1. Introduction
In recent decades, the food industry has been characterized by the large volume of waste it produces. Approximately one third of the food produced for human consumption, around 1.3 billion tons per year, is lost or wasted worldwide. The most significant loss is observed in fruits and vegetables, around 0.5 billion tons. In developing countries, much of this loss occurs during processing. However, losses during post-harvest and distribution stages are also significant.
[1,2]
In Brazil, food processing activities have expanded intensely to meet the market demand for processed foods.
[3] The processing of fruits and vegetables generates a substantial amount of waste, including peels, seeds, pulp, and bagasse. These by-products are known to be rich sources of nutrients, mainly biopolymers (such as polysaccharides and dietary fiber) and bioactive compounds.
[4] In this context, it is important to find applications for the residue of camu-camu, especially considering that the fruit is rich in vitamin C.
Biopolymers, such as polysaccharides and proteins, derived from agricultural byproducts have been proposed for the formulation of biodegradable materials. They are susceptible to biodegradation (except when subjected to severe chemical modifications), originate from renewable sources, and are non-toxic to the soil and environment.
[5] Due to these environmental concerns, significant attention has been directed towards the development of edible/biodegradable films. Edible films are packaging materials made from non-toxic macromolecules designed to control barriers like moisture permeability and other factors that accelerate food degradation, such as methylene, oxygen, and light exposure. Additionally, these films can incorporate additives such as antimicrobials and antioxidants, aimed at expanding the range of potential applications for these materials.
[6]These films can also enhance nutritional value and improve sensory characteristics when consumed along with the product they are applied. Moreover, they can be used as an alternative to petroleum-derived raw materials, reinforcing the sustainability of the process.
[7]
Films derived from vegetables exhibit low to moderate oxygen permeability and acceptable mechanical properties. For instance, films incorporated with kale pulp are suggested as an alternative for the production of edible packaging and coatings.
[8] However, there are still limited studies on the use of fruit and vegetable processing residues in the development of biodegradable films.
[9,10] In studies conducted by Rangaraj, Rambabu, Banat, and Mittal,
[11] the effect of incorporating extract derived from date syrup waste into gelatin films for food preservation was examined. It was reported that the extract, slowly and in a controlled manner, releases antioxidant molecules within the films, thereby preventing the formation of peroxides and consequent degradation reactions, such as lipid auto-oxidation. Another study by Santos and collaborators
[12] reported antioxidant activities in gelatin films incorporated with Malpighia emarginata waste extract. The addition of 4% residue was shown to reduce the formation of malonaldehyde and total carbonyl compounds. Consequently, the resulting packaging exhibits promising properties for preserving meat products, potentially extending their shelf life.
In an effort to find a beneficial application for camu-camu residue, considering the fruit’s richness in vitamins, the residue was incorporated into a gelatin and glycerin matrix, and its characteristics were analyzed. The fruit is primarily sold on a small scale at local markets in the production region, with the majority being sold as frozen pulp. Although relatively unknown domestically, it is highly sought after by the Japanese, Americans, and Europeans, and is exported in refrigerated containers.
[13] It is important to highlight that this was the first study on gelatin and glycerin films incorporated with camu-camu residues. In studies where camu-camu residues were integrated into polymeric film matrices, it was noted that the bioactive compounds of camu-camu could impart antioxidant properties to the packaging. This observation was confirmed by Ju and Song,
[14]who developed biodegradable films based on starch and camu-camu with these specific characteristics.
In terms of food packaging in Brazil, according to the Brazilian Packaging Association (ABRE), plastics account for 37.47% of the total value of packaging production. This material consequently represents a similar volume of improper disposal, in addition to the emission of toxic and polluting gases during production stages. However, due to their diversity and versatility, petroleum-derived polymers provide technological advancements, energy savings, and several other benefits to society through the production of a variety of products. Bioplastics, such as edible films developed from biopolymers such as hydroxypropyl methylcellulose (HPMC), chitosan, alginates, and gelatins, are non-toxic natural alternatives that are extremely promising for replacing conventional packaging. Some of these are also abundant sources of antimicrobial action.
[9,10,15]
One of the main characteristics of gelatin is its high hygroscopic nature. When in contact with cold water, its particles swell, increasing their mass by about ten times. When heated above its denaturation temperature (40 °C), gelatin forms a colloidal solution with water and gels after cooling. This behavior allows films made from this material to be more likely to dissolve when immersed in water above this temperature.
[16,17]
Given the current scenario, considering the escalating generation of waste from fruit and vegetable processing and their potential in the synthesis of new products, coupled with the environmental impact caused by plastic packaging widely used in the food industry, this paper aimed to check the potential of using fruit waste in the production of a biodegradable film and its potential application, particularly in their use for managing and protecting solid foods with high water activity and a significant presence of phenolic compounds and lipids in their composition. Simultaneously, by appreciating the utilization of an Amazonian fruit, we aim to showcase the potential of Amazonian raw materials. Demonstrating how their comprehensive exploitation can not only address environmental challenges but also stimulate the local circular economy.
2. Materials and Methods
2.1. Material
Gelatin powder P.A. (bovine) (CAS [9000-70-8] Dinâmica (Indaiatuba – SP); Bi-distilled Glycerol U.S.P (P.M. 92.09) Synth (Diadema – SP); Camu-camu residue dried at 50ºC for 24 hours and ground at the State University of Maringá-PR, the residue was obtained from the unused fruit parts leftover from pulp production for ice cream. It is rich in fibers and may contain a considerable amount of vitamin C and antioxidants.
2.2. Preparation of the Gelatin Filmogenic Solution
A subjective analysis and a pH analysis were conducted for the selection of the best film, focusing on a positive results characterization. Gelatin solutions at 2% (w.v-1) (the film matrix) were produced with 1 g of powdered gelatin hydrated in 49 g of distilled water for one hour. After hydration, the sample was conditioned in a water bath, monitoring the temperature up to 50 ºC for complete dissolution. Finally, the solution was stirred on a magnetic stirrer (500 rpm) until cooling down to 30 ºC. Bi-distilled glycerin was used at 25% and 35% (w.w-1) relative to the mass of the polymer. The amount of glycerin was chosen based on studies conducted previously by the research group. This compound was added to improve the manageability of the samples.
2.3. Preparation and Weighing of Camu-Camu Extract
The camu-camu extract was dried at 50 ºC for 24 hours. The extract was then macerated manually in a porcelain crucible to obtain fine grains to maximize homogenization in the matrix. Two concentrations were used for the characterizations (0.25 g and 0.5 g).
2.4. Incorporation of the Extract into the Matrix
The films were produced using the casting technique, incorporating glycerin and camu-camu extract. The Casting involves the deposition of the filmogenic solution onto a polyester support, on a leveled bench, and then dried at room temperature. This method is widely disseminated by the research group for obtaining this type of packaging.
[18]
The experimental planning consisted of samples 1, 2, 3, and 4: sample 1 contains 0.25 g of extract and 0.25 g of glycerin incorporated into the matrix; sample 2 contains 0.5 g of extract and 0.25 g of glycerin; sample 3 contains 0.25 g of extract and 0.35 g of glycerin; and sample 4 contains 0.5 g of extract and 0.35 g of glycerin. All samples were tested with a Mod.752 magnetic stirrer (Fisatom Brasil) for two hours at a speed of 500 rpm, and also with an Ultra-Turrax homogenizer (Marconi-MA102) for 30 minutes at the speed of 12000 rpm.
Results will also present the samples: pure gelatin with 0.25g of glycerin (named “pure 25%”); and pure gelatin with 0.35g of glycerin (“pure 35%”).
2.5. Determination of Thickness
The thickness of the films was obtained using a digital micrometer (No. 7326, Mitutoyo Manufacturing, Japan). Five analyses were conducted in different areas of the film.
2.6. Scanning Electron Microscopy (SEM)
The morphology analyses of the films were conducted in a computerized scanning electron microscope (ZEISS model EVO LS15), with a voltage of 12 kV. The films were fixed on a double-sided carbon tape and metallized by a Sputter Coater (Quorum, model Q150 T) for 1.5 minute for the deposition of a thin layer of gold on them.
For this analysis, the samples were placed in a glass desiccator with silica for 24 hours to remove the moisture present in the film. Only after this period were the films cut for analysis, into square shapes, each with a length of 5cm.
[19]
2.7. Quantification of Total Phenolic Compounds and Antioxidant Capacity
Samples 3, 4, and pure 35% were analyzed immediately after preparation (1:3 w/v with methanol), after extraction. The extracts were obtained by homogenization using centrifugation (15 min, 4,000 rpm) and filtration (filter paper).
[20]
Total phenolic compounds were determined by the Folin-Ciocalteau colorimetric method
[21] with minor modifications. The results were expressed in milligrams of gallic acid equivalent per 100 g of sample on a dry basis (mg GAE g-1 dry basis).
The free radical scavenging potential was determined by the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay according to Thaipong et al.
[22] The free radical scavenging by the 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical was determined according to Re et al., (1999).
[23] The FRAP assay was performed according to the methodology of Benzie & Strain, (1996).
[24] All antioxidant activities were expressed in micromoles of Trolox equivalent per gram of dry weight (µmol TE g-1 dry weight).
2.8. Water Vapor Permeability (WVP)
For the WVP analyses, the method adapted from ASTM (1980) was used,
[25] as described by McHugh and collaborated.
[26]
Plates with the films were placed in an oven with a controlled temperature (25 ± 2 °C) and humidity (50 ± 3 %). Silica was used to control internal humidity. Plates were weighed hourly to determine the WVP. To calculate the WVP, Equations (1)–(3) were used.
Where: mw is the molar mass of water (18 g.mol-1), D is the diffusivity of water vapor through air at 298 K (0.102 m2.s-1), P is the total pressure (1 atm), p1 is the vapor pressure at 298 K, R is the gas constant (82.1 x 10-6 m3.atm/g.mol.K), z is the average height the inert gas reaches, p2 (Partial vapor pressure at the bottom of the film), p3 (Partial vapor pressure at the top of the film), and y (Average film thickness).
2.9. Soil Biodegradation
To evaluate the soil biodegradation of the developed films, a commercial plant substrate from the brand Carolina Soil was used. Its composition includes peat, vermiculite, class A agro-industrial organic residue (roasted rice husk), and limestone. The substrate characterization was described on the label.
According to the methodology applied by Pavoni et al.,
[27] film samples of 25 mm² were arranged in a plastic mesh, in triplicate, and buried in the substrate at a depth of approximately 20 mm in a polypropylene (PP) container with an internal diameter of 14 cm. The biodegradation test occurred at room temperature, and deionized water was sprayed daily to maintain humidity. Commercial films of low-density polyethylene (LDPE) and cellulose were tested as controls.
As reported by Abe et al.,
[28] biodegradation measurements by weight loss of bioplastics are extremely difficult to perform. Therefore, the samples were carefully removed from the substrate after 3, 5, 10, 15, and 30 days to evaluate the disintegration process by visual inspection.
2.10. Morphological Analysis
For the visual analysis, photomicrographs were taken (Zeiss Optical Stereo Microscope, model Stemi 508), in addition to photographs (Nikon, model Coolpix P510). The morphological analysis was carried out on a scanning electron microscope (JEOL, model JSM-6510LV). The sample preparation included drying at 60ºC in an oven without air circulation for 1 hour. Subsequently, the samples were glued onto a copper support with carbon tape and vacuum metallization with gold was carried out for 3 minutes (Denton Vacuum, model Desk V). All samples were kept in a desiccator before analyses.
2.11. Seed Germination in the Presence of Degraded Film
The methodology developed by Harada and collaborated
[29] was adapted to evaluate the toxicity of the substrate for plant growth after the degradation of the films over a period of 30 days. Black common bean Phaseolus vulgaris (L.) was planted (7 seeds per container), using the plant substrate that was not exposed to the samples during the biodegradation experiment as a control. The seeds were watered with deionized water daily for 10 days. They were then cleaned and the parameters of germination (%), growth (cm), and total biomass (g) were evaluated.
3. Results and Discussion
3.1. Gelatin Films (2%) Incorporated with Glycerin and Camu-Camu Residue
A plan was implemented for the production of films that, once ready, underwent a subjective evaluation to classify them based on their continuity (absence of fracture or rupture after drying), homogeneity (absence of particles visible to the naked eye, opacity) and handling (ability to handle the film without risk of breakage. [
30]
In the subjective analysis, the films with the best continuity, homogeneity, and handling were those produced with a 2% (w/v) gelatin matrix, classified as samples 3 and 4. The films titled as samples 1 and 2 came in second place. Based on this analysis, the films named as samples 1 and 2 were eliminated from the preparation process due to their poor handling, roughness, and brittleness. In all samples prepared for analysis, the insoluble residue was used in the incorporation.
3.2. pH Analysis of Gelatin Films (2%) Incorporated with Glycerin and Camu-Camu Residue
In accordance with the pH analysis (
Table 1) was also conducted to verify the influence of the residue on the film matrix in terms of viscosity and acidification.
Analyzing the results in the table, it can be stated that the presence of camu-camu residue slightly acidifies the filmogenic solution. This is an expected result, as astringent fruits have this characteristic, which contributes to the formation of films as the solution becomes more viscous. It is already reported in the literature that viscosity influences the formation of films. [
30]
3.3. Film Thickness
The average thickness of the films with a precision of 0.001mm in five different areas of the film, ranged between 0.020 and 0.080 mm. This result is compatible with other published results. [
31]
It is worth mentioning that because these are films incorporated with residues that are not completely dissolved, the thickness varies depending on the presence of insoluble residues. This suggests that the surface presents a heterogeneity in relation to the thickness.
3.4. Scanning Electron Microscopy (SEM)
Based on the surface morphology of the films presented in
Figure 1, as well as the cryogenic fractures of the cross-section of the 35% pure matrix films, are shown in
Figure 1d, and sample films 3 and 4 are shown in
Figure 1d,e, respectively. The images in
Figure 1d show a smooth and homogeneous surface, without phase separation or formation of agglomerates. The images in
Figure 1d,e, which show the surfaces of the polymer matrices and cryogenic fractures, display surfaces with some agglomerates, showing high similarity in all samples, taking into account the regions containing non-solubilized residue.
This change in the texture of the surface and interior of the film when camu-camu residue is added is possibly related to the formation of agglomerates caused by the interaction between the components of the gelatin film and the residue.
It is possible to identify that there was a change in surface roughness and formation of agglomerates with the addition of the camu-camu residue, due to the approximation of the polar chains that occurs in the polymeric matrix. However, as can be seen in
Figure 1e,f, the addition of camu-camu residue caused changes in the microstructure of the films. It is possible to verify the presence of pores in the films, indicating, according to de Andrade, [
32] who studied the addition of vegetable flour in edible films. This occurs due to the incorporation of a hydrophobic component, with a high amount of fiber, into a hydrophilic phase. Possibly, the addition of the camu-camu residue caused some disturbance between the interactions of the polymeric network, making it difficult for the chains to align and, consequently, causing heterogeneity in the system, as shown in the cross-sectional micrographs.
3.5. Quantification of Antioxidant Capacity
Table 2 presents the analysis of antioxidant capacity using DPPH, FRAP and ABTS methods expressed in μg Trolox/g of sample and total phenolic compounds in mg EAG/g of sample.
Considering the values presented, we can infer that in the analysis of pure residue, a very satisfactory result was obtained. When compared with the films incorporated with residue, even at lower concentrations, we can affirm the presence of antioxidants and phenolic compounds. This justifies the result when not detected on the film that does not contain residue. Sample 4 presented 56% more total phenolic compounds and on average 60% more antioxidant content than sample 3.
3.6. Water Vapor Permeability (WVP)
Are presented in
Table 3, the results obtained for the water vapor permeability of the films produced. The water vapor permeability (WVP) occurs due to a difference in relative humidity (RH) inside the cells used. This analysis measures water vapor’s ability to diffuse into a region with lower water vapor pressure from a region with higher water vapor pressure. [
33]
It is possible to observe that the film produced only from 2% gelatin with 25% and 35% glycerin presents significantly lower water vapor permeability values than those of the other two formulations of 2% gelatin with 35% glycerin embedded with 0.25 g and 0.5 g of camu-camu.
As shown in
Table 3, the WVP value increased from 1.146 to 2.72 g-mm/kPa-h-m2 when the residue was incorporated with 0.25 g of camu-camu, and from 1.146 to 2.712 g-mm/ kPa-h-m
2 when the residue was incorporated with 0.5 g of camu-camu.
This increase can be attributed to the presence of glucose found in fruits and in greater quantities in these two formulations, impairing the barrier properties of the material. However, further analyses need to be performed to confirm this hypothesis. [
1,
2]
Films of the same concentration were compared, 2% gelatin with 35% glycerin, incorporated with 0.25 g of camu-camu, using the same production process. When added to the polyester base for drying, the insoluble substrate was not used. It was left at the bottom of the beaker.
It is observed that the film without residue had a WVP of approximately 20% difference, making it clear that the incorporation of the insoluble substrate in the matrix during the drying process, aids in water vapor passage. This confirms the presented difference in WVP of the pure gelatin matrix with samples incorporated with camu-camu substrate, analyzed in
Table 3.
These results may be favorable depending on their applications and are in line with the results reported by Altiok et al., [
34] and Kavoosi et al. [
35] For example, if the application is in a food that can allow this passage from WVP.
3.7. Biodegradation of Samples
Figure 2 shows samples before biodegradation and after some periods. The visual aspects analysis carried out through photographs indicated that the samples acquired greater opacity than initially, in addition to becoming rigid and brittle. The films showed changes from the third day of the experiment. At the end of the analyzed period, they did not completely degrade, possibly due to the plastic mesh support used, preventing full contact between the bottom surface of the samples and the substrate.
When comparing different materials over a period of 30 days, it appears that Samples 03 and 04 were those that degraded most, with two of the three triplicate samples being the most affected.
In other photographs illustrating the degradation of sample 04 after 15 and 30 days, as presented in the supplementary material of this work, a more detailed observation of the sample’s mass loss and darkening can be observed. This darkening likely resulted from hydrolysis caused by enzymes and microorganisms present in the degradation environment, given the nutrient-rich and fiber-rich composition of the material. These modifications may have occurred due to substrate particles that adhered to the surface, in addition to the material’s structure’s undergoing degradation due to contact with the plant substrate, environmental microorganisms, and humidity. [
4]
3.8. Seeds Germinated with the Presence of Degraded Films
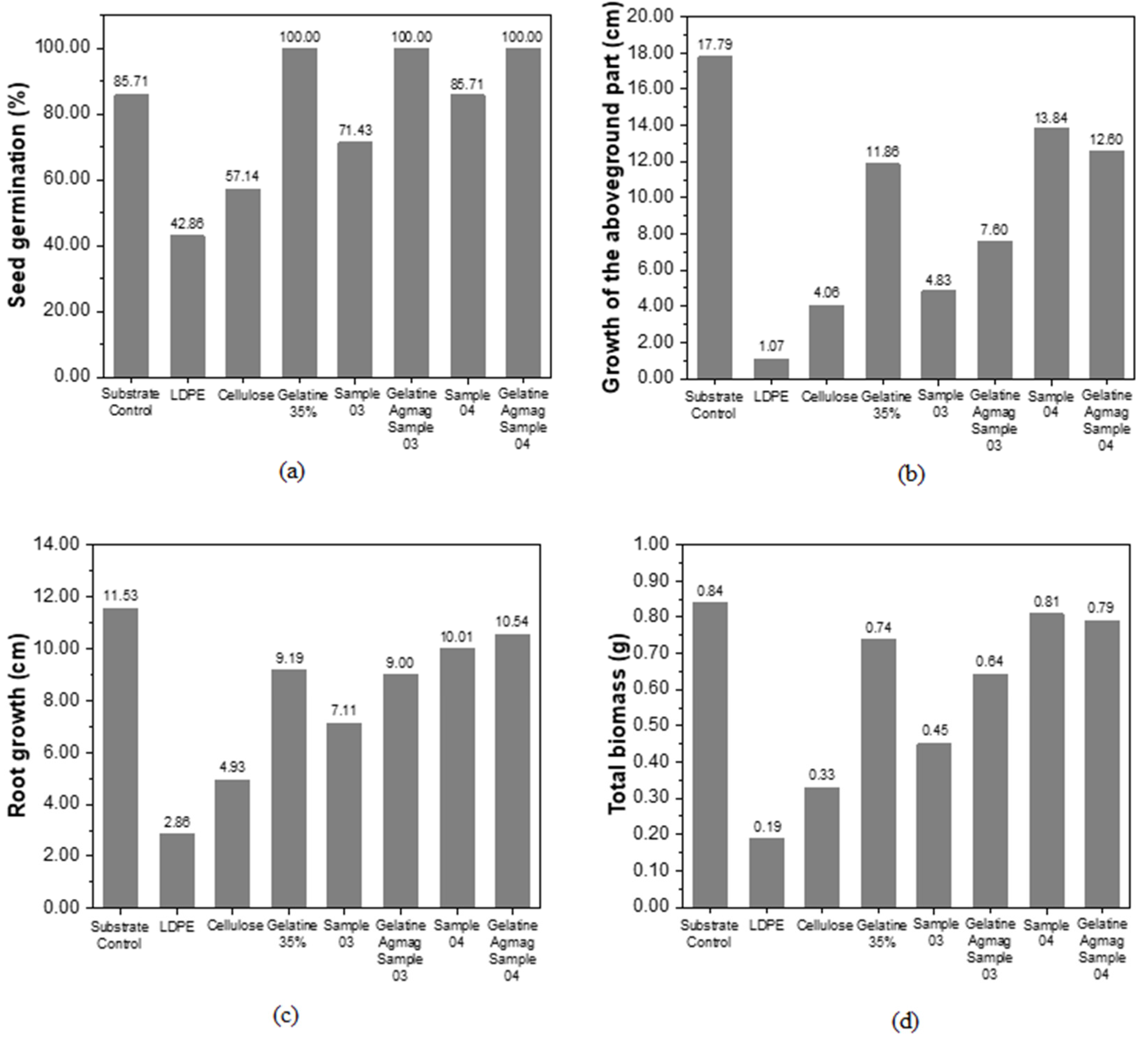
Figure 3 presents the results of germinations inserted in different situations, where the presence of degraded films presents great potential when used as substrates for planting.
Despite the observed reduction in aerial part growth, root growth, and total biomass in
Figure 3b–d, in terms of the germination percentage presented in
Figure 3a, it was demonstrated that in the presence of products resulting from the degradation of Gelatine 35% samples, Gelatine Agmag Sample 03, and Gelatine Agmag Sample 04, all seeds germinated. This value is higher than that presented by the control with pure substrate and Sample 04, which had a germination rate of 85.71%. This increase in germination and the addition in the growth of the aerial part, root, and total biomass compared to seeds with cellulose and LDPE.
The germination and growth of plants related to Sample 04 showed a satisfactory development compared to other samples, as an example, the growth results of the upper part of the seedlings obtained the highest value (13.84 cm), indicating that the samples developed in this study may contribute to plant growth.
Seed germination after 10 days showed a very satisfactory development, indicating that the residue contributed to seed growth.
With the analysis of the graphs, it is clear that the presence of camu-camu residue greatly aids in seed germination, and shows that the degraded film is beneficial for seed growth. Based on the results discussed in sections 3.7 and 3.8, it can be concluded that this new material is not only biodegradable but also the products resulting from its degradation would not have a negative impact on the environment. On the contrary, they could potentially be utilized as compostable material, unlike conventional petroleum-based packaging.
4. Conclusion
Gelatin and glycerin films were obtained by incorporating dried camu-camu residues at concentrations of 0.25 g and 0.50 g. The films produced with the incorporation of camu-camu residue proved to be manageable and visually homogeneous, with the presence of some non-solubilized residues, but having a bright and well-presented appearance.
The gelatin and glycerin films incorporated with camu-camu residues, with a mass of 0.5 g, showed the best interaction between the particles and the polymeric matrix because of the presence of fibers in the waste.
Through the experimental planning and subjective analysis used in the preparation of the films, it was possible to produce films with good homogeneity, continuity, and handling.
It was observed that the film without insoluble residue had a WVP approximately 20% lower than the film with insoluble residue, indicating that the incorporation of the insoluble residue in the matrix collaborates with the passage of water vapor. This confirmed the difference in WVP of the pure gelatin matrix (35%) with samples 3 and 4, which were incorporated with camu-camu residue. This result was important for the study because, for future applications, the presence of non-solubilized residues in the film causes a slight separation in the polymeric molecule.
In the scanning electron microscopy (SEM) analysis, it was identified that there was a change in surface roughness and the formation of agglomerates with the immersion of the camu-camu residue. Due to the approximation of the polar chains that occur in the polymeric matrix, the immersion of the residue of camu-camu caused changes in the microstructure of the films.
The findings presented in this article suggest that films made of gelatin and camu-camu offer a cost-effective option with antioxidant properties that can inhibit certain reactions known to reduce the shelf life of food. This preservation of organoleptic characteristics ensures the quality of the product.
Authors’ contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were also performed by all authors. The first draft of the manuscript was written by Huéberton Barbosa Naves and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. The research and investigation process, specifically the execution of experiments, data collection, and evidence gathering, were carried out by Huéberton Barbosa Naves and by Marcia R. de Moura. Ana Paula Stafussa and Grasiele Scaramal Madrona conducted analyses of biodegradation and seeds germinated with the presence of degraded films. The development of methodology and creation of models were validated by Fabrício C. Tanaka, Grasiele Scaramal Madrona, Fauze Ahmad Aouada, and Marcia R. de Moura. The responsibility for research management and coordination was taken on by Marcia R. de Moura.
Declaration of competing interest
Nothing to declare or report.
Aknowledgements
The authors would like to thank the USP - Universidade de Sao Paulo (University of São Paulo) and São Paulo State University (UNESP), São Paulo Research Foundation (FAPESP) (2018/18697-1; 2013/03643-0; 2013/07296-2), and National Council for Scientific and Technological Development (CNPq) (MRM 315513/2021-7; FAA 312414/2018-8; 316174/2021-1; MCTIC Grant #406973/2022-9 through INCT/Polysaccharides – National Technology-Science Institute for Polysaccharides). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – “Finance Code 001.”
Declaration of generative AI in scientific writing
The authors declare that they did not use AI tools to analyze and extract insights from data as part of the research process.
References
- BETT, K.L.; INGRAM, D.A.; GRIMM, C.C.; LLOYD, S.W.; SPANIER, A.M.; MILLER, J.M.; GROSS, K.C.; BALDWIN, E.A.; VINYARD, B.T. FLAVOR OF FRESH-CUT GALA APPLES IN BARRIER FILM PACKAGING AS AFFECTED BY STORAGE TIME. J Food Qual 2001, 24, 141–156. [Google Scholar] [CrossRef]
- Nazmi, N.N.; Isa, M.I.N.; Sarbon, N.M. Preparation and Characterization of Chicken Skin Gelatin/CMC Composite Film as Compared to Bovine Gelatin Film. Food Biosci 2017, 19, 149–155. [Google Scholar] [CrossRef]
- Chandika, P.; Ko, S.-C.; Jung, W.-K. Marine-Derived Biological Macromolecule-Based Biomaterials for Wound Healing and Skin Tissue Regeneration. Int J Biol Macromol 2015, 77, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Liao, N.; Unnithan, A.R.; Joshi, M.K.; Tiwari, A.P.; Hong, S.T.; Park, C.-H.; Kim, C.S. Electrospun Bioactive Poly (ɛ-Caprolactone)–Cellulose Acetate–Dextran Antibacterial Composite Mats for Wound Dressing Applications. Colloids Surf A Physicochem Eng Asp 2015, 469, 194–201. [Google Scholar] [CrossRef]
- Momoh, F.U.; Boateng, J.S.; Richardson, S.C.W.; Chowdhry, B.Z.; Mitchell, J.C. Development and Functional Characterization of Alginate Dressing as Potential Protein Delivery System for Wound Healing. Int J Biol Macromol 2015, 81, 137–150. [Google Scholar] [CrossRef] [PubMed]
- V, A.K.; Hasan, M.; Mangaraj, S.; M, P.; Verma, D.K.; Srivastav, P.P. Trends in Edible Packaging Films and Its Prospective Future in Food: A Review. Applied Food Research 2022, 2. [Google Scholar] [CrossRef]
- Zahran, M.K.; Ahmed, H.B.; El-Rafie, M.H. Surface Modification of Cotton Fabrics for Antibacterial Application by Coating with AgNPs–Alginate Composite. Carbohydr Polym 2014, 108, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Eweis, M.; Elkholy, S.S.; Elsabee, M.Z. Antifungal Efficacy of Chitosan and Its Thiourea Derivatives upon the Growth of Some Sugar-Beet Pathogens. Int J Biol Macromol 2006, 38, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Forato, L.A.; Yushmanov, V.E.; Colnago, L.A. Interaction of Two Prolamins with 1- 13 C Oleic Acid by 13 C NMR. Biochemistry 2004, 43, 7121–7126. [Google Scholar] [CrossRef]
- de Brito Nogueira, T.B.; da Silva, T.P.M.; de Araújo Luiz, D.; de Andrade, C.J.; de Andrade, L.M.; Ferreira, M.S.L.; Fai, A.E.C. Fruits and Vegetable-Processing Waste: A Case Study in Two Markets at Rio de Janeiro, RJ, Brazil. Environmental Science and Pollution Research 2020, 27, 18530–18540. [Google Scholar] [CrossRef]
- Rangaraj, V.M.; Rambabu, K.; Banat, F.; Mittal, V. Effect of Date Fruit Waste Extract as an Antioxidant Additive on the Properties of Active Gelatin Films. Food Chem 2021, 355. [Google Scholar] [CrossRef]
- da Nóbrega Santos, E.; Cesar de Albuquerque Sousa, T.; Cassiano de Santana Neto, D.; Brandão Grisi, C.V.; Cardoso da Silva Ferreira, V.; Pereira da Silva, F.A. Edible Active Film Based on Gelatin and Malpighia Emarginata Waste Extract to Inhibit Lipid and Protein Oxidation in Beef Patties. LWT 2022, 154. [Google Scholar] [CrossRef]
- REVISTA BRASILEIRA DE FRUTICULTURA A CULTURA DE CAMU-CAMU NO BRASIL.
- Ju, A.; Song, K. Bin Development of Teff Starch Films Containing Camu-Camu (Myrciaria Dubia Mc. Vaugh) Extract as an Antioxidant Packaging Material. Ind Crops Prod 2019, 141. [Google Scholar] [CrossRef]
- Nedorostova, L.; Kloucek, P.; Kokoska, L.; Stolcova, M.; Pulkrabek, J. Antimicrobial Properties of Selected Essential Oils in Vapour Phase against Foodborne Bacteria. Food Control 2009, 20, 157–160. [Google Scholar] [CrossRef]
- Mariod, A.; Fadul, H. Review: Gelatin, Source, Extraction and Industrial Applications. ACTA Scientiarum Polonorum Technol. Aliment. 2013, 12, 135–147. [Google Scholar]
- Qiao, C.; Wang, X.; Zhang, J.; Yao, J. Influence of Salts in the Hofmeister Series on the Physical Gelation Behavior of Gelatin in Aqueous Solutions. Food Hydrocoll 2021, 110, 106150. [Google Scholar] [CrossRef]
- Nunes, J.C.; Melo, P.T.S.; Lorevice, M.V.; Aouada, F.A.; de Moura, M.R. Effect of Green Tea Extract on Gelatin-Based Films Incorporated with Lemon Essential Oil. J Food Sci Technol 2021, 58, 1–8. [Google Scholar] [CrossRef]
- Saranti, T.F. da S.; Melo, P.T.S.; Cerqueira, M.A.; Aouada, F.A.; de Moura, M.R. Performance of Gelatin Films Reinforced with Cloisite Na+ and Black Pepper Essential Oil Loaded Nanoemulsion. Polymers (Basel) 2021, 13, 4298. [Google Scholar] [CrossRef] [PubMed]
- Vital, A.C.P.; Guerrero, A.; Monteschio, J. de O.; Valero, M.V.; Carvalho, C.B.; de Abreu Filho, B.A.; Madrona, G.S.; do Prado, I.N. Effect of Edible and Active Coating (with Rosemary and Oregano Essential Oils) on Beef Characteristics and Consumer Acceptability. In PLOS ONE; Chamani, J., Ed.; 2016; Vol. 11, p. e0160535.
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am J Enol Vitic 1965, 16, 144–158. [Google Scholar] [CrossRef]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Hawkins Byrne, D. Comparison of ABTS, DPPH, FRAP, and ORAC Assays for Estimating Antioxidant Activity from Guava Fruit Extracts. Journal of Food Composition and Analysis 2006, 19, 669–675. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant Activity Applying an Improved ABTS Radical Cation Decolorization Assay. Free Radic Biol Med 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal Biochem 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
-
ASTM - Standard Test Method for Water Vapor Transmission of Materials; PA: ASTM: Philadelphia, USA, 1980.
- McHUGH, T.H.; AVENA-BUSTILLOS, R.; KROCHTA, J.M. Hydrophilic Edible Films: Modified Procedure for Water Vapor Permeability and Explanation of Thickness Effects. J Food Sci 1993, 58, 899–903. [Google Scholar] [CrossRef]
- Pavoni, J.M.F.; Luchese, C.L.; Tessaro, I.C. Impact of Acid Type for Chitosan Dissolution on the Characteristics and Biodegradability of Cornstarch/Chitosan Based Films. Int J Biol Macromol 2019, 138, 693–703. [Google Scholar] [CrossRef]
- Abe, M.M.; Branciforti, M.C.; Nallin Montagnolli, R.; Marin Morales, M.A.; Jacobus, A.P.; Brienzo, M. Production and Assessment of the Biodegradation and Ecotoxicity of Xylan- and Starch-Based Bioplastics. Chemosphere 2022, 287, 132290. [Google Scholar] [CrossRef] [PubMed]
- Harada, J.; de Souza, A.G.; de Macedo, J.R.N.; Rosa, D.S. Soil Culture: Influence of Different Natural Fillers Incorporated in Biodegradable Mulching Film. J Mol Liq 2019, 273, 33–36. [Google Scholar] [CrossRef]
- Porto, D.S.; de Faria, C.M.G.; Inada, N.M.; Frollini, E. Polyurethane Films Formation from Microcrystalline Cellulose as a Polyol and Cellulose Nanocrystals as Additive: Reactions Favored by the Low Viscosity of the Source of Isocyanate Groups Used. Int J Biol Macromol 2023, 236, 124035. [Google Scholar] [CrossRef]
- Rezende Barbosa TURBIANI, F.; Guenter KIECKBUSCH, T. Propriedades Mecânicas e de Barreira de Filmes de Alginato de Sódio Reticulados Com Benzoato de Cálcio e/Ou Cloreto de Cálcio. Brazilian Journal of Food Technology 2011, 14, 82–90. [Google Scholar] [CrossRef]
- de Andrade, R.M.S. BIOACESSIBILIDADE, CAPACIDADE ANTIOXIDANTE E POTENCIAL PREBIÓTICO DE SUBPRODUTO DE FRUTAS E HORTALIÇAS. Doctoral thesis, UNIVERSIDADE FEDERAL DO ESTADO DO RIO DE JANEIRO: Rio de Janeiro, 2020.
- Chambi, H.N.M.; Grosso, C.R.F. Mechanical and Water Vapor Permeability Properties of Biodegradables Films Based on Methylcellulose, Glucomannan, Pectin and Gelatin. Ciência e Tecnologia de Alimentos 2011, 31, 739–746. [Google Scholar] [CrossRef]
- Altiok, D.; Altiok, E.; Tihminlioglu, F. Physical, Antibacterial and Antioxidant Properties of Chitosan Films Incorporated with Thyme Oil for Potential Wound Healing Applications. J Mater Sci Mater Med 2010, 21, 2227–2236. [Google Scholar] [CrossRef]
- Kavoosi, G.; Rahmatollahi, A.; Mohammad Mahdi Dadfar, S.; Mohammadi Purfard, A. Effects of Essential Oil on the Water Binding Capacity, Physico-Mechanical Properties, Antioxidant and Antibacterial Activity of Gelatin Films. LWT - Food Science and Technology 2014, 57, 556–561. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).