Submitted:
11 May 2024
Posted:
13 May 2024
You are already at the latest version
Abstract
Keywords:
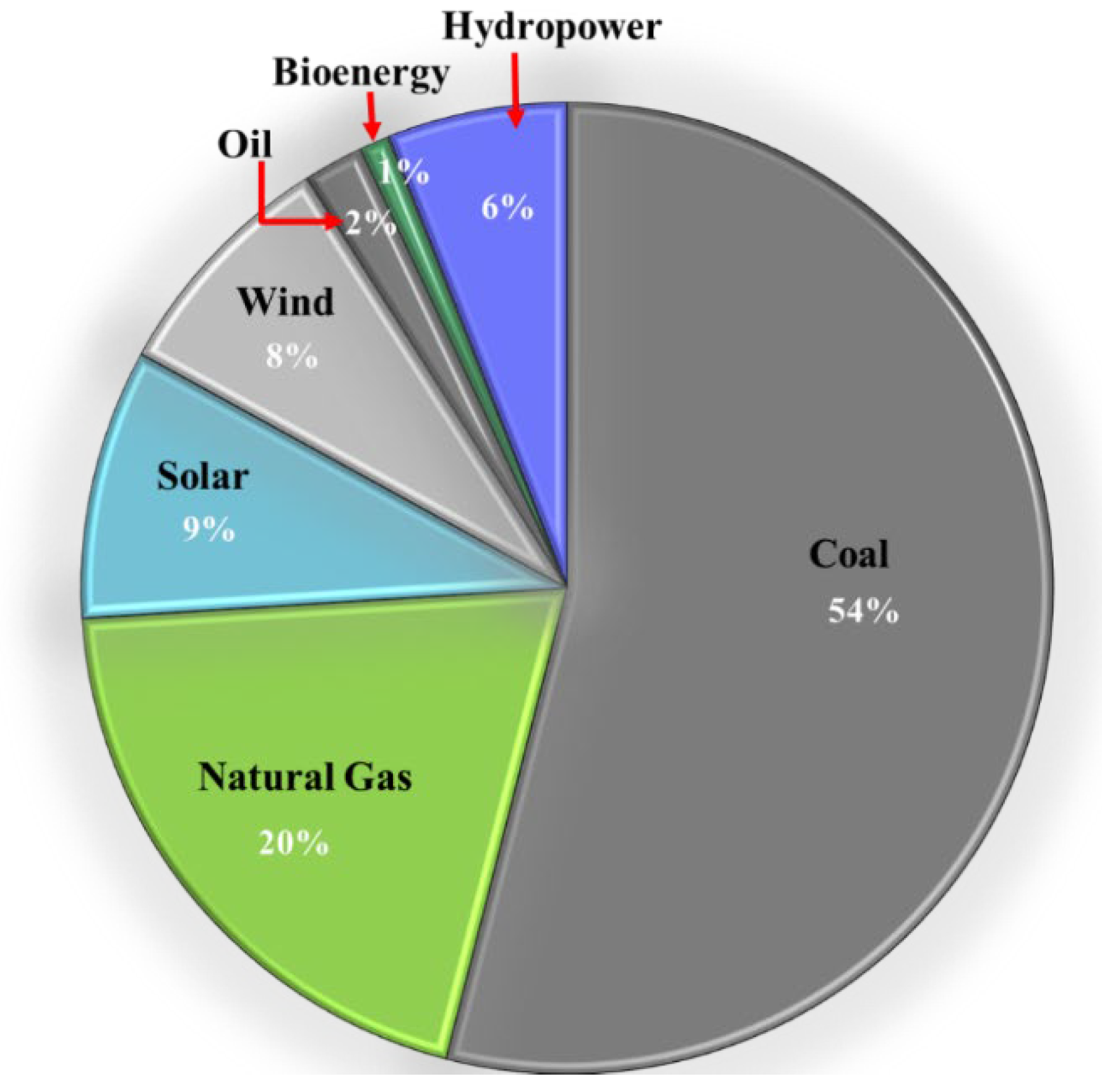
1. Introduction
1.1. Background
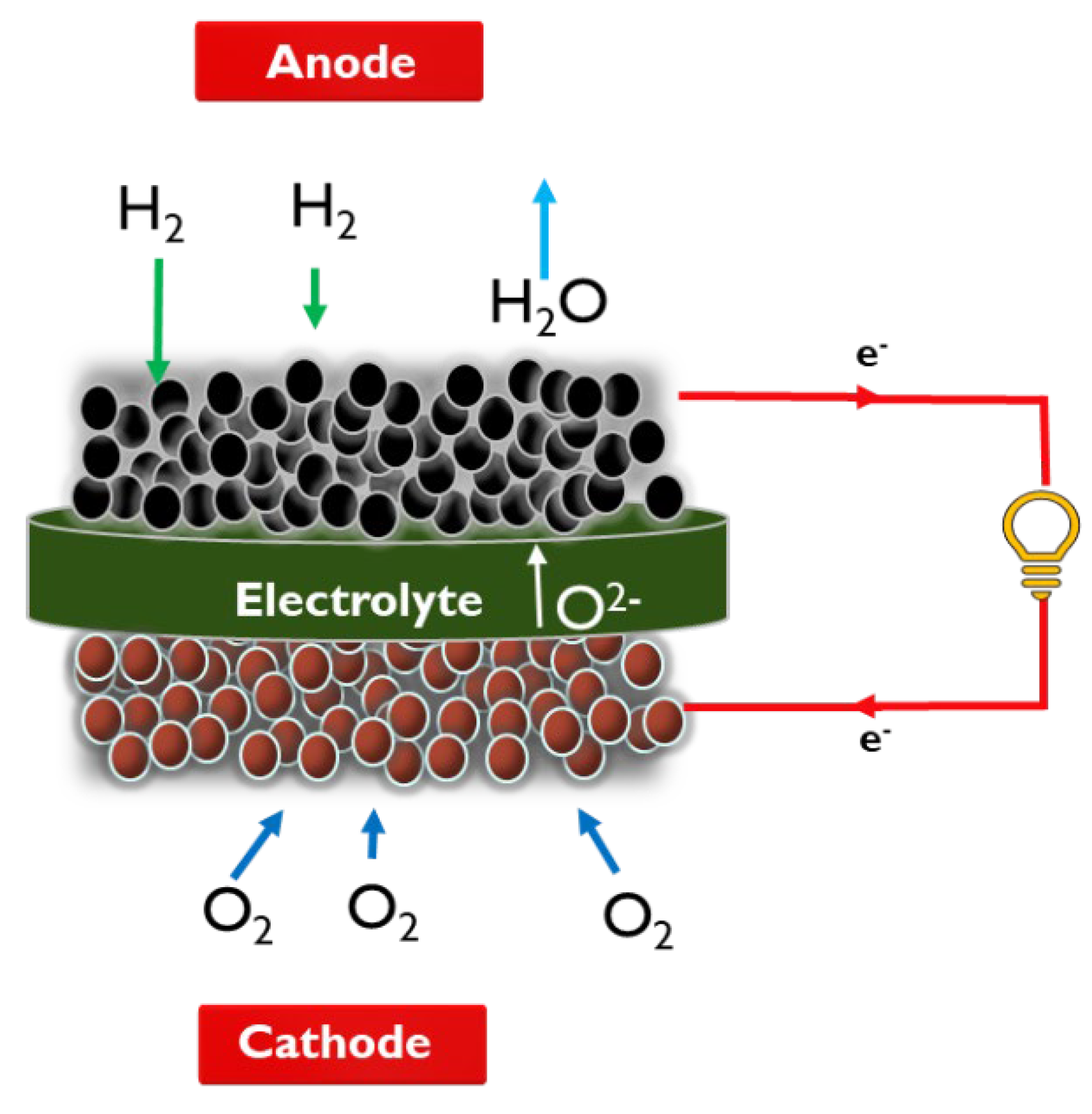
1.2. Fundamentals of SOFC
2. Materials and Requirements of Different Cell Components
2.1. Anode
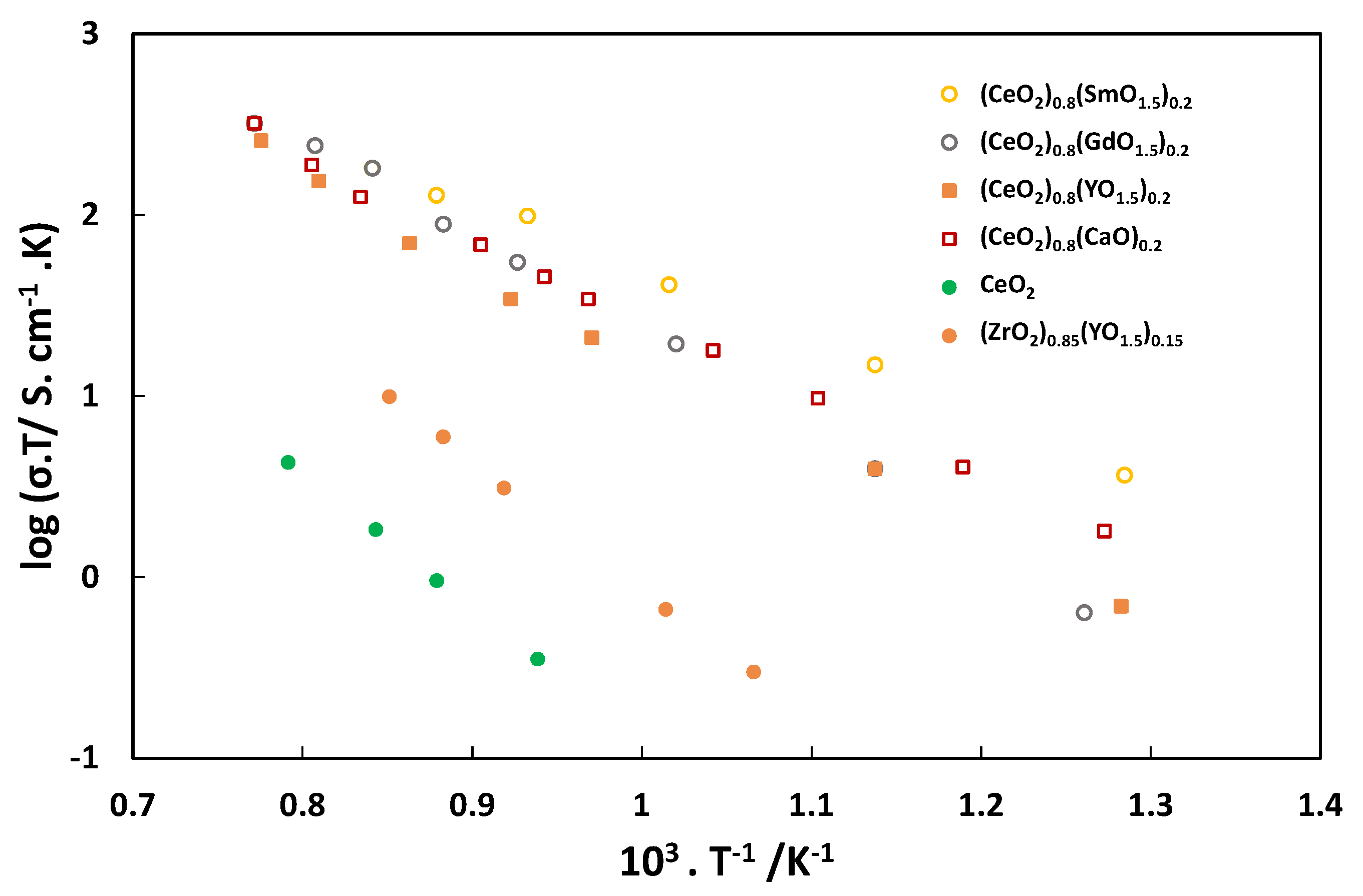
2.2. Electrolyte
2.3. Cathode
3. Strategies for Improvement in the Performance of Cathode Materials
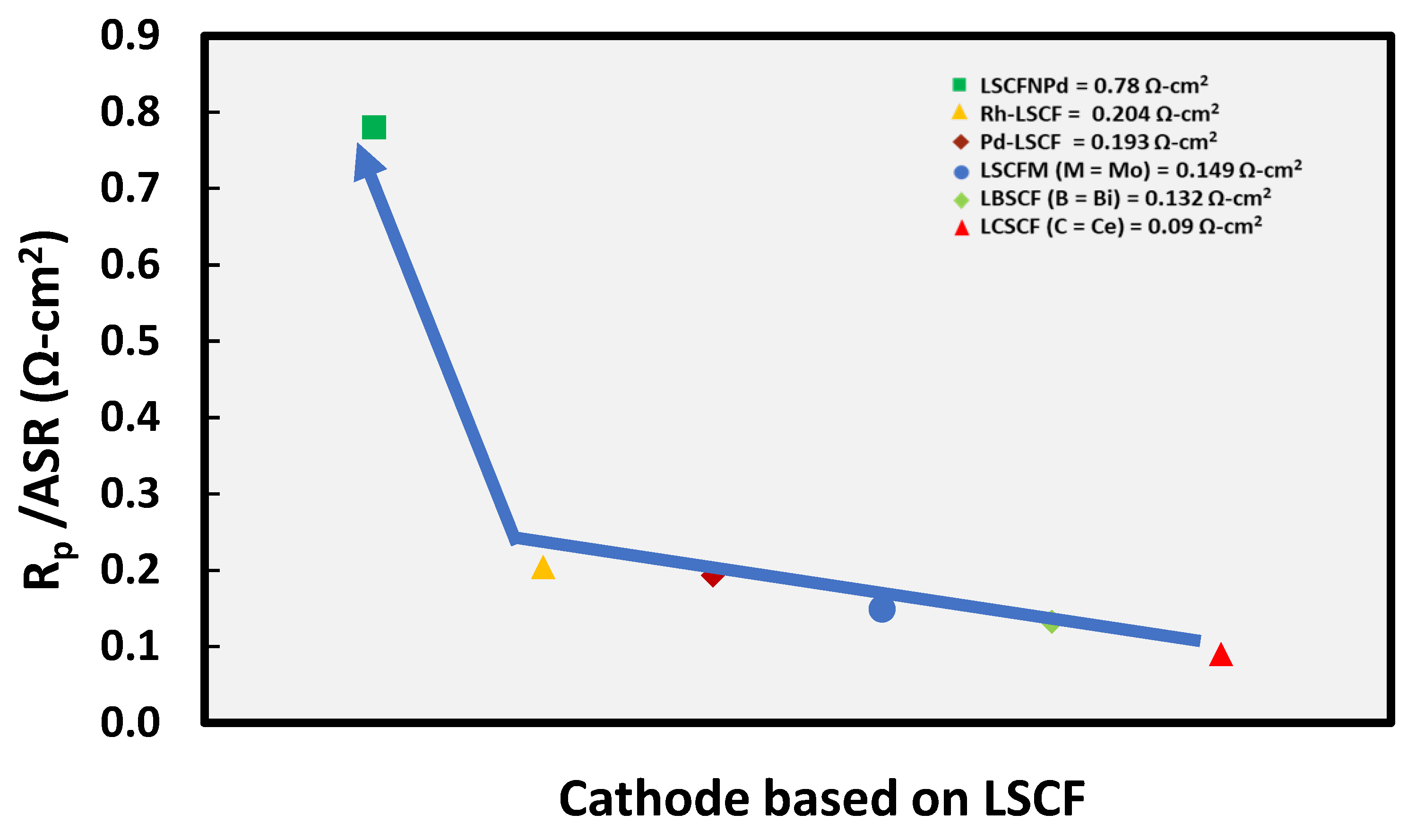
3.1. Doping
Effect on Structural Stability
Effect on Conductivity
Effect on Coefficient of Thermal Expansion (TEC)
3.2. Composites
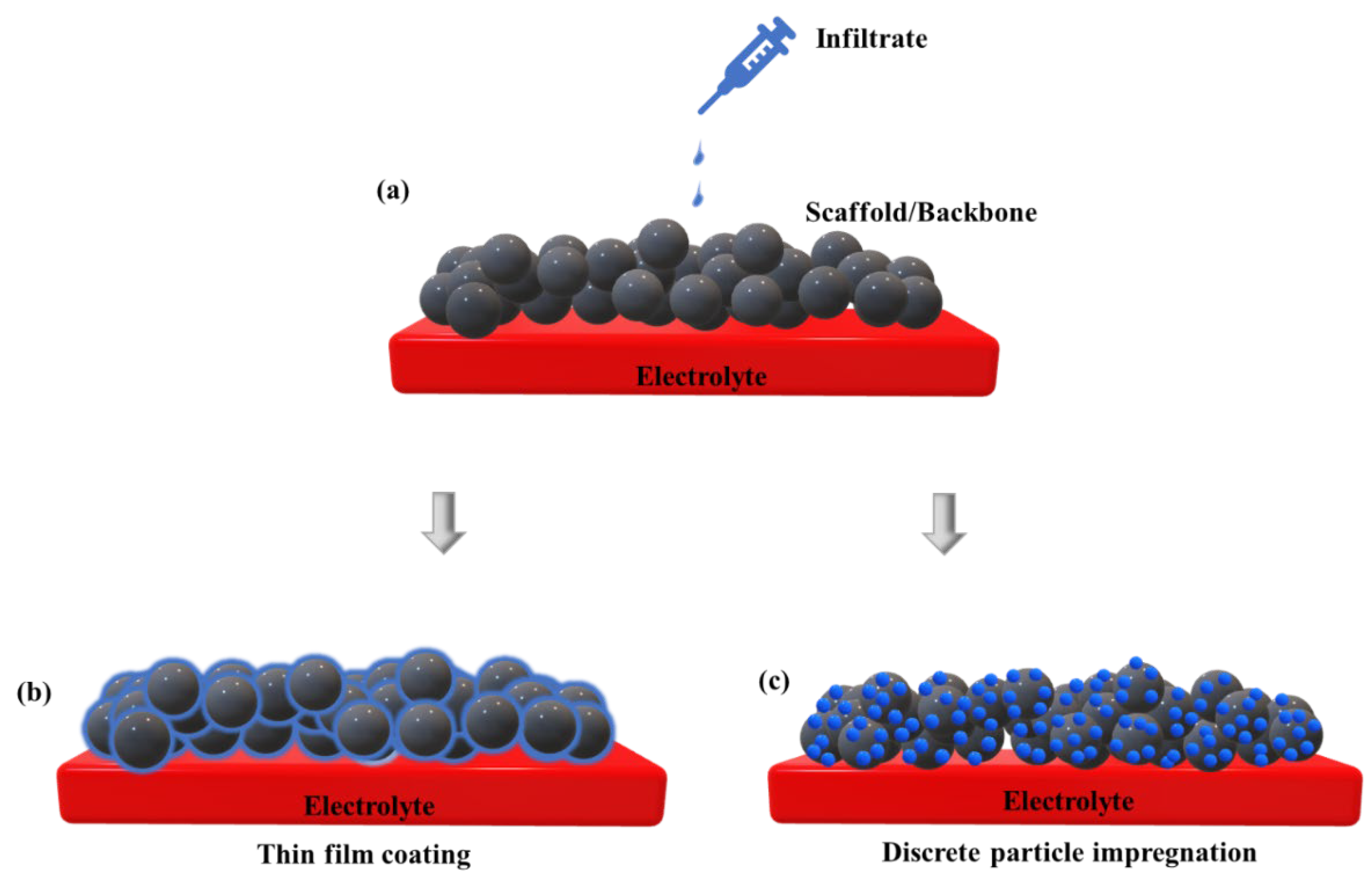
3.3. Infiltration/Impregnation
Effect of Infiltration on TPB
3.4. Core-Shell Composites
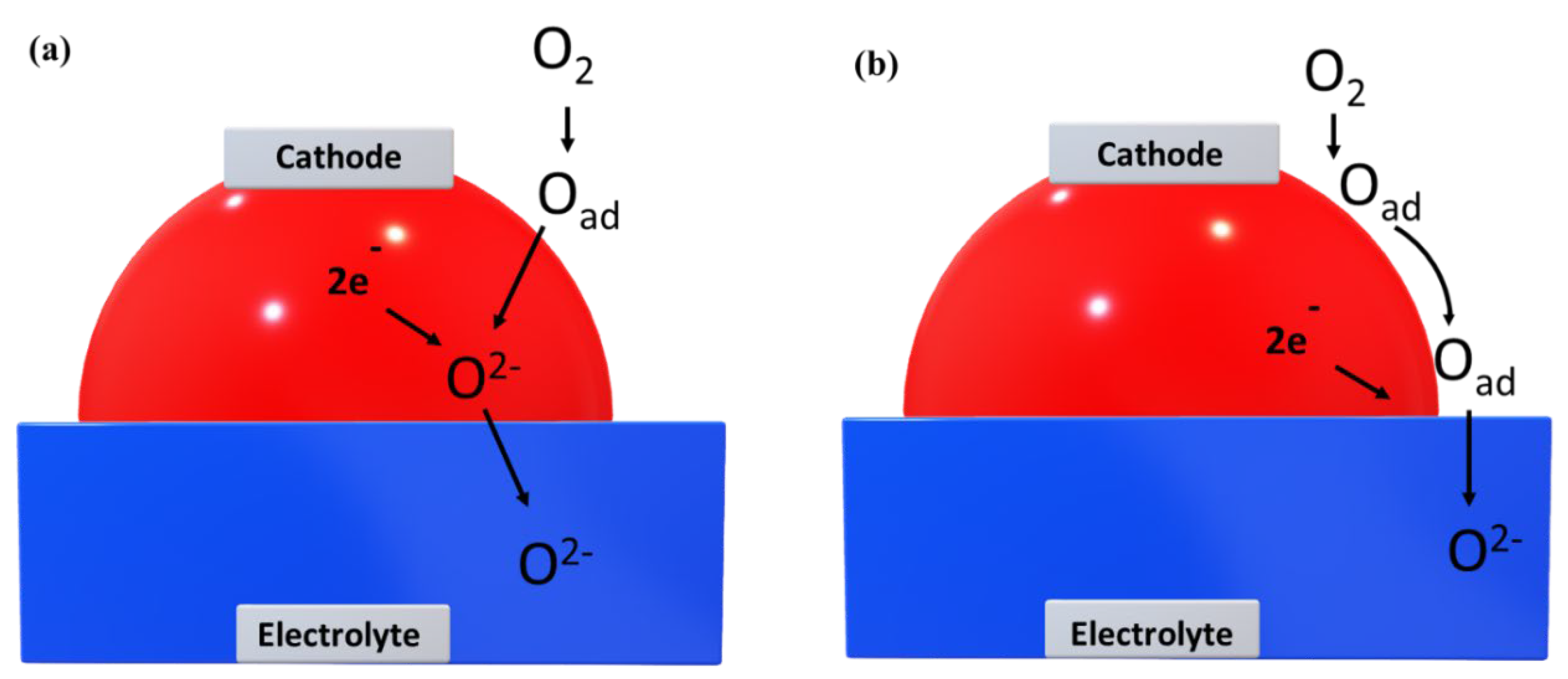
3.5. Interface /Interfacial Layer Modifications
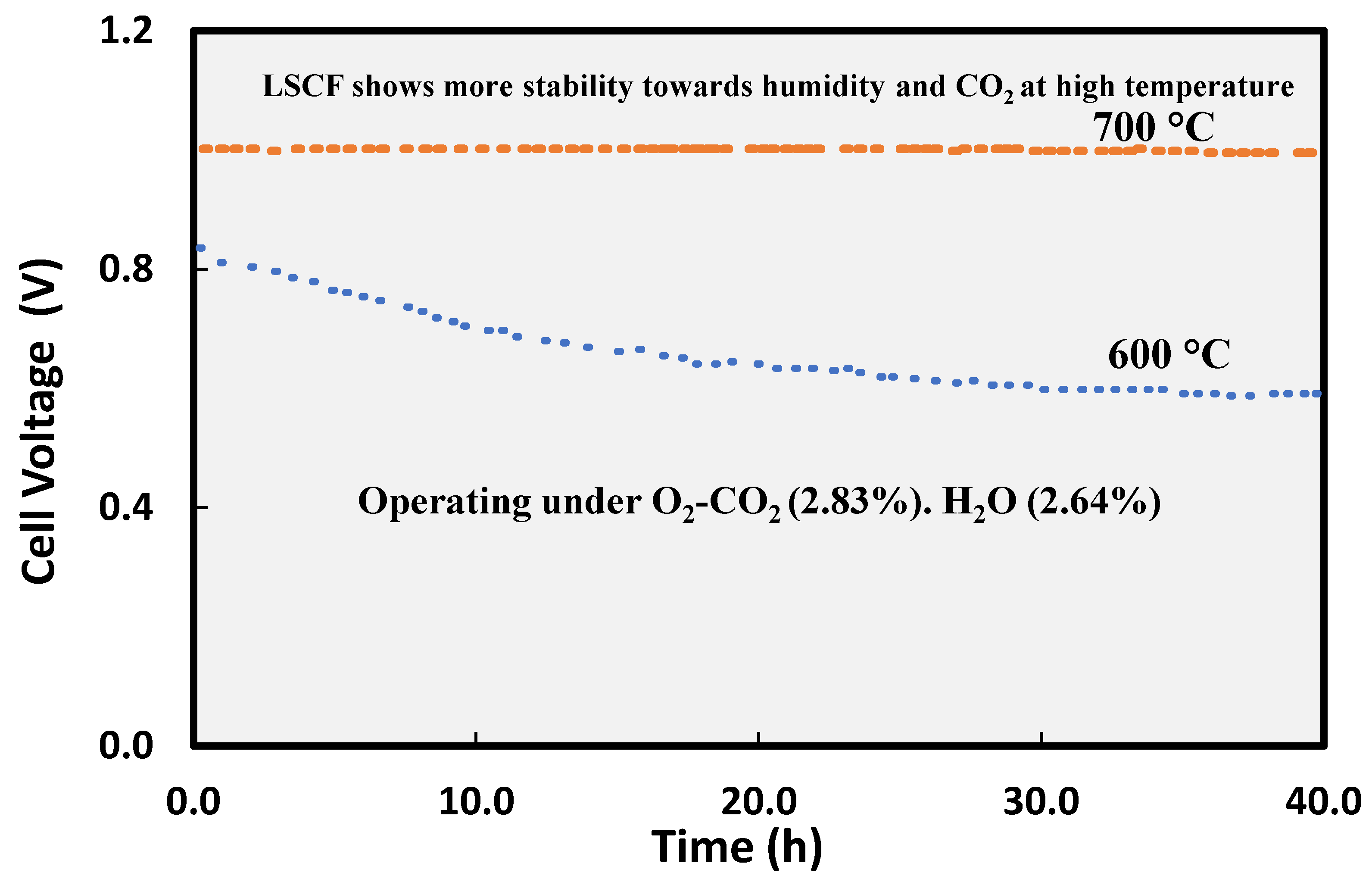
4. Modifications to Cathode to Mitigate Carbon Dioxide (CO2) and Humidity Effect
5. Conclusion
Acknowledgment
Abbreviation
| SOFC | Solid Oxide Fuel Cell |
| HT-SOFC | High-Temperature solid oxide fuel cell |
| IT-SOFC | Intermediate Temperature solid oxide fuel cell |
| SOEC | Solid oxide electrolysis cell |
| PEMFC | Polymer electrolyte membrane fuel cell |
| ORR | Oxygen reduction reaction |
| TPB | Triple phase boundary |
| NOx | Oxides of nitrogen |
| SOx | Oxides of Sulphur |
| DES | Distributed Energy system |
| BOP | Balance of plant |
| NG | Natural gas |
| OCV | Open circuit voltage |
| ABO3 | A general formula for Perovskites |
| MIEC | Mixed ionic and electronic conductor |
| Ni-YSZ | Nickle-Yttrium stabilized zirconia |
| YSZ | Yttrium stabilized zirconia |
| ScSZ | Scandium stabilized zirconia |
| GDC | Gadolinium doped ceria |
| SDC | Samarium doped ceria |
| LSGM | Strontium magnesium doped Lanthanum gallate |
| LSM | Lanthanum strontium manganite |
| LSCF | Lanthanum (La) Strontium (Sr) Cobalt (Co) ferrite |
| BSCF | Barium (Ba) Strontium (Sr) Cobalt (Co) ferrite |
| SSC | Samarium (Sm) doped Strontium (Sr) cobalt oxide |
| R&D | Research and development |
| ASR | Area-specific resistance |
| TGA | Thermal Gravitational Analysis |
| SEM | Scanning Electron Microscope |
| SIMS | Secondary Ion Mass Spectroscopy |
| EDS | Energy Dispersive Spectroscopy |
| Symbols | |
| δ | Oxygen non-stoichiometry |
| K−1 | Per Kelvin |
| S.cm−1 | Siemens per centimetre |
| Vf | Free oxygen volume |
| D* | Electronic conductivity |
| Rp | Cathode Polarization/resistance |
| Rohm | Electrolyte polarization/resistance |
| Ea | Activation energy |
| R | Ionic radius |
| a,b,c | Lattice parameters of the cubic system along with three the dimensions |
| Ω.cm2 | Ohms centimetre square |
| W.cm−2 | Watt per centimetre square |
| mW.cm−2 | Milliwatt per centimetre square |
| Ln | Rare earth metals |
References
- Australia’s Department of Industry, Innovation, and Science, Office of the Chief Economist, Australian Energy Update 2021, pg 27-30.
- Chen, J.M.P.; Ni, M. Economic analysis of a solid oxide fuel cell cogeneration/trigeneration system for hotels in Hong Kong. Energy Build. 2014, 75, 160–169. [Google Scholar] [CrossRef]
- Baldi, F.; Wang, L.; Pérez-Fortes, M.; Maréchal, F. A Cogeneration System Based on Solid Oxide and Proton Exchange Membrane Fuel Cells With Hybrid Storage for Off-Grid Applications. Front. Energy Res. 2019, 6. [Google Scholar] [CrossRef]
- Song, B.; Ruiz-Trejo, E.; Bertei, A.; Brandon, N.P. Quantification of the degradation of Ni-YSZ anodes upon redox cycling. J. Power Sources 2018, 374, 61–68. [Google Scholar] [CrossRef]
- Jiao, Z.; Lee, G.; Shikazono, N.; Kasagi, N. Quantitative Study on the Correlation Between Solid Oxide Fuel Cell Ni-YSZ Composite Anode Performance and Sintering Temperature Based on Three-dimensional Reconstruction. J. Electrochem. Soc. 2012, 159, F278–F286. [Google Scholar] [CrossRef]
- Iwata, T. Characterization of Ni-YSZ Anode Degradation for Substrate-Type Solid Oxide Fuel Cells. J. Electrochem. Soc. 1996, 143, 1521–1525. [Google Scholar] [CrossRef]
- Liu, Y.; Shao, Z.; Mori, T.; Jiang, S.P. Development of nickel based cermet anode materials in solid oxide fuel cells – Now and future. Mater. Rep. Energy 2021, 1, 100003. [Google Scholar] [CrossRef]
- Vafaeenezhad, S.; Hanifi, A.R.; A Laguna-Bercero, M.; Etsell, T.H.; Sarkar, P. Microstructure and long-term stability of Ni–YSZ anode supported fuel cells: a review. Mater. Futur. 2022, 1, 042101. [Google Scholar] [CrossRef]
- Heidarpour, A.; Saidi, A.; Abbasi, M.; Choi, G. In situ fabrication mechanism of a dense Sr and Ca doped lanthanum chromite interconnect on Ni-YSZ anode of a solid oxide fuel cell during co-sintering. Ceram. Int. 2013, 39, 1821–1826. [Google Scholar] [CrossRef]
- Horita, T.; Tsunoda, T.; Yamaji, K.; Sakai, N.; Kato, T.; Yokokawa, H. Microstructures and oxygen diffusion at the LaMnO3 film/yttria-stabilized zirconia interface. Solid State Ionics 2002, 152-153, 439–446. [Google Scholar] [CrossRef]
- Hussain, S.; Yangping, L. Review of solid oxide fuel cell materials: cathode, anode, and electrolyte. Energy Transitions 2020, 4, 113–126. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, M.; Huang, J.; Song, Z.; Fu, Y.; Chang, Y.; Li, C.; He, T. Improved thermal expansion and electrochemical performances of Ba0.6Sr0.4Co0.9Nb0.1O3−δ–Gd0.1Ce0.9O1.95 composite cathodes for IT-SOFCs. Int. J. Hydrogen Energy 2014, 39, 7972–7979. [Google Scholar] [CrossRef]
- Yin, S.; Li, M.; Zeng, Y.; Li, C.; Chen, X.; Ye, Z. Study of Sm0.2Ce0.8O1.9 (SDC) electrolyte prepared by a simple modified solid-state method. J. Rare Earths 2014, 32, 767–771. [Google Scholar] [CrossRef]
- Fan, B.; Yan, J.; Yan, X. The ionic conductivity, thermal expansion behavior, and chemical compatibility of La0.54Sr0.44Co0.2Fe0.8O3-δ as SOFC cathode material. Solid State Sci. 2011, 13, 1835–1839. [Google Scholar] [CrossRef]
- Jia, W.; Huang, Z.; Sun, W.; Wu, L.; Zheng, L.; Wang, Y.; Huang, J.; Yang, X.; Lv, M.; Ge, L. Flexible A-site doping La0.6-xMxSr0.4Co0.2Fe0.8O3 (M=Ca, Ba, Bi; x=0, 0.1, 0.2) as novel cathode material for intermediate-temperature solid oxide fuel cells: A first-principles study and experimental exploration. J. Power Sources 2021, 490, 229564. [Google Scholar] [CrossRef]
- Jung, W. and H.L. Tuller, Investigation of surface Sr segregation in model thin film solid oxide fuel cell perovskite electrodes. Energy & Environmental Science, 2012. 5(1): p. 5370-5378.
- Liu, Q. , et al., Perovskite Sr2Fe1.5Mo0.5O6−δ as electrode materials for symmetrical solid oxide electrolysis cells. International Journal of Hydrogen Energy, 2010. 35(19): p. 10039-10044.
- Dey, S. , et al., Synthesis and characterization of Nanocrystalline Ba0·6Sr0·4Co0·8Fe0·2O3 for application as an efficient anode in solid oxide electrolyser cell. International Journal of Hydrogen Energy, 2020. 45(7): p. 3995-4007.
- Stevenson, J.W. , et al., Electrochemical Properties of Mixed Conducting Perovskites La1 − x M x Co1 − y Fe y O 3 − δ (M = Sr, Ba, Ca). Journal of The Electrochemical Society, 1996. 143(9): p. 2722-2729.
- Anbo, Y.; Tian, X.; Liping, S.; Qiang, L.; Lihua, H.; Hui, Z. Effects of rare earth doping on electrochemical properties of NdBaCo2O6- cathode materials. J. Alloy. Compd. 2020, 837. [Google Scholar] [CrossRef]
- Juliao, P.S.B. A-site cation influences on performance, structure and conductivity of a lanthanide-based perovskite electrode for symmetrical solid oxide fuel cells. J. Power Sources 2020, 450, 227723. [Google Scholar] [CrossRef]
- Yang, X. , et al., Improving stability and electrochemical performance of Ba0.5Sr0.5Co0.2Fe0.8O3-δ electrode for symmetrical solid oxide fuel cells by Mo doping. Journal of Alloys and Compounds, 2020. 831.
- Xu, J. , et al., Characterization of high–valence Mo–doped PrBaCo2O5+ cathodes for IT–SOFCs. Journal of Alloys and Compounds, 2020. 842.
- Sowjanya, C. , et al., Effect of B-site substitution on the crystal structure, electrical conductivity and oxygen transport properties of La0.5Sr0.5M0.2Fe0.8O3-δ (M = Co, Al, and Zn) perovskite. Journal of Solid State Chemistry, 2020. 285.
- Zhou, F.; Liu, Y.; Zhao, X.; Tang, W.; Yang, S.; Zhong, S.; Wei, M. Effects of cerium doping on the performance of LSCF cathodes for intermediate temperature solid oxide fuel cells. Int. J. Hydrogen Energy 2018, 43, 18946–18954. [Google Scholar] [CrossRef]
- Chen, K.; He, S.; Li, N.; Cheng, Y.; Ai, N.; Chen, M.; Rickard, W.D.; Zhang, T.; Jiang, S.P. Nb and Pd co-doped La0.57Sr0.38Co0.19Fe0.665Nb0.095Pd0.05O3-δ as a stable, high performance electrode for barrier-layer-free Y2O3-ZrO2 electrolyte of solid oxide fuel cells. J. Power Sources 2018, 378, 433–442. [Google Scholar] [CrossRef]
- Serra, J.M.; Buchkremer, H.-P. On the nanostructuring and catalytic promotion of intermediate temperature solid oxide fuel cell (IT-SOFC) cathodes. J. Power Sources 2007, 172, 768–774. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, F.; Chen, X.; Wang, C.; Zhong, S. Enhanced electrochemical activity and stability of LSCF cathodes by Mo doping for intermediate temperature solid oxide fuel cells. J. Appl. Electrochem. 2021, 51, 425–433. [Google Scholar] [CrossRef]
- Zhu, Y. , et al., A permeation model study of oxygen transport kinetics of BaxSr1-xCo0.8Fe0.2O3-δ. AlChE journal, 2020. 66(9).
- Weber, V.; Meffert, M.; Wagner, S.; Störmer, H.; Unger, L.-S.; Ivers-Tiffée, E.; Gerthsen, D. Influence of B-site doping with Ti and Nb on microstructure and phase constitution of (Ba0.5Sr0.5)(Co0.8Fe0.2)O3−δ. J. Mater. Sci. 2019, 55, 947–966. [Google Scholar] [CrossRef]
- Zeng, Q. , et al., A Zn-Doped Ba0.5Sr0.5Co0.8Fe0.2O3-δ Perovskite Cathode with Enhanced ORR Catalytic Activity for SOFCs. Catalysts, 2020. 10(2).
- Mushtaq, N. , et al., Perovskite SrFe1-xTixO3-δ (x < = 0.1) cathode for low temperature solid oxide fuel cell. Ceramics International, 2018. 44(9): p. 10266-10272.
- Navarrete, L. , et al., Tailoring La2-XAXNi1-YBYO4+δcathode performance by simultaneous A and B doping for IT-SOFC. International Journal of Hydrogen Energy, 2020. 45(31): p. 15589-15599.
- Zhou, Q. , et al., Preparation and electrochemical properties of an La-doped Pr2Ni0.85Cu0.1Al0.05O4+δcathode material for an IT-SOFC. Journal of Alloys and Compounds, 2020. 824.
- Huang, Y.; Ding, J.; Xia, Y.; Miao, L.; Li, K.; Zhang, Q.; Liu, W. Ba0.5Sr0.5Co0.8-xFe0.2NbxO3-δ (x≤ 0.1) as cathode materials for intermediate temperature solid oxide fuel cells with an electron-blocking interlayer. Ceram. Int. 2020, 46, 10215–10223. [Google Scholar] [CrossRef]
- Wan, Y. , et al., Thermal cycling durability improved by doping fluorine to PrBaCo2O5+δ as oxygen reduction reaction electrocatalyst in intermediate-temperature solid oxide fuel cells. Journal of Power Sources, 2018. 402: p. 363-372.
- Wang, W.; Zhang, X.; Zhang, D.; Zeng, Q.; Jiang, Y.; Lin, B. Highly promoted performance of triple-conducting cathode for YSZ-based SOFC via fluorine anion doping. Ceram. Int. 2020, 46, 23964–23971. [Google Scholar] [CrossRef]
- Wang, J.; Saccoccio, M.; Chen, D.; Gao, Y.; Chen, C.; Ciucci, F. The effect of A-site and B-site substitution on BaFeO3−δ: An investigation as a cathode material for intermediate-temperature solid oxide fuel cells. J. Power Sources 2015, 297, 511–518. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, L.; Guan, K.; Zhang, X.; Meng, J.; Wang, H.; Liu, X.; Meng, J. Effective promotion of oxygen reduction activity by rare earth doping in simple perovskite cathodes for intermediate-temperature solid oxide fuel cells. J. Power Sources 2020, 446, 227360. [Google Scholar] [CrossRef]
- Wang, S.; Yang, H.; Zheng, Y.; Ge, L.; Chen, H.; Guo, L. Effect of electrolyte composite on the performance of SmBa0.5Sr0.25Ca0.25CoFeO5+δ cathode for IT-SOFCs. Ionics 2019, 26, 281–291. [Google Scholar] [CrossRef]
- Gao, J.; Li, Q.; Guo, M.; Sun, L.; Huo, L.; Zhao, H. Improved electrochemical activity of Bi0.5Sr0.5FeO3-–Ce0.9Gd0.1O1.95composite cathode electrocatalyst for solid oxide fuel cells. Ceram. Int. 2020, 47, 748–754. [Google Scholar] [CrossRef]
- Eguchi, K.; Setoguchi, T.; Inoue, T.; Arai, H. Electrical properties of ceria-based oxides and their application to solid oxide fuel cells. Solid State Ionics 1992, 52, 165–172. [Google Scholar] [CrossRef]
- Ishihara, T.; Xie, J.; Shin, T.H.; Ju, Y.-W.; Ida, S.; Kilner, J.A. Bi doped Pr6O11 as fluorite oxide cathode for all-fluorite solid oxide fuel cells. J. Power Sources 2015, 275, 167–174. [Google Scholar] [CrossRef]
- Eksioglu, A.; Arslan, L.C.; Sezen, M.; Ow-Yang, C.W.; Buyukaksoy, A. Formation of Nanocomposite Solid Oxide Fuel Cell Cathodes by Preferential Clustering of Cations from a Single Polymeric Precursor. ACS Appl. Mater. Interfaces 2019, 11, 47904–47916. [Google Scholar] [CrossRef]
- Antipinskaya, E.A. , et al., Electrochemical performance and superior CO2 endurance of PrBaCo2O6–δ–PrBaCoTaO6 composite cathode for IT-SOFCs. Electrochimica Acta, 2021. 365: p. 137372.
- Ding, X.F. , et al., Enhanced oxygen reduction activity on surface-decorated perovskite La0.6Ni0.4FeO3 cathode for solid oxide fuel cells. ELECTROCHIMICA ACTA, 2015. 163: p. 204-212.
- Shen, J. , et al., Impregnated LaCo0.3Fe0.67Pd0.03O3-delta as a promising electrocatalyst for “symmetrical” intermediate-temperature solid oxide fuel cells. JOURNAL OF POWER SOURCES, 2016. 306: p. 92-99.
- Giuliano, A. , et al., Infiltration, Overpotential and Ageing Effects on Cathodes for Solid Oxide Fuel Cells: La0.6Sr0.4Co0.2Fe0.8O3-delta versus Ba0.5Sr0.5Co0.8Fe0.2O3-delta. JOURNAL OF THE ELECTROCHEMICAL SOCIETY, 2017. 164(10): p. F3114-F3122.
- Endler-Schuck, C.; Joos, J.; Niedrig, C.; Weber, A.; Ivers-Tiffée, E. The chemical oxygen surface exchange and bulk diffusion coefficient determined by impedance spectroscopy of porous La0.58Sr0.4Co0.2Fe0.8O3−δ (LSCF) cathodes. Solid State Ionics 2015, 269, 67–79. [Google Scholar] [CrossRef]
- Shah, M.; Voorhees, P.W.; Barnett, S.A. Time-dependent performance changes in LSCF-infiltrated SOFC cathodes: The role of nano-particle coarsening. Solid State Ionics 2011, 187, 64–67. [Google Scholar] [CrossRef]
- Song, X.; Lee, S.; Chen, Y.; Gerdes, K. Electrochemically influenced cation inter-diffusion and Co3O4 formation on La0.6Sr0.4CoO3 infiltrated into SOFC cathodes. Solid State Ionics 2015, 278, 91–97. [Google Scholar] [CrossRef]
- Ovtar, S.; Chen, M.; Samson, A.J.; Kiebach, R. In-situ formed Ce0.8Gd0.2O1.9 barrier layers on yttria stabilized zirconia backbones by infiltration - A promising path to high performing oxygen electrodes of solid oxide cells. Solid State Ionics 2017, 304, 51–59. [Google Scholar] [CrossRef]
- Hu, B.; Wang, Y.; Xia, C. Effects of Ceria Conductivity on the Oxygen Incorporation at the LSCF-SDC-Gas Three-Phase Boundary. J. Electrochem. Soc. 2015, 162, F33–F39. [Google Scholar] [CrossRef]
- Nie, L. , et al., La0.6Sr0.4Co0.2Fe0.8O3−δ cathodes infiltrated with samarium-doped cerium oxide for solid oxide fuel cells. Journal of Power Sources, 2010. 195(15): p. 4704-4708.
- Muneeb Irshad, K.S. , Rizwan Raza, et al., A Brief Description of High Temperature Solid Oxide Fuel Cell’s Operation, Materials, Design, Fabrication Technologies and Performance. Vol. 6. 2016, ; : Appl. Sci.
- Tomov, R.I. , et al., Performance optimization of LSCF/Gd:CeO2 composite cathodes via single-step inkjet printing infiltration. Journal of Applied Electrochemistry, 2017. 47(5): p. 641-651.
- Song, Y.-H. , et al., Facile surface modification of LSCF/GDC cathodes by epitaxial deposition of Sm0.5Sr0.5CoO3via ultrasonic spray infiltration. Journal of materials chemistry. A, Materials for energy and sustainability, 2020. 8(7): p. 3967-3977.
- Lee, S.; Lee, K.; Kang, S.; Kang, J.; Song, S.; Bae, J. Investigation of electrospun Ba0.5Sr0.5Co0.8Fe0.2O3−δ-Gd0.1Ce0.9O1.95 cathodes for enhanced interfacial adhesion. Int. J. Hydrogen Energy 2018, 43, 21535–21546. [Google Scholar] [CrossRef]
- Ai, N., K. Chen, and S.P. Jiang, A La0.8Sr0.2MnO3/La0.6Sr0.4Co0.2Fe0.8O3−δ core–shell structured cathode by a rapid sintering process for solid oxide fuel cells. International Journal of Hydrogen Energy, 2017. 42(10): p. 7246-7251.
- Kamlungsua, K.; Lee, T.-H.; Lee, S.; Su, P.-C.; Yoon, Y.-J. Inkjet-printed Ag@SDC core-shell nanoparticles as a high-performance cathode for low-temperature solid oxide fuel cells. Int. J. Hydrogen Energy 2021, 46, 30853–30860. [Google Scholar] [CrossRef]
- Yang, T.; Ramasamy, D.; Queirós, R.P.; Loureiro, F.J.A.; de Almeida, C.M.R.; Julião, P.S. The annealing influence on the microstructure and performance of Au@Ni core-shell bimetal as the cathode of low-temperature solid oxide fuel cells. Int. J. Hydrogen Energy 2015, 40, 4980–4988. [Google Scholar] [CrossRef]
- Yang, J.; Wang, J.; Fu, L.; Wu, K.; Liu, Z.; Wu, K.; Zhou, J. Electrospun Core–Shell Fibers for High-Efficient Composite Cathode-Based Solid Oxide Fuel Cells. Energy Fuels 2021, 35, 1768–1778. [Google Scholar] [CrossRef]
- Ding, X.; Zhao, X.; Yan, Y.; Li, M. Enhancing oxygen reduction activity of perovskite cathode decorated with core@shell nano catalysts. Int. J. Hydrogen Energy 2019, 44, 22122–22128. [Google Scholar] [CrossRef]
- Qiu, P. , et al., Promoted CO2-poisoning resistance of La0.8Sr0.2MnO3−δ-coated Ba0.5Sr0.5Co0.8Fe0.2O3−δ cathode for intermediate temperature solid oxide fuel cells. Journal of Power Sources, 2016. 327: p. 408-413.
- Lee, D.; Jung, I.; Lee, S.O.; Hyun, S.H.; Jang, J.H.; Moon, J. Durable high-performance Sm0.5Sr0.5CoO3–Sm0.2Ce0.8O1.9 core-shell type composite cathodes for low temperature solid oxide fuel cells. Int. J. Hydrogen Energy 2011, 36, 6875–6881. [Google Scholar] [CrossRef]
- Wang, L.; Wang, P.; Geng, C.; Cao, H.; Xu, C.; Cheng, J.; Hong, T. A novel core-shell LSCF perovskite structured electrocatalyst with local hetero-interface for solid oxide fuel cells. Int. J. Hydrogen Energy 2020, 45, 11824–11833. [Google Scholar] [CrossRef]
- Li, J. , et al., Microstructure optimization for high-performance PrBa0.5Sr0.5Co1.5Fe0.5O5+δ-La2NiO4+δ core-shell cathode of solid oxide fuel cells. Journal of Power Sources, 2018. 379: p. 206-211.
- Qiu, P. , et al., LaCoO3-δ coated Ba0.5Sr0.5Co0.8Fe0.2O3-δ cathode for intermediate temperature solid oxide fuel cells. Electrochimica Acta, 2019. 319: p. 981-989.
- Li, D.; Zhang, X.; Liang, C.; Jin, Y.; Fu, M.; Yuan, J.; Xiong, Y. Study on durability of novel core-shell-structured La0.8Sr0.2Co0.2Fe0.8O3-δ@Gd0.2Ce0.8O1.9 composite materials for solid oxide fuel cell cathodes. Int. J. Hydrogen Energy 2021, 46, 28221–28231. [Google Scholar] [CrossRef]
- Li, F. , et al., LaSrCoO4±@La0.5Sr0.5CoO3- core-shell hybrid as the cathode materials for solid oxide fuel cells. Journal of Alloys and Compounds, 2020. 819.
- Kim, C. , et al., Ba0.5Sr0.5Co0.8Fe0.2O3-δ / Gd0.1Ce0.9O2-δ Core/shell Nanofiber Via One-Step Electrospinning for Cathode of LT-SOFCs, in ECS Trans. 2017, The Electrochemical Society, Inc. p. 637-641.
- Chen, Y.; Bu, Y.; Zhao, B.; Zhang, Y. X.; Ding, D.; Hu, R.; Wei, T.; Rainwater, B.; Ding, Y.; Chen, F.; et al. A durable, high-performance hollow-nanofiber cathode for intermediate-temperature fuel cells. Nano Energy 2016, 26, 90–99. [Google Scholar] [CrossRef]
- Zhang, W. , et al., La0.6Sr0.4Co0.2Fe0.8O3−δ/CeO2 Heterostructured Composite Nanofibers as a Highly Active and Robust Cathode Catalyst for Solid Oxide Fuel Cells. ACS Applied Materials & Interfaces, 2019. 11.
- Horita, T.; Nishi, M.; Shimonosono, T.; Kishimoto, H.; Yamaji, K.; Brito, M.E.; Yokokawa, H. Visualization of oxide ionic diffusion at SOFC cathode/electrolyte interfaces by isotope labeling techniques. Solid State Ionics 2014, 262, 398–402. [Google Scholar] [CrossRef]
- Li, Z.-P.; Mori, T.; Auchterlonie, G.J.; Zou, J.; Drennan, J. Two types of diffusions at the cathode/electrolyte interface in IT-SOFCs. J. Solid State Chem. 2011, 184, 2458–2461. [Google Scholar] [CrossRef]
- Jang, I.; Kim, S.; Kim, C.; Lee, H.; Yoon, H.; Song, T.; Paik, U. Interface engineering of yttrium stabilized zirconia/gadolinium doped ceria bi-layer electrolyte solid oxide fuel cell for boosting electrochemical performance. J. Power Sources 2019, 435, 226776. [Google Scholar] [CrossRef]
- Park, S.-Y.; Ahn, J.H.; Jeong, C.-W.; Na, C.W.; Song, R.-H.; Lee, J.-H. Ni–YSZ-supported tubular solid oxide fuel cells with GDC interlayer between YSZ electrolyte and LSCF cathode. Int. J. Hydrogen Energy 2014, 39, 12894–12903. [Google Scholar] [CrossRef]
- Zhao, Z.; Liu, L.; Zhang, X.; Wu, W.; Tu, B.; Ou, D.; Cheng, M. A comparison on effects of CO2 on La0.8Sr0.2MnO3+ and La0.6Sr0.4CoO3− cathodes. J. Power Sources 2013, 222, 542–553. [Google Scholar] [CrossRef]
- Almar, L. , et al., Improved Phase Stability and CO2 Poisoning Robustness of Y-Doped Ba0.5Sr0.5Co0.8Fe0.2O3−δ SOFC Cathodes at Intermediate Temperatures. ACS Applied Energy Materials, 2018. 1(3): p. 1316-1327.
- Hu, X.; Xie, Y.; Wan, Y.; Yang, Y.; Wu, X.; Xia, C. Antimony-doped strontium cobalt oxide as promising cathode for low-temperature solid oxide fuel cell with excellent carbon dioxide tolerance. Appl. Catal. B: Environ. 2021, 286, 119901. [Google Scholar] [CrossRef]
- Huang, Y.L.; Pellegrinelli, C.; Wachsman, E.D. Fundamental Impact of Humidity on SOFC Cathode ORR. J. Electrochem. Soc. 2015, 163, F171–F182. [Google Scholar] [CrossRef]
- Zhao, Z. , et al., High- and low- temperature behaviors of La0.6Sr0.4Co0.2Fe0.8O3−δ cathode operating under CO2/H2O-containing atmosphere. International Journal of Hydrogen Energy, 2013. 38(35): p. 15361-15370.
- Nielsen, J.; Hagen, A.; Liu, Y. Effect of cathode gas humidification on performance and durability of Solid Oxide Fuel Cells. Solid State Ionics 2010, 181, 517–524. [Google Scholar] [CrossRef]
- Liu, R.; Kim, S.; Taniguchi, S.; Oshima, T.; Shiratori, Y.; Ito, K.; Sasaki, K. Influence of water vapor on long-term performance and accelerated degradation of solid oxide fuel cell cathodes. J. Power Sources 2011, 196, 7090–7096. [Google Scholar] [CrossRef]
- Chen, Y.; Fan, Y.; Lee, S.; Hackett, G.; Abernathy, H.; Gerdes, K.; Song, X. Interface and grain boundary degradation in LSM-YSZ composite Solid Oxide Fuel Cell cathodes operated in humidified air. J. Power Sources 2019, 438. [Google Scholar] [CrossRef]
- Hardy, J.S.; Coyle, C.A.; Bonnett, J.F.; Templeton, J.W.; Canfield, N.L.; Edwards, D.J.; Mahserejian, S.M.; Ge, L.; Ingram, B.J.; Stevenson, J.W. Evaluation of cation migration in lanthanum strontium cobalt ferrite solid oxide fuel cell cathodes via in-operando X-ray diffraction. J. Mater. Chem. A 2018, 6, 1787–1801. [Google Scholar] [CrossRef]
- Wang, R.; Sun, Z.; Lu, Y.; Gopalan, S.; Basu, S.N.; Pal, U.B. Comparison of chromium poisoning between lanthanum strontium manganite and lanthanum strontium ferrite composite cathodes in solid oxide fuel cells. J. Power Sources 2020, 476, 228743. [Google Scholar] [CrossRef]
- Egger, A. , et al., Effect of Microstructure on the Degradation of La0.6Sr0.4CoO3-δ electrodes in dry and humid Atmosphere. Fuel cells, 2019.
- Wang, J. , et al., Effect of humidity on La0.4Sr0.6Co0.2Fe0.7Nb0.1O3−δ cathode of solid oxide fuel cells. International Journal of Hydrogen Energy, 2018. 44.
- Hagen, A., K. Neufeld, and Y.-l.J.J.o.T.E.S. Liu, Effect of Humidity in Air on Performance and Long-Term Durability of SOFCs. 2010. 157.


| Perovskites/Double Perovskites | Doping | Opt. value of dopant (%) |
Doping Site | Cell- configuration Electrolyte/ Anode (Cell type) |
Test-ing temp. (℃) | Rp (Ω. cm2) |
Power density mW.cm−2 (of cathode material) |
TEC (10−6 K−1) | Cell Test-ing Time (hr) |
Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| Nd1-xLnxBaCo2O6 | Ln = La, Sm & Gd | La = 0.1 | A - site | YSZ/ (CGO Buffer layer)/NiO-Zr0.85Y0.15O1.95(Single cell) | 700 | 0.083 | 1.045 W.cm−2 | - | 100 hrs | [20] |
| Ba0.5Sr0.5Co0.2Fe0.7Mo0.1O 3-d | Mo | Mo = 0.1 | B - site | YSZ/ Ba0.5Sr0.5Co0.2Fe0.7Mo0.1O3-d (BSCFM) (Symmetrical cell) |
800 | 0.035 | 418 (At 700 ℃ in H2 with 3% water) |
- | 115 hrs | [22] |
| PrBaCo2-xMoxO5+ƿ | Mo (x = 0-0.07) | Mo = 0.03 | B- site | SDC/NiO-SDC (Single Cell) |
700 | 0.067 | 339 | 18.1 | 42 hrs | [23] |
| PrBaCo2O5+Fx (O-site doping) |
x = 0- 0.2 % | F- = 0.1 | O - site | GDC/Ni-GDC (Single cell) |
650 | 0.062 | 0.679 W.cm−2 |
17.46 | 100 hrs | [36] |
| BaFeO3-δ | La+3, Sm+3, Gd+3 (A-Site) Zr+4 and Ce+4 (B-Site) |
La+3 and Zr+4 = 0.005 | A - site and B - site | SDC/Ba0.9La0.05 FeO3-δ (BFO) (A-site Symmetrical cell) SDC/BaFe0.95Zr0.05O3-δ (BFO) (B-site Symmetrical cell) |
700 | 0.029 (La+3 at A-Site) 0.020 (Zr+4 B-Site) |
- | - | - | [38] |
| La0.6-xMxSr0.4Co0.2Fe0.8O3 | M = Ca, Ba & Bi | Bi = 0.2 % | A - site | YSZ/La0.4Bi0.2Sr0.4Co0.2Fe0.8O3(LBSCF (Symmetrical cell) |
750 | 0.132 | 1.002 W.cm −2 | 18.1 | 100 hrs at 600 °C | [15] |
| LnxBa1-xCo0.5Fe0.3O3-δ (LnxBCF) | Ln = La, Pr, Nd | Pr = 0.1% | A -site | SDC/Pr0.1Ba0.9Co0.5Fe0.3O3-δ (Symmetrical cell) |
700 | 0.026 (ASR) |
1236 | 16.2 | 150 hrs | [39] |
| La2-xAxNi1-yByO4+δ (LNO) | x = Pr y = Co |
Pr =0.5% Co = 0.2% |
A-site and B-site | YSZ/La1.5Pr0.5Ni0.8Co0.2O4+δ (LPNCO) (Symmetrical cell) |
600 | 1.95 | 400 | - | - | [33] |
| Pr2-xLaxNi0.85Cu0.1Al0.05O4+δ | x = La | La = 1 % | A-site | LSGM/PrLaNi0.85Cu0.1Al0.05O4+δ (Symmetrical cell) |
700 | 0.037 (ASR at 800 °C) |
341 (at 700 °C) | 14.6 | 72 hrs | [34] |
| Ba0.5Sr0.5(Co0.8Fe0.2)1-xZnxO3-δ (BSCFZ) | x = Zn+2 | Zn+2 = 0.04% | B - site | YSZ/Ba0.5Sr0.5 (Co0.8Fe0.2)0.96Zn0.04O3-δ (BSCFZ) (Symmetrical cell) |
600 | 0.23 | 0.58 W.cm−2 | - | 140 hrs | [31] |
| Ba0.5Sr0.5Co0.8-xFe0.2NbxO3-δ (BSCFNb) | y = Nb | Nb = 0.05 % | B - site | SDC/NiO-BaZr0.1 Ce0.7Y0.2O3-δ (Single cell) |
700 | 1.14 | 882 | 18.74 | 70 hrs | [35] |
| La1-xCexSr0.4Co0.2Fe0.8O3 (LCSCF) | x = Ce+3 | Ce+3 = 0.6 % | A -site | GDC/Pt (Single cell) |
750 | 0.09 | - | - | - | [25] |
| Core shell Cathode | Electrolyte | Temperature (℃) | Rp (Ω.cm2) | Power density (W.cm−2) | Reference |
|---|---|---|---|---|---|
| Ag@SDC | SDC | 450 | 0.7 | 0.0562 | [60] |
| Au@Ni | GDC | 500 | 0.25 | 0.464 | [61] |
| YCo0.5Fe0.5O3(YCF)@Gd0.1Ce0.9O1.95(GDC) | GDC | 550 | 0.66 | 0.4265 | [62] |
| Ag @ Pr0.2Ce0.8O2-δ -LSCF |
SDC | 550 | 0.65 | - | [63] |
| La0.8Sr0.2MnO3-δ (LSM)@Ba0.5Sr0.5Co0.8Fe0.2O3-δ(BSCF) | GDC | 600 | 1.141 | - | [64] |
| Sm0.5Sr0.5CoO3 (SSC)@Sm0.2Ce0.8O1.9 | SDC | 650 | 0.098 | 1.07 (600 ℃) | [65] |
| La0.6Sr0.4Co0.2Fe0.8O3-δ(LSCF)@La0.6Sr1.4Co0.2Fe0.8O4-δ (LSCF) | SDC | 650 | 0.17 | 0.57 | [66] |
| La0.8Sr0.2MnO3 (LSM)@La0.6Sr0.4Co0.2Fe0.8O3-δ (LSCF) |
YSZ | 700 | 2.1 | - | [59] |
| PrBa0.5Sr0.5Co1.5Fe0.5O5+δ (PBSCF)-La2NiO4+δ (LN) |
YSZ | 700 | 0.51 | 0.71 | [67] |
| LaCoO3-d (LC)@Ba0.5Sr0.5Co0.8Fe0.2O3-δ (BSCF) |
GDC | 750 | 0.020 | 0.929 | [68] |
| La0.8Sr0.2Co0.2Fe0.8O3-δ (LSCF)@Gd0.2Ce0.8O1.9 (GDC) |
GDC | 750 | 0.16 | - | [69] |
| LaSrCoO4±δ(LSC) @La0.5Sr0.5CoO 3-δ (LSC) |
LSGM | 800 | 0.03 | 0.87 | [70] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




