Submitted:
22 July 2024
Posted:
22 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Ethical Statements
2.3. Fibroblasts Culture
2.4. Drug Screening
2.5. Quantitative Real-Time PCR (qPCR)
2.6. Immunoblotting
2.7. Prussian Blue staining
2.8. Sudan Black staining
2.9. TEM Analysis
2.10. PDH and KGDH Activities
2.11. Immunofluorescence Microscopy
2.12. Measurement of Membrane Potential
2.13. Bioenergetics
2.14. Mitochondrial Complexes Activity
2.15. SIRT3 Activity
2.16. NAD+/NADH Levels
2.17. Immunoprecipitation
2.18. Cell Transfection with Human LIPT1 Plasmid
2.19. Measurement of Cell Membrane and Mitochondrial Membrane Lipid Peroxidation
2.20. Direct Reprogramming
2.21. Statistical Analysis
4. Results.
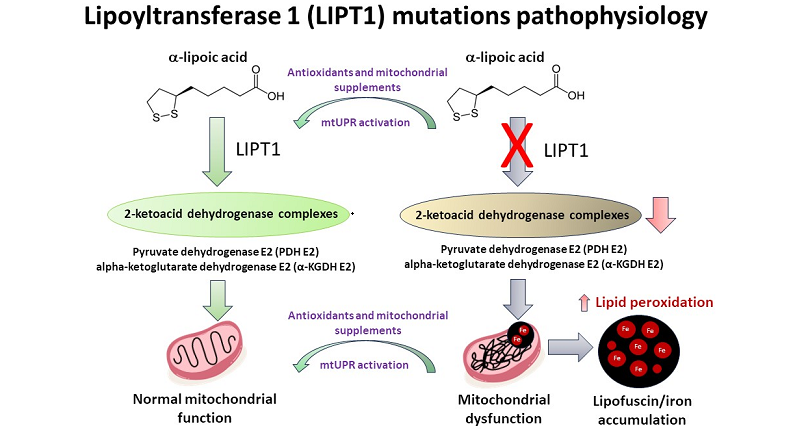
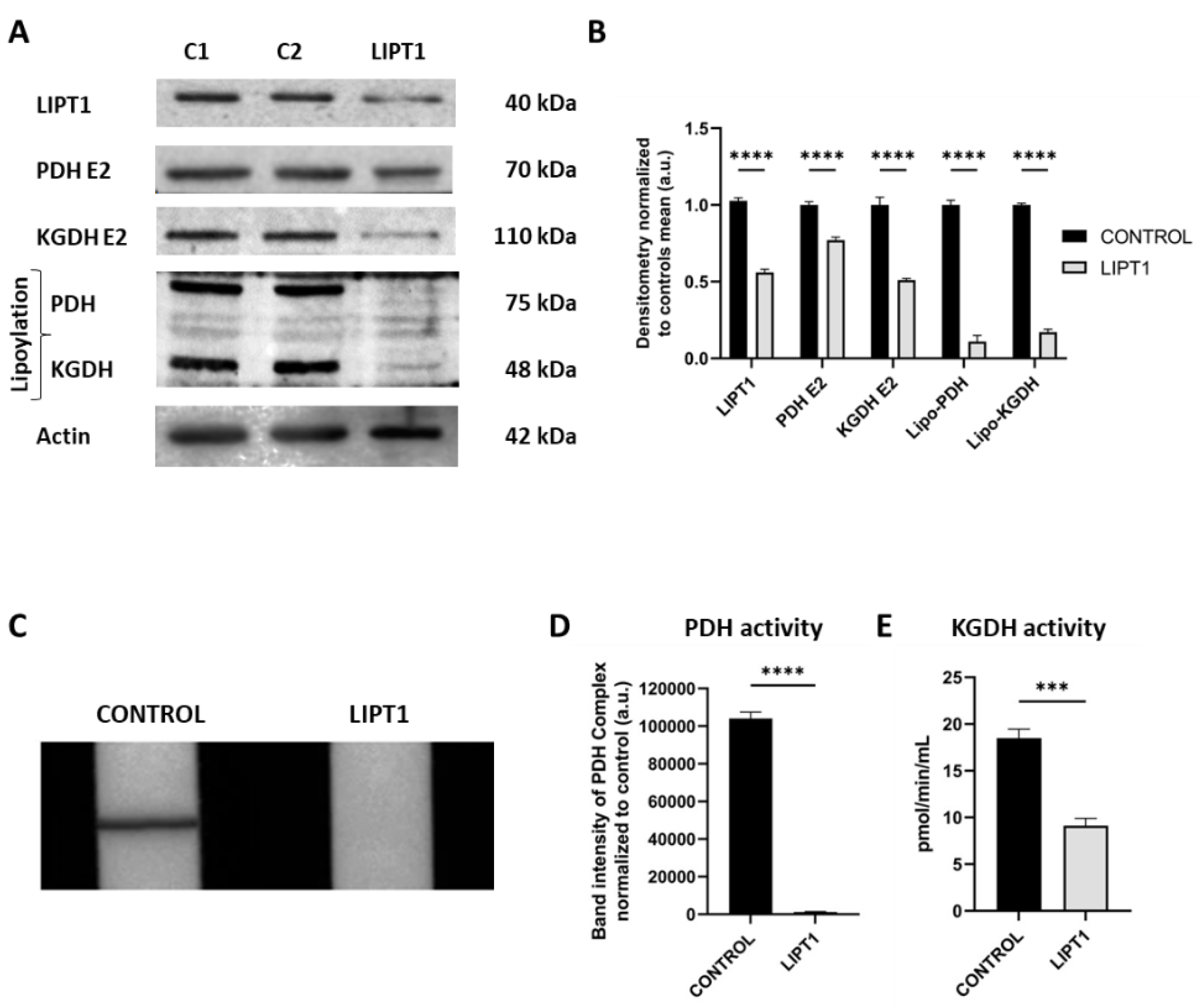
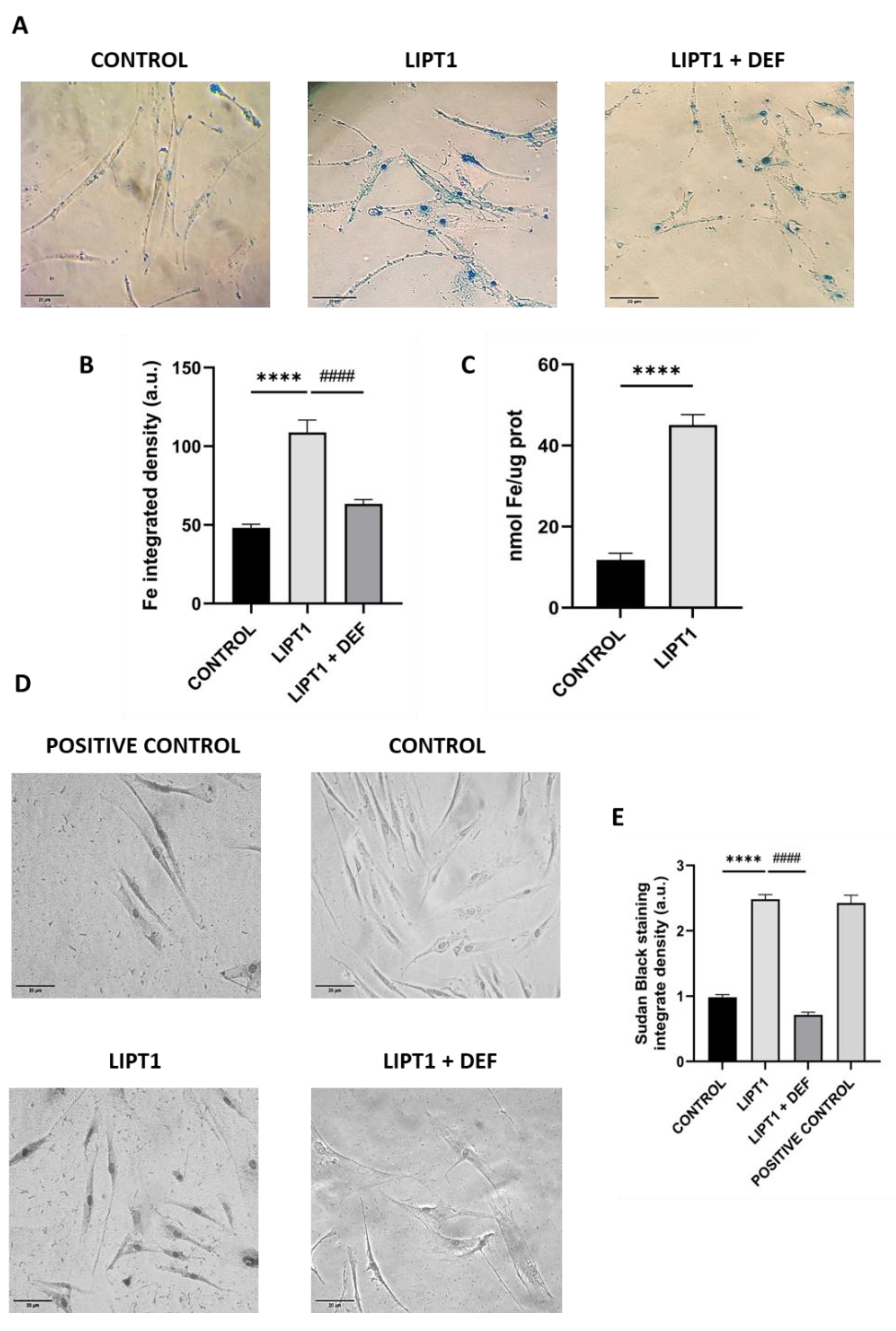
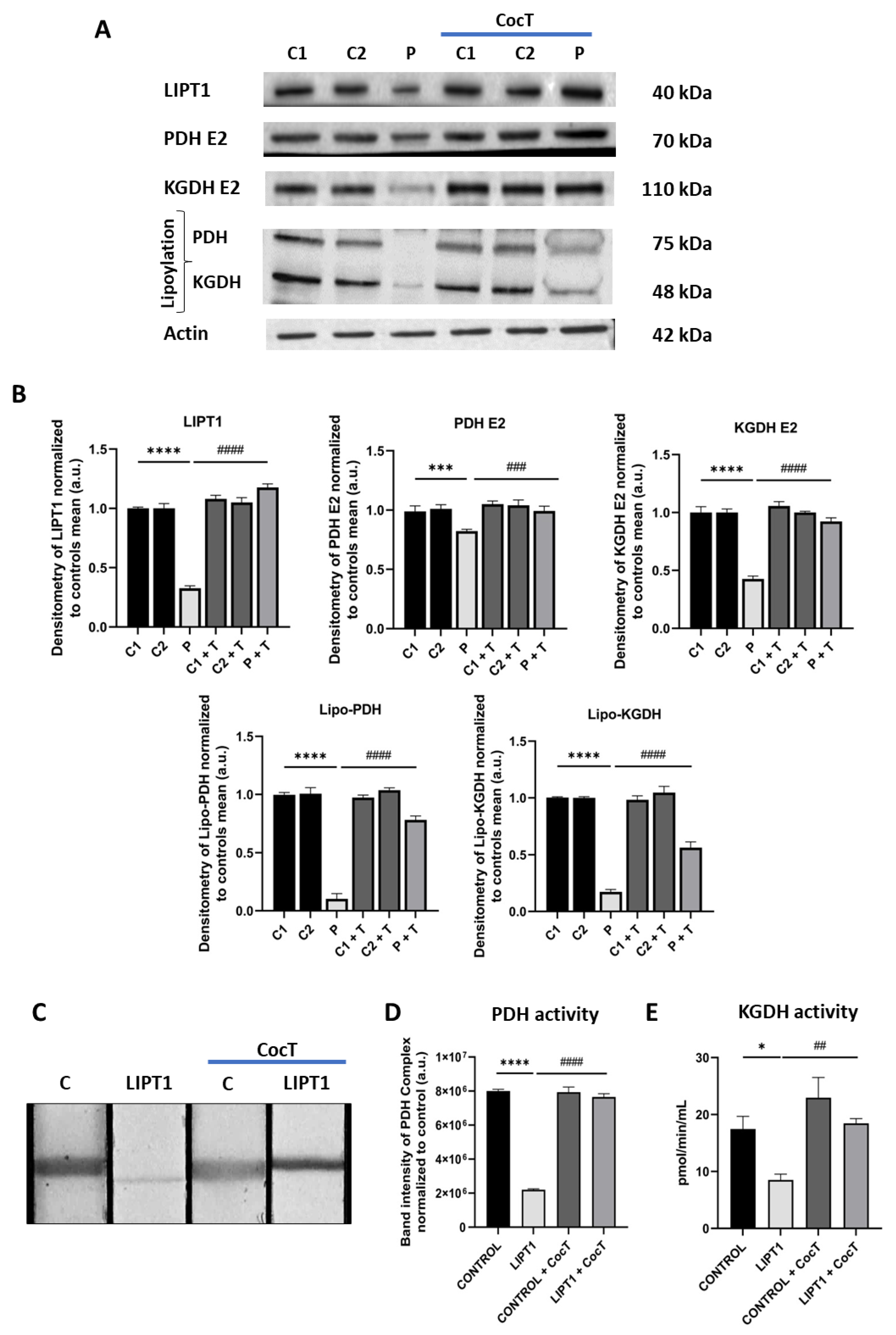
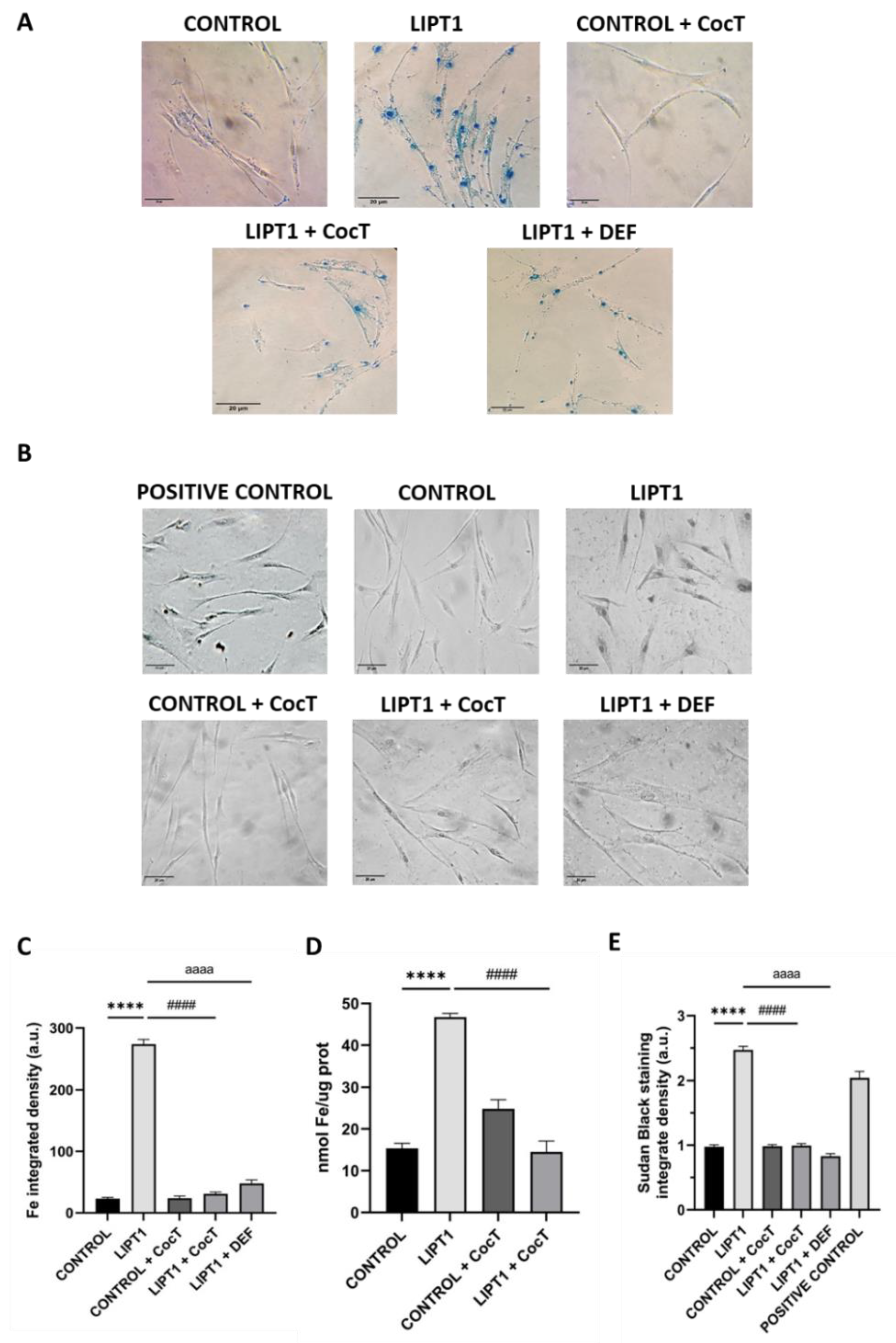
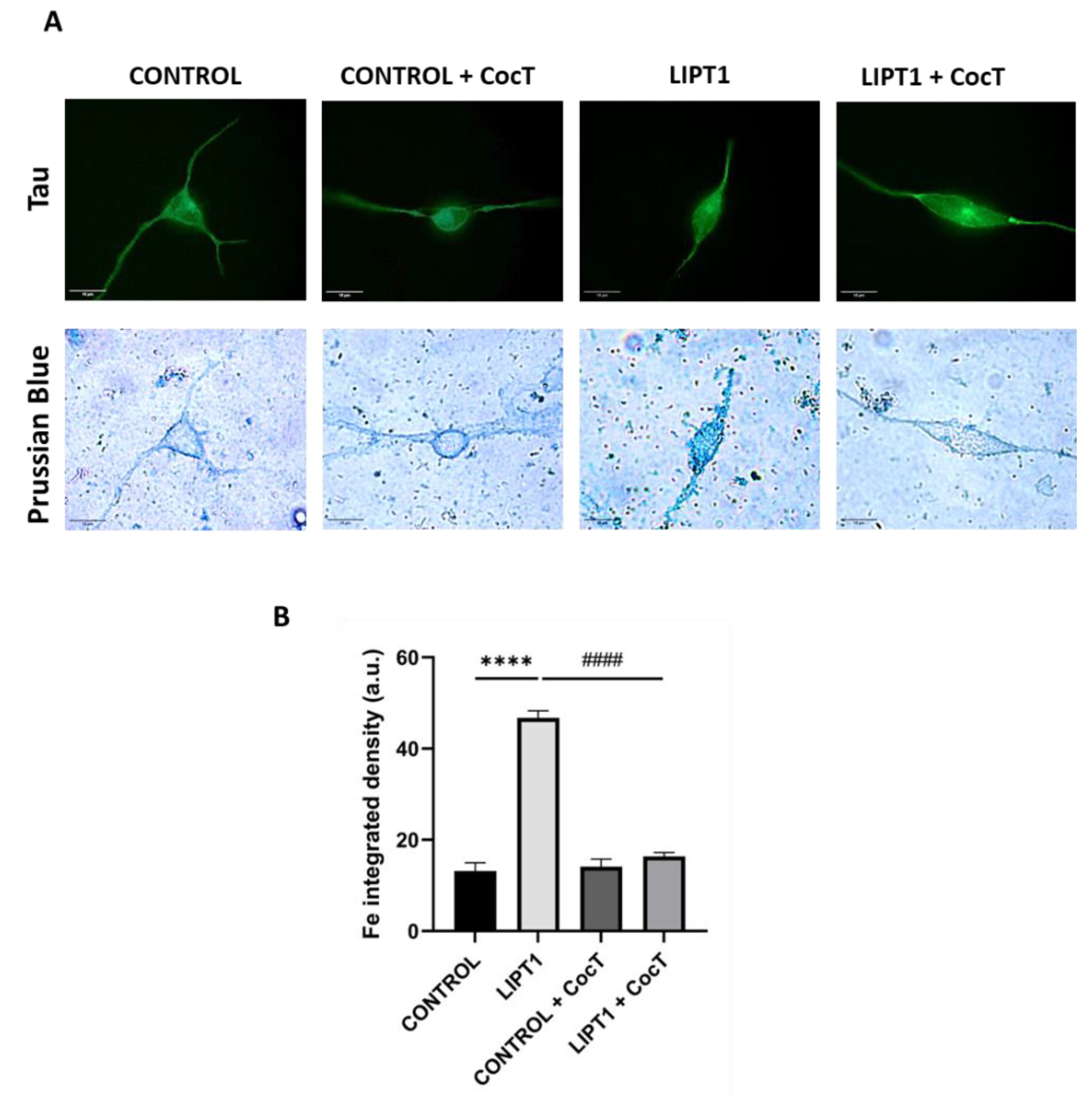
4.1. LIPT1 Mutation Causes Deficiency Of Lipoylation Of Pdh And Kgdh E2 Subunits, Impaired 2-Ketoacid Dehydrogenase Enzymes Activities And Iron Accumulation In Mutant Fibroblasts
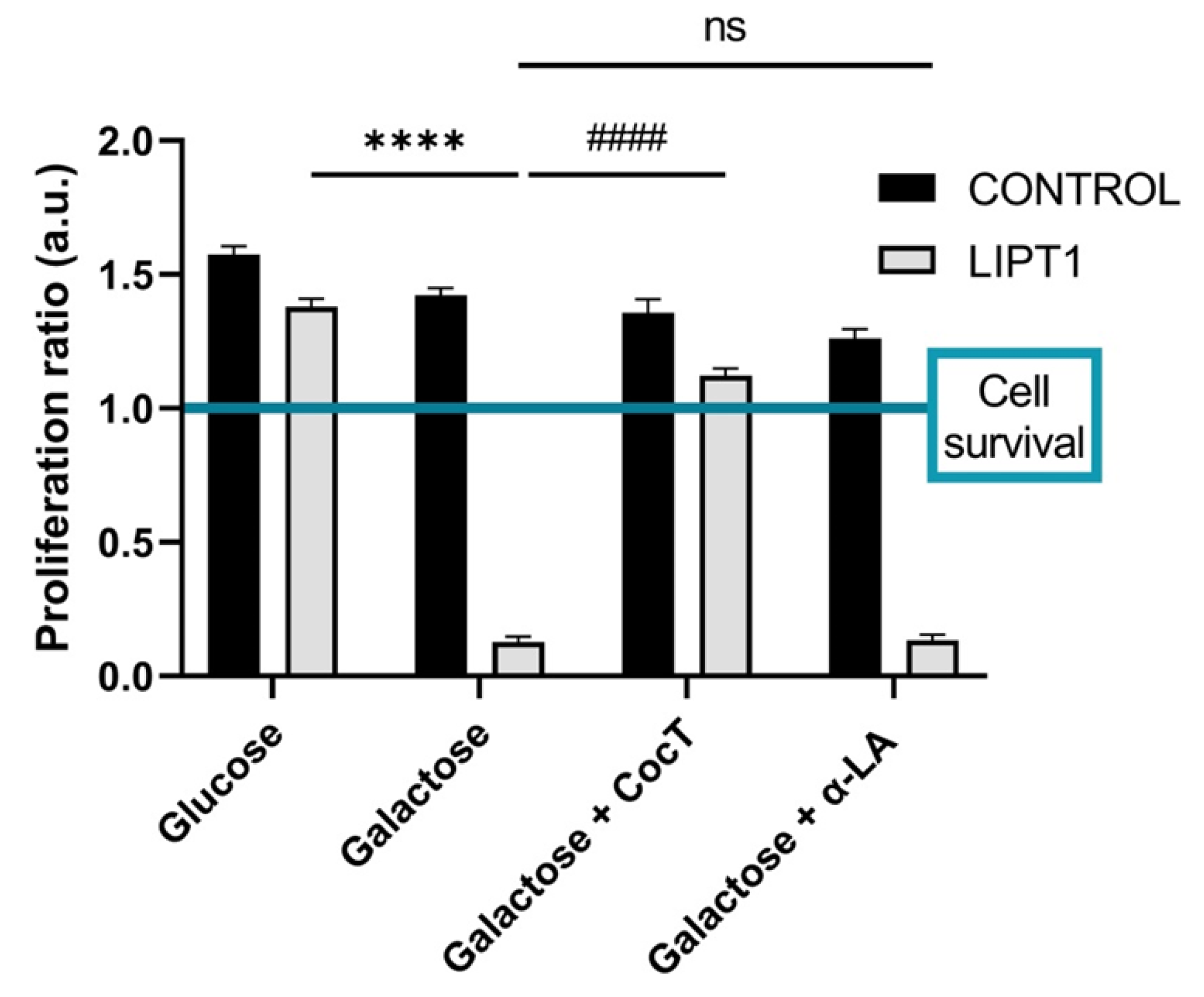
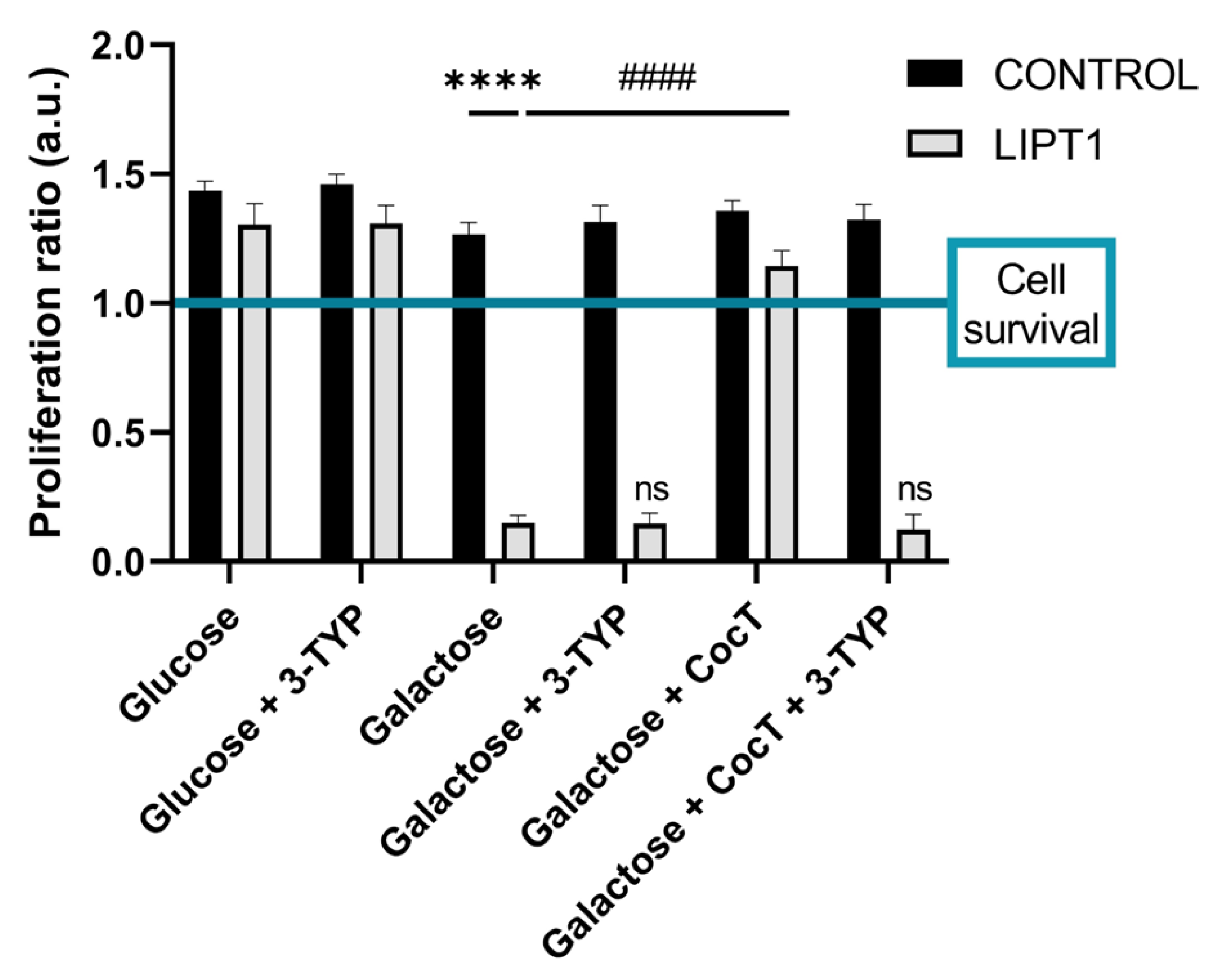
4.2. Pharmacological Screening in Galactose Medium
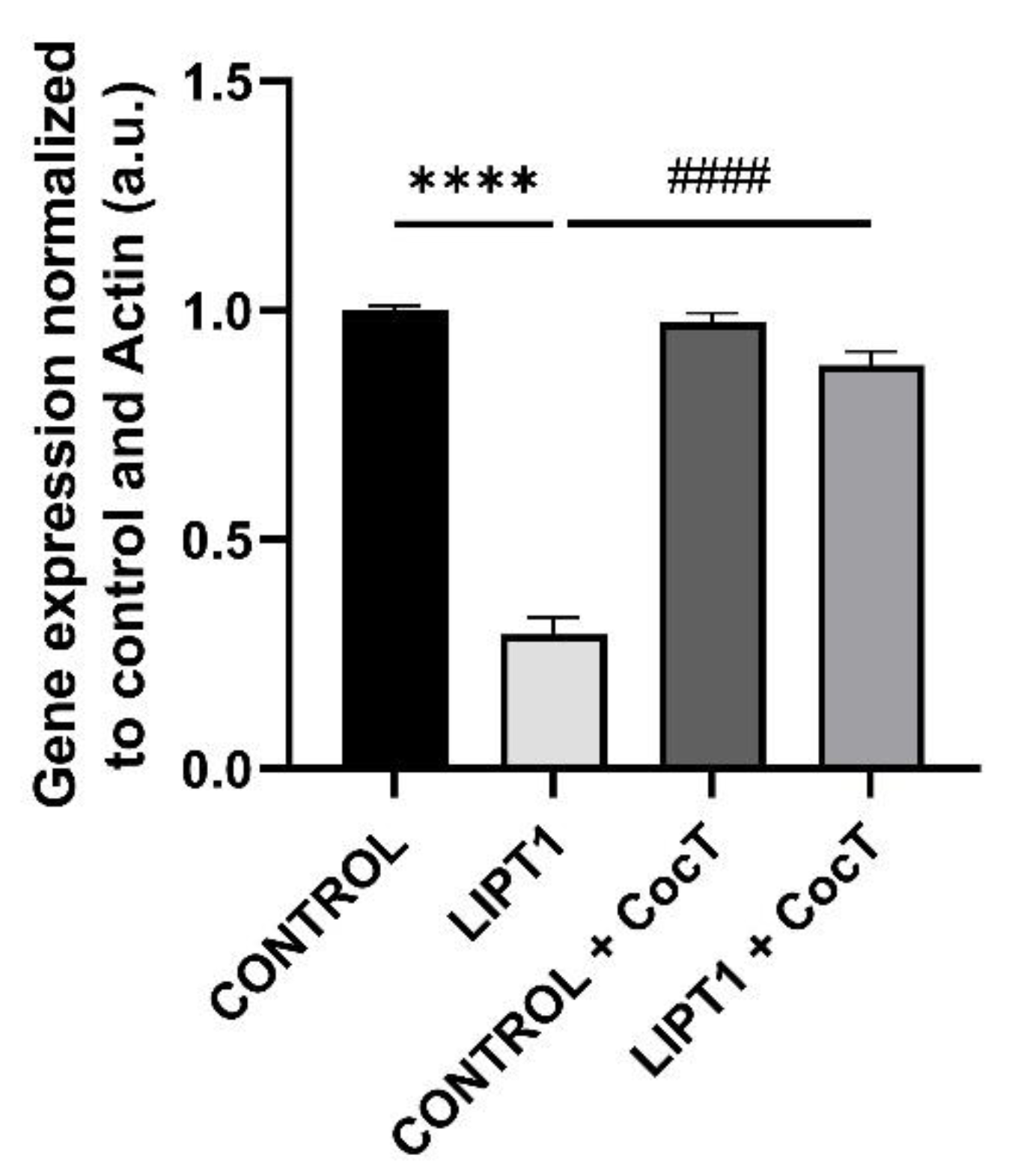
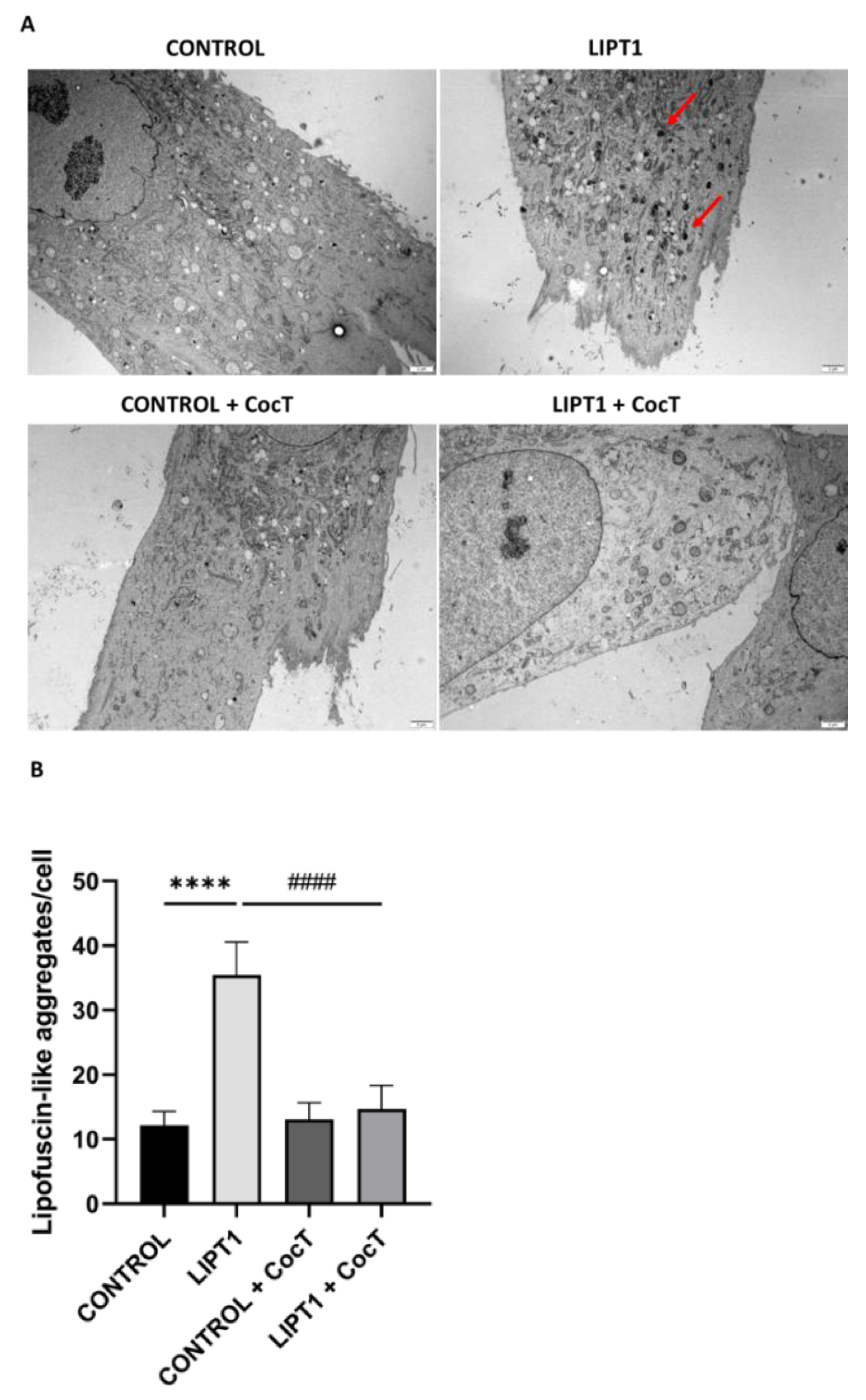
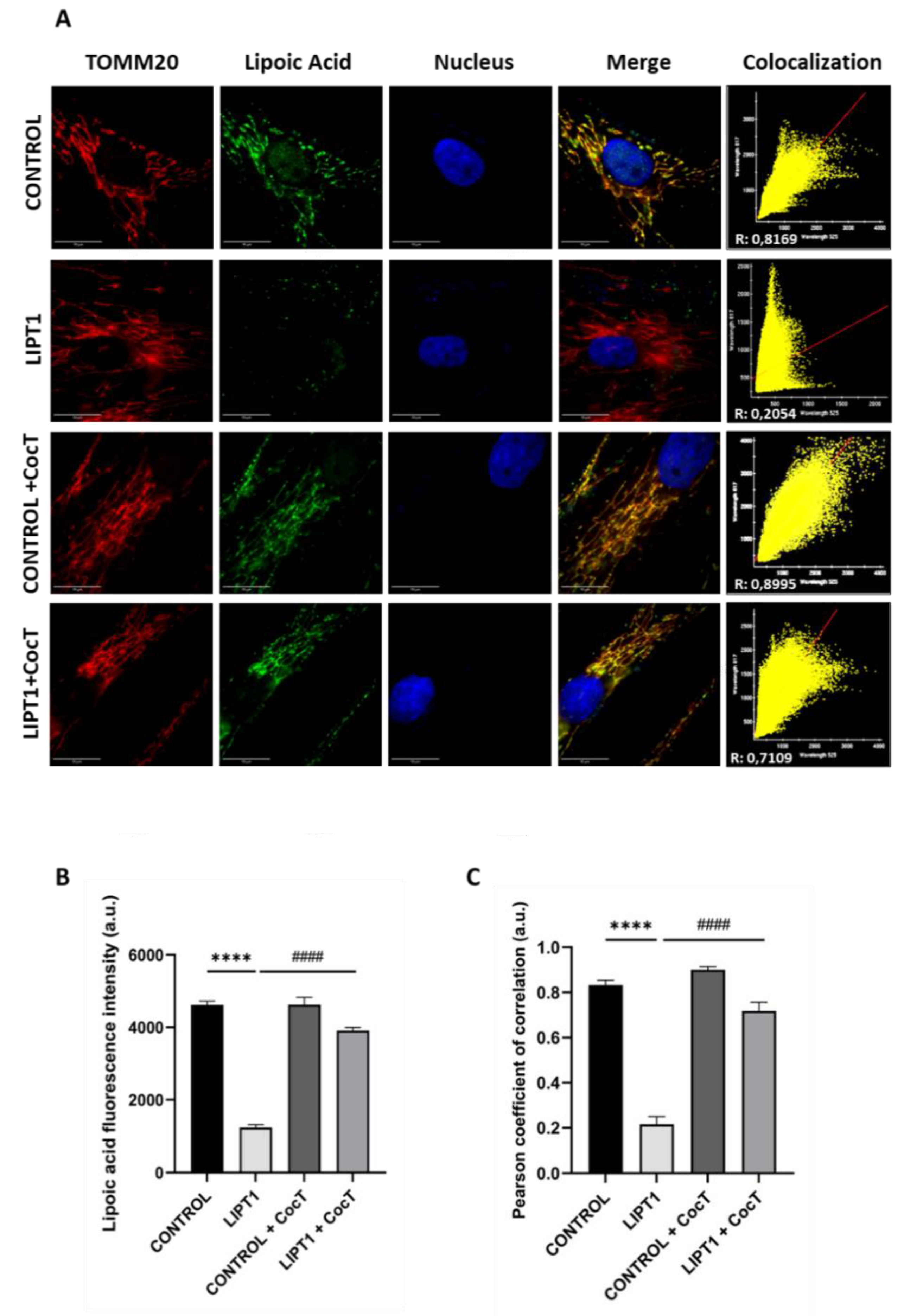
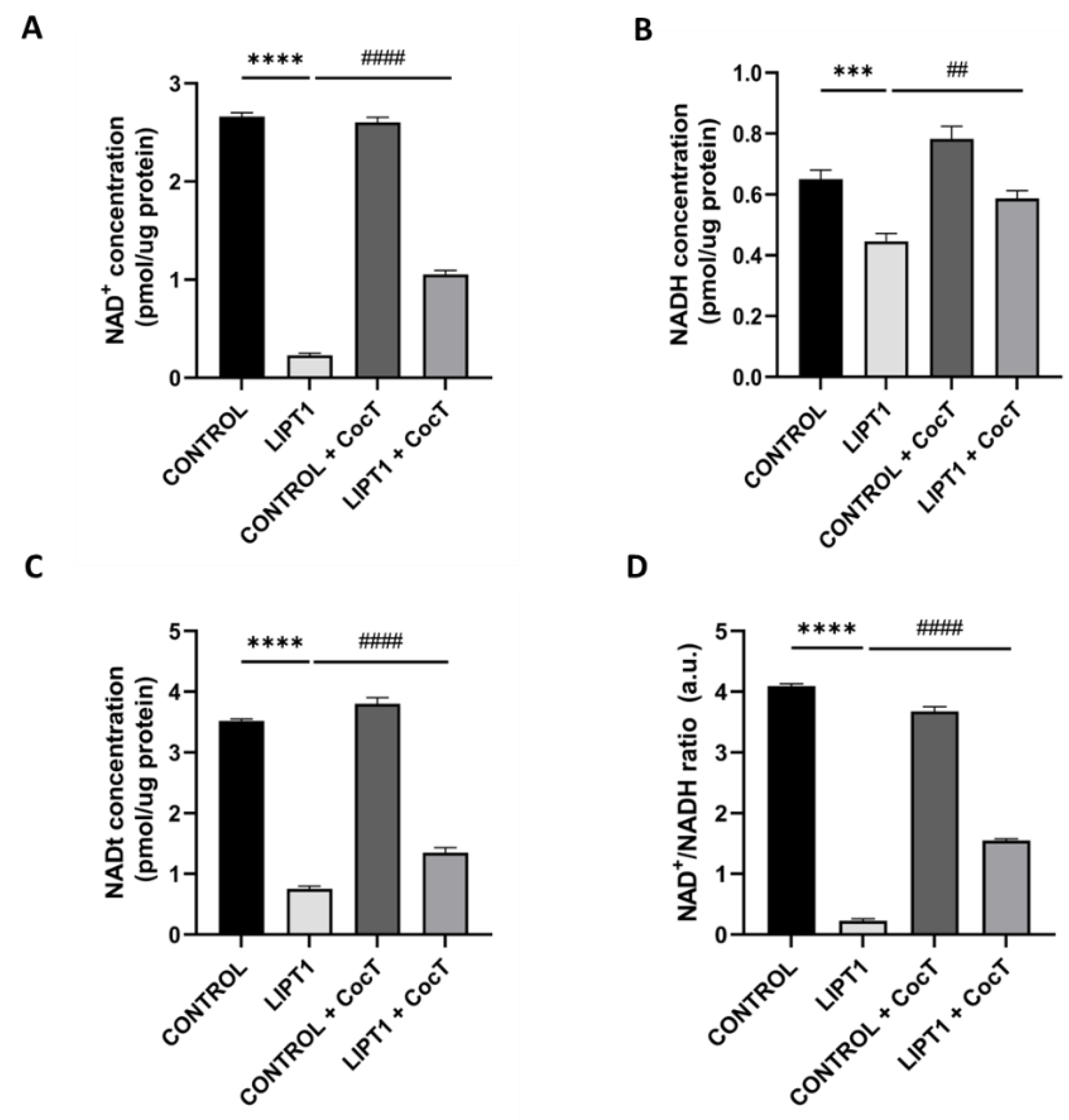
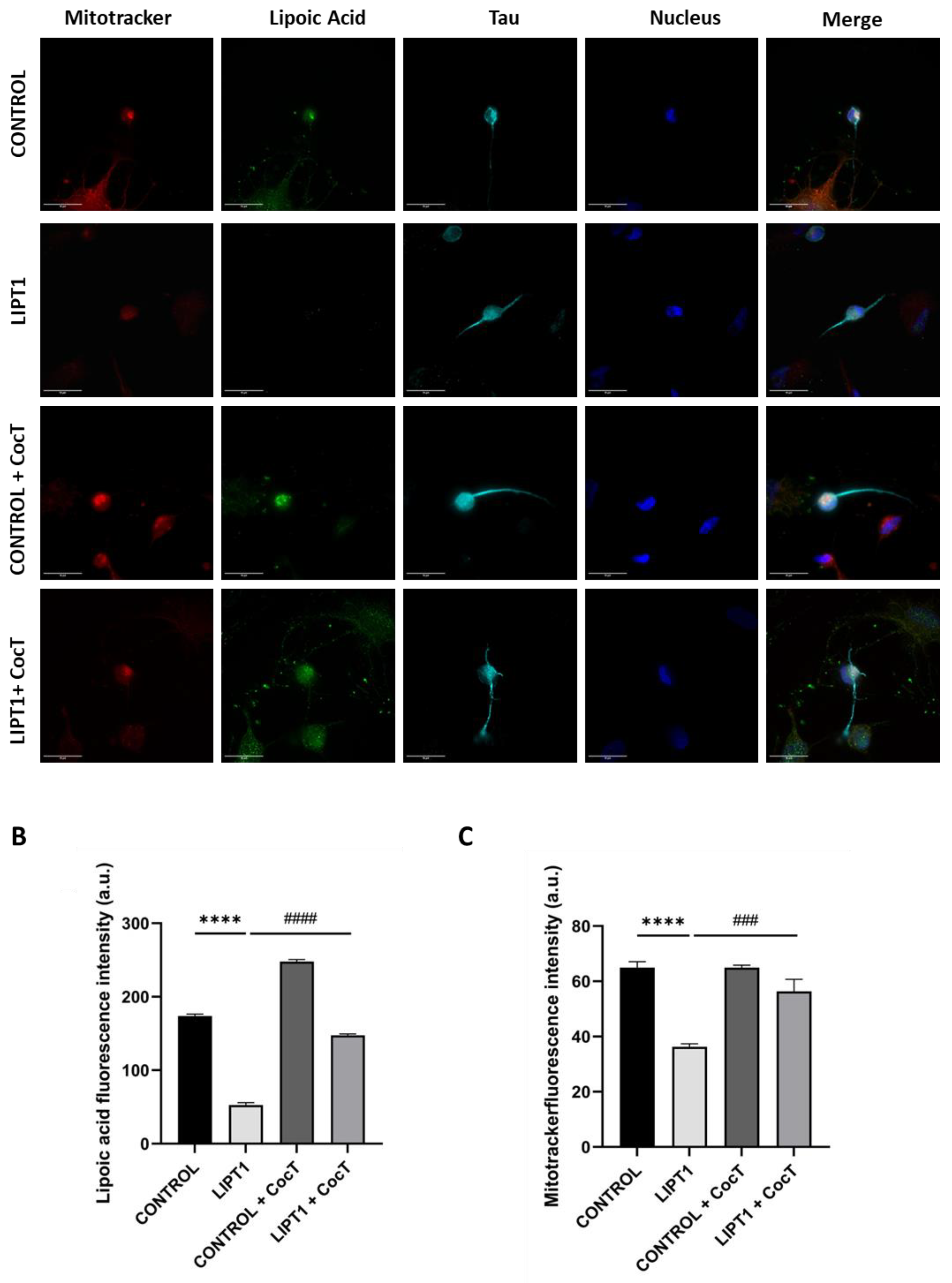
4.3. The supplementation with CocT increases LIPT1 Transcript Levels And Corrects Mutant Fibroblasts Pathophysiology.
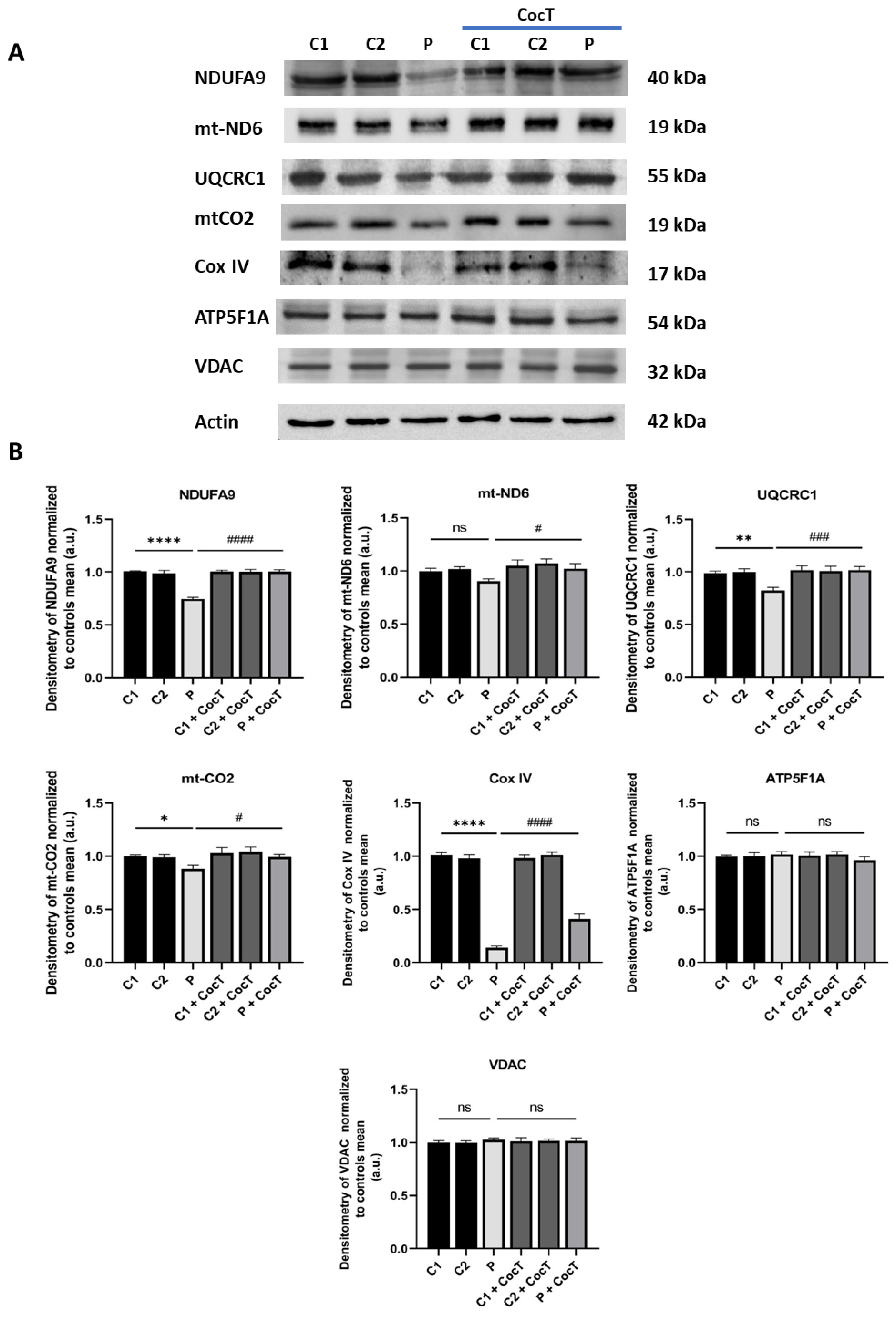
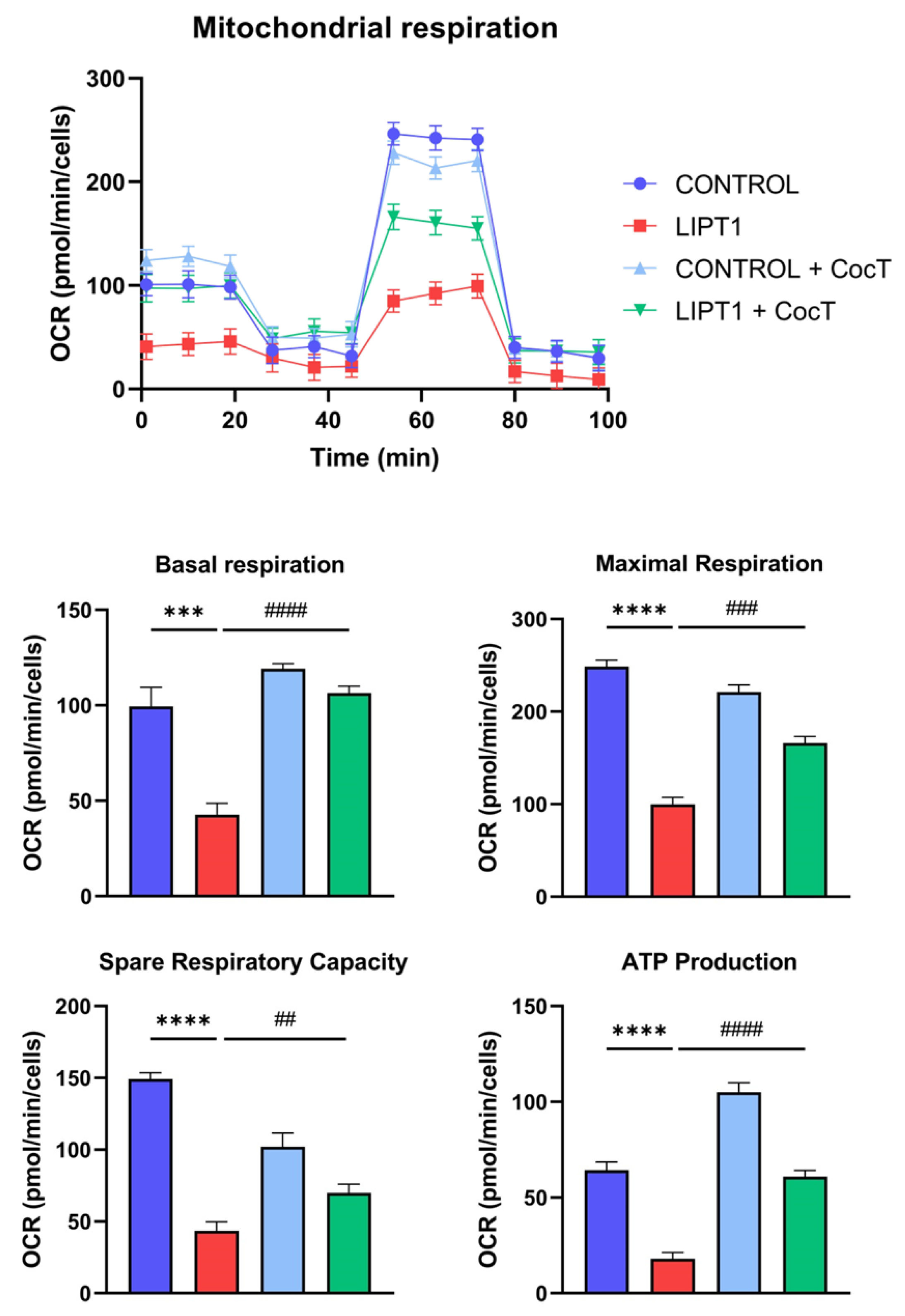
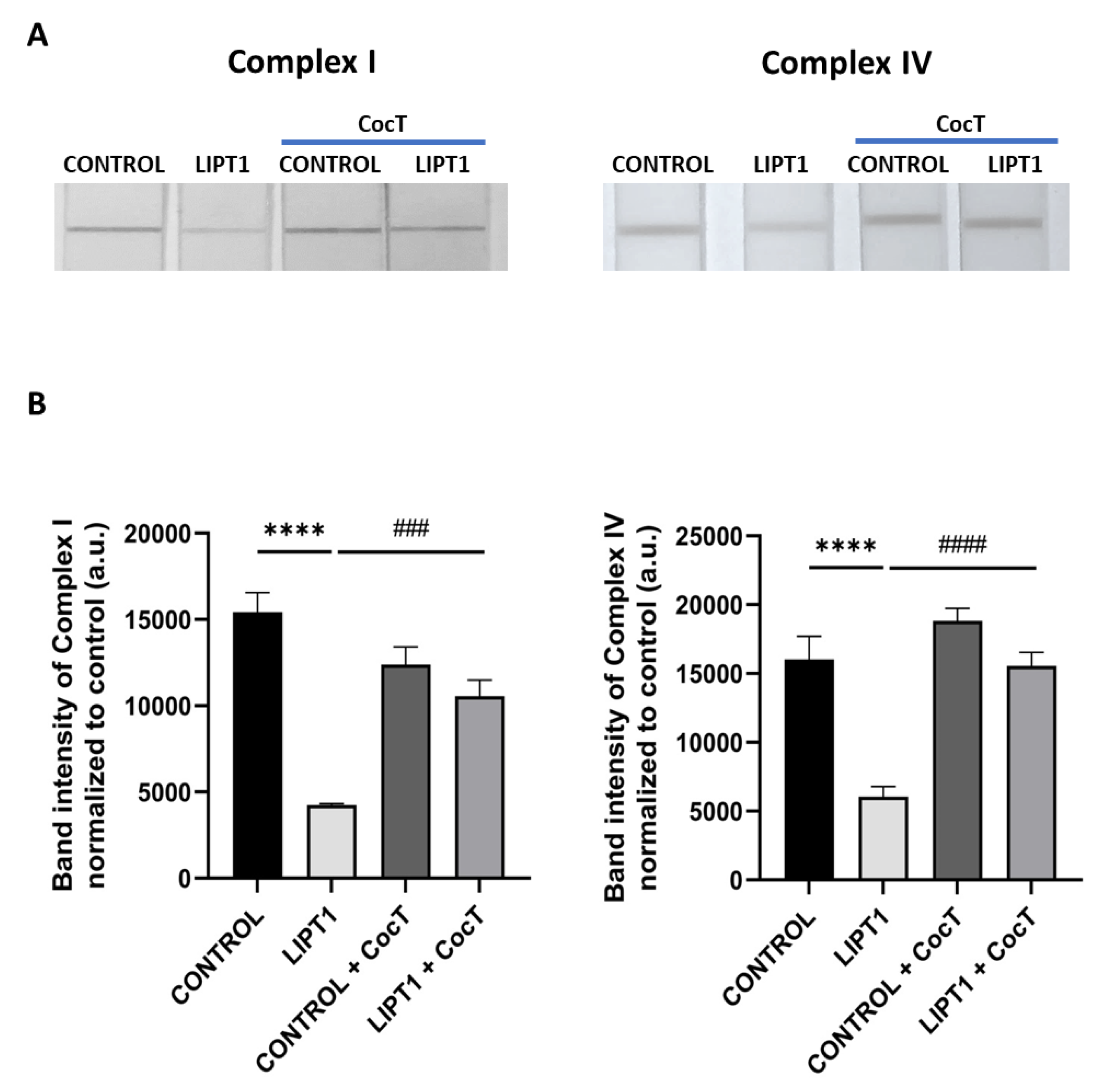
4.4. CocT Supplementation Increases Mitochondrial Proteins Expression Levels And Improves Mitochondrial Bioenergetics Of Mutant LIPT1 Cells
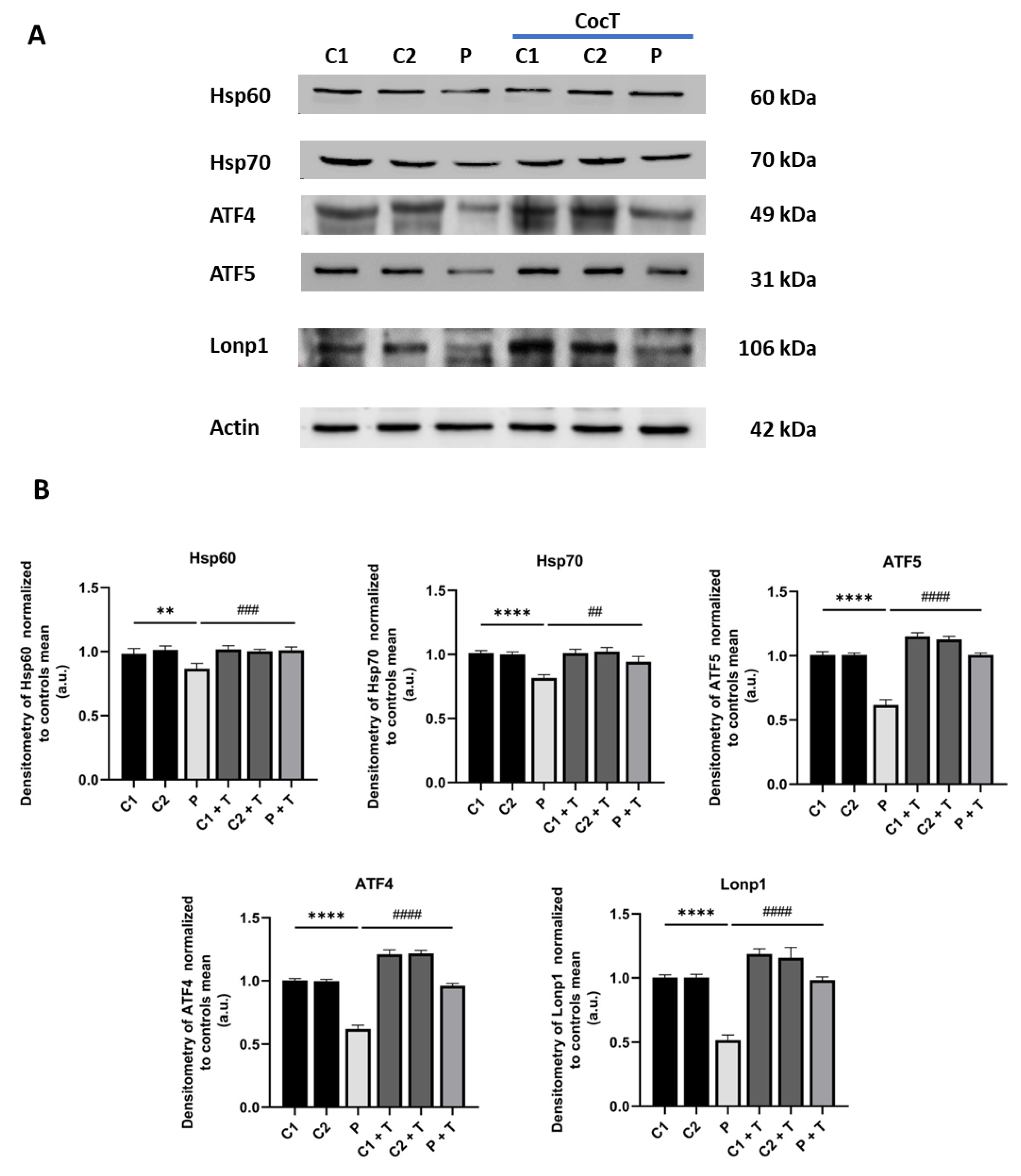
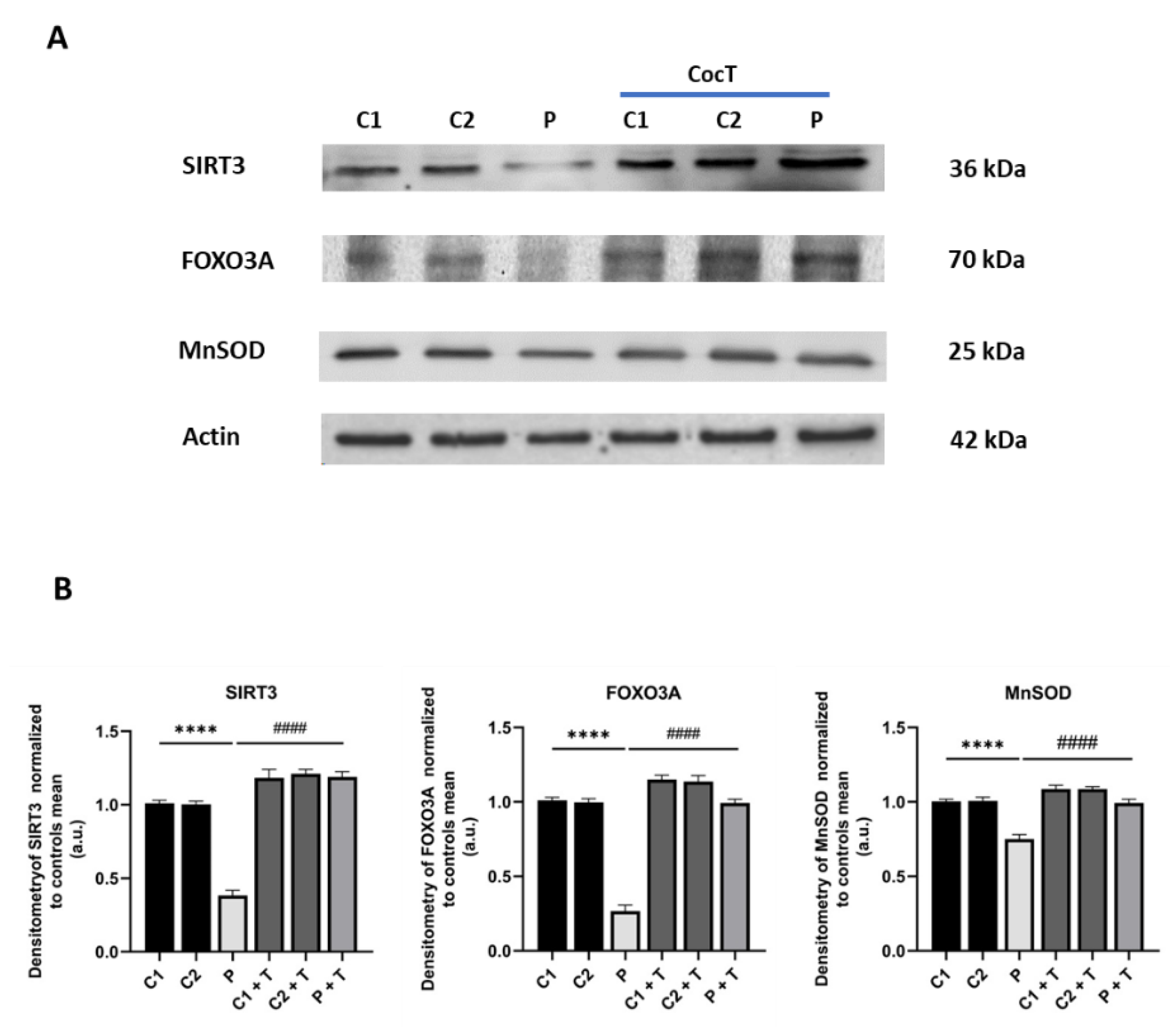
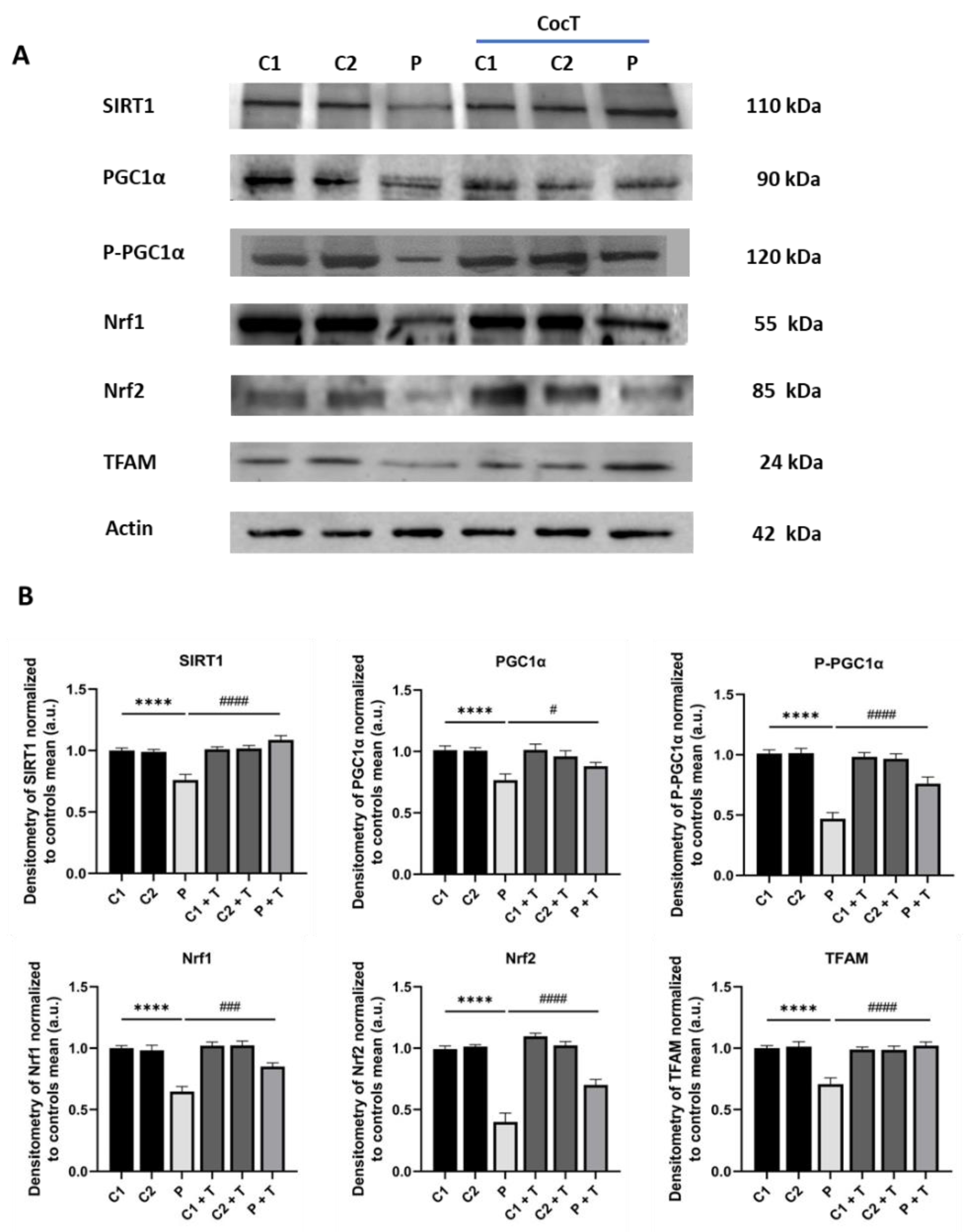
4.5. Supplementation with CocT Activates The Mitochondrial Unfolded Protein Response (mtUPR) and Mitochondrial Biogenesis
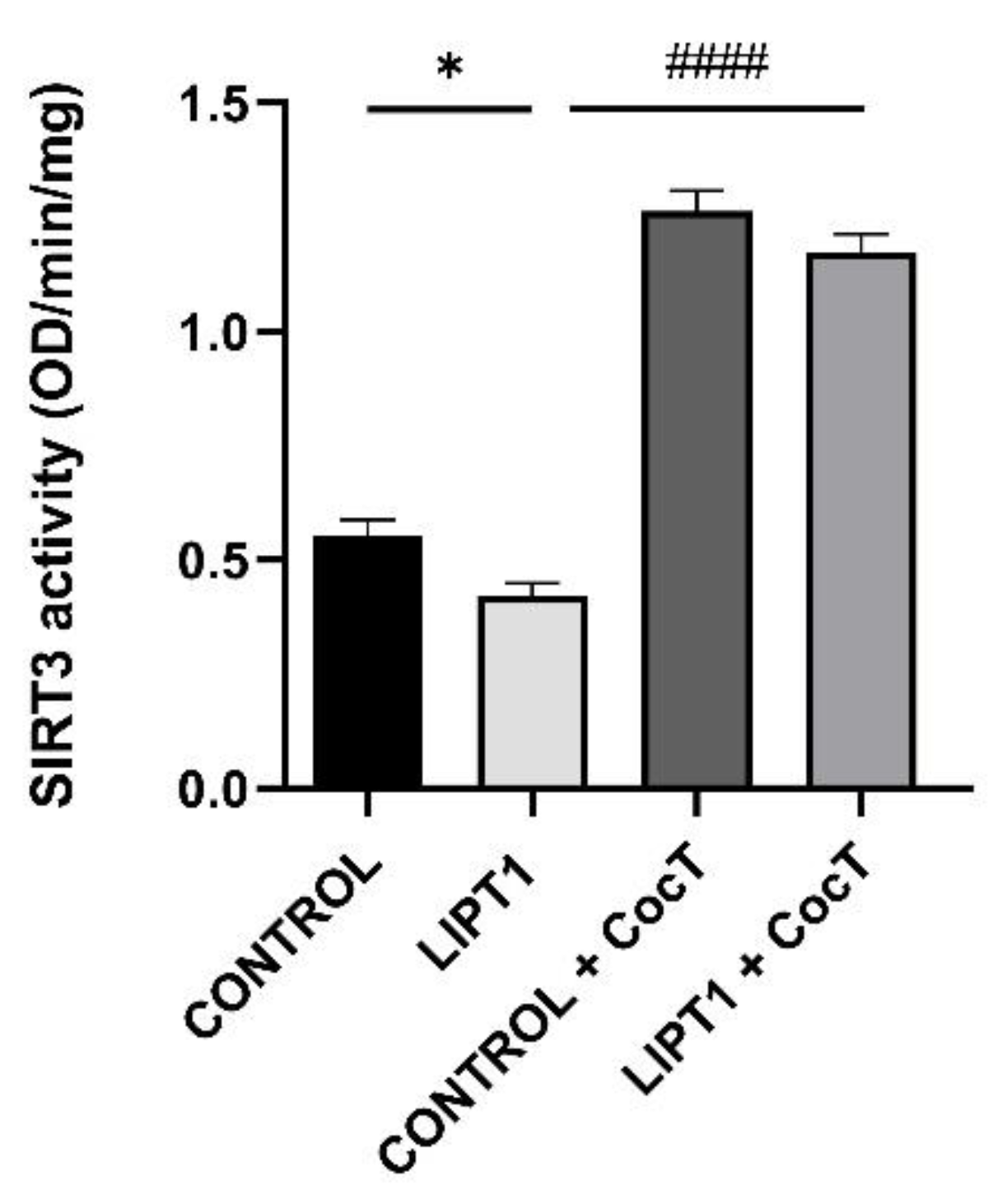
4.6. 3-TYP, a Specific SIRT3 Inhibitor, Blocks The Effect of CocT
4.7. Induced Neurons
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 3-TYP | 3-(1H-1,2,3-triazol-4-yl) pyridine |
| α-LA | alpha-lipoic acid |
| α-KGDH | alpha-ketoglutarate dehydrogenase |
| ACP | acyl carrier protein |
| ACSM2A | acyl-CoA synthethase medium chain family member 2A |
| AMP | adenosin monophosphate |
| AMPK | protein kinase AMP-activated |
| ASCL1 | Achaete-Scute Family BHLH Transcription Factor 1 |
| ATF4 | activating transcription factor 4 |
| ATF5 | activating transcription factor 5 |
| ATP5F1A | ATP synthase F1 subunit 1 alpha |
| BCKDH | branched chain ketoacid dehydrogenase |
| BRN2 | POU class 3 homeobox 2 |
| BSA | bovine serum albumin |
| CLPP | caseinolytic mitochondrial matrix peptidase proteolytic subunit |
| CoA | coenzyme A |
| CoPAN | CoA synthase protein-associated neurodegeneration |
| Cox IV | cytochrome C oxidase subunit IV |
| CPI-613 | 6,8-bis (benzylthiol)-octanoic acid |
| DAPI | 4′,6-diamidino-2-phenylindole |
| DLDH | lipoamide dehydrogenase |
| DMEM | Dulbecco's modified eagle medium |
| DMSO | Dimethyl Sulfoxide |
| EEG | electroencephalography |
| FAO | fatty acids oxidation |
| FASII | fatty acid synthesis type II |
| FBS | fetal bovine serum |
| FCCP | Carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone |
| FOX03A | forkhead box O3 |
| GCS | glycine cleavage system |
| GCSH | glycine cleavage system H protein |
| H2AX | H2A histone family member X |
| HEPES | 4-(2-hydroxyethyl)-1-piperazine ethanesulfonic acid |
| HRP | horseradish peroxidase |
| Hsp60 | heat shock protein 60 |
| Hsp70 | heat shock protein 70 |
| IC50 | half-maximal inhibitory concentration |
| ICP-MS | inductively coupled plasma mass spectrometry |
| IEM | inborn error of metabolism |
| iNs | induced neurons |
| LIAS | lipoic acid synthase |
| lip3 | lipoate protein ligase 3 |
| LIPT1 | lipoyltransferase 1 |
| LIPT2 | lipoyltransferase 2 |
| Lonp1 | lon peptidase 1 |
| LpIA | lipoate-protein ligase A |
| MePAN | mitochondrial enoyl CoA reductase protein-associated neurodegeneration |
| MnSOD | manganese superoxide dismutase |
| MRI | magnetic resonance imaging |
| mt-CO2 | mitochondrially encoded cytochrome C oxidase subunit II |
| mtDNA | mitochondrial DNA |
| mtETC | mitochondrial electron transport chain |
| mtUPR | mitochondrial Unfolded Protein Response |
| NAD+ | nicotinamide adenine dinucleotide |
| NAMPT | nicotinamide phosphoribosyltransferase |
| NBIA | neurodegeneration with brain iron accumulation |
| nDNA | nuclear DNA |
| NDUFA9 | NADH:ubiquinone oxidorreductase subunit A9 |
| Nrf1 | nuclear respiratory factor 1 |
| Nrf2 | nuclear respiratory factor 2 |
| OADH | 2-oxoadipate dehydrogenase |
| OCR | oxygen comsumption rate |
| OXPHOS | oxidative phosphorylation |
| PDH | pyruvate dehydrogenase |
| PFA | paraformaldehyde |
| PGC-1α | peroxisome proliferator-activated receptor-γ coactivator 1-α |
| P-PGC-1α | phosphorylated peroxisome proliferator-activated receptor-γ coactivator 1α |
| PGK | phosphoglycerate kinase |
| UQCRC1 | ubiquinol-cytochrome C reductase core protein 1 |
| PKAN | pantothenate kinase associated neurodegeneration |
| ROS | reactive oxygen species |
| SIRT1 | sirtuin 1 |
| SIRT3 | sirtuin 3 |
| TCA | tricarboxylic acids cycle |
| TEM | transmission electron microscope |
| TOMM20 | translocase of the outer mitochondrial membrane 20 |
| TFAM | mitochondrial transcription factor A |
| UQCRC1 | ubiquinol-cytochrome C reductase core protein 1 |
| VDAC | voltage-dependent anion channel |
| WES | whole exome sequencing |
References
- Patterson, E.L.; et al. Crystallization of a Derivative of Protogen-B. Journal of the American Chemical Society 2002, 73, 5919–5920. [Google Scholar] [CrossRef]
- Reed, L.J.; et al. Crystalline alpha-lipoic acid; a catalytic agent associated with pyruvate dehydrogenase. Science 1951, 114, 93–94. [Google Scholar] [CrossRef] [PubMed]
- Goraca, A.; et al. Lipoic acid - biological activity and therapeutic potential. Pharmacol Rep 2011, 63, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Mayr, J.A.; et al. Lipoic acid biosynthesis defects. J Inherit Metab Dis 2014, 37, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Reed, L.J. A trail of research from lipoic acid to alpha-keto acid dehydrogenase complexes. J Biol Chem 2001, 276, 38329–38336. [Google Scholar] [CrossRef] [PubMed]
- Solmonson, A.; DeBerardinis, R.J. Lipoic acid metabolism and mitochondrial redox regulation. J Biol Chem 2018, 293, 7522–7530. [Google Scholar] [CrossRef] [PubMed]
- Rochette, L.; et al. Alpha-lipoic acid: Molecular mechanisms and therapeutic potential in diabetes. Can J Physiol Pharmacol 2015, 93, 1021–1027. [Google Scholar] [CrossRef] [PubMed]
- Cronan, J.E. Assembly of Lipoic Acid on Its Cognate Enzymes: An Extraordinary and Essential Biosynthetic Pathway. Microbiol Mol Biol Rev 2016, 80, 429–450. [Google Scholar] [CrossRef] [PubMed]
- Schonauer, M.S.; et al. Lipoic acid synthesis and attachment in yeast mitochondria. J Biol Chem 2009, 284, 23234–23242. [Google Scholar] [CrossRef]
- Morris, T.W.; Reed, K.E.; Cronan, J.E., Jr. Identification of the gene encoding lipoate-protein ligase A of Escherichia coli. Molecular cloning and characterization of the lplA gene and gene product. J Biol Chem 1994, 269, 16091–16100. [Google Scholar]
- Fujiwara, K.; et al. Purification, characterization, and cDNA cloning of lipoate-activating enzyme from bovine liver. J Biol Chem 2001, 276, 28819–28823. [Google Scholar] [CrossRef] [PubMed]
- DeBerardinis, R.J.; Thompson, C.B. Cellular metabolism and disease: What do metabolic outliers teach us? Cell 2012, 148, 1132–1144. [Google Scholar] [CrossRef]
- Stowe, R.C.; Sun, Q.; Elsea, S.H.; Scaglia, F. LIPT1 deficiency presenting as early infantile epileptic encephalopathy, Leigh disease, and secondary pyruvate dehydrogenase complex deficiency. Am J Med Genet A. 2018, 176. [Google Scholar] [CrossRef]
- Ni, M.; et al. Functional Assessment of Lipoyltransferase-1 Deficiency in Cells, Mice, and Humans. Cell Rep 2019, 27, 1376–1386 e6. [Google Scholar] [CrossRef]
- Tort, F.; Ferrer-Cortes, X.; Ribes, A. Differential diagnosis of lipoic acid synthesis defects. J Inherit Metab Dis 2016, 39. [Google Scholar] [CrossRef] [PubMed]
- Soreze, Y.; et al. Mutations in human lipoyltransferase gene LIPT1 cause a Leigh disease with secondary deficiency for pyruvate and alpha-ketoglutarate dehydrogenase. Orphanet J Rare Dis 2013, 8, 192. [Google Scholar] [CrossRef]
- Quinn, J. Lipoylation of acyltransferase components of 2-oxo acid dehydrogenase complexes. Methods Enzymol 1997, 279, 193–202. [Google Scholar]
- Kamalian, L.; et al. Acute Metabolic Switch Assay Using Glucose/Galactose Medium in HepaRG Cells to Detect Mitochondrial Toxicity. Curr Protoc Toxicol 2019, 80, e76. [Google Scholar] [CrossRef]
- Coelho, A.I.; Berry, G.T.; Rubio-Gozalbo, M.E. Galactose metabolism and health. Curr Opin Clin Nutr Metab Care 2015, 18, 422–427. [Google Scholar] [CrossRef]
- Ghio, A.J.; Roggli, V.L. Perls' Prussian Blue Stains of Lung Tissue, Bronchoalveolar Lavage, and Sputum. J Environ Pathol Toxicol Oncol 2021, 40, 1–15. [Google Scholar]
- Tarohda, T.; Ishida, Y.; Kawai, K.; Yamamoto, M.; Amano, R. Regional distributions of manganese, iron, copper, and zinc in the brains of 6-hydroxydopamine-induced parkinsonian rats. Anal Bioanal Chem 2005, 383, 224–234. [Google Scholar] [CrossRef]
- Georgakopoulou, E.A.; Tsimaratou, K.; Evangelou, K.; Fernandez Marcos, P.J.; Zoumpourlis, V.; Trougakos, I.P.; et al. Specific lipofuscin staining as a novel biomarker to detect replicative and stress-induced senescence. A method applicable in cryo-preserved and archival tissues. Aging (Albany NY) 2013, 5. [Google Scholar] [CrossRef]
- Evangelou, K.; Gorgoulis, V.G. Sudan Black B, The Specific Histochemical Stain for Lipofuscin: A Novel Method to Detect Senescent Cells. Methods Mol Biol 2017. [Google Scholar] [CrossRef]
- Alcocer-Gomez, E.; et al. Metformin and caloric restriction induce an AMPK-dependent restoration of mitochondrial dysfunction in fibroblasts from Fibromyalgia patients. Biochim Biophys Acta 2015, 1852, 1257–1267. [Google Scholar] [CrossRef]
- Pap, E.H.; et al. Ratio-fluorescence microscopy of lipid oxidation in living cells using C11-BODIPY(581/591). FEBS Lett 1999, 453, 278–282. [Google Scholar] [CrossRef]
- Nakamura, S.; et al. Ionomycin-induced calcium influx induces neurite degeneration in mouse neuroblastoma cells: Analysis of a time-lapse live cell imaging system. Free Radic Res 2016, 50, 1214–1225. [Google Scholar] [CrossRef]
- Legault, E.M.; Drouin-Ouellet, J. Generation of Induced Dopaminergic Neurons from Human Fetal Fibroblasts. Methods Mol Biol 2021, 2352, 97–115. [Google Scholar]
- Bruzelius, A.; et al. Reprogramming Human Adult Fibroblasts into GABAergic Interneurons. Cells 2021, 10. [Google Scholar] [CrossRef]
- Zufferey, R.; et al. Multiply attenuated lentiviral vector achieves efficient gene delivery in vivo. Nat Biotechnol 1997, 15, 871–875. [Google Scholar] [CrossRef]
- Le Gall, J.Y.; et al. [Human iron metabolism]. Bull Acad Natl Med 2005, 189, 1635–1647. [Google Scholar]
- Miller, J.R.; Busby, R.W.; Jordan, S.W.; Cheek, J.; Henshaw, T.F.; Ashley, G.W.; et al. Escherichia coli LipA is a lipoy synthase: In vitro biosynthesis of lipoylated pyruvate dehydrogenase complex from octanoyl-acyl carrier protein. Biochemistry 2000, 39. [Google Scholar] [CrossRef]
- Lill, R.; Freibert, S.A. Mechanisms of Mitochondrial Iron-Sulfur Protein Biogenesis. Annu Rev Biochem 2020, 89, 471–499. [Google Scholar] [CrossRef]
- Said, H.M. Biotin: Biochemical, physiological and clinical aspects. Subcell Biochem 2012, 56, 1–19. [Google Scholar]
- Pekala, J.; et al. L-carnitine--metabolic functions and meaning in humans life. Curr Drug Metab 2011, 12, 667–678. [Google Scholar] [CrossRef]
- Canto, C.; et al. The NAD(+) precursor nicotinamide riboside enhances oxidative metabolism and protects against high-fat diet-induced obesity. Cell Metab 2012, 15, 838–847. [Google Scholar] [CrossRef]
- Miyazawa, T.; et al. Vitamin E: Regulatory Redox Interactions. IUBMB Life 2019, 71, 430–441. [Google Scholar] [CrossRef]
- Depeint, F.; et al. Mitochondrial function and toxicity: Role of the B vitamin family on mitochondrial energy metabolism. Chem Biol Interact 2006, 163, 94–112. [Google Scholar] [CrossRef]
- Salehi, B.; et al. Insights on the Use of alpha-Lipoic Acid for Therapeutic Purposes. Biomolecules 2019, 9. [Google Scholar] [CrossRef]
- Talaveron-Rey, M.; et al. Alpha-lipoic acid supplementation corrects pathological alterations in cellular models of pantothenate kinase-associated neurodegeneration with residual PANK2 expression levels. Orphanet J Rare Dis 2023, 18, 80. [Google Scholar] [CrossRef]
- Martinus, R.D.; Garth, G.P.; Webster, T.L.; Cartwright, P.; Naylor, D.J.; Hoj, P.B.; Hoogenraad, N.J. Selective induction of mitochondrial chaperones in response to loss of the mitochondrial genome. Eur J Biochem 1996, 240. [Google Scholar] [CrossRef]
- Munch, C. The different axes of the mammalian mitochondrial unfolded protein response. BMC Bio. 2018, 16. [Google Scholar] [CrossRef]
- Cilleros-Holgado, P.; Gómez-Fernández, D.; Piñero-Pérez, R.; Reche-López, D.; Álvarez-Córdoba, M.; Munuera-Cabeza, M.; et al. mtUPR Modulation as a Therapeutic Target for Primary and Secondary Mitochondrial Diseases. International Journal of Molecular Sciences 2023. [Google Scholar] [CrossRef]
- Canto, C.; Menzies, K.J.; Auwerx, J. NAD(+) Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell Metab 2015, 22, 31–53. [Google Scholar] [CrossRef]
- Ismail, F.Y.; Mitoma, H.; Fatemi, A. Metabolic ataxias. Handb Clin Neurol 2018, 155, 117–127. [Google Scholar]
- Molnar, M.J.; Kovacs, G.G. Mitochondrial diseases. Handb Clin Neurol 2017, 145, 147–155. [Google Scholar]
- Feng, D.; Witkowski, A.; Smith, S. Down-regulation of Mitochondrial Acyl Carrier Protein in Mammalian Cells Compromises Protein Lipoylation and Respiratory Complex I and Results in Cell Death. J Biol Chem 2009, 284. [Google Scholar] [CrossRef]
- Cronan, J.E. Biotin and Lipoic Acid: Synthesis, Attachment, and Regulation. EcoSal Plus 2014, 6. [Google Scholar] [CrossRef]
- Lavatelli, A.; de Mendoza, D.; Mansilla, M.C. Defining Caenorhabditis elegans as a model system to investigate lipoic acid metabolism. J Biol Chem 2020, 295. [Google Scholar] [CrossRef]
- Head, R.A.; et al. Clinical and genetic spectrum of pyruvate dehydrogenase deficiency: Dihydrolipoamide acetyltransferase (E2) deficiency. Ann Neurol 2005, 58, 234–241. [Google Scholar] [CrossRef]
- Leoni, V.; et al. Metabolic consequences of mitochondrial coenzyme A deficiency in patients with PANK2 mutations. Mol Genet Metab 2012, 105, 463–471. [Google Scholar] [CrossRef]
- McWilliam, C.A.; et al. Pyruvate dehydrogenase E2 deficiency: A potentially treatable cause of episodic dystonia. Eur J Paediatr Neurol 2010, 14, 349–353. [Google Scholar] [CrossRef]
- Alvarez-Cordoba, M.; et al. Pantothenate Rescues Iron Accumulation in Pantothenate Kinase-Associated Neurodegeneration Depending on the Type of Mutation. Mol Neurobiol 2019, 56, 3638–3656. [Google Scholar] [CrossRef]
- Alvarez-Cordoba, M.; et al. Down regulation of the expression of mitochondrial phosphopantetheinyl-proteins in pantothenate kinase-associated neurodegeneration: Pathophysiological consequences and therapeutic perspectives. Orphanet J Rare Dis 2021, 16, 201. [Google Scholar] [CrossRef]
- Lambrechts, R.A.; et al. CoA-dependent activation of mitochondrial acyl carrier protein links four neurodegenerative diseases. EMBO Mol Med 2019, 11, e10488. [Google Scholar] [CrossRef]
- Tai, S.; et al. Alpha-Lipoic Acid Mediates Clearance of Iron Accumulation by Regulating Iron Metabolism in a Parkinson's Disease Model Induced by 6-OHDA. Front Neurosci 2020, 14, 612. [Google Scholar] [CrossRef]
- Gray, D.A.a.W.J. Lipofuscin and Aging: A Matter of Toxic Waste. Sci Aging Knowledge Environ 2005. [Google Scholar] [CrossRef]
- Moreno-García, A.; Kun, A.; Calero, O.; Medina, M.; Calero, M. An Overview of the Role of Lipofuscin in Age-Related Neurodegeneration. Front. Neurosci 2018, 12. [Google Scholar] [CrossRef]
- onig, J.; Ott, C.; Hugo, M.; Jung, T.; Bulteau, A.-L.; et al. Mitochondrial contribution to lipofuscin formation. Redox Biol 2017. [Google Scholar]
- Villalon-Garcia, I.; et al. Vicious cycle of lipid peroxidation and iron accumulation in neurodegeneration. Neural Regen Res 2023, 18, 1196–1202. [Google Scholar]
- Suarez-Rivero, J.M.; et al. UPR(mt) activation improves pathological alterations in cellular models of mitochondrial diseases. Orphanet J Rare Dis 2022, 17, 204. [Google Scholar] [CrossRef]
- Suarez-Rivero, J.M.; et al. Pterostilbene in Combination With Mitochondrial Cofactors Improve Mitochondrial Function in Cellular Models of Mitochondrial Diseases. Front Pharmacol 2022, 13, 862085. [Google Scholar] [CrossRef]
- Rochette, L.; et al. Direct and indirect antioxidant properties of alpha-lipoic acid and therapeutic potential. Mol Nutr Food Res 2013, 57, 114–125. [Google Scholar] [CrossRef]
- Park, S.; et al. Physiological effect and therapeutic application of alpha lipoic acid. Curr Med Chem 2014, 21, 3636–3645. [Google Scholar] [CrossRef]
- Zhou, B.; et al. Alpha Lipoamide Ameliorates Motor Deficits and Mitochondrial Dynamics in the Parkinson's Disease Model Induced by 6-Hydroxydopamine. Neurotox Res 2018, 33, 759–767. [Google Scholar] [CrossRef]
- Dziedziak, J.; Kasarello, K.; Cudnoch-Jedrzejewska, A. Dietary Antioxidants in Age-Related Macular Degeneration and Glaucoma. Antioxidants (Basel) 2021, 10. [Google Scholar] [CrossRef]
- Molz, P.; Schroder, N. Potential Therapeutic Effects of Lipoic Acid on Memory Deficits Related to Aging and Neurodegeneration. Front Pharmacol 2017, 8, 849. [Google Scholar] [CrossRef]
- Chen, P.; et al. Effects of alpha-lipoic acid on expression of iron transport and storage proteins in BV-2 microglia cells. Pharmacol Rep 2017, 69, 1–5. [Google Scholar] [CrossRef]
- Wang, Y.; et al. The Neuroprotection of Lysosomotropic Agents in Experimental Subarachnoid Hemorrhage Probably Involving the Apoptosis Pathway Triggering by Cathepsins via Chelating Intralysosomal Iron. Mol Neurobiol 2015, 52, 64–77. [Google Scholar] [CrossRef]
- Wu, Q.J.; et al. The sirtuin family in health and disease. Signal Transduct Target Ther 2022, 7, 402. [Google Scholar]
- Carafa, V.; et al. Sirtuin functions and modulation: From chemistry to the clinic. Clin Epigenetics 2016, 8, 61. [Google Scholar] [CrossRef]
- Rutanen, J.; et al. SIRT1 mRNA expression may be associated with energy expenditure and insulin sensitivity. Diabetes 2010, 59, 829–835. [Google Scholar] [CrossRef]
- de Kreutzenberg, S.V.; et al. Downregulation of the longevity-associated protein sirtuin 1 in insulin resistance and metabolic syndrome: Potential biochemical mechanisms. Diabetes 2010, 59, 1006–1015. [Google Scholar] [CrossRef]
- Imai, S.; Guarente, L. NAD+ and sirtuins in aging and disease. Trends Cell Biol 2014, 24, 464–471. [Google Scholar]
- Xin, T.; Lu, C. SirT3 activates AMPK-related mitochondrial biogenesis and ameliorates sepsis-induced myocardial injury. Aging (Albany NY) 2020, 12, 16224–16237. [Google Scholar] [CrossRef]
- Zhang, J.; et al. Mitochondrial Sirtuin 3: New emerging biological function and therapeutic target. Theranostics 2020, 10, 8315–8342. [Google Scholar] [CrossRef]
- Verdin, E. NAD(+) in aging, metabolism, and neurodegeneration. Science 2015, 350, 1208–1213. [Google Scholar] [CrossRef]
- Kim, T.S.; et al. SIRT3 promotes antimycobacterial defenses by coordinating mitochondrial and autophagic functions. Autophagy 2019, 15, 1356–1375. [Google Scholar] [CrossRef]
- Imai, S. Dissecting systemic control of metabolism and aging in the NAD World: The importance of SIRT1 and NAMPT-mediated NAD biosynthesis. FEBS Lett 2011, 585, 1657–1662. [Google Scholar] [CrossRef]
- Chen, C.; et al. SIRT1 and aging related signaling pathways. Mech Ageing Dev 2020, 187, 111215. [Google Scholar] [CrossRef]
- Katsyuba, E.; et al. NAD(+) homeostasis in health and disease. Nat Metab 2020, 2, 9–31. [Google Scholar] [CrossRef]
- van de Ven, R.A.H.; Santos, D.; Haigis, M.C. Mitochondrial Sirtuins and Molecular Mechanisms of Aging. Trends Mol Med 2017, 23, 320–331. [Google Scholar] [CrossRef]
- Silaghi, C.N.; Farcas, M.; Craciun, A.M. Sirtuin 3 (SIRT3) Pathways in Age-Related Cardiovascular and Neurodegenerative Diseases. Biomedicines 2021, 9. [Google Scholar] [CrossRef]
- Finley, L.W.; et al. Succinate dehydrogenase is a direct target of sirtuin 3 deacetylase activity. PLoS ONE 2011, 6, e23295. [Google Scholar] [CrossRef]
- Meng, H.; et al. SIRT3 Regulation of Mitochondrial Quality Control in Neurodegenerative Diseases. Front Aging Neurosci 2019, 11, 313. [Google Scholar] [CrossRef]
- Hiromasa, Y.; et al. Organization of the cores of the mammalian pyruvate dehydrogenase complex formed by E2 and E2 plus the E3-binding protein and their capacities to bind the E1 and E3 components. J Biol Chem 2004, 279, 6921–6933. [Google Scholar] [CrossRef]
- Wang, C.; et al. Protective effects of metformin against osteoarthritis through upregulation of SIRT3-mediated PINK1/Parkin-dependent mitophagy in primary chondrocytes. Biosci Trends 2019, 12, 605–612. [Google Scholar] [CrossRef]
- Hirschey, M.D.; et al. SIRT3 regulates mitochondrial fatty-acid oxidation by reversible enzyme deacetylation. Nature 2010, 464, 121–125. [Google Scholar] [CrossRef]
- Sakakibara, I.; et al. Fasting-induced hypothermia and reduced energy production in mice lacking acetyl-CoA synthetase 2. Cell Metab 2009, 9, 191–202. [Google Scholar] [CrossRef]
- Schwer, B.; et al. Reversible lysine acetylation controls the activity of the mitochondrial enzyme acetyl-CoA synthetase 2. Proc Natl Acad Sci U S A 2006, 103, 10224–10229. [Google Scholar] [CrossRef]
- Shimazu, T.; et al. SIRT3 deacetylates mitochondrial 3-hydroxy-3-methylglutaryl CoA synthase 2 and regulates ketone body production. Cell Metab 2010, 12, 654–661. [Google Scholar] [CrossRef]
- Lombard, D.B.; et al. Mammalian Sir2 homolog SIRT3 regulates global mitochondrial lysine acetylation. Mol Cell Biol 2007, 27, 8807–8814. [Google Scholar] [CrossRef]
- Hallows, W.C.; Lee, S.; Denu, J.M. Sirtuins deacetylate and activate mammalian acetyl-CoA synthetases. Proc Natl Acad Sci U S A 2006, 103, 10230–10235. [Google Scholar] [CrossRef]
- Cimen, H.; et al. Regulation of succinate dehydrogenase activity by SIRT3 in mammalian mitochondria. Biochemistry 2010, 49, 304–311. [Google Scholar] [CrossRef]
- Fritz, K.S.; et al. Ethanol metabolism modifies hepatic protein acylation in mice. PLoS ONE 2013, 8, e75868. [Google Scholar] [CrossRef]
- Ahn, B.H.; et al. A role for the mitochondrial deacetylase Sirt3 in regulating energy homeostasis. Proc Natl Acad Sci U S A 2008, 105, 14447–14452. [Google Scholar] [CrossRef]
- Cheng, A.; et al. Mitochondrial SIRT3 Mediates Adaptive Responses of Neurons to Exercise and Metabolic and Excitatory Challenges. Cell Metab 2016, 23, 128–142. [Google Scholar] [CrossRef]
- Mattson, M.P.; Gleichmann, M.; Cheng, A. Mitochondria in neuroplasticity and neurological disorders. Neuron 2008, 60, 748–766. [Google Scholar] [CrossRef]
- Cui, X.X.; et al. SIRT3 deacetylated and increased citrate synthase activity in PD model. Biochem Biophys Res Commun 2017, 484, 767–773. [Google Scholar] [CrossRef]
- Huang, W.; et al. SIRT3 Expression Decreases with Reactive Oxygen Species Generation in Rat Cortical Neurons during Early Brain Injury Induced by Experimental Subarachnoid Hemorrhage. Biomed Res Int 2016, 2016, 8263926. [Google Scholar] [CrossRef]
- Tseng, A.H.; Shieh, S.S.; Wang, D.L. SIRT3 deacetylates FOXO3 to protect mitochondria against oxidative damage. Free Radic Biol Med 2013, 63, 222–234. [Google Scholar] [CrossRef]
- Cilleros-Holgado, P.; et al. mtUPR Modulation as a Therapeutic Target for Primary and Secondary Mitochondrial Diseases. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Gerhart-Hines, Z.; et al. Metabolic control of muscle mitochondrial function and fatty acid oxidation through SIRT1/PGC-1alpha. EMBO J 2007, 26, 1913–1923. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



