Submitted:
13 July 2024
Posted:
16 July 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Instruments
2.3. Methods
2.3.1. Formula One (F1). AgNP Synthesis Using Trisodium Citrate as a Reducing Agent
2.3.2. Formula Two (F2). AgNP Synthesis Using Sodium Borohydride as Reducing Agent
2.3.3. Formula Three (F3). AgNP Synthesis by a Combined Method Using Sodium Borohydride as Primary Reductant and Trisodium Citrate as Secondary Reductant
2.3.4. Formula Four (F4). AgNP Synthesis by a Combined Method Using Trisodium Citrate and Sodium Borohydride as Reducing Agent in the Presence of Hydrogen Peroxide
2.3.5. Formula Five (F5). AgNP Synthesis Using Polyethylene Glycol as a Reducing Agent
2.4. Purification
2.5. Characterization
2.6. Microbiological Studies
2.6.1. Bacterial Strains Collection
2.6.2. Well Diffusion Test
2.6.3. Determination of Minimum Bactericidal Concentration (MBC)
3. Results and Discussions
3.1. Chemical Characterization
3.1.1. Quantitative Analysis
3.1.2. Thermogravimetric Analysis
3.1.3. Morphological Analysis
3.1.4. Stability Analysis
3.2. Microbiological Study
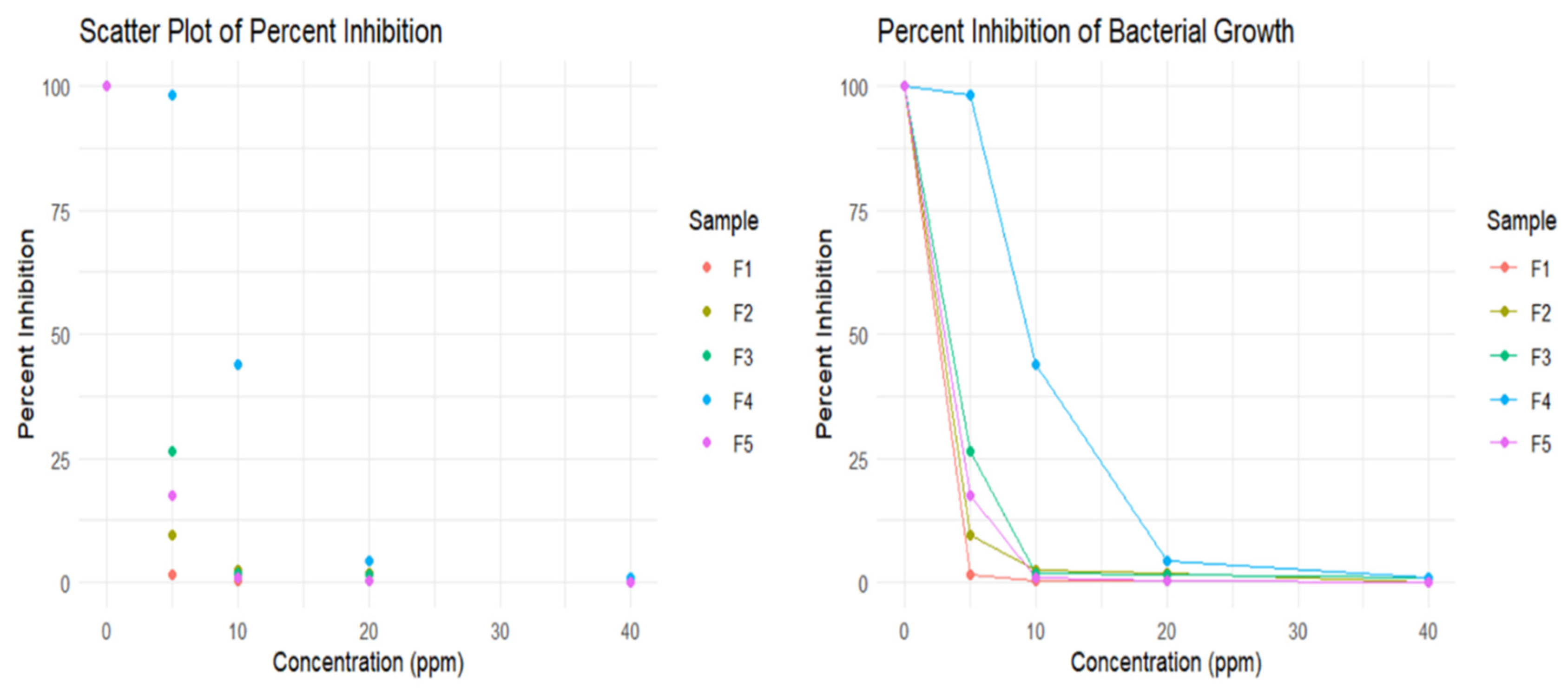
3.2.1. Determination of MBC
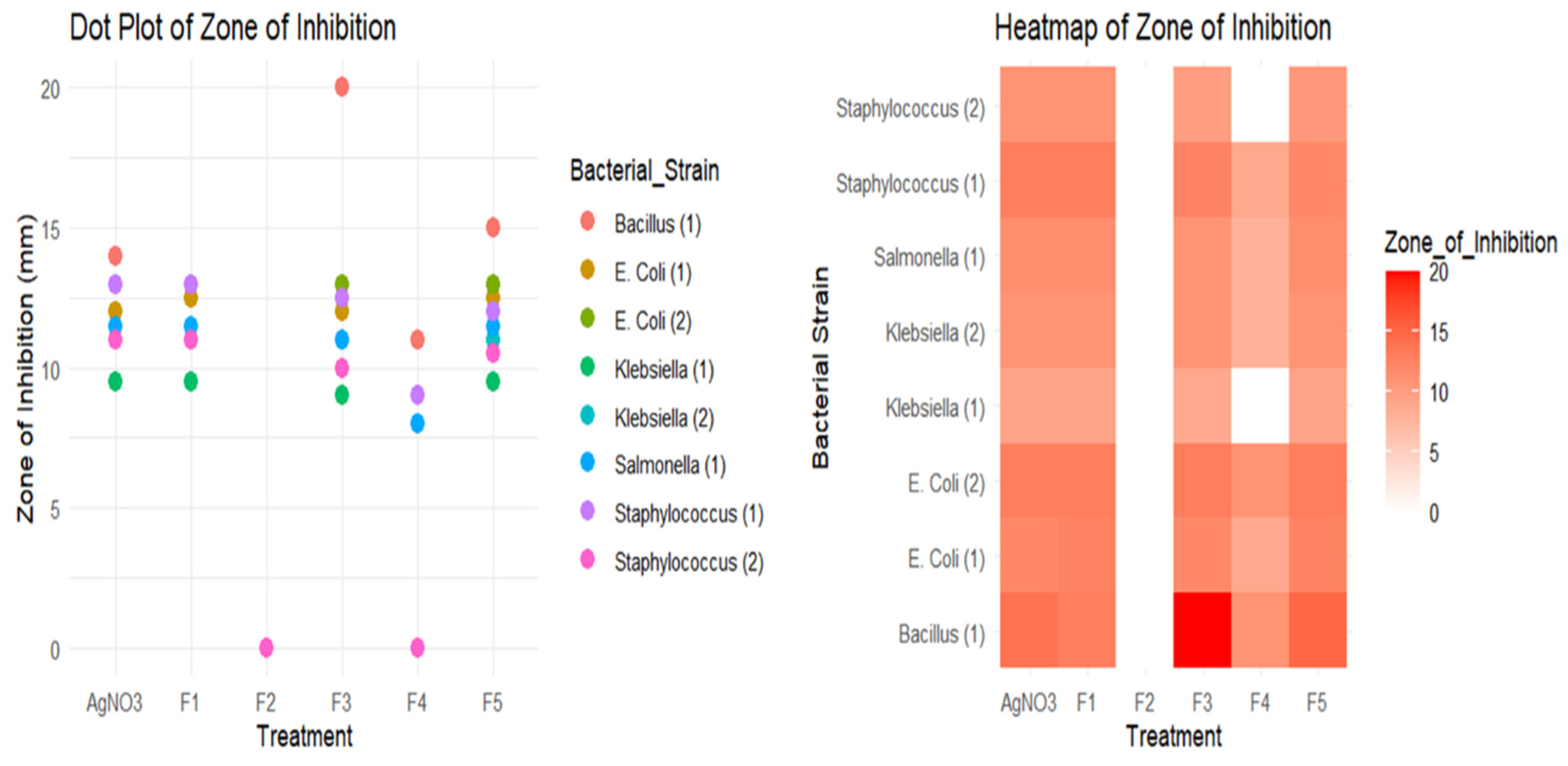
3.2.2. Well Diffusion Test
4. Conclusion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Chaturvedi, S.; Dave, P.N.; Shah, N.K. Applications of nano-catalyst in new era. Journal of Saudi Chemical Society 2012, 16, 307–325. [Google Scholar] [CrossRef]
- Liu, W.T. Nanoparticles and their biological and environmental applications. J Biosci Bioeng. 2006, 102, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Blackman, J.A.; Binns, C. Chapter 1 Introduction. In Handbook of Metal Physics; Blackman, J.A., Ed.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 1–16. [Google Scholar]
- Mulvaney, P. Nanoscience vs Nanotechnology—Defining the Field. ACS Nano 2015, 9, 2215–2217. [Google Scholar] [CrossRef] [PubMed]
- Meyers, M.A.; Mishra, A.; Benson, D.J. Mechanical properties of nanocrystalline materials. Progress in materials science 2006, 51, 427–556. [Google Scholar] [CrossRef]
- Murphy, C.J.; Sau, T.K.; Gole, A.M.; Orendorff, C.J.; Gao, J.; Gou, L.; et al. Anisotropic metal nanoparticles: Synthesis, assembly, and optical applications. The Journal of Physical Chemistry B 2005, 109, 13857–13870. [Google Scholar] [CrossRef]
- Banerjee, D.; Harfouche, R.; Sengupta, S. Nanotechnology-mediated targeting of tumor angiogenesis. Vascular cell. 2011, 3, 3. [Google Scholar] [CrossRef]
- Thiruvengadam, M.; Rajakumar, G.; Chung, I.M. Nanotechnology: Current uses and future applications in the food industry. 3 Biotech. 2018, 8, 74. [Google Scholar] [CrossRef] [PubMed]
- Bayda, S.; Adeel, M.; Tuccinardi, T.; Cordani, M.; Rizzolio, F. The History of Nanoscience and Nanotechnology: From Chemical-Physical Applications to Nanomedicine. Molecules 2019, 25, 112. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, M.; Hara, K.; Kudo, J. Bactericidal actions of a silver ion solution on Escherichia coli, studied by energy-filtering transmission electron microscopy and proteomic analysis. Applied and environmental microbiology. 2005, 71, 7589–7593. [Google Scholar] [CrossRef]
- Kim, J.S.; Kuk, E.; Yu, K.N.; Kim, J.H.; Park, S.J.; Lee, H.J.; et al. Antimicrobial effects of silver nanoparticles. Nanomedicine nanotechnology, biology, and medicine 2007, 3, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Souza, A.D.; Mehta, D.S.; Leavitt, R.W. Bactericidal activity of combinations of Silver-Water Dispersion™ with 19 antibiotics against seven microbial strains. Curr Sci. 2006, 91, 926–929. [Google Scholar]
- Durán, N.; Marcato, P.D.; De Souza, G.I.H.; Alves, O.L.; Esposito, E. Antibacterial Effect of Silver Nanoparticles Produced by Fungal Process on Textile Fabrics and Their Effluent Treatment. Journal of Biomedical Nanotechnology. 2007, 3, 203–208. [Google Scholar] [CrossRef]
- Jia, J.; Duan, Y.-Y.; Wang, S.-H.; Zhang, S.-F.; Wang, Z.-Y. Preparation and characterization of antibacterial silver-containing nanofibers for wound dressing applications. Journal of US-China Medical Science. 2007, 4, 52–54. [Google Scholar]
- Baker, C.; Pradhan, A.; Pakstis, L.; Pochan, D.J.; Shah, S.I. Synthesis and antibacterial properties of silver nanoparticles. Journal of nanoscience and nanotechnology. 2005, 5, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Shahverdi, A.R.; Fakhimi, A.; Shahverdi, H.R.; Minaian, S. Synthesis and effect of silver nanoparticles on the antibacterial activity of different antibiotics against Staphylococcus aureus and Escherichia coli. Nanomedicine Nanotechnology, Biology and Medicine. 2007, 3, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Ji, X.; Wu, H.; Zhao, L.; Li, J.; Yang, W. Shape Control of Silver Nanoparticles by Stepwise Citrate Reduction. The Journal of Physical Chemistry C. 2009, 113, 6573–6576. [Google Scholar] [CrossRef]
- Mulfinger, L.; Solomon, S.D.; Bahadory, M.; Jeyarajasingam, A.V.; Rutkowsky, S.A.; Boritz, C. Synthesis and Study of Silver Nanoparticles. Journal of Chemical Education. 2007, 84, 322. [Google Scholar] [CrossRef]
- Brown, A.N.; Smith, K.; Samuels, T.A.; Lu, J.; Obare, S.O.; Scott, M.E. Nanoparticles functionalized with ampicillin destroy multiple-antibiotic-resistant isolates of Pseudomonas aeruginosa and Enterobacter aerogenes and methicillin-resistant Staphylococcus aureus. Applied and environmental microbiology. 2012, 78, 2768–2774. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, N.; Goebl, J.; Lu, Z.; Yin, Y. A systematic study of the synthesis of silver nanoplates: Is citrate a “magic” reagent? Journal of the American Chemical Society 2011, 133, 18931–18939. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, J.F.A.; Saito, A.; Bido, A.T.; Kobarg, J.; Stassen, H.K.; Cardoso, M.B. Defeating Bacterial Resistance and Preventing Mammalian Cells Toxicity Through Rational Design of Antibiotic-Functionalized Nanoparticles. Sci Rep. 2017, 7, 1326. [Google Scholar] [CrossRef] [PubMed]
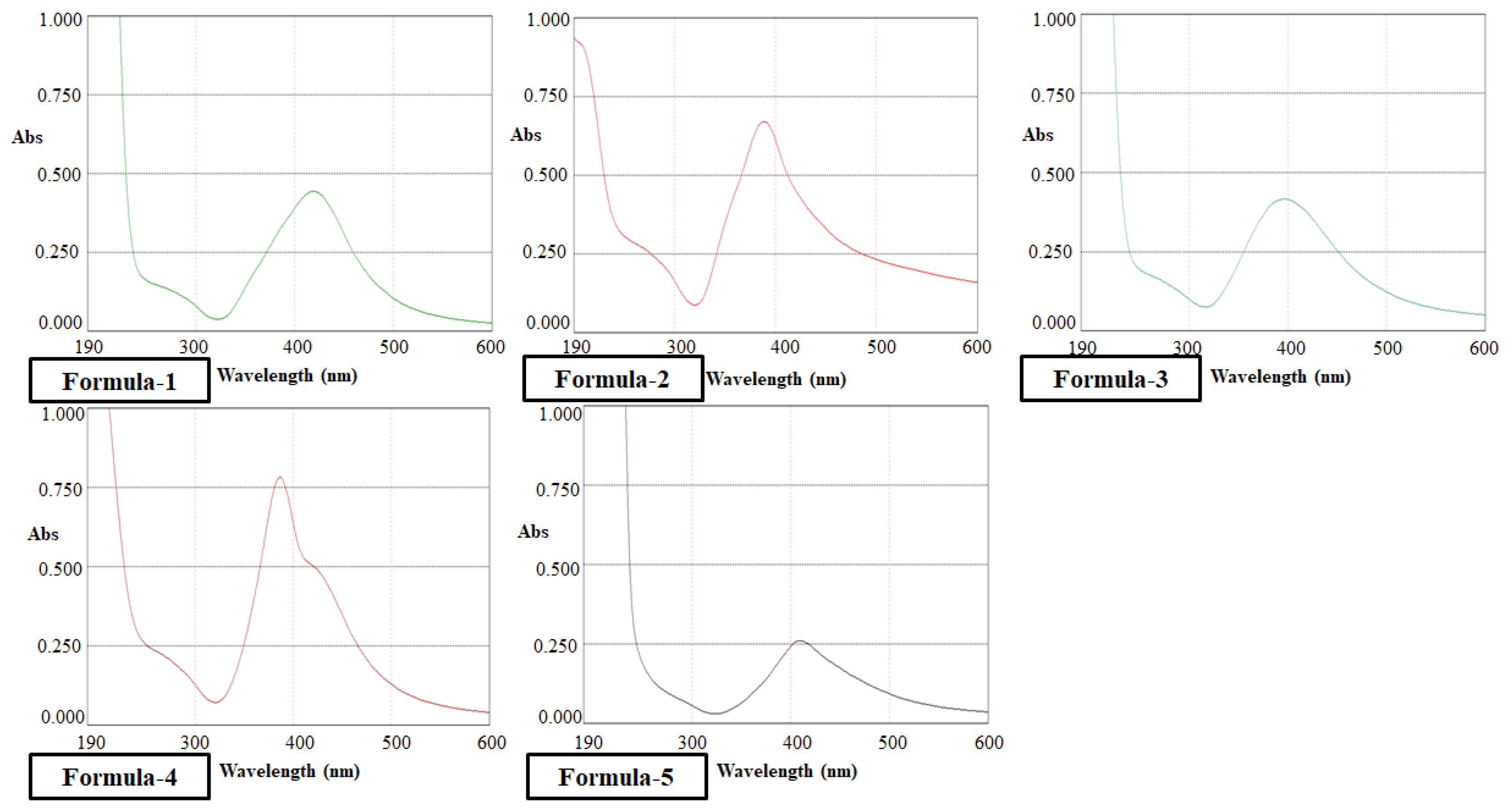
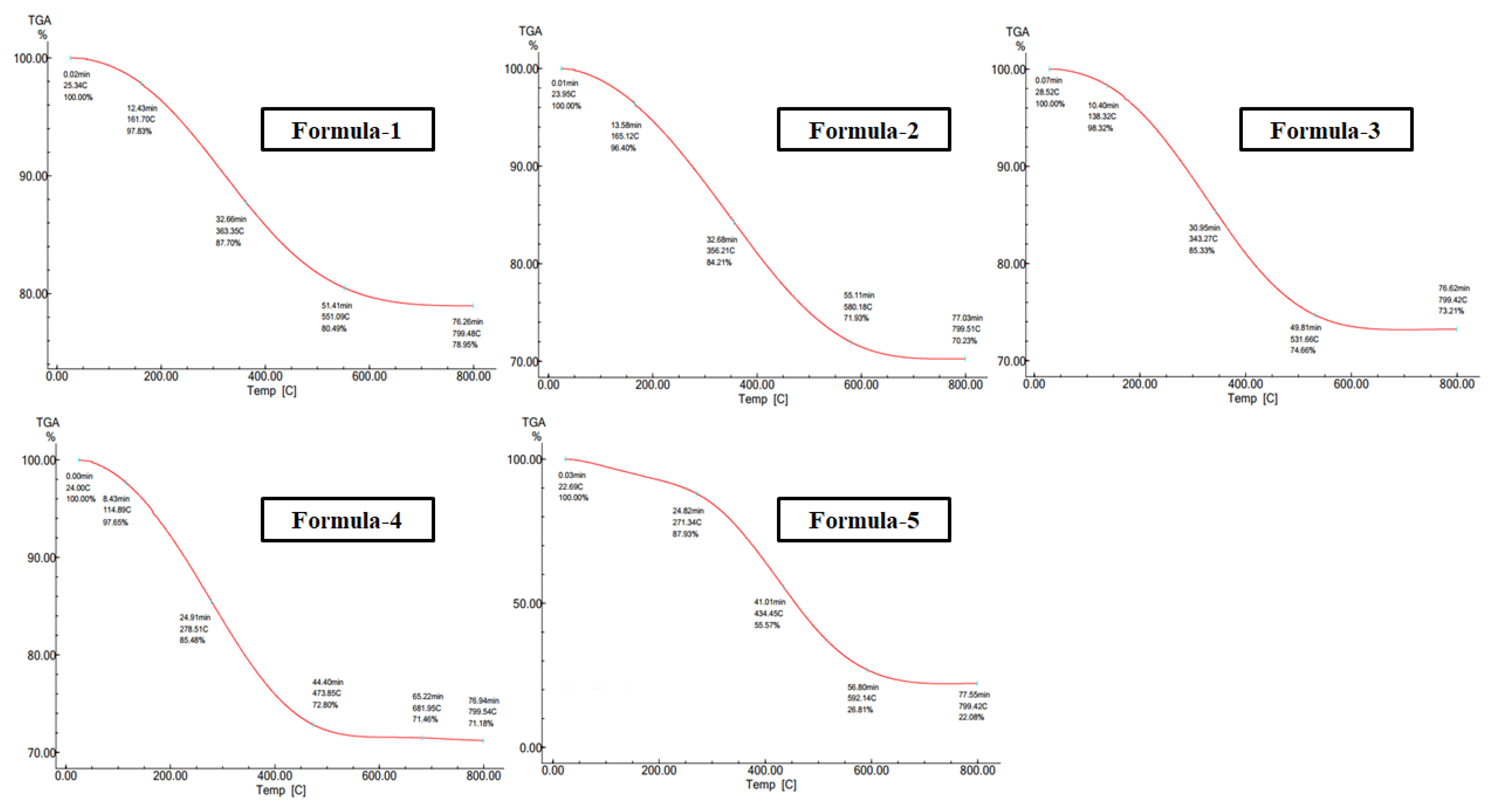

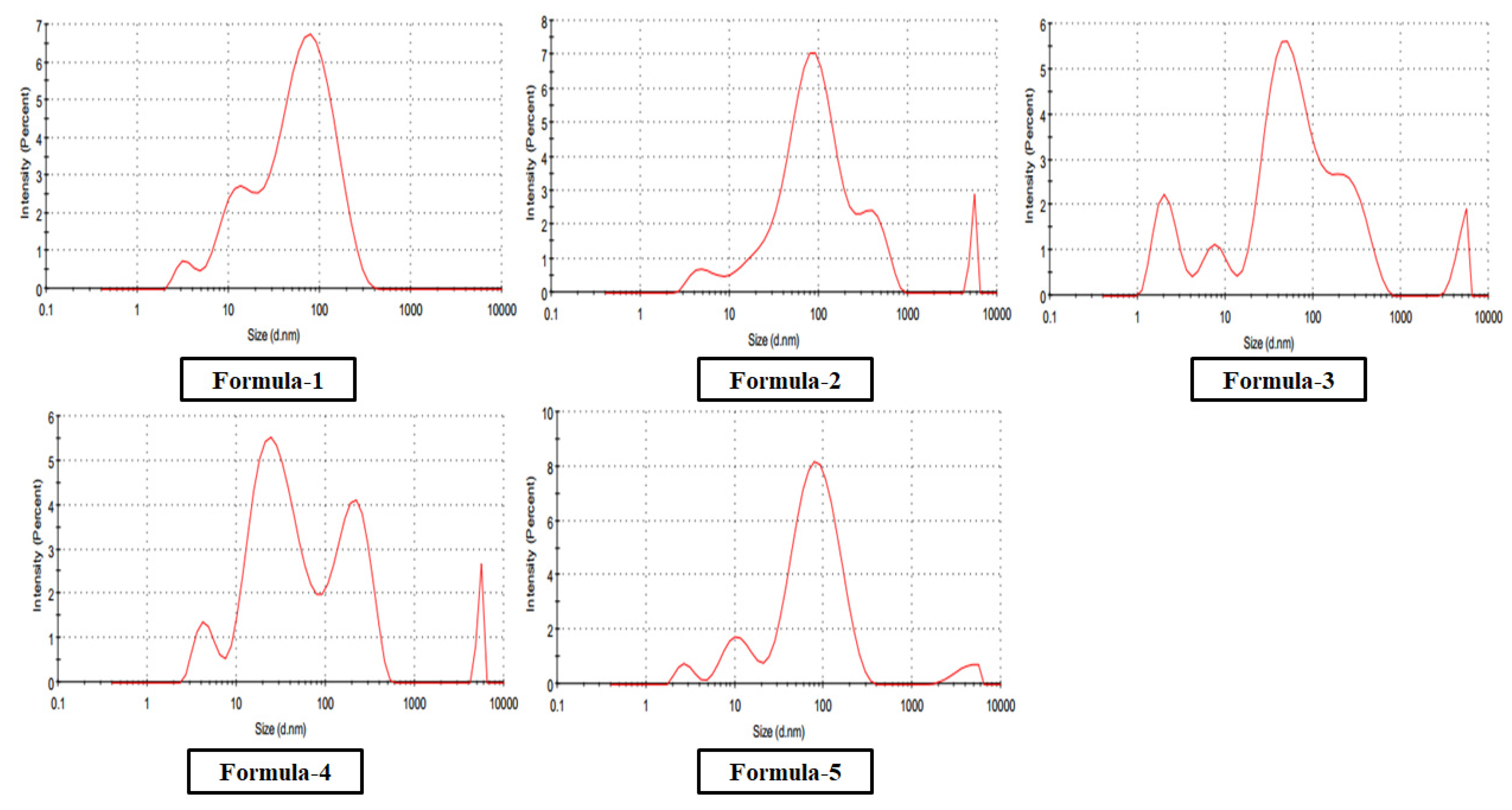
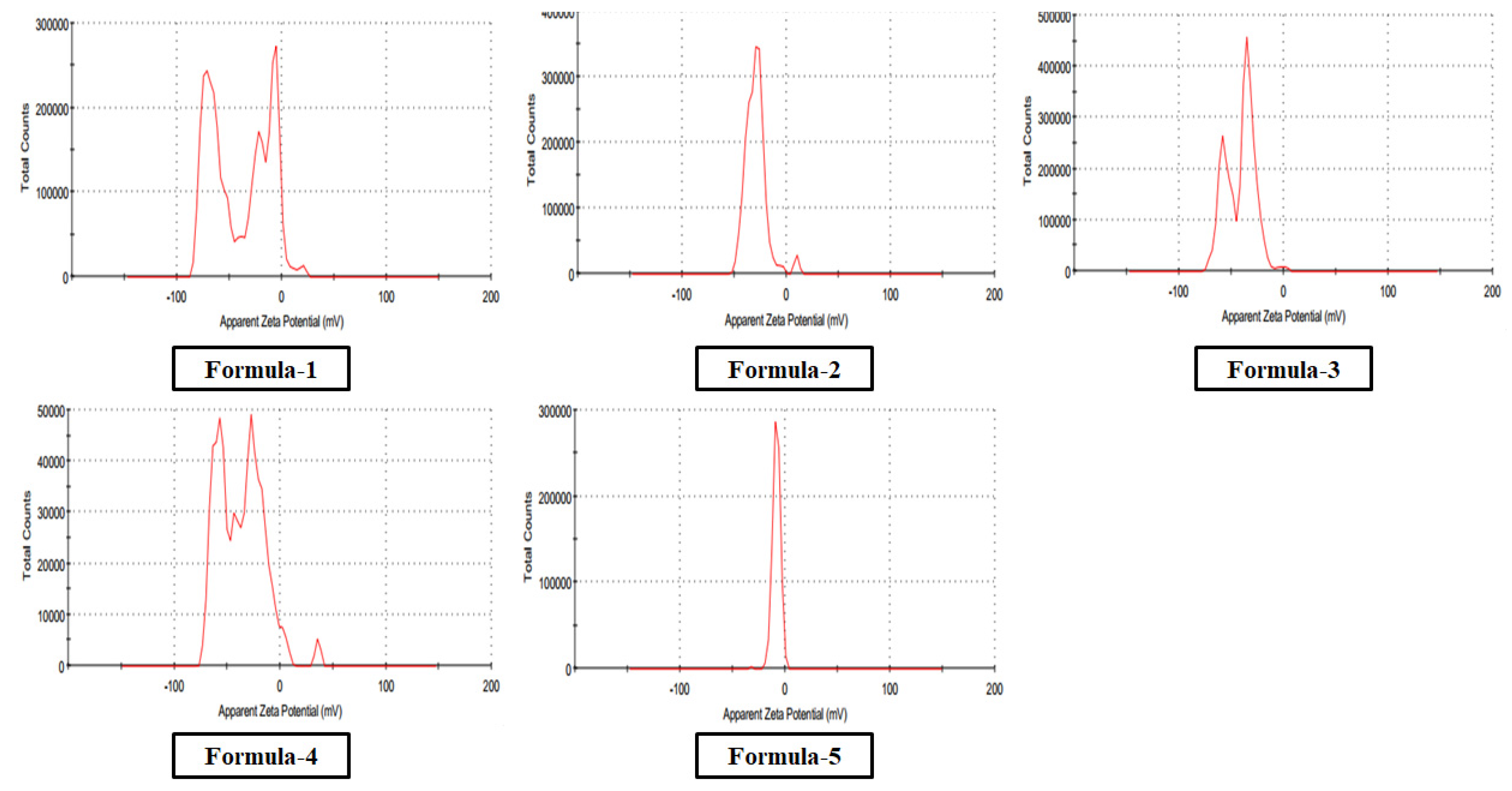
| Formula (Reducing Agent) |
Electrical Conductivity | Total Dissolved Solid (Solution Volume = 30 ml) |
Zeta Potential | Particle Size (Diameter) |
|---|---|---|---|---|
| Formula-1 (TSC) |
818 µS | 314 ppm | -39.4 mV | 87.15 nm |
| Formula-2 (NaBH4) |
848 µS | 176 ppm | -29.1 mV | 91.71 nm |
| Formula-3 (NaBH4 & TSC) |
779 µS | 297 ppm | -41.1 mV | 65.07 nm |
| Formula-4 (NaBH4 & TSC, H2O2) |
758 µS | 609 ppm | -36.7 mV | 30.91 nm |
| Formula-5 (PEG & PVP) |
871 µS | 191 ppm | -8.0 mV | 93.85 nm |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




