Submitted:
12 July 2024
Posted:
15 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Section
2.1. Materials and Measurements
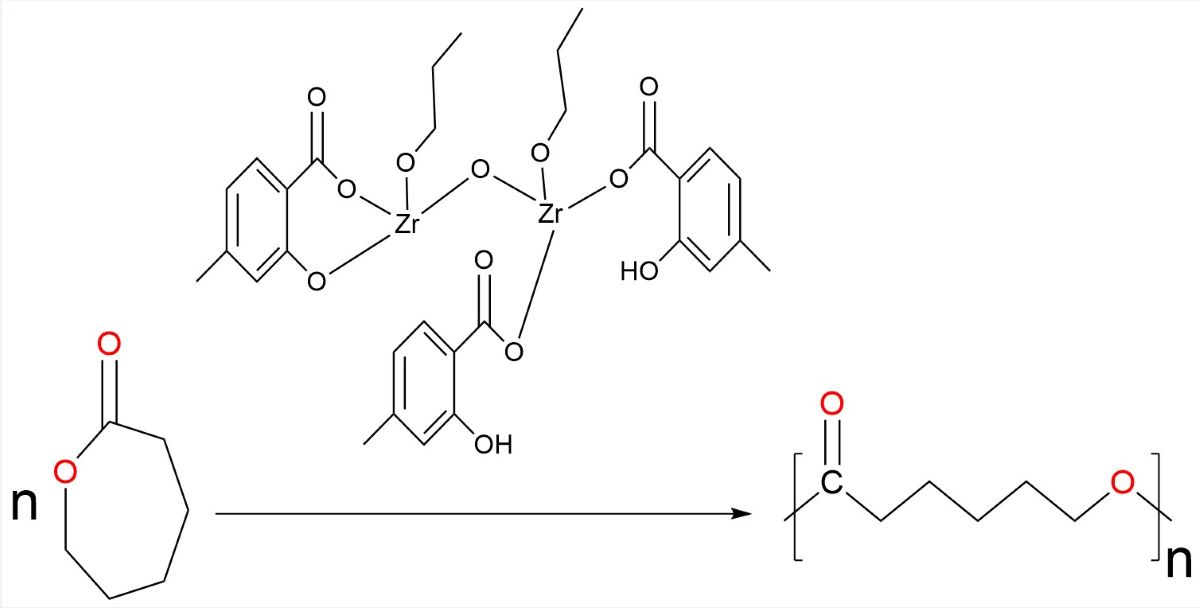
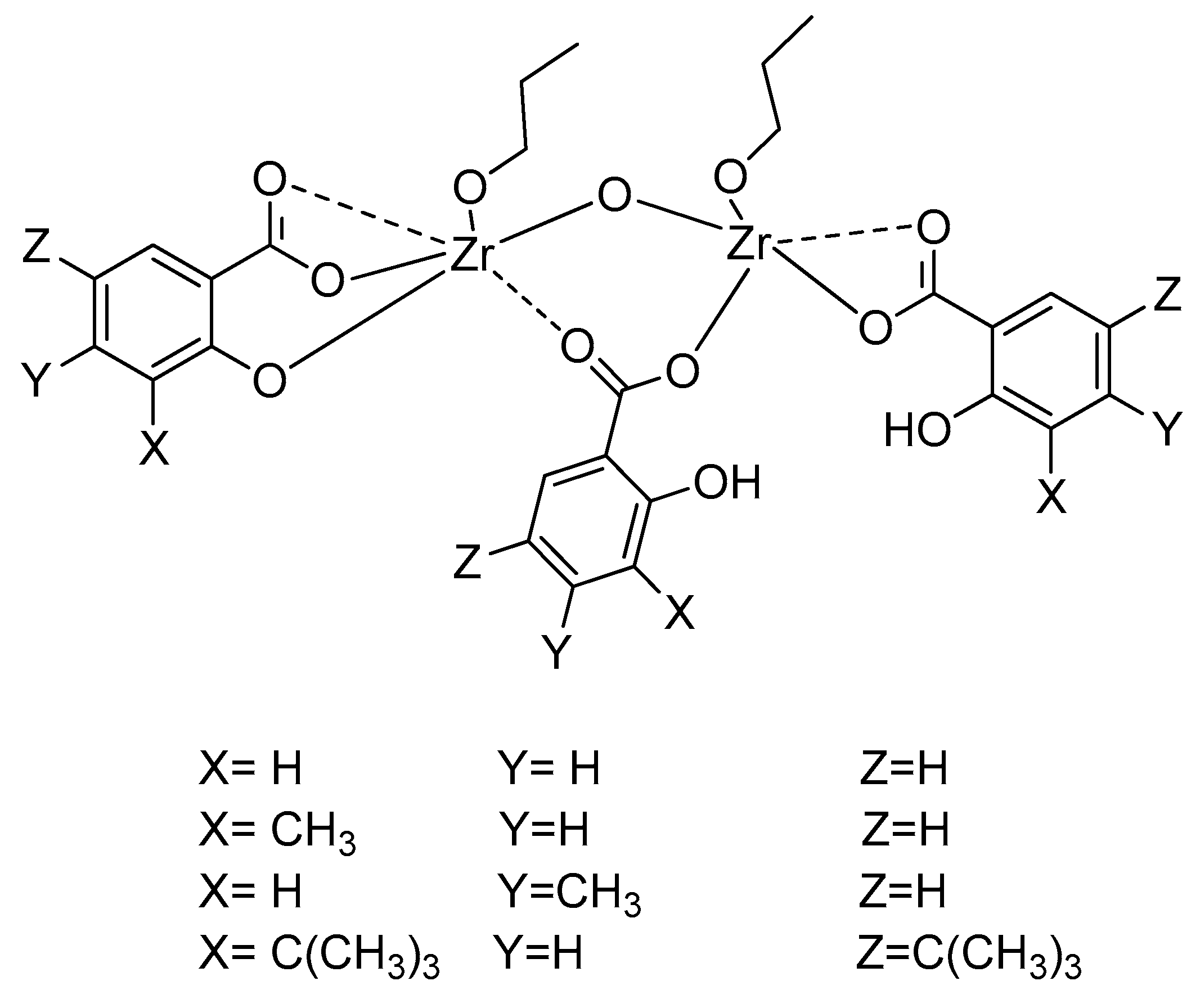
2.2. Preparation of [(SA-OH)2(SA)Zr2(OnPr)2O] (C1)
2.3. Preparation of [(3-Me-SA-OH)2(3-Me-SA)Zr2(OnPr)2O] (C2)
2.4. Preparation of [(4-Me-SA-OH)2(4-Me-SA)Zr2(OnPr)2O] (C3)
2.5. Preparation of [(3,5-But2-SA-OH)2(3,5-But2-SA)Zr2(OnPr)2O] (C4)
2.6. Preparation of [(SA-OH)2(SA)Zr2(OnBu)2O] (C5)
2.7. Preparation of [(3-Me-SA-OH)2(3-Me-SA)Zr2(OnBu)2O] (C6)
2.8. Preparation of [(4-Me-SA-OH)2(4-Me-SA)Zr2(OnBu)2O] (C7)
2.9. Preparation of [(3,5-But2-SA-OH)2(3,5-But2-SA)Zr2(OnBu)2O] (C8)
2.10. ROP of Ɛ-caprolactone with the catalyst [(4-Me-SA-OH)2(4-Me-SA)Zr2(OnBu)2O]
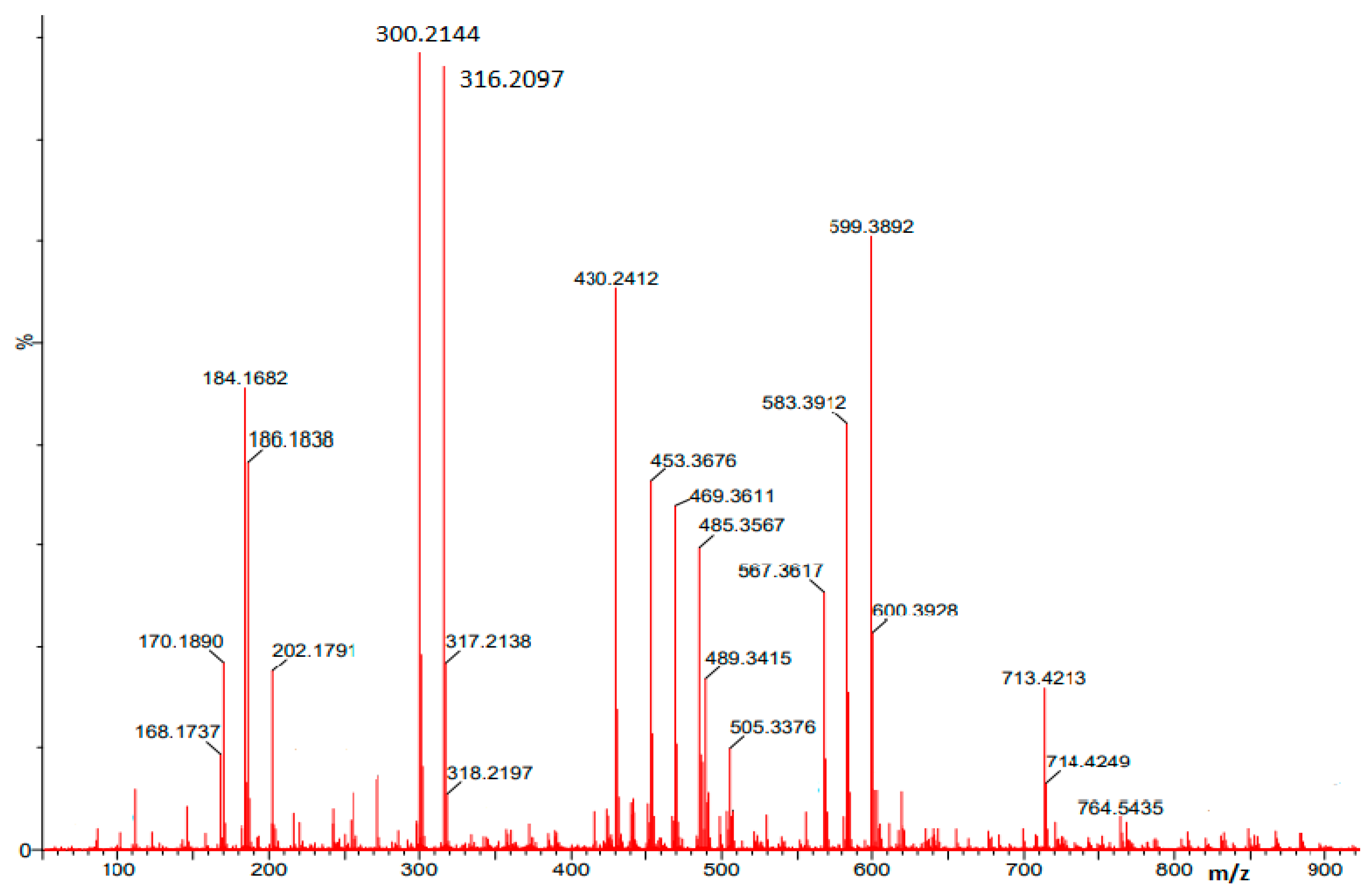
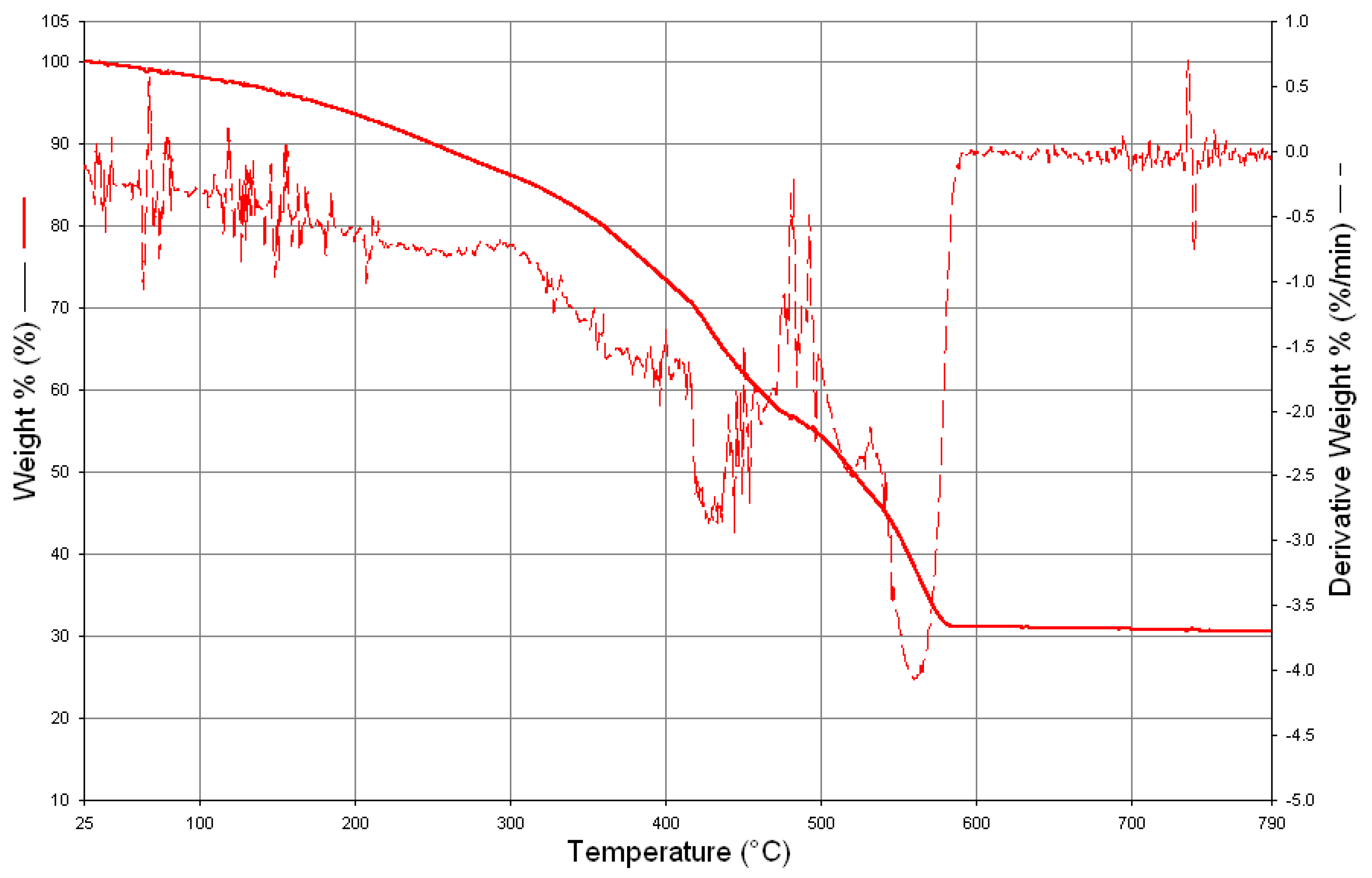
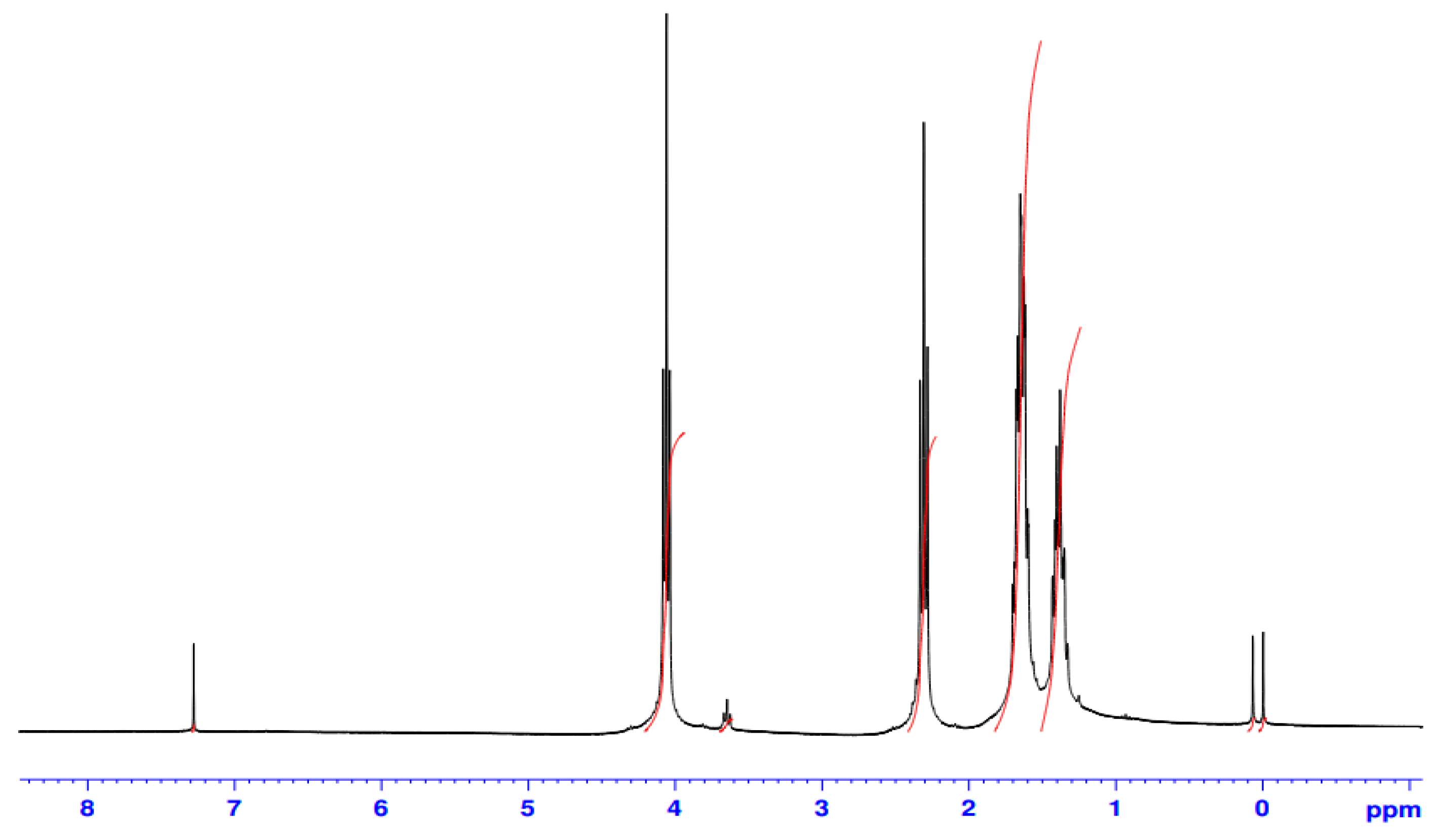
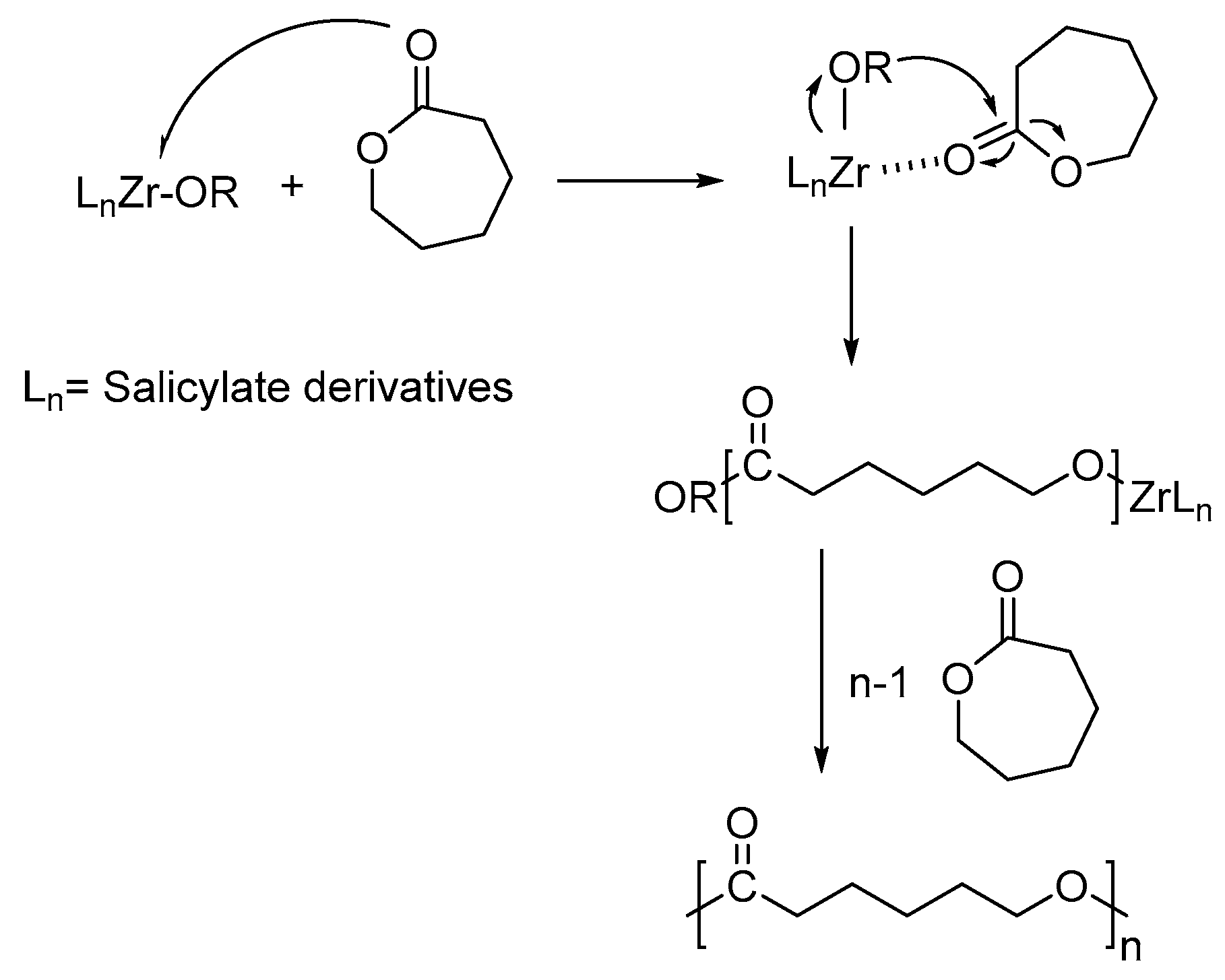
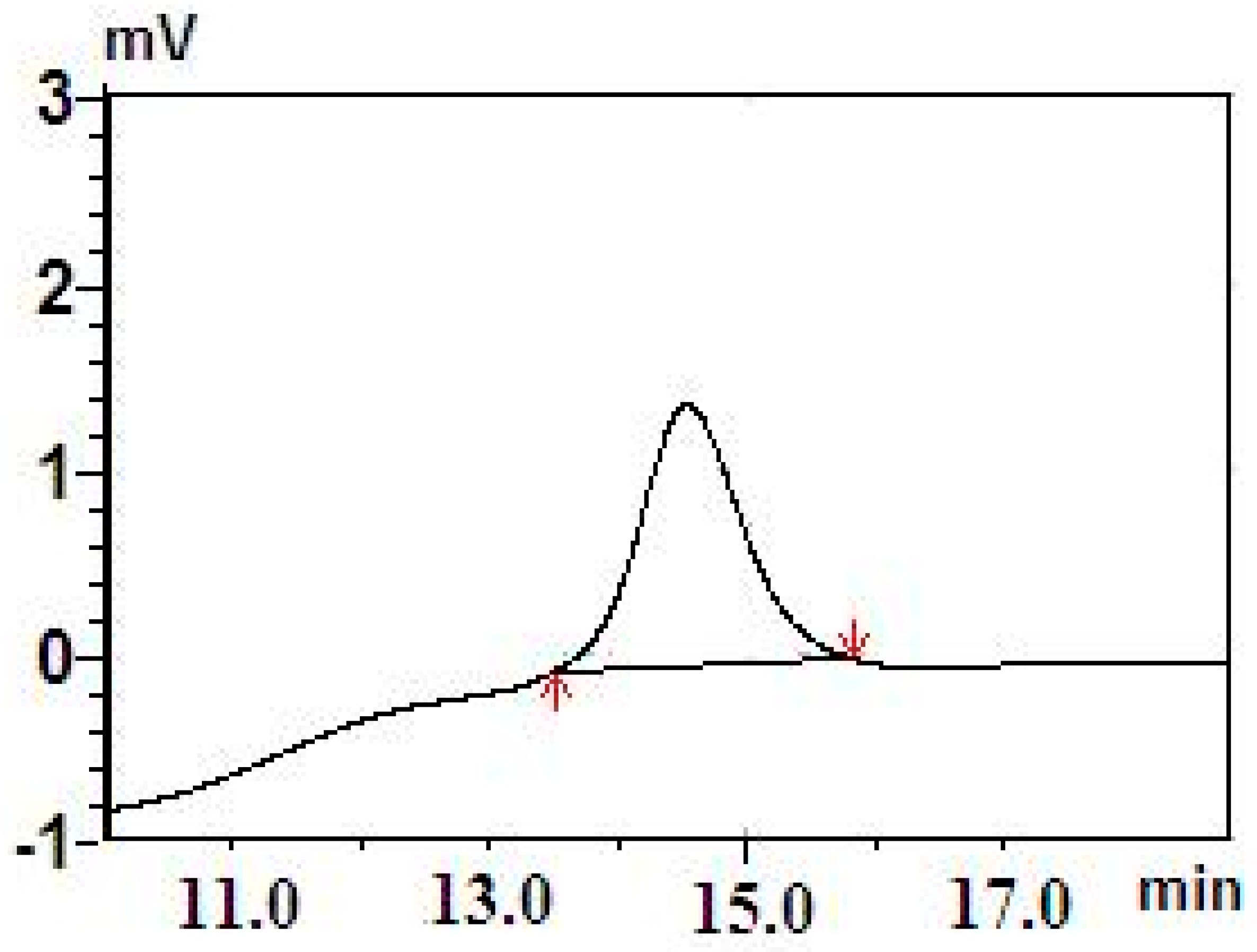
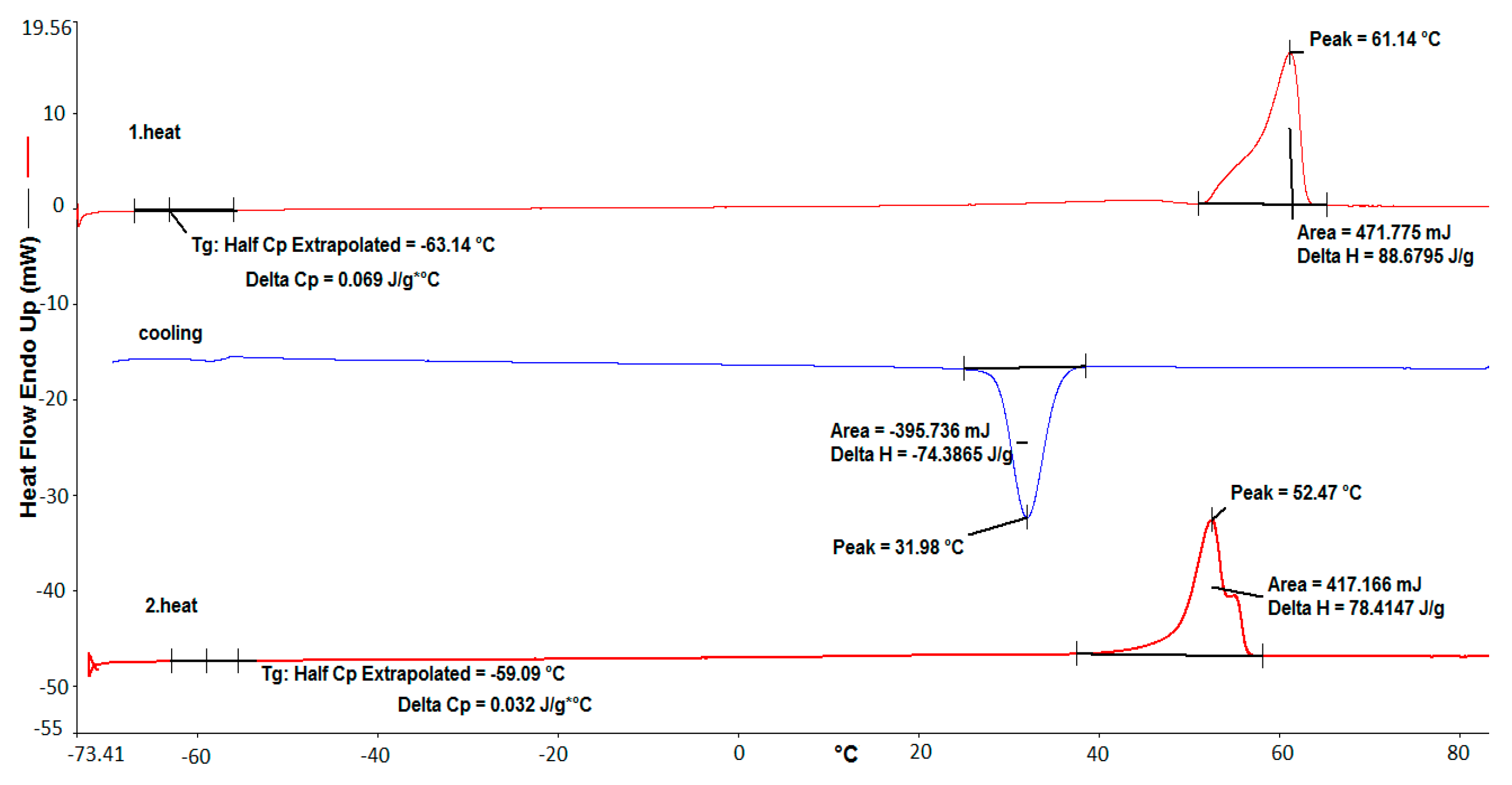
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kayan, G.Ö.; Kayan, A. Polycaprolactone Composites/Blends and Their Applications Especially in Water Treatment. ChemEng. 2023, 7, 104. [Google Scholar] [CrossRef]
- Pawar, R.; Pathan, A.; Nagaraj, S.; Kapare, H.; Giram, P.; Wavhale, R. Polycaprolactone and its derivatives for drug delivery. Polym. Adv. Technol. 2023, 34, 3296–3316. [Google Scholar] [CrossRef]
- Mandal, P.; Shunmugam, R. Polycaprolactone: a biodegradable polymer with its application in the field of self-assembly study. J. Macromol. Sci. A 2020, 58, 111–129. [Google Scholar] [CrossRef]
- Liu, Z.H.; Li, Y.; Zhang, C.J.; Zhang, Y.Y.; Cao, X.H.; Zhang, X.H. Synthesis of high-molecular-weight poly (ε-caprolactone) via heterogeneous zinc-cobalt (III) double metal cyanide complex. Giant 2020, 3, 100030. [Google Scholar] [CrossRef]
- El Yousfi, R.; Brahmi, M.; Dalli, M.; Achalhi, N.; Azougagh, O.; Tahani, A.; El Idrissi, A. Recent advances in nanoparticle development for drug delivery: A comprehensive review of polycaprolactone-based multi-arm architectures. Polymers 2023, 15, 1835. [Google Scholar] [CrossRef] [PubMed]
- Dias, J.R.; Sousa, A.; Augusto, A.; Bártolo, P.J.; Granja, P.L. Electrospun polycaprolactone (PCL) degradation: An in vitro and in vivo study. Polymers 2022, 14, 3397. [Google Scholar] [CrossRef] [PubMed]
- Yalcin, G.; Yildiz, U.; Kayan, A. Preparation of Al, Ti, Zr-perfluoroheptanoate compounds and their use in ring opening polymerization. Appl. Catal. A: Gen. 2012, 423, 205–210. [Google Scholar] [CrossRef]
- Kayan, A. Recent studies on single site metal alkoxide complexes as catalysts for ring opening polymerization of cyclic compounds. Catal. Surv. Asia 2020, 24, 87–103. [Google Scholar] [CrossRef]
- Jansen, J.H.; Powell, A.B.; Specht, S.E.; Gerislioglu, S.; Hermans, I. Understanding the structure and reactivity of mixed titanium (IV) alkoxide and tin (II)/(IV) carboxylates as esterification catalysts. ACS Sustain. Chem. Eng. 2022, 10, 2484–2493. [Google Scholar] [CrossRef]
- Brown, S.E.; Mantaloufa, I.; Andrews, R.T.; Barnes, T.J.; Lees, M.R.; De Proft, F.; Pike, S.D. Photoactivation of titanium-oxo cluster [Ti6O6(OR)6(O2CtBu)6]: mechanism, photoactivated structures, and onward reactivity with O2 to a peroxide complex. Chem. Sci. 2023, 14, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Schubert, U. En route from metal alkoxides to metal oxides: metal oxo/alkoxo clusters. J. Sol-Gel Sci. Technol. 2023, 105, 587–595. [Google Scholar] [CrossRef]
- Kayan, G.Ö.; Kayan, A. Inorganic–organic hybrid materials of zirconium and aluminum and their usage in the removal of methylene blue. J. Inorg. Organomet. Polym. Mater. 2021, 31, 3613–3623. [Google Scholar] [CrossRef]
- Du, Q.; Rao, R.; Bi, F.; Yang, Y.; Zhang, W.; Yang, Y.; Zhang, X. Preparation of modified zirconium-based metal-organic frameworks (Zr-MOFs) supported metals and recent application in environment: a review and perspectives. Surf. Interfaces 2022, 28, 101647. [Google Scholar] [CrossRef]
- Zhang, H.; Xiong, P.; Li, G.; Liao, C.; Jiang, G. Applications of multifunctional zirconium-based metal-organic frameworks in analytical chemistry: Overview and perspectives. TrAC Trends Anal. Chem. 2020, 131, 116015. [Google Scholar] [CrossRef]
- Peprah, F.; Tarantola, G.E.; Plaman, A.S.; Vu, E.L.; Huynh, A.B.; Durr, C.B. Synthesis and catalytic activity of single-site group V alkoxide complexes for the ring-opening polymerization of ε-caprolactone. Dalton Trans. 2024, 53, 7073–7080. [Google Scholar] [CrossRef] [PubMed]
- Su, R.R.; Ganta, P.K.; Cheng, C.A.; Hu, Y.T.; Chang, Y.C.; Chang, C.J.; Wu, K.H. Ring-opening polymerization of ε-caprolactone and L-lactide using ethyl salicylate-bearing zinc complexes as catalysts. Mol. Catal. 2023, 537, 112965. [Google Scholar] [CrossRef]
- Buchard, A.; Davidson, M.G.; Gobius du Sart, G.; Jones, M.D.; Kociok-Köhn, G.; McCormick, S.N.; Mckeown, P. Coordination of ε-Caprolactone to a Cationic Niobium (V) Alkoxide Complex: Fundamental Insight into Ring-Opening Polymerization via Coordination–Insertion. Inorg. Chem. 2023, 62, 15688–15699. [Google Scholar] [CrossRef] [PubMed]
- Mankaev, B.N.; Karlov, S.S. Metal complexes in the synthesis of biodegradable polymers: achievements and prospects. Materials 2023, 16, 6682. [Google Scholar] [CrossRef] [PubMed]
- Mert, O.; Kayan, A. Synthesis and characterization of substituted salicylate zirconium compounds and their catalytic activity over ε-caprolactone. J. Incl. Phenom. Macrocycl. Chem. 2014, 80, 409–416. [Google Scholar] [CrossRef]
- Stefanov, B.I. Optically Transparent TiO2 and ZnO Photocatalytic Thin Films via Salicylate-Based Sol Formulations. Coatings 2023, 13, 1568. [Google Scholar] [CrossRef]
- Mao, Y.; Chen, G.H.; Yi, X.; Kang, Y.; Zhang, J.; Zhang, L. Preparation and visible-light response of salicylate-stabilized heterobimetallic Pb–Ti–Oxo clusters initiated via auxiliary quaternary ammonium salts and a solvent effect. Inorg. Chem. 2022, 61, 5017–5024. [Google Scholar] [CrossRef] [PubMed]
- Kickelbick, G.; Schubert, U. J. Chem. Soc. Dalton Trans. 1999, 1301–1305.
- Tuancharoensri, N.; Ross, G.M.; Kongprayoon, A.; Mahasaranon, S.; Pratumshat, S.; Viyoch, J.; Ross, S. In situ compatibilized blends of PLA/PCL/CAB melt-blown films with high elongation: Investigation of miscibility, morphology, crystallinity and modelling. Polymers 2023, 15, 303. [Google Scholar] [CrossRef] [PubMed]
- Limwanich, W.; Meepowpan, P.; Dumklang, M.; Funfuenha, W.; Rithchumpon, P.; Punyodom, W. Non-isothermal kinetics of the organocatalytic ring-opening polymerization of ε-caprolactone with metal-free α-hydroxy acids: Eco-friendly and facile synthesis process. Thermochim. Acta 2024, 736, 179734. [Google Scholar] [CrossRef]
- Rosa, R.P.; Ferreira, F.V.; Lona, L.M. Modeling of Ring Opening Polymerization: A short review with insights on how to develop the method of moments. Chem. Eng. Sci. 2021, 246, 116934. [Google Scholar] [CrossRef]
- Yildiz, B.C.; Kayan, A. Ti (IV)-silyliminophenolate catalysts for ϵ-caprolactone and L-Lactide polymerization. Sustain. Chem. Pharm. 2021, 21, 100416. [Google Scholar] [CrossRef]
- Limwanich, W.; Rakbamrung, N.; Meepowpan, P.; Funfuenha, W.; Kongsuk, J.; Punyodom, W. Solvent-free ring-opening polymerization of ε-caprolactone initiated by Mg (II), Sn (II), Zn (II), Al (III), and Sn (IV) derivatives: a comparative study. React. Kinet. Mech. Catal. 2023, 136, 381–395. [Google Scholar] [CrossRef]
- Malikmammadov, E.; Tanir, T.E.; Kiziltay, A.; Hasirci, V.; Hasirci, N. PCL and PCL-based materials in biomedical applications. J. Biomater. Sci. Polym. Ed. 2018, 29, 863–893. [Google Scholar] [CrossRef] [PubMed]
- Steinman, N.Y.; Bentolila, N.Y.; Domb, A.J. Effect of Molecular Weight on Gelling and Viscoelastic Properties of Poly (caprolactone)–b-Poly (ethylene glycol)–b-Poly (caprolactone)(PCL–PEG–PCL) Hydrogels. Polymers 2020, 12, 2372. [Google Scholar] [CrossRef] [PubMed]
- Mahović Poljaček, S.; Priselac, D.; Tomašegović, T.; Elesini, U.S.; Leskovšek, M.; Leskovac, M. Effect of the Addition of Nano-Silica and Poly (ε-caprolactone) on the Mechanical and Thermal Properties of Poly (lactic acid) Blends and Possible Application in Embossing Process. Polymers 2022, 14, 4861. [Google Scholar] [CrossRef] [PubMed]
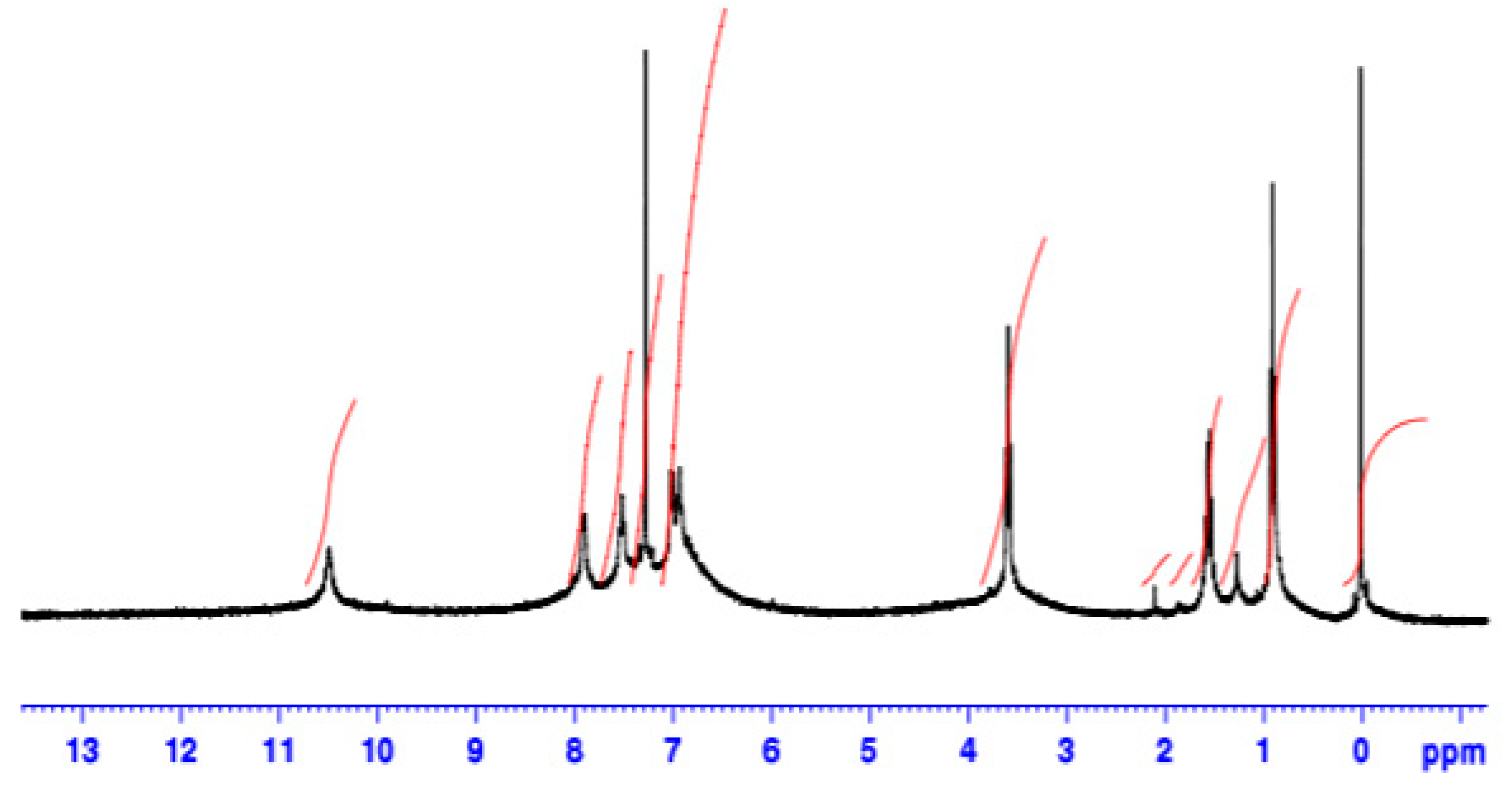
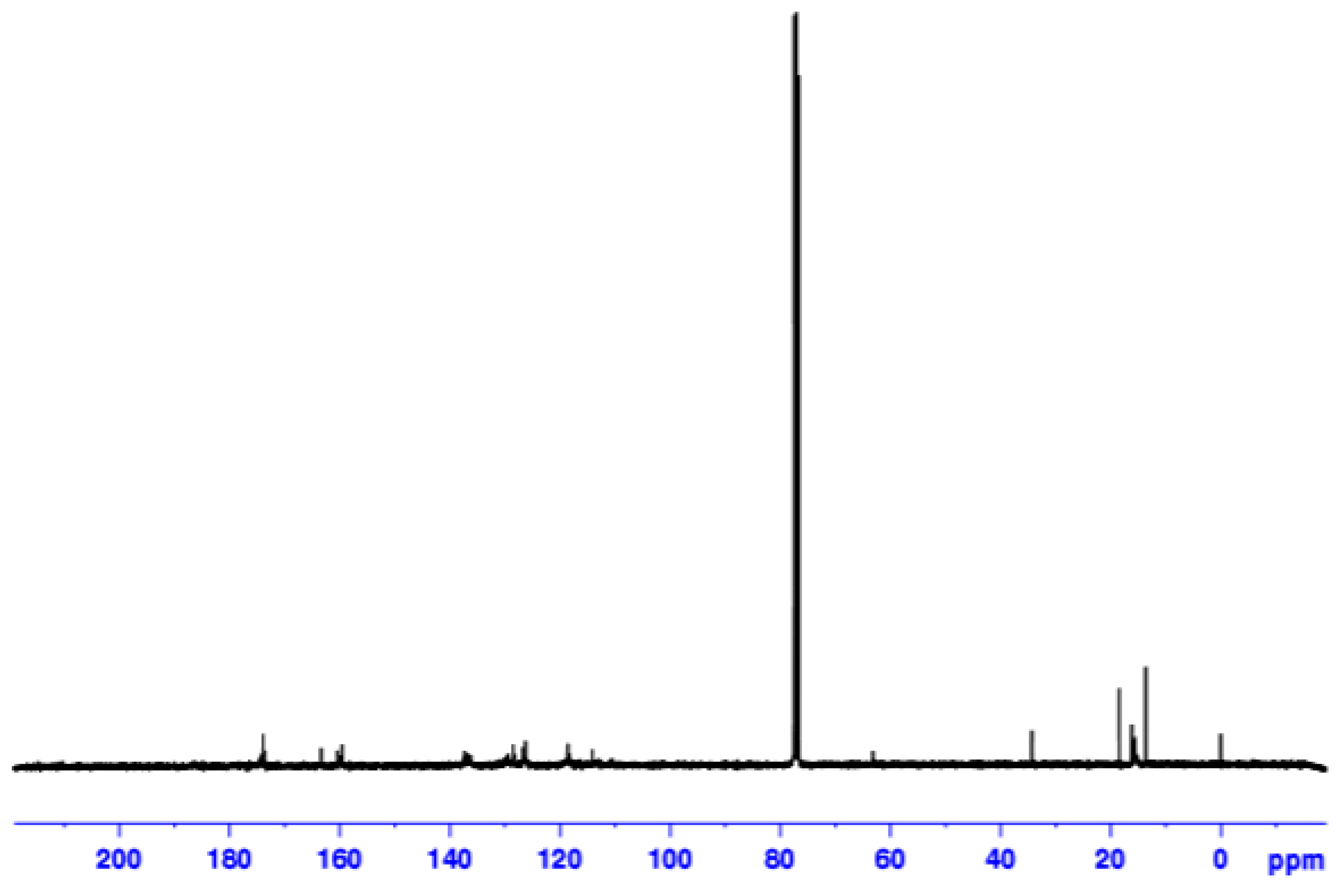
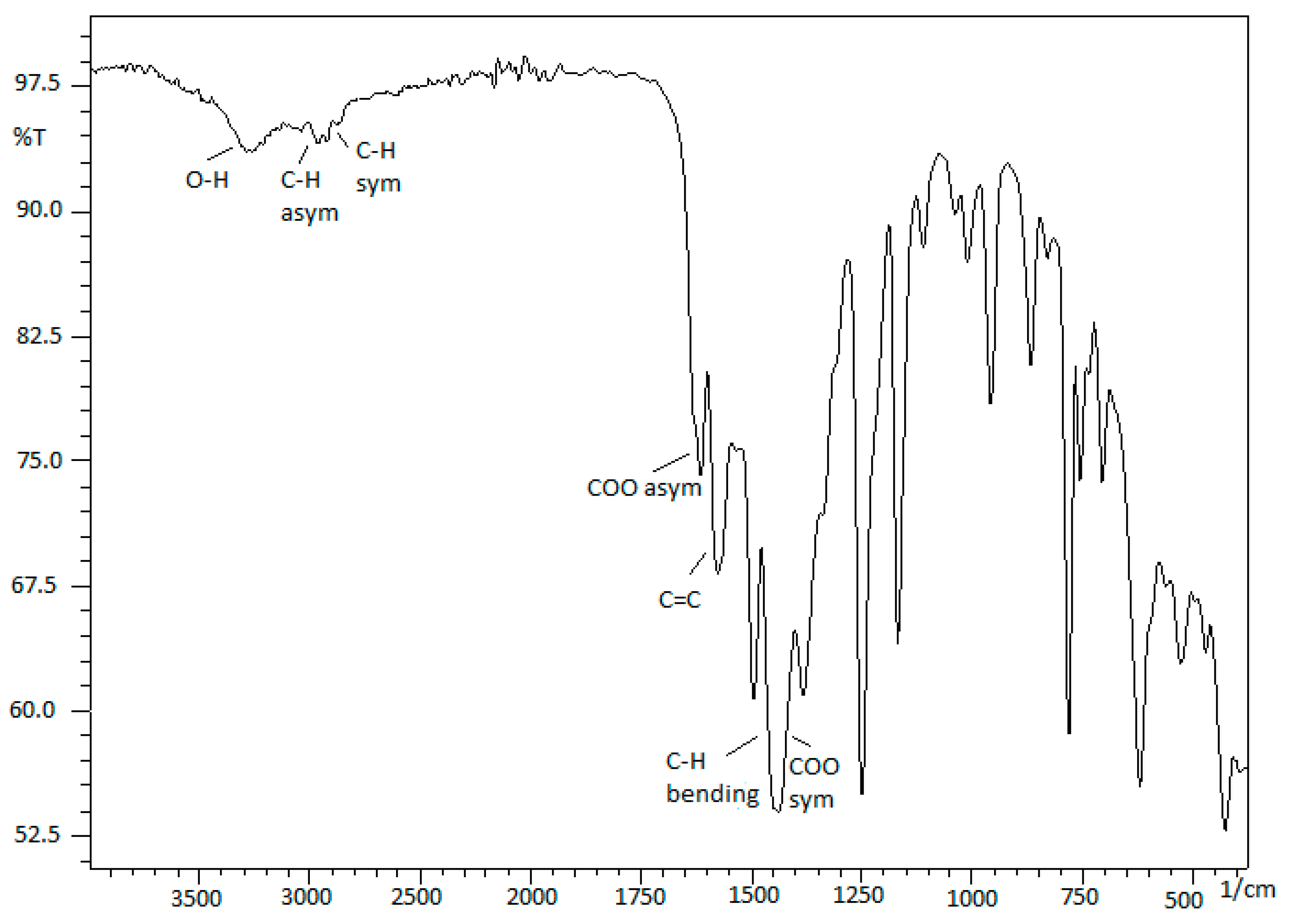
| Catalyst | T, oC | Time, h | Mw | Mn | (Mw/Mn) | Conversion (%) |
|---|---|---|---|---|---|---|
| C1 | 90 | 24 | 2710 | 2610 | 1.04 | 100 |
| C2 | 90 | 24 | 3450 | 3335 | 1.03 | 100 |
| C3 | 90 | 24 | 7420 | 7210 | 1.03 | 100 |
| C4 | 90 | 24 | 2020 | 1825 | 1.11 | 100 |
| C5 | 90 | 24 | 7240 | 6920 | 1.05 | 100 |
| C6 | 90 | 24 | 2665 | 2560 | 1.04 | 100 |
| C7 | 90 | 24 | 7490 | 7275 | 1.03 | 100 |
| C8 | 90 | 24 | 1520 | 1220 | 1.23 | 100 |
| Catalyst | T, oC | Time, h | Mw | Mn | (Mw/Mn) | Conversion (%) |
|---|---|---|---|---|---|---|
| C1 | 110 | 24 | 18740 | 15530 | 1.21 | 100 |
| C2 | 110 | 24 | 9150 | 8620 | 1.06 | 100 |
| C3 | 110 | 24 | 17510 | 17530 | 1.14 | 100 |
| C4 | 110 | 24 | 18415 | 16695 | 1.10 | 100 |
| C5 | 110 | 7 | 14650 | 12940 | 1.13 | 98 |
| C6 | 110 | 24 | 5110 | 4950 | 1.03 | 100 |
| C7 | 110 | 8 | 6770 | 6330 | 1.07 | 98 |
| C7 | 110 | 18 | 15900 | 12880 | 1.23 | 98 |
| C7 | 110 | 24 | 18905 | 14617 | 1.29 | 100 |
| C8 | 110 | 24 | 3140 | 2980 | 1.05 | 98 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




