Submitted:
26 April 2024
Posted:
28 April 2024
You are already at the latest version
Abstract
Keywords:
General Morphological Characteristics of the Skin
Functions of the Skin
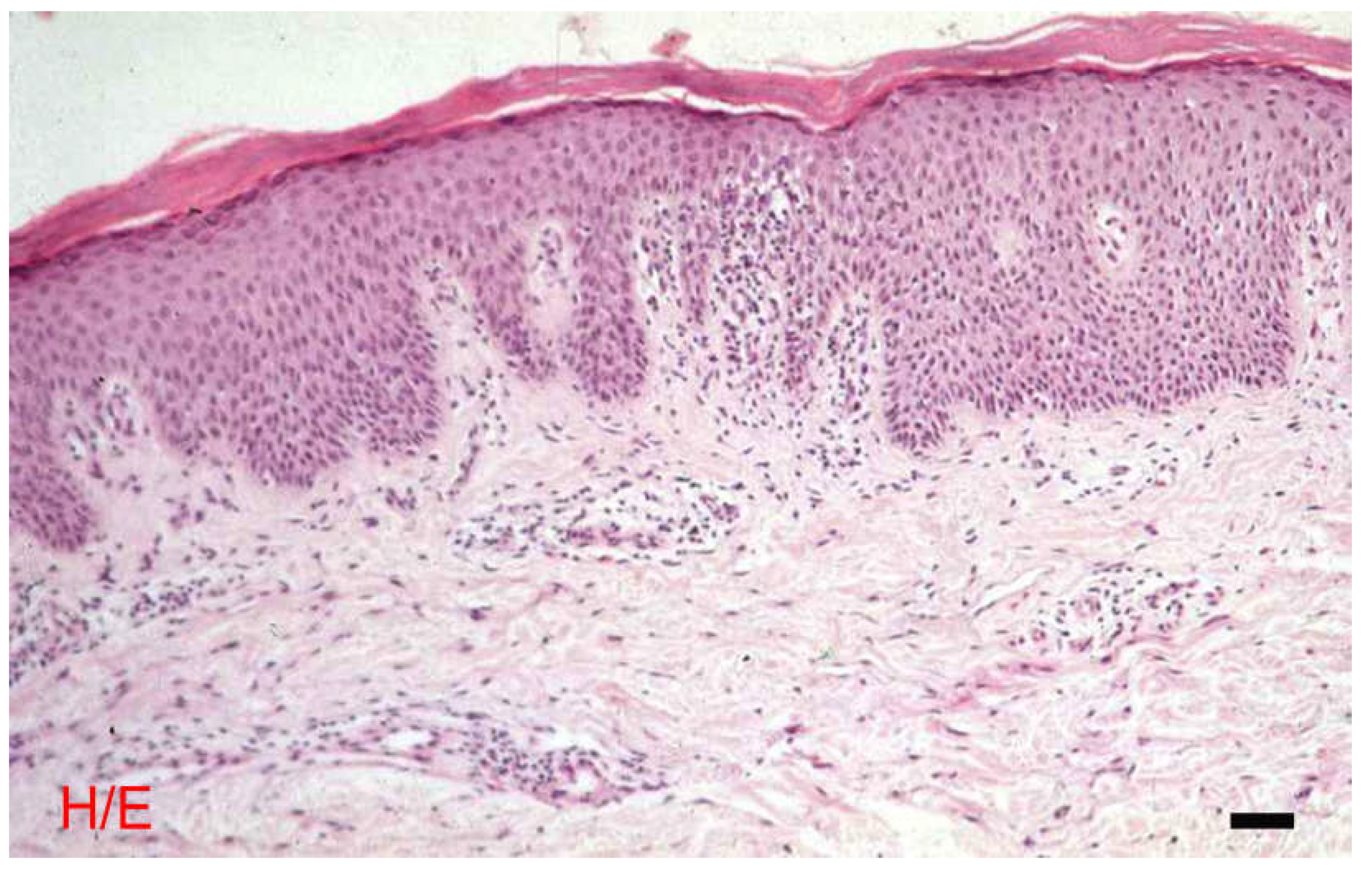
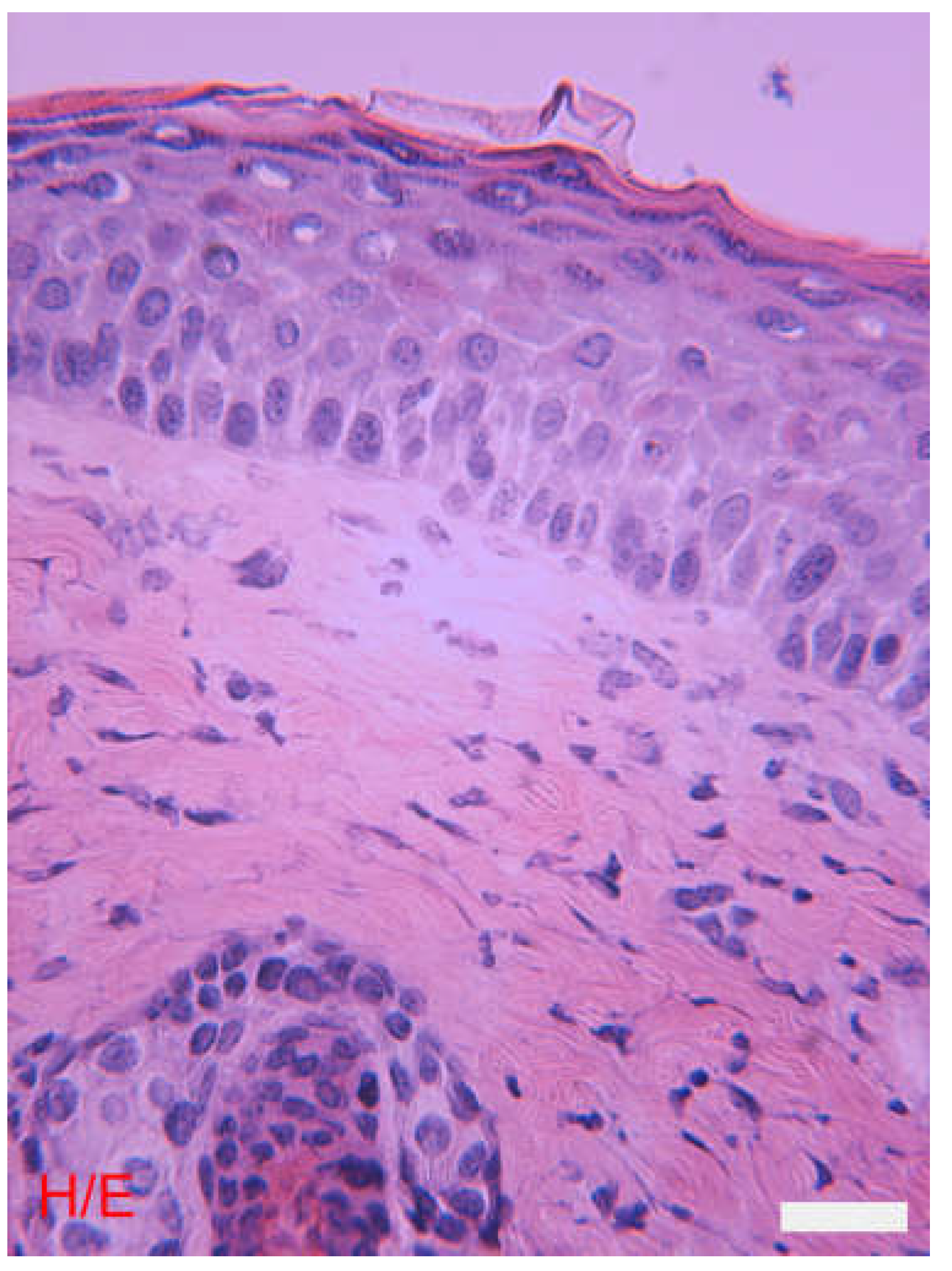
The Epidermis
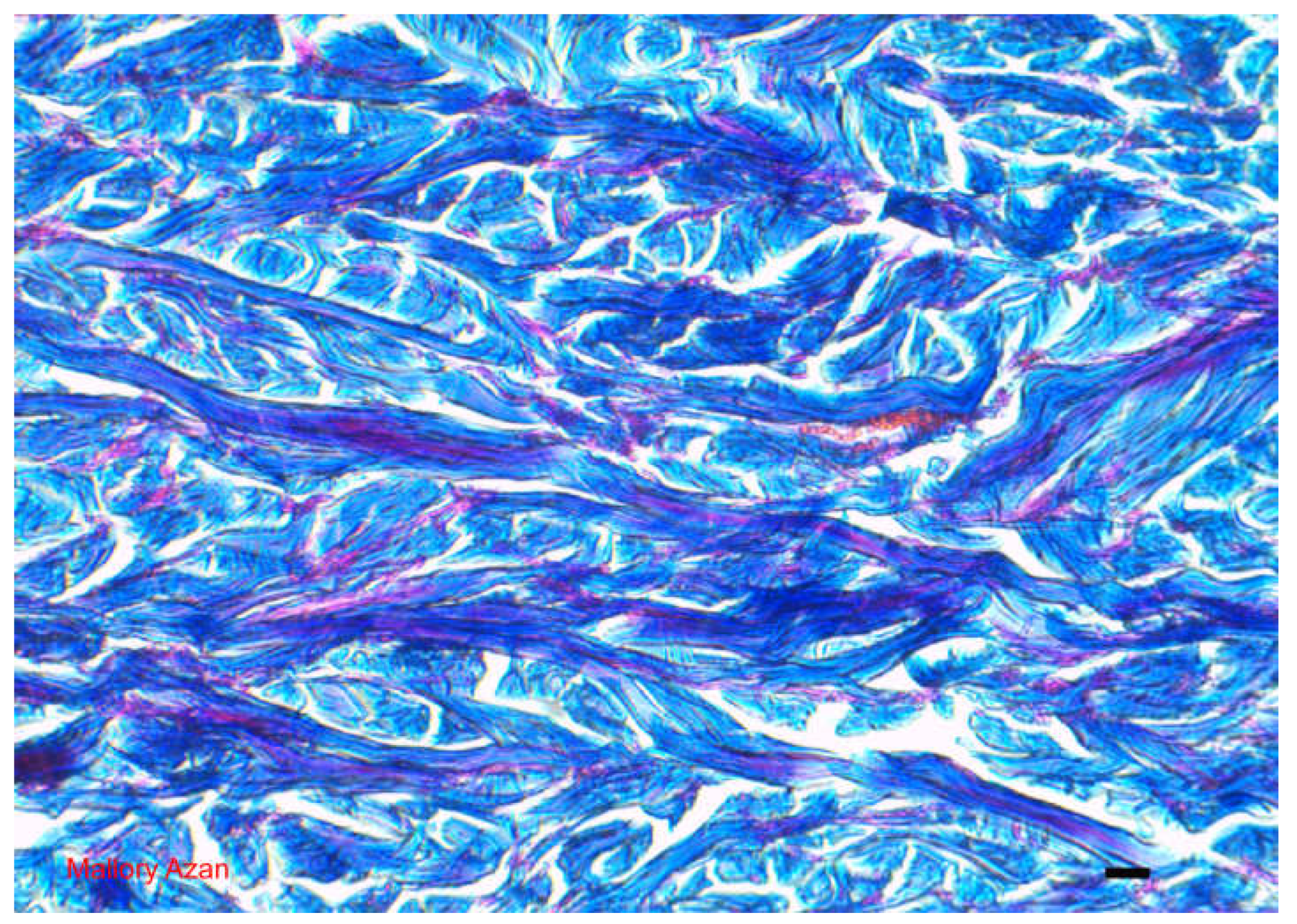
The Dermis
Subcutaneous
Vascularization and Innervation of the Skin
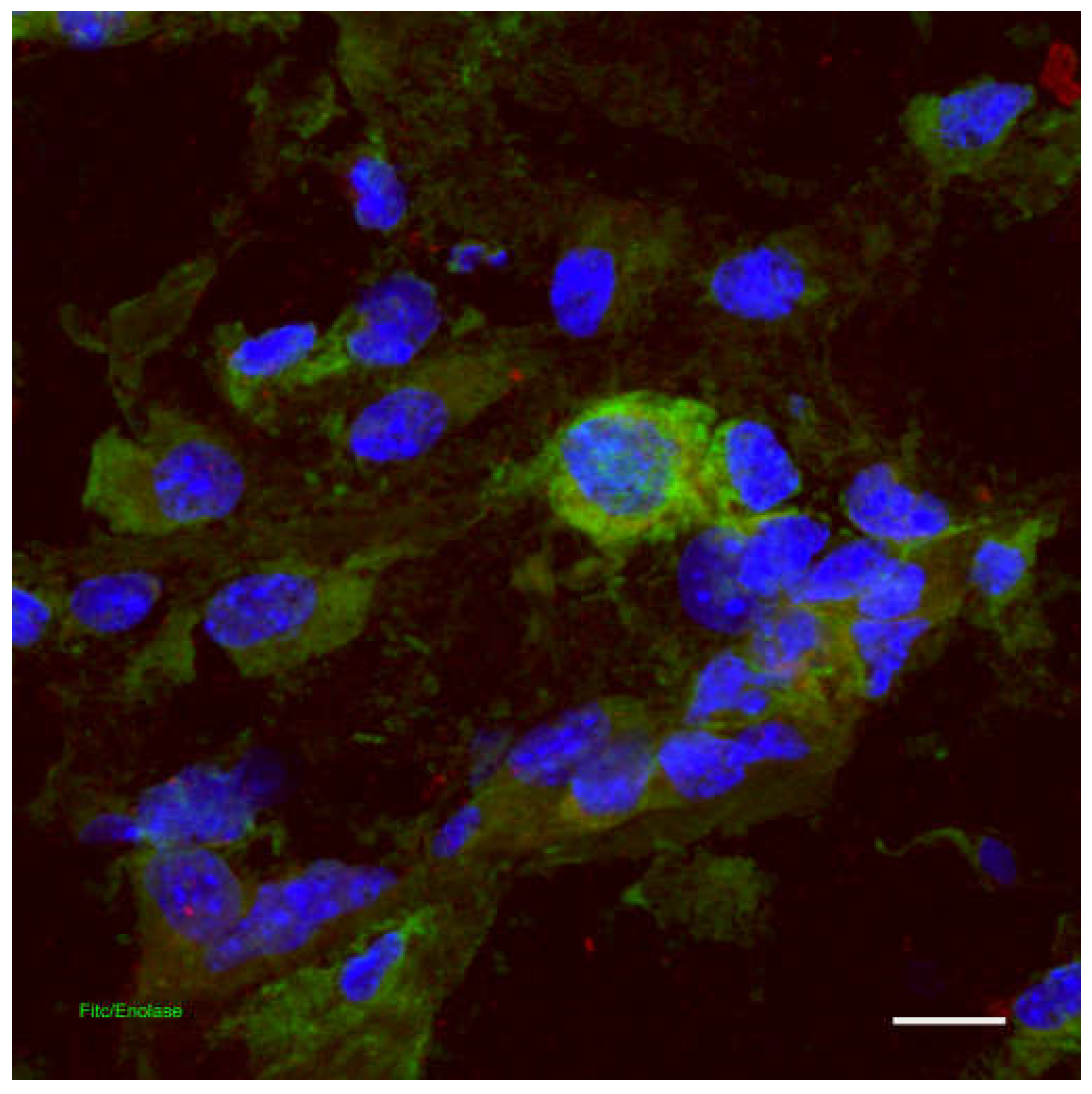
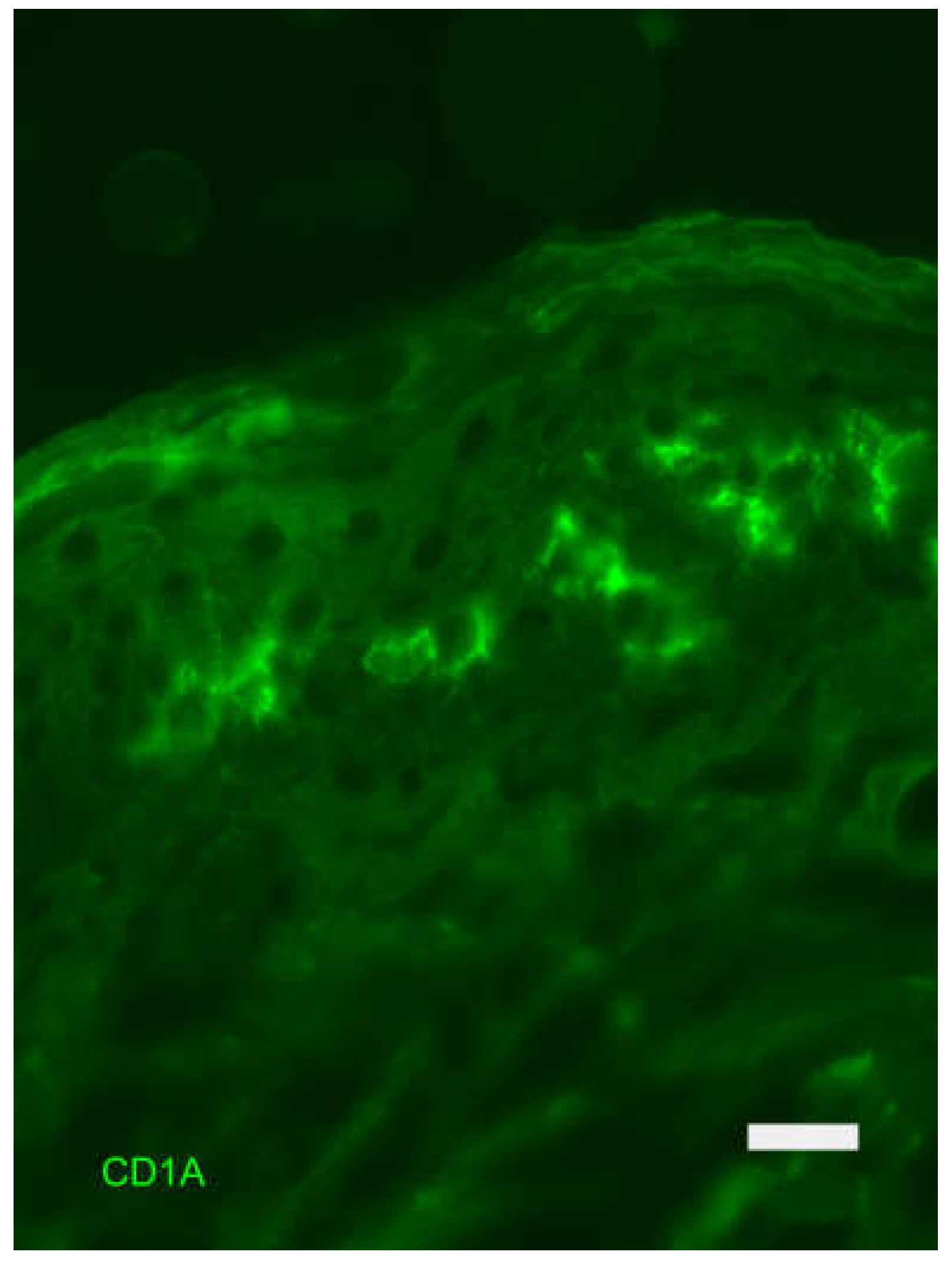
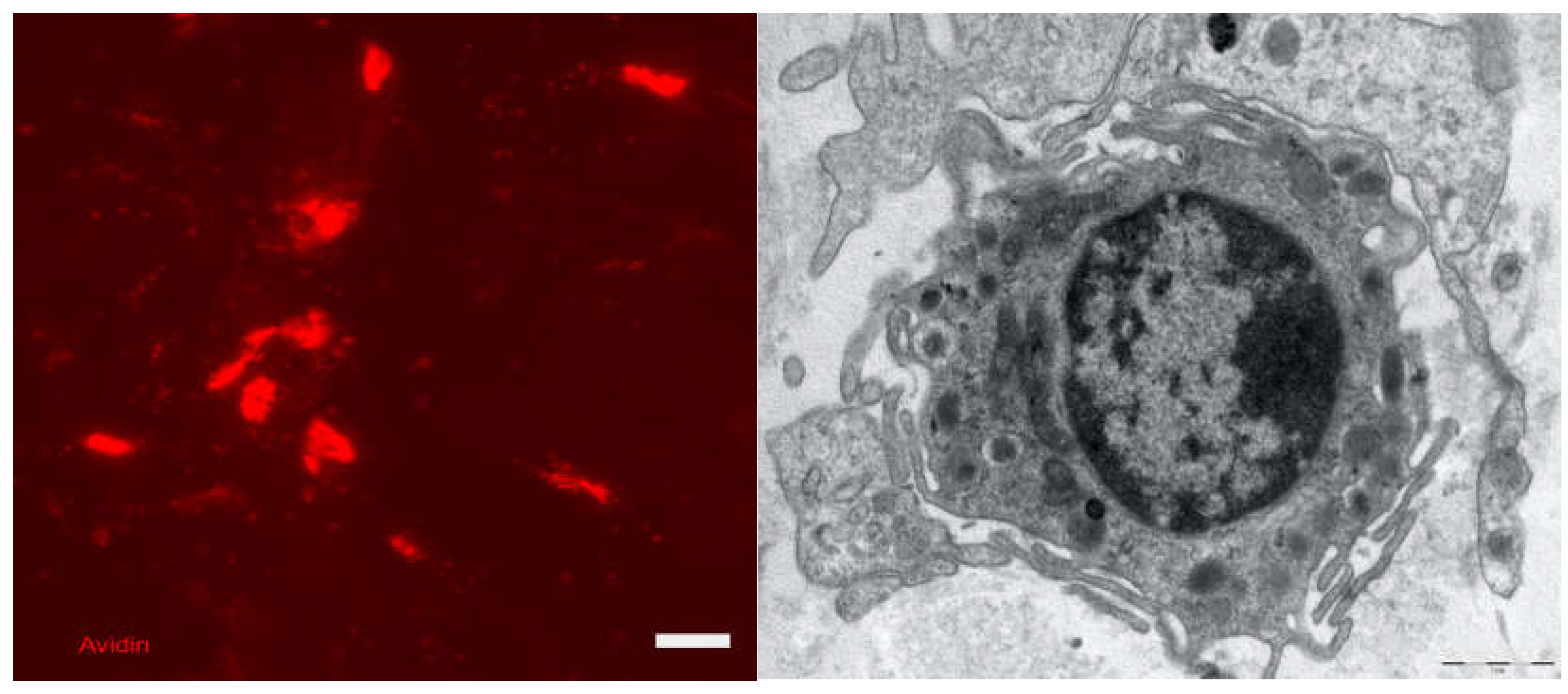
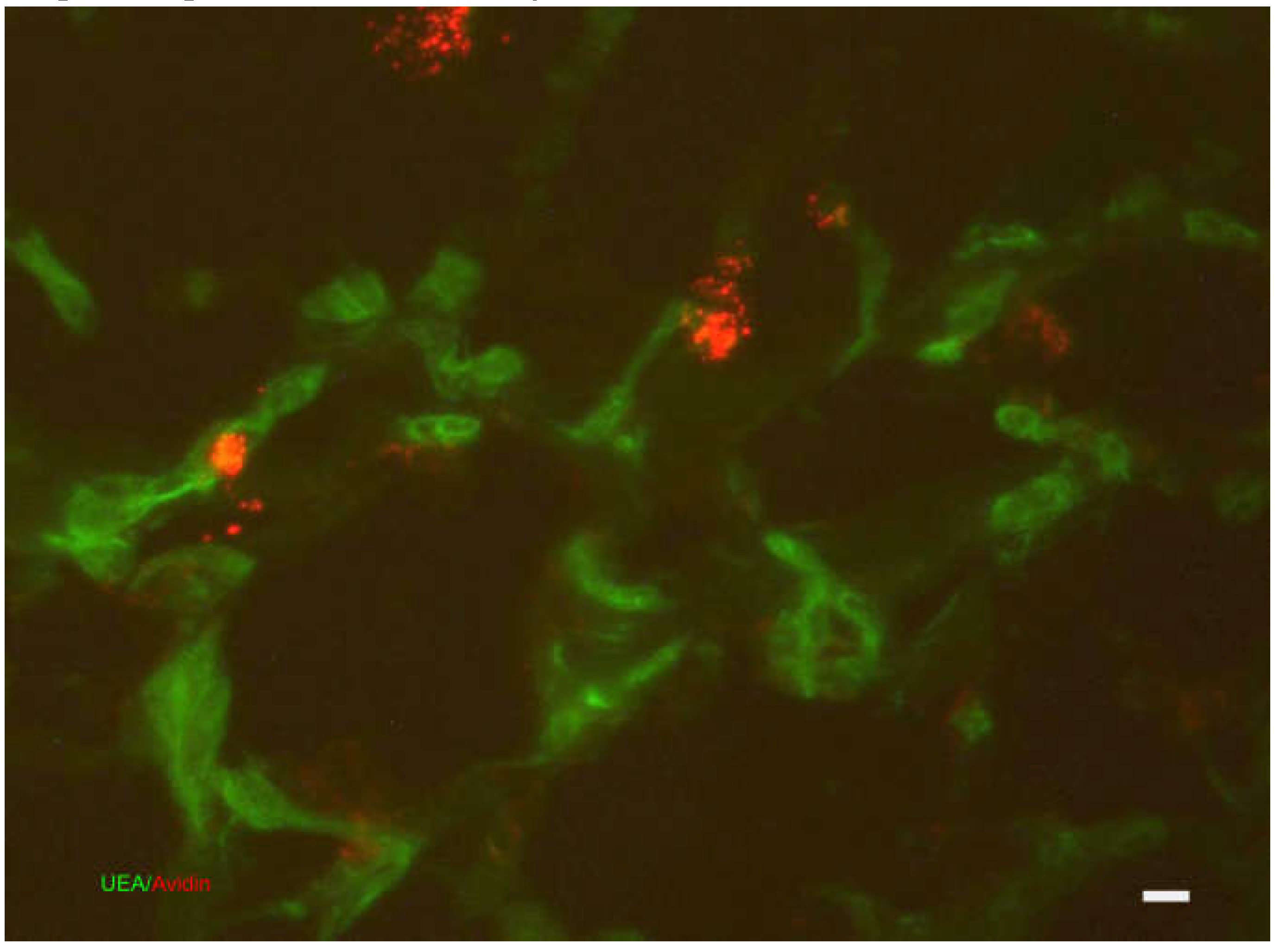
Skin Immune System
Skin pH
Skin Development
Disturbances in the Skin Homeostasis: The History of Wound Healing
Historical Aspect of a Wound
Vertebrates and Wound Healing
Wound Healing Modalities
Wounds Can Heal in Three Different Ways
Gross stages of Wound Healing
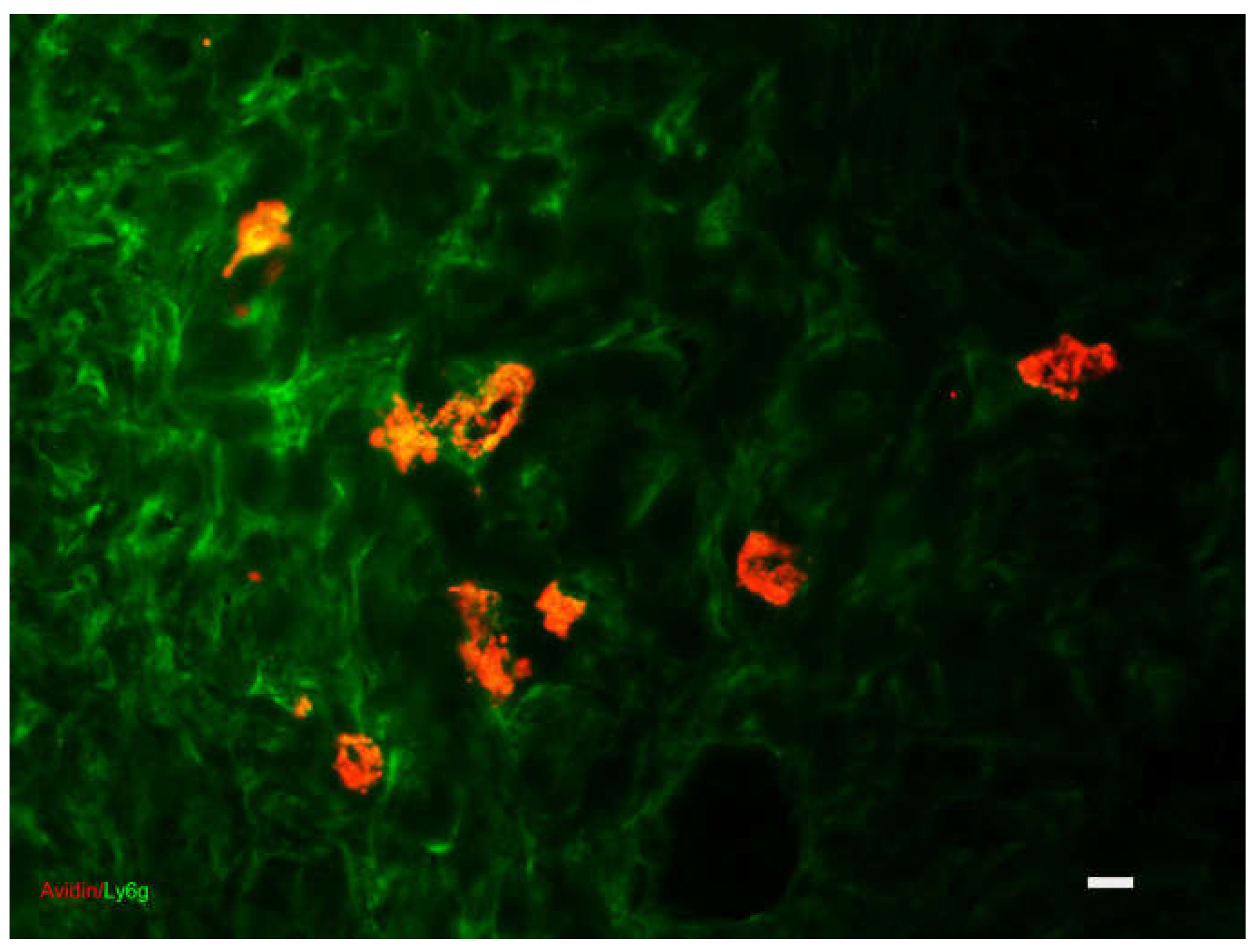
Acute Inflammatory Reaction
Summary of Principal Events in Wound Healing
The Scar
| Time | Processes | Associated visible changes and events | Cells | Cell adhesion molecules | ECM components | Key mediators/regulators |
|---|---|---|---|---|---|---|
| 0-15 minutes | Hemostasis | Whitening, Formation of the fibrin clot |
Endothelial cells, platelets | Fibrin, fibronectin | Epinephrine, prostaglandins, thromboxanes, thrombin | |
| 15 minutes-6 days | Inflammation | Redness, swelling, heat of the inflamed part, pain, functional alteration, Vasodilation, diapedesis, inflammatory response, phagocytosis | Endothelial cells, MC, macrophages, T lymphocytes | Temporary matrix formed by fibrin, complement proteins, PDGF, IL-8, IL-1 alpha, IL-1 beta, IL-6 and TNF-alpha | Sympathetic nervous system, histamine, kinins, leukotrienes, thrombin, | |
| 24-48 hours | Proliferation | Detachment of the eschar, epithelialization, | Keratinocytes | Beta1-integrins | Collagen, fibronectin, vitronectin and tenescinE (temporary ECM) | MMP/TIMP, FGF-2, FGF-7, FGF-10, GM-CSF, NO, TGF-beta, NGF, HGF, HB-EGF, IL-6, Leptina, sAPP |
| 4-7 days |
Proliferation | Granulation tissue, Angiogenesis | Fibroblasts, Endothelial cells | Alphabeta3 integrins |
Collagen, GAGs and Proteoglycans (Provisional ECM) | MMP/TIMP, VEGF, FGF, angiopoietin, TGF-beta, PDGF. |
| 4-14 days | Maturation | Approaching the edges of the wound (wound contaction) | myofibroblasts | Collagen | PDGF, TGF-beta, NGF |
|
| 3-21 days | Maturation | Scar Deposition of the collagen network | Fibroblasts | Integrins | EGF, IGF-1, FGF-2 | |
| From 2 days to several weeks | Maturation | Scar Deposition of the collagen network | PDGF, TGF-beta, NGF |
Differences with Fetal Wound Healing
Alterations in the Normal Wound Healing Process
Chronic Skin Lesions
Keloids
Hypertrophic Scars
Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Joey, E.; Cheong, L.; McGrath, J.A. Structure and function of skin, hair and nails. Medicine 2021, 49, 337–342. [Google Scholar] [CrossRef]
- Fore, J. A review of skin and the effects of aging on skin structure and function. Ostomy/wound management 2006, 52, 24–37. [Google Scholar] [PubMed]
- Farage, M.A.; Miller, K.W.; Elsner, P.; Maibach, H.I. Structural characteristics of the aging skin: a review. Cutan Ocular Toxicol 2007, 26, 343–357. [Google Scholar] [CrossRef] [PubMed]
- Sorg, H.; Sorg, C.G.G. Skin wound healing: of players, patterns, and processes. Eur Surg Res 2023, 64, 141–157. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.; Geyer, S.; Weninger, W.; Guimberteau, J.C.; Wong, J.K. The dynamic anatomy and patterning of skin. Exper Dermat 2016, 25, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Kolarsick Paul, A.J.; Kolarsick, M.A.; Goodwin, C. Anatomy and physiology of the skin. JDNA 2011, 3, 203–213. [Google Scholar] [CrossRef]
- Furuse, M.; Hata, M.; Furuse, K.; Yoshida, Y.; Haratake, A.; Sugitani, Y.; Noda, T.; Kubo, A.; Tsukita, S. Claudin-based tight junctions are crucial for the mammalian epidermal barrier: a lesson from claudin-1-deficient mice. J Cell Biol 2002, 156, 1099–1111. [Google Scholar] [CrossRef] [PubMed]
- Madison, K.C. Barrier function of the skin: "la raison d'être" of the epidermis. J Invest Dermatol 2003, 121, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Johansson, J.A.; Headon, D.J. Regionalisation of the skin. Stem Cell Dev Biology 2014, 26, 3–10. [Google Scholar] [CrossRef]
- Mc Grath, J.A.; Llitto, J. Anatomy and organization of human skin. In: Rook’s textbook of dermatology, 8th ed. Burns, T., Breathnach, S., Cox, N., Griffiths, C. (Eds).; Hoboken, NY: Wiley Blachwell; Chapter 3, 2010.
- Fernandez-Flores, A. Regional variations in the histology of the skin. Am J Dermatopahol 2015, 37, 737–754. [Google Scholar] [CrossRef]
- Khavkin, J.; Ellis, D.A. Aging skin: histology, physiology, and pathology. Facial plast surg clin North Am 2015, 19, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Laverdet, B.; Danigo, A.; Girard, D.; Magy, L.; Demiot, C.; Desmoulière, A. Skin innervation: important roles during normal and pathological cutaneous repair. Histol Histopathol 2015, 30, 875–892. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, M.; Baguneid, M.; Bayat, A. The role of neuromediators and innervation in cutaneous wound healing. Acta Derm Venereol 2016, 96, 587–594. [Google Scholar] [CrossRef]
- Nguyen, A.V.; Soulika, A.M. The dynamics of the skin's immune system. Int J Mol Sci 2019, 20, 1811. [Google Scholar] [CrossRef] [PubMed]
- Quaresma, J.A.S. Organization of the skin immune system and compartmentalized immune responses in infectious diseases. Clin Microbiol Rev 2019, 10, 1128. [Google Scholar] [CrossRef]
- Zhang, C.; Merana, G.R.; Harris-Tryon, T.; Scharschmidt, T.C. Skin immunity: dissecting the complex biology of our body's outer barrier. Mucosal Immunol 2022, 15, 551–561. [Google Scholar] [CrossRef]
- Zanna, M.Y.; Yasmin, A.R.; Omar, A.R.; Arshad, S.S.; Mariatulqabtiah, A.R.; Nur-Fazila, S.H.; Mahiza, M.I.N. Review of dendritic cells, their role in clinical immunology, and distribution in various animal species. Int J Mol Sci 2021, 22, 8044. [Google Scholar] [CrossRef]
- Bacci, S.; Nakamura, T.; Streilein, J.W. Failed antigen presentation after UVB radiation correlates with modifications of Langerhans cell cytoskeleton. J Invest Dermatol 1996, 107, 838–843. [Google Scholar] [CrossRef]
- Bacci, S.; Romagnoli, P.; Streilein, J.W. Reduction in number and morphologic alterations of Langerhans cells after UVB radiation in vivo are accompanied by an influx of monocytoid cells into the epidermis. J Invest Dermatol 1998, 111, 1134–1139. [Google Scholar] [CrossRef]
- Bacci, S.; Alard, P.; Streilein, J.W. Evidence that ultraviolet B radiation transiently inhibits emigration of Langerhans cells from exposed epidermis, thwarting contact hypersensitivity induction. Eur J Immunol 2001, 31, 3588–3594. [Google Scholar] [CrossRef]
- Focardi, M.; Bugelli, V.; Venturini, M.; Bianchi, I.; Defraia, B.; Pinchi, V.; Bacci, S. (2022). Increased expression of iNOS by Langerhans cells in hanging marks. Aust J Forensic Sci 2022, 54, 177–186. [Google Scholar] [CrossRef]
- Focardi, M.; Puliti, E.; Grifoni, R.; Palandri, M.; Bugelli, V.; Pinchi, V.; Norelli, G.A.; Bacci, S. Immunohistochemical localization of Langerhans cells as a tool for vitality in hanging mark wounds: a pilot study. Aust J Forensic Sc 2020, 52, 393–405. [Google Scholar] [CrossRef]
- Bacci, S.; Alard, P.; Dai, R.; Nakamura, T.; Streilein, J.W. High and low doses of haptens dictate whether dermal or epidermal antigen-presenting cells promote contact hypersensitivity. Eur J Immunol 1997, 27, 442–448. [Google Scholar] [CrossRef]
- Bacci, S.; Defraia, B.; Cinci, L.; Calosi, L.; Guasti, D.; Pieri, L.; Lotti, V.; Bonelli, A.; Romagnoli, P. Immunohistochemical analysis of dendritic cells in skin lesions: correlations with survival time. Forensic Sci Int 2014, 244, 179–185. [Google Scholar] [CrossRef]
- Bacci, S. Fine regulation during wound healing by mast cells, a physiological role not yet clarified. Int J Mol Sci, 2022, 23 1820. [Google Scholar] [CrossRef]
- Martel, J.L.; Miao, J.H.; Badri, T. Anatomy, Hair Follicle. [Updated 2022 Oct 10]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470321/.
- Lousada, M.B.; Lachnit, T.; Edelkamp, J.; Rouillé, T; Ajdic, D.; Uchida, Y.; Di Nardo, A.; Bosch, T.C.G.; Paus, R. Exploring the human hair follicle microbiome. Br J Dermatol 2021, 184, 802–815. [Google Scholar] [CrossRef]
- de Berker, D. Nail anatomy. Clin Dermatol 2013, 31, 509–515. [Google Scholar] [CrossRef]
- Okoro, O.E.; Camera, E.; Flori, E.; Ottaviani, M. Insulin and the sebaceous gland function. Front Physiol 2023, 14, 1252972. [Google Scholar] [CrossRef] [PubMed]
- Makrantonaki, E.; Ganceviciene, R.; Zouboulis, C. An update on the role of the sebaceous gland in the pathogenesis of acne. Dermatoendocrinol 2011, 3, 41–49. [Google Scholar] [CrossRef]
- Baker, L.B. Physiology of sweat gland function: The roles of sweating and sweat composition in human health. Temperature (Austin) 2019, 17, 211–259. [Google Scholar] [CrossRef] [PubMed]
- Kurata, R.; Futaki, S.; Nakano, I.; Fujita, F.; Tanemura, A.; Murota, H.; Katayama, I.; Okada, F.; Sekiguchi, K. Three-dimensional cell shapes and arrangements in human sweat glands as revealed by whole-mount immunostaining. PLoS One 2017, 12, e0178709. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Zhang, L.; Chen, J.; Wang, C.; Zhao, J.; Liu, X.; Yan, Y.; Tang, Y.; Chen, Z.; Li, H. Differential antigen expression between human apocrine sweat glands and eccrine sweat glands. Eur J Histochem 2023, 67, 3559. [Google Scholar] [CrossRef]
- Saga, K. Histochemical and immunohistochemical markers for human eccrine and apocrine sweat glands: an aid for histopathologic differentiation of sweat gland tumors. J Investig Dermatol Symp Proc 2001, 6, 49–53. [Google Scholar] [CrossRef]
- Ferreira, T.; Gama, A.; Seixas, F.; Faustino-Rocha, A.I.; Lopes, C.; Gaspar, V.M.; Mano, J.F.; Medeiros, R.; Oliveira, P.A. Mammary glands of women, female dogs and female rats: similarities and differences to be considered in breast cancer research. Vet Sci 2023, 10, 379. [Google Scholar] [CrossRef] [PubMed]
- Musumeci, G.; Castrogiovanni, P.; Szychlinska, M.A.; Aiello, F.C.; Vecchio, G.M.; Salvatorelli, L.; Magro, G.; Imbesi, R. Mammary gland: From embryogenesis to adult life. Acta Histochem 2015, 117, 379–85. [Google Scholar] [CrossRef] [PubMed]
- Proksch, E. pH in nature, humans and skin. J Dermatol 2018, 45, 1044–1052. [Google Scholar] [CrossRef]
- Blaak, J.; Staib, P. The relation of pH and skin cleansing. Curr Probl Dermatol 2018, 54, 132–142. [Google Scholar] [CrossRef]
- Visscher, M.O.; Adam, R.; Brink, S.; Odio, M. Newborn infant skin: physiology, development, and care. Clin Dermatol 2015, 33, 271–80. [Google Scholar] [CrossRef]
- Veltri, A.; Lang, C.; Lien, W.H. Concise review: wnt signaling pathways in skin development and epidermal stem cells. Stem Cells 2018, 36, 22–35. [Google Scholar] [CrossRef] [PubMed]
- Michalak, M.; Pierzak, M.; Kręcisz, B.; Suliga, E. Bioactive compounds for skin health: a review. Nutrients 2021, 113, 203. [Google Scholar] [CrossRef]
- Tobin, D.J.; Veysey, E.C.; Finlay, A.Y. Aging and the skin. In: Brocklehurst's Textbook of Geriatric Medicine and Gerontology Fillit HM, 8th ed.; Rockwood, K., Young J, Eds; Publisher: Elsevier; Philadelphia, PA, 2017: Chap 25.
- Chhabra, S.; Chhabra, N.; Kaur, A.; Gupta, N. Wound healing concepts in clinical practice of OMFS. J Maxillofac Oral Surg 2017, 16, 403–423. [Google Scholar] [CrossRef]
- Gupta, S.; Andersen, C.; Black, J.; de Leon, J.; Fife, C.; Lantis Ii, J.C.; Niezgoda, J.; Snyder, R.; Sumpio, B.; Tettelbach, W.; Treadwell, T.; Weir, D.; Silverman, R.P. Management of chronic wounds: diagnosis, preparation, treatment, and follow-up. Wounds 2017, 29, S19–S36. [Google Scholar] [PubMed]
- Babalska, Z.L.; Korbecka-Paczkowska, M.; Karpiński, T.M. Wound antiseptics and european guidelines for antiseptic application in wound treatment. Pharmaceuticals (Basel) 2021, 14, 1253. [Google Scholar] [CrossRef]
- Nuutila, K.; Katayama, S.; Vuola, J.; Kankuri, E. Human wound-healing research: issues and perspectives for studies using wide-scale analytic platforms. Adv wound care (New Rochelle) 2014, 3, 264–271. [Google Scholar] [CrossRef]
- Monika, P.; Chandraprabha, M.N.; Rangarajan, A.; Waiker, P.V.; Chidambara Murthy, K.N. Challenges in healing wound: role of complementary and alternative medicine. Front Nutr 2022, 8, 791899. [Google Scholar] [CrossRef]
- Mahmoud, M.; Gould, L.J. Opportunities and challenges of the management of chronic wounds: A multidisciplinary viewpoint. Chronic Wound Care Management and Research 2020, 7, 27–36. [Google Scholar] [CrossRef]
- Han, G.; Ceilley, R. Chronic wound healing: A review of current management and treatments. Adv Ther 2017, 7, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Bacci, S. Cellular mechanisms and therapies in wound healing: looking toward the future. Biomedicines 2021, 9, 1611. [Google Scholar] [CrossRef]
- Takeo, M.; Lee, W.; Ito, M. Wound healing and skin regeneration. Cold Spring Harb Perspect Med 2015, 5, a023267. [Google Scholar] [CrossRef] [PubMed]
- Arenas Gómez, C.M.; Sabin, K.Z.; Echeverri, K. Wound healing across the animal kingdom: crosstalk between the immune system and the extracellular matrix. Dev Dynam 2020, 249, 834–846. [Google Scholar] [CrossRef]
- Richardson, R.J. Parallels between vertebrate cardiac and cutaneous wound healing and regeneration. npj Regen Med, 2018, 3, 21. [Google Scholar] [CrossRef]
- Belacortu, Y.; Paricio, N. Drosophila as a model of wound healing and tissue regeneration in vertebrates. Dev Dyn 2011, 240, 2379–2404. [Google Scholar] [CrossRef]
- Seifert, A.W.; Maden, M. New insights into vertebrate skin regeneration. Int Review Cell Mol Biol 2014, 310, 129–169. [Google Scholar] [CrossRef] [PubMed]
- Ozgok Kangal, M.K.; Regan, J.P. Wound Healing 2023: In StatPearls. StatPearls Publishing.
- Healing by intention. Advances in skin & wound care, 2017, 30, 246–247. [CrossRef]
- Childs, D.R.; Murthy, A.S. Overview of wound healing and management. Surg Clin North Am 2017, 97, 189–207. [Google Scholar] [CrossRef]
- Braiman-Wiksman, L.; Solomonik, I.; Spira, R.; Tennenbaum, T. Novel insights into wound healing sequence of events. Toxicol Pathol 2007, 35, 767–779. [Google Scholar] [CrossRef]
- Grey, J.E.; Enoch, S.; Harding, K.G. Wound assessment. BMJ 2006, 332, 285–288. [Google Scholar] [CrossRef]
- Madkour, F.F.; Hassan, M.M.; Abdo, W.; Khalil, W.F. Wound healing activity of brown algae plus polyherbal extract in normal and alloxan-induced diabetic rats. Journal of Advanced Veterinary Research 2013, 3102–3108. [Google Scholar]
- Annoodee, S.; Nasuruddin, D.N. Acute Inflammatory Response. [Updated 2022 Nov 14]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih. 5560. [Google Scholar]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget 2017, 9, 7204–7218. [Google Scholar] [CrossRef]
- Palmblad, J. The acute inflammatory reaction: new concepts for old cells. J Intern Med 2010, 332, 1–4. [Google Scholar] [CrossRef]
- Fioranelli, M.; Roccia, M.G.; Flavin, D.; Cota, L.; Cota, L. Regulation of inflammatory reaction in health and disease. Int J Mol Sci 2021, 332, 5277. [Google Scholar] [CrossRef]
- Canedo-Dorantes, L.; Canedo-Ayala, M. Skin acute wound healing: a comprehensive review. Int J Inflam 2019, 2019, 3706315. [Google Scholar] [CrossRef] [PubMed]
- Tyavambiza, C.; Meyer, M.; Meyer, S. Cellular and molecular events of wound healing and the potential of silver based nanoformulations as wound healing agents. Bioengineering 2022, 9, 712. [Google Scholar] [CrossRef] [PubMed]
- Sorg, H.; Sorg, C.G.G. Skin wound healing: of players, patterns, and processes. Eur Surg Res 2023, 64, 141–157. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Guarino, M.; Hernández-Bule, M.L.; Bacci, S. Cellular and molecular processes in wound healing. Biomedicines 2023, 11, 2526. [Google Scholar] [CrossRef] [PubMed]
- Raziyeva, K.; Kim, Y.; Zharkinbekov, Z.; Kassymbek, K.; Jimi, S.; Saparov, A. Immunology of acute and chronic wound healing. Biomolecules 2021, 11, 700. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, H.N.; Hardman, M.J. Wound healing: cellular mechanisms and pathological outcomes. Open Biol 2020, 10, 200223. [Google Scholar] [CrossRef] [PubMed]
- Banu, S.A., K. Sharun, M. Mamachan, L. Abualigah, R. Kumar, A.M. Pawde, K. Dhama, S.K. Maiti, A. Wound healing and skin regeneration: present status and future directions. Journal of Experimental Biology and Agricultural Sciences 2023, 11, 871–83. [Google Scholar] [CrossRef]
- Wilgus, T.A. Fetal wound healing. In Wound healing, Tissue Repair, and Regeneration in Diabetes; Bagchi, D., Das, A., Roy, Eds.; Academic Press: Cambridge, MA, 2020; Chapter 27, 579-591. [Google Scholar]
- Yagi, L.H.; Watanuki, L.M.; Isaac, C.; Gemperli, R.; Nakamura, M.Y.; Ladeira, P.R.S. Human fetal wound healing a review of molecular and cellular aspects. Eur J Plast Surg 2016, 39, 239–246. [Google Scholar] [CrossRef]
- Harding, K.G.; Morris, H.L.; Patel, G.K. Science, medicine and the future, Healing chronic wounds. BrMed J 2002, 324, 160–163. [Google Scholar] [CrossRef]
- Toporcer, T.; Lakyová,, L. ; Radonak, J. Venous ulcer-present view on aetiology, diagnostics and therapy. Cas Lek Cesk, 2008, 147, 199–205. [Google Scholar]
- Günter, C.I.; Machens, H.G. New strategies in clinical care of skin wound healing. Eur Surg Res 2012, 49, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Ceilley, R. Chronic wound healing: a review of current management and treatments. Adv Ther 2017, 34, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K. Human wounds and its burden: an updated compendium of estimates. Adv Wound Care (New Rochelle) 2019, 8, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Kyaw, B.M.; Järbrink, K.; Martinengo, L.; Car, J.; Harding, K.; Schmidtchen, A. Need for improved definition of chronic wounds in clinical studies. Acta Derm Venereol 2018, 12, 157–158. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Guarino, M.; Bacci, S.; Pérez González, L.A.; Bermejo-Martínez, M.; Cecilia-Matilla, A.; Hernández-Bule, M.L. The role of physical therapies in wound healing and assisted scarring. Int J Mol Sci 2023, 19, 7487. [Google Scholar] [CrossRef]
- Martin, P.; Nunan, R. Cellular and molecular mechanisms of repair in acute and chronic wound healing. Br J Dermatol 2015, 173, 370–378. [Google Scholar] [CrossRef]
- Grandi, V.; Corsi, A.; Pimpinelli, N.; Bacci, S. Cellular mechanisms in acute and chronic wounds after PDT Therapy: An Update. Biomedicines 2022, 10, 1624. [Google Scholar] [CrossRef] [PubMed]
- Falanga, V.; Isseroff, R.R.; Soulika, A.M.; Romanelli, M.; Margolis, D.; Kapp, S.; Granick, M.; Harding, K. Chronic wounds. Nature Rev Dis Primers, 2022, 8, 50. [Google Scholar] [CrossRef]
- Gonzalez, A.C.; Costa, T.F.; Andrade, Z.A.; Medrado, A.R. Wound healing - A literature review. Anais brasileiros de dermatologia 2016, 91, 614–620. [Google Scholar] [CrossRef]
- Tottoli, E.M.; Dorati, R.; Genta, I.; Chiesa, E.; Pisani, S.; Conti, B. Skin wound healing process and new emerging technologies for skin wound care and regeneration. Pharmaceutics 2020, 12, 735. [Google Scholar] [CrossRef]
- Steinman, L. Elaborate interactions between the immune and nervous systems. Nature Immunol. 2004, 5, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Grandi, V.; Paroli, G.; Puliti, E.; Bacci, S.; Pimpinelli, N. Single ALA-PDT irradiation induces increase in MC degranulation and neuropeptide acute response in chronic venous ulcers: A pilot study. Photodiagnosis Photodyn Ther 2021, 34, 102222. [Google Scholar] [CrossRef] [PubMed]
- Notari, L.; Nardini, P.; Grandi, V.; Corsi, A.; Pimpinelli, N.; Bacci, S. Neuroimmunomodulation in chronic wound healing after treatment with Photodynamic Therapy: the role of iNOs. Med Sci Forum 2023, 21, 44. [Google Scholar] [CrossRef]
- Nardini, P.; Notari, L.; Magazzini, M.; Mariani, B.; Rossi, F.; Rossi, S.; Van Aardt, E.; Marszalek, K.; Grandi, V.; Corsi, A.; Pimpinelli, N.; Bacci, S. (2024). Neuroimmunomodulatory Effect of NO on Chronic Wound Healing After Photodynamic Therapy. Photodiagnosis Photodyn Therapy, 104078. Advance online publication. [CrossRef]
- Corsi, A.; Lecci, P.P.; Bacci, S.; Cappugi, P.; Pimpinelli, N. Early activation of fibroblasts during PDT treatment in leg ulcers. G.Ital Dermatol Venereol 2016, 151, 223–229. [Google Scholar] [PubMed]
- Grandi, V.; Bacci, S.; Corsi, A.; Sessa, M.; Puliti, E.; Murciano, N.; Scavone, F.; Cappugi, P.; Pimpinelli, N. ALA-PDT exerts beneficial effects on chronic venous ulcers by inducing changes in inflammatory microenvironment, especially through increased TGF-beta release: A pilot clinical and translational study. Photodiagnosis Photodyn Ther 2018, 21, 252–256. [Google Scholar] [CrossRef]
- Kadam, S.; Nadkarni, S.; Lele, J.; Sakhalkar, S.; Mokashi, P.; Kaushik, K.S. Bioengineered platforms for chronic wound infection studies: how can we make them more human-relevant? Front Bioeng Biotechnol 2019, 7, 418. [Google Scholar] [CrossRef] [PubMed]
- Babalska, Z.Ł.; Korbecka-Paczkowska, M.; Karpiński, T.M. Wound antiseptics and european guidelines for antiseptic application in wound treatment. Pharmaceuticals (Basel, Switzerland) 2021, 14, 1253. [Google Scholar] [CrossRef] [PubMed]
- Limandjaja, G.C.; Niessen, F.B.; Scheper, R.J.; Gibbs, S. The keloid disorder: heterogeneity, histopathology, mechanisms and models. Front Cell Dev Biol 2020, 8, 360. [Google Scholar] [CrossRef]
- Huang, C.; Ogawa, R. Keloidal pathophysiology: current notions. Scars Burn Heal 2021, 7. [Google Scholar] [CrossRef]
- Magni, G.; Banchelli, M.; Cherchi, F.; Coppi, E.; Fraccalvieri, M.; Rossi, M.; Tatini, F.; Pugliese, A.M.; Rossi Degl'Innocenti, D.; Alfieri, D.; Matteini, P.; Pini, R.; Pavone, F.S.; Rossi, F. Experimental study on blue light interaction with human keloid-derived fibroblasts. Biomedicines 2020, 8, 573. [Google Scholar] [CrossRef]
- Boyce, D.E.; Ciampolini, J.; Ruge, F.; Murison, M.S.; Harding, K.G. Inflammatory-cell subpopulations in keloid scars. Br J Plast Surg 2001, 54, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Bagabir, R.; Byers, R.J.; Chaudhry, I.H.; Muller, W.; Paus, R.; Bayat, A. Site-specific immunophenotyping of keloid disease demonstrates immune upregulation and the presence of lymphoid aggregates. Br J Dermatol 2012, 167, 1053–1066. [Google Scholar] [CrossRef]
- van der Veer, W.M.; Bloemen, M.C.; Ulrich, M.M.; Molema, G.; van Zuijlen, P.P.; Middelkoop, E.; Niessen, F.B. Potential cellular and molecular causes of hypertrophic scar formation. Burns 2009, 35, 15–29. [Google Scholar] [CrossRef]
- Mony, M.P.; Harmon, K.A.; Hess, R.; Dorafshar, A.H.; Shafikhani, S.H. An updated review of hypertrophic scarring. Cells 2023, 12, 678. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.; Kosaric, N.; Bonham, C.A.; Gurtner, G.C. Wound Healing: A cellular Perspective. Physiol reviews 2019, 99, 665–706. [Google Scholar] [CrossRef]
- Rossi, F.; Tatini, F.; Pini, R.; Bacci, S.; De Siena, G.; Cicchi RPavone, S.; Alfieri, D. Improved wound healing in blue LED treated superficial abrasions, Proc. SPIE (Medical Laser Applications and Laser-Tissue Interactions VI), 2013, 88030S.; [CrossRef]
- Cicchi, R.; Rossi, F.; Alfieri, D.; Bacci, S.; Tatini, F.; De Siena, G.; Paroli, G.; Pini, R.; Pavone, F.S. (2016). Observation of an improved healing process in superficial skin wounds after irradiation with a blue-LED haemostatic device. J Biophotonics 2016, 9, 645–655. [Google Scholar] [CrossRef]
- Magni, G.; Tatini, F.; Siena, G.; Pavone, F.S.; Alfieri, D.; Cicchi, R.; Rossi, M.; Murciano, N.; Paroli, G.; Vannucci, C.; Sistri, G.; Pini, R.; Bacci, S.; Rossi, F. Blue-LED-light photobiomodulation of inflammatory responses and new tissue formation in mouse-skin wounds. Life (Basel, Switzerland), 2022, 12, 1564. [Google Scholar] [CrossRef]
- Magni, G.; Tatini, F.; Bacci, S.; Paroli, G.; De Siena, G.; Cicchi, R.; Pavone, F.S.; Pini, R.; Rossi, F. Blue LED light modulates inflammatory infiltrate and improves the healing of superficial wounds. Photodermatol photoimmunol photomed 2020, 36, 166–168. [Google Scholar] [CrossRef]
- Hernández-Bule, M.L.; Naharro-Rodríguez, J.; Bacci, S.; Fernández-Guarino, M. Unlocking the Power of Light on the Skin: A Comprehensive Review on Photobiomodulation. Int. J. Mol. Sci. 2024, 25, 4483. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




