Submitted:
11 June 2024
Posted:
13 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
2.1 Tumor Cell Diffusion
- is the diffusion coefficient,
- is the proliferation rate,
- is the carrying capacity.
2.2. Angiogenesis and Nutrient Availability
- represents the nutrient concentration,
- is the nutrient diffusion coefficient,
- is the nutrient consumption rate by tumor cells,
- is the tumor cell density,
- is the angiogenesis rate,
- is the maximum nutrient concentration.
2.3. Molecular Signaling Pathways
- EGFR, PI3K, and MET represent the concentrations of the respective signaling molecules,
- are the activation rates,
- is the tumor cell density,
- Apoptosis Resistance is the resistance to apoptosis,
- is the apoptosis resistance factor,
- Migration Rate is the migration rate of tumor cells,
- is the migration rate factor.
2.4. Blood-Brain Barrier Interactions
- represents the BBB integrity,
- is the BBB diffusion coefficient,
- is the formation rate,
- is the degradation rate by tumor cells,
- is the tumor cell density.
2.5. Numerical Simulation
2.6. Parameter Estimation and Sensitivity Analysis
2.7. Model Validation and Prediction
3. Results
3.1. Graphs Explanation
- Tumor Cell Density: The first graph (Figure 1) depicts the spatial distribution of tumor cell density at the end of the simulation (t = 120 days). The color map represents the density of tumor cells, with warmer colors indicating higher densities. The plot clearly shows the growth and spread of the tumor from the initial starting point at the center of the domain (50, 50). The tumor exhibits a compact, dense core surrounded by a diffuse rim of infiltrating cells. This pattern is consistent with the invasive nature of glioblastoma, where tumor cells migrate into the surrounding brain tissue heterogeneously.
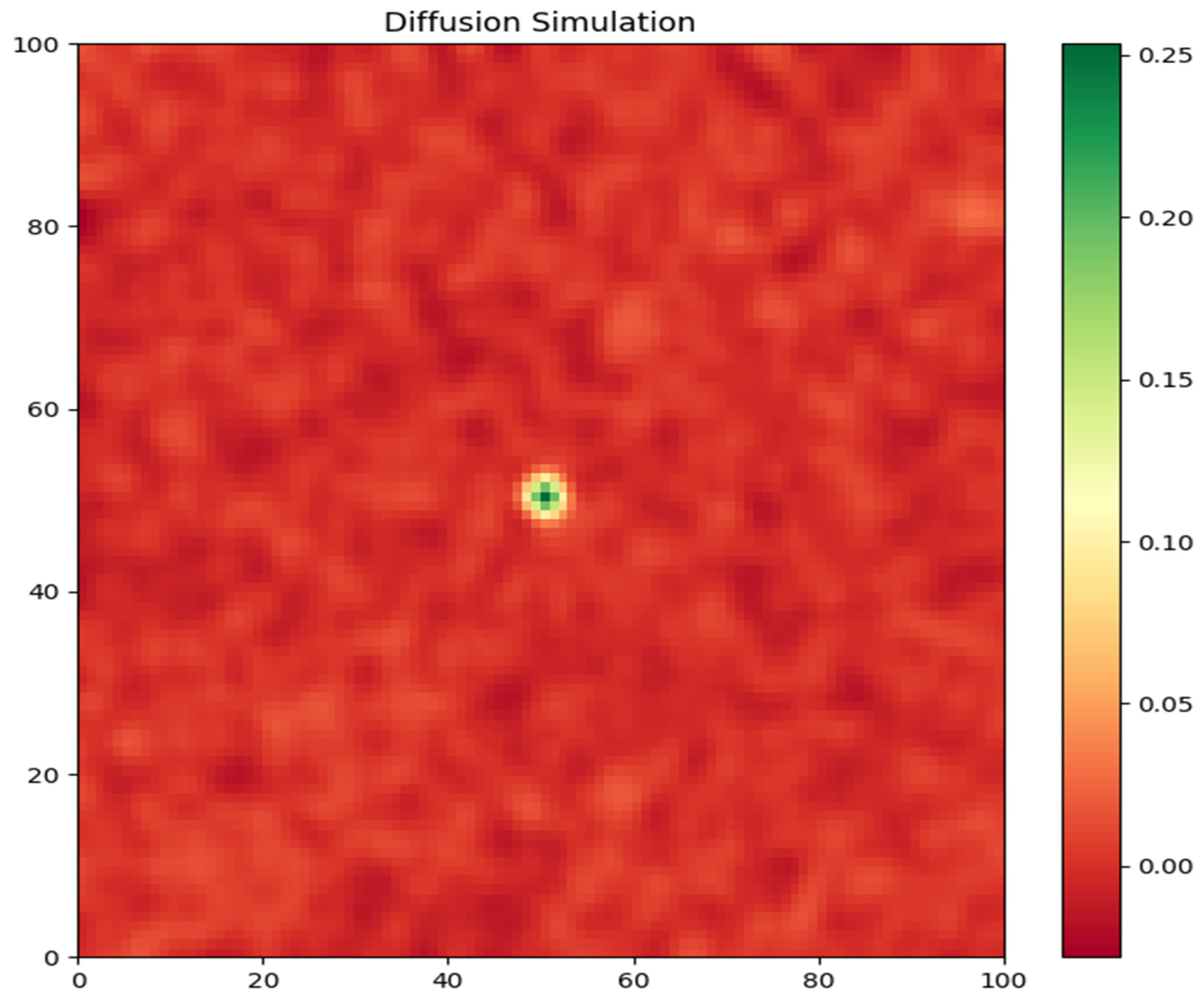
- Nutrient Concentration: The second graph (Figure 2) illustrates the spatial distribution of nutrient concentration at the end of the simulation. The color map represents the nutrient levels, with warmer colors indicating higher concentrations. The plot reveals a heterogeneous distribution of nutrients within the tumor region. The nutrients are more abundant in the peripheral areas, where the tumor cells are less dense, and are depleted in the tumor core due to the high consumption rate by the tightly packed tumor cells. This nutrient gradient is a key factor driving tumor cell migration and invasion.
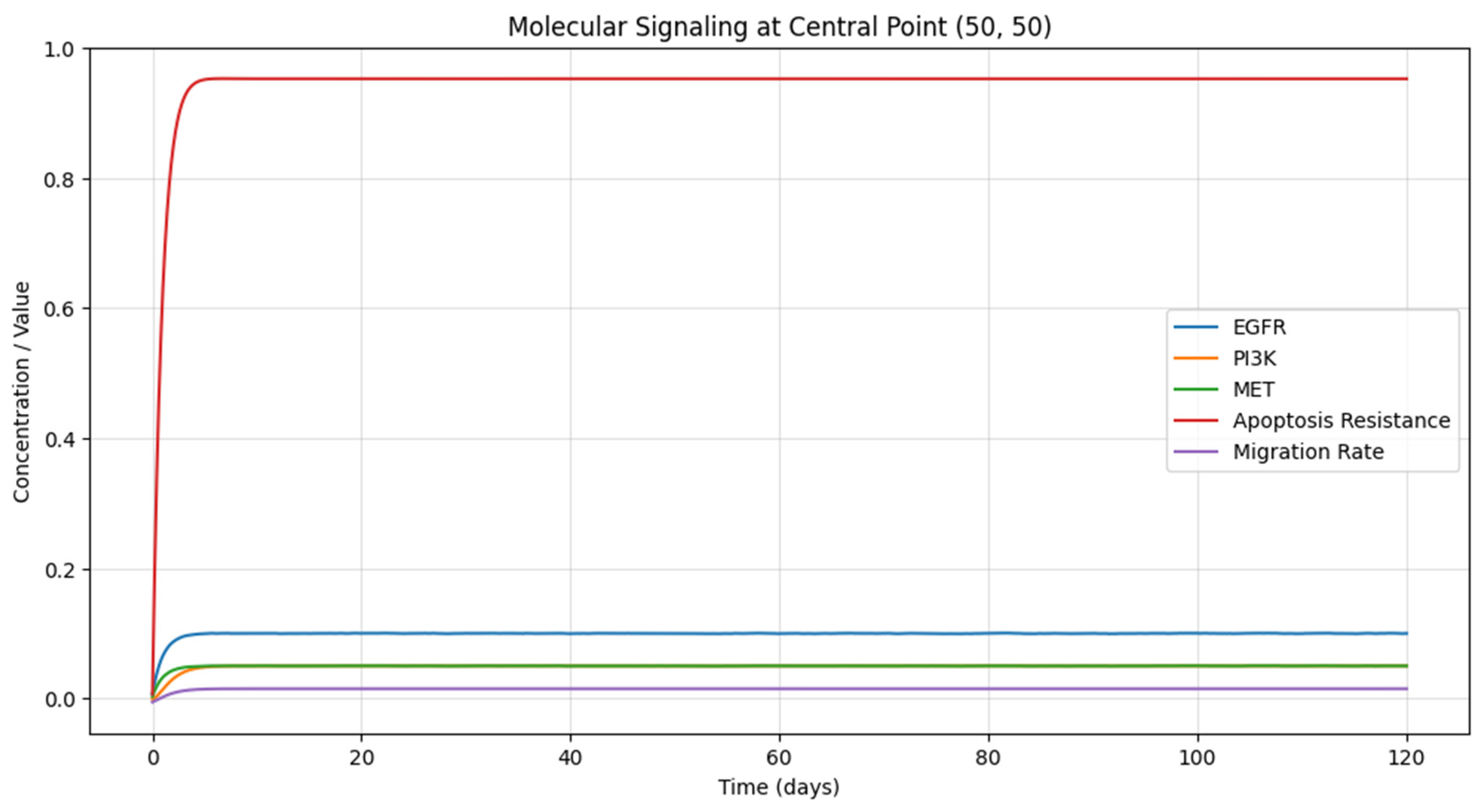
- Molecular Signaling Pathways: The third graph (Figure 3) presents the temporal evolution of the concentrations of key signaling molecules (EGFR, PI3K, and MET) at a specific location within the tumor (50, 50). The plot shows the relative levels of these molecules over time, with each line representing a different signaling pathway. The concentrations of EGFR and MET increase rapidly in the early stages of tumor growth, indicating their activation by the increasing tumor cell density. The PI3K concentration follows a similar trend, albeit with a slight delay, as it is activated by EGFR. These signaling pathways play crucial roles in regulating tumor cell survival, proliferation, and migration (RM, Montgomery et al., 2015). The graph also displays the temporal evolution of cellular processes, specifically apoptosis resistance and migration rate, at the same location within the tumor (50, 50). The plot shows how these processes change over time in response to the underlying signaling pathways. Apoptosis resistance increases as the PI3K pathway becomes more active, allowing tumor cells to evade programmed cell death. The migration rate also rises as the EGFR and MET pathways are activated, promoting tumor cell infiltration into the surrounding tissue.
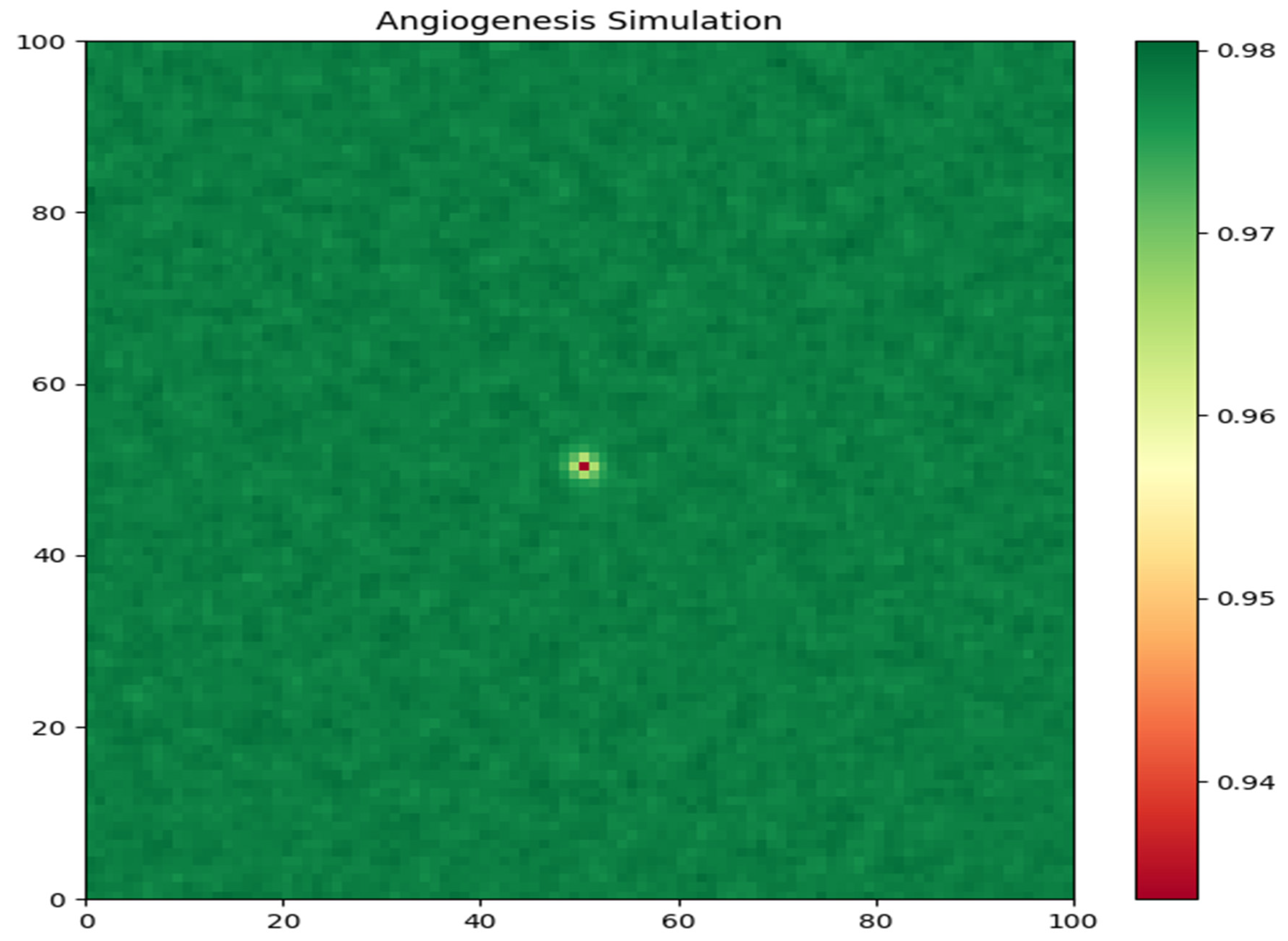
- Blood-Brain Barrier Integrity: The fourth graph (Figure 4) depicts the spatial distribution of blood-brain barrier (BBB) integrity at the end of the simulation. The color map represents the level of BBB integrity, with warmer colors indicating a more intact barrier. The plot reveals a heterogeneous pattern of BBB breakdown, with the most significant disruption occurring in the regions of high tumor cell density. The degradation of the BBB is mediated by the tumor cells, which secrete factors that compromise the barrier’s integrity. The loss of BBB integrity facilitates the invasion of tumor cells into the surrounding brain tissue and enhances the delivery of nutrients to the tumor.
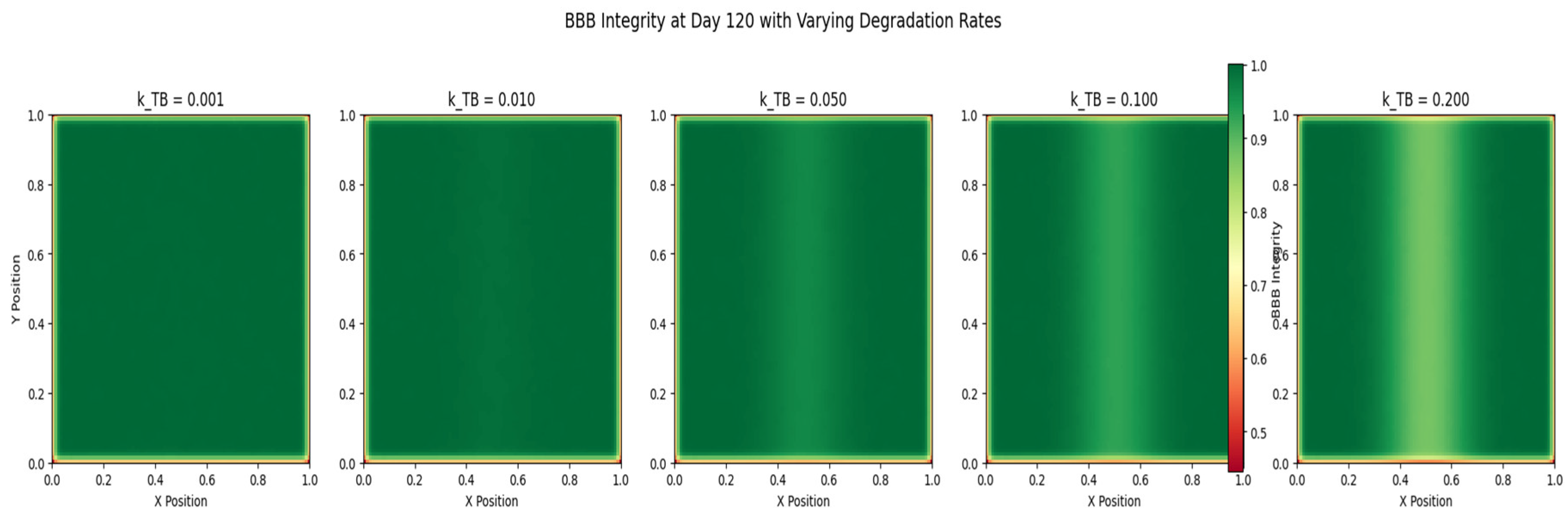
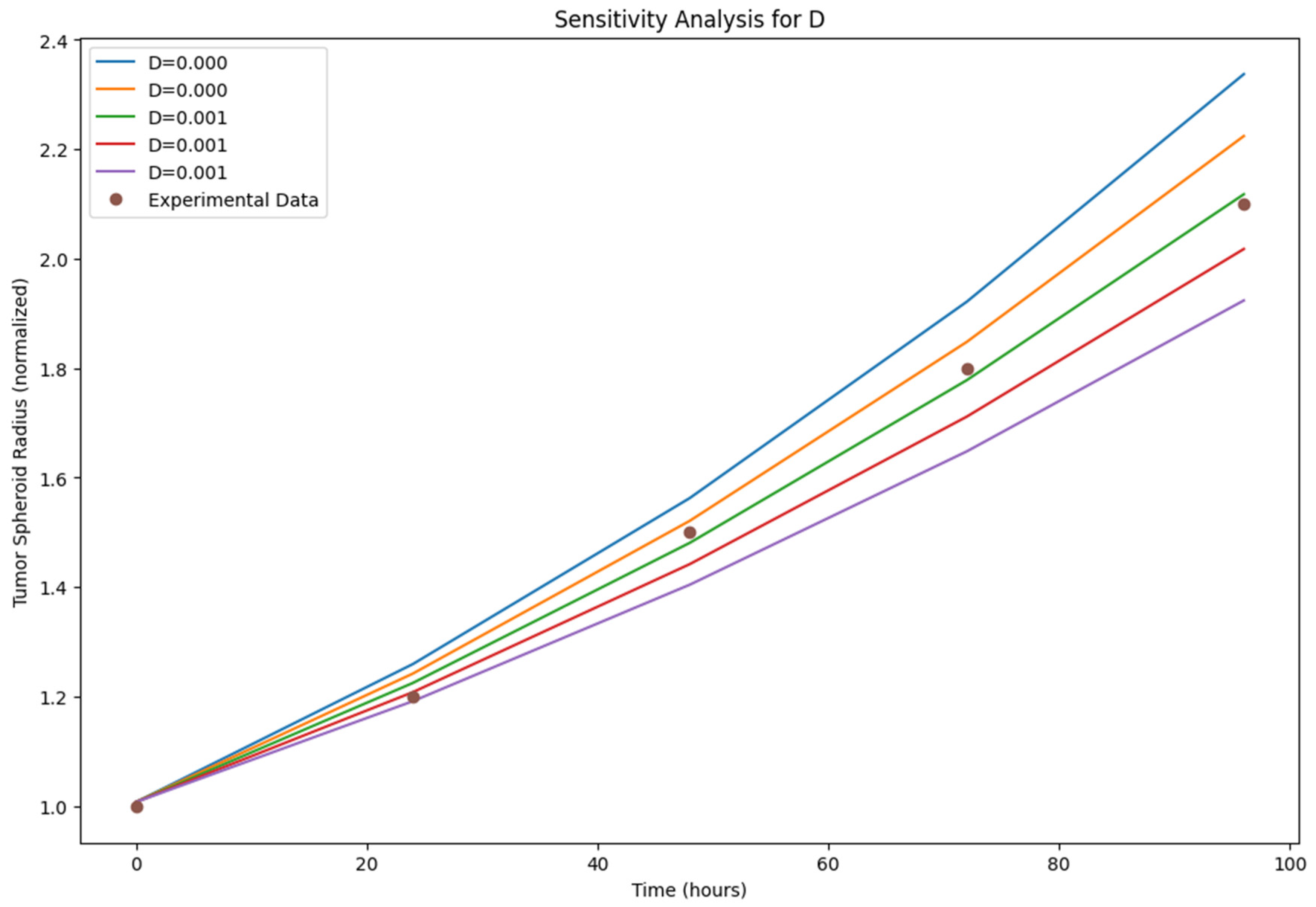
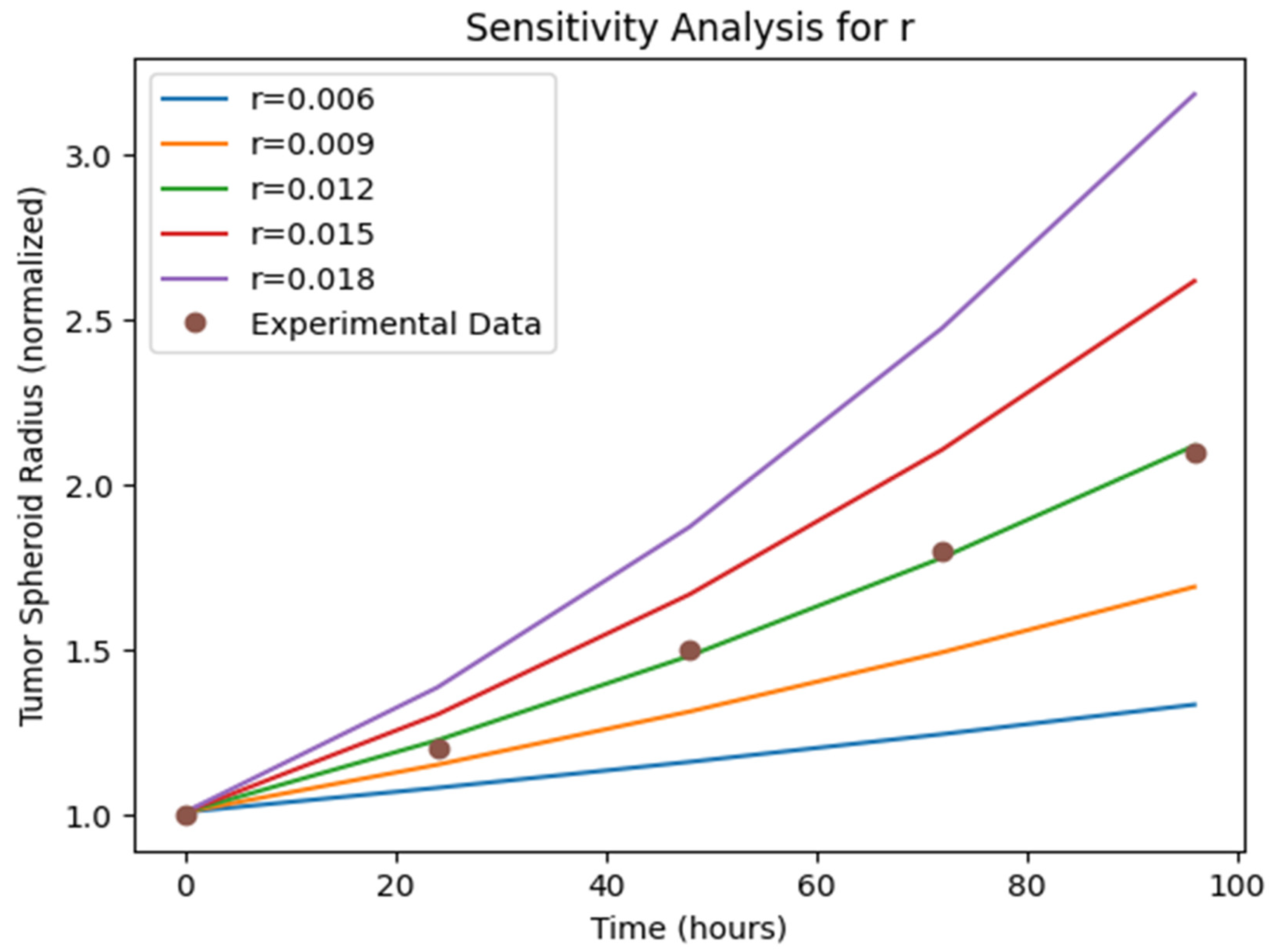
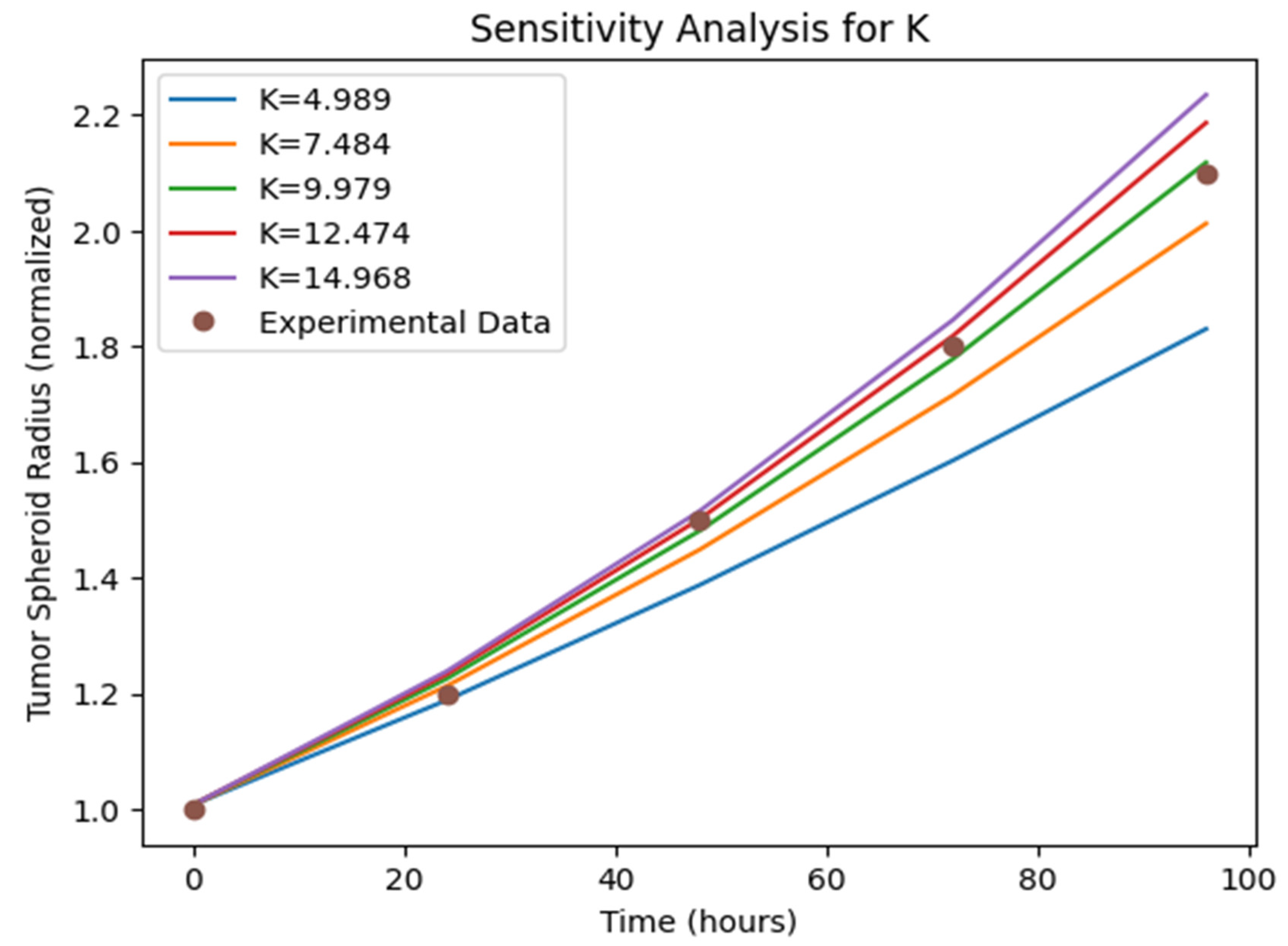
- Sensitivity Analysis: The sensitivity analysis results (Figure 5, Figure 6 and Figure 7)) provide insights into the relative importance of different model parameters in shaping the tumor growth dynamics. The plots show the impact of varying each parameter on the tumor volume, nutrient concentration, and BBB integrity. The analysis reveals that parameters such as the tumor cell diffusion coefficient (D), proliferation rate (r), and angiogenesis rate (α) have a significant influence on tumor growth and spread. Other parameters, such BBB degradation rate K, also play important roles in modulating tumor progression.
4. Discussion
5. Conclusion
6. Attachments
References
- Aldape, K., Brindle, K. M., Chesler, L., Chopra, R., Gajjar, A., Gilbert, M. R., Gottardo, N., Gutmann, D. H., Hargrave, D., Holland, E. C., Jones, D. T. W., Joyce, J. A., Kearns, P., Kieran, M. W., Mellinghoff, I. K., Merchant, M., Pfister, S. M., Pollard, S. M., Ramaswamy, V., ... Gilbertson, R. J. (2019). Challenges to curing primary brain tumours. Nature Reviews Clinical Oncology, 16(8), 509-520. [CrossRef]
- Alfonso, J. C. L., Talkenberger, K., Seifert, M., Klink, B., Hawkins-Daarud, A., Swanson, K. R., Hatzikirou, H., & Deutsch, A. (2017). The biology and mathematical modelling of glioma invasion: a review. Journal of the Royal Society Interface, 14(136), 20170490. [CrossRef]
- Alarcón, T., Byrne, H. M., & Maini, P. K. (2004). A mathematical model of the effects of hypoxia on the cell-cycle of normal and cancer cells. Journal of theoretical biology, 229(3), 395-411. [CrossRef]
- Anderson, A. R. A., & Quaranta, V. (2008). Integrative mathematical oncology. Nature Reviews Cancer, 8(3), 227-234. [CrossRef]
- Arvanitis, C. D., Ferraro, G. B., & Jain, R. K. (2020). The blood–brain barrier and blood–tumour barrier in brain tumours and metastases. Nature Reviews Cancer, 20(1), 26-41. [CrossRef]
- Batchelor, T. T., Sorensen, A. G., di Tomaso, E., Zhang, W. T., Duda, D. G., Cohen, K. S., Kozak, K. R., Cahill, D. P., Chen, P. J., Zhu, M., Ancukiewicz, M., Mrugala, M. M., Plotkin, S., Drappatz, J., Louis, D. N., Ivy, P., Scadden, D. T., Benner, T., Loeffler, J. S., ... Jain, R. K. (2007). AZD2171, a pan-VEGF receptor tyrosine kinase inhibitor, normalizes tumor vasculature and alleviates edema in glioblastoma patients. Cancer Cell, 11(1), 83-95. [CrossRef]
- Boujelben, A., Watson, M., McDougall, S., Yen, Y. F., Gerstner, E. R., Catana, C., Deisboeck, T., Batchelor, T. T., Boas, D., Rosen, B., Kalpathy-Cramer, J., & Chaplain, M. A. J. (2016). Multimodality imaging and mathematical modelling of drug delivery to glioblastomas. Interface Focus, 6(5), 20160039. [CrossRef]
- Bueno-Orovio, A., Pérez-García, V. M., & Fenton, F. H. (2012). Spectral methods for partial differential equations in irregular domains: the spectral smoothed boundary method. SIAM Journal on Scientific Computing, 28(3), 886-900. [CrossRef]
- Cai, Y., Wu, J., Li, Z., & Long, Q. (2021). Mathematical modelling of a brain tumour initiation and early development: A coupled model of glioblastoma growth, pre-existing vessel co-option, angiogenesis and blood perfusion. PLoS One, 11(3), e0150296. [CrossRef]
- Cloughesy, T. F., Cavenee, W. K., & Mischel, P. S. (2014). Glioblastoma: from molecular pathology to targeted treatment. Annual Review of Pathology: Mechanisms of Disease, 9, 1-25. [CrossRef]
- Eikenberry, S. E., Sankar, T., Preul, M. C., Kostelich, E. J., Thalhauser, C. J., & Kuang, Y. (2009). Virtual glioblastoma: growth, migration and treatment in a three-dimensional mathematical model. Cell proliferation, 42(4), 511-528. [CrossRef]
- Furnari, F. B., Fenton, T., Bachoo, R. M., Mukasa, A., Stommel, J. M., Stegh, A., ... & Cavenee, W. K. (2007). Malignant astrocytic glioma: genetics, biology, and paths to treatment. Genes & development, 21(21), 2683-2710. [CrossRef]
- Gerstner, E. R., & Fine, R. L. (2007). Increased permeability of the blood-brain barrier to chemotherapy in metastatic brain tumors: establishing a treatment paradigm. Journal of clinical oncology, 25(16), 2306-2312. [CrossRef]
- Giese, A., Bjerkvig, R., Berens, M. E., & Westphal, M. (2003). Cost of migration: invasion of malignant gliomas and implications for treatment. Journal of clinical oncology, 21(8), 1624-1636.
- Giese, A., Loo, M. A., Tran, N., Haskett, D., Coons, S. W., & Berens, M. E. (1996). Dichotomy of astrocytoma migration and proliferation. International journal of cancer, 67(2), 275-282. [CrossRef]
- Gimple, R. C., Bhargava, S., Dixit, D., & Rich, J. N. (2019). Glioblastoma stem cells: lessons from the tumor hierarchy in a lethal cancer. Genes & Development, 33(11-12), 591-609. [CrossRef]
- Hanahan, D., & Weinberg, R. A. (2011). Hallmarks of cancer: the next generation. cell, 144(5), 646-674.
- Harpold, H. L., Alvord, E. C., & Swanson, K. R. (2007). The evolution of mathematical modeling of glioma proliferation and invasion. Journal of Neuropathology & Experimental Neurology, 66(1), 1-9. [CrossRef]
- Jackson, P. R., Juliano, J., Hawkins-Daarud, A., Rockne, R. C., & Swanson, K. R. (2015). Patient-specific mathematical neuro-oncology: using a simple proliferation and invasion tumor model to inform clinical practice. Bulletin of mathematical biology, 77(5), 846-856. [CrossRef]
- Jain, R. K., di Tomaso, E., Duda, D. G., Loeffler, J. S., Sorensen, A. G., & Batchelor, T. T. (2007). Angiogenesis in brain tumours. Nature Reviews Neuroscience, 8(8), 610-622. [CrossRef]
- Kargiotis, O., Rao, J. S., & Kyritsis, A. P. (2006). Mechanisms of angiogenesis in gliomas. Journal of Neuro-Oncology, 78(3), 281-293. [CrossRef]
- Kim, Y., & Roh, S. (2013). A hybrid model for cell proliferation and migration in glioblastoma. Discrete & Continuous Dynamical Systems-B, 18(4), 969-1015. [CrossRef]
- Konstorum, A., Vella, A. T., Adler, A. J., & Laubenbacher, R. (2018). Addressing current challenges in cancer immunotherapy with mathematical and computational modelling. Journal of the Royal Society Interface, 15(144), 20180150. [CrossRef]
- Laird, A. K. (1964). Dynamics of tumor growth. British journal of cancer, 18(3), 490.
- Leder, K., Pitter, K., LaPlant, Q., Hambardzumyan, D., Ross, B. D., Chan, T.
- Montgomery RM, Queiroz LS, Rogerio F. (2015). EGFR, p53, IDH-1 and MDM2 immunohistochemical analysis in glioblastoma: therapeutic and prognostic correlation. Arquivos de neuropsiquiatria, 2015 - SciELO Brasil.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



