Submitted:
15 May 2024
Posted:
15 May 2024
You are already at the latest version
Abstract

Keywords:
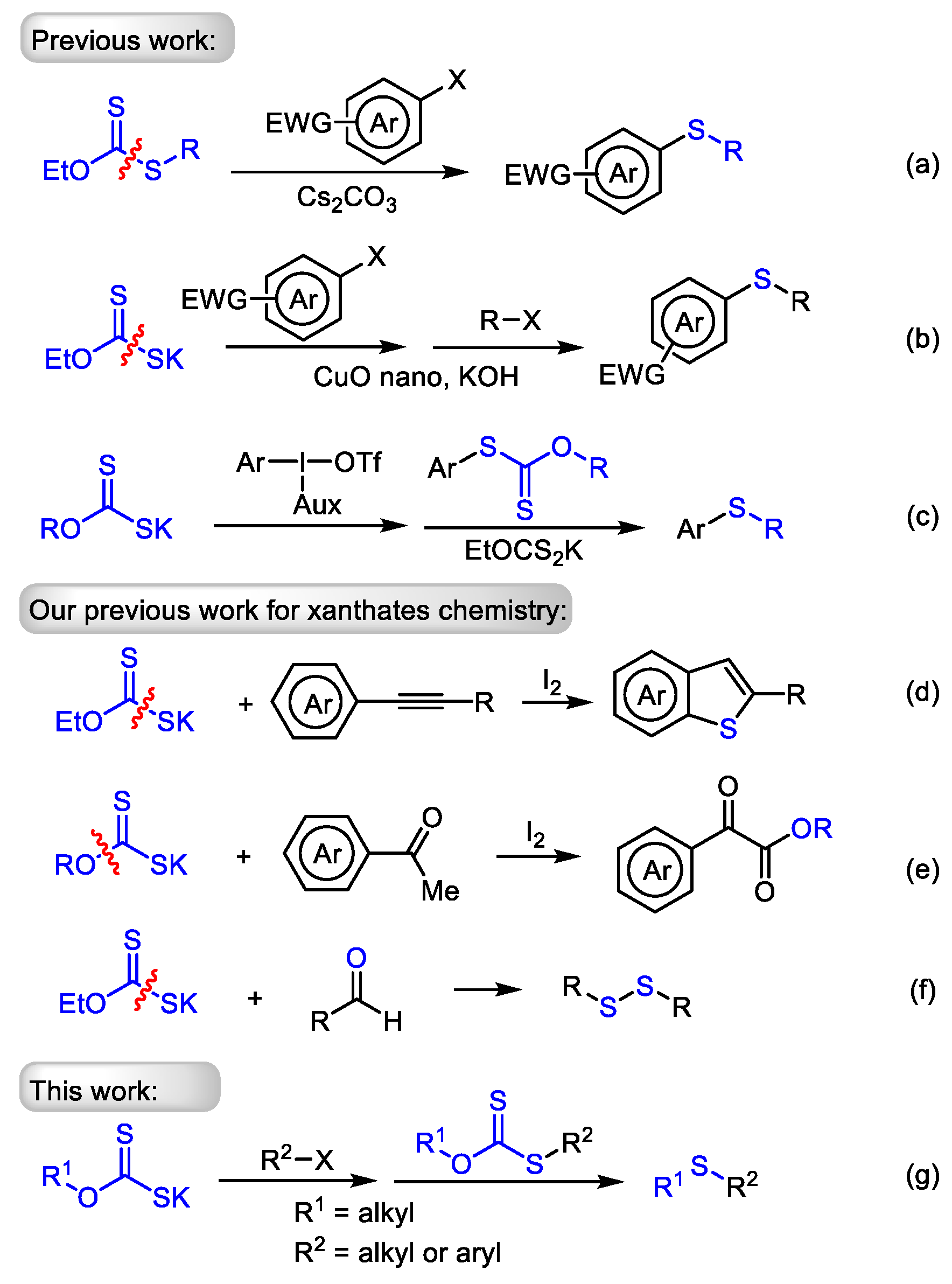
1. Introduction
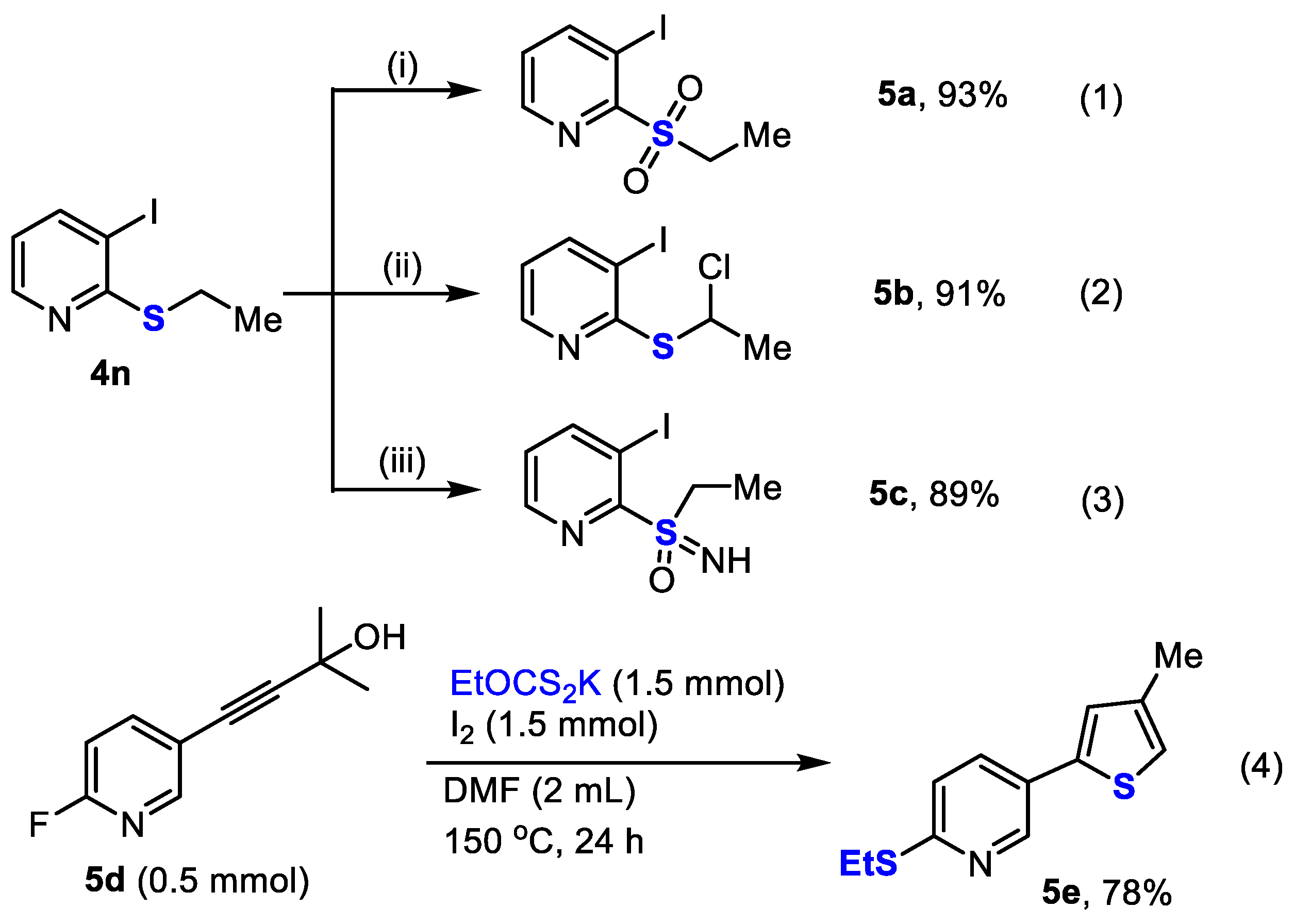
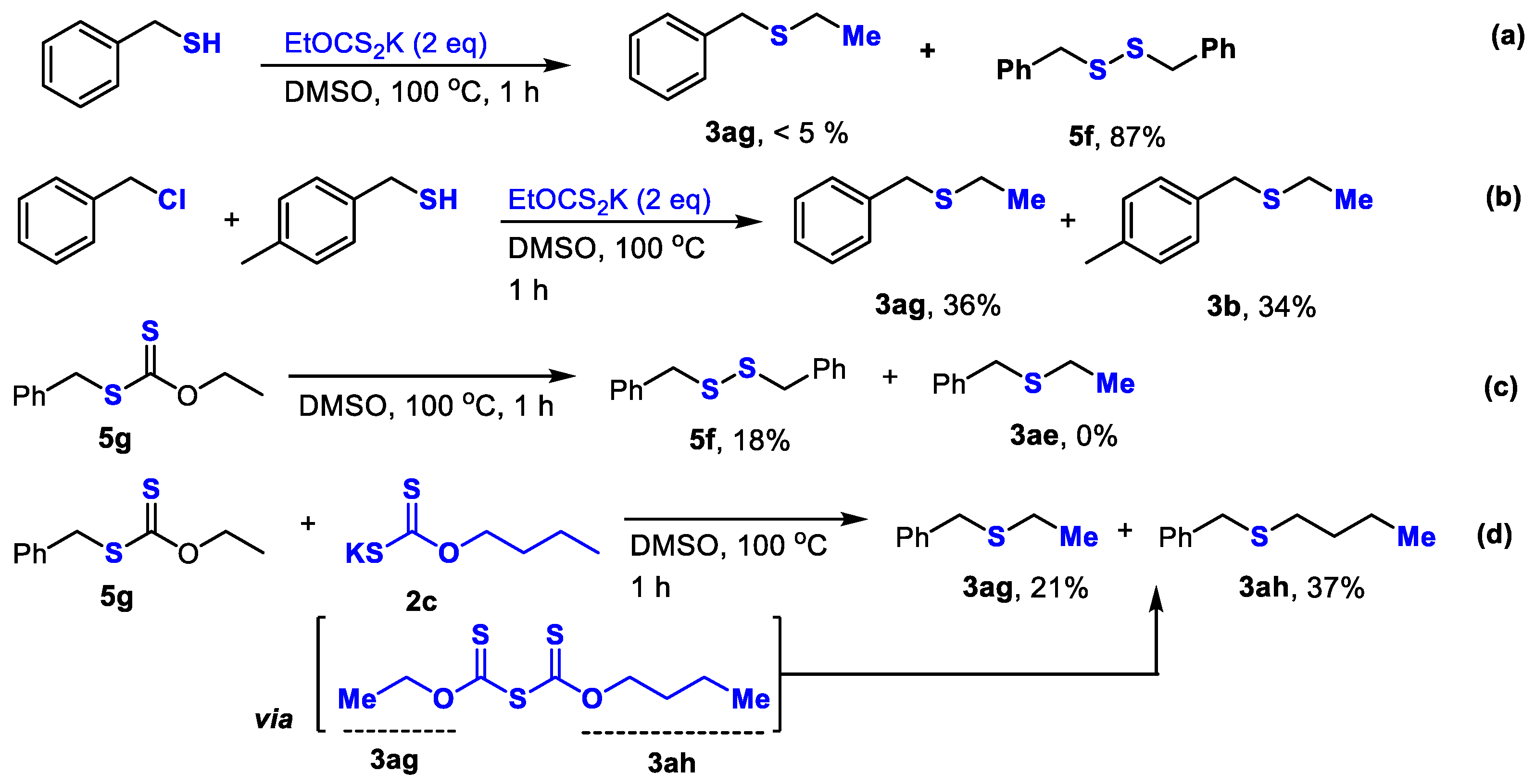
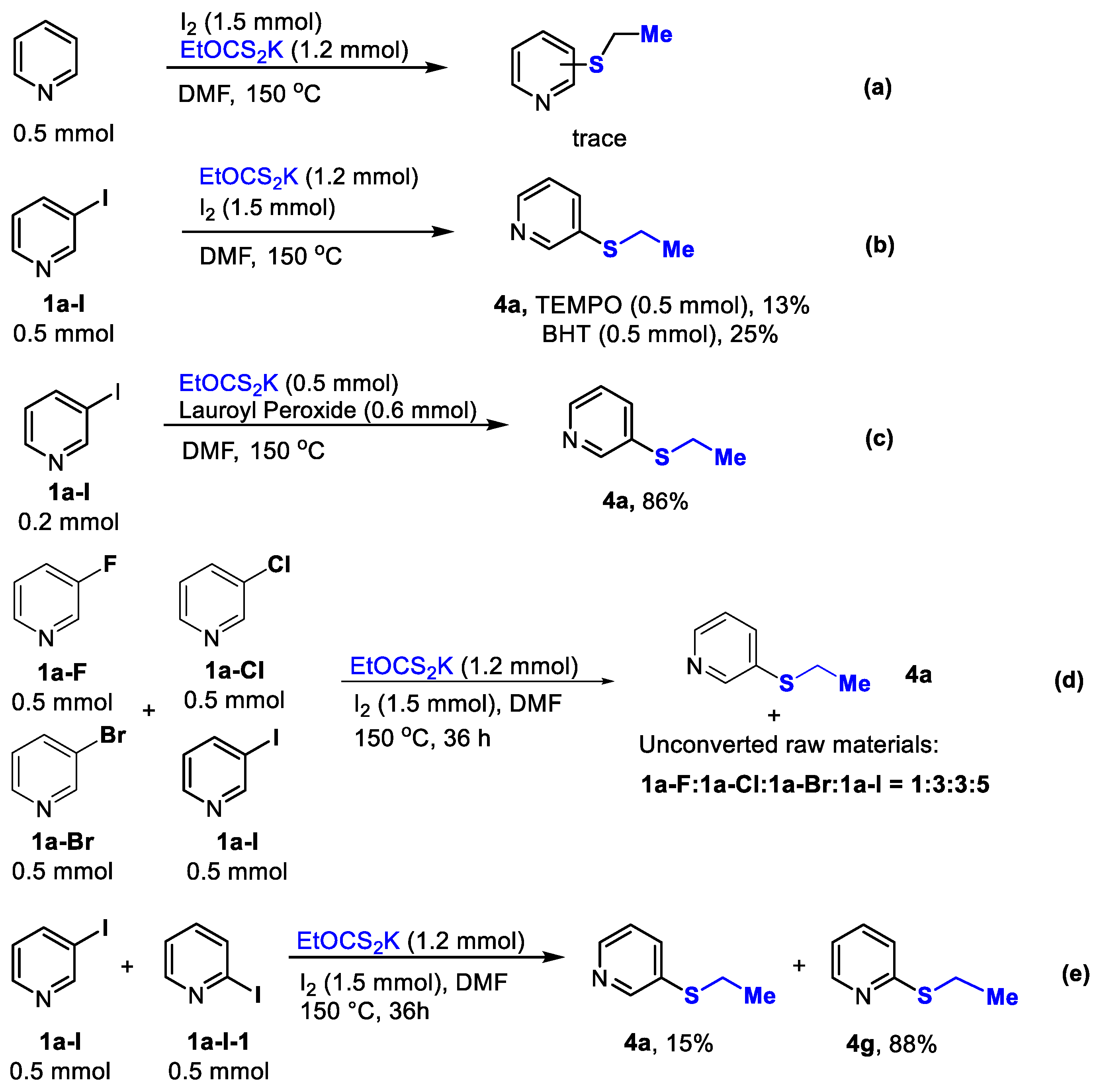
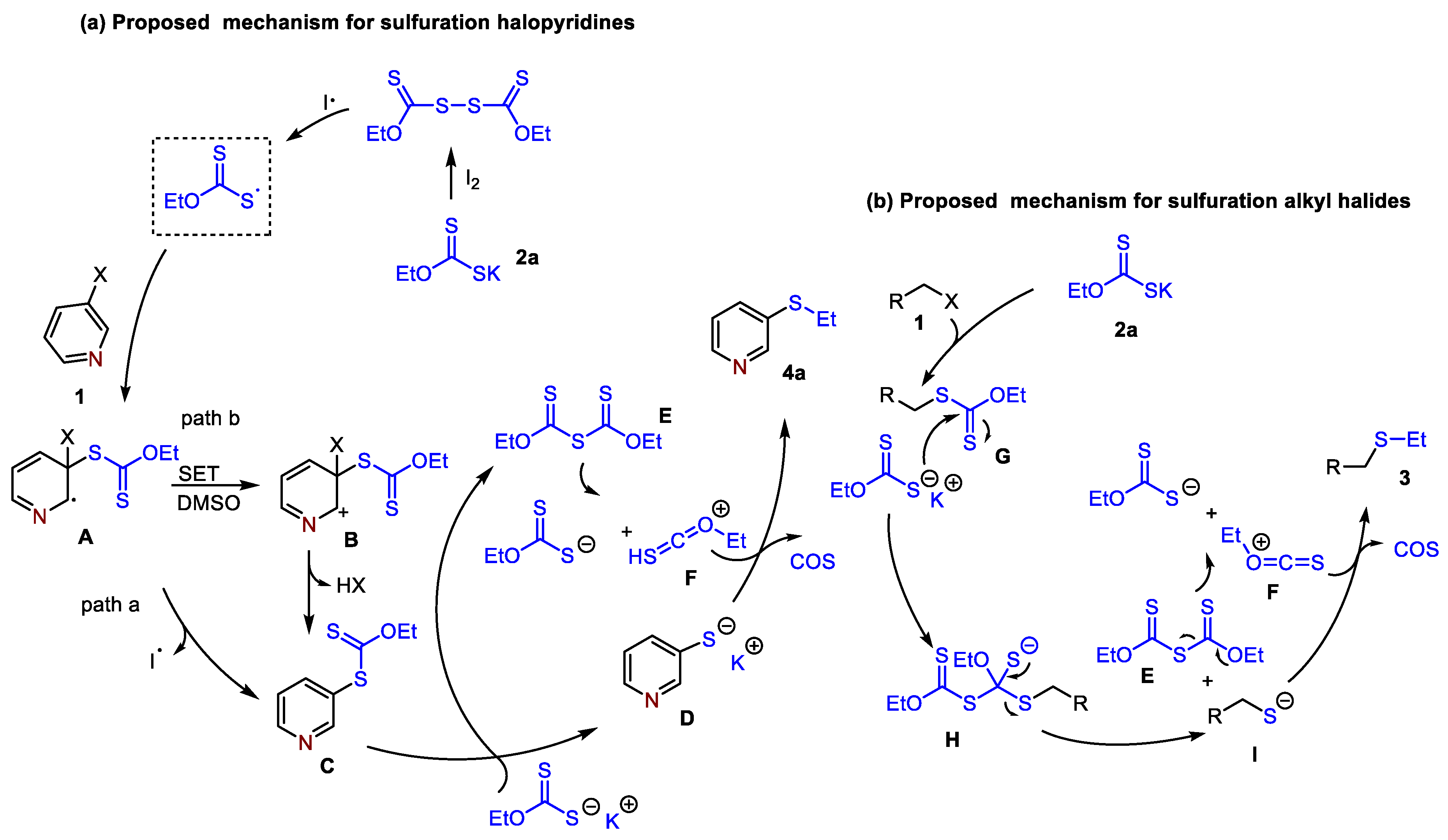
2. Results and Discussion
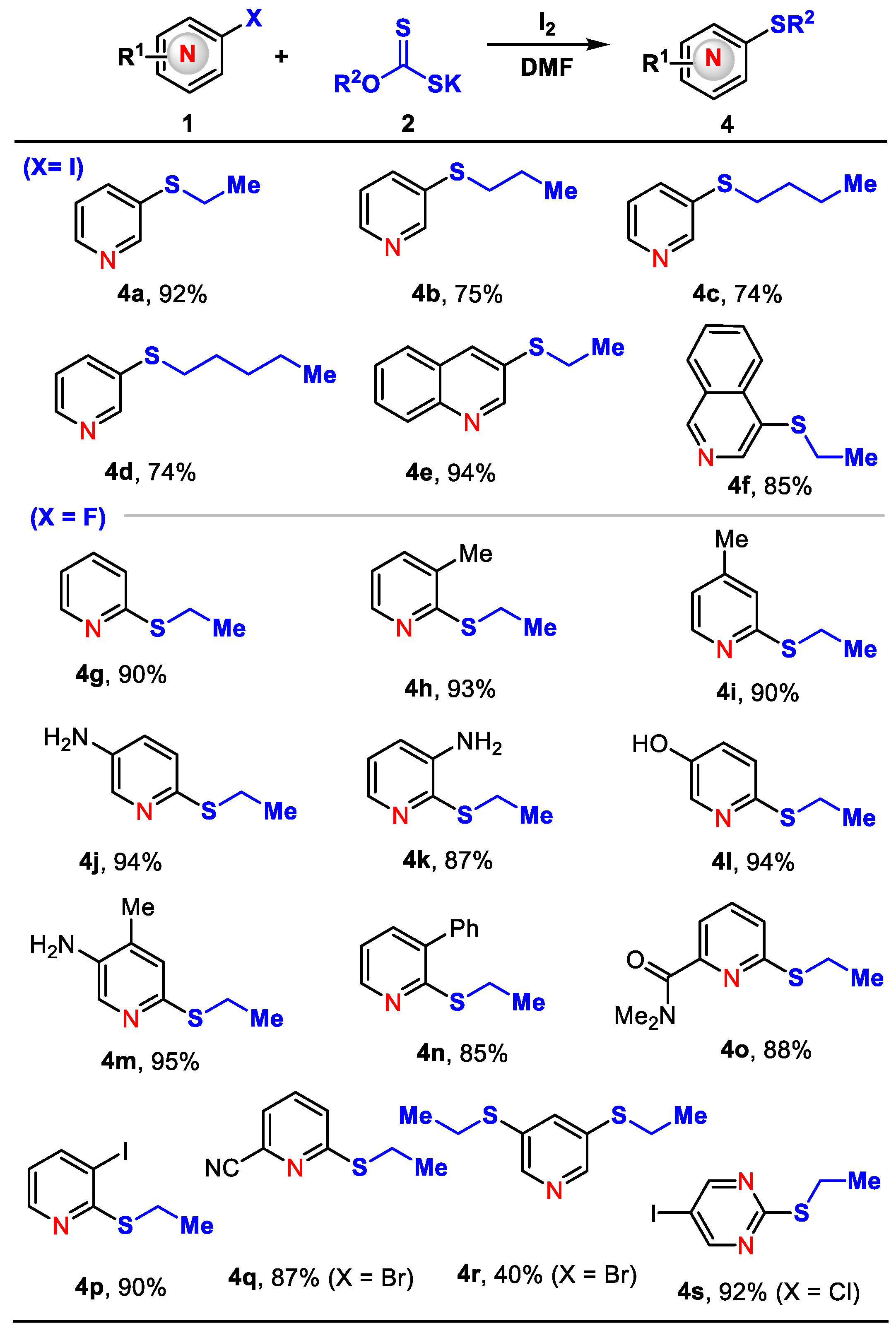
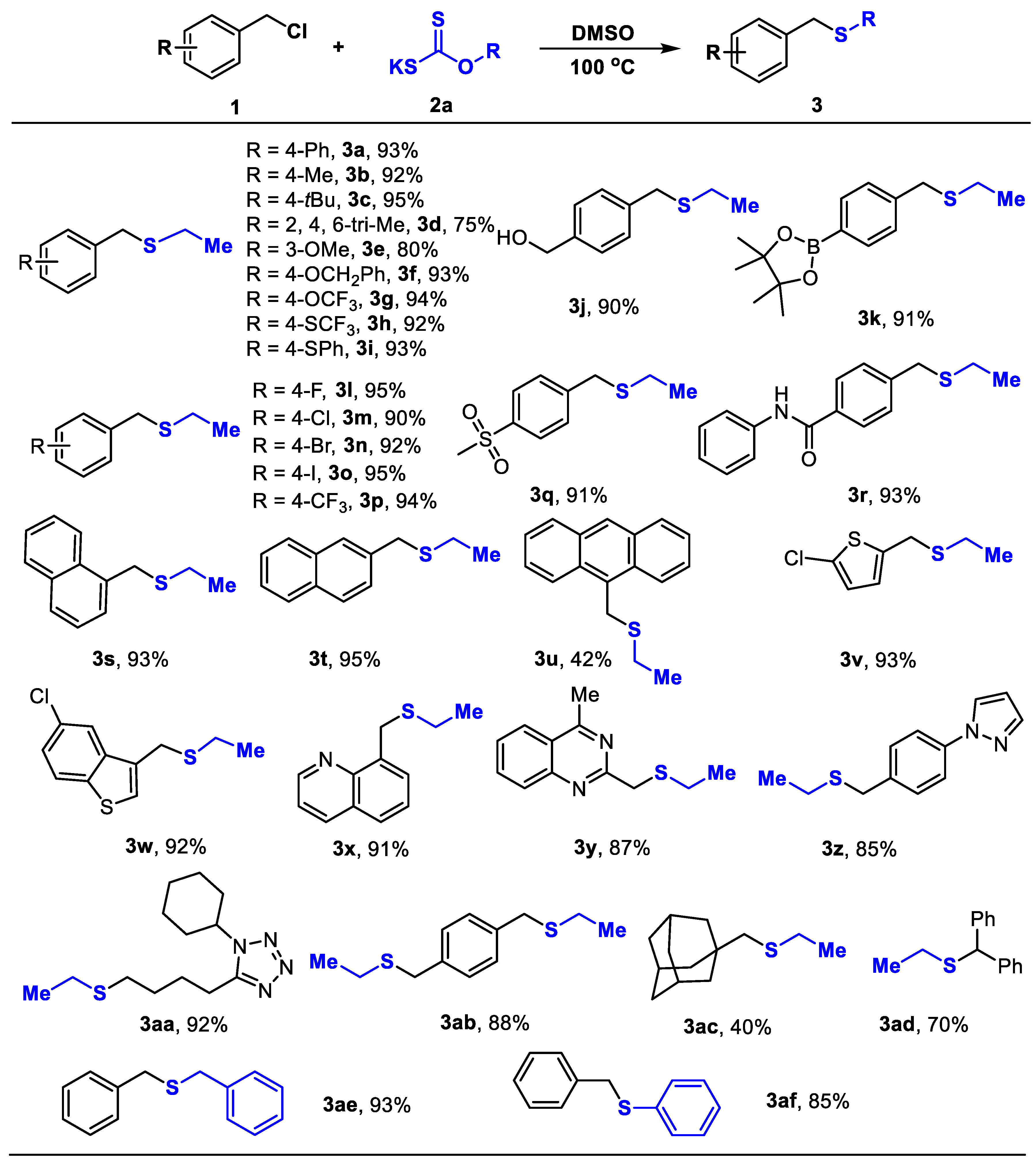
3. Materials and Methods
General Methods (Chemistry)
General Procedures for the Preparation of Compounds 3a–3af.
General Procedures for the Preparation of Compounds 4a–4s.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Dvoral, C. A.; Schmitz, W. D.; Poon, D. J.; Pryde, D. C.; Lawson, J. P.; Amos, R. A.; Meyers, A. I. The Synthesis of Streptogramin Antibiotics: (−)-Griseoviridin and Its C-8 Epimer. Angew. Chem. Int. Ed. 2000, 39, 1664–1666. [Google Scholar] [CrossRef]
- Kaldor, S. W. , Kalish, V. J., Davies, J. F., Shetty, B. V., Fritz, J. E., Appelt, K., Burgess, J. A.; Campanale K. M.; Chirgadze, N. Y.; Clawson D. K.; Dressman B. A.; Hatch S. D.; Khalil D. A.; Kosa M. B.; Lubbehusen P. P.; Muesing M. A.; Patick A. K.; Reich, S. H.; Su, K. S.; Tatlock, J. H. Viracept (Nelfinavir Mesylate, AG1343): a Potent, Orally Bioavailable Inhibitor of HIV-1 Protease. J. Med. Chem. 1997, 40, 3979–3985. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J. J; Rogers, J. D.; Holland, S. D.; Larson, P.; Amin, R. D.; Haesen, R.; Freeman, A.; Seiberling, M.; Merz, M.; Cheng, H. Pharmacokinetics and Bioavailability of Montelukast Sodium (MK-0476) in Healthy Young and Elderly Volunteers, Pharm Research, 1996, 13, 445-448. [CrossRef]
- Kahan, F. M.; Kropp, H.; Sumdelof, J. G.; Bormbaum, J. Thienamycin: Development of Imipenem-Cilastatin. J. Antimicrob. Chemoth 1983, 12, 1–35. [Google Scholar] [CrossRef] [PubMed]
- Kondo, T.; Mitsudo, T. Metal-Catalyzed Carbon−Sulfur Bond Formation. Chem. Rev. 2000, 100, 3205–3220. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ding, Y. Recent Advances in the Synthesis of Thioether, Mini-Reviews in Organic Chemistry, 2017, 14, 407-431. [CrossRef]
- Xiao, F.; Chen, S.; Li, C.; Huang, H.; Deng, G. Copper-Catalyzed Three-Component One-Pot Synthesis of Aryl Sulfides with Sulfur Powder under Aqueous Conditions. Adv. Syn. Catal. 2016, 358, 3881–3883. [Google Scholar] [CrossRef]
- Huang, H.; Qu, Z.; Ji, X.; Deng, G. J. Three-component Bs-heterocycliation for Synthesis of 2-Aminobenzo[4,5]thieno[3,2-d]thiazoles. Org. Chem. Front. 2019, 6, 1146–1150. [Google Scholar] [CrossRef]
- Li, J.; Li, C.; Yang, S.; An, Y.; Wu, W.; Jiang, H. Assembly of 3-Sulfenylbenzofurans and 3-Sulfenylindoles by Palladium-Catalyzed Cascade Annulation/Arylthiolation Reaction. J. Org. Chem. 2016, 81, 2875–2887. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T. B. Recent Advances in Organic Reactions Involving Elemental Sulfur. Adv. Syn. Catal. 2017, 359, 1066–1130. [Google Scholar] [CrossRef]
- Qiao, Z.; Wei, J.; Jiang, X. Direct Cross-Coupling Access to Diverse Aromatic Sulfide: Palladium-Catalyzed Double C–S Bond Construction Using Na2S2O3 as a Sulfurating Reagent. Org Lett. 2014, 16, 1212–1215. [Google Scholar] [CrossRef]
- Reeves, J. T.; Camara, K.; Han, Z. S.; Xu, Y.; Lee, H.; Busacca, C. A.; Senanayake, C. H. The Reaction of Grignard Reagents with Bunte Salts: A Thiol-Free Synthesis of Sulfides. Org. Lett. 2014, 16, 1196–1199. [Google Scholar] [CrossRef]
- Li, Y.; Wang, M.; Jiang, X. Controllable Sulfoxidation and Sulfenylation with Organic Thiosulfate Salts via Dual Electron- and Energy-Transfer Photocatalysis, ACS Catal. 2017, 7, 7587–7592. [CrossRef]
- Luo, F.; Pan, C.; Li, L.; Chen, F.; Cheng, J. Copper-Mediated Methylthiolation of Aryl Halides with DMSO. Chem. Commun 2011, 47, 5304–5036. [Google Scholar] [CrossRef]
- Cao, L.; Luo, S. H.; Wu, H. Q.; Chen, L. Q.; Jiang, K.; Hao Z., F.; Wang, Z. Y. ; Copper(I)-Catalyzed Alkyl- and Arylsulfenylation of 3,4-Dihalo-2(5H)-Furanones (X=Br, Cl) with Sulfoxides under Mild Conditions. Adv. Synth. Catal. 2017, 359, 2961–2971. [Google Scholar] [CrossRef]
- Wang, F.; Rao, W.; Wang, S. Nickel-Catalyzed Reductive Thiolation of Unactivated Alkyl Bromides and Arenesulfonyl Cyanides. J. Org. Chem. 2021, 86, 8970–8979. [Google Scholar] [CrossRef]
- Zhang, C.; Zhou, Y.; Huang, J.; Tu, C.; Zhou, X.; Yin, G. Cesium Carbonate-Promoted Synthesis of Aryl Methyl Sulfides Using S-Methylisothiourea Sulfate under Transition-Metal-Free Conditions. Org. Biomol. Chem. 2018, 16, 6316–6321. [Google Scholar] [CrossRef]
- Saravanan, P.; Anbarasan, P. Palladium Catalyzed Aryl (Alkyl)Thiolation of Unactivated Arenes. Org. Lett. 2014, 16, 848–851. [Google Scholar] [CrossRef]
- Hostier, T.; Ferey, V.; Ricci, G.; Pardo, D. G.; Janine Cossy, J. Synthesis of Aryl Sulfides: Metal-Free C–H Sulfenylation of Electron-Rich Arenes. Org. Lett. 2015, 17, 3898–3901. [Google Scholar] [CrossRef]
- Arisawa M.; Yamagushi, M. Rhodium-Catalyzed Synthesis of Organosulfur Compounds Involving S-S Bond Cleavage of Disulfides and Sulfur, Molecules, 2020, 25, 3595. [CrossRef]
- Bao, Y.; Yang, X.; Dai, Z.; Ji, S.; Zhou, Q.; Yang, F. Iodine-Promoted Tunable Synthesis of 2-Naphthyl Thioethers and 1-Naphthyl Thioethers. Adv. Syn. Catal 2019, 361, 2154–2158. [Google Scholar] [CrossRef]
- Duan, Y.; Guo, Z.; Zheng, T.; Lu, Y.; XU, J.; Liu, J.; Yang, F. Iodine-Promoted Reductive Sulfenylation Using Ketones as Hydride Donors. J. Org. Chem. 2024, 89, 5851–5856. [Google Scholar] [CrossRef]
- Chen, P.; Tian, L.; Ji. X.; Deng, G. J.; Huang, H. Copper-Catalyzed 1,2-Sulfonyletherification of 1,3-Dienes. Org. Lett., 2024, 26, 2939–2944. [CrossRef]
- Wang, Y.; Zhang, X.; Liu, H.; Chen, H.; Huang, H. Nickel-catalyzed Direct Formation of the C–S Bonds of aryl Sulfides from Arylsulfonyl Chlorides and Aryl Iodides using Mn as a Reducing Agent. Org. Chem. Front. 2017, 4, 31–36. [Google Scholar] [CrossRef]
- Zhong, S.; Zhou, Z.; Zhao, F.; Mao, G.; Deng, G. J.; Huang, H. Deoxygenative C–S Bond Coupling with Sulfinates via Nickel/Photoredox Dual Catalysis, Org. Lett., 2022, 24, 9, 1865–1870. [CrossRef]
- Qiao, Z.; Ge, N.; Jiang, X. CO2-promoted Oxidative Cross-coupling Reaction for C–S bond Formation via Masked Strategy in an Odourless Way. Chem. Commun 2015, 51, 10295–10298. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Cui, W.; Song, X.; Xu, G.; Jiang, M.; Sun, K.; Lv, J.; Yang, D. Sulfonylation of Aryl Halides by Visible Light/Copper Catalysis. Org. Lett. 2021, 23, 3663–3668. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Gui, Y.; Yu, B.; Jin, Y.; Tian, S. K. By-Product-Catalyzed Redox-Neutral Sulfenylation/Deiodination/Aromatization of Cyclic Alkenyl Iodides with Sulfonyl Hydrazides. Adv. Syn.Catal. 2016, 358, 3368–3372. [Google Scholar] [CrossRef]
- Saini, Y.; Khungar, B. Recyclable Imidazolium Ion-tagged Nickel Catalyst for Microwave-assisted C–S Cross-coupling in Water using Sulfonyl Hydrazide as the Sulfur Source. New J. Chem. 2018, 42, 12796–12801. [Google Scholar] [CrossRef]
- Bulmer, G.; Mann, F. G. The Chemistry of Xanthic Acid Derivatives. Part I. The Preparation and Comparative Properties of Isomeric Xanthates and Dithiocarbonates, J. Chem. Soc., 1945, 666-674. [CrossRef]
- Zard, S. Z. The Xanthate Route to Ketones: When the Radical Is Better than the Enolate. Acc. Chem. Res. 2018, 51, 1722–1733. [Google Scholar] [CrossRef]
- Debien, L.; Quiclet-Sire, B.; Zard, S. Z. Allylic Alcohols: Ideal Radical Allylating Agents? Acc. Chem. Res. 2015, 48, 1237–1253. [Google Scholar] [CrossRef] [PubMed]
- Soleiman-Beigi, M.; Arzehgar, Z. A Novel Method for the Direct Synthesis of Symmetrical and Unsymmetrical Sulfides and Disulfides from Aryl Halides and Ethyl Potassium Xanthogenate, Synlett 2018; 29, 986-992. [CrossRef]
- SoKolov, A. I.; Mikhaylov, A. A.; Baleeva,N. S.; Baranov, M. S. Xanthates as Thiol Surrogates for Nucleophilic Substitution with Aryl Halides. Eur J Org Chem. 2021, 30, 4350–4357. [CrossRef]
- Akkilagunta V., K.; Kakulapati, R. R. Synthesis of Unsymmetrical Sulfides Using Ethyl Potassium Xanthogenate and Recyclable Copper Catalyst under Ligand-Free Conditions. J. Org. Chem. 2011, 76, 6819–6824. [Google Scholar] [CrossRef]
- Volkov, A. A.; Bugaenko, D. I.; Bogdanov, A. V.; Karchava, A. V. Visible-Light-Driven Thioesterification of Aryl Halides with Potassium Thiocarboxylates: Transition-Metal Catalyst-Free Incorporation of Sulfur Functionalities into an Aromatic Ring. J. Org. Chem., 2022, 87, 8170–8182. [Google Scholar] [CrossRef]
- Bugaenko, D. T.; Volkov, A. A.; Karchava. A. V. A Thiol-Free Route to Alkyl Aryl Thioethers. J. Org. Chem. 2023, 88, 9968–9972. [CrossRef]
- Degani, I.; Fochi, R.; Regondi. Phase-Transfer Synthesis of Symmetrical S,S-dialkyl Dithiocarbonates via o-Methyl S-alkyl Dithiocarbonates. Synthesis 1980, 5, 375–378. [CrossRef]
- He, R.; Liu, Y.; Feng, Y.; Chen, L.; Huang, Y.; Xie, F.; Li, Y. Access to Thienopyridine and Thienoquinoline Derivatives via Site-selective C–H Bond Functionalization and Annulationm. Org. Lett. 2022, 24, 3167–3172. [Google Scholar] [CrossRef]
- Luo, X.; He, R.; Liu, Q.; Gao, Y.; Li, J.; Chen, X.; Zhu, Z.; Huang, Y.; Li, Y. Metal-free Oxidative Esterification of Ketones and Potassium Xanthates: Selective Synthesis of α-Ketoesters and Esters, J. Org. Chem., 2020, 85, 5220-5230. [CrossRef]
- Feng, Y.; Nie, J.; Sijie Xie, S.; He, Z.; Hong, H.; Li, J.; Huang, Y.; Chen, L; Li, Y. Potassium Xanthate–promoted Reductive Sulfuration Reaction: from Aldehydes to Thiol, Disulfide, and Thioester Derivatives. Chem. Commun. 2024, 59, 1140–1143. [CrossRef]
- Harada, T.; Karasawa, A.; Oku, A. A new Synthetic Route to Vinyl Sulfides Utilizing the Reaction of (Phenylthio)carbenes with Nitrile Anions. J. Org. Chem. 1986, 51, 842–846. [Google Scholar] [CrossRef]
- Hua, Y.; Zhang, W.; Wang, X.; Ge, Z.; Li, R. Synthesis of novel five-membered endocyclic sulfoximines-1-aryl-3-(arylimino)-3,5-dihydro-1,4,2-dithiazole 1-oxides, Tetrahedron, 2017, 73, 4387-4391. [CrossRef]
- Li, J.; Liu, Y.; Chen, Z.; Li, J.; Ji, X.; Chen, L.; Huang, Y.; Liu, Q.; Li, Y. Synthesis of Substituted Thiophenes through Dehydration and Heterocyclization of Alkynols. J. Org. Chem 2022, 87, 3555–3566. [Google Scholar] [CrossRef]
- Li, Y.; Cheng, L.; Liu, X.; Li, B.; Sun, N. Copper-promoted Hydration and Annulation of 2-Fluorophenylacetylene Derivatives: from Alkynes to Benzo[b]furans and benzo[b]thiophenes. Beilstein J. Org. Chem 2014, 10, 2886–2891. [Google Scholar] [CrossRef]
- Khan, M. Y.; Cho, M. S.; Kwark, Y. J. Dual Roles of a Xanthate as a Radical Source and Chain Transfer Agent in the Photoinitiated RAFT Polymerization of Vinyl Acetate, Macromolecules 2014, 47, 1929-1934. [CrossRef]
- Erian, A. W.; Reid, D. L.; Warkentin, J. Reactions of Dimethoxycarbene with Xanthates. J. Sulfur Chem 2005, 26, 203–209. [Google Scholar] [CrossRef]
- Liu, Q.; Ci, C.; Zhao, H.; Xie, R.; Jiang, H.; Zhang, M. Direct Access to Functional Phenazines via Oxidative Annulation of Anilines and o-Phenylenediamines with a Reusable cobalt Catalyst. Green Chem 2023, 25, 678–683. [Google Scholar] [CrossRef]
- Tan, Z.; Liang, Y.; Yang, L.; Cao, L.; Jiang, H. Tan, Z.; Liang, Y.; Yang, L.; Cao, L.; Jiang, H. Zhang, M. Site-Specific Oxidative C−H Chalcogenation of (Hetero)Aryl-Fused Cyclic Amines Enabled by Nanocobalt Oxides. Org. Lett. 2018, 20, 6554–6558. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Wu, Q.; Gong, X.; Ao, G.; Liu, F. A Facile Method for Hydroxytrifluoromethylation of Alkenes with Langlois Reagent and DMSO. Green Chem 2019, 21, 2983–2987. [Google Scholar] [CrossRef]
- Cao, X. H.; Pan, X.; Zhou, P. J.; Asekun, O. T. Manganese(iii)-mediated direct Csp2–H Radical Trifluoromethylation of Coumarins with Sodium Trifluoromethanesulfinate. Chem. Commun. 2014, 50, 3359–3362. [Google Scholar] [CrossRef]
- Itoh, T.; Mase, T. A General Palladium-Catalyzed Coupling of Aryl Bromides/Triflates and Thiols. Org. Lett. 2004, 6, 4587–4590. [Google Scholar] [CrossRef]
- Chen, B.; Wang, J.; Sun, W.; Pan, Y.; Ding, A.; Liu, W.; Guo, H. Thioxanthone-TfOH Complex (9-HTXTF) Photoredox Enabled Reduction of Sulfoxides, Tetrahedron Letters, 2024, 139, 155007. [CrossRef]
- Liu, Y.; Kim, J.; Seo, H.; Park, S.; Chae, J. Copper(II)-Catalyzed Single-Step Synthesis of Aryl Thiols from Aryl Halides and 1,2-Ethanedithiol. Adv. Synth. Catal. 2015, 357, 2205–2212. [Google Scholar] [CrossRef]
- Ryota, I.; Miki, B.; Kei, M.; Junichiro, Y. Ni-Catalyzed Aryl Sulfide Synthesis through an Aryl Exchange Reaction. J. Am. Chem. Soc., 2021, 143, 10333–10340. [Google Scholar] [CrossRef]
- Zhang, H.; Cao, W.; Ma, D. L-Proline-Promoted CuI-Catalyzed C-S Bond Formation between Aryl Iodides and Thiols. Synth. Commun. 2007, 37, 25–35. [Google Scholar] [CrossRef]
- Anuradha, N.; Imran, K.; Somraj, G.; Govindasamy, S. Visible-Light-Driven Halogen-Bond-Assisted Direct Synthesis of Heteroaryl Thioethers Using Transition-Metal-Free One-Pot C–I Bond Formation/C–S Cross-Coupling Reaction, J. Org. Chem., 2021, 86, 2570−2581. [CrossRef]
- Benjamin, H.; Moritz, B.; Paul, K. Thiolation of Pyridine-2-sulfonamides using Magnesium Thiolates, Synthesis, 2019, 51, 4452-4462. [CrossRef]
- Forrest, H. S.; Walker, J. Chemotherapeutic agents of the Sulphone type. Part V. 2: 5-Disubstituted Derivatives of Pyridine, J. Chem. Soc., 1948, 1939-1945. [CrossRef]
- Beugelmansm, R.; Bois-Choussy, M.; Boudet, B. Etude des Reactions de srn1-partie 10: Action de Sulfanions sur les Halogenures d'aryle Fonctionnalises. Synthese Directe de Benzothiophenes et Thienopyridines, Tetrahedron, 1983, 39, 4153-4161. [CrossRef]
- M. Obe; K. Tanaka; K. Nishiyama; W. Ando. Aerobic Oxidation of Thiols to Disulfides Catalyzed by Diaryl Tellurides under Photosensitized Conditions, J. Org. Chem., 2011, 76, 4173-4177. [CrossRef]
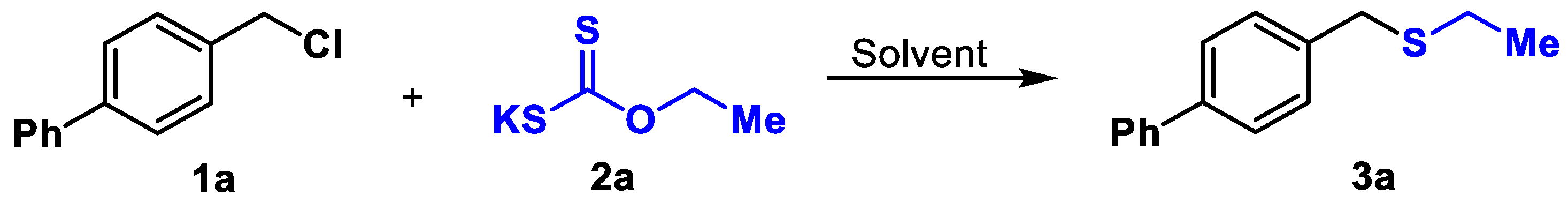
| Entry | EtOCS2K (mmol) | Solvent (mL) | T (°C) | T (h) | Yield (%) |
| 1 | EtOCS2K (1) | DMF (2) | 150 | 6 | 77 |
| 2 | EtOCS2K (1) | o-xylene (2) | 150 | 6 | <5 |
| 3 | EtOCS2K (1) | DMSO (2) | 150 | 6 | 95 |
| 4 | EtOCS2K (1) | DMAc (2) | 150 | 6 | 57 |
| 5 | EtOCS2K (1) | NMP (2) | 150 | 6 | 45 |
| 6 | EtOCS2K (1) | DMSO (1) | 150 | 6 | 94 |
| 7 | EtOCS2K (0.8) | DMSO (1) | 150 | 6 | 78 |
| 8 | EtOCS2K (1) | DMSO (1) | 130 | 6 | 95 |
| 9 | EtOCS2K (1) | DMSO (1) | 100 | 6 | 94 |
| 10 | EtOCS2K (1) | DMSO (1) | 80 | 6 | 89 |
| 11 | EtOCS2K (1) | DMSO (1) | 100 | 3 | 94 |
| 12 | EtOCS2K (1) | DMSO (1) | 100 | 1 | 93 |
| 13 | EtOCS2K (1) | DMSO (1) | 100 | 0.5 | 87 |
| Entry | EtOCS2K (mmol) | Additive (mmol) | Solvent | Time (h) | Yield (%) |
| 1 | EtOCS2K (1.2) | I2 (1.5) | DMF | 24 | 37 |
| 2 | EtOCS2K (1.2) | I2 (1.5) | DMF | 30 | 45 |
| 3 | EtOCS2K (1.2) | I2 (1.5) | DMF | 36 | 92 |
| 4 | EtOCS2K (0.8) | I2 (1.5) | DMF | 48 | 69 |
| 5 | EtOCS2K (1.2) | I2 (0.7) | DMF | 36 | 10 |
| 6 | EtOCS2K (1.2) | NH4I (1.5) | DMF | 36 | <5 |
| 7 | EtOCS2K (1.2) | HI (1.5) | DMF | 36 | 15 |
| 8 | EtOCS2K (1.2) | I2 (1.5) | DMSO | 36 | <5 |
| 9 | EtOCS2K (1.2) | I2 (1.5) | NMP | 36 | <5 |
| 10 | EtOCS2K (1.2) | I2 (1.5) | DMAc | 36 | 43 |
| 11c | EtOCS2K (1.2) | I2 (1.5) | DMF | 36 | 39 |
| 12 | EtOCS2K (1.2) | - | DMF | 36 | <5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




