Submitted:
26 April 2024
Posted:
26 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Conjugation of RNase A to Carboxy Paramagnetic Beads
2.2. In Vitro SELEX Selection of Anti-RNase A Aptamers
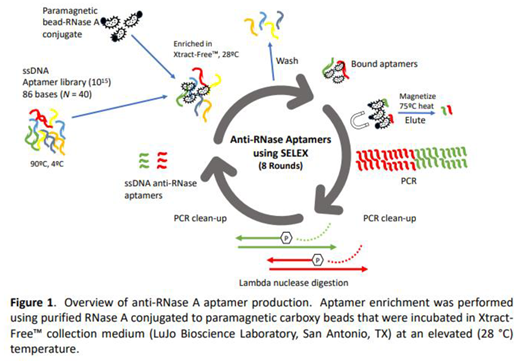
2.3. Polymerase Chain Reaction Amplification
2.4. Lambda Nuclease Digestion
2.5. Next-Generation Sequencing of SELEX Aptamer Rounds
2.6. Evaluation of ssDNA Aptamers Using Substrate Nuclease Detection
2.7. Evaluation of ssDNA Anti-RNase Aptamers Using qPCR Analysis
3. Results
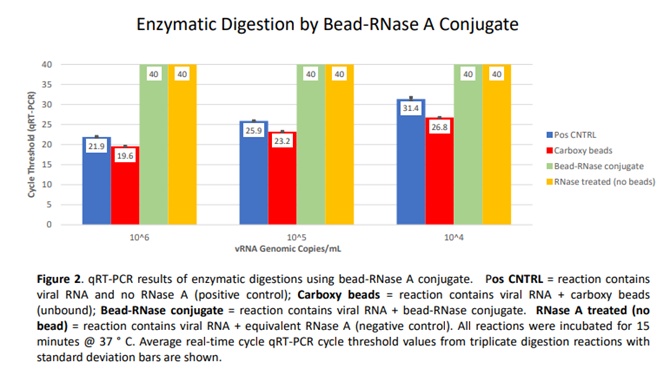
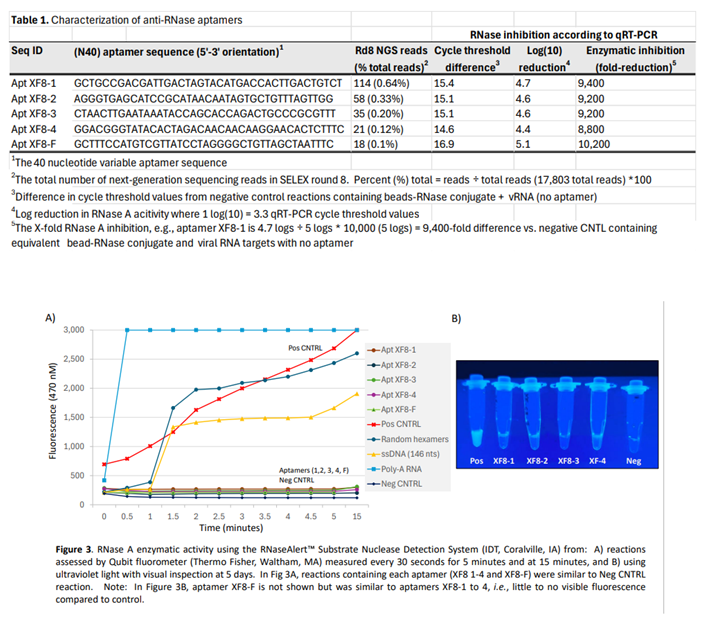
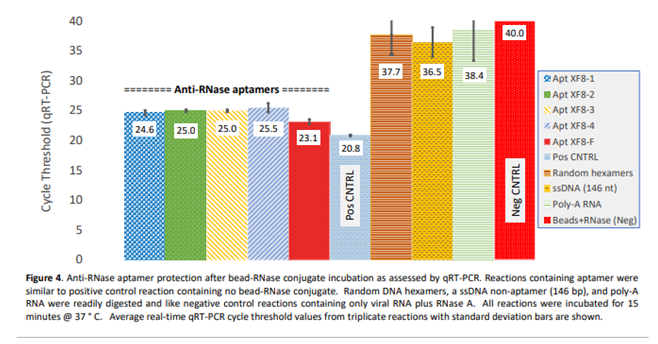
4. Discussion
5. Conclusions
References
- Ellington, A., Szostak, J. In vitro selection of RNA molecules that bind specific ligands. Nature 346, 818–822 (1990). [CrossRef]
- Wang T, Chen C, Larcher LM, Barrero RA, Veedu RN. Three decades of nucleic acid aptamer technologies: Lessons learned, progress and opportunities on aptamer development. Biotechnol Adv. 2019 Jan-Feb;37(1):28-50. [CrossRef]
- Zhang Y, Lai BS, Juhas M. Recent Advances in Aptamer Discovery and Applications. Molecules. 2019 Mar 7;24(5):941. [CrossRef]
- Kolm C, Cervenka I, Aschl UJ, Baumann N, Jakwerth S, Krska R, Mach RL, Sommer R, DeRosa MC, Kirschner AKT, Farnleitner AH, Reischer GH. DNA aptamers against bacterial cells can be efficiently selected by a SELEX process using state-of-the art qPCR and ultra-deep sequencing. Sci Rep. 2020 Dec 1;10(1):20917. [CrossRef]
- Citartan, M., Tang, TH., Tan, SC. et al. Conditions optimized for the preparation of single-stranded DNA (ssDNA) employing lambda exonuclease digestion in generating DNA aptamer. World J Microbiol Biotechnol 27, 1167–1173 (2011). [CrossRef]
- Kohlberger M, Gadermaier G. SELEX: Critical factors and optimization strategies for successful aptamer selection. Biotechnol Appl Biochem. 2022 Oct;69(5):1771-1792. [CrossRef]
- Afrasiabi S, Pourhajibagher M, Raoofian R, Tabarzad M, Bahador A. Therapeutic applications of nucleic acid aptamers in microbial infections. J Biomed Sci. 2020 Jan 3;27(1):6. [CrossRef]
- Sefah K, Phillips JA, Xiong X, Meng L, Van Simaeys D, Chen H, Martin J, Tan W. Nucleic acid aptamers for biosensors and bio-analytical applications. Analyst. 2009 Sep;134(9):1765-75. [CrossRef]
- Kim TH, Lee SW. Aptamers for Anti-Viral Therapeutics and Diagnostics. Int J Mol Sci. 2021 Apr 17;22(8):4168. [CrossRef]
- González VM, Martín ME, Fernández G, García-Sacristán A. Use of Aptamers as Diagnostics Tools and Antiviral Agents for Human Viruses. Pharmaceuticals (Basel). 2016 Dec 16;9(4):78. [CrossRef]
- Yang, W. Nucleases: Diversity of structure, function and mechanism. Q. Rev. Biophys. 2011, 44, 1–93. [CrossRef]
- Rosenberg HF. RNase A ribonucleases and host defense: an evolving story. J Leukoc Biol. 2008 May;83(5):1079-87. doi: 10.1189/jlb.1107725. Epub 2008 Jan 22. PMID: 18211964; PMCID: PMC2692241. [CrossRef]
- McPherson A, Brayer G, Cascio D, Williams R. The mechanism of binding of a polynucleotide chain to pancreatic ribonuclease. Science. 1986 May 9;232(4751):765-8. doi: 10.1126/science.3961503. PMID: 3961503. [CrossRef]
- Nichols NM, Yue D. Ribonucleases. Curr Protoc Mol Biol. 2008 Oct; Chapter 3: Unit3.13.
- Neira JL, Rico M. Folding studies on ribonuclease A, a model protein. Fold Des. 1997;2(1):R1-11. [CrossRef]
- Strom SP. Fundamentals of RNA Analysis on Biobanked Specimens. Methods Mol Biol. 2019; 1897:345-357.
- Bai H, Zhao J, Ma C, Wei H, Li X, Fang Q, Yang P, Wang Q, Wang D, Xin L. Impact of RNA degradation on influenza diagnosis in the surveillance system. Diagn Microbiol Infect Dis. 2021 Aug;100(4):115388. [CrossRef]
- Fleige S., Pfaffl M.W. RNA integrity and the effect on the real-time qRT-PCR performance. Mol. Aspects Med. 2006; 27:126–139.
- Lennette WEH, Halonen P, Murphy FA, et al. Laboratory Diagnosis of Infectious Diseases Principles and Practice_ VOLUME II Viral, Rickettsial, and Chlamydial Disease, n.d.).
- Schaeffer M. Manual of clinical microbiology. Am. J. Trop. Med. Hyg. 1971;20(3):508–508. doi: 10.4269/ajtmh.1971. [CrossRef]
- Daum LT, Worthy SA, Yim KC, Nogueras M, Schuman RF, Choi YW, Fischer GW. A clinical specimen collection and transport medium for molecular diagnostic and genomic applications. Epidemiology and Infection. 2011 Nov;139(11):1764-1773. [CrossRef]
- Banik S, Saibire K, Suryavanshi S, Johns G, Chakravorty S, Kwiatkowski R, Alland D, Banada PP. Inactivation of SARS-CoV-2 virus in saliva using a guanidium based transport medium suitable for RT-PCR diagnostic assays. PLoS One. 2021 Jun 11;16(6): e0252687.
- Mosscrop L, Watber P, Elliot P, Cooke G, Barclay W, Freemont PS, Rosadas C, Taylor GP. Evaluation of the impact of pre-analytical conditions on sample stability for the detection of SARS-CoV-2 RNA. J Virol Methods. 2022 Nov; 309:114607.
- Drygin YF, Butenko KO, Gasanova TV. Environmentally friendly method of RNA isolation. Anal Biochem. 2021 May 1; 620:114113. [CrossRef]
- Ertell. K. A Review of Toxicity and Use and Handling Considerations for Guanidine, Guanidine Hydrochloride, and Urea. 2006. Available at: www.pnnl.gov/main/publications/external/technical_reports/PNNL-15747.pdf.
- Crotti. N. (June, 2020). FDA warns of cyanide gas danger with improper use of Hologic COVID-19 tests. Available at: www.massdevice.com/fda-warns-of-cyanide-gas-danger-with-improper-use-of-hologic-covid-19-tests/.
- Centers for Disease Control and Prevention (CDC). CDC’s Laboratory Outreach Communication System (LOCS). Available at: www.cdc.gov/locs/2020/mtm_and_cyanide_gas.html.
- Daum, LT; Rodriguez, JD.; Ward, SR.; Chambers, JP. Extraction-Free Detection of SARS-CoV-2 Viral RNA Using LumiraDx’s RNA Star Complete Assay from Clinical Nasal Swabs Stored in a Novel Collection and Transport Medium. Diagnostics 2023, 13, 3010.
- Daum LT, Canas LC, Arulanandam BP, Niemeyer D, Valdes JJ, Chambers JP. Real-time RT-PCR assays for type and subtype detection of influenza A and B viruses. Influenza Other Respir Viruses. 2007 Jul;1(4):167-75. [CrossRef]
- Kim N, Kwon A, Roh EY, Yoon JH, Han MS, Park SW, Park H, Shin S. Effects of Storage Temperature and Media/Buffer for SARS-CoV-2 Nucleic Acid Detection. Am J Clin Pathol. 2021 Feb 4;155(2):280-285.
- Miller JM, Binnicker MJ, Campbell S, et al. A guide to utilization of the microbiology laboratory for diagnosis of infectious diseases: 2018 update by the Infectious Diseases Society of America and the American Society for Microbiology. Clin Infect Dis. 2018;67: e1-e94. [CrossRef]
- World Health Organization, 2020. Laboratory testing for coronavirus disease (COVID-19) in suspected human cases: interim guidance, 19 March 2020. World Health Organization. Accessed 6th April 2021.
- Dsa, O. C., Kadni, T. S., & N, S. (2023). From cold chain to ambient temperature: transport of viral specimens- a review. Annals of Medicine, 55(2). [CrossRef]
- Shaw KJ, Thain L, Docker PT, Dyer CE, Greenman J, Greenway GM, Haswell SJ. The use of carrier RNA to enhance DNA extraction from microfluidic-based silica monoliths. Anal Chim Acta. 2009 Oct 12;652(1-2):231-3. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



