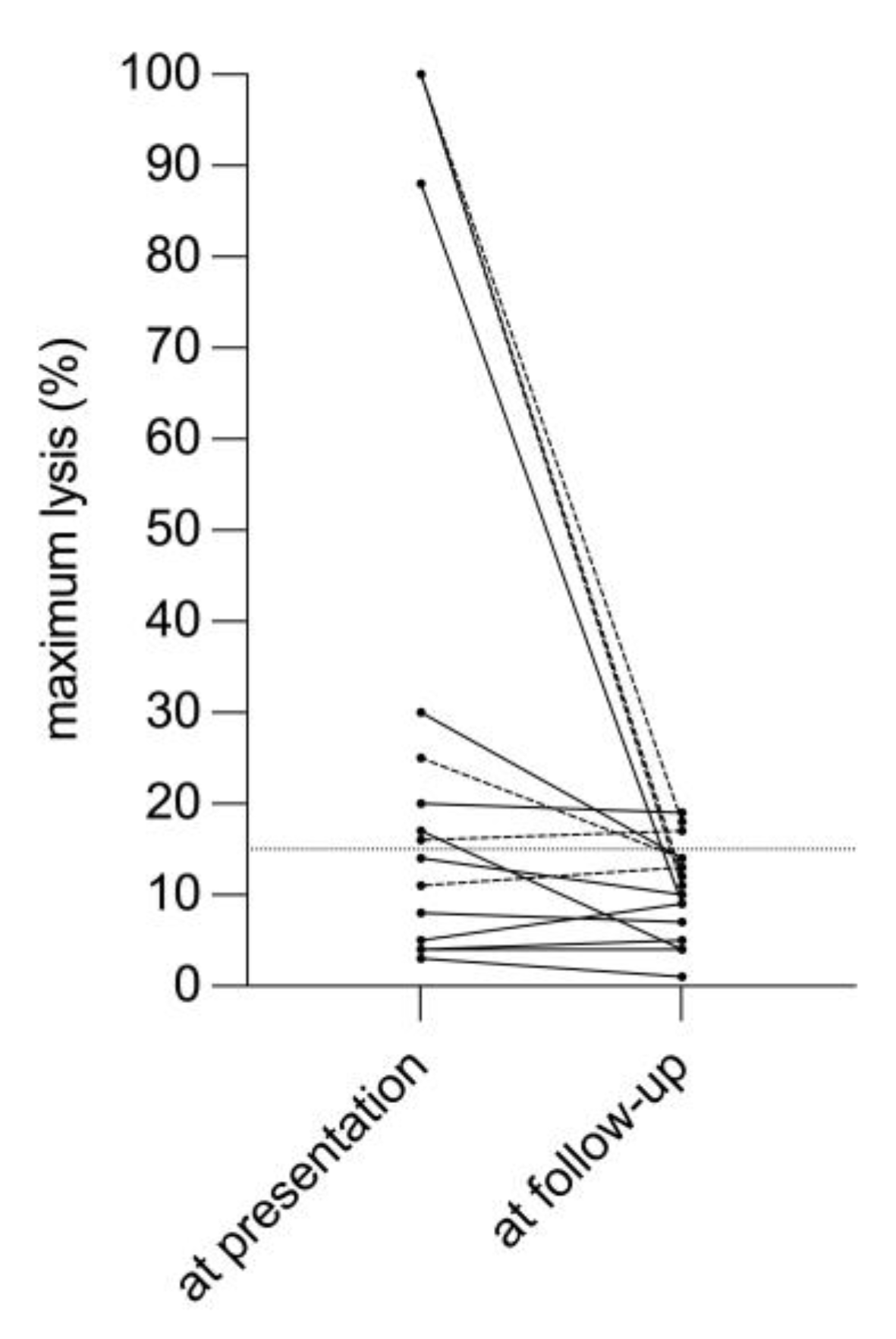
3. Results
3.1. Primary Analysis
3.2. Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mazzi, G.; Raineri, A.; Lacava, E.; De Roia, D.; Santarossa, L.; Orazi, B.M. Primary hyperfibrinogenolysis in a patient with anaphylactic shock. Haematologica 1994, 79, 283-285.
- Iqbal, A.; Morton, C.; Kong, K.-L. Fibrinolysis during anaphylaxis, and its spontaneous resolution, as demonstrated by thromboelastography. Br J Anaesth 2010, 105, 168-171. [CrossRef]
- Kienbacher, C.L.; Schoergenhofer, C.; Ruzicka, G.; Grafeneder, J.; Hufnagl, C.; Jilma, B.; Schwameis, M.; Herkner, H. Thromboelastography in acute immunologic reactions. A prospective pilot study. Res Pract Thromb Haemost 2024, 102425. [CrossRef]
- Longstaff, C. Measuring fibrinolysis: from research to routine diagnostic assays. J Thromb Haemost 2018, 16, 652-662. [CrossRef]
- Bernstein, J.A.; Cremonesi, P.; Hoffmann, T.K.; Hollingsworth, J. Angioedema in the emergency department: a practical guide to differential diagnosis and management. Int J Emerg Med 2017, 10, 15-15. [CrossRef]
- Muraro, A.; Worm, M.; Alviani, C.; Cardona, V.; DunnGalvin, A.; Garvey, L.H.; Riggioni, C.; de Silva, D.; Angier, E.; Arasi, S.; et al. EAACI guidelines: Anaphylaxis (2021 update). Allergy 2022, 77, 357-377. [CrossRef]
- National Institute for Health and Care Excellence. Anaphylaxis: assessment to confirm an anaphylactic episode and the decision to refer after emergency treatment for a suspected anaphylactic episode. Available online: https://www.nice.org.uk/guidance/cg134/documents/anaphylaxis-full-guideline2 (accessed on 6 September 2023).
- Reese, I.; Ballmer-Weber, B.; Beyer, K.; Fuchs, T.; Kleine-Tebbe, J.; Klimek, L.; Lepp, U.; Niggemann, B.; Saloga, J.; Schäfer, C.; et al. German guideline for the management of adverse reactions to ingested histamine. Allergo Journal International 2017, 26, 72-79. [CrossRef]
- Simons, F.E.R.; Ardusso, L.R.F.; Bilò, M.B.; El-Gamal, Y.M.; Ledford, D.K.; Ring, J.; Sanchez-Borges, M.; Senna, G.E.; Sheikh, A.; Thong, B.Y.; et al. World Allergy Organization guidelines for the assessment and management of anaphylaxis. World Allergy Organ J 2011, 4, 13-37. [CrossRef]
- Kemp, S.F. The post-anaphylaxis dilemma: how long is long enough to observe a patient after resolution of symptoms? Curr Allergy Asthma Rep 2008, 8, 45-48. [CrossRef]
- Lin, R.Y.; Schwartz, L.B.; Curry, A.; Pesola, G.R.; Knight, R.J.; Lee, H.-S.; Bakalchuk, L.; Tenenbaum, C.; Westfal, R.E. Histamine and tryptase levels in patients with acute allergic reactions: an emergency department–based study. J Allergy Clin Immunol 2000, 106, 65-71. [CrossRef]
- Schwartz, L.B.; Metcalfe, D.D.; Miller, J.S.; Earl, H.; Sullivan, T. Tryptase levels as an indicator of mast-cell activation in systemic anaphylaxis and mastocytosis. N Engl J Med 1987, 316, 1622-1626. [CrossRef]
- Werfen. ROTEM® delta. Available online: https://www.werfen.com/na/en/coagulation-testing-rotem-delta (accessed on 6 September 2023).
- Kuiper, G.J.; Kleinegris, M.C.; van Oerle, R.; Spronk, H.M.; Lancé, M.D.; Ten Cate, H.; Henskens, Y.M. Validation of a modified thromboelastometry approach to detect changes in fibrinolytic activity. Thromb J 2016, 14, 1. [CrossRef]
- Spiel, A.O.; Mayr, F.B.; Firbas, C.; Quehenberger, P.; Jilma, B. Validation of rotation thrombelastography in a model of systemic activation of fibrinolysis and coagulation in humans. J Thromb Haemost 2006, 4, 411-416. [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol 2008, 61, 344-349. [CrossRef]
- Gelbenegger, G.; Buchtele, N.; Schoergenhofer, C.; Grafeneder, J.; Schwameis, M.; Schellongowski, P.; Denk, W.; Jilma, B. Disseminated intravascular coagulation in anaphylaxis. Semin Thromb Hemost 2023, 569-579. [CrossRef]
| All patients, N=16 (100%) | Patients with persistent symptoms, n=6 (38%) | Asymptomatic patients, n=10 (63%) | |
| Female, n (%) | 10 (63) | 2 (33) | 8 (80) |
| Age, years, mean (SD) | 50 (14) | 50 (16) | 51 (11) |
| Suspected trigger, n (%) | |||
| Medication | 7 (44) | 4 (67) | 3 (30) |
| Insect venom | 4 (25) | 1 (17) | 4 (40) |
| Food | 1 (6) | 1 (17) | 0 (0) |
| Unknown | 3 (19) | 0 (0) | 3 (30) |
| Severe symptoms at presentation, n(%) | 8 (50) | 6 (100) | 2 (20) |
| Symptom onset to ED presentation, minutes, median (IQR) | 89 (31 – 279) | 25 (16 – 44) | 120 (80 – 375) |
| Time between initial and follow-up thromboelastography, median (IQR) | 136 (104 – 151) | 125 (105 – 177) | 136 (103 – 150) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




