Submitted:
25 May 2024
Posted:
27 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Recruitment and Clinical Analyses
2.2. Statistical Analysis
2.3. Genetic Analyses
2.4. Extraction of RNA and cDNA Synthesis
2.5. RT-PCR and Next Generation Sequencing (NGS)
2.6. Exonic Splicing Enhancer Sequence (ESE) Analysis
3. Results
3.1. Clinical Evaluation of KIZ Patient
3.2. Characterization of the Expression Pattern of KIZ in Patient-Derived Skin Fibroblasts
3.3. Identification of Exonic Splicing Enhancer (ESE) Sequences in KIZ-Exon 3
3.4. Characterization of KIZ Expression in Normal Mice and Sheep Retina
3.5. Analysis of Cilia Generation and Length in Controls and Patients
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schneider, N.; Sundaresan, Y.; Gopalakrishnan, P.; Beryozkin, A.; Hanany, M.; Levanon, E.Y.; Banin, E.; Ben-Aroya, S.; Sharon, D. Inherited Retinal Diseases: Linking Genes, Disease-Causing Variants, and Relevant Therapeutic Modalities. Prog Retin Eye Res 2021, 101029. [Google Scholar] [CrossRef] [PubMed]
- Hartong, D.T.; Berson, E.L.; Dryja, T.P. Retinitis Pigmentosa. Lancet 2006, 368, 1795–1809. [Google Scholar] [CrossRef] [PubMed]
- Ayuso, C.; Millan, J.M. Retinitis Pigmentosa and Allied Conditions Today: A Paradigm of Translational Research. Genome Med 2010, 2, 34. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; Welby, E.; Li, T.; Swaroop, A. Retinal Disease in Ciliopathies: Recent Advances with a Focus on Stem Cell-Based Therapies. Transl Sci Rare Dis 2019, 4, 97–115. [Google Scholar] [CrossRef] [PubMed]
- Tatour, Y.; Ben-Yosef, T. Syndromic Inherited Retinal Diseases: Genetic, Clinical and Diagnostic Aspects. Diagnostics 2020, 10, 779. [Google Scholar] [CrossRef]
- Chen, H.Y.; Kelley, R.A.; Li, T.; Swaroop, A. Primary Cilia Biogenesis and Associated Retinal Ciliopathies. Semin Cell Dev Biol 2021, 110, 70–88. [Google Scholar] [CrossRef] [PubMed]
- El Shamieh, S.; Neuillé, M.; Terray, A.; Orhan, E.; Condroyer, C.; Démontant, V.; Michiels, C.; Antonio, A.; Boyard, F.; Lancelot, M.-E.; et al. Whole-Exome Sequencing Identifies KIZ as a Ciliary Gene Associated with Autosomal-Recessive Rod-Cone Dystrophy. Am J Hum Genet 2014, 94, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, K.; Duncan, J.L.; Biswas, P.; Soto-Hermida, A.; Matsui, H.; Jakubosky, D.; Suk, J.; Telenti, A.; Frazer, K.A.; Ayyagari, R. Whole Genome Sequencing Revealed Mutations in Two Independent Genes as the Underlying Cause of Retinal Degeneration in an Ashkenazi Jewish Pedigree. Genes (Basel) 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- El Shamieh, S.; Méjécase, C.; Bertelli, M.; Terray, A.; Michiels, C.; Condroyer, C.; Fouquet, S.; Sadoun, M.; Clérin, E.; Liu, B.; et al. Further Insights into the Ciliary Gene and Protein KIZ and Its Murine Ortholog PLK1S1 Mutated in Rod-Cone Dystrophy. Genes (Basel) 2017, 8. [Google Scholar] [CrossRef]
- Méjécase, C.; Kozak, I.; Moosajee, M. The Genetic Landscape of Inherited Eye Disorders in 74 Consecutive Families from the United Arab Emirates. Am J Med Genet C Semin Med Genet 2020, 184, 762–772. [Google Scholar] [CrossRef]
- Jauregui, R.; Chan, L.; Oh, J.K.; Cho, A.; Sparrow, J.R.; Tsang, S.H. Disease Asymmetry and Hyperautofluorescent Ring Shape in Retinitis Pigmentosa Patients. Sci Rep 2020, 10, 3364. [Google Scholar] [CrossRef] [PubMed]
- Sharon, D.; Ben-Yosef, T.; Goldenberg- Cohen, N.; Pras, E.; Gradstein, L.; Soudry, S.; Mezer, E.; Zur, D.; Abbasi, A.H.; Zeitz, C.; et al. A Nation-wide Genetic Analysis of Inherited Retinal Diseases in Israel as Assessed by the Israeli Inherited Retinal Disease Consortium (IIRDC). Hum Mutat 2019, 41, 140–149. [Google Scholar] [CrossRef]
- Weisschuh, N.; Obermaier, C.D.; Battke, F.; Bernd, A.; Kuehlewein, L.; Nasser, F.; Zobor, D.; Zrenner, E.; Weber, E.; Wissinger, B.; et al. Genetic Architecture of Inherited Retinal Degeneration in Germany: A Large Cohort Study from a Single Diagnostic Center over a 9-Year Period. Hum Mutat 2020, 41, 1514–1527. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Xu, C.L.; Breazzano, M.P.; Tanaka, A.J.; Ryu, J.; Levi, S.R.; Yao, K.; Sparrow, J.R.; Tsang, S.H. Progressive RPE Atrophy and Photoreceptor Death in KIZ-Associated Autosomal Recessive Retinitis Pigmentosa. Ophthalmic Genet 2020, 41, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Villafuerte-de la Cruz, R.A.; Garza-Garza, L.A.; Garza-Leon, M.; Rodriguez-De la Torre, C.; Parra-Bernal, C.; Vazquez-Camas, I.; Ramos-Gonzalez, D.; Rangel-Padilla, A.; Espino Barros-Palau, A.; Nava-García, J.; et al. Spectrum of Variants Associated with Inherited Retinal Dystrophies in Northeast Mexico. BMC Ophthalmol 2024, 24, 60. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Coussa, R.G.; DeBenedictis, M.J.M.; Traboulsi, E.I. Retinal Dystrophy Associated with a Kizuna (KIZ) Mutation and a Predominantly Macular Phenotype. Ophthalmic Genet 2019, 40, 455–460. [Google Scholar] [CrossRef] [PubMed]
- El Shamieh, S.; Neuillé, M.; Terray, A.; Orhan, E.; Condroyer, C.; Démontant, V.; Michiels, C.; Antonio, A.; Boyard, F.; Lancelot, M.-E.; et al. Whole-Exome Sequencing Identifies KIZ as a Ciliary Gene Associated with Autosomal-Recessive Rod-Cone Dystrophy. Am J Hum Genet 2014, 94, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Oshimori, N.; Ohsugi, M.; Yamamoto, T. The Plk1 Target Kizuna Stabilizes Mitotic Centrosomes to Ensure Spindle Bipolarity. Nat Cell Biol 2006, 8, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- El Shamieh, S.; Méjécase, C.; Bertelli, M.; Terray, A.; Michiels, C.; Condroyer, C.; Fouquet, S.; Sadoun, M.; Clérin, E.; Liu, B.; et al. Further Insights into the Ciliary Gene and Protein KIZ and Its Murine Ortholog PLK1S1 Mutated in Rod-Cone Dystrophy. Genes (Basel) 2017, 8. [Google Scholar] [CrossRef]
- Beryozkin, A.; Khateb, S.; Idrobo-Robalino, C.A.; Khan, M.I.; Cremers, F.P.M.; Obolensky, A.; Hanany, M.; Mezer, E.; Chowers, I.; Newman, H.; et al. Unique Combination of Clinical Features in a Large Cohort of 100 Patients with Retinitis Pigmentosa Caused by FAM161A Mutations. Sci Rep 2020, 10. [Google Scholar] [CrossRef]
- Kimchi, A.; Khateb, S.; Wen, R.; Guan, Z.; Obolensky, A.; Beryozkin, A.; Kurtzman, S.; Blumenfeld, A.; Pras, E.; Jacobson, S.G.; et al. Nonsyndromic Retinitis Pigmentosa in the Ashkenazi Jewish Population. Genetic and Clinical Aspects. Ophthalmology 2018, 125, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Beryozkin, A.; Khateb, S.; Idrobo-Robalino, C.; Khan, M.; Cremers, F.; Obolensky, A.; Hanany, M.; Mezer, E.; Chowers, I.; Newman, H.; et al. Unique Combination of Clinical Features in a Large Cohort of 100 Patients with Retinitis Pigmentosa Caused by FAM161A Mutations. Sci Rep 2020, 10, 15156. [Google Scholar] [CrossRef] [PubMed]
- Sundaresan, Y.; Banin, E.; Sharon, D. Exonic Variants That Affect Splicing - An Opportunity for “Hidden” Mutations Causing Inherited Retinal Diseases. Adv Exp Med Biol 2023, 1415, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Hanany, M.; Rivolta, C.; Sharon, D. Worldwide Carrier Frequency and Genetic Prevalence of Autosomal Recessive Inherited Retinal Diseases. Proceedings of the National Academy of Sciences 2020, 117, 2710–2716. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Xu, C.L.; Breazzano, M.P.; Tanaka, A.J.; Ryu, J.; Levi, S.R.; Yao, K.; Sparrow, J.R.; Tsang, S.H. Progressive RPE Atrophy and Photoreceptor Death in KIZ-Associated Autosomal Recessive Retinitis Pigmentosa. Ophthalmic Genet 2020, 41, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Kramer, F.; Mohr, N.; Kellner, U.; Rudolph, G.; Weber, B.H. Ten Novel Mutations in VMD2 Associated with Best Macular Dystrophy (BMD). Hum Mutat 2003, 22, 418. [Google Scholar] [CrossRef] [PubMed]
- Finkel, R.S.; Flanigan, K.M.; Wong, B.; Bonnemann, C.; Sampson, J.; Sweeney, H.L.; Reha, A.; Northcutt, V.J.; Elfring, G.; Barth, J.; et al. Phase 2a Study of Ataluren-Mediated Dystrophin Production in Patients with Nonsense Mutation Duchenne Muscular Dystrophy. PLoS One 2013, 8, e81302. [Google Scholar] [CrossRef] [PubMed]
- Wilschanski, M.; Miller, L.L.; Shoseyov, D.; Blau, H.; Rivlin, J.; Aviram, M.; Cohen, M.; Armoni, S.; Yaakov, Y.; Pugatsch, T.; et al. Chronic Ataluren (PTC124) Treatment of Nonsense Mutation Cystic Fibrosis. Eur Respir J 2011, 38, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Samanta, A.; Stingl, K.; Kohl, S.; Nagel-Wolfrum, K.; Ries, J.; Linnert, J. Ataluren for the Treatment of Usher Syndrome 2A Caused by Nonsense Mutations. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Mendonca, C.A.; Yukselen, O.; Muneeruddin, K.; Ren, L.; Liang, J.; Zhou, C.; Xie, J.; Li, J.; et al. AAV-Delivered Suppressor TRNA Overcomes a Nonsense Mutation in Mice. Nature 2022, 604, 343–348. [Google Scholar] [CrossRef]
- Albers, S.; Allen, E.C.; Bharti, N.; Davyt, M.; Joshi, D.; Perez-Garcia, C.G.; Santos, L.; Mukthavaram, R.; Delgado-Toscano, M.A.; Molina, B.; et al. Engineered TRNAs Suppress Nonsense Mutations in Cells and in Vivo. Nature 2023, 618, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Hentze, M.W.; Kulozik, A.E. A Perfect Message: RNA Surveillance and Nonsense-Mediated Decay. Cell 1999, 96, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Schneider, N.; Steinberg, R.; Ben-David, A.; Valensi, J.; David-Kadoch, G.; Rosenwasser, Z.; Banin, E.; Levanon, E.Y.; Sharon, D.; Ben-Aroya, S. A Pipeline for Identifying Guide RNA Sequences That Promote RNA Editing of Nonsense Mutations That Cause Inherited Retinal Diseases. Mol Ther Nucleic Acids 2024, 35, 102130. [Google Scholar] [CrossRef] [PubMed]
- Beryozkin, A.; Samanta, A.; Gopalakrishnan, P.; Khateb, S.; Banin, E.; Sharon, D.; Nagel-Wolfrum, K. Translational Read-Through Drugs (TRIDs) Are Able to Restore Protein Expression and Ciliogenesis in Fibroblasts of Patients with Retinitis Pigmentosa Caused by a Premature Termination Codon in FAM161A. Int J Mol Sci 2022, 23, 3541. [Google Scholar] [CrossRef]
- Vössing, C.; Owczarek-Lipska, M.; Nagel-Wolfrum, K.; Reiff, C.; Jüschke, C.; Neidhardt, J. Translational Read-through Therapy of Rpgr Nonsense Mutations. Int J Mol Sci 2020, 21, 1–16. [Google Scholar] [CrossRef]
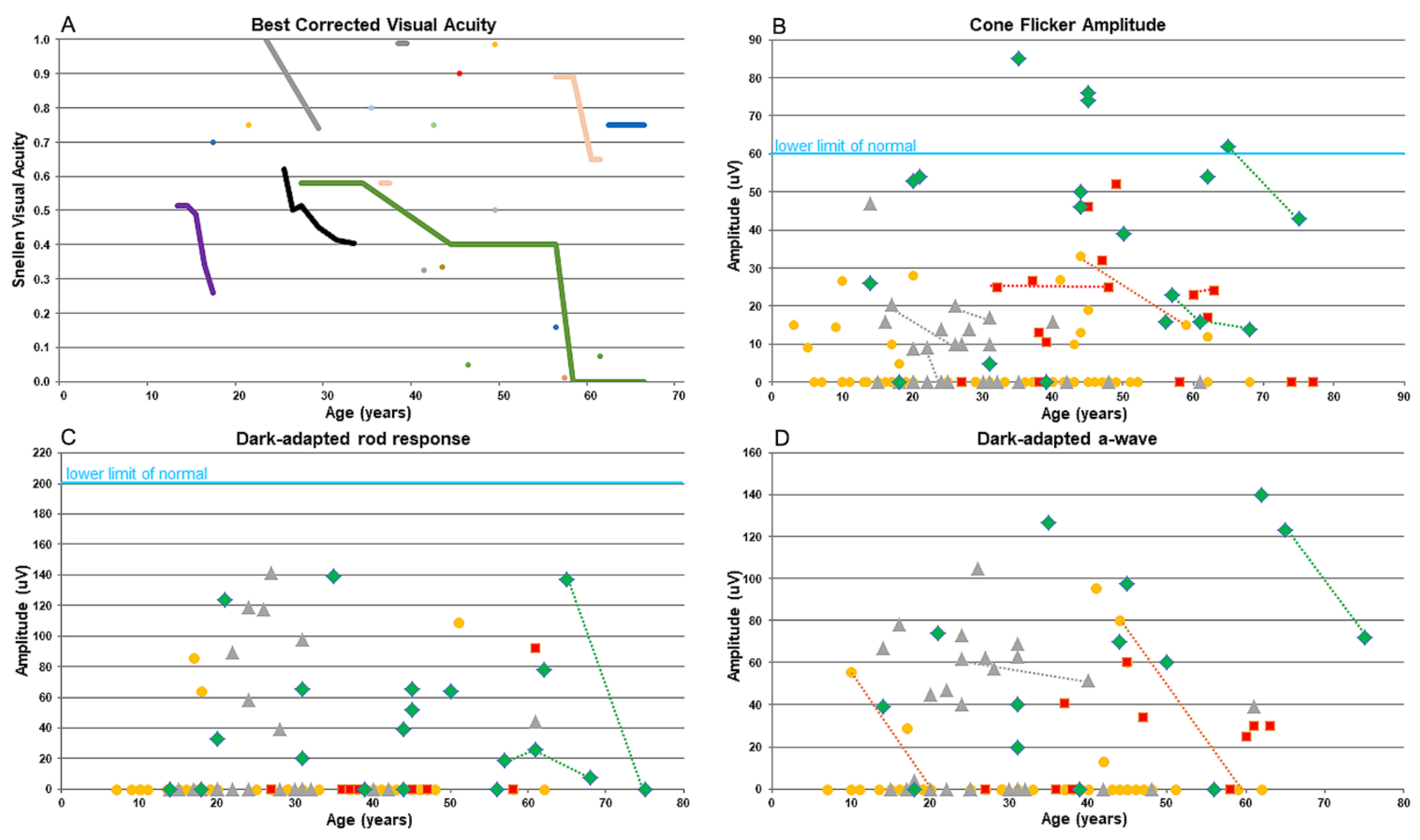
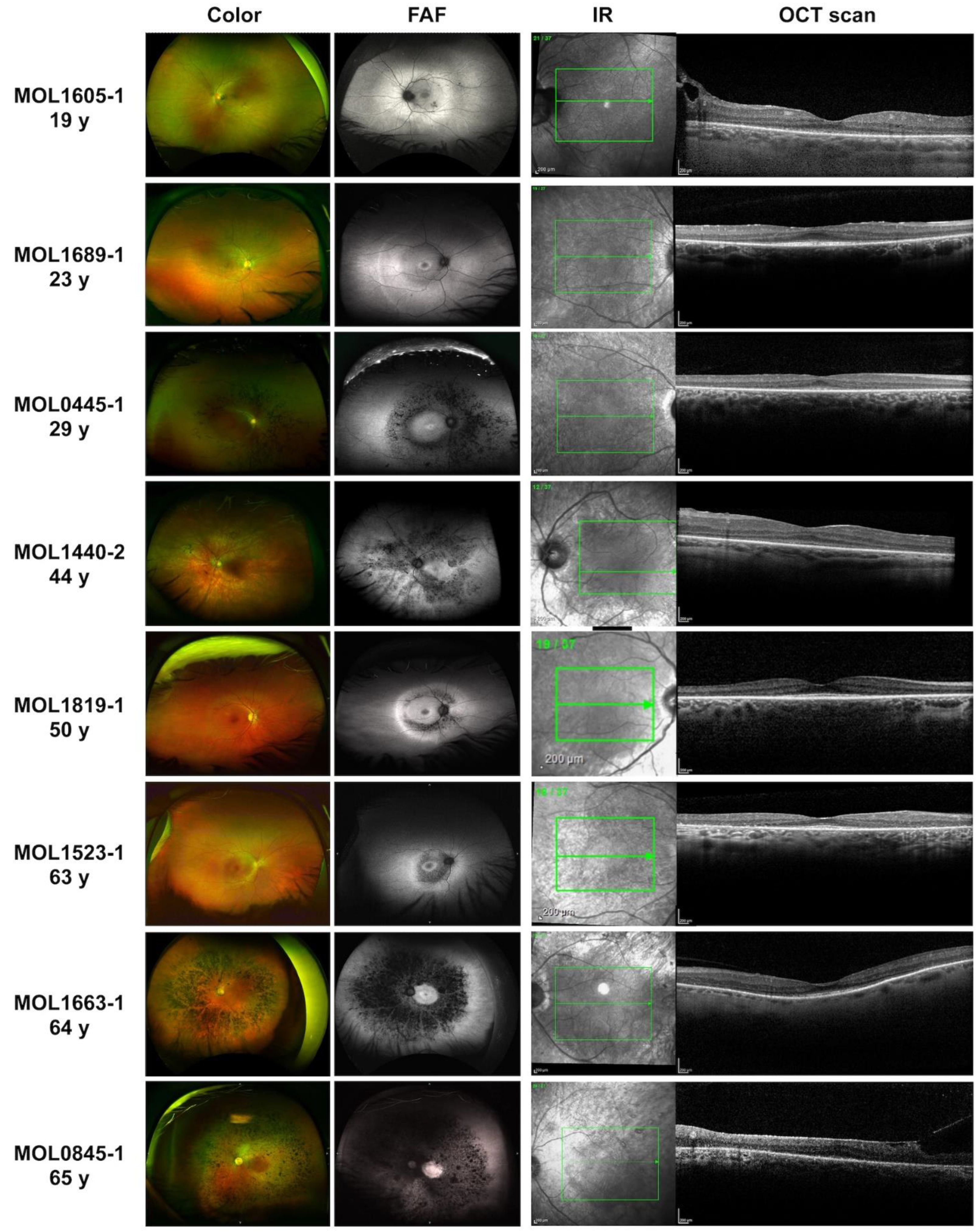
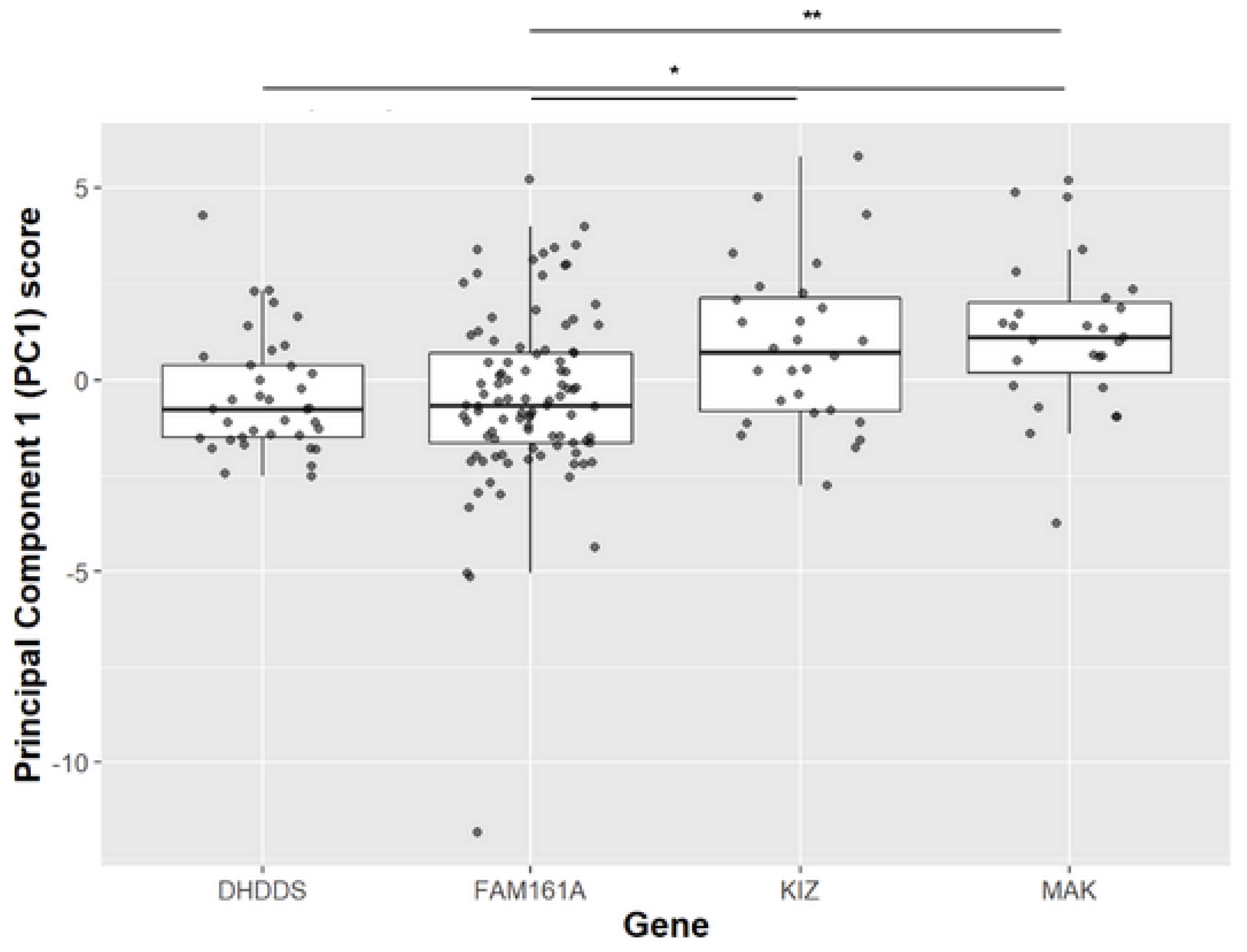
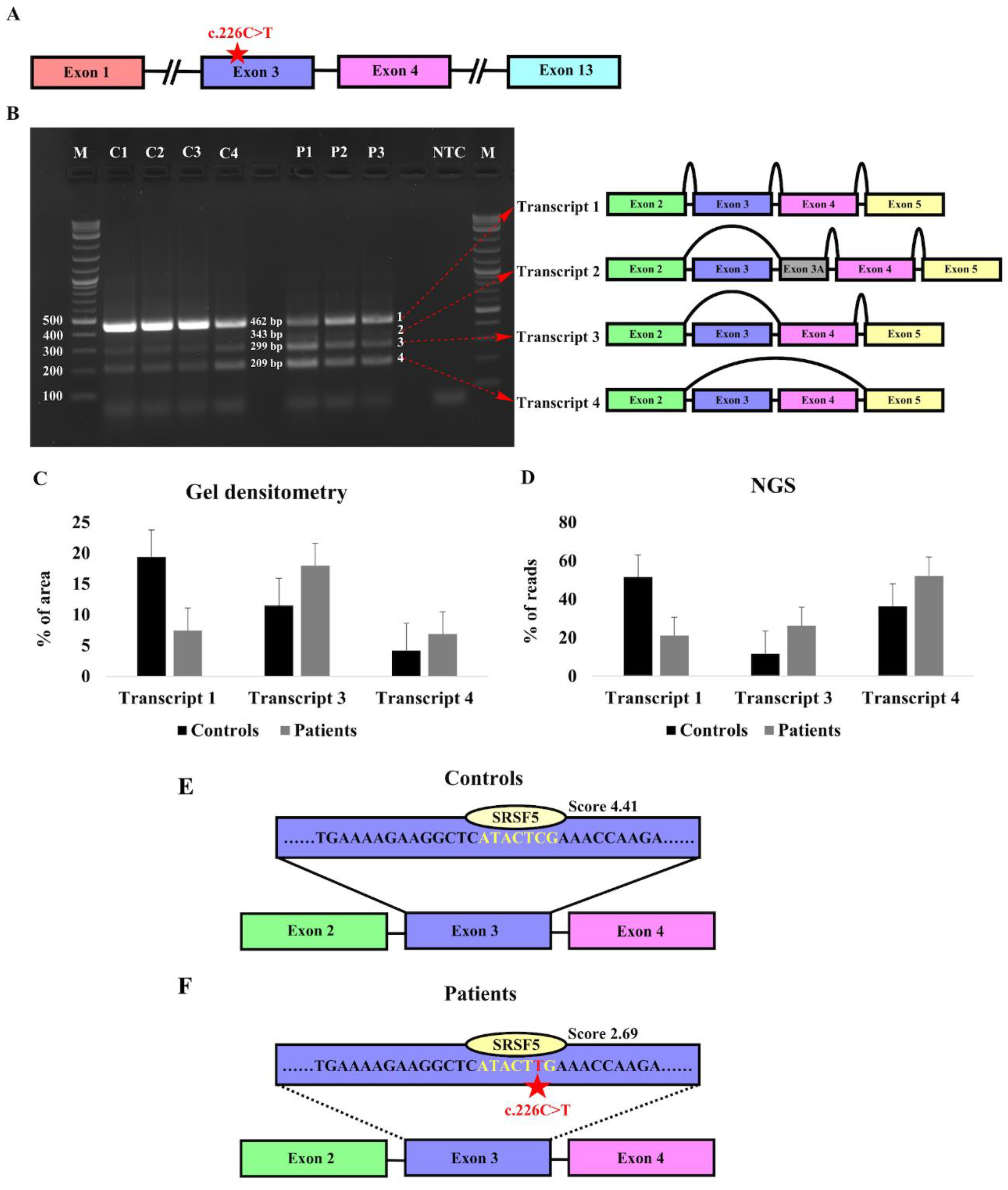
| Family Number | Origin | Consanguinity | Mutation 1 | Mutation 2 | # affected recruited | # affected- not recruited |
|---|---|---|---|---|---|---|
| MOL0289 | Turkish Jew | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL0336 | NAJ | 2:3 | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL0445 | NAJ | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL0588 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 1 |
| MOL0610 | ASH | 3:3 | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL0845 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL1015 | ASH/Turkish Jew | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL1156 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL1236 | AM | 2:2 | c.247C>T, p.R83* | c.247C>T, p.R83* | 1 | 0 |
| MOL1329 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL1440 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 2 | 0 |
| MOL1523 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL1605 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 1 |
| MOL1621 | ASH/Iraq | No | c.226C>T, p.R76* | c.3G>A, p.M1? | 1 | 1 |
| MOL1663 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL1684 | ASH | 3:3 | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL1689 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL1720 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| MOL1819 | NAJ | No | c.226C>T, p.R76* | c.3G>A, p.M1? | 1 | 0 |
| MOL2026 | NAJ | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| TB240 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| TB244 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| TB338 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| TB675 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| TB711 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| TB736 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| TB928 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| TB980 | NAJ | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| TB1044 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
| TB1212 | ASH | No | c.226C>T, p.R76* | c.226C>T, p.R76* | 1 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




