Introduction
Radioisotope bone-targeted therapy can be divided into calcium analogs such as
223Ra and strontium-89, and bisphosphonate derivatives such as rhenium-186 etidronate.
223Ra, a calcium mimetic drug, is incorporated into bone by osteoblasts, introducing a targeted alpha therapy for the treatment of CRPC-BM in clinical practice [
1,
2,
3,
4].
In 2013, the ALSYMPCA study defined a significant improvement in OS of 3.6 months in CRPC-BM, with respect to placebo, which was unique for a radiopharmaceutical therapy [
5]. Following this publication, the Food and Drugs Administration and European Medicament Agency (EMA) approved
223Ra as a treatment option for symptomatic CRPC-BM patients with limited extraosseous disease. However, in 2018, ERA-223, a phase 3 randomized study promoted by EMA, investigating the effectiveness of
223Ra in combination with Abiraterone in CRPC compared to a control group with placebo, reported a 29% increase in the number of fractures compared to the placebo group [
6].
223Ra, as a calcium analogue, is incorporated into bone by osteoblasts through the same pathway as calcium [
7]. In a same manner, BS with diphosphonates spots the bone disease locations where
223Ra will act. However, it is well known, that metastatic CRPC, as an already advanced tumor disease, promotes tumor heterogeneity, with two implications: increasing chance of resistance to different therapies and limiting the disease detection in one step molecular imaging, supporting the use of additional diagnostic procedures.
The higher diagnostic accuracy of choline analogues-PET/CT compared to standard BS in detecting BM, as well as the ability to diagnose extraosseous disease in the same scan moved us to develop a prospective and multicenter study (ChoPET-Rad) [
8,
9]. We used the unique PET radiotracer available and authorized for prostate cancer use in Spain, the FCH PET, for patient selection before
223Ra and treatment response based on in clinical practice, monitoring
223Ra treatment rely on clinical and biochemical markers, while assessment of response with imaging techniques remains a controversial topic [
10,
11].
In addition, based on not all patients obtain benefit from
223Ra, patient selection is the cornerstone of the therapy’s effectiveness; however, it is a continuous challenge. In the last decade, despite efforts, no predictive factors have been established in clinical practice [
12,
13,
14,
15]. For this purpose, the development of a novel nomogram that includes both clinical and imaging variables, considering findings in BS and FCH PET/CT, for the prediction of therapeutic failure, could be useful for the optimal selection of those CRPC-BM patients eligible for this treatment.
Therefore, we aimed to study the value of FCH PET/CT and BS in patient selection and response assessment. Additionally, based on the scarcely evidence regarding the prognostic factors potentially able to select patients most likely to benefit from
223Ra [
16,
17], we aimed to obtain a nomogram system including clinical and radiomic variables able to predict therapeutic failure, bone events and OS in patients, with CRPC-BM, underwent
223Ra therapy.
Material and Methods
The present study (ChoPET-Rad) was designed as a prospective, multicentre (six centers) and non-randomized, and approved by an Ethical Committee (internal code:C-52/2016). Informed consent was obtained from all patients.
Patients
Patients with CRPC-BM who met all the inclusion criteria and none of the exclusion criteria for 223Ra treatment were included between January 2015 and December 2022. The inclusion criteria for initiating 223Ra treatment were: (a) patients with CRPC with symptomatic bone metastases and a negative or inconclusive CT for adenopathic involvement larger than 3 cm or visceral metastatic disease performed in the previous 6 weeks to request 223Ra treatment; (b) patients with a good bone marrow reserve that fulfilled the hematologic criteria necessary to administer 223Ra and (c) Eastern Cooperative Oncology Group (ECOG) performance status of 0–2 and life expectancy greater than 6 months.
The exclusion criteria were: (a) patients who denied participating in the study or (b) who did not fulfill any of the inclusion criteria or (c) were diagnosed of visceral or diffuse bone marrow involvement on baseline FCH PET/CT and/or BS.
Patients were scheduled for treating with 223Ra (55 KBq/kg, intravenously) in a 4-week cycle. Patients who had completed a total of six cycles of 223Ra were defined as treatment completion and those who did not finish the complete treatment protocol because of clinical progression or any other cause were considered as treatment failure. Patients maintained androgen deprivation therapy.
Clinical Assessment
Each patient was clinical, hematological and biochemically evaluated before each 223Ra administration and bimonthly or monthly, after the last 223Ra administration, depending on the patient clinical status and the subsequent therapeutic lines.
Clinical variables studied were: age, Gleason score, prostate-specific antigen (PSA), AP and lactate dehydrogenase (LDH) levels, time of evolution of prostate cancer, time of evolution of BM, therapeutic line that 223Ra represented, number and type of treatments received before 223Ra, castration-resistance date, bone events before, while or after 223Ra treatment, have received a bone protective medication like zoledronic acid and ECOG) performance status before 223Ra. LDH and AP were considered as pathological when their values were higher than 333 and 147 U/L, respectively. Treatments received after 223Ra were collected.
Bone or skeletal events (SE) were considered when BM required analgesic treatment with radiotherapy (RT) or orthopedic surgery, as well as the detection of pathological fractures or spinal cord compression syndrome, with or without the need for palliative RT treatment.
Clinical progression was assessed following the Radiographic Assessments for Detection of Advanced Recurrence (RADAR) II group recommendation [
18]. when at least two of the following indicators were reached: (1) convincing and consistent rise in PSA, defined as three consecutive rises, resulting in two 50% increases over the basal PSA value, (2) diagnostic imaging progression evidence, or (3) status performance worsening or appearance of clinical symptoms while the patient was on therapy. Diagnostic imaging progression was stablished when any of imaging techniques (BS or FCH PET/CT) defined progression.
223Ra treatment stopping underwent when clinical progression was addressed. However, the decision to stopping treatment, based on early progression (after the third or fourth 223Ra doses), was made by a multidisciplinary team (oncologist and nuclear medicine physician).
Biochemical PSA response was considered when a decrease in absolute PSA value of ≥ 30% between baseline PSA and interim (1 month later the third dose) or end-treatment (1 month later the sixth dose) was observed, and was defined as early or delayed response, respectively. Stability was considered for the rest of biochemical changes. AP and LDH progression were defined as an increase of ≥ 25% from last available determination during treatment with respect to baseline and response as a reduction of ≥ 30%. The rest of the conditions were considered as stability.
Clinical endpoints were: (a) treatment failure, defined as an incomplete 223Ra administration (less than six cycles) because of clinical progression of the disease or hematological toxicity and/or other clinical impairment, (b) PFS, attending to PSA evolution, defined as three consecutive rises in PSA, resulting in two ≥ 50% increases over the basal PSA value and (c) OS, defined as the elapsed time between the date of the start of 223Ra and the date of either death or the last follow-up. Last follow-up was performed in February 2024.
Patients that received less than three doses due to bone marrow failure or constitutional syndrome, among other causes, earlier to assess disease progression attending to PSA values, were excluded from the response assessment group. Each cause of failure was studied and noted individually, although, some of them were due to causes unrelated to the treatment.
Image Acquisition
FCH PET/CT and BS were performed, within a time interval of 4 weeks, before the first administration (baseline), before the fourth (interim) and after the sixth (end-treatment) 223Ra dose. BS was acquired 3 hours after injection of 740 MBq of 99mTc-methylene diphosphonate (MDP) in three reference centers. FCH PET/CT was performed in a unique reference hospital, 5–15 min after intravenous administration of 2–4 MBq/kg, in three-dimensional acquisition mode for 3 min per bed position, from skull to proximal legs. Low dose CT (120 kV, 80 mA) without contrast was performed for attenuation correction and as an anatomical map. The emission data was corrected for scatter, random coincidence events, and system dead time using the provided software.
Imaging Evaluation
Two independent observers visually evaluated FCH PET/CT and BS. In case of discordances, a third observer reviewed the studies to reach a consensus. BM extension was assessed on baseline FCH PET/CT and BS, considering maximum intensity projection in the former and planar images in the latter. Depending on the number of lesions in BS and FCH PET/CT, BM disease was classified as oligometastatic (≤ 5 lesions) or polimetastatic (> 5 lesions). Also, referring to the extension of the bone disease 4 grades were established, understanding grade I as single or oligometastatic disease, grade II between 6 and 20 metastases, grade III more than 20 metastases and grade IV superscan pattern. When more than 4 bone lesions were observed in BS or FCH PET/CT, with at least one extra-axial location, it was considered a high tumor burden. Furthermore, it was compared whether the predominant activity was osteogenic (BS dominant) or metabolic (PET dominant). Finally, morphological translation in the CT portion of FCH PET/CT was assessed, exclusively in pathological locations, and was classified as: predominantly osteoblastic (>50% of blastic lesions), osteolytic (>50% lytic lesions) or mixed (both blastic and lytic combined in a similar proportion).
Definition criteria for bone positivity was the presence of focal tracer uptake higher than background, regardless of its intensity, with or without any underlying lesion in CT and that could not be explained by a benign lesion like osteophytes or radiotracer excretion locations.
Special care was taken analyzing baseline FCH PET/CT where regions of interest were placed obtaining the maximum standardized uptake value (SUVmax) of the most hypermetabolic location and the average SUVmax of the five lesions with the highest FCH activity. Relation between SUVmax of pathological locations and the hepatic one was also compared.
For lymph node evaluation, any node with visually detectable uptake (higher than background) on FCH PET/CT, despite its size, was considered suspicious of malignancy. Rest of pelvic organs as prostate, bladder and seminal vesicles or visceral locations (lung and liver) were evaluated in the same way. Any of these was considered as soft tissue involvement (STI), except to visceral metastases that caused the exclusion of the patient for receiving 223Ra.
For response assessment, interim and end-treatment BS and FCH PET/CT were compared with respect to the previous one, evaluating all the included anatomical areas (preferable axial skeleton and proximal third of extremities), in order to establish response (baseline and interim, respectively), following the criteria for response formulated by the National Prostatic Cancer Treatment Group, formerly called the National Prostatic Cancer Project (NPCP) [
19]. and according to criteria of the European Organization for Research and Treatment of Cancer (EORTC), respectively [
20]. For progression assessment, the same criteria were used for BS and FCH PET/CT, consisted of the appearance of at least 2 new lesions.
Concordance between FCH PET/CT and BS baseline, interim and end-treatment, understanding this as a similar BM distribution between both techniques, was evaluated by visual inspection and classified as good (≥75% of the lesions), moderate (approximately between 75 and 25% of lesions) and bad (≤25% of lesions).
Results
Treatment with 223Ra was requested for 117 patients; however after clinical and imaging data derived from BS, CT and FCH PET/CT, 17 were dismissed for different reasons: 3 due to visceral metastatic disease (pulmonary, hepatic, and cerebral, respectively), 3 with locoregional infiltration (seminal vesicles, bladder, and pelvic lymph nodes, respectively), 4 due to extensive bone marrow infiltration observed by BS and/or FCH PET/CT, 2 with hematological toxicity (anemia and pancytopenia, respectively), 1 with a possible second primary tumor (hypernephroma), 2 for a deteriorated clinical condition with ECOG > 2, 1 for medullary canal infiltration visualized by FCH PET/CT, and 1 due to having a single bone metastatic lesion.
Finally, 100 patients were enrolled in the current study. Clinical and disease characteristics of patients are summarized in
Table 1 and 2. Most of them (n=97) had a good clinical status (ECOG 0-1) before the initiation of
223Ra treatment with a Gleason score ≥ 8 in 45 patients. Only 53 patients underwent previous prostate cancer radical treatment: 26 patients underwent prostatectomy (4 of them received adjuvant RT due to affected surgical margins on surgical specimen) and 27 patients were treated with radical RT. The remaining treatments received prior to
223Ra are described in
Table 2. Regarding the line of treatment,
223Ra was administered within the first three therapeutic lines in 80 patients, so in the global list of treatments,
223Ra represented the third line (median).
44 patients completed 6 doses of 223Ra therapy. PSA progression was detected in 70 patients during treatment with 223Ra, 54 experiencing it within the first 3 months from the start of treatment (early progression). In 3 other patients was not possible to assess if there was PSA progression as they died before this parameter could be elevated.
Skeletal events before 223Ra initiation occurred in 23 patients and included: 5 pathological fractures, 7 spinal cord compression syndromes, 3 of which were treated with RT, and 11 lesions treated with RT for pain. SE during and after 223Ra were documented in 3 and 26 patients, respectively; and included: 15 lesions treated with RT, 4 pathological fractures, one of which was treated with orthopedic surgery, and 7 cord compressive syndromes, 3 of which were treated with RT. 2 cases of spinal cord compression during 223Ra treatment lead treatment discontinuation.
Regarding bone protective treatments, 90 patients received Denosumab, Zoledronic acid, or both during their disease management (22, 58, and 10, respectively). 44 patients maintained them before, during, and after 223Ra treatment, while the rest only in some of them.
Median follow-up was 73 months with a median PFS and OS of 4 and 14 months, respectively. 97 patients experienced PSA progression during their follow-up, occurring in 70 during treatment with 223Ra, and more specifically in 54 within the first 3 months since the start of treatment. 83 received at least one treatment after 223Ra, with a mean of 2 subsequent lines received. Received treatments included abiraterone, enzalutamide, and one or more lines of chemotherapy in 26, 28, 41, and 28 of them, respectively. For those patients who completed 223Ra treatment, the median OS was 20 months compared to 9 months for those who did not complete it. During the follow-up 92 patients died.
According to the analysis of baseline parameters of imaging techniques, only a single patient did not undergo a baseline BS prior to treatment, while 87 underwent baseline FCH PET/CT. The detailed analysis of baseline parameters derived from the results of these techniques was described in
Table 1 and
Table 3. They all showed avidity in their lesions for the injected radiopharmaceuticals. Additionally, high tumor burden was observed in 61% of BS and 48% of FCH PET/CT. Furthermore, FCH PET/CT detected STI in sixteen patients, 14 with nodal involvement, 3 with local prostatic disease, and prostatic and nodal disease in 2 patients.
If we focus on the assessment of treatment response, 13 patients were excluded for receiving less than 3 doses of 223Ra: 5 due to hematological intolerance, 2 for symptomatic bone progression, 2 for visceral progression, 2 for primarily digestive intolerance, and the last 2 due to other causes unrelated to 223Ra (traumatic fracture and pulmonary embolism).
Only 7 patients experienced PSA response during 223Ra treatment, 4 of them early responses during the first 3 doses that kept during the rest of treatment and the rest delayed ones, during the second 3 doses.
AP progression was observed in 15 cases, and LDH progression in 17. AP and LDH responses were detected in 22 and 10 cases, respectively.
Regarding binary imaging response (progression vs. no progression), progression was more frequently observed in FCH PET/CT scans both at interim and end-treatment compared to BS. According to the progression pattern in interim FCH PET/CT, in most cases, progression was osseous, but in 15 of them soft tissue involvement was observed: 3 exclusively nodal, 7 nodal and osseous, and 5 visceral and osseous. The proportion of patients who progressed at the end of treatment FCH PET/CT was lower (51.3% vs 64.4% in the interim): 13 exclusively osseous, 3 nodal, 1 visceral, 2 osseous and nodal, and 1 osseous and visceral. The degree of agreement between interim BS and FCH PET/CT was weak (k: 0.349; p<0.001), being higher in the case of binary response assessment (progression vs. no progression) (k=0.447; p<0.001). No agreement was observed between end-of-treatment studies (k=0.157; p=0.120 and k=0.211; p=0.075 in binary response). The distribution of results is shown in
Table 4.
The results of the Chi-square analysis between the different variables and therapeutic failure rate and SE during or posterior
223Ra are summarized in
Table 5 and
Table 6. The number of patients who did not receive complete treatment with
223Ra was associated with those with poorer performance status (ECOG > 0) (p= 0.038), those who did not receive prior bone protective treatment (p= 0.040), or with pathological baseline AP levels (p= 0.030) or LDH and p= 0.039), among others.
In the case of SE, only the variables of additional treatments after
223Ra, OS (≤12 months vs >12 months), and a high tumor burden on FCH PET/CT showed significant associations (
Table 6). Paradoxically, patients with a higher number of lines of treatment following
223Ra showed a statistically significant occurrence of new SE during or after
223Ra treatment (p< 0.001). In fact, any additional therapeutic line increased the risk in 42.7%. However, in multivariate analysis, only OS showed significant association with SE as patients with OS longer than 12 months had 10.795 times an increased risk of SE (p= 0.003). This result was inconsistent for a nomogram design based on the higher the OS; the higher is the probability of receiving subsequent therapeutic lines and suffering SE.
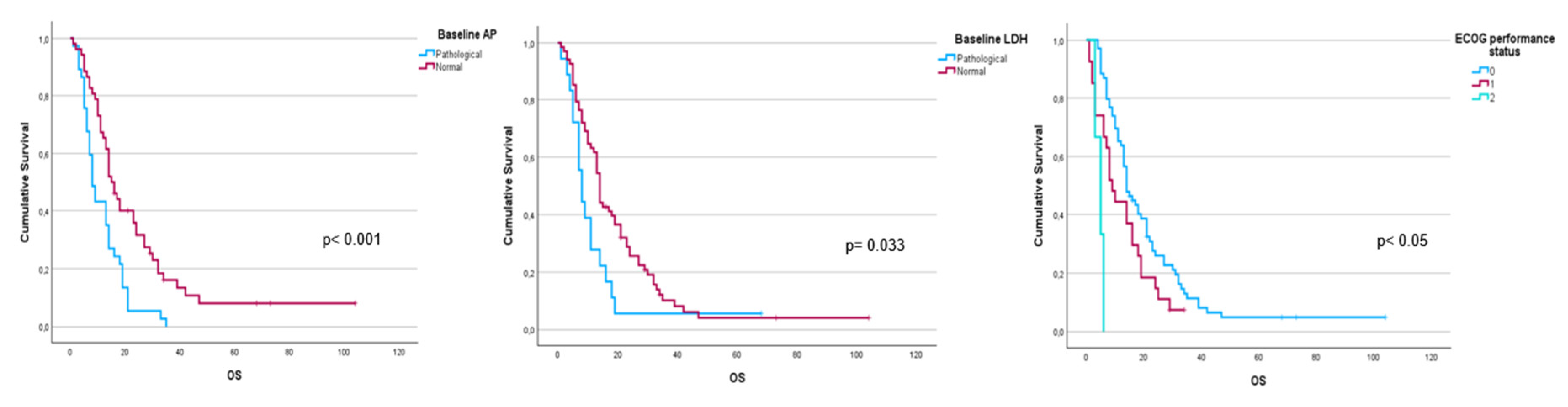
Performance status before to
223Ra (p< 0.001) and AP baseline levels (p< 0.001) and LDH (p= 0.033) were found to have a significant impact on OS (
Figure 1), as well as other factors such as completion of treatment with
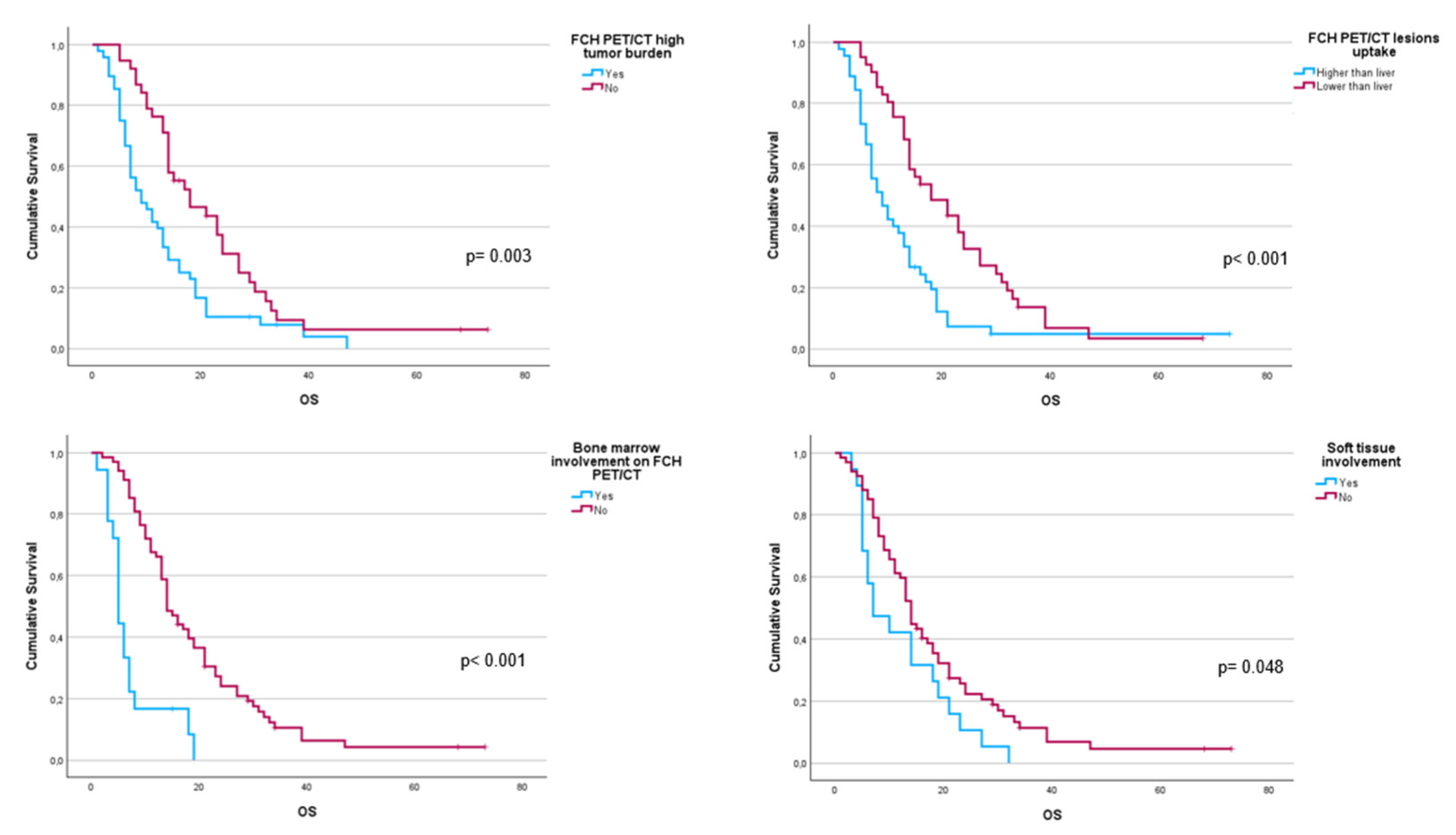
223Ra (p< 0.001). Regarding imaging variables, both the Chi-square test and the Log-Rank test showed that derived FCH PET/CT variables had statistical significance. Thus, high tumor burden (p= 0.003), uptake of the most hipermetabolic lesion above the liver (p< 0.001), or STI in FCH PET/CT study (p= 0.048) were associated with a lower median OS (
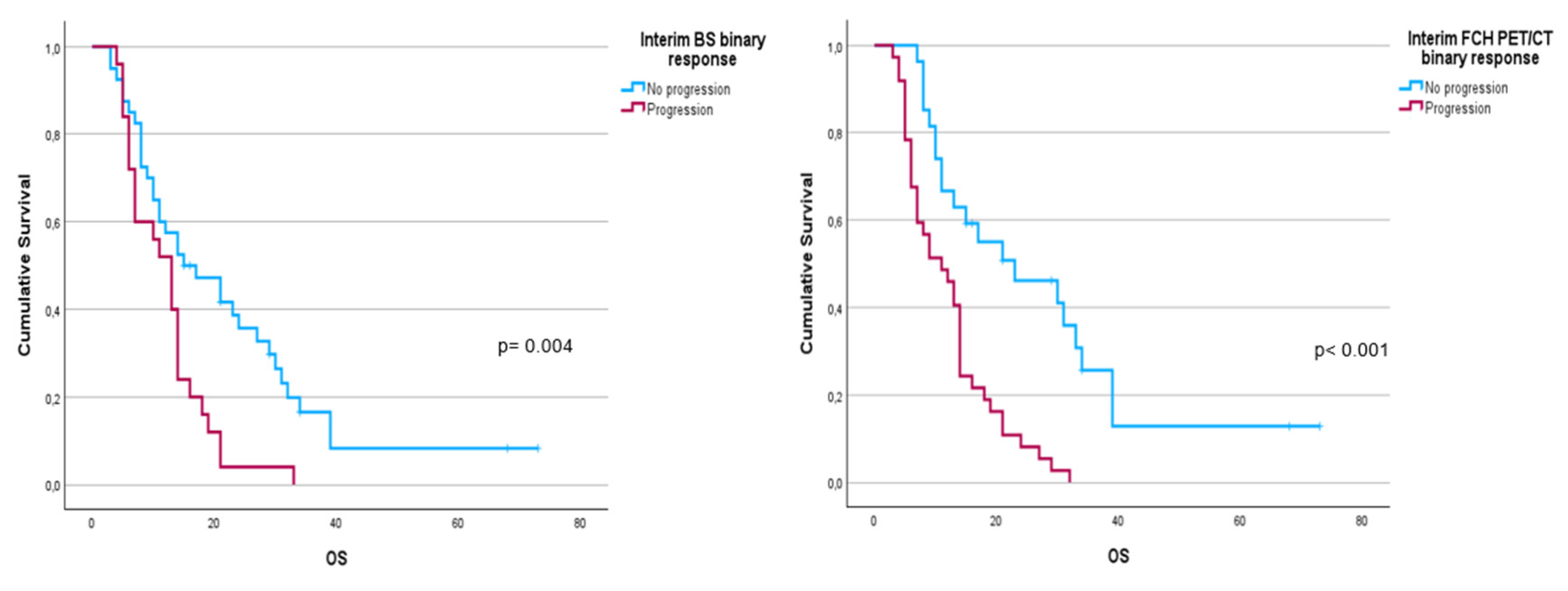
Figure 2). Focusing on treatment response assessment, only interim studies (BS and FCH PET/CT) showed a significant association with OS, with data extracted from FCH PET/CT being slightly more robust compared to BS (p< 0.001 vs p= 0.004, respectively) (
Figure 3). The significant variables in the univariate analysis were included in multivariate analysis for the OS and therapeutic failure variable. In the former, the association of uptake of lesions above the liver (p = 0.011), therapeutic failure (p = 0.001), or pathological baseline AP (p = 0.011) was highlighted as leading to lower OS.
Table 7 details the results of the univariate Cox regression and
Table 8,
Table 9 and
Table 10 the results of the Log-Rank test regarding OS.
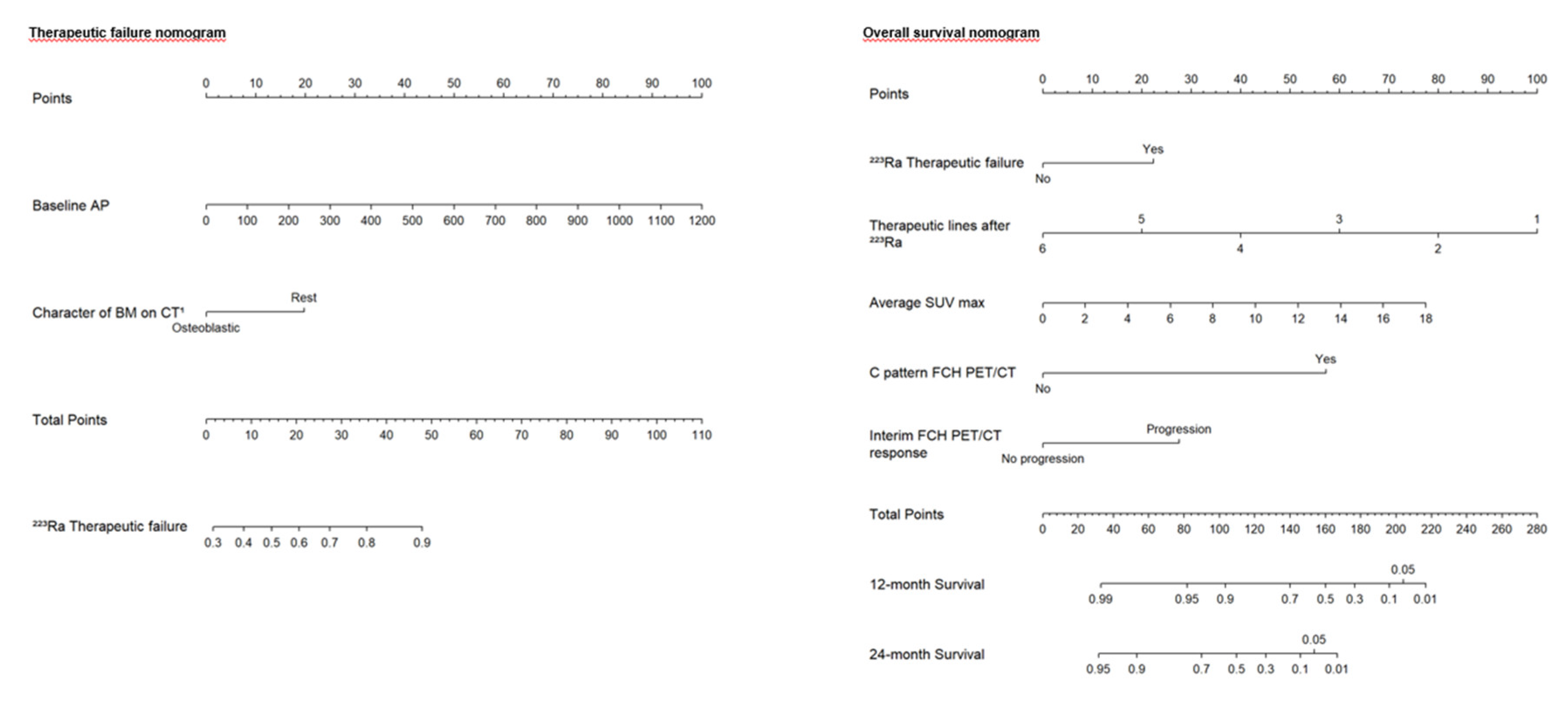
Based on the independent risk factors obtained from multivariate logistic regression analyses, two nomograms were constructed: one to predict the percentage of therapeutic failure and the other to predict the 1- and 2-year survival rates in patients treated with
223Ra. A point scale from 0 to 100 was used to score each variable, and then the sum of all scores was calculated. The risk of therapeutic failure and death can be predicted by observing the total points. In the first nomogram (therapeutic failure), the variable characteristics of bone metastases on the CT were divided into osteoblastic or rest of lesions (lytic or mixed), and the units of measurement for baseline AP were IU/L. In the second nomogram (OS), the variable lines of treatment after
223Ra were divided into 6 to 1 lines and for the mean SUVmax from 0 to 18. In the case of the response variable on the FCH PET/CT, it was divided into progression and non-progression, and for the rest of the variables, no and yes (
Figure 4).
Figure 1.
Kaplan-Meier OS curves of baseline AP levels (upper left panel), LDH levels (upper right panel), and ECOG performance status (lower panel).
Figure 1.
Kaplan-Meier OS curves of baseline AP levels (upper left panel), LDH levels (upper right panel), and ECOG performance status (lower panel).
Figure 2.
Kaplan-Meier OS curves of baseline FCH PET/CT radiomics: high tumor burden (upper left panel), uptake higher than liver for the most hypermetabolic bone metastases (upper right panel), bone marrow involvement (lower left panel) and soft tissue involvement (lower right panel).
Figure 2.
Kaplan-Meier OS curves of baseline FCH PET/CT radiomics: high tumor burden (upper left panel), uptake higher than liver for the most hypermetabolic bone metastases (upper right panel), bone marrow involvement (lower left panel) and soft tissue involvement (lower right panel).
Figure 3.
Kaplan-Meier OS curves of binary response in interim FCH PET/CT scans (left panel) and BS (right panel).
Figure 3.
Kaplan-Meier OS curves of binary response in interim FCH PET/CT scans (left panel) and BS (right panel).
Figure 4.
Designed nomograms to predict therapeutic failure (left panel) and the probability of survival at 12 and 24 months (right panel). The nomograms were developed based on the 100 patients in our population with CRPC-BM who received 223Ra therapy. To obtain the probability of therapeutic failure and survival, the values for each variable of the patients included in each nomogram are marked. Then, a straight vertical line is drawn up to the “Points” line at the top of the nomogram. This determines how many points are attributed to each variable. Once this is done for each variable, the sum of all the points obtained is calculated and added to the “Total Points” line at the bottom of the nomogram. This value is then used to assess the individual probability of predicting the risk of therapeutic failure (left panel) and survival at 12 and 24 months (right panel).
Figure 4.
Designed nomograms to predict therapeutic failure (left panel) and the probability of survival at 12 and 24 months (right panel). The nomograms were developed based on the 100 patients in our population with CRPC-BM who received 223Ra therapy. To obtain the probability of therapeutic failure and survival, the values for each variable of the patients included in each nomogram are marked. Then, a straight vertical line is drawn up to the “Points” line at the top of the nomogram. This determines how many points are attributed to each variable. Once this is done for each variable, the sum of all the points obtained is calculated and added to the “Total Points” line at the bottom of the nomogram. This value is then used to assess the individual probability of predicting the risk of therapeutic failure (left panel) and survival at 12 and 24 months (right panel).
Figure 5.
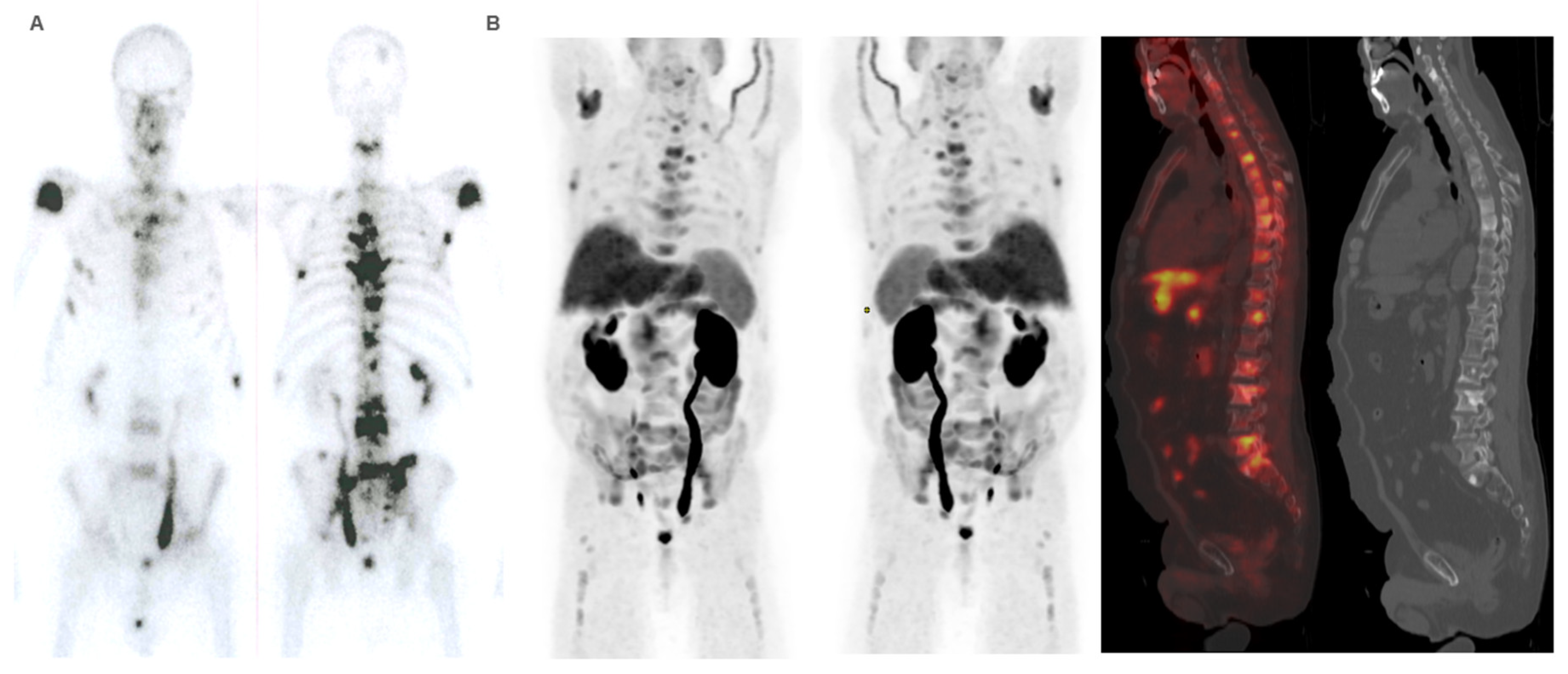
Patient ≠ 1. 71-year-old man, diagnosed with prostate adenocarcinoma Gleason Score 9 (4+5), underwent prostatectomy followed by adjuvant radiotherapy to the surgical bed due to early PSA progression. Three years after diagnosis, bone metastases were detected, prompting the start of systemic treatment lines (Enzalutamide and Docetaxel). 223Ra was administered as third line. At the start of treatment, the patient was in good general condition (ECOG 0), experienced pain, and had baseline PSA levels of 50.7 ng/dL, AP of 377 IU/L, and LDH of 377 IU/L. Baseline BS (A) shows a polymetastatic disease (> 20 lesions) affecting both the axial and extra-axial skeleton. Baseline FCH PET/CT (B) shows the presence of mixed characteristic BM, bone marrow infiltration, uptake of the most hypermetabolic BM higher than liver and soft tissue involvement at the pelvic lymph nodes. The concordance between both studies was moderate, defining FCH PET/CT more BM with respect to BS.
Figure 5.
Patient ≠ 1. 71-year-old man, diagnosed with prostate adenocarcinoma Gleason Score 9 (4+5), underwent prostatectomy followed by adjuvant radiotherapy to the surgical bed due to early PSA progression. Three years after diagnosis, bone metastases were detected, prompting the start of systemic treatment lines (Enzalutamide and Docetaxel). 223Ra was administered as third line. At the start of treatment, the patient was in good general condition (ECOG 0), experienced pain, and had baseline PSA levels of 50.7 ng/dL, AP of 377 IU/L, and LDH of 377 IU/L. Baseline BS (A) shows a polymetastatic disease (> 20 lesions) affecting both the axial and extra-axial skeleton. Baseline FCH PET/CT (B) shows the presence of mixed characteristic BM, bone marrow infiltration, uptake of the most hypermetabolic BM higher than liver and soft tissue involvement at the pelvic lymph nodes. The concordance between both studies was moderate, defining FCH PET/CT more BM with respect to BS.
Figure 6.
Patient ≠ 1. PSA and AP showed a steadily increase after 223Ra initiation. Clinical deterioration was observed after third 223Ra administration. Interim BS (A) and FCH PET/CT (B) show disease progression. Patient died 6 months after starting treatment with 223Ra.
Figure 6.
Patient ≠ 1. PSA and AP showed a steadily increase after 223Ra initiation. Clinical deterioration was observed after third 223Ra administration. Interim BS (A) and FCH PET/CT (B) show disease progression. Patient died 6 months after starting treatment with 223Ra.
Figure 7.
Patient ≠ 2. 88-year-old men diagnosed with metastatic prostate cancer, Gleason Score 7 (4+3), from the onset. He received first-line treatment with Enzalutamide until biochemical and bone progression after 5 years. Treatment with 223Ra was proposed as a second-line. Baseline BS and FCH PET/CT (A) shows oligometastatic disease with only axial involvement and predominantly osteoblastic. At the start of 223Ra treatment, patient was in very good general condition (ECOG 0) with baseline PSA levels of 14.9 ng/mL, AP of 73 IU/L, and LDH of 450 IU/L. The patient completed 6 doses of 223Ra, remaining stable in the interim BS and FCH PET/CT studies (B) but showed bone progression at the end of treatment in both the BS and FCH PET/CT (C). His PSA levels increased during the treatment, while AP levels remained stable. After 223Ra treatment, patient received Docetaxel and experienced a bone event that consisted of a painful metastatic bone at 17 months, treated with palliative vertebral radiotherapy. He ultimately died with an OS of 30 months.
Figure 7.
Patient ≠ 2. 88-year-old men diagnosed with metastatic prostate cancer, Gleason Score 7 (4+3), from the onset. He received first-line treatment with Enzalutamide until biochemical and bone progression after 5 years. Treatment with 223Ra was proposed as a second-line. Baseline BS and FCH PET/CT (A) shows oligometastatic disease with only axial involvement and predominantly osteoblastic. At the start of 223Ra treatment, patient was in very good general condition (ECOG 0) with baseline PSA levels of 14.9 ng/mL, AP of 73 IU/L, and LDH of 450 IU/L. The patient completed 6 doses of 223Ra, remaining stable in the interim BS and FCH PET/CT studies (B) but showed bone progression at the end of treatment in both the BS and FCH PET/CT (C). His PSA levels increased during the treatment, while AP levels remained stable. After 223Ra treatment, patient received Docetaxel and experienced a bone event that consisted of a painful metastatic bone at 17 months, treated with palliative vertebral radiotherapy. He ultimately died with an OS of 30 months.
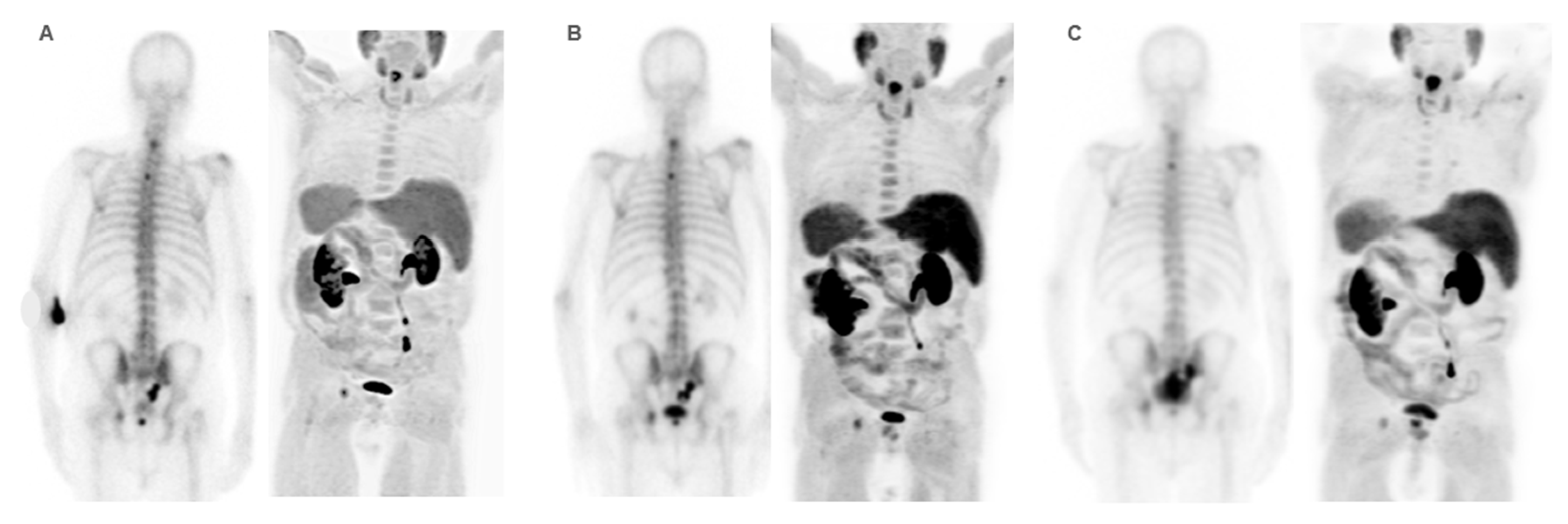
Figure 8.
Patient ≠ 3. 69-year-old man diagnosed with prostate cancer Gleason score 8 (4+4) stage IV T3N1M1 (hepatic, pulmonary, nodal, and bone infiltration). Patient achieved a complete radiologic response after received first-line Docetaxel with and anti-androgen blockade. After progression, Abiraterone-Prednisone was administered with partial biochemical response. 223Ra was administered as third line. Baseline BS and FCH PET/CT (A) shows oligometastatic axial and extra-axial BM on the BS and polymetastatic (6-20 lesions) axial and extra-axial involvement on the FCH PET/CT without soft tissue involvement, with some BM showing lytic characteristics and higher uptake than the liver. Despite this, the concordance between both techniques was good, with metabolic activity predominating over osteogenic. At the start of treatment, the patient was in very good general condition (ECOG 0) with pain and baseline PSA values of 5 ng/dL, AP of 87 IU/L, and LDH of 363 IU/L. He received 6 doses of 223Ra, with PSA values progressing from the start of treatment, although AP and LDH values did not. The interim BS study shows rib deposits interpreted as bone progression while the FCH PET/CT showed stability, explained by the interpretation of rib deposits as probable fractures. However, the end-treatment scans (C) show bone and nodal progression of FCH PET/CT with stability in the BS. Subsequently, treatment with Abiraterone-Prednisone was resumed, followed, shortly after, of Cabazitaxel. Patient died 10 months after 223Ra initiation.
Figure 8.
Patient ≠ 3. 69-year-old man diagnosed with prostate cancer Gleason score 8 (4+4) stage IV T3N1M1 (hepatic, pulmonary, nodal, and bone infiltration). Patient achieved a complete radiologic response after received first-line Docetaxel with and anti-androgen blockade. After progression, Abiraterone-Prednisone was administered with partial biochemical response. 223Ra was administered as third line. Baseline BS and FCH PET/CT (A) shows oligometastatic axial and extra-axial BM on the BS and polymetastatic (6-20 lesions) axial and extra-axial involvement on the FCH PET/CT without soft tissue involvement, with some BM showing lytic characteristics and higher uptake than the liver. Despite this, the concordance between both techniques was good, with metabolic activity predominating over osteogenic. At the start of treatment, the patient was in very good general condition (ECOG 0) with pain and baseline PSA values of 5 ng/dL, AP of 87 IU/L, and LDH of 363 IU/L. He received 6 doses of 223Ra, with PSA values progressing from the start of treatment, although AP and LDH values did not. The interim BS study shows rib deposits interpreted as bone progression while the FCH PET/CT showed stability, explained by the interpretation of rib deposits as probable fractures. However, the end-treatment scans (C) show bone and nodal progression of FCH PET/CT with stability in the BS. Subsequently, treatment with Abiraterone-Prednisone was resumed, followed, shortly after, of Cabazitaxel. Patient died 10 months after 223Ra initiation.
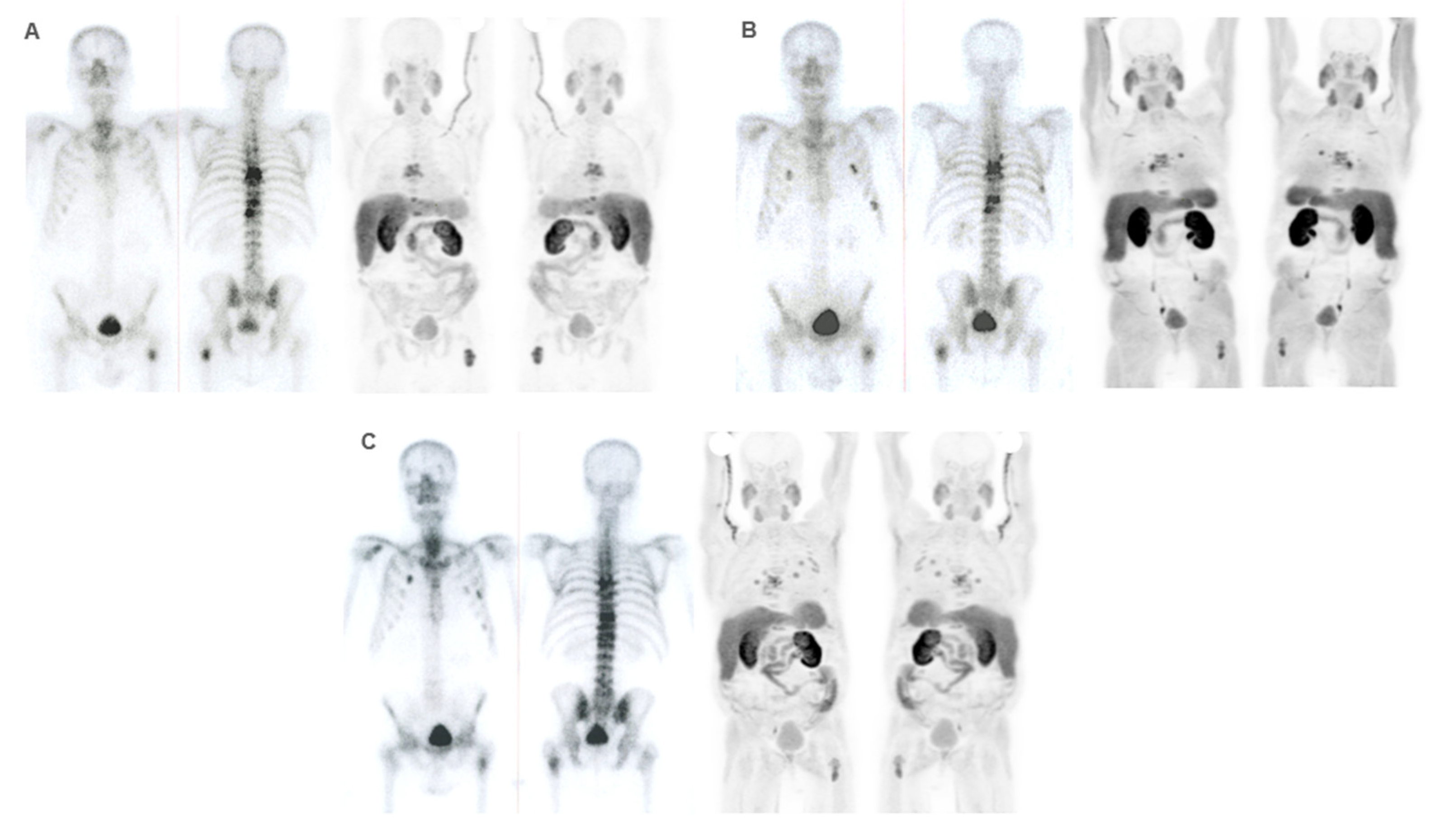
Table 1.
Patient and tumor characteristics.
Table 1.
Patient and tumor characteristics.
| Baseline quantitative variables |
Mean ± IQR |
| Age (years) |
72.72 ± 8.50 |
| Baseline PSA (ng/ml) |
188.53 ± 913.94 |
| Baseline AP (U/l) |
176.78 ± 187.26 |
| Baseline LDH (U/l) |
400.46 ± 192.37 |
| SUVmax |
8.62 ± 5.37 |
| Average SUVmax |
6.82 ± 4.01 |
| Time variables |
Median (Q1 – Q3) |
| Time of evolution of prostate cancer (months) |
62.50 (75.75 – 108.00) |
| Time of evolution of bone metastases (months) |
28.00 (20.00 – 48.00) |
| Time of castration resistance (months) |
21 (12.25–43.00) |
Table 2.
Patients and tumour qualitative and categorical characteristics (n=100).
Table 2.
Patients and tumour qualitative and categorical characteristics (n=100).
| Clinical characteristics |
n and % |
| Gleason |
|
| ≤ 7 |
46 |
| ≥ 8 |
45 |
| n.a. |
9 |
| Baseline AP |
|
| Pathological |
37 |
| Normal |
53 |
| n.a. |
10 |
| Baseline LDH |
|
| Pathological |
18 |
| Normal |
69 |
| n.a. |
13 |
| Bone events before to 223Ra |
|
| Yes |
23 |
| No |
77 |
| Bone events during or after to 223Ra |
|
| Yes |
26 |
| No |
74 |
| Bone protective treatment |
|
| Yes |
90 |
| No |
10 |
| Death (*) |
|
| Yes |
92 |
| No |
8 |
| Progression (*) |
|
| Yes |
97 |
| No |
3 |
| Therapeutic failure |
|
| Yes |
56 |
| No |
44 |
| ECOG performance status |
|
| 0 |
70 |
| 1 |
27 |
| 2 |
3 |
| Enzalutamide before to 223Ra |
|
| Yes |
42 |
| No |
58 |
| Abiraterone before to 223Ra |
|
| Yes |
66 |
| No |
34 |
| ChT (**) |
|
| First line Docetaxel |
71 |
| 2nd line ChT |
21 |
| Xofigo line |
|
| 1 |
3 |
| 2 |
20 |
| 3 |
57 |
| 4 |
13 |
| 5 |
6 |
| 7 |
1 |
| PSA progression during 223Ra |
|
| Yes |
70 |
| No |
27 |
| n.a. |
3 |
| PSA progression during 223Ra (detailed 1) |
|
| First 3 months |
54 |
| 4-6 dose |
16 |
| n.a. |
30 |
| PSA progression during 223Ra (detailed 2) |
|
| First 3 months |
54 |
| Rest |
43 |
| n.a. |
3 |
Table 3.
Results of baseline imaging techniques (n=100).
Table 3.
Results of baseline imaging techniques (n=100).
| Baseline imaging characteristics |
n and % |
| Lesion localization on BS |
|
| Axial |
39 |
| Axial and other |
60 |
| n.a. |
1 |
| Number of BM on BS |
|
| Oligomts ≤5 |
27 |
| Polimts >5 |
72 |
| n.a. |
1 |
| Extent of BM on BS |
|
| 1-5 lesions |
27 |
| 6-20 lesions |
35 |
| >20 lesions |
30 |
| Superscan |
7 |
| n.a. |
1 |
| High tumour burden on BS |
|
| Yes |
61 |
| No |
38 |
| n.a. |
1 |
| Lesion localisation on FCH PET/CT |
|
| Axial |
39 |
| Axial and other |
48 |
| n.a. |
13 |
| Number of BM on FCH PET/CT |
|
| Oligomts ≤5 |
27 |
| Polimts >5 |
60 |
| n.a. |
13 |
| Extent of BM on FCH PET/CT |
|
| 1-5 lesions |
27 |
| 6-20 lesions |
33 |
| >20 lesions |
21 |
| Superscan |
6 |
| n.a. |
13 |
| High tumour burden on FCH PET/CT |
|
| Yes |
48 |
| No |
39 |
| n.a. |
13 |
| BM characteristics on CT (*) |
|
| Osteoblastic |
56 |
| Osteolitic |
12 |
| Mixed |
19 |
| n.a. |
13 |
| Soft tissue involvement on FCH PET/CT |
|
| Yes |
19 |
| No |
68 |
| n.a. |
13 |
| Concordance BS/ FCH PET/CT |
|
| Good |
58 |
| Moderate |
12 |
| Bad |
16 |
| n.a. |
14 |
| A pattern on BS/ FCH PET/CT |
|
| 1 (predominant metabolic activity) |
22 |
| 2 (predominant osteogenic activity) |
54 |
| 3 (similar uptake in both) |
10 |
| n.a. |
14 |
| B pattern (bone marrow involvement on BS) |
|
| Yes |
23 |
| No |
76 |
| n.a. |
1 |
| C pattern (bone marrow involvement on FCH PET/CT) |
|
| Yes |
18 |
| No |
69 |
| n.a. |
13 |
| D pattern (FCH PET/CT uptake) |
|
| 1 (higher than liver) |
45 |
| 2 (lower than liver) |
42 |
| n.a. |
13 |
Table 4.
Response results of interim and end-treatment bone scan and FCH PET/CT.
Table 4.
Response results of interim and end-treatment bone scan and FCH PET/CT.
| |
Interim FCH PET/CT |
| |
|
PR |
S |
P |
Interim BS
(n=63)
|
PR |
1 |
0 |
1 |
| S |
7 |
16 |
14 |
| P |
1 |
2 |
21 |
| |
End-treatment FCH PET/CT |
| |
|
CR |
PR |
E |
P |
| End-treatment BS (n=38) |
PR |
1 |
0 |
2 |
1 |
| E |
1 |
1 |
14 |
12 |
| P |
0 |
0 |
1 |
5 |
Table 5.
Association of clinical and imaging variables with therapeutic failure.
Table 5.
Association of clinical and imaging variables with therapeutic failure.
| Variables |
χ² |
p value |
| Clinical and disease characteristics before 223Ra |
| Bone events before to 223Ra (categorical) |
1.030 |
0.310 |
| Gleason |
3.438 |
0.931 |
| Gleason (categorical) |
0.566 |
0.452 |
| ECOG |
6.188 |
0.038 |
| Prostatectomy (radical prostate cancer treatment) |
4.386 |
0.036 |
| RT (radical prostate cancer treatment) |
0.230 |
0.632 |
| Enzalutamide (before to 223Ra) |
0.038 |
0.845 |
| Abiraterone (before to 223Ra) |
0.167 |
0.683 |
| Docetaxel (before to 223Ra) |
0.114 |
0.736 |
| 2nd line ChT (before to 223Ra) |
0.376 |
0.540 |
| Xofigo line |
3.797 |
0.620 |
| PSA progression during 223Ra |
19.378 |
< 0.001 |
| PSA progression during 223Ra detailed 1 (i) |
5.145 |
0.023 |
| PSA progression during 223Ra detailed 2 (ii) |
18.564 |
< 0.001 |
| Baseline AP |
4.735 |
0.030 |
| Baseline LDH |
4.247 |
0.039 |
| Clinical and disease characteristics after 223Ra |
| Bone events during or after to 223Ra (categorical) |
0.410 |
0.840 |
| Bone protective treatment (categorical) |
5.213 |
0.040 |
| Death (during follow up) |
11.335 |
< 0.001 |
| Progression (during follow up) |
2.43 |
0.253 |
| PFS (≤6 months vs >6 months) |
23.522 |
< 0.001 |
| OS (≤12 months vs >12 months) |
18.446 |
< 0.001 |
| Baseline imaging techniques results |
| BM location on BS |
2.304 |
0.129 |
| Number of BM on BS |
3.300 |
0.069 |
| Extent of BM on baseline BS |
4.637 |
0.205 |
| High tumor burden of BM on BS |
1.674 |
0.196 |
| BM location on FCH PET/CT |
6.584 |
0.010 |
| Number of BM on FCH PET/CT |
10.321 |
0.001 |
| Extent of BM on baseline FCH PET/CT |
14.998 |
0.001 |
| BM characteristics on CT portion of FCH PET/CT |
7.299 |
0.025 |
| BM characteristics on CT portion FCH PET/CT (categorical) (iii) |
7.016 |
0.008 |
| Soft tissue involvement |
4.141 |
0.042 |
| Concordance BS/ FCH PET/CT (iv) |
1.729 |
0.421 |
| A pattern (BS vs FCH PET/CT) |
3.972 |
0.146 |
| B pattern (BS) |
0.343 |
0.558 |
| C pattern (FCH PET/CT) |
3.434 |
0.064 |
| D pattern (FCH PET/CT) |
8.318 |
0.004 |
Table 6.
Association of clinical and imaging variables with skeletal events during or after 223Ra treatment.
Table 6.
Association of clinical and imaging variables with skeletal events during or after 223Ra treatment.
| Variables |
χ² |
p value |
|
| Patient and clinical characteristics before 223Ra |
|
| |
Bone events before to 223Ra (yes/no) |
1.198 |
0.274 |
| |
Bone events before to 223Ra (type) |
8.473 |
0.029 |
| |
Gleason |
7.450 |
0.395 |
| |
ECOG |
2.422 |
0.375 |
| |
223Ra line |
3.624 |
0.651 |
| |
PSA progression during 223Ra |
0.498 |
0.480 |
| |
PSA progression during 223Ra detailed 1 |
0.048 |
1.000 |
| |
PSA progression during 223Ra detailed 2 |
0.256 |
0.613 |
| |
Baseline AP |
1.455 |
0.228 |
| |
Baseline LDH |
0.164 |
0.759 |
| Clinical and disease characteristics after 223Ra |
|
| |
Aditional treatments (categorical) |
2.157 |
0.225 |
| |
Additional treatments (lines number) |
18.544 |
0.001 |
| |
Additional treatment (Abiraterone) |
3.835 |
0.050 |
| |
Additional treatment (Enzalutamide) |
1.127 |
0.288 |
| |
Additional treatment (ChT) |
5.963 |
0.058 |
| |
Bone protective treatment (categorical) |
0.092 |
1.000 |
| |
Bone protective treatment (descriptive) |
2.816 |
0.250 |
| |
Bone protective treatment (moment) |
1.117 |
0.771 |
| |
Death during follow-up |
0.851 |
0.449 |
| |
Progression during follow-up |
0.086 |
1.000 |
| |
PFS (≤6 months vs >6 months) |
0.837 |
0.360 |
| |
OS (≤12 months vs >12 months) |
7.787 |
0.005 |
| Baseline imaging techniques |
|
| |
BM location on BS |
0.013 |
0.910 |
| |
Number of BM on BS |
0.217 |
0.641 |
| |
Extent of BM on baseline BS |
0.316 |
0.972 |
| |
BM high tumour burden on BS |
0.000 |
0.992 |
| |
BM on FCH PET/CT |
3.253 |
0.071 |
| |
Number of BM on FCH PET/CT |
0.205 |
0.651 |
| |
Extent of BM on FCH PET/CT |
1.270 |
0.752 |
| |
BM high tumour burden on FCH PET/CT |
5.256 |
0.022 |
| |
BM characteristics on CT portion of FCH PET/CT (descriptive) |
4.036 |
0.134 |
| |
BM characteristics on CT portion of FCH PET/CT (categorical) |
0.368 |
0.618 |
| |
Soft tissue involvement on FCH PET/CT (categorical) |
1.417 |
0.234 |
| |
Soft tissue involvement of FCH PET/CT (descriptive) |
1.273 |
0.718 |
| |
Concordance BS/ FCH PET/CT 1 |
0.842 |
0.622 |
| |
Concordance BS/ FCH PET/CT 2 |
0.031 |
1.000 |
| |
A pattern (BS vs FCH PET/CT) |
1.674 |
0.457 |
| |
B pattern (BS) |
1.218 |
0.270 |
| |
C pattern (FCH PET/CT) |
5.088 |
0.033 |
| |
D pattern (FCH PET/CT) |
3.594 |
0.058 |
| Interim imaging techniques |
|
| |
Interim BS response |
0.105 |
0.904 |
| |
Interim BS response (categorical) |
0.101 |
0.751 |
| |
Interim FCH PET/CT response |
0.115 |
1.000 |
| |
Interim FCH PET/CT response (categorical) |
0.028 |
0.867 |
| |
Progression localisation |
2.856 |
0.463 |
| |
Concordance BS/ FCH PET/CT 1 |
0.523 |
0.768 |
| |
Concordance BS/ FCH PET/CT 2 |
0.651 |
0.522 |
| End-treatment imaging techniques |
|
| |
End-treatment BS response |
3.288 |
0.213 |
| |
End-treatment BS response |
1.081 |
0.427 |
| |
End-treatment FCH PET/CT response |
3.934 |
0.287 |
| |
End-treatment FCH PET/CT response |
2.438 |
0.155 |
| |
Progression location |
2.526 |
0.906 |
| |
Concordance BS/ FCH PET/CT 1 |
0.891 |
0.646 |
| |
Concordance BS/ FCH PET/CT 2 |
0.107 |
1.000 |
Table 7.
Results of the Cox regression index of clinical and imaging quantitative variables with OS.
Table 7.
Results of the Cox regression index of clinical and imaging quantitative variables with OS.
| Variables |
HR |
95% CI of HR |
p value |
| Doses of 223Ra (number) |
0.699 |
0.606-0.807 |
< 0.001 |
| Patient age |
1.005 |
0.981-1.030 |
0.666 |
| Time from diagnosis of pCa to 223Ra |
0.122 |
0.995-1.003 |
0.727 |
| Time of evolution of BM to 223Ra |
0.999 |
0.993-1.007 |
0.925 |
| Time of CRPC to 223Ra |
1.003 |
0.994-1.012 |
0.479 |
| Baseline PSA |
1.000 |
1.0001-1.0004 |
0.028 |
| Baseline AP |
1.002 |
1.001-1.004 |
< 0.001 |
| Baseline LDH |
1.000 |
0.999-1.001 |
0.511 |
| SUVmax |
1.049 |
1.006-1.092 |
0.023 |
| Average SUVmax |
1.070 |
1.012-1.132 |
0.017 |
Table 8.
Long rank (Mantel-Cox) results of association of clinical and tumor characteristics with overall survival.
Table 8.
Long rank (Mantel-Cox) results of association of clinical and tumor characteristics with overall survival.
| Variables |
Median (mo) |
χ² |
p value |
| Bone events before to 223Ra |
Yes |
16 |
0.022 |
0.883 |
| No |
13 |
| Bone events during or after to 223Ra |
Yes |
19 |
4.998 |
0.025 |
| No |
12 |
| Any treatments after 223Ra |
Yes |
14 |
25.699 |
< 0.001 |
| No |
5 |
| Abiraterone after 223Ra |
Yes |
23 |
6.614 |
0.010 |
| No |
13 |
| Enzalutamide after 223Ra |
Yes |
23 |
11.312 |
<0.001 |
| No |
13 |
| Bone protective treatment |
Yes |
14 |
0.113 |
0.737 |
| No |
14 |
| Bone protective treatment (Denosumab) |
Zolendronic |
16 |
4.228 |
0.040 |
| Both |
8 |
0.329 |
0.566 |
| Bone protective treatment (Zolendronic) |
Denosumab |
13 |
4.228 |
0.040 |
| Both |
8 |
1.506 |
0.220 |
| Therapeutic failure |
Yes |
8 |
29.1 |
< 0.001 |
| No |
21 |
| Gleason (categorical) |
≥ 8 |
14 |
0.036 |
0.850 |
| Rest |
14 |
| ECOG 0 |
1 |
9 |
4.253 |
0.039 |
| 2 |
5 |
23.337 |
<0.001 |
| ECOG 1 |
2 |
5 |
3.874 |
0.049 |
| Enzalutamide before to 223Ra |
Yes |
10 |
1.081 |
0.299 |
| No |
16 |
| Abiraterone before to 223Ra |
Yes |
13 |
2.663 |
0.103 |
| No |
18 |
| 2nd line ChT before to 223Ra |
Yes |
9 |
0.119 |
0.730 |
| No |
14 |
| 1st Docetaxel before to 223Ra |
Yes |
14 |
0.958 |
0.328 |
| No |
14 |
|
223Ra line 1 |
2 |
15 |
4.168 |
0.041 |
| 3 |
11 |
0.851 |
0.356 |
| 4 |
14 |
0.243 |
0.622 |
| 5 |
7 |
5.279 |
0.022 |
| 7 |
8 |
3.000 |
0.083 |
|
223Ra line 2 |
3 |
11 |
0.051 |
0.821 |
| 4 |
14 |
0.563 |
0.453 |
| 5 |
7 |
5.171 |
0.023 |
| 7 |
8 |
3.725 |
0.054 |
|
223Ra line 3 |
4 |
14 |
0.766 |
0.381 |
| 5 |
7 |
1.715 |
0.190 |
| 7 |
8 |
0.551 |
0.458 |
|
223Ra line 4 |
5 |
7 |
3.208 |
0.073 |
| 7 |
8 |
1.548 |
0.213 |
|
223Ra line 5 |
7 |
8 |
0.141 |
0.707 |
|
223Ra line (categorical 1) |
First 3 |
14 |
0.015 |
0.902 |
| 4rd and beyond |
11 |
|
223Ra line (categorical 2) |
First 2 |
19 |
0.360 |
0.549 |
| 3rd and beyond |
11 |
| PSA progression during 223Ra |
Yes |
12 |
8.508 |
0.004 |
| No |
19 |
| PSA progression detailed 1 |
A |
12 |
0.122 |
0.726 |
| B |
11 |
| PSA progression detailed 2 |
A |
12 |
5.693 |
0.017 |
| C |
16 |
| Baseline AP |
Pathological |
8 |
12.581 |
< 0.001 |
| Normal |
15 |
| Baseline LDH |
Pathological |
8 |
4.526 |
0.033* |
| Normal |
14 |
Table 9.
Long rank (Mantel-Cox) results of association of baseline imaging variables with overall survival.
Table 9.
Long rank (Mantel-Cox) results of association of baseline imaging variables with overall survival.
| Baseline imaging variables |
Median (mo) |
χ² |
p value |
| BM location on BS |
Axial |
15 |
1.414 |
0.234 |
| Axial and other |
11 |
| BM number on BS |
Oligometastatic |
17 |
2.534 |
0.111 |
| Polimetastasic |
11 |
| Number of BM on BS (1-5 lesions) |
6-20 lesions |
13 |
0.715 |
0.398 |
| > 20 lesions |
8 |
3.454 |
0.063 |
| Superscan |
5 |
2.531 |
0.112 |
| Number of BM on BS (6-20 lesions) |
> 20 lesions |
8 |
1.289 |
0.256 |
| Superscan |
5 |
0.703 |
0.402 |
| Number of BM on BS (> 20 lesions) |
Superscan |
5 |
0.117 |
0.732 |
| High burden of BM on BS |
Yes |
11 |
1.691 |
0.193 |
| No |
16 |
| BM location on FCH PET/CT |
Axial |
21 |
14.769 |
< 0.001 |
| Axial and other |
8 |
| Number of BM on FCH PET/CT |
Oligometastatic |
18 |
4.814 |
0.028 |
| Polimetastasic |
10 |
| Number of BM on FCH PET/CT (1-5) |
6-20 |
13 |
1.215 |
0.270 |
| > 20 |
8 |
7.259 |
0.007 |
| Superscan |
5 |
16.751 |
<0.001 |
| Number of BM on FCH PET/CT (6 -20) |
> 20 |
8 |
3.577 |
0.059 |
| Superscan |
5 |
9.494 |
0.002 |
| Number of BM on FCH PET/CT (> 20) |
Superscan |
5 |
2.081 |
0.149 |
| FCH PET/CT high tumour burden of BM |
Yes |
9 |
9.016 |
0.003 |
| No |
18 |
| BM characteristics (i) on CT (osteoblastic) |
Osteolytic |
13 |
0.196 |
0.658 |
| Mixed |
9 |
3.944 |
0.047 |
| BM characteristics on CT (osteolytic) |
Mixed |
9 |
0.953 |
0.329 |
| BM characteristics on CT (categorical) |
Osteoblastic |
14 |
2.620 |
0.106 |
| Rest |
12 |
| Soft tissue involvement on FCH PET/CT |
Yes |
7 |
3.911 |
0.048 |
| No |
14 |
| BS/PET concordance (good) |
Moderate |
8 |
4.988 |
0.026 |
| Bad |
13 |
0.896 |
0.344 |
| BS/PET concordance (moderate) |
Bad |
13 |
5.211 |
0.022 |
| A pattern |
Osteogenic activity predominance |
15 |
3.718 |
0.054 |
| Similar uptake |
7 |
0.193 |
0.660 |
| A pattern |
Similar uptake |
7 |
0.879 |
0.348 |
| B pattern |
Yes |
7 |
2.626 |
0.105 |
| No |
14 |
| C pattern |
Yes |
5 |
27.266 |
< 0.001 |
| No |
14 |
| D pattern |
Higher than liver |
9 |
13.170 |
< 0.001 |
| Lower than liver |
18 |
Table 10.
Long rank (Mantel-Cox) results of association of treatment response imaging variables with overall survival.
Table 10.
Long rank (Mantel-Cox) results of association of treatment response imaging variables with overall survival.
| Variables |
Median (mo) |
χ² |
p value |
| Interim imaging techniques |
| BS interim response (partial response, 12 mo) |
Stability |
17 |
0.018 |
0.892 |
| Progression |
13 |
0.836 |
0.361 |
| BS interim response (stability) |
Progression |
13 |
8.723 |
0.003 |
| BS interim response categorical |
No progression |
15 |
8.371 |
0.004 |
| Progression |
13 |
| Interim PET response (partial response, 21 mo) |
Stability |
23 |
0.011 |
0.917 |
| Progression |
11 |
7.044 |
0.008 |
| Interim PET response (stability) |
Progression |
11 |
13.759 |
<0.001 |
| Interim PET response categorical |
No progression |
23 |
17.026 |
< 0.001 |
| Progression |
11 |
| Interim PET progression (bone, 8 mo) |
Adenopathic |
14 |
1.543 |
0.214 |
| Bone and adenopathic |
14 |
0.420 |
0.517 |
| Bone and visceral |
9 |
0.066 |
0.797 |
| Interim PET progression (adenopathic) |
Bone and adenopathic |
14 |
0.347 |
0.556 |
| Bone and visceral |
9 |
0.656 |
0.418 |
| Interim PET progression (bone and adenopathic) |
Bone and visceral |
9 |
1.101 |
0.294 |
| End-treatment images techniques |
| End-treatment BS response (partial response 23 mo) |
Stability |
21 |
0.005 |
0.946 |
| Progression |
16 |
2.598 |
0.107 |
| End-treatment BS response (stability) |
Progression |
16 |
2.944 |
0.086 |
| End-treatment BS response categorical |
No progression |
23 |
3.470 |
0.063 |
| Progression |
16 |
| End-treatment PET response (complete response, 24 mo) |
Partial response |
34 |
0.059 |
0.808 |
| Stability |
17 |
0.128 |
0.720 |
| Progression |
19 |
0.553 |
0.457 |
| End-treatment PET response (partial response) |
Stability |
17 |
0.089 |
0.765 |
| Progression |
19 |
0.373 |
0.542 |
| End-treatment PET response (stability) |
Progression |
19 |
0.264 |
0.607 |
| End-treatment PET response categorical |
No progression |
23 |
1.327 |
0.249 |
| Progression |
18 |
| End-treatment PET progression (bone, 15 mo) |
Adenopathic |
18 |
0.022 |
0.883 |
| Visceral |
10 |
2.762 |
0.097 |
| Bone and adenopathic |
19 |
1.185 |
0.276 |
| Bone and visceral |
47 |
2.166 |
0.141 |
| End-treatment PET progression (adenopathic) |
Visceral |
10 |
3.000 |
0.083 |
| Bone and adenopathic |
19 |
1.182 |
0.277 |
| Bone and visceral |
47 |
1.779 |
0.182 |
| End-treatment PET progression (visceral) |
Bone and adenopathic |
19 |
2.000 |
0.157 |
| Bone and visceral |
47 |
1.000 |
0.317 |
| End-treatment PET progression (bone and adenopathic) |
Bone and visceral |
47 |
1.471 |
0.225 |