Submitted:
13 April 2024
Posted:
15 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
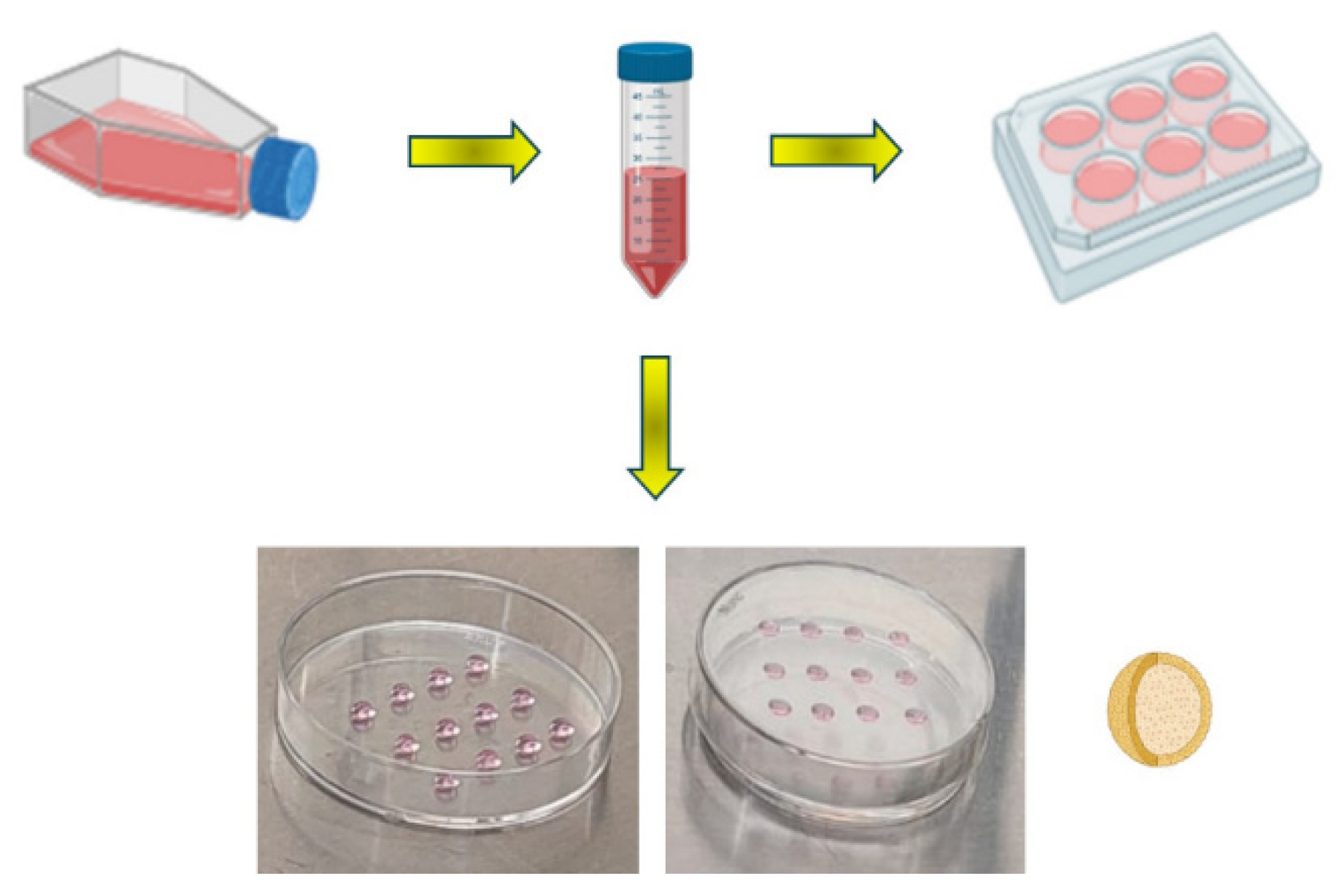
2.1. 2D and 3D Cell Cultures
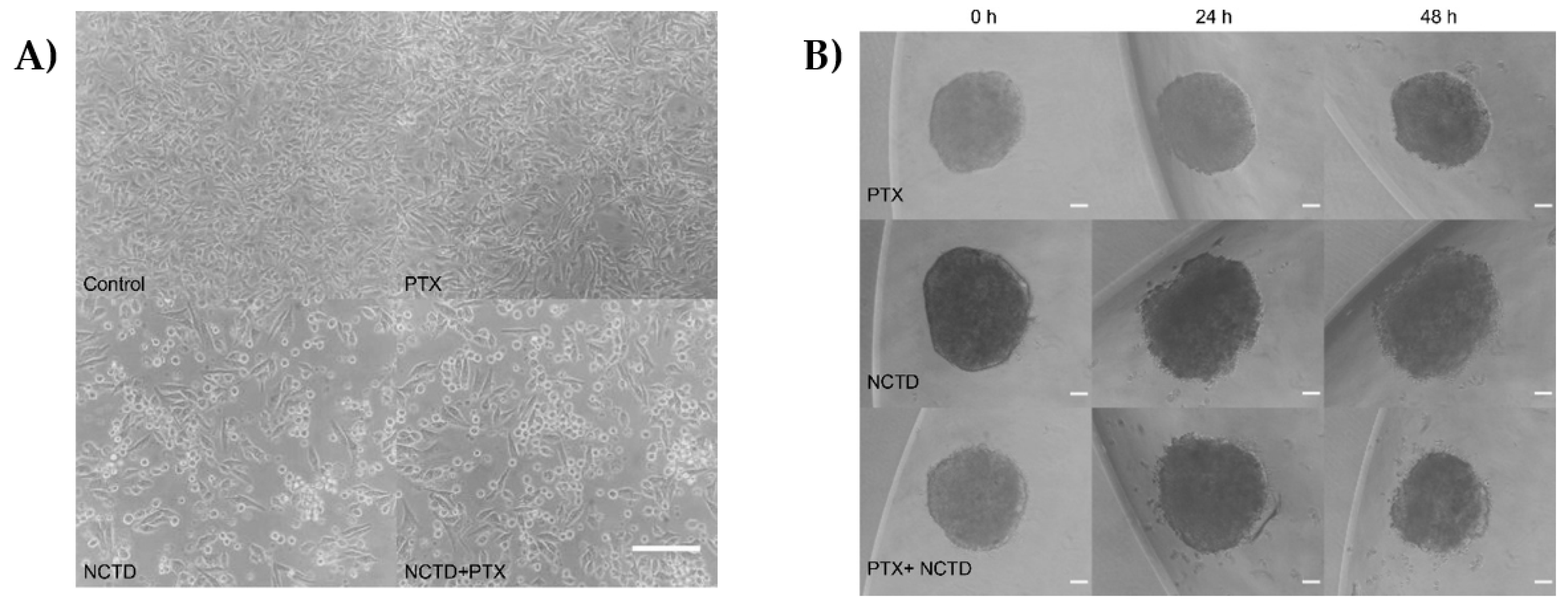
2.2. Treatment of 2D and 3D Cell Cultures with PXT and NCTD
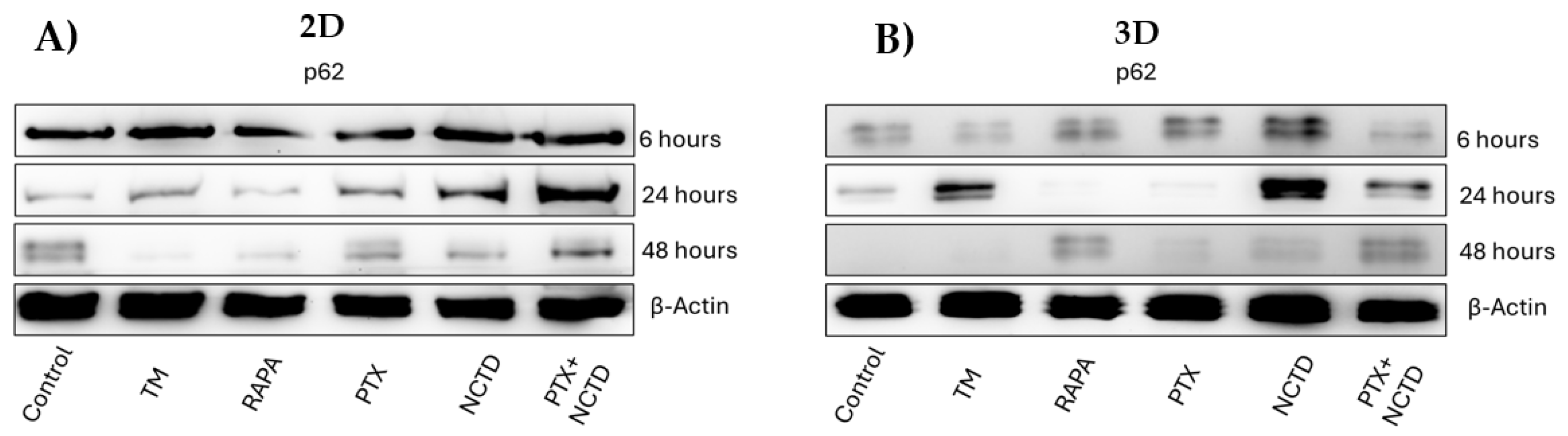
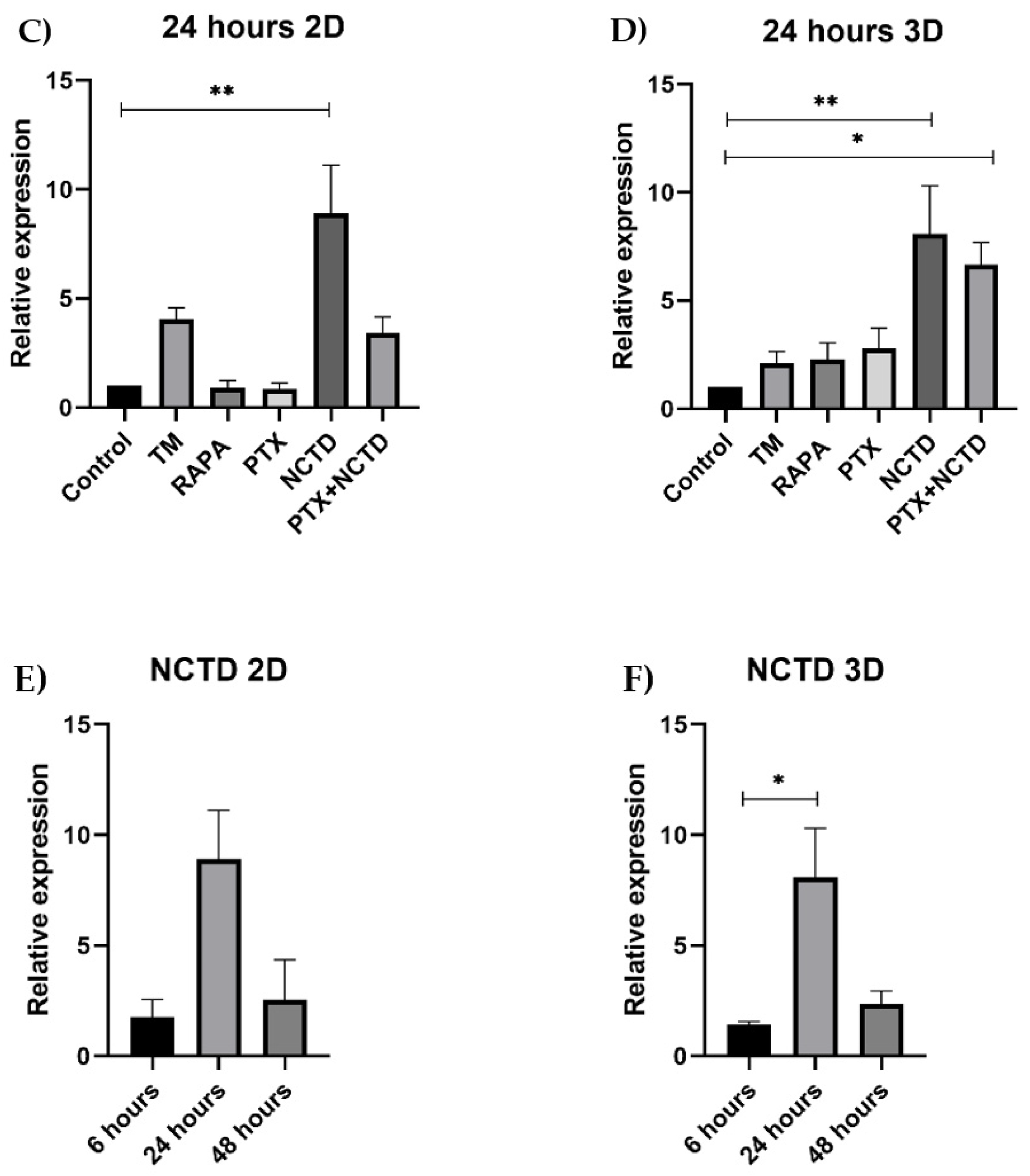
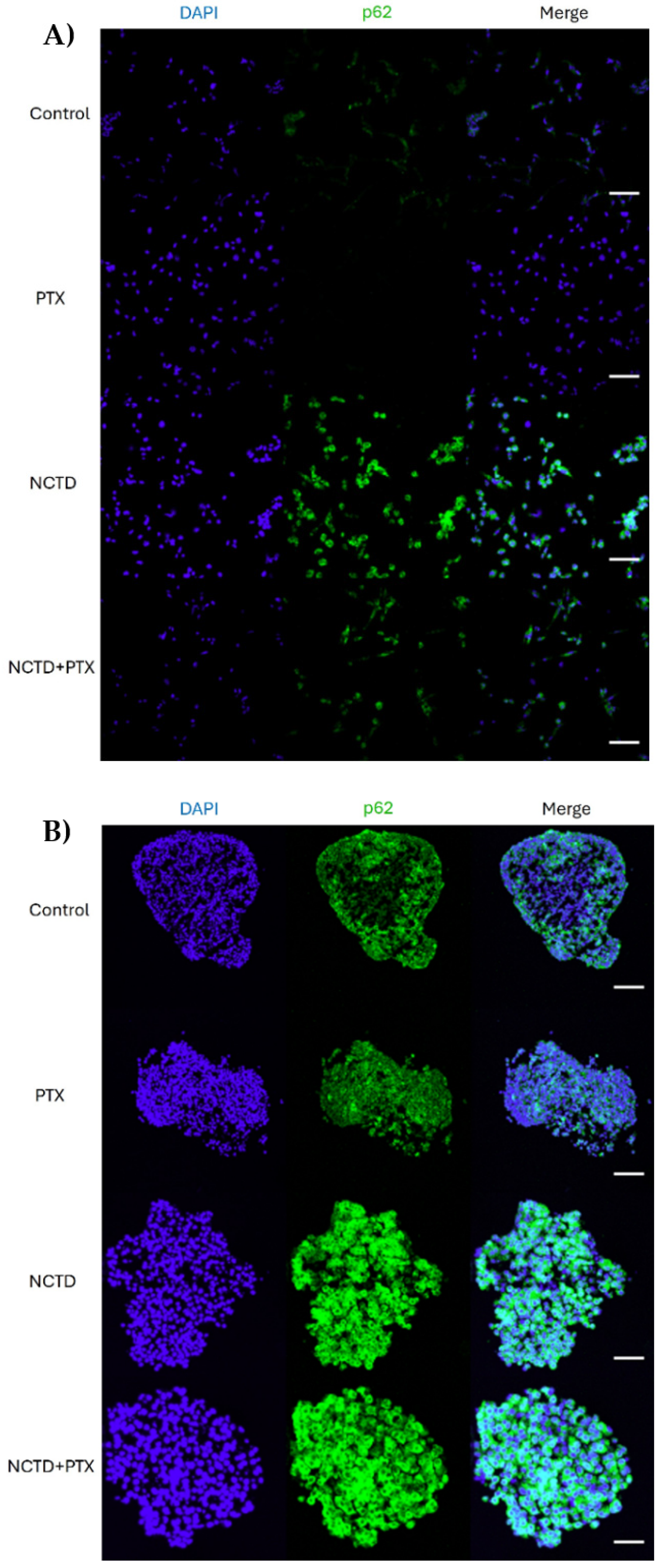
2.3. Autophagy p62 Expression in 2D and 3D Cultures
2.4. Expression of p62 in 2D and 3D Cultures by Immunofluorescence
3. Discussion
4. Materials and Methods
4.1. Two-Dimensional (2D) and Three-Dimensional (3D) Cell Culture Models
4.2. Western Blot Analysis
4.2. Slices of the 3D Cultures of the Spheroids
4.3. Immunofluorescence of 2D and 3D Cultures
4.3. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dhanyamraju P.K.; Patel T.N. Melanoma therapeutics: a literature review. J Biomed Res. 2022, 36, 77–97. [CrossRef]
- Patton E.E.; Mueller K.L.; Adams D.J.; Anandasabapathy N.; Aplin A.E. Melanoma models for the next generation of therapies. Cancer Cell. 2021, 39, 610–631. [CrossRef]
- Brancato V.; Oliveira J.M.; Correlo V.M.; Reis R.L.; Kundu S.C. Could 3D models of cancer enhance drug screening? Biomaterials, 232, 119744. [CrossRef]
- Sherman H.; Gitschier H.J.; Rossi A.E. A Novel Three-Dimensional Immune Oncology Model for High-Throughput Testing of Tumoricidal Activity. Front. Immunol. 2018, 9, 857. [CrossRef]
- Kapałczyńska M.; Kolenda T.; Przybyła W.; Zajączkowska M.; Teresiak A.; Filas V.; Ibbs M.; Bliźniak R.; Łuczewski L.; Lamperska K. 2D and 3D cell cultures – a comparison of different types of cancer cell cultures. Arch Med Sci. 2018, 14, 910–919. [CrossRef]
- Cave D.D; Rizzo R.; Sainz B., Jr.; Gigli G.; del Mercato L.L.; Lonardo E. The Revolutionary Roads to Study Cell–Cell Interactions in 3D In Vitro Pancreatic Cancer Models. Cancers, 2021, 13, 930. [CrossRef]
- Khandia R.; Dadar M.; Munjal A.; Dhama K.; Karthik K.; Tiwari R.; Yatoo M.I.; Iqbal H.M.N; Singh K.P.; Joshi S.K; Chaicumpa W. A Comprehensive Review of Autophagy and Its Various Roles in Infectious, Non-Infectious, and Lifestyle Diseases: Current Knowledge and Prospects for Disease Prevention, Novel Drug Design, and Therapy. Cells. 2019, 8, 674. [CrossRef]
- Brier L.W.; Ge L.; Stjepanovic G.; Thelen A.M.; Hurley J.H.; Schekman R. Regulation of LC3 Lipidation by the Autophagy-Specific Class III phosphatidylinositol-3 Kinase Complex. Mol Biol Cell., 2019, 30, 1098-1107. [CrossRef]
- Nath S.; Dancourt J.; Shteyn V.; Puente G.; Fong W.M.; Nag S.; Bewersdorf J.; Yamamoto A.; Antonny B.; Melia T.J. Lipidation of the LC3/GABARAP family of autophagy proteins relies upon a membrane curvature-sensing domain in Atg3. Nat Cell Biol., 2014, 16, 415–424. [CrossRef]
- Lin X.; Li S.; Zhao Y.; Ma X.; Zhang K.; He X.; Wang Z. Interaction domains of p62: a bridge between p62 and selective autophagy. DNA Cell Biol. 2013, 32, 220-227. [CrossRef]
- Pankiv S.; Clausen T.H.; Lamark T.; Brech A.; Bruun J.A.; Outzen H.; Øvervatn A.; Bjørkøy G.; Johansen T. p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J Biol Chem. 2007, 282, 24131-24145. [CrossRef]
- Yun C.W.; Lee S.H. The Roles of Autophagy in Cancer. Int J Mol Sci., 2018, 19(11), 3466. [CrossRef]
- Frampton J.E.; Brogden R.N. Pentoxifylline (Oxpentifylline) A Review of its Therapeutic Efficacy in the Management of Peripheral Vascular and Cerebrovascular Disorders. Drugs & Aging, 1995, 7, 480-503. [CrossRef]
- Golunski G.; Woziwodzka A.; Piosik J. Potential Use of Pentoxifylline in Cancer Therapy. Curr Pharm Biotechnol., 2018, 19, 206-216. [CrossRef]
- Pratibha D.; Yuvraj N.; Rajiv P.G. Pentoxifylline: A Potent Inhibitor of Angiogenesis via Blocking STAT3 Signaling in B16F10 Melanoma. Int. J. Tumor Ther., 2013, 2, 1-9. [CrossRef]
- Mohammad Z.K.; Rajiv P.G. Pentoxifylline Inhibits Melanoma Tumor Growth and Angiogenesis by Targeting STAT3 Signaling Pathway. Biomed Pharmacother, 2013, 67, 399-405. [CrossRef]
- Sharma K.; Ishaq M.; Sharma G.; Khan M.A.; Dutta R.K.; Majumdar S. Pentoxifylline triggers autophagy via ER stress response that interferes with Pentoxifylline induced apoptosis in human melanoma cells. Biochem Pharmacol., 2016, 103, 17-28. [CrossRef]
- Wang G.S. Medical uses of Mylabris in ancient China and recent studies. J Ethnopharmacol, 1989, 26, 147-162. [CrossRef]
- An W.W.; Wang M.W.; Tashiro S.; Onodera S.; Ikejima T. Norcantharidin induces human melanoma A375-S2 cell apoptosis through mitochondrial and caspase pathways. J Korean Med Sci., 2004, 19, 560–566. [CrossRef]
- Liu S.; Yu H.; Kumar S.M.; Martin J.S.; Bing Z.; Sheng W.; Bosenberg M.; Xu X. Norcantharidin induces melanoma cell apoptosis through activation of TR3 dependent pathway. Cancer Biol Ther., 2011, 12, 1005-1014. [CrossRef]
- Liu Z.; Li B.; Cao M.; Jiang J. Norcantharidin triggers apoptotic cell death in non-small cell lung cancer via a mitophagy-mediated autophagy pathway. Ann Transl Med., 2021, 9, 971. [CrossRef]
- Müller I.; Kulms D. A 3D Organotypic Melanoma Spheroid Skin Model. J Vis Exp. 2018, 135, 57500. [CrossRef]
- Ocampo-Godinez J.M.; Gonzalez-Quiroz J.L.; Cote-Palafox H.; George E.; Vergara-Lope Nuñez J.A.; Villagomez-Olea G.; Vazquez-Vazquez F.C.; Lopez-Villegas E.O.; Leon-Avila G.; Dominguez-Lopez M.L.; Alvarez-Perez M.A. Primary explants of the postnatal thymus allow the expansion of clonogenic thymic epithelial cells that constitute thymospheres. Stem Cell Res Ther., 2023 ,14, 312. [CrossRef]
- Nayak P.; Bentivoglio V.; Varani M.; Signore A. Three-Dimensional In Vitro Tumor Spheroid Models for Evaluation of Anticancer Therapy: Recent Updates. Cancers., 2023, 15, 4846. [CrossRef]
- Zhou J.; Ren Y.; Tan L.; Song X.; Wang M.; Li Y.; Cao Z.; Guo C. Norcantharidin: research advances in pharmaceutical activities and derivatives in recent years. Biomed Pharmacother. 2020, 131, 110755. [CrossRef]
- Xiao W.; Dai B.; Zhu Y.; Ye D. Norcantharidin induces autophagy-related prostate cancer cell death through Beclin-1 upregulation by miR-129-5p suppression. Tumour Biol., 2015. [CrossRef]
- Han Z.; Li B.; Wang J.; Zhang X.; Li Z.; Dai L.; Cao M.; Jiang J. Norcantharidin Inhibits SK-N-SH Neuroblastoma Cell Growth by Induction of Autophagy and Apoptosis. Technol Cancer Res Treat., 2017, 16, 33-44. [CrossRef]
- Xu L.; Su B.; Mo L.; Zhao C.; Zhao Z.; Li H.; Hu Z.; Li J. Norcantharidin Induces Immunogenic Cell Death of Bladder Cancer Cells through Promoting Autophagy in Acidic Culture. Int J Mol Sci., 2022, 23, 3944. [CrossRef]
- Pangilinan C.; Xu X.; Herlyn M.; Liang C. Autophagy Paradox: Strategizing Treatment Modality in Melanoma. Curr Treat Options Oncol., 2023, 2, 130-145. [CrossRef]
- Kumar A.V.; Mills J.; Lapierre L.R. Selective Autophagy Receptor p62/SQSTM1, a Pivotal Player in Stress and Aging. Front Cell Dev Biol., 2022, 10, 793328. [CrossRef]
- Li H.C.; Xia Z.H.; Chen Y.F.; Yang F.; Feng W.; Cai H.; Mei Y.; Jiang Y.M.; Xu K.; Feng D.X. Cantharidin Inhibits the Growth of Triple-Negative Breast Cancer Cells by Suppressing Autophagy and Inducing Apoptosis in Vitro and in Vivo. Cell Physiol Biochem., 2017, 43, 1829-1840. [CrossRef]
- Yun C.W.; Jeon J.; Go G.; Lee J.H.; Lee S.H. The Dual Role of Autophagy in Cancer Development and a Therapeutic Strategy for Cancer by Targeting Autophagy. Int. J. Mol. Sci., 2021, 22, 179. [CrossRef]
- Bingel C.; Koeneke E.; Ridinger. Three-dimensional tumor cell growth stimulates autophagic flux and recapitulates chemotherapy resistance. Cell Death Dis., 2017, 8, e3013. [CrossRef]
- Imamura Y.; Mukohara T.; Shimono Y.; Funakoshi Y.; Chayahara N.; Toyoda M.; Kiyota N.; Takao S.; Kono S.; Nakatsura T.; Minami H. Comparison of 2D- and 3D-culture models as drug-testing platforms in breast cancer. Oncol Rep., 2015, 33, 1837-1843. [CrossRef]
- Islam M.A.; Sooro M.A.; Zhang P. Autophagic Regulation of p62 is Critical for Cancer Therapy. Int J Mol Sci., 2018, 19, 1405. [CrossRef]
- Philipson E.; Engström C.; Naredi P.; Bourghardt Fagman J. High expression of p62/SQSTM1 predicts shorter survival for patients with pancreatic cancer. BMC Cancer. 2022, 22, 347. [CrossRef]
- Moscat J.; Karin M.; Diaz-Meco M.T. p62 in Cancer: Signaling Adaptor Beyond Autophagy. Cell., 2016, 167, 606-609. [CrossRef]
- Tang D.Y.; Ellis R.A.; Lovat P.E. Prognostic Impact of Autophagy Biomarkers for Cutaneous Melanoma. Front Oncol., 2016, 6, 236. [CrossRef]
- Ward A.; Clissold S.P. Pentoxifylline. A review of its pharmacodynamic and pharmacokinetic properties, and its therapeutic efficacy. Drugs. 1987, 34, 50-97. [CrossRef]
- Dua P.; Gude R.P. Antiproliferative and antiproteolytic activity of pentoxifylline in cultures of B16F10 melanoma cells. Cancer Chemother Pharmacol., 2006, 58, 195-202. [CrossRef]
- Kamran M.Z.; Gude R.P. Preclinical evaluation of the antimetastatic efficacy of Pentoxifylline on A375 human melanoma cell line. Biomed Pharmacother. 2012, 66, 617-26. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



