1. Introduction
A notable uptick in world exports of major tropical fruit crops was observed in 2023, with an estimated 12% increase compared to previous years, resulting in a total value exceeding USD 11 billion [
1]. This surge marks the highest level recorded thus far, highlighting the growing demand for tropical fruit in the global market. Fruit crops constitute a significant portion of the global agricultural output, accounting for 17% of the total production value of crops in 2021 [
2]. This sector not only fulfills domestic food requirements but also sustains employment, engaging approximately 5 million workers in Brazil, and generating around 190 million jobs in China & India [
3,
4].
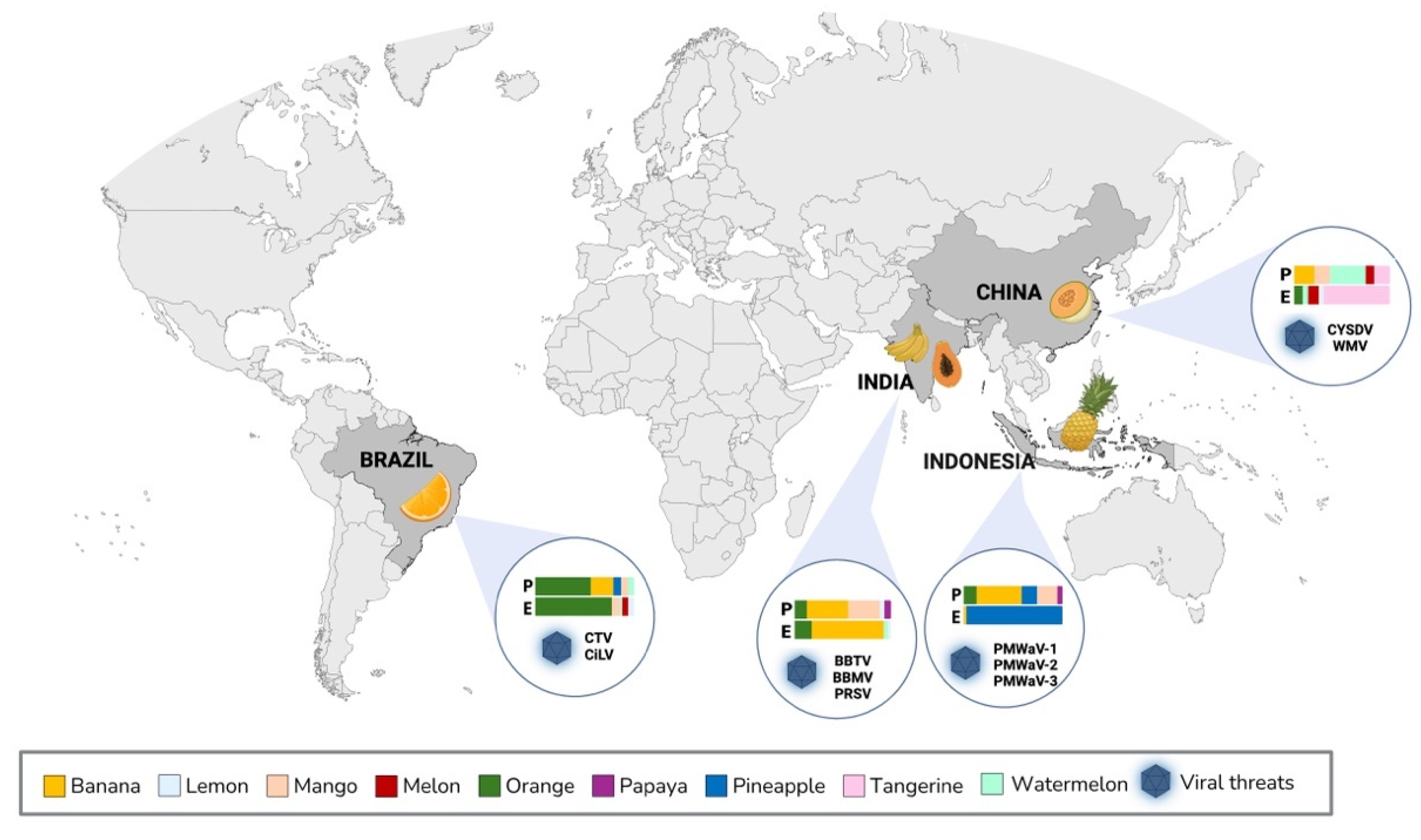
Countries in Asia and in the Americas are collectively responsible for 76.2% of global production of tropical fruit crops [
5]. The major crops cultivated worldwide are avocado, banana, citrus (orange, tangerine, lemon and lime), mango, melon, papaya, pineapple and watermelon [
1,
6,
7].
Plants viruses in tropical fruits can cause annual losses exceeding $30 billion [
8]
. Bananas, pineapple, papaya, melon and citrus appear to be particularly susceptible to viral infections, representing economic challenges for the main producing and exporting countries (
Figure 1).
Specific export projections anticipate a 4% expansion in global pineapple exports, reaching 3.2 million tonnes in 2023; conversely, papaya exports are forecasted to decrease by 3%, amounting to 365,000 tonnes [
1]. Despite an overall positive supply outlook, concerns persist regarding adverse weather events and the spread of plant diseases, particularly impacting papaya cultivation. These challenges highlight the importance of improving mitigation strategies and adopting sustainable practices to protect tropical fruit production and trade.
Most plant viruses feature RNA as their genetic material, with geminiviruses (ssDNA viruses) representing a notable exception; in fruit crops, Umer et al. reviewed that over 70% of the economically importatnt viruses have (+)ssRNA genomes [
9]. Genetic diversity relies on the constant changes occurring in DNA or RNA viral genomes, which generate new variants through mechanisms such as mutation and recombination [
10]. Fruit crops are exposed to several viral infections, and coinfections (a necessary occurrence to generate recombinants) are common in the field.
Biotechnology emerges as an essential ally to face the challenge of controlling viral diseases in the fruit sector, employing precise scientific methodologies to confer resistance, combat infections in orchards, and improve new cultivars [
11]. This review emphasizes the use of contemporary gene-editing techniques, notably CRISPR/Cas9, while providing historical insights into classical methods such as genetically modified organisms (“transgenics”). Considering the genetic diversity exhibited by viruses, we initially explore the impact of viral infections on tropical fruit crops, examine the correlation between viral modes of action and the extensive possibilities for manipulating host genomes, and conclude by presenting products which have been approved by national regulatory bodies and are available for consumers.
2. Viral Infections in Tropical Fruit Crops
Plants face constant threats from pathogens like fungi, bacteria, and viruses, resulting in an annual economic toll of around US
$220 billion; notably, viral infections alone make up half of the emerging and reemerging diseases caused by microorganisms [
12]. Until 2019, the International Committee on Taxonomy of Viruses (ICTV) had identified 1484 plant viruses, among which those with RNA genomes stand out as the most detrimental to crops [
13].
Table 1 details the tropical fruit crops highlighted in this review and their main viral diseases.
The infection process begins with the transmission of viral particles to the plant via vectors such as mites, aphids, leafhoppers, whiteflies, and scale insects. Cellular susceptibility occurs when the virus bypasses the defense-response system of plant cells [
14], which can be physical, such as cell wall and cuticle, or induced after pathogen exposure, such as R genes [
15,
16]. Viral infections cause several cell wall modifications that may lead to resistance, such as the production of endoxylanases during potyvirus attack, which loosens cell wall structure [
17]. Each crop differs in terms of symptomatic response, and in some crops, such as pineapple and papaya, the diagnosis of a single disease is challenging due to the existence of viral complexes.
2.1. Banana
Banana bract mosaic virus (BBrMV) is transmitted through suckers and aphids vectors, and constitute a major problem in southern India, Philippines, Sri Lanka and Hawaii [
18]. BBrMV encodes the three proteases typical of potyviruses, named P1, HC-Pro and NIa-Pro. HC-Pro is a major RNA silencing suppressor protein, acting as a pathogenicity factor in most potyvirus infections. It has also been suggested by in silico analysis that BBrMV may encode miRNAs, which could affect the expression of host genes, but this requires experimental validation [
19].
Banana bunchy top virus (BBTV), also transmitted by aphids, causes extensive losses in banana crops which, if not controlled, can reach 70% of the production [
20]. This virus encodes three proteins, CP, MP and Clink, that act as RNA silencing suppressors, and another designated NSP, a virulence factor that blocks the host’s transmembrane receptor kinase activity [
21,
22]. After being challenged by BBTV infection, transcriptomic analysis revealed differentially expressed genes (DEG) of wild susceptible
Musa balbisiana and a resistant phenotype [
23]. They shared a similar pattern of expression of 62 DEGs, 151 were exclusively to susceptible cultivar and 99 belonged to resistant
M. balbisiana. The profiles of DEGs were also different. The host’s protein machinery was upregulated in the wild cultivar, such as small ribosomal subunit 40S, translation elongation factor (eEF1A) and eukaryotic translation initiation factor (eIF5A); also, RNA polymerase sigma factor 70 and phosphorolytic exoribonuclease PNP were upregulated, highlighting the essential role of these proteins to the pathogen. On the other hand, the study showed that the resistant cultivar expressed a set of different genes, such as the up and downregulation of specific protein kinases, downregulation of phytohormones – auxin efflux carrier component 1a and abscisic acid signaling, and changes in reactive oxygen species (ROS) and secondary metabolites production levels. These findings are crucial for understanding the viral mechanisms, which can lead to new disease control strategies.
In banana crops, bunchy top and streak disease (caused by banana streak virus – BSV) play an important economic role, reducing its yield and productivity [
24]. BSV transmission occurs by mealybugs, but its spread through crops also occurs via endosomal BSV (eBSV) activation. eBSV is incorporated into the
M. balbisiana (B) genome, and therefore is present in hybrids containing the B genome (eg, AAB). Although
M. balbisiana is resistant to infection, AAB hybrids are susceptible [
25]. Under stress conditions, the viral replication pathway is activated and the new infective BSV particles are able to avoid DNA cytosine methylation and transcriptional gene silencing, even after facing accumulation of siRNAs produced by the defense-response mechanism; this is supposed to explain why eBSV persist in its integrated form in plants [
24,
26], a challenge to biotechnological approaches.
2.2. Citrus
Citrus leprosis virus C (CiLV-C) infects orange, grapefruit, and tangerines, and the infection is restricted to the area where the vector (mites of the genus
Brevipalpus) attacks, an intriguing process that relies on the production of reactive oxygen species by the host, activating a hypersensible response [
27]. Arena et al. described that 4 hours after the beginning of infection, the replication rate is increased, and after 6 hours occurs a reprogramming of citrus transcription, with downregulation of the jasmonate/ethylene (JA/ET) pathways, which is involved in anti-herbivory defense response [
28]. The authors suggest that this could be a viral strategy to increase the vector’s fitness, and it may be a future target for gene edited-based modifications.
Citrus tristeza virus (CTV), transmitted by aphids (eg,
Toxoptera citricida), has the one of the largest RNA genomes in plants, comprising 12 open reading frames (ORFs) in 19.3 kb [
29]. At the 5’ proximal end, ORF1a encodes a 349-kDa polyprotein, and eventually the translation continues to the ORF1b. At the 3’ proximal region, subgenomic RNAs (sgRNAs) direct the translation of viral proteins responsible for the assembly of virus particles, virus movement and RNA silecing suppression [
30]. CTV encodes three RNA silencing suppressors, p20, p23 and the CP, each one with a distinct mode of action. p23 is a local (intracellular) suppressor, while p20 suppresses both local and systemic silencing. The CP is also a systemic (intercellular) supressor but does not act locally, which is a distinct mode of action from all other known viral suppressor proteins [
31]. Collectively, the three CTV suppressors interfere with multiple points of the RNA silencing pathway, which may be necessary for a virus with such a long RNA genome and which causes long-term, persistent infections in perennial hosts. In addition to its repertoire of RNA silencing suppressors, CTV also produces a non-conding sgRNAs named low-molecular-weight tristeza 1 (LMT1), which down-regulates the salicylic acid-based defense response, the only know case of a long, non-coding viral RNA that interferes with plant defense [
32].
The citrus psorosis disease is widely spread in North and South America, specially Argentina and Uruguay [
33]. Citrus psorosis virus (CPsV) is transmitted by vegetative propagation, and the bark symptoms are frequently observed in sweet orange, mandarin and grapefruit [
34]. Belabess et al. reported that CPsV downregulates the RNAi machinery in
Citrus sinensis and upregulated gene
Scarecrow-like 6 (SCL6), activating programmed cell death and reducing chlorophyll synthesis [
35]. The 24K viral protein, encoded by ORF1 of RNA1, interacts with the miRNA synthesys machinery and negatively affects their accumulation, altering the expression of their targets [
36].
2.3. Pineapple
Mealybug wilt of pineapple (MWP) is described as the most significant viral disease of pineapple. It is caused by a complex composed of three viruses: pineapple mealybug-associated virus 1 (PMWaV-1), pineapple mealybug-associated virus 2 (PMWaV-2), and pineapple mealybug-associated virus 3 (PMWaV-3). Dey et al. report that the etiology of the disease, regarding symptomatic expression, depends on the interaction of ants with scale insects (vectors of the virus) and also on the pineapple genotype [
37]. Furthermore, Green et al. state that only pineapples infested by
Dysmicoccus brevipes and
D. neobrevipes and infected by PMWaV-2 develop typical symptoms, such as root drying, wilting, and gradual leaf discoloration; PMWaV-2 is responsible for encoding RNA silencing suppressor proteins, which similarly to those encoded by CTV act locally and systemically to prevent the degradation of viral mRNAs by the host’s RNA silencing machinery [
38].
2.4. Papaya
Viral infections affecting papaya constitute a phytosanitary and economic problem for growers, who often resort to roguing, an effective control measures by removing the infected plants. Delayed implementation of roguing can result in losses of up to 100% in the state of Espírito Santo, the main papaya-growing region of Brazil [
39]. Papaya ringspot virus papaya-strain (PRSV-p, a potyvirus) is the etiological agent of the most devastating disease of papaya crop, named ringspot/mosaic disease, since the symptoms constitute the appearance of a mosaic in leaf tissues, and a yellow-circular spot on the fruits [
40,
41]. During potyviruses infection, the host defense response and innate immune system are activated [
42], and specifically to PRSV, a protein interaction between viral coat protein (CP) and papaya identified 23 proteins grouped in four groups, as: transcription factors and cell division; hormones and stress; mitochondrial electron transferase and respiration; and proteasome [
43].
Papaya sticky disease (PSD) is caused by a viral complex including papaya meleira virus 1 (PMeV-1), and papaya meleira virus 2 (PMeV-2). Symptoms include reduced latex viscosity with subsequent aqueous latex exudation, causing the fruit to become “sticky”, and is often followed by necrosis of leaf tips [
44]. Interestingly, symptom expression is dependent on the plant’s developmental stage, occurring only after flowering. PSD is present in Brazil [
45], Mexico [
46], Ecuador [
47] and Australia [
48], and its management imposes additional challenges to growers.
The interactions between the PMeV complex and papaya plants have not been characterized in detail, but it is known that the viruses are located primarily, if not solely, in cells of the laticiferous vessels, responsible for storing latex and having a predominance of proteases. In an attempt to escape the viral attack, papaya cells activate defense mechanisms before the flowering period, altering the expression of genes related to the synthesis of growth regulators and encoding proteins related to the chloroplast [
49]. The study also demonstrated that two regions of the putative CP ORF of PMeV-1, CP2 and CP4, interact with ribosomal proteins, specifically the 50S ribosomal protein L17 (RPL17). RPL17 is downregulated during the pre-flowering period, which is consistent with the above-mentioned symptom expression occurring only after flowering
2.5. Melon
The Cucurbitaceae family includes a wide range of crop plants, such as cucumber, squash, melon and watermelon, and an important virus disease of the group is caused by the potyvirus watermelon mosaic virus (WMV). WMV infects more than 170 plant species, and in
Cucumis melo (melon), a mixed infection, with the phloem-limited cucurbit yellow stunting disorder virus (CYSDV), is common. An extensive work by Domingo-Calap et al. studied the mixed infection with WMV and CYSDV [
50,
51]. CYSDV had higher titers during the first 60 days of infection, while WMV had a lower concentration; however, a positive interaction was observed as aphid vector displayed a –preference– by plants with the two viruses over those with a single infection by WMV. At the molecular level, the authors identified relevant interactions among four proteins: WMV P1 HC-Pro (with P1 modulating HC-Pro silecing suppressing activity), and CYSDV P22-P25 (P22 modulating P25 suppressor activity). When a mixed infection occurs, P22 does not influence HCPro functions, but P1 has a negative effect on P25, reducing its suppressor activity in a dose-dependent fashion.
Looking to obtaining GM, WMV-resistant cultivars, the
rwm1 gene was reported as a recessive resistance gene for WMV in
Arabidopsis [
52]. Expression of the dominant alele, causes the plant to be susceptible to viral infection by interaction of the
rwm1 product (the chloroplast-encoded phosphoglycerate kinase cPGK2) with the WMV VPg protein, but if a single amino acid substitution occurs in the cPGK2 protein, the interaction no longer occurs and WMV infection is prevented. This was the first case of recessive resistant to a potyvirus where the product of the resistance gene is not an isoform of the translation initiation factors eIF4E or eIF4F, and raised new possibilities of successfully engineering resistance to potyviruses using a host-encoded gene.
Melons can also be infected by two other potyviruses, zucchini yellow mosaic virus (ZYMV) and papaya ringspot virus – watermelon (PRSV-w). PRSV-w is closely related to PRSV-p, but is able to infect only cucurbits. PRSV-w can be transmitted by at least 32 aphids species and the disease development constitutes a significant factor for crop productivity [
53,
54]. The PRSV-w cylindrical inclusion protein interacts with the NBS2 domain of the Prv protein, a typical TIR-NLR resistace (R) protein. This interaction is hypothesized to be essential for virus infection in melon [
55].
3. Management Strategies
Plants are constantly exposed to infection by plant viruses and due to the obligatory intracellular lifestyle of these pathogens, control methods must be based on risk-reducing and preventive measures [
56]. Viral infectious processes are established through the complete development of a replication cycle within the host cell and the production of new infectious progeny. Control strategies can be of chemical, physical, biological or genetic nature.
Because insects act as vectors of several plant viruses, chemical control of the vector is often an effective way of managing virus diseases. However, insecticides cause environmental harm, generate residues in crops, and their long-term use can select for resistant vector populations [
57]. Moreover, chemical control of the insect vector is ineffective to control nonpersistently-transmitted plant viruses. Physical strategies include roguing (removal of the entire symptomatic plant) or pruning (selective removal of symptomatic portions of the plant). These methods are common in some crops, such as passion fruit, in which roguing is used to control cowpea aphid-borne mosaic virus [
58], papaya, in which it is used to control PRSV and PMeV [
59,
60], and banana, in which it is a preferred technique to control BBTV [
61].
Genetic modifications came to solve problems that the methods above could not: identify, at the molecular level, how the host cell interacts with the invader agent, and how to interfere with this interaction to block the viral infection or improve the defense response. Genetically modified organisms (GMOs) were developed that brought important benefits for agriculture, such as new virus-resistant varieties of tomato [
62], banana [
63], apple [
64] and pineapple [
65]. The recently developed gene editing technology (GEd) has already generated virus-resistant varieties of tomato [
66], banana [
67], apple [
68], orange [
69] and melon [
70].
4. A success Story: Genetic Engineering of Papaya for Resistance to Papaya Ringspot Virus (PRSV-p)
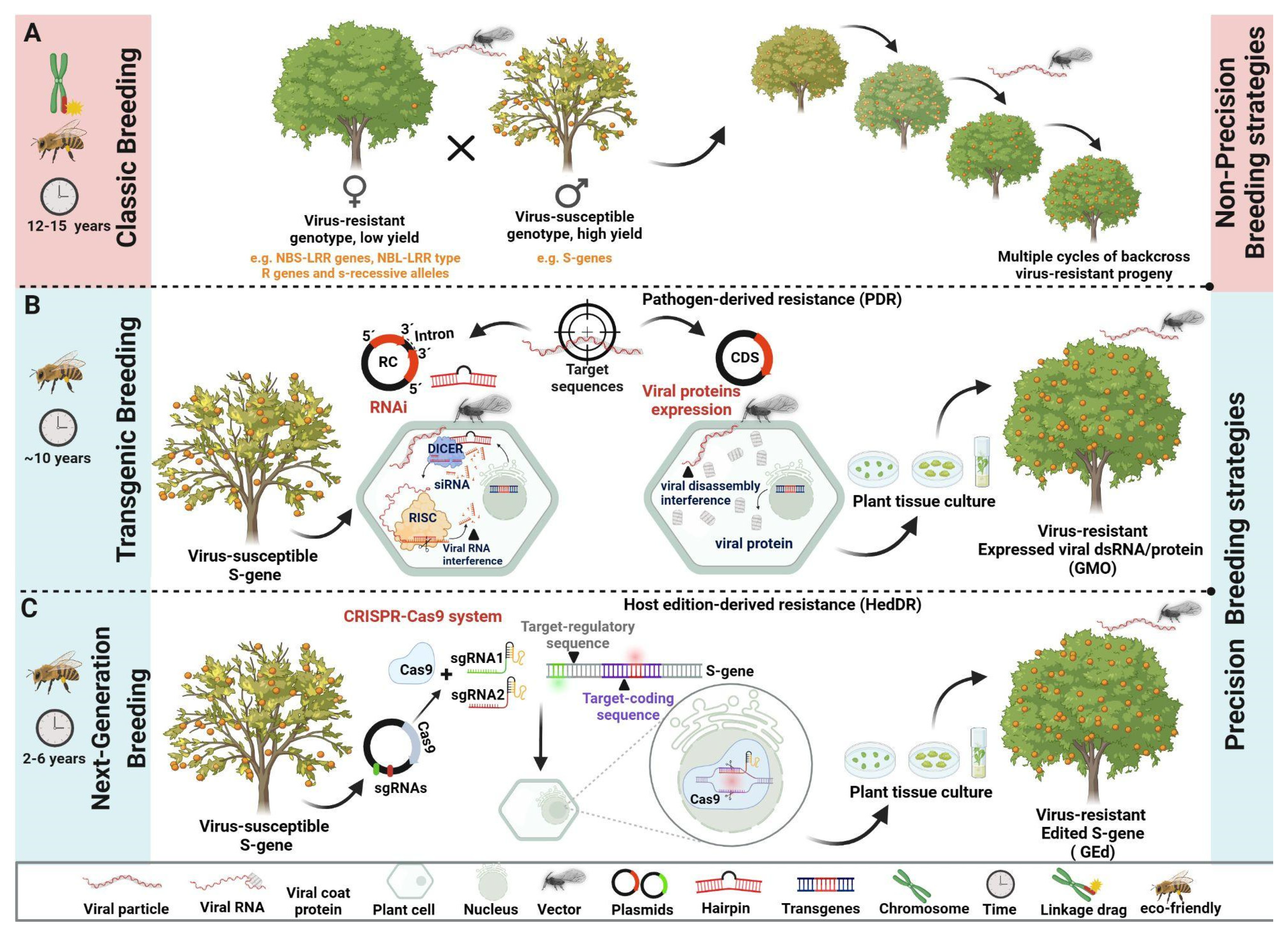
The production of virus-resistant genetically-modified (GM) plants was one of the greatest achievements of plant biotechnology in the late 20th century, and consitutes a virtually assured way of controling viral diseases in both annual and perennial crops. The main strategy for generating such plants is the expression of a non-coding, virus-derived RNA which will form a hairpin and activate the RNA silencing machinery in advance of the arrival of the virus. One of the first crops to be engineered was a tropical fruit crop, papaya, for resistance to PRSV-p.
The two GM cultivars of papaya were obtained, named “SunUp” and “Rainbow”, the first engineered using an untranslatable version of the PRSV-p HA 5-1 coat protein gene, and the latter a hybrid form “SunUp” and “Kahopo” [
71]. Planting of GM papaya started in 1998 in the Puna district of the island of Hawaii, and saved the papaya industry in that region. According to Gonsalves
et al., initially 76% of farmers in the Puna district grew transgenic seeds, and the next year, 56% of papaya fields were transgenic [
72]. Despite the great results after a field trial, the authors noticed that “SunUp” was susceptible to PRSV-p strains from outside Hawaii, and that resistance in “Rainbow” was age-dependent for Hawaiian PRSV-p, mainly found in plants that were younger than 8 weeks. Even facing this problem, “Rainbow” had its commercial market ranging from 70% of the Hawaiian papaya industry, and evolved from 32% in 2001 to 77% of the market in 2009 [
73]. Presently, more than 90% of the papaya production in Hawaii is GM, with no breakdown in resistance and no harmful environmental effects.
Successful control of papayaa ringspot using GM papaya also occurred in China. The transgenic construct was designed to provide resistance against the four PRSV-p variants identified in China, named Ys, Vb, Sm, and Lc, and was based on an untranslatable version of the Ys NIb (replicase) gene, which is more conserved than the CP gene [
74]. In 1998, the Huanong No. 1 GM papaya was obtained and its commercial release was approved in 2006. Large scale planting of Huanong No. 1 confirmed resistance to the four variants, and by 2010, 85% of the cultivated area was comprised of the GM cultivar.
5. Disease-Resistant Crops: Research Trends in Tropical Fruits Virology
In agricultural practice, virus-resistant cultivars offer both economic and social advantages by significantly reducing losses caused by viral diseases without harmful environmental effects. Recent advances in plant biotechnology allow for direct, precise modifications to the organisms’ genome, such as the introduction, removal, or editing of specific genes, significantly accelerating and efficiently obtaining desirable traits in crops [
75]. Techniques such as heterologous expression, RNA interference (RNAi), and gene editing through the clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein (CRISPR/Cas) system enable genome alteration within the same species and facilitate genetic material exchange among different species, expanding the range of genes available for use.
Plant resistance mediated by genetic engineering follows two main pathways. In pathogen-derived resistance (PDR) part of the viral genome is inserted into the host plant to induce a resistance response [
76]. This is the strategy used in the GM papaya, plants expressing the PRSV-p coat protein commercialized in Hawaii and China [
77].
It is also possible to utilize characteristics of the host genome itself to induce a resistance phenotype, denominated as heterologous organism derived resistance (HoDR). The eukaryotic translation initiation factors constitute an important susceptibility/resistance pathway in several crops facing potyvirus infection, specially the eIF4E family.
eIF4E expression is upregulated during the soybean mosaic virus (SMV) and WMV in soybean crops, but after the knot-out of the gene by RNAi, plants showed resistance phenotype [
78]. The CRISPR/Cas9 approach was used to silence the same gene to confer resistance to potyviruses in tomato [
79] and potato [
80]. Studies encompassing the use of the host genome as a source of resistance are predominant in identifying susceptible genes, thus facilitating gene modification/editing and promoting the resistance phenotype. However, with the discovery of CRISPR/Cas9, this landscape is changing in current research.
Currently, basic studies such as functional genomics are capable of indicating regions in the genome or target genes that assist in the production of genetically-modified plants [
81]. Techniques such as knockout of susceptible genes [
82], knockin of genes conferring resistance to stresses [
83], and alteration of gene expression regulation [
84] are highly effective (
Figure 2). Regardless of the strategy used, whether it is pathogen-derived or host-derived resistance, targeted genetic modifications and editing mediated by biotechnological tools hold promising prospects for combating viral disease (as well as other biotic stresses) in plants.
5.1. Pathogen-Derived Resistance by RNA Silencing
RNA interference (RNAi)-mediated post-transcriptional gene silencing (PTGS), or RNA silencing, refers to an antiviral defense response based on small (20-24 nt) RNA molecules, and involving Dicer-like (DCL), Argonaute (AGO), and RNA-dependent RNA polymerase (RDR) proteins [
85]. DCL proteins (type III RNases) process double-stranded RNA (dsRNA) into small interfering RNA (siRNA) molecules. These siRNAs are then integrated into AGO endonucleases, forming the RNA-induced silencing complex (RISC). The RISC complex is guided by siRNAs to selectively bind to target mRNA molecules, cleaving the target mRNA and thus, suppressing protein production. Additionally, the cleaved target RNA can be recognized by RNA-dependent RNA (RDR) proteins, responsible for amplifying dsRNA, enhancing the gene silencing effect. RDRs significantly increase the efficiency of the RNAi mechanism and contribute to more robust gene silencing [
86,
87].
Plant-infecting viral pathogens exhibit a wide diversity of genomes, including different types of genetic material such as single-stranded RNA (ssRNA), double-stranded RNA (dsRNA), single-stranded DNA (ssDNA), or double-stranded DNA (dsDNA) [
88,
89]. In RNA viruses, viral-derived siRNAs (vsiRNAs) are produced from DCL-mediated cleavage of dsRNA replication intermediates. In DNA viruses, siRNAs are produced by RNA-dependent RNA (Pol II), responsible for copying regions of the viral DNA on both strands (sense and antisense), forming a perfect double-stranded RNA molecule to be cleaved by DCLs [
90].
Plants engineered with viral-derived DNA sequences containing inverted repeats will express dsRNAs, which will be targeted by the RNA silencing machinery to produce vsiRNAs [
91]. This has been the primary strategy to obtain virus-resistant GM plants. However, plant viruses encode RNA silencing suppressor proteins (VSRs) to bypass the siRNA-based antiviral defense response. These proteins can inhibit siRNA production, capture siRNAs, or block the spread of RNA silencing signals [
92]. These evolutionary strategies allow viruses to circumvent plant immune responses and successfully establish infection. Therefore, the constant refinement of technologies for engineering virus resistance in plants is essential for successful combat against biotic stresses of viral origin in agricultural crops.
By exploring proteins responsible for viral replication (Rep), transgenic banana plants fully resistant to banana bunchy top virus (BBTV) were developed by Shekhawat et al. using RNA silencing [
93]. In addition to the previously mentioned GM papayas expressing the PRVV-p coat protein, Kung et al. generated transgenic papaya lines resistant to two viruses, PRSV-p and papaya leaf distortion mosaic virus (PLDMV) [
94]. Although other proteins can be used to induce resistance, the predominant strategy is based on expressing the viral CPs proteins [
95].
Although PDR based on RNA silencing is falling out of favour due to the rise of CRISP-Cas, this technology remains a viable option to generate virus resistant plants due to its efficiency, durability, increasingly simplified production and cost reduction [
96]. Significant efforts are being devoted to expanding and optimizing the arsenal of RNA silencing-based tools for topical applications, including dsRNAs, synthetic microRNAs (amiRNAs), non-coding RNAs (tasiRNAs), and miRNA mimics [
97].
5.2. CRISPR/Cas
The CRISPR/Cas technology is a modern biotechnological tool that employs nucleases (Cas proteins) guided by RNA to alter specific sequences in the genome with high precision [
98], and which has emerged as one of the most prominent approaches due to its simplicity, effectiveness, flexibility, accuracy, and multiplexing capability [
99]. Based on the adaptive immune system of
Streptococcus pyogenes, the Cas protein forms a complex by binding to CRISPR RNA (crRNA) and trans-activating crRNA (tracrRNA) molecules, which when fused for biotechnological purposes is termed single guide RNA (sgRNA) [
100].
This complex identifies specific genome locations which are complementary to the 5’ leader sequence of the crRNA by recognizing the PAM (protospacer adjacent motif) sequence and initiates editing upstream of the sequence [
101] after forming an RNA/DNA heteroduplex structure between the crRNA and the host DNA strand [
102]. The PAM sequence may vary depending on the origin of the Cas protein used in the system. In the case of the
Streptococcus pyogenes Cas9 protein, the sequence is NGG or NAG [
103]. Gene editing involves insertions and deletions (indels) or substitutions of nucleotides, promoted by repair processes after double-strand breaks (DSBs). DSB repair can be mediated by homology-directed repair (HDR) or by non-homologous end joining repair (NHEJ) [
104].
Compared to RNAi, CRISPR/Cas technology offers distinct advantages in engineering virus resistance in plants. Instead of post-transcriptional gene silencing, it enables targeting susceptible genes necessary for viral spread or the manipulation of active sites of gene expression [
105]. Among all biotechnological methods involved in plant transformation, the gene editing method via CRISPR/Cas has significantly increased in use in recent years.
Several studies have demonstrated the effectiveness of the CRISPR/Cas9 system in conferring resistance to DNA and RNA viruses in plants [
106,
107,
108]. Furthermore, the CRISPR/Cas method allows for the exclusion of exogenous DNA persistence in the plant transformation process.
New research covering the various applications of the CRISPR/Cas9 methodology will be essential for managing viral diseases in tropical fruit crops. The CRISPR/Cas9 system has already been applied in fruit crops grown in tropical and subtropical regions, such as banana [
109,
110,
111], orange [
112,
113], melon [
114,
115], apple [
116,
117], and papaya [
118]. However, studies on some major tropical fruits crops are either scarce (papaya) or non-existant (pineapple). To mitigate the effects of viral diseases, these versatile methodologies are adopted in both basic [
119,
120] and applied [
121,
122] research. But even with these gaps, CRISPR/Cas is quickly becoming a powerful tool in the development of transgenic and non-transgenic plant varieties resistant to viral diseases, preventing agricultural production loss and aiding food security.
6. Conclusions and Future Perspectives
The investigation into plant viral diseases is crucial given their substantial impact on agricultural production, including tropical fruit crops. Genetic modification is an indispensable tool in the development of virus-resistant plants by targeting specific regions critical for viral infection. Nevertheless, recent research emphasizes how viral evolution can surpass these modifications, presenting persistent challenges to biotechnological endeavors [
123,
124].
Future research should focus on exploring alternative genetic targets for modification, integrating multiple resistance mechanisms, and leveraging emerging biotechnological tools such as CRISPR-based technologies. Additionally, there is a need for increased surveillance and monitoring of viral diversity in tropical fruit crops to anticipate and mitigate the emergence of new viral variants.
Ultimately, by advancing our understanding of viral biology and genomics and leveraging innovative biotechnological solutions, we can mitigate the impact of plant viral diseases on global food security and ensure the sustainable production of tropical fruit crops.
Author Contributions
Conceptualization, P.M.B.F., F.M.Z., M.S.V. and A.S.X.; Literature Review and Writing—Original Draft Preparation, M.S.V. and R.L.R.C.; Writing—Review and Editing, P.M.B.F., A.S.X., L.F. and L.S.M. Figures Conceptualization, L.F., A.S.X., L.S.M., R.L.R.C. Figures development, L.F., L.S.M. and A.S.X.. Funding acquisition, P.M.B.F. All authors have read and agreed to the published version of the manuscript.
Funding
Grants were awarded from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) to LSM (research program to support the permanence of young scientists in Brazil #150429/2023-1), to FMZ and PMBF (research productivity #312493/2018-5 and 308306/2021-0, respectively). MSV acknowledges the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for her PhD scholarship (grant #88887.920990/2023-00). LFS and RLRC acknowledges the Fundação de Amparo à Pesquisa do Espírito Santo (FAPES) for her postdoctoral scholarship (grant #725/2022) and his PhD scholarship (grant #299/2023), respectively.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created in this study.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Food and Agriculture Organization of the United Nations. Major Tropical Fruits Market Review – Preliminary Results 2023. 2024. Rome, Italy.
- Food and Agriculture Organization of the United Nations. Fruit and vegetables – your dietary essentials. The International Year of Fruits and Vegetables. 2021. Rome, Italy.
- Associação Brasileira dos Produtores Exportadores de Frutas e Derivados. In A Revolução da Fruticultura Sustentável: Tendências e Inovações. 2023.
- Food and Agriculture Organization of the United Nations. World Food and Agriculture – Statistical Yearbook 2022. 2022. Rome, Italy.
- Food and Agriculture Organization of the United Nations. Agricultural Production Statistics 2020-2022. FAOSTAT ANALYTICAL BRIEF 2022, 79.
- Food and Agriculture Organization of the United Nations. Banana Market Review – Preliminary results 2023. 2023. Rome, Italy.
- Food and Agriculture Organization of the United Nations. Citrus Fruit Statistical Compendium 2020. 2021. Rome, Italy.
- Nicaise, V. Crop immunity against viruses: outcomes and future challenges. Front. Plant Sci. 2014, 5, 660. [CrossRef]
- Umer, M.; Liu, J.; You, H.; Xu, C.; Dong, K.; Luo, N.; Kong, L.; Li, X.; Hong, N.; Wang, G.; et al. Genomic, Morphological and Biological Traits of the Viruses Infecting Major Fruit Trees. Viruses 2019, 11, 515. [CrossRef]
- Ellegren, H.; Galtier, N. Determinants of Genetic Diversity. Nat. Rev. Genet. 2016 177 2016, 17 (7), 422–433.
- Fernandes, P.M.B.; Favaratto, L.; Fernandes, A.A.R.; Vicien, C.; Capalbo, D.M.F.; Zerbini, F.M. To become more sustainable organic agriculture needs genome editing technology. Front. Bioeng. Biotechnol. 2022, 10, 912793. [CrossRef]
- Jones, R.A.; Naidu, R.A. Global Dimensions of Plant Virus Diseases: Current Status and Future Perspectives. Annu. Rev. Virol. 2019, 6, 387–409. [CrossRef]
- He, S.; Creasey Krainer, K. M. Pandemics of People and Plants: Which Is the Greater Threat to Food Security? Mol. Plant 2020, 13 (7), 933–934.
- Jin, X.; Cao, X.; Wang, X.; Jiang, J.; Wan, J.; Laliberté, J.-F.; Zhang, Y. Three-Dimensional Architecture and Biogenesis of Membrane Structures Associated with Plant Virus Replication. Front. Plant Sci. 2018, 9, 57. [CrossRef]
- Underwood, W. The Plant Cell Wall: A Dynamic Barrier Against Pathogen Invasion. Front. Plant Sci. 2012, 3, 85. [CrossRef]
- Sett, S.; Prasad, A.; Prasad, M. Resistance genes on the verge of plant-virus interaction. Trends Plant Sci. 2022, 27, 1242–1252. [CrossRef]
- Koziel, E.; Otulak-Koziel, K.; Bujarski, J.J. Plant Cell Wall as a Key Player During Resistant and Susceptible Plant-Virus Interactions. Front. Microbiol. 2021, 12. [CrossRef]
- Sinijadas, K.; Paul, A.; Radhika, N.S.; Johnson, J.M.; Manju, R.V.; Anuradha, T. Piriformospora indica suppresses the symptoms produced by Banana bract mosaic virus by inhibiting its replication and manipulating chlorophyll and carotenoid biosynthesis and degradation in banana. 3 Biotech 2024, 14, 1–19. [CrossRef]
- Sankaranarayanan, R.; Palani, S.N.; Kumar, A.; S., P.S.A.; Tennyson, J. Prediction and experimental confirmation of banana bract mosaic virus encoding miRNAs and their targets. ExRNA 2020, 2, 1–8. [CrossRef]
- Chabi, M.; Dassou, A.G.; Adoukonou-Sagbadja, H.; Thomas, J.; Omondi, A.B. Variation in Symptom Development and Infectivity of Banana Bunchy Top Disease among Four Cultivars of Musa sp.. Crops 2023, 3, 158–169. [CrossRef]
- Qazi, J. Banana bunchy top virus and the bunchy top disease. J. Gen. Plant Pathol. 2015, 82, 2–11. [CrossRef]
- Ji, X.-L.; Yu, N.-T.; Qu, L.; Li, B.-B.; Liu, Z.-X. Banana bunchy top virus (BBTV) nuclear shuttle protein interacts and re-distributes BBTV coat protein in Nicotiana benthamiana. 3 Biotech 2019, 9, 1–6. [CrossRef]
- Lantican, D.V.; Nocum, J.D.L.; Manohar, A.N.C.; Mendoza, J.-V.S.; Gardoce, R.R.; Lachica, G.C.; Gueco, L.S.; Cueva, F.M.D. Comparative RNA-seq analysis of resistant and susceptible banana genotypes reveals molecular mechanisms in response to banana bunchy top virus (BBTV) infection. Sci. Rep. 2023, 13, 1–14. [CrossRef]
- Tripathi, L.; Ntui, V.O.; Tripathi, J.N.; Kumar, P.L. Application of CRISPR/Cas for Diagnosis and Management of Viral Diseases of Banana. Front. Microbiol. 2021, 11. [CrossRef]
- Nasim, N.; Dey, N. Pararetroviruses: Plant Infecting dsDNA Viruses. Plant Mol. Biol. Rep. 2021, 40, 106–118. [CrossRef]
- Rajeswaran, R.; Seguin, J.; Chabannes, M.; Duroy, P.-O.; Laboureau, N.; Farinelli, L.; Iskra-Caruana, M.-L.; Pooggin, M.M.; Scaturro, P.; Trist, I.M.L.; et al. Evasion of Short Interfering RNA-Directed Antiviral Silencing in Musa acuminata Persistently Infected with Six Distinct Banana Streak Pararetroviruses. J. Virol. 2014, 88, 11516–11528. [CrossRef]
- Xia, X.-J.; Zhou, Y.-H.; Shi, K.; Zhou, J.; Foyer, C.H.; Yu, J.-Q. Interplay between reactive oxygen species and hormones in the control of plant development and stress tolerance. J. Exp. Bot. 2015, 66, 2839–2856. [CrossRef]
- Arena, G.D.; Ramos-González, P.L.; Falk, B.W.; Casteel, C.L.; Freitas-Astúa, J.; Machado, M.A. Plant Immune System Activation Upon Citrus Leprosis Virus C Infection Is Mimicked by the Ectopic Expression of the P61 Viral Protein. Front. Plant Sci. 2020, 11, 1188. [CrossRef]
- Folimonova, S. Y.; Sun, Y.-D. Annual Review of Virology Citrus Tristeza Virus: From Pathogen to Panacea. 2022.
- Folimonova, S.Y. Citrus tristeza virus: A large RNA virus with complex biology turned into a valuable tool for crop protection. PLOS Pathog. 2020, 16, e1008416. [CrossRef]
- Lu, R.; Folimonov, A.; Shintaku, M.; Li, W.-X.; Falk, B.W.; Dawson, W.O.; Ding, S.-W. Three distinct suppressors of RNA silencing encoded by a 20-kb viral RNA genome. Proc. Natl. Acad. Sci. 2004, 101, 15742–15747. [CrossRef]
- Kang, S.-H.; Sun, Y.-D.; Atallah, O.O.; Huguet-Tapia, J.C.; Noble, J.D.; Folimonova, S.Y. A Long Non-Coding RNA of Citrus tristeza virus: Role in the Virus Interplay with the Host Immunity. Viruses 2019, 11, 436. [CrossRef]
- Reyes, C.A.; De Francesco, A.; Peña, E.J.; Costa, N.; Plata, M.I.; Sendin, L.; Castagnaro, A.P.; García, M.L. Resistance to Citrus psorosis virus in transgenic sweet orange plants is triggered by coat protein–RNA silencing. J. Biotechnol. 2011, 151, 151–158. [CrossRef]
- Zhou, C.; da Graça, J. V.; Freitas-Astúa, J.; Vidalakis, G.; Duran-Vila, N.; Lavagi, I. Citrus Viruses and Viroids. The Genus Citrus 2020, 391–410.
- Belabess, Z.; Sagouti, T.; Rhallabi, N.; Tahiri, A.; Massart, S.; Tahzima, R.; Lahlali, R.; Jijakli, M.H. Citrus Psorosis Virus: Current Insights on a Still Poorly Understood Ophiovirus. Microorganisms 2020, 8, 1197. [CrossRef]
- Reyes, C.A.; Ocolotobiche, E.E.; Marmisollé, F.E.; Luna, G.R.; Borniego, M.B.; Bazzini, A.A.; Asurmendi, S.; García, M.L. Citrus psorosis virus 24K protein interacts with citrus miRNA precursors, affects their processing and subsequent miRNA accumulation and target expression. Mol. Plant Pathol. 2015, 17, 317–329. [CrossRef]
- Dey, K.K.; Green, J.C.; Melzer, M.; Borth, W.; Hu, J.S. Mealybug Wilt of Pineapple and Associated Viruses. Horticulturae 2018, 4, 52. [CrossRef]
- Green, J.C.; Rwahnih, M.A.; Olmedo-Velarde, A.; Melzer, M.J.; Hamim, I.; Borth, W.B.; Brower, T.M.; Wall, M.; Hu, J.S. Further genomic characterization of pineapple mealybug wilt-associated viruses using high-throughput sequencing. Trop. Plant Pathol. 2020, 45, 64–72. [CrossRef]
- Galeano, E.A.V.; Martins, D.S.; Barros, F.L.S.; Ventura, J.A.; Queiroz, R.B. Cadeia produtiva do mamão no Espírito Santo. Vitória, ES. Incaper 2022, 1-172.
- Premchand, U.; Mesta, R.K.; Devappa, V.; Basavarajappa, M.P.; Venkataravanappa, V.; Reddy, L.R.C.N.; Shankarappa, K.S. Survey, Detection, Characterization of Papaya Ringspot Virus from Southern India and Management of Papaya Ringspot Disease. Pathogens 2023, 12, 824. [CrossRef]
- Hamim, I.; Borth, W.B.; Marquez, J.; Green, J.C.; Melzer, M.J.; Hu, J.S. Transgene-mediated resistance to Papaya ringspot virus: challenges and solutions. Phytoparasitica 2018, 46, 1–18. [CrossRef]
- Vargas-Mejía, P.; Vega-Arreguín, J.; Chávez-Calvillo, G.; Ibarra-Laclette, E.; Silva-Rosales, L. Differential Accumulation of Innate- and Adaptive-Immune-Response-Derived Transcripts during Antagonism between Papaya Ringspot Virus and Papaya Mosaic Virus. Viruses 2020, 12, 230. [CrossRef]
- Siriwan, W.; Roytrakul, S.; Chowpongpang, S.; Sawwa, A. Carica Papaya Proteins. J. Plant Interact. 2021, 16 (1), 474–480.
- Antunes, T.F.S.; Maurastoni, M.; Madronero, L.J.; Fuentes, G.; Santamaria, J.M.; Ventura, J.A.; Abreu, E.F.; Fernandes, A.A.R.; Fernandes, P.M.B. Battle of Three: The Curious Case of Papaya Sticky Disease. Plant Dis. 2020, 104, 2754–2763. [CrossRef]
- Antunes, T.F.S.; Amaral, R.J.V.; Ventura, J.A.; Godinho, M.T.; Amaral, J.G.; Souza, F.O.; Zerbini, P.A.; Zerbini, F.M.; Fernandes, P.M.B. The dsRNA Virus Papaya Meleira Virus and an ssRNA Virus Are Associated with Papaya Sticky Disease. PLOS ONE 2016, 11, e0155240–e0155240. [CrossRef]
- García-Cámara, I.; Tapia-Tussell, R.; Magaña-Álvarez, A.; Velázquez, A.C.; Martín-Mex, R.; Moreno-Valenzuela, O.A.; Pérez-Brito, D. Empoasca papayae (Hemiptera: Cicadellidae)-Mediated Transmission of Papaya Meleira Virus-Mexican Variant in Mexico. Plant Dis. 2019, 103, 2015–2023. [CrossRef]
- Cornejo-Franco, J.F.; Alvarez-Quinto, R.A.; Quito-Avila, D.F. Transmission of the umbra-like Papaya virus Q in Ecuador and its association with meleira-related viruses from Brazil. Crop. Prot. 2018, 110, 99–102. [CrossRef]
- Puthiyaparambil, J.; Pagie, M.; Teressita, S.; Jay, P.; Bongani, N.; Paul, F.; Candy, M.; Mark, M.; Marion, M.; Ian, M.; et al. Rapid papaya crop improvement through accelerated in vitro breeding and molecular diagnostics. Acta Hortic. 2023, 499–506. [CrossRef]
- Maurastoni, M.; Antunes, T.F.S.; Abreu, E.F.M.; Ribeiro, S.G.; Mehta, A.; Sanches, M.M.; Fontes, W.; Kitajima, E.W.; Cruz, F.T.; Santos, A.M.C.; et al. A Capsid Protein Fragment of a Fusagra-like Virus Found in Carica papaya Latex Interacts with the 50S Ribosomal Protein L17. Viruses 2023, 15, 541. [CrossRef]
- Domingo-Calap, M.L.; Moreno, A.B.; Pendón, J.A.D.; Moreno, A.; Fereres, A.; López-Moya, J.J. Assessing the Impact on Virus Transmission and Insect Vector Behavior of a Viral Mixed Infection in Melon. Phytopathology® 2020, 110, 174–186. [CrossRef]
- Domingo-Calap, M.L.; Chase, O.; Estapé, M.; Moreno, A.B.; López-Moya, J.J. The P1 Protein of Watermelon mosaic virus Compromises the Activity as RNA Silencing Suppressor of the P25 Protein of Cucurbit yellow stunting disorder virus. Front. Microbiol. 2021, 12. [CrossRef]
- Ouibrahim, L.; Mazier, M.; Estevan, J.; Pagny, G.; Decroocq, V.; Desbiez, C.; Moretti, A.; Gallois, J. L.; Caranta, C. Cloning of the Arabidopsis rwm1 Gene for Resistance to Watermelon Mosaic Virus Points to a New Function for Natural Virus Resistance Genes. Plant J. 2014, 79 (5), 705–716.
- Ali, A. Epidemiology and Evolution of Poytviruses Infecting Cucurbits. Appl. Plant Virol. Adv. Detect. Antivir. Strateg. 2020, 405–417.
- Kesh, H.; Kaushik, P. Advances in melon (Cucumis melo L.) breeding: An update. Sci. Hortic. 2021, 282, 110045. [CrossRef]
- Normantovich, M.; Amitzur, A.; Offri, S.; Pashkovsky, E.; Shnaider, Y.; Nizan, S.; Yogev, O.; Jacob, A.; Taylor, C.G.; Desbiez, C.; et al. The melon Fom-1–Prv resistance gene pair: Correlated spatial expression and interaction with a viral protein. Plant Direct 2024, 8, e565. [CrossRef]
- Tatineni, S.; Hein, G.L. Plant Viruses of Agricultural Importance: Current and Future Perspectives of Virus Disease Management Strategies. Phytopathology® 2023, 113, 117–141. [CrossRef]
- Manjunatha, L.; Rajashekara, H.; Uppala, L.S.; Ambika, D.S.; Patil, B.; Shankarappa, K.S.; Nath, V.S.; Kavitha, T.R.; Mishra, A.K. Mechanisms of Microbial Plant Protection and Control of Plant Viruses. Plants 2022, 11, 3449. [CrossRef]
- Spadotti, D.M.A.; Favara, G.M.; Novaes, Q.S.; Mello, A.P.O.A.; Freitas, D.M.S.; Molina, J.P.E.; Rezende, J.A.M. Long-lasting systematic roguing for effective management of CABMV in passion flower orchards through maintenance of separated plants. Plant Pathol. 2019, 68, 1259–1267. [CrossRef]
- Rezende, J. A. M.; Kitajima, E. W. Vírus e viroides. In: Manual de Fitopatologia – Princípios e Conceitos 2018, 1, São Paulo, Brazil, 369–76.
- Ventura, J.A.; Costa, H.; Tatagiba, J.S.; Andrade, J.S.; Martins, D.S. Meleira do mamoeiro: Etiologia, sintomas e epidemiologia. In Papaya Brasil: Qualidade do Mamão Para o Mercado Interno; Martins, D.S., Ed.Incaper: Vitória, Brazil, 2003267–276.
- Abiola, A.; Zandjanakou-Tachin, M.; Aoudji, K.N.A.; Avocevou-Ayisso, C.; Kumar, P.L. Adoption of Roguing to Contain Banana Bunchy Top Disease in South-East Bénin: Role of Farmers’ Knowledge and Perception. Int. J. Fruit Sci. 2019, 20, 720–736. [CrossRef]
- Krishna, R.; Karkute, S. G.; Waquar, ·; Ansari, A.; Durgesh, ·; Jaiswal, K.; Prakash Verma, J.; Singh, M. Transgenic Tomatoes for Abiotic Stress Tolerance: Status and Way Ahead. Biotech. 2019, 9, 143.
- Jin, Y.; Goodman, R.E.; Tetteh, A.O.; Lu, M.; Tripathi, L. Bioinformatics analysis to assess potential risks of allergenicity and toxicity of HRAP and PFLP proteins in genetically modified bananas resistant to Xanthomonas wilt disease. Food Chem. Toxicol. 2017, 109, 81–89. [CrossRef]
- Stowe, E.; Dhingra, A. Development of the Arctic® Apple. Plant Breed. Rev. 2020, 273–296.
- Mhatre, M. Agrobacterium-Mediated Genetic Transformation of Pineapple (Ananas Comosus L., Merr.). Methods Mol. Biol. 2012, 994, 435–453.
- Waltz, E. GABA-enriched tomato is first CRISPR-edited food to enter market. Nat. Biotechnol. 2021, 40, 9–11. [CrossRef]
- Tripathi, L.; Ntui, V.O.; Tripathi, J.N. CRISPR/Cas9-based genome editing of banana for disease resistance. Curr. Opin. Plant Biol. 2020, 56, 118–126. [CrossRef]
- Malnoy, M.; Viola, R.; Jung, M.-H.; Koo, O.-J.; Kim, S.; Kim, J.-S.; Velasco, R.; Kanchiswamy, C.N. DNA-Free Genetically Edited Grapevine and Apple Protoplast Using CRISPR/Cas9 Ribonucleoproteins. Front. Plant Sci. 2016, 7, 1904. [CrossRef]
- Jia, H.; Nian, W. Targeted Genome Editing of Sweet Orange Using Cas9/SgRNA. PLoS One 2014, 9 (4).
- Parsa, H.S.; Sabet, M.S.; Moieni, A.; Shojaeiyan, A.; Dogimont, C.; Boualem, A.; Bendahmane, A. CRISPR/Cas9-Mediated Cytosine Base Editing Using an Improved Transformation Procedure in Melon (Cucumis melo L.). Int. J. Mol. Sci. 2023, 24, 11189. [CrossRef]
- Ferreira, S.A.; Pitz, K.Y.; Manshardt, R.; Zee, F.; Fitch, M.; Gonsalves, D. Virus Coat Protein Transgenic Papaya Provides Practical Control of Papaya ringspot virus in Hawaii. Plant Dis. 2002, 86, 101–105. [CrossRef]
- Gonsalves, D.; Gonsalves, C.; Ferreira, C.; Fitch, M. Transgenic Virus-Resistant Papaya: From Hope to Reality in Controlling Papaya Ringspot Virus in Hawaii.. APSnet Featur. Artic. 2004. [CrossRef]
- Gonsalves, D. Hawaii’s Transgenic Papaya Story 1978-2012: A Personal Account. Genet. Genomics Papaya 2014, 115–142.
- Wu, Z.; Mo, C.; Zhang, S.; Li, H. Characterization of Papaya Ringspot Virus Isolates Infecting Transgenic Papaya ‘Huanong No.1’ in South China. Sci. Reports 2018, 8 (1), 1–11.
- Christou, P. Plant genetic engineering and agricultural biotechnology 1983–2013. Trends Biotechnol. 2013, 31, 125–127. [CrossRef]
- Filho, P.E.M.; Vilarinhos, A.D.; Oliveira, V.J.d.S.d.; Silva, D.d.C.S.d.; Santos, V.d.S.; Dantas, J.L.L. Resistance of transgenic papaya trees to papaya ringspot in Brazil. Pesqui. Agropecu. Bras. 2021, 56. [CrossRef]
- Ye, Changming, and Huaping Li. 20 Years of transgenic research in China for resistance to Papaya ringspot virus. Transgenic Plant J., 2010, 4, 58-63.
- Gao, L.; Luo, J.; Ding, X.; Wang, T.; Hu, T.; Song, P.; Zhai, R.; Zhang, H.; Zhang, K.; Li, K.; et al. Soybean RNA interference lines silenced for eIF4E show broad potyvirus resistance. Mol. Plant Pathol. 2019, 21, 303–317. [CrossRef]
- Atarashi, H.; Jayasinghe, W.H.; Kwon, J.; Kim, H.; Taninaka, Y.; Igarashi, M.; Ito, K.; Yamada, T.; Masuta, C.; Nakahara, K.S. Artificially Edited Alleles of the Eukaryotic Translation Initiation Factor 4E1 Gene Differentially Reduce Susceptibility to Cucumber Mosaic Virus and Potato Virus Y in Tomato. Front. Microbiol. 2020, 11. [CrossRef]
- Lucioli, A.; Tavazza, R.; Baima, S.; Fatyol, K.; Burgyan, J.; Tavazza, M. CRISPR-Cas9 Targeting of the eIF4E1 Gene Extends the Potato Virus Y Resistance Spectrum of the Solanum tuberosum L. cv. Desirée. Front. Microbiol. 2022, 13, 873930. [CrossRef]
- Zulfiqar, S.; Farooq, M.A.; Zhao, T.; Wang, P.; Tabusam, J.; Wang, Y.; Xuan, S.; Zhao, J.; Chen, X.; Shen, S.; et al. Virus-Induced Gene Silencing (VIGS): A Powerful Tool for Crop Improvement and Its Advancement towards Epigenetics. Int. J. Mol. Sci. 2023, 24, 5608. [CrossRef]
- Sharma, N.; Prasad, M. Silencing AC1 of Tomato leaf curl virus using artificial microRNA confers resistance to leaf curl disease in transgenic tomato. Plant Cell Rep. 2020, 39, 1565–1579. [CrossRef]
- Kim, J.; Lee, S.; Baek, K.; Jin, E. Site-Specific Gene Knock-Out and On-Site Heterologous Gene Overexpression in Chlamydomonas reinhardtii via a CRISPR-Cas9-Mediated Knock-in Method. Front. Plant Sci. 2020, 11, 306. [CrossRef]
- Lee, J.E.; Neumann, M.; Duro, D.I.; Schmid, M. CRISPR-based tools for targeted transcriptional and epigenetic regulation in plants. PLOS ONE 2019, 14, e0222778. [CrossRef]
- Khalid, A.; Zhang, Q.; Yasir, M.; Li, F. Small RNA Based Genetic Engineering for Plant Viral Resistance: Application in Crop Protection. Front. Microbiol. 2017, 8, 43. [CrossRef]
- van Esse, H.P.; Reuber, T.L.; van der Does, D. Genetic modification to improve disease resistance in crops. New Phytol. 2019, 225, 70–86. [CrossRef]
- Zhao, Y.; Yang, X.; Zhou, G.; Zhang, T. Engineering plant virus resistance: from RNA silencing to genome editing strategies. Plant Biotechnol. J. 2019, 18, 328–336. [CrossRef]
- Huang, J.; Yang, M.; Zhang, X. The function of small RNAs in plant biotic stress response. J. Integr. Plant Biol. 2016, 58, 312–327. [CrossRef]
- Yang, Z.; Li, Y. Dissection of RNAi-based antiviral immunity in plants. Curr. Opin. Virol. 2018, 32, 88–99. [CrossRef]
- Aregger, M.; Borah, B.K.; Seguin, J.; Rajeswaran, R.; Gubaeva, E.G.; Zvereva, A.S.; Windels, D.; Vazquez, F.; Blevins, T.; Farinelli, L.; et al. Primary and Secondary siRNAs in Geminivirus-induced Gene Silencing. PLOS Pathog. 2012, 8, e1002941. [CrossRef]
- Yu, H.; Wang, Y.; Fu, F.; Li, W. Transgenic Improvement for Biotic Resistance of Crops. Int. J. Mol. Sci. 2022, 23, 14370. [CrossRef]
- Csorba, T.; Kontra, L.; Burgyán, J. viral silencing suppressors: Tools forged to fine-tune host-pathogen coexistence. Virology 2015, 479-480, 85–103. [CrossRef]
- Shekhawat, U.K.S.; Ganapathi, T.R.; Hadapad, A.B. Transgenic banana plants expressing small interfering RNAs targeted against viral replication initiation gene display high-level resistance to banana bunchy top virus infection. J. Gen. Virol. 2012, 93, 1804–1813. [CrossRef]
- Kung, Y.-J.; Yu, T.-A.; Huang, C.-H.; Wang, H.-C.; Wang, S.-L.; Yeh, S.-D. Generation of hermaphrodite transgenic papaya lines with virus resistance via transformation of somatic embryos derived from adventitious roots of in vitro shoots. Transgenic Res. 2009, 19, 621–635. [CrossRef]
- Collinge, D.B.; Sarrocco, S. Transgenic approaches for plant disease control: Status and prospects 2021. Plant Pathol. 2021, 71, 207–225. [CrossRef]
- Niehl, A.; Soininen, M.; Poranen, M.M.; Heinlein, M. Synthetic biology approach for plant protection using dsRNA. Plant Biotechnol. J. 2018, 16, 1679–1687. [CrossRef]
- Taliansky, M.; Samarskaya, V.; Zavriev, S.K.; Fesenko, I.; Kalinina, N.O.; Love, A.J. RNA-Based Technologies for Engineering Plant Virus Resistance. Plants 2021, 10, 82. [CrossRef]
- Mushtaq, M.; Sakina, A.; Wani, S.H.; Shikari, A.B.; Tripathi, P.; Zaid, A.; Galla, A.; Abdelrahman, M.; Sharma, M.; Singh, A.K.; et al. Harnessing Genome Editing Techniques to Engineer Disease Resistance in Plants. Front. Plant Sci. 2019, 10, 550. [CrossRef]
- Scheben, A.; Wolter, F.; Batley, J.; Puchta, H.; Edwards, D. Towards CRISPR/Cas crops – bringing together genomics and genome editing. New Phytol. 2017, 216, 682–698. [CrossRef]
- Prado, G.S.; Rocha, D.C.; dos Santos, L.N.; Contiliani, D.F.; Nobile, P.M.; Martinati-Schenk, J.C.; Padilha, L.; Maluf, M.P.; Lubini, G.; Pereira, T.C.; et al. CRISPR technology towards genome editing of the perennial and semi-perennial crops citrus, coffee and sugarcane. Front. Plant Sci. 2024, 14, 1331258. [CrossRef]
- Schiml, S.; Puchta, H. Revolutionizing plant biology: multiple ways of genome engineering by CRISPR/Cas. Plant Methods 2016, 12, 1–9. [CrossRef]
- Anzalone, A. V.; Koblan, L. W.; Liu, D. R. Genome editing with CRISPR–Cas nucleases, base editors, transposases and prime editors. Nature Biotechnology, 2020, 38, 824-844.
- Tripathi, L.; Ntui, V.O.; Tripathi, J.N.; Kumar, P.L. Application of CRISPR/Cas for Diagnosis and Management of Viral Diseases of Banana. Front. Microbiol. 2021, 11. [CrossRef]
- Jinek, M.; Chylinski, K.; Fonfara, I.; Hauer, M.; Doudna, J.A.; Charpentier, E. A Programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 2012, 337, 816–821. [CrossRef]
- Khan, Z.A.; Kumar, R.; Dasgupta, I. CRISPR/Cas-Mediated Resistance against Viruses in Plants. Int. J. Mol. Sci. 2022, 23, 2303. [CrossRef]
- Pyott, D. E.; Sheehan, E.; Molnar, A. Engineering of CRISPR/Cas9-mediated potyvirus resistance in transgene-free Arabidopsis plants. Molecular Plant Pathology, 2016, 17, 1276-1288.
- Kis, A.; Hamar, .; Tholt, G.; Bán, R.; Havelda, Z. Creating highly efficient resistance against wheat dwarf virus in barley by employing CRISPR/Cas9 system. Plant Biotechnol. J. 2019, 17, 1004–1006. [CrossRef]
- Mehta, D.; Stürchler, A.; Anjanappa, R.B.; Zaidi, S.S.-e.-A.; Hirsch-Hoffmann, M.; Gruissem, W.; Vanderschuren, H. Linking CRISPR-Cas9 interference in cassava to the evolution of editing-resistant geminiviruses. Genome Biol. 2019, 20, 80. [CrossRef]
- Tripathi, J.N.; Ntui, V.O.; Ron, M.; Muiruri, S.K.; Britt, A.; Tripathi, L. CRISPR/Cas9 editing of endogenous banana streak virus in the B genome of Musa spp. overcomes a major challenge in banana breeding. Commun. Biol. 2019, 2, 1–11. [CrossRef]
- Ntui, V. O.; Tripathi, J. N.; Tripathi, L. Robust CRISPR/Cas9 mediated genome editing tool for banana and plantain (Musa spp.). Current Plant Biology, 2020, 21, 100-128.
- Tripathi, L.; Ntui, V.O.; Tripathi, J.N. Control of Bacterial Diseases of Banana Using CRISPR/Cas-Based Gene Editing. Int. J. Mol. Sci. 2022, 23, 3619. [CrossRef]
- Jia, H.; Orbović, V.; Wang, N. CRISPR-LbCas12a-mediated modification of citrus. Plant Biotechnology Journal, 2019, 17, 1928-1937.
- Jia, H.; Omar, A. A.; Orbović, V.; Wang, N. Biallelic editing of the LOB1 promoter via CRISPR/Cas9 creates canker-resistant ‘Duncan’ grapefruit. Phytopathology®, 2022, 112, 308-314.
- Hooghvorst, I.; López-Cristoffanini, C.; Nogués, S. Efficient knockout of phytoene desaturase gene using CRISPR/Cas9 in melon. Sci. Rep. 2019, 9, 1–7. [CrossRef]
- Nizan, S.; Amitzur, A.; Dahan-Meir, T.; Benichou, J.I.C.; Bar-Ziv, A.; Perl-Treves, R. Mutagenesis of the melon Prv gene by CRISPR/Cas9 breaks papaya ringspot virus resistance and generates an autoimmune allele with constitutive defense responses. J. Exp. Bot. 2023, 74, 4579–4596. [CrossRef]
- Nishitani, C.; Hirai, N.; Komori, S.; Wada, M.; Okada, K.; Osakabe, K.; Yamamoto, T.; Osakabe, Y. Efficient Genome Editing in Apple Using a CRISPR/Cas9 system. Sci. Rep. 2016, 6, 31481. [CrossRef]
- Osakabe, Y.; Liang, Z.; Ren, C.; Nishitani, C.; Osakabe, K.; Wada, M.; Komori, S.; Malnoy, M.; Velasco, R.; Poli, M.; et al. CRISPR–Cas9-mediated genome editing in apple and grapevine. Nat. Protoc. 2018, 13, 2844–2863. [CrossRef]
- Brewer, S.E.; Chambers, A.H. CRISPR/Cas9-mediated genome editing of phytoene desaturase in Carica papaya L. J. Hortic. Sci. Biotechnol. 2022, 97, 580–592. [CrossRef]
- Jagram, N.; Dasgupta, I. Principles and practice of virus induced gene silencing for functional genomics in plants. Virus Genes 2022, 59, 173–187. [CrossRef]
- Tuo, D.; Ma, C.; Yan, P.; Kong, H.; Zhou, P.; Guo, A.; Zhao, H.; Shen, W. Genetic transformation and gene delivery strategies in Carica papaya L.. Trop. Plants 2023, 2. [CrossRef]
- Chen, T.-Y.; Pai, H.; Hou, L.-Y.; Lee, S.-C.; Lin, T.-T.; Chang, C.-H.; Hsu, F.-C.; Hsu, Y.-H.; Lin, N.-S. Dual resistance of transgenic plants against Cymbidium mosaic virus and Odontoglossum ringspot virus. Sci. Rep. 2019, 9, 1–12. [CrossRef]
- Cheng, H.-W.; Lin, T.-T.; Huang, C.-H.; Raja, J.A.J.; Yeh, S.-D. Modification of Papaya Ringspot Virus HC-Pro to Generate Effective Attenuated Mutants for Overcoming the Problem of Strain-Specific Cross Protection. Plant Dis. 2023, 107, 1757–1768. [CrossRef]
- LaTourrette, K.; Garcia-Ruiz, H. Determinants of Virus Variation, Evolution, and Host Adaptation. Pathogens 2022, 11, 1039. [CrossRef]
- Desbiez, C.; Domingo-Calap, M.L.; Pitrat, M.; Wipf-Scheibel, C.; Girardot, G.; Ferriol, I.; Lopez-Moya, J.J.; Lecoq, H. Specificity of Resistance and Tolerance to Cucumber Vein Yellowing Virus in Melon Accessions and Resistance Breaking with a Single Mutation in VPg. Phytopathology® 2022, 112, 1185–1191. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).