Submitted:
07 July 2024
Posted:
08 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
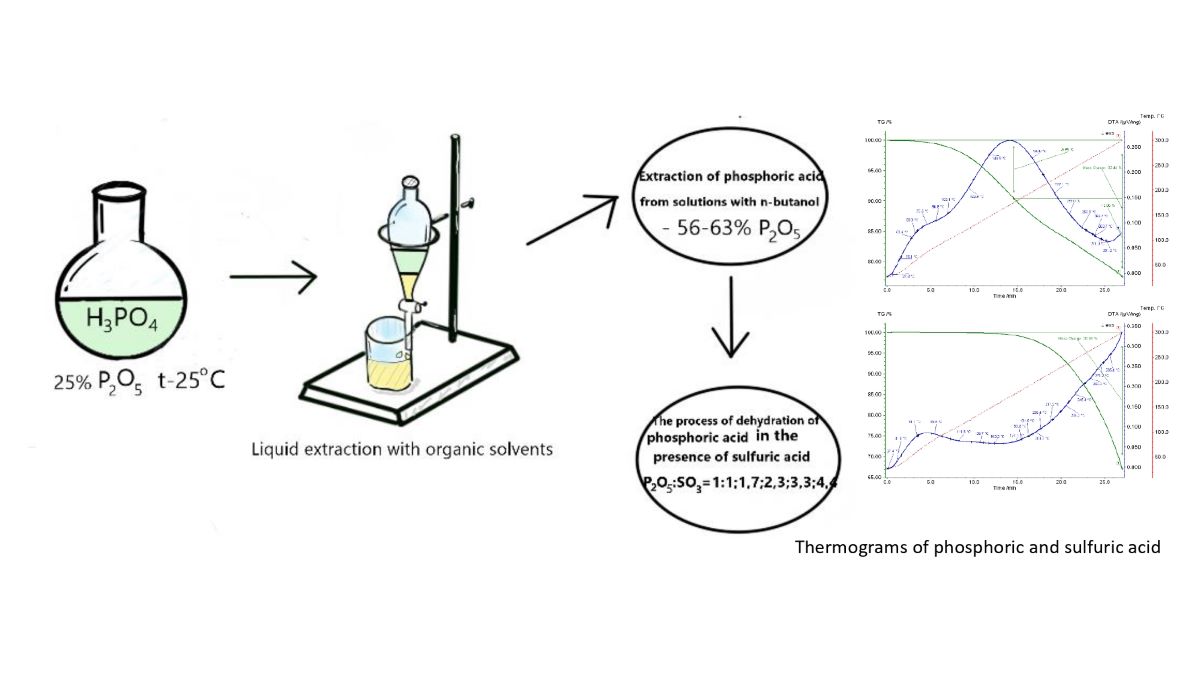
2. Research Objects and Methods
3. Results and Discussions
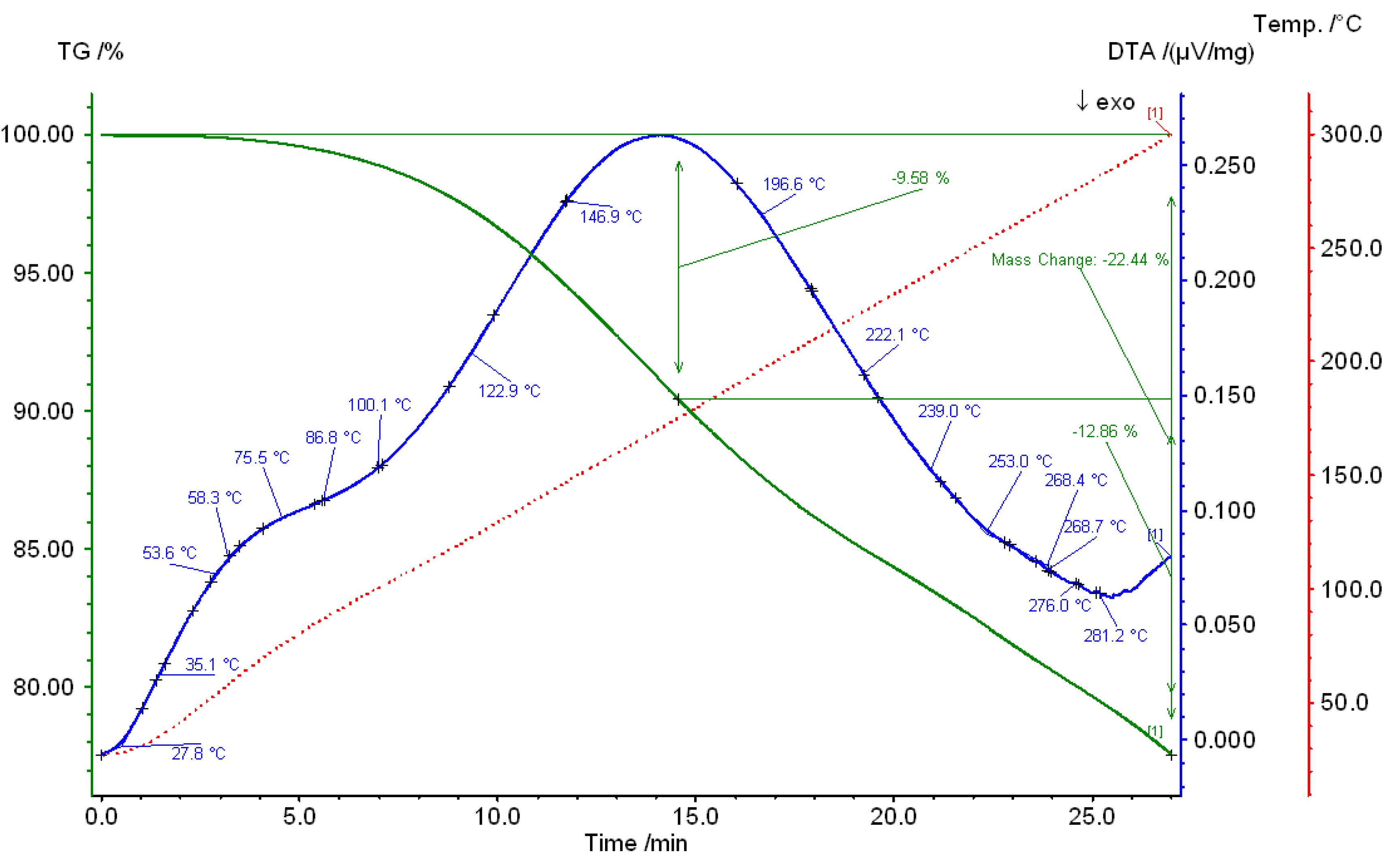
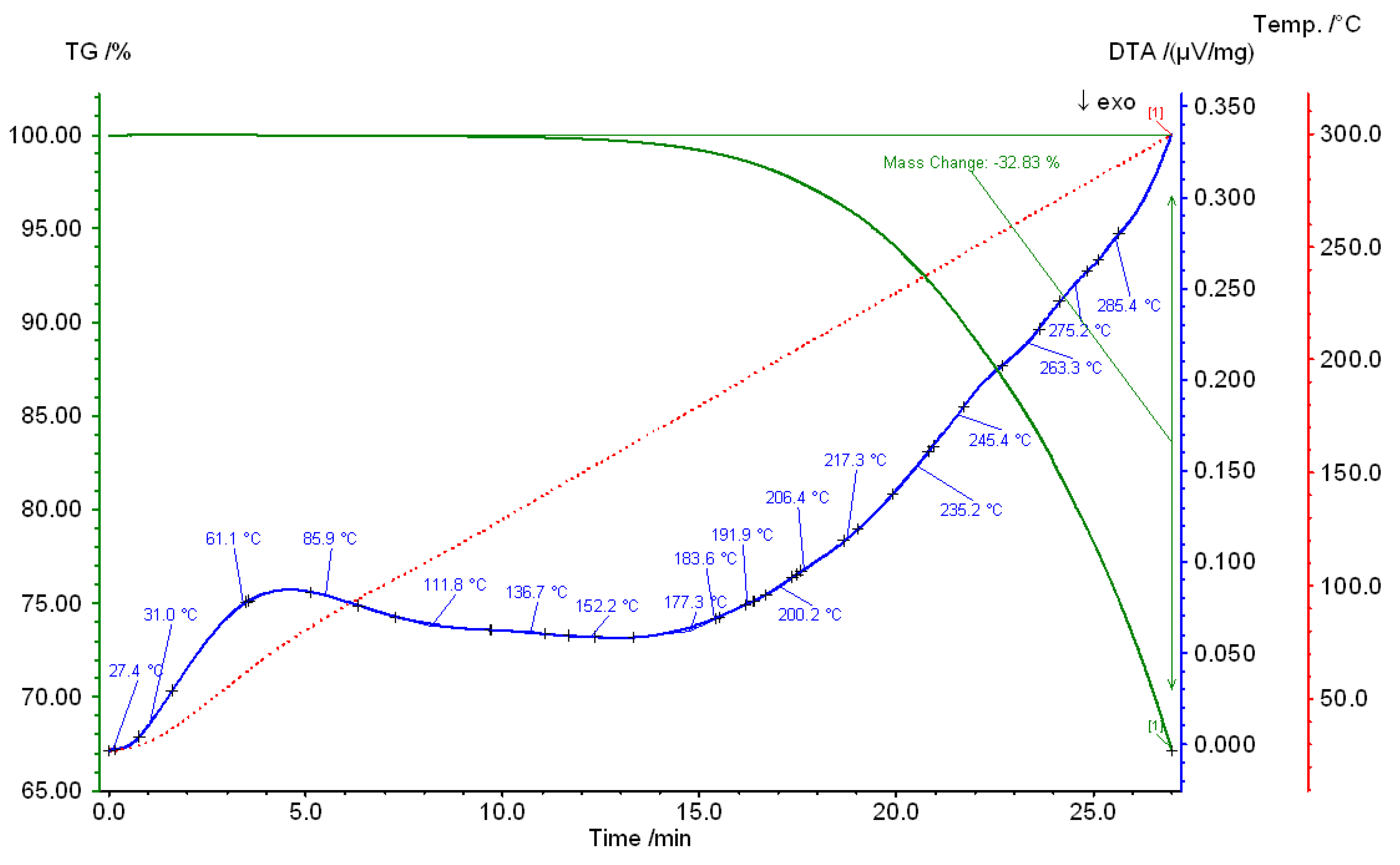
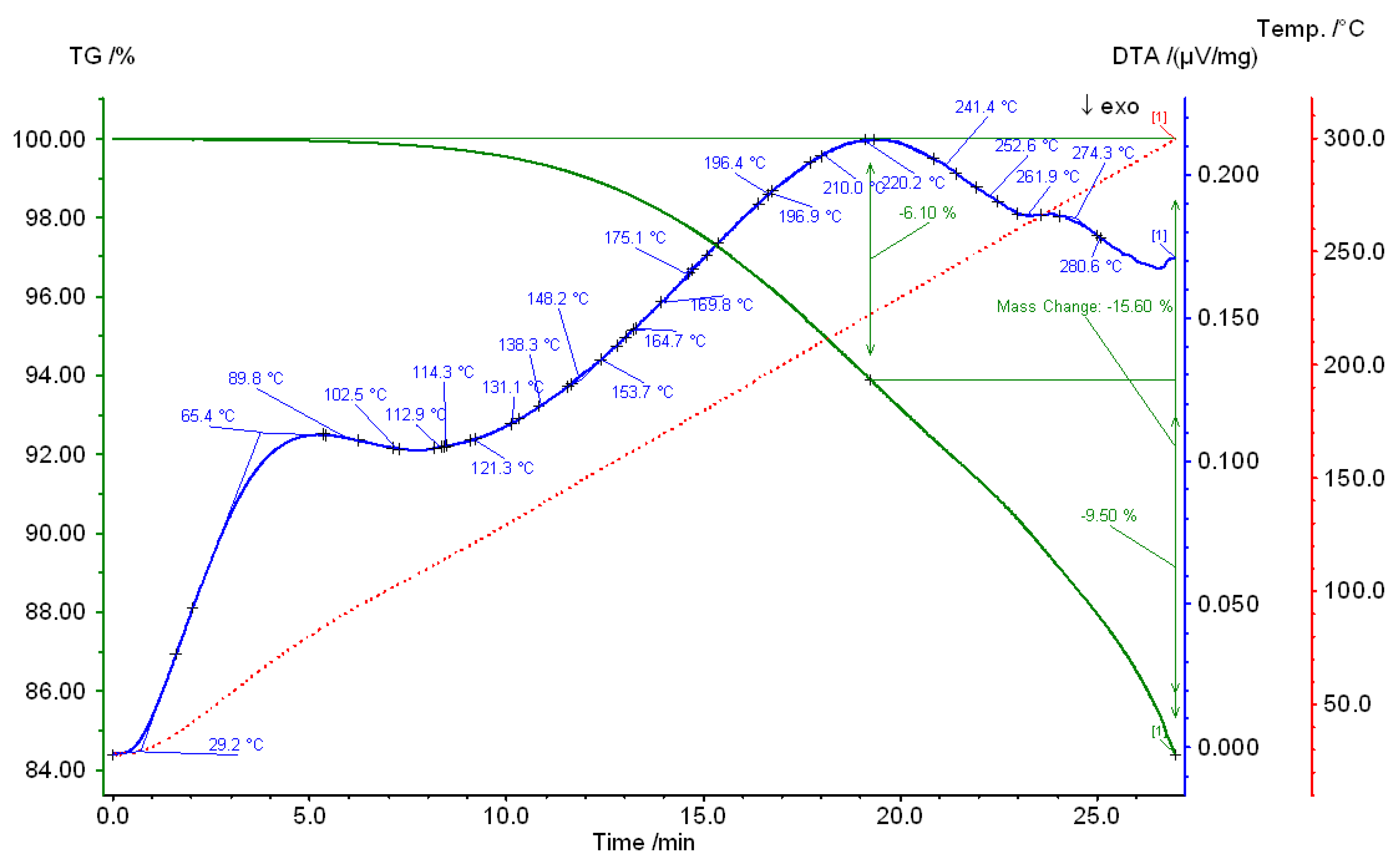
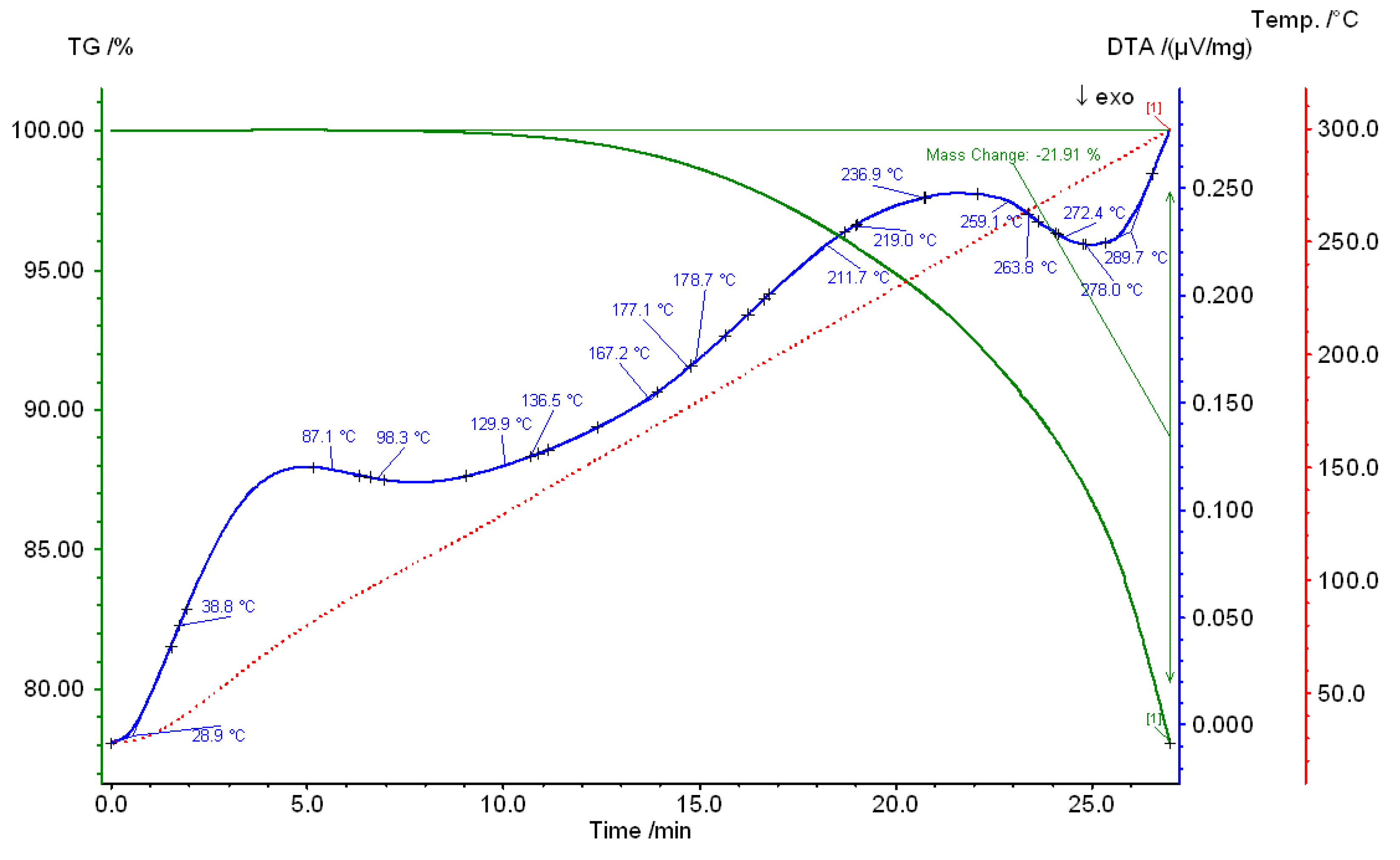
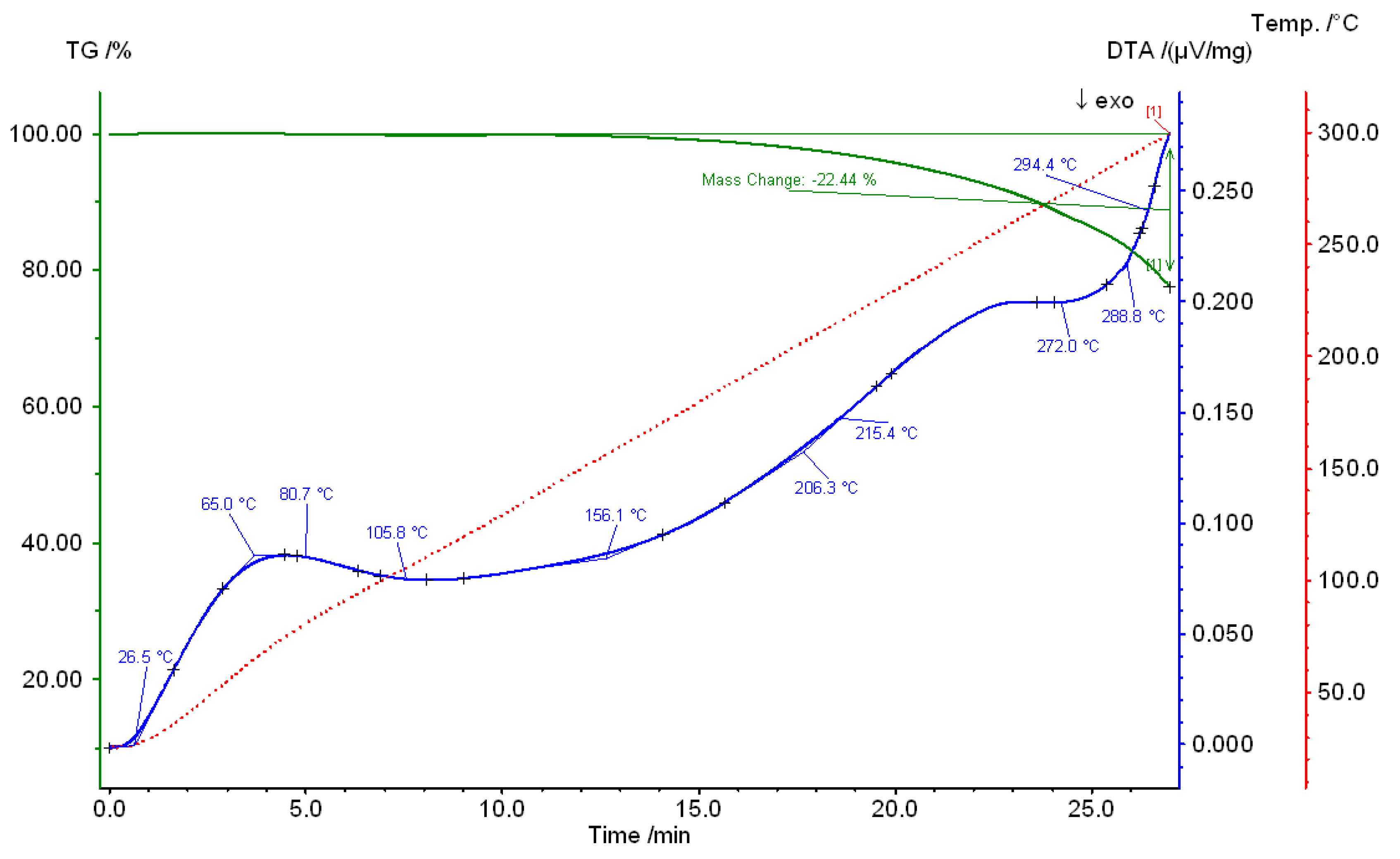
4. Conclusions
Author Contributions
Conflict of interests
References
- U. Ryszko, P. Rusek, D. Kołodyńska, Quality of Phosphate Rocks from Various Deposits Used in Wet Phosphoric Acid and P-Fertilizer Production, Materials, 16 (2023) 2. 793. [CrossRef]
- S.V. Vakal, E. Karpovich, H, Turgumbaeva, Investigation of the process of concentration of phosphoric acid obtained from Karatau phosphorite, Chemical Journal of Kazakhstan, 1 (2013) 104-109. [in Russian].
- B. M. Rakishev, The role and prospects of mineral resources in the development of the economy of Kazakhstan, News of the National Academy of Sciences of the Republic of Kazakhstan, Series of Geology and Technical Sciences, 2 (2016) 29-39. [in Russian].
- B. Numonov et al. Low-waste process of complex fertilizer based on sulphuric acid processing thermic calcinated phosphorite concentrate. Journal of Chemical Technology & Metallurgy, 4 (2020) 55. 4.
- D. Guelfi, A.P.P.Nunes, L.F.Sarkis, D.P. Oliveira, Innovative Phosphate Fertilizer Technologies to Improve Phosphorus Use Efficiency in Agriculture, Sustainability, 14 (2022) 14266. [CrossRef]
- M.A. de Boer, L. Wolzak, J.C. Slootweg, Phosphorus: Reserves, Production, and Applications. In: Ohtake, H., Tsuneda, S. (eds) Phosphorus Recovery and Recycling. Springer, Singapore, (2019). [CrossRef]
- S. Matta, K. Stephan, J. Stephan, R. Lteif, C. Goutaudier, J. Saab, Phosphoric acid production by attacking phosphate rock with recycled hexafluosilicic acid, International Journal of Mineral Processing, 161 (2017) 21-27, ISSN 0301-7516, . [CrossRef]
- Ibtissem Boumnijel, Hedi Ben Amor, Cheker Chtara, Effect of calcinated and activated perlite on improving efficiency of dihydrate process for phosphoric acid, International Journal of Mineral Processing, 125 (2013) 112-117. [CrossRef]
- M. Schorr et al. Phosphate ore processing for phosphoric acid production: classical and novel technology, Mineral Processing and Extractive Metallurgy, 119 (2010) 3, 125-129. [CrossRef]
- A. Mizane, A. Boumerah, N. Dadda, R. Rehamnia, S.Belhait, Obtaining the partially acidulated phosphate rocks by means of intermediate-grade phosphate and diluted phosphoric acid: Influence of some parameters, Polish Journal of Chemical Technology, 18 (2016) 39-43. [CrossRef]
- N.S. Awwad, Y.A. El-Nadi, M.M. Hamed, Successive processes for purification and extraction of phosphoric acid produced by wet process, Chemical Engineering and Processing: Process Intensification, 74 (2013) 69-74. [CrossRef]
- G. Kodirova, I. Shamshidinov, B. Sultonov, R. Najmiddinov, B. Mamurov, Investigation of the Process of Purification of Wet-Process Phosphoric Acid and Production of Concentrated Phosphoric Fertilizers Based on it. Chemical Science International Journal, 30(1) (2021) 1-10. [CrossRef]
- N.I. Khurramov, T.I. Nurmurodov, A.U. Erkaev, Investigation of the process of extraction phosphoric acid production from washed dried phosphate rock, Universum: technical sciences: electron. nauchn. zhurn. 2 (2021) 83. [in Russian]. [CrossRef]
- E.D. Pluzhnikova, R.R. Yakubova, M.M. Eskendirova, Study of sulfuric acid decomposition of phosphorite of Zhanatas deposit, Bulletin of Science of South Kazakhstan, 2 (2020) 134-138. [in Russian].
- M. Daryani, N. Jodeiri, E. Fatehifar, Shahbazi, J. Optimization of operating conditions in purification of wet process phosphoric acid in a liquid-liquid extraction column, Chemical Engineering Communications, 209(8) (2021). 1082–1095. [CrossRef]
- B. Wang, Q. Zhou, C. Chen, H. Liu, L. Yang, Separation of phosphoric acid and magnesium from wet process phosphoric acid by solvent extraction. Canadian Metallurgical Quarterly, 62(4) (2022) 791–802. [CrossRef]
- S. Zhang, Y. Chen, T. Zhang, L. Lv, D. Zheng, B. Zhong, S. Tang, Separation of H3PO4 from HCl-wet-processing phosphate rocks leach liquor by TBP, Extraction equilibria and mechanism study, Separation and Purification Technology, 249 (2020) 117156, . [CrossRef]
- L. Xinxin, F. Wu, G. Qu, C. Jin, Y, Liu, L. Kuang, H. Li, X. Chen, Z. Wang, Y. Cheng, Application prospect of advanced oxidation technology in wet process phosphoric acid production, Journal of Environmental Chemical Engineering, 10 (2022) 6, 108868, . [CrossRef]
- Y. Jin et al., Extraction Kinetics of Phosphoric Acid from the Phosphoric Acid Calcium Chloride Solution by Tri-n-butyl Phosphate, Industrial & Engineering Chemistry Research, 54, 1 (2015) 108-116. [CrossRef]
- U.K. Alimov, A.M. Reimov, S.S. Namazov, et al. The insoluble part of phosphorus fertilizers, obtained by processing of phosphorites of central Kyzylkum with partially ammoniated extraction phosphoric acid, Russ J. Appl. Chem, 83, (2010). 545–552 . [CrossRef]
- M. Lassis, A. Mizane, N. Dadda, R. Rehamnia, Dissolution of Djebel Onk phosphate ore using sulfuric acid, Environmental Nanotechnology, Monitoring & Management, 4 (2015) 12-16, . [CrossRef]
- R. Gilmour, Phosphoric Acid. Purification, Uses, Technology and Econimics, CRC Press, USA. (2013). [CrossRef]
- A.S. Egorkin et al. Mathematical modeling of the half-hydrate stage of the dihydrate-semihydrate process of extraction phosphoric acid production from phosphate rock, Fundamental Research, 6 (2013) 33-37. [in Russian].
- Samir, Abu-Eishah, M. Nizar Abu-Jabal, Parametric study on the production of phosphoric acid by the dihydrate process, Chemical Engineering Journal, 81 (2001) 1–3, 231-250. [CrossRef]
- S. P. Kochetkov, N. N. Smirnov, A. P. Ilyin, V. M. Lembrikov, S.V. Khromov, Methods for activating the processes of dehydration and defluorination of extraction phosphoric acid. News of higher educational institutions, Chemistry and Chemical Technology, 50 (5) (2007) 41-47. [in Russian].
- R. Kijkowska, D. Pawlowska-Kozinska, Z. Kowalski, M. Jodko, Z. Wzorek, Wet-process phosphoric acid obtained from Kola apatite. Purification from sulphates, fluorine, and metals, Separation and Purification Technology, 28, 3 (2002) 197-205, . [CrossRef]
- H. Li, W. Ge, J. Zhang, R. M. Kasomo, J. Leng, X. Weng, Q. Chen, Q. Gao, S. Song, L. Xiao, C. Tian, Control foaming performance of phosphate rocks used for wet-process of phosphoric acid production by phosphoric acid, Hydrometallurgy, 195 (2020) 105364, . [CrossRef]
- Device and process used for restoring fluorine from smoke after phosphorus absorption by hydration in burning process in furnace for producing phosphoric acid. МПК B01D 53/68 №201514455.5 Hou Yonghe (CN), Wei Shifa (CN), Wei Chenjuan (CN)19.12.2017.
- Magda, A. et al., Liquid-liquid extraction technique for purification of Egyptian WET process phosphoric acid, Periodica Polytechnica Chemical Engineering, 54, 2 (2010.0), 57–62. [CrossRef]
- M.I. Amin, M.M. Ali, H.M. Kamal, A.M. Youssef, M.A. Akl, Recovery of high grade phosphoric acid from wet process acid by solvent extraction with aliphatic alcohols, Hydrometallurgy, 105, 1–2, (2010) 115-119, . [CrossRef]
- M. Chen, J. Li, Y. Jin, J. Luo, X. Zhu, D. Yu, Efficient solvent extraction of phosphoric acid with dibutyl sulfoxide, J. Chem. Technol. Biotechnol, 93 (2018) 467-475. [CrossRef]
- H. Ghanadzadeh, A. Ghanadzadeh, Z. Aghajani, S. Abbasnejad, S. Shekarsaraee, (Liquid+liquid) equilibria in ternary aqueous mixtures of phosphoric acid with organic solvents at T=298.2K, The Journal of Chemical Thermodynamics, 42, 6 (2010), 695-699, . [CrossRef]
- M.S. Mokhort et al. Investigation of the peculiarities of decofluorination of extraction phosphoric acid. Neftegazokhimiya, Proceedings of the V International Scientific and Technical Forum on Chemical Technologies and Oil and Gas Processing, Minsk, BGTU, (2022) 64-68. [in Russian].
- 34. M. Basha, Chromatography, In: Analytical Techniques in Biochemistry, Springer Protocols Handbooks. Humana, New York, NY. (2022). [CrossRef]
- C. Schick, D. Lexa, L. Leibowitz, Differential scanning calorimetry and differential thermal analysis, Characterization of materials. John Wiley & Sons Inc, New York, (2012) 483-495. [CrossRef]
- А. Cheremysinova, I. Sknar, Y. Kozlov, O. Sverdlikovska, O. Sigunov, Study of thermal dehydration of sodium orthophosphate monosubstituted. Sigunov, Study of thermal dehydration of sodium orthophosphate monosubstituted. East European Journal of Advanced Technology, 3 (6) (2017) 60-66. [in Russian]. [CrossRef]
- A.N. Strashko Thermal analysis: methodological instructions for laboratory works on the course "Physico-chemical methods of analysis" for students of IV year, studying in the direction 240501 "Chemical technology of materials of modern energy", Tomsk Polytechnic University, (2014) 16. [in Russian].
| Raw material – Karatau phosphorite | Content, mas. % | |||||||
| Р2О5 | К2О | СаО | MgO | R2O3 | F | SO3 | insoluble residue | |
| 26,81 | 41,68 | 1,96 | 0,79 | 17,05 | ||||
| Phosphoric acid | 25 | 20,58 | - | 2,87 | 1,17 | 1,51 | 3,64 | - |
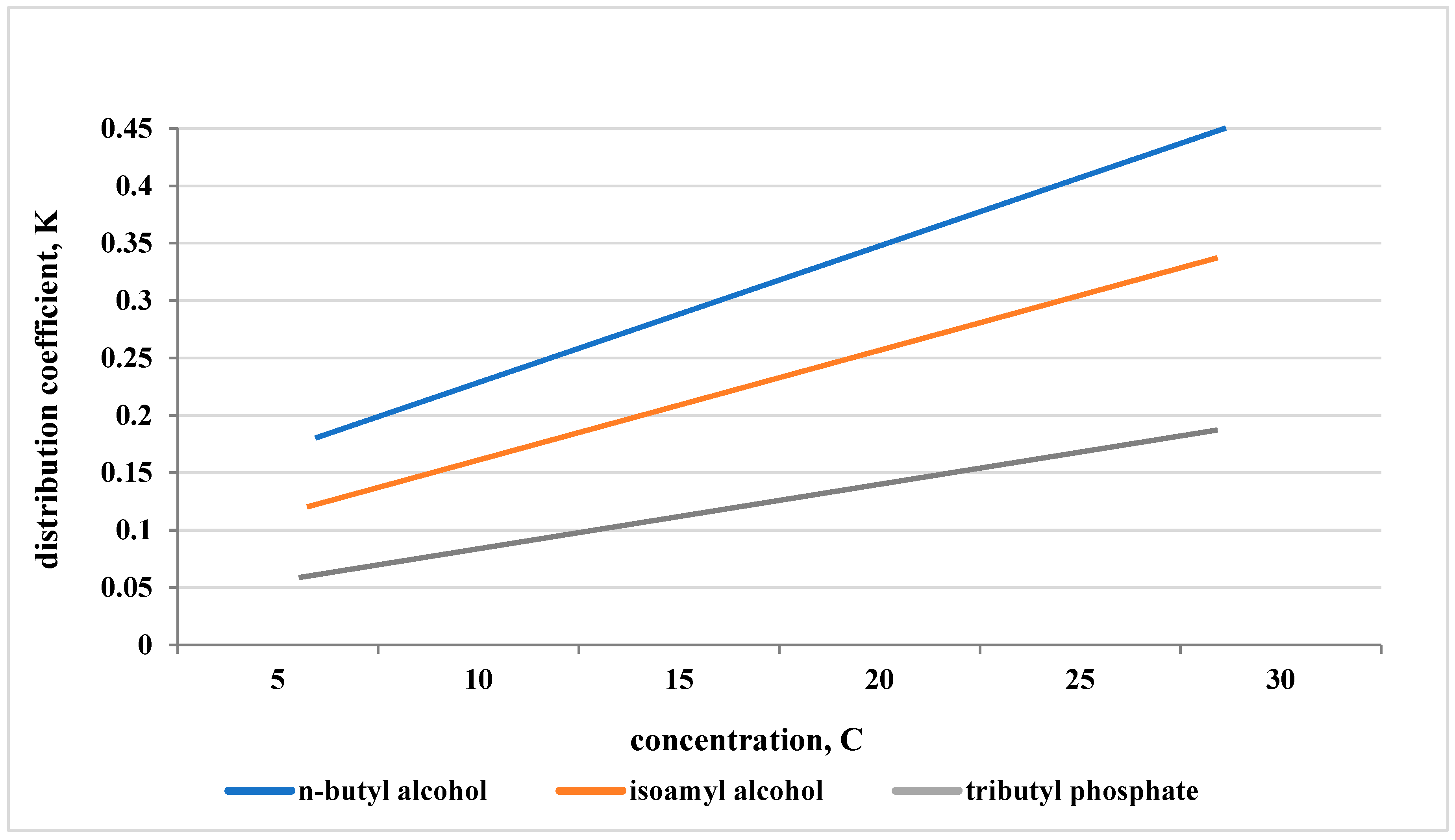
| MgO content in the initial solution, mas. % | n-Butyl alcohol | Isoamyl alcohol | Tributyl phosphate |
||||||
| Сorg. | Сaque. | Кd | Сorg. | Сaque. | Кd | Сorg. | Сaque. | Кd | |
| % P2О5 |
% P2О5 |
% P2О5 |
% P2О5 |
% P2О5 |
% P2О5 |
||||
| MgO in the form of MgSO4 | |||||||||
| 0,63 | 7,43 | 22,23 | 0,334 | 4,07 | 23,72 | 0,170 | 6,45 | 21,73 | 0,297 |
| 1,05 | 8,67 | 21,03 | 0,412 | 4,09 | 23,60 | 0,170 | 6,57 | 21,48 | 0,304 |
| 1,63 | 8,59 | 20,41 | 0,421 | 4,37 | 23,41 | 0,190 | 6,75 | 21,16 | 0,319 |
| 2,10 | 8,73 | 22,16 | 0,394 | 4,68 | 23,45 | 0,201 | 7,05 | 21,19 | 0,331 |
| 2,64 | 9,11 | 21,60 | 0,422 | 5,00 | 23,54 | 0,209 | 7,36 | 21,24 | 0,347 |
| MgO in the form of Mg(Н2РО4)2 | |||||||||
| 0,56 | 8,79 | 23,87 | 0,370 | 4,15 | 26,09 | 0,154 | 6,66 | 23,28 | 0,286 |
| 1,02 | 7,47 | 24,84 | 0,301 | 3,62 | 26,13 | 0,135 | 6,67 | 23,28 | 0,287 |
| 1,48 | 6,58 | 26,19 | 0,247 | 3,14 | 26,19 | 0,119 | 5,83 | 23,65 | 0,248 |
| 2,04 | 5,57 | 25,02 | 0,221 | 2,67 | 25,94 | 0,105 | 5,37 | 24,44 | 0,217 |
| 2,44 | 4,55 | 25,21 | 0,184 | 2,19 | 26,15 | 0,084 | 4,81 | 24,79 | 0,195 |
| Known concentration of SO3, % |
Equilibrium concentration of phosphoric acid, mas. % P2О5 | Кd | |
| Сorg. | Сaque. | ||
| n-Butyl alcohol | |||
| 0,14 | 8,42 | 22,14 | 0,379 |
| 2,01 | 9,47 | 21,47 | 0,442 |
| 3,57 | 10,54 | 20,91 | 0,501 |
| 4,77 | 10,77 | 20,28 | 0,534 |
| 6,15 | 11,82 | 19,97 | 0,590 |
| Isoamyl alcohol | |||
| 0,13 | 4,78 | 23,14 | 0,204 |
| 2,02 | 5,82 | 22,98 | 0,251 |
| 3,57 | 6,85 | 22,65 | 0,301 |
| 4,75 | 7,37 | 21,89 | 0,335 |
| 6,12 | 8,19 | 21,60 | 0,378 |
| Tributyl phosphate | |||
| 0,13 | 6,98 | 21,10 | 0,330 |
| 2,02 | 7,90 | 2,92 | 0,377 |
| 3,58 | 8,28 | 20,55 | 0,402 |
| 4,77 | 8,49 | 19,95 | 0,425 |
| 6,15 | 8,91 | 20,02 | 0,443 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




