1. Introduction
Maternal care is a pivotal factor in shaping offspring development, encompassing a myriad of behavioral, physiological, and neurobiological processes [
1]. A series of experiments dedicated to measuring the impact of reduced maternal care in rats has demonstrated important detrimental effects on central nervous system (CNS) development and adult brain function [
2,
3]. Such impacts on brain development were also observed across nonhuman primates and humans [
4,
5,
6,
7]. Among these processes, the intricate interplay between maternal care, brain inflammation, and microglial dynamics has emerged as a focal point in understanding neurodevelopmental outcomes [
8,
9].
Microglia, the resident immune cells of the central nervous system, play multifaceted roles in modulating brain homeostasis, synaptic pruning, and [
10] profound influences on microglial function and neuroinflammatory states, thereby impacting offspring's neurobiological trajectories [
11,
12,
13]. Microglia exhibit remarkable plasticity and responsiveness to environmental stimuli, including maternal care quality and quantity. Moreover, microglial activation and subsequent inflammatory responses are intricately linked to neurodevelopmental outcomes, with implications for synaptic pruning, neurogenesis, and cognitive function [
8,
9,
10].
Maternal care encompasses a spectrum of nurturing behaviors, including grooming, nursing, and protection, crucial for offspring survival and development [
14]. Studies have elucidated the crucial role of maternal care in regulating microglial morphology, distribution, and activity during critical periods of neurodevelopment. Moreover, maternal care has been implicated in modulating neuroinflammatory processes, with implications for neuroplasticity and cognitive function in the offspring [
10,
15,
16].
Intriguingly, maternal care dynamics are not uniform across reproductive contexts, with litter size representing a critical determinant. Variations in litter size introduce differential demands on maternal resources and caregiving behaviors, thereby influencing offspring outcomes. While larger litter sizes often necessitate increased maternal investment, smaller litter sizes may afford heightened maternal attention and resources per offspring. Consequently, investigating the neurobiological consequences of varying litter sizes provides a unique lens into the interplay between maternal care, microglial function, and neuroinflammation.
In rodent models, studies have revealed lasting metabolic changes and epigenetic implications associated with extreme litter sizes, ranging from as few as three or four pups per litter [
17,
18,
19] to as high as 16 to 18 pups per litter [
20,
21,
22]. A profound influence of litter size on the quantity of maternal care allocated per pup has been demonstrated, exhibiting notable effects on metabolic traits, behavioral patterns, and immune responses [
23,
24].
Nonetheless, the understanding of the long-term effects of litter size on the interface between the neural and immune systems remains limited [
23,
25], particularly on the CNS immune cells of adult and aged animals [
26]. While existing evidence highlights the impact of litter size on corticosterone levels during early postnatal stages [
20,
27], substantial gaps persist in our knowledge regarding the enduring consequences of microglial response in the CNS [
28,
29,
30,
31].
Furthermore, recent studies have begun elucidating the role of voluntary exercise as a modulator of microglial activity and neuroinflammation in the context of maternal care. Exercise-induced alterations in microglial phenotype and function may interact with maternal care dynamics to shape offspring neurodevelopment [
32,
33]. This multifaceted enrichment approach fosters the expression of neurotrophins and prompts epigenetic modifications, thereby aiding in alleviating developmental deficits within the central nervous system (CNS) [
34].
Voluntary exercise has been found to mitigate microglial proliferation and inhibit their activation within the hippocampus of stress-induced depressed rats [
35]. Additionally, in aged animals, exercise promotes a pro-neurogenic phenotype in microglia [
36], while moderate physical training has shown promise in mitigating the effects of perinatal undernutrition on the peripheral immune system [
37]. Thus, unraveling the interconnected pathways linking maternal care, microglial activity, inflammation, and voluntary exercise holds promise for elucidating mechanisms underlying neurodevelopmental plasticity and resilience.
Our previous studies underscored the dentate gyrus' increased vulnerability to the effects of aging and sedentary lifestyles, especially observed in rats reared in larger litters. [
31]. Moreover, we have shown that environmental enrichment contributes to partially restoring age-related memory impairments and associated astroglia changes in mice [
38,
39]. This study explores the enduring impacts of two distinct litter sizes (6 and 12 pups/dam) on microglia's quantity and laminar distribution in the dentate gyrus.
We also scrutinize the object recognition and spatial memories of adult mature rats (4 months old) and aged rats (23 months old) that remained sedentary throughout their lives or engaged in five weeks of exercise later in life. Employing an optical fractionator, we quantified microglial numbers in the dentate gyrus to test the hypothesis that litter size and a sedentary lifestyle could alter microglia's quantity and laminar distribution. Additionally, we investigated potential associations between these alterations and impairments in object recognition and spatial memory. Furthermore, we explored whether five weeks of exercise later in life could mitigate the changes in microglial numbers and cognitive decline.
2. Materials and Methods
All procedures undertaken in this investigation received approval from the institutional animal care committee of the Federal University of Pernambuco, Brazil. These procedures were conducted strictly with the "Principles of Laboratory Animal Care" outlined by the NIH (National Institutes of Health).
2.1. Experimental Groups
The study employed offspring from an outbred colony strain of Wistar rats obtained from the Department of Nutrition at the Federal University of Pernambuco. Female Wistar rats were fed ad libitum with a rodent laboratory chow diet (Purina do Brazil Ltd) containing 23% protein and were housed in groups of 2 or 3. Upon mating and gestation, female Wistar rats typically gave birth to litters ranging from 7 to 12 pups. To manipulate maternal care and suckling competition levels, we established two pup-to-dam ratios: 6:1 (representing small litter size, N = 20) and 12:1 (representing larger litter size, N = 20) within 48 hours post-birth.
Within the same timeframe, pups from different litters underwent random redistribution among litters. Offspring from different dams were pooled, mixed, and then randomly distributed among dams in litters of six or twelve pups per dam. This method aimed to mitigate the potential influence of genetic traits specific to individual litters. All male pups were chosen and allocated among the litters, while female pups were included only if the number of male pups was insufficient to meet the predetermined litter size during lactation. Following weaning, exclusively male rats were chosen as experimental subjects.
The objective was to investigate whether litter size affects milk access competition and maternal care [
40,
41,
42]. To achieve this, we meticulously observed and quantified maternal care provided by each dam across small and large litters from the 3rd to the 21st postnatal day (weaning period). Additionally, body weights were recorded at various intervals to monitor subjects' growth under different experimental conditions.
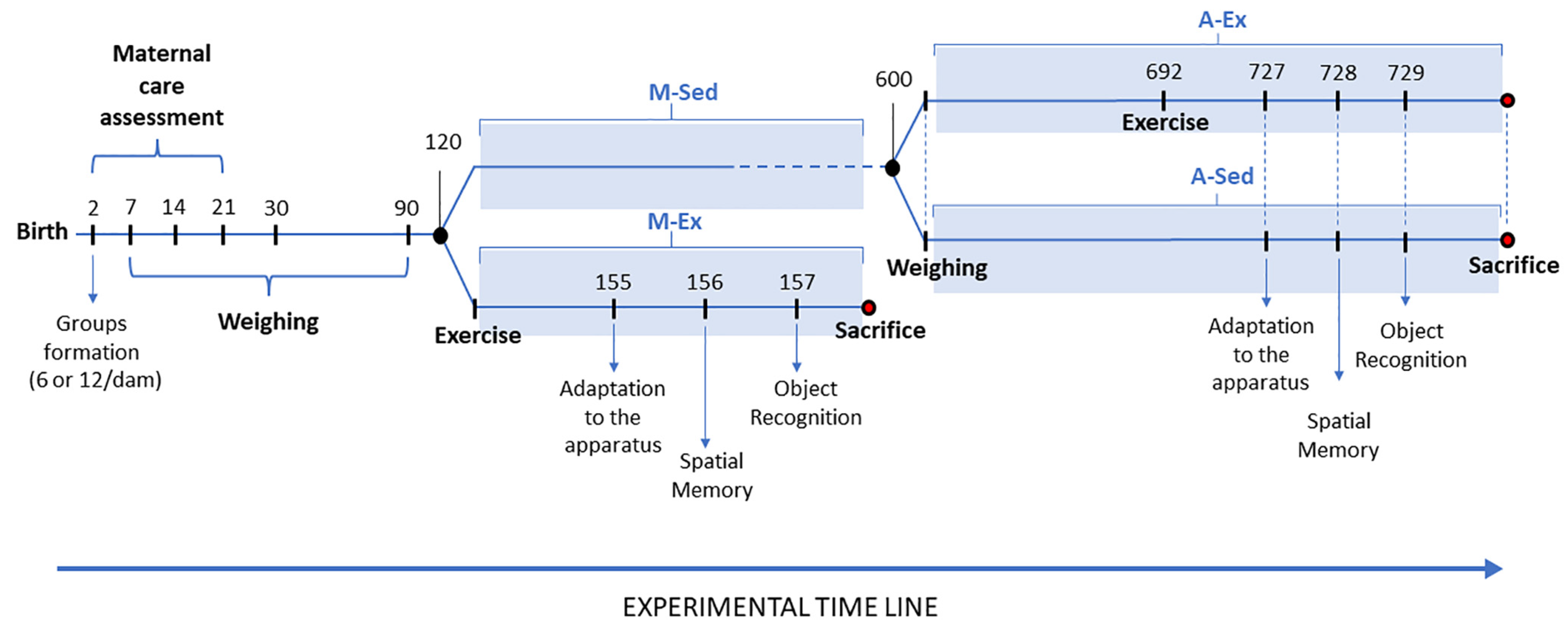
Figure 1 illustrates the timeline of the experimental procedures involving pre- and post-weaned Wistar rats.
2.2. Maternal Care Assessment
To assess the variation in maternal care between dams with large and small litters, we systematically observed maternal behavior using a predefined protocol. Each observation session lasted for one hour[
43,
44] with consistent monitoring at specific intervals. These observations occurred four times during the light phase (09:00, 11:00, 13:00, and 15:00) and twice during the dark phase of the light-dark cycle (19:00 and 21:00) performed at defined intervals: four times during the light phase (09:00, 11:00, 13:00, and 15:00) and twice during the dark phase of the light-dark cycle (19:00 and 21:00).
The behaviors recorded encompassed the following:
- a)
Mother's absence from pups.
- b)
Mother engaging in licking/grooming behavior towards pups, including both general body grooming and specific attention to the anogenital region.
- c)
Mother nursing pups in an arched-back or low-back posture, often referred to as the 'blanket' posture where the mother lays over the pups.
- d)
Mother assuming a passive posture, lying either on her back or side while the pups nurse.
- e)
Nest-building behavior exhibited by the mother.
- f)
Mother carrying or retrieving pups to the nest.
- g)
Pups being away from the nest.
- h)
Mother displaying passive behavior within the nest.
- i)
Mother being away from pups while eating.
2.3. Environment, Exercise, and Sedentary Conditions
After the suckling period, all experimental cohorts were granted unrestricted access to an identical rodent laboratory chow diet (Purina do Brazil Ltd) containing 23% protein. They were then housed in polypropylene cages (51 × 35.5 × 18.5 cm) in groups of 2 or 3 animals, ensuring an environment consistent with standard laboratory conditions: a 12-hour light-dark cycle (lights on at 6 a.m.) and a room temperature maintained at 23 ± 1°C. These housing parameters remained consistent for all animals post-weaning until the time of sacrifice.
Subsequently, either after 4 or 17 months, half of each experimental cohort (n=10) commenced a progressive exercise regimen on a treadmill for five weeks, following the specified parameters outlined in
Table 1. The treadmill utilized (Insight Equipamentos Ltda, Ribeirão Preto, São Paulo, Brazil) offered precise control over the duration and speed of the moving platform. As a control measure, the sedentary animals were transferred each morning to an inactive treadmill for an equivalent duration.
2.4. Behavioral Assessment and Testing Procedures
Following the exercise regimen, all mature (4-month-old) and aged (23-month-old) rats, whether sedentary or engaged in exercise, underwent spatial memory and object recognition tests across all experimental groups.
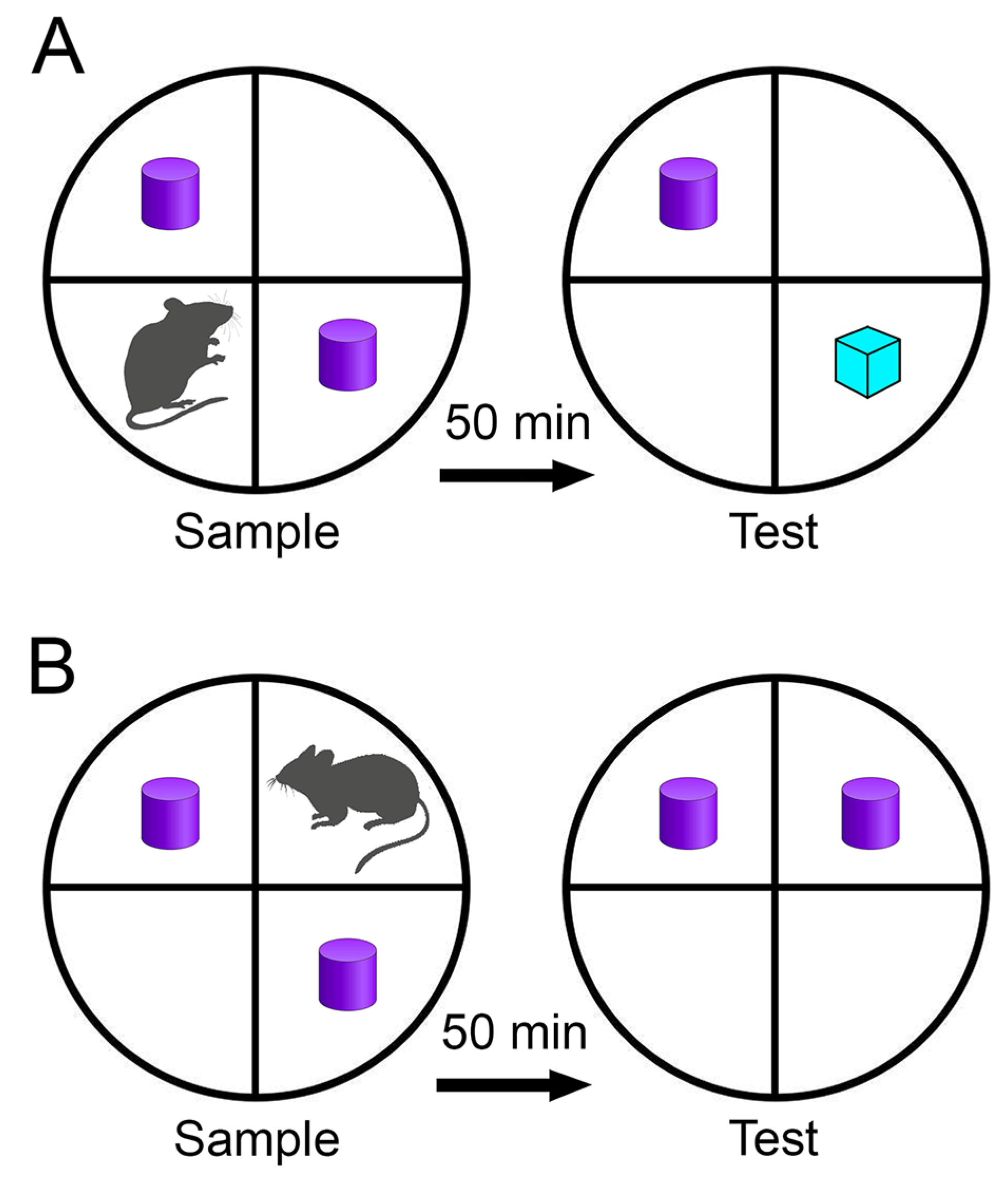
Figure 2 illustrates a schematic diagram of the object recognition and object placement apparatus along with the corresponding test procedure. This study employed single-trial tests to evaluate object identity and placement recognition memories.
Apparatus Description and Test Protocol:
The apparatus utilized for the single-trial object recognition and spatial memory tests consisted of a painted, varnished wood circular container measuring 1 meter in diameter. The floor featured marked lines to demarcate four quadrants, while the luminance at the center of the circular box floor was measured at 2.4 cd/m². Detailed protocols and rationales for test selections have been previously documented [
45,
46,
47].
Behavioral assessments spanned five days, with one day allocated for open field habituation, two days for object habituation, and two days for testing purposes — dedicating one day to each specific test.
To minimize the influence of innate preferences for specific objects or materials, objects made from the same material yet featuring distinct geometries were chosen. These objects were deliberately selected for their ease of discrimination and offered similar potential for interaction [
48]. Constructed from plastic, the objects varied in shapes, heights, and colors. Before introducing each rat into the arena, both the arena and objects underwent cleaning using 75% ethanol to minimize any discernible olfactory cues.
Testing Procedures:
Open-field habituation: Each animal explored the arena for 5 minutes without any objects present.
Object habituation: Animals encountered two identical objects (not used during test days) placed at distinct quadrants within the arena for 5 minutes, repeated three times with 50-minute intervals.
One-trial recognition tests:
a. Object identity test: Rats explored two identical objects during a 5-minute sample trial. After a 50-minute intermission, a second 5-minute test trial introduced a "novel" object alongside a "familiar" one.
b. Object identity recognition test: Mirroring the previous procedure, except one identical object was relocated during the test trial, termed the "displaced" object.
Behavioral Data Analysis:
Behavioral data underwent analysis using parametric statistics. A two-tailed t-test for dependent groups discerned significant differences in exploration time [
49]. Performance was evaluated based on the exploration time for each object, expressed as a proportion of the total exploration time. Distinctions were deemed significant when one object's exploration time exceeded 60% compared to the other. The threshold for significance was set at p<0.05.
2.5. Immunohistochemical Analysis
After the completion of behavioral tests, rats underwent weighing and anesthesia induction via intraperitoneal injection of 2,2,2-tribromoethanol (0.04 ml/g of body weight). Following anesthesia, transcardial perfusion commenced, initially with heparinized saline, followed by 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.2–7.4). Subsequently, alternate series of sections (70 µm thickness) were obtained using a Vibratome (Micron) and subjected to immunolabeling using a polyclonal antibody targeting ionized calcium-binding adapter molecule 1 (IBA-1) for microglia and/or macrophage detection (anti-Iba1, #019-19741; Wako Pure Chemical Industries Ltd., Osaka, Japan). Chemicals utilized in this process were sourced from Sigma-Aldrich (Poole, UK) or Vector Labs (Burlingame, CA, USA).
During the immunolabeling procedure, the free-floating sections underwent a series of treatments. Initially, they were pre-treated with 0.2 M boric acid (pH 9) at a temperature range of 65–70°C for 60 minutes to enhance antigen retrieval. Subsequent steps involved washing the sections in 5% phosphate-buffered saline (PBS) and then immersing them in 10% normal goat serum (for IBA-1 immunolabeling; Vector Laboratories) for 20 minutes.
Following this, the sections were incubated with anti-Iba1 (2 µg/ml in PBS) diluted in 0.1 M PBS (pH 7.2–7.4) for three days at 4°C with continuous, gentle agitation. After primary incubation, the sections were washed and incubated overnight with a biotinylated secondary antibody (goat anti-rabbit for IBA-1, 1:250 in PBS, Vector Laboratories).
Subsequently, the sections were immersed in a 3% H2O2 solution in PBS to inhibit endogenous peroxidases. Following PBS washing, the sections were transferred to a solution of avidin–biotin–peroxidase complex (VECTASTAIN ABC kit; Vector Laboratories) for 1 hour. After another round of washing, the sections were incubated in 0.1 M acetate buffer (pH 6.0) for 3 minutes and then developed in a solution comprising 0.6 mg/ml diaminobenzidine, 2.5 mg/ml ammonium nickel chloride, and 0.1 mg/ml glucose oxidase [
50].
To validate the specificity of the immunohistochemical pattern, a negative control was conducted by omitting the primary antibody. This control revealed the absence of immunoreactivity across all structures, confirming specificity [
51].
2.6. Quantification Using the Optical Fractionator Method
The optical fractionator method represents a precise stereological approach for quantifying cellular populations, amalgamating the functionalities of an optical dissector and a fractionator. Its efficacy in assessing cell numbers across diverse brain regions has been well-documented in numerous studies [
52,
53,
54]. Notably, the optical fractionator remains unaffected by histological alterations, shrinkage, or injury-induced expansion (West et al. 1991). Our aim was to investigate the hypothesis that aging, and a sedentary lifestyle exacerbate microglial alterations associated with variations in litter size.
Within the histological sections, we meticulously delineated the layers of the region of interest (dentate gyrus) by positioning counting probes and digitally capturing images using a low-resolution 4× objective on a NIKON Eclipse 80i microscope (Nikon, Japan) equipped with a motorized stage (MAC200, Ludl Electronic Products, Hawthorne, NY, USA). This setup interfaced with a computer running the StereoInvestigator software (MicroBrightField, Williston, VT, USA), facilitating the storage and analysis of digitized x, y, and z coordinates of points.
To ensure accurate detection of microglia using the dissector probe, we replaced the low-resolution objective with a high-resolution 100× oil immersion plan fluoride objective (Nikon, NA 1.3, DF = 0.19µm). This adjustment enabled unambiguous identification and counting of microglia.
The thickness of each section was meticulously assessed at every counting site using the high-resolution objective, enabling precise delineation of the immediate layers at the section's top and bottom. Considering the variability in thickness and cell distribution across sections, the total count of objects of interest was adjusted based on section thickness. All microglial cell bodies clearly visible within the counting frame were meticulously counted and added to the total marker count. This encompassed cell bodies entirely within the counting frame or intersecting the acceptance line without contacting the rejection line, adhering to established criteria elsewhere [
55]. Counting boxes were consistently positioned within a grid in a randomized yet systematic manner to ensure comprehensive coverage and unbiased sampling.
2.7. Volume Estimations of Dentate Gyrus Using Planimetric Techniques
Planimetric assessments were performed utilizing the Stereo Investigator software to gauge the volumes of the unilateral dentate gyrus across all experimental conditions. Leveraging the optical fractionator method as the foundational approach for volume calculation, the software ensured consistent section-to-section distances throughout the sequence.
Area estimates from multiple sections were amalgamated to derive a comprehensive estimate of the total volume, while also computing the coefficient of error (CE). This methodology proved crucial in estimating volumes based on the actual thickness of histologically prepared sections. Given that shrinkage induced by these processes typically exhibits nonlinearity and predominantly affects the z-axis due to dehydration, all volume estimations were conducted without adjustments for shrinkage.
2.8. Photomicrography and Processing
Digital photomicrographs were acquired using a digital camera (MicroFire, Optronics, Goleta, CA, USA) connected to a NIKON Eclipse 80i microscope. Adobe Photoshop software was employed for post-capture processing, encompassing adjustments such as scaling and fine-tuning brightness and contrast levels across the entire image.
From the array of micrographs obtained, specific sections from each experimental group were meticulously selected. These selections were based on their proximity to the mean value of microglia number within the region of interest, ensuring that the chosen micrographs offered a representative depiction.
2.9. Statistical Analyses
Data are presented as the mean ± standard error of the mean.
Supplementary Tables S2–S6 provide detailed summaries of experimental parameters and average counting results obtained from the optical fractionator. Our grid size was meticulously tailored to achieve an acceptable coefficient of error (CE). To evaluate the CE of total microglial counts per rat, we employed the one-stage systematic sampling procedure (Scheaffer CE), a method utilized in previous studies [
56].
The acceptable level of CE for stereological estimations was determined as the ratio between the inherent error introduced by the methodology and the coefficient of variation (CV) [
56,
57]. For this study, a CE ≤ 0.05 was considered appropriate, as the variance introduced by the estimation procedure minimally contributed to the observed group variance [
56]. While the ratio between CE2/CV2 ideally should not exceed 0.5, certain exceptions were identified in our investigation. In these cases, despite higher CE2/CV2 values than recommended, the biological variance and methodology-induced CE were exceedingly low, rendering strict adherence to this rule neither meaningful nor practical [
58].
A multifactorial analysis of variance (ANOVA) was performed using ezANOVA, a freely available statistical software, to compare stereological estimations among all groups. This was applied as Design 3 Between Subject Factors, followed by pairwise comparisons employing Tukey's honestly significant difference test (HSD). This test aims to control for multiple comparisons while expressing a standardized Q score. Significance levels were set at p < 0.05 to determine statistical significance across comparisons.
3. Results
3.1. Body Weights and Dentate Gyrus Volumes
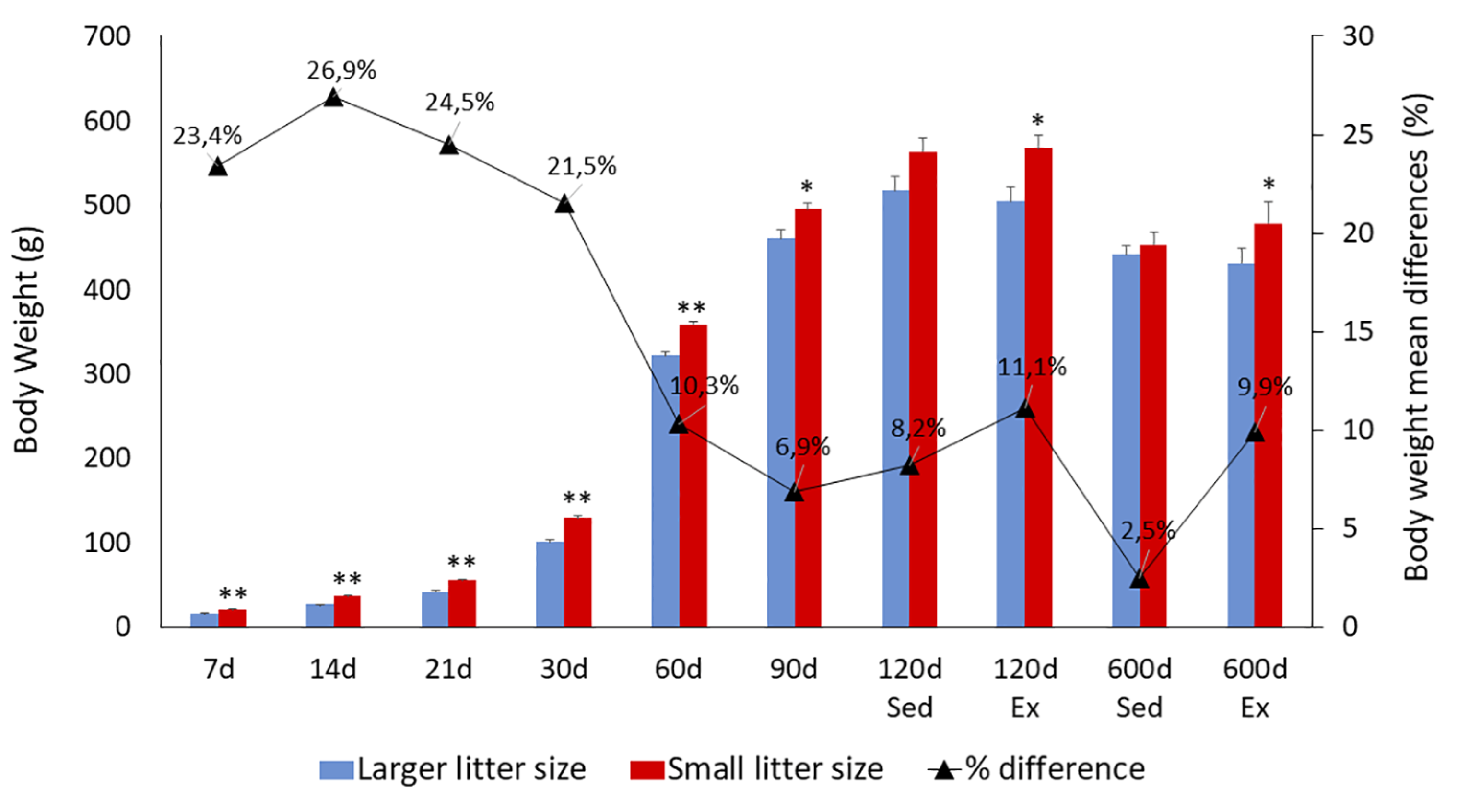
Figure 3 provides a comprehensive overview of mean body weight values alongside their respective standard errors across multiple postnatal days (7, 14, 21, 30, 60, 90, 120, and 600). A progressive and significant increase in body weight, irrespective of litter size, was observed until reaching maturity, followed by a decline during aging. Notably, animals from larger litters exhibited significantly lower mean body weights than those from smaller litters between the 7th and 90th postnatal days. However, beyond this period, the mean body weights of rats from both large and small litters became comparable during both mature (120 days) and aged (600 days) stages, particularly among sedentary groups. Interestingly, exercised rats from smaller litters displayed higher body weights during these mature and aged periods when compared to exercised rats from larger litters.
Regarding dentate gyrus volumes, while mature exercised rats from smaller litters showcased higher values, and aged sedentary rats from larger litters exhibited smaller volumes on average, no statistically significant differences were detected among the experimental groups. An exception was noted in aged rats from smaller litters raised sedentarily, which displayed a significant reduction compared to age-matched exercised animals raised in similar-sized litters (F1,32 = 6.16; p < 0.018, three-way ANOVA, pairwise comparisons [Q = Tukey HSD: t(8) = 2.40, p < 0.0434]).
3.2. Impact of Litter Size on Maternal Care
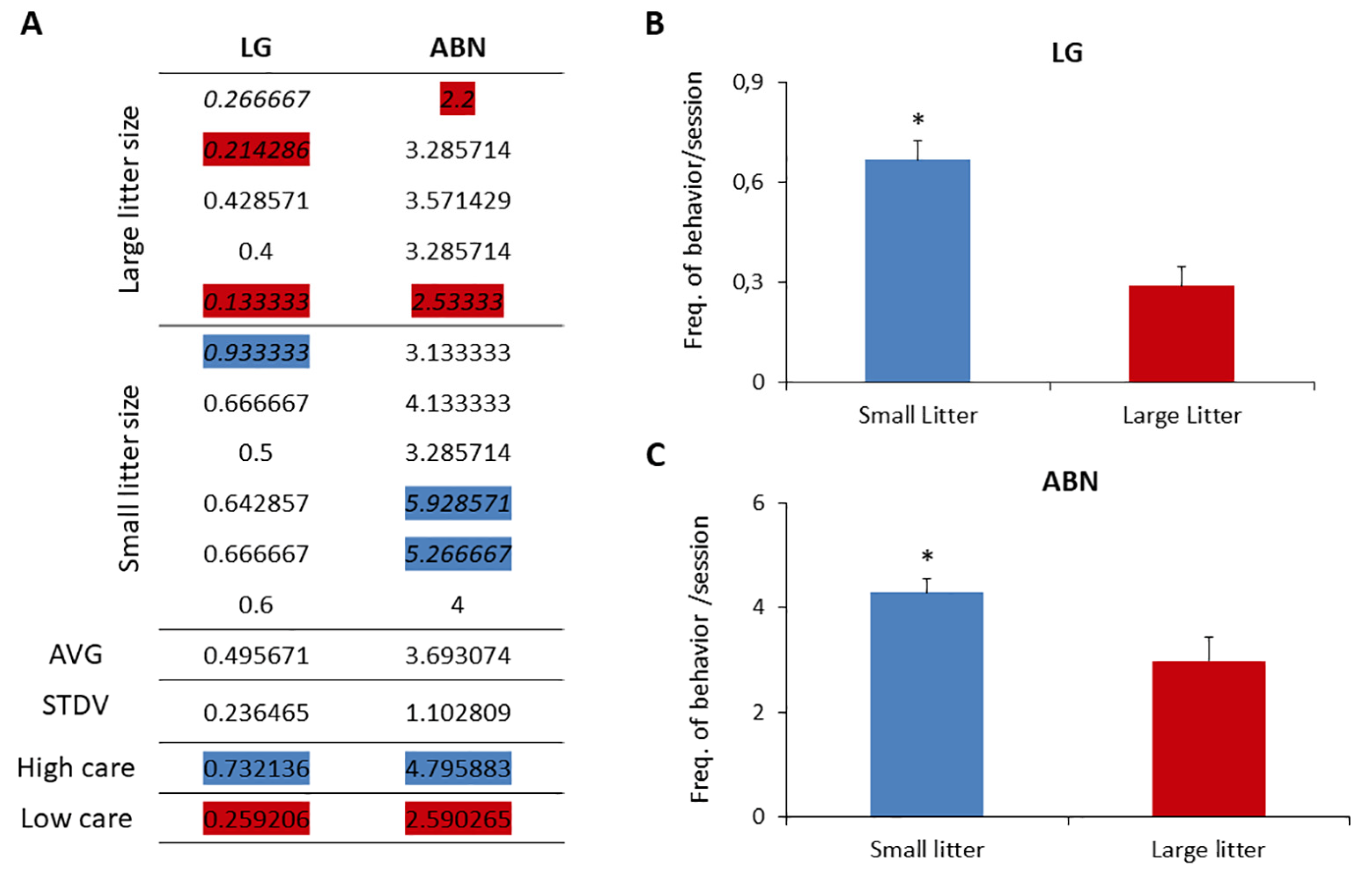
Observations of dams tending to large and small litters unveiled a distinct variance in maternal care, contingent upon litter size. Our investigation primarily focused on behaviors crucially identified by Meaney and colleagues as discriminators between high-care and low-care mothers, specifically, licking/grooming behavior (LG) and arched-back nursing behavior (ABN) [
59].
Figure 4 illustrates the average occurrences of LG and ABN behaviors per session per dam, complemented by the population mean and standard deviation. Mothers exhibiting high and low care were categorized based on LG and ABN scores ±1 standard deviation above (high) or below (low) the mean for the entire group. The distribution of licking behavior across the cohort displayed a normal pattern, with dams classified as high and low caregivers predominantly belonging to the small and large litter groups, respectively.
Statistical analysis revealed a significant difference in the averages of LG and ABN behaviors between large litters (0.28 ± 0.12 LG and 2.97 ± 0.57 ABN) and small litters (0.66 ± 0.14 LG and 4.29 ± 1.1 ABN) (t-test, p < 0.05). Additionally, the frequency of pups being out of the nest was notably higher in large litters compared to small litters.
3.3. Behavioral Assays
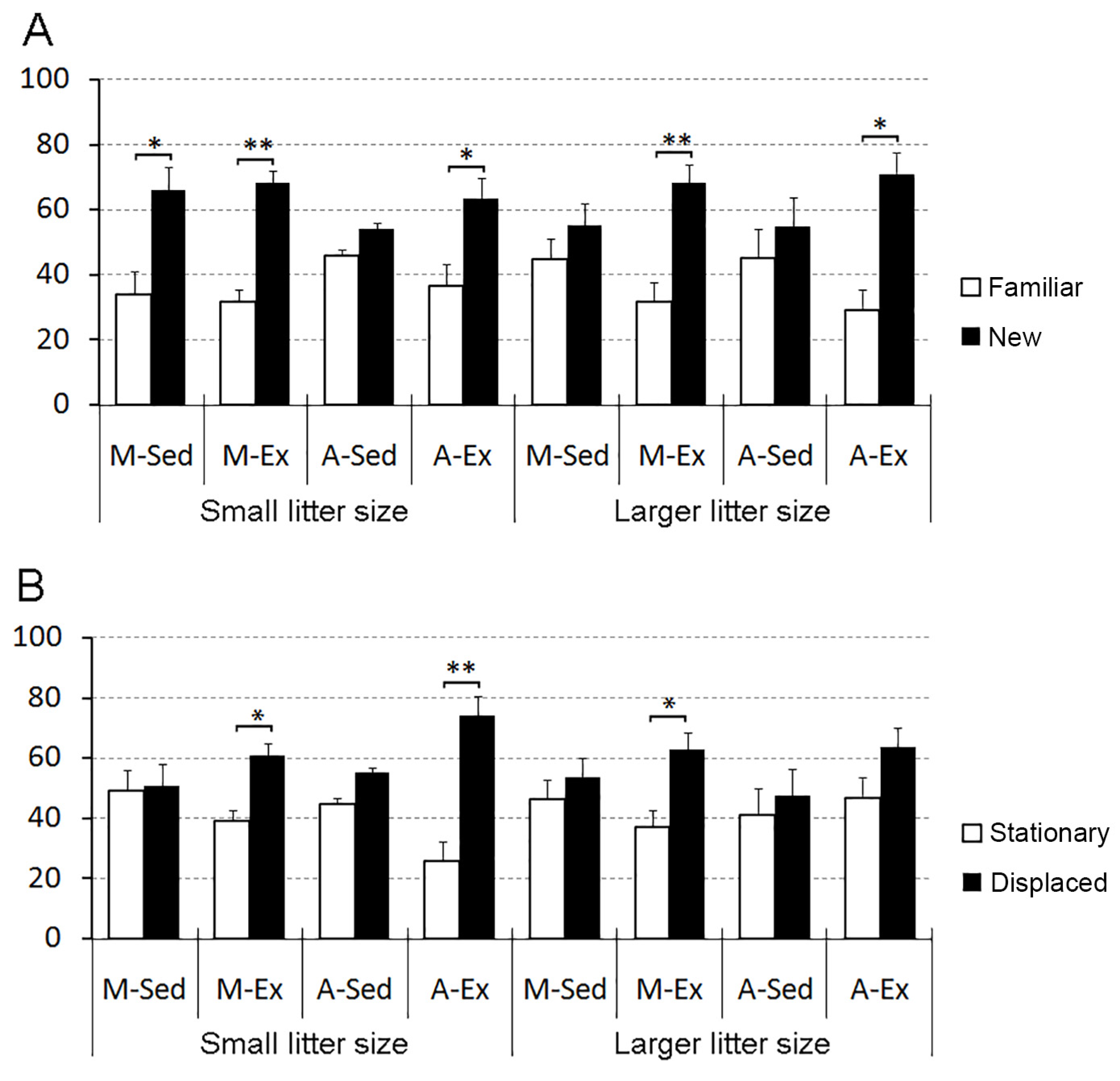
Figure 5 displays the outcomes of the object recognition and spatial memory tests.
Our findings underscore the detrimental impact of a sedentary lifestyle on spatial memory, specifically in both young and aged rats, independent of litter size. However, the exercise proved effective in counteracting these effects among aged rats from smaller litters but did not yield the same benefits in those from larger litters. Conversely, only sedentary-aged rats from both larger and smaller litters display impaired identity object recognition memory, a deficit that was ameliorated by exercise regardless of litter size.
In terms of one-trial object identity recognition, sedentary rats from larger litters, regardless of their age, faced challenges in object discrimination. Yet, exercise emerged as a significant contributor to enhancing their object discrimination abilities, underscoring its consistent impact in mitigating the repercussions of larger litter size, irrespective of the rats' age (
Figure 5A).
Concerning one-trial object placement recognition, all sedentary rats struggled to distinguish stationary from displaced objects regardless of litter size and age. Conversely, exercised rats from the mature and aged groups from small litters, as well as exercised mature (but not aged) animals, could differentiate between stationary and displaced objects (
Figure 5B).
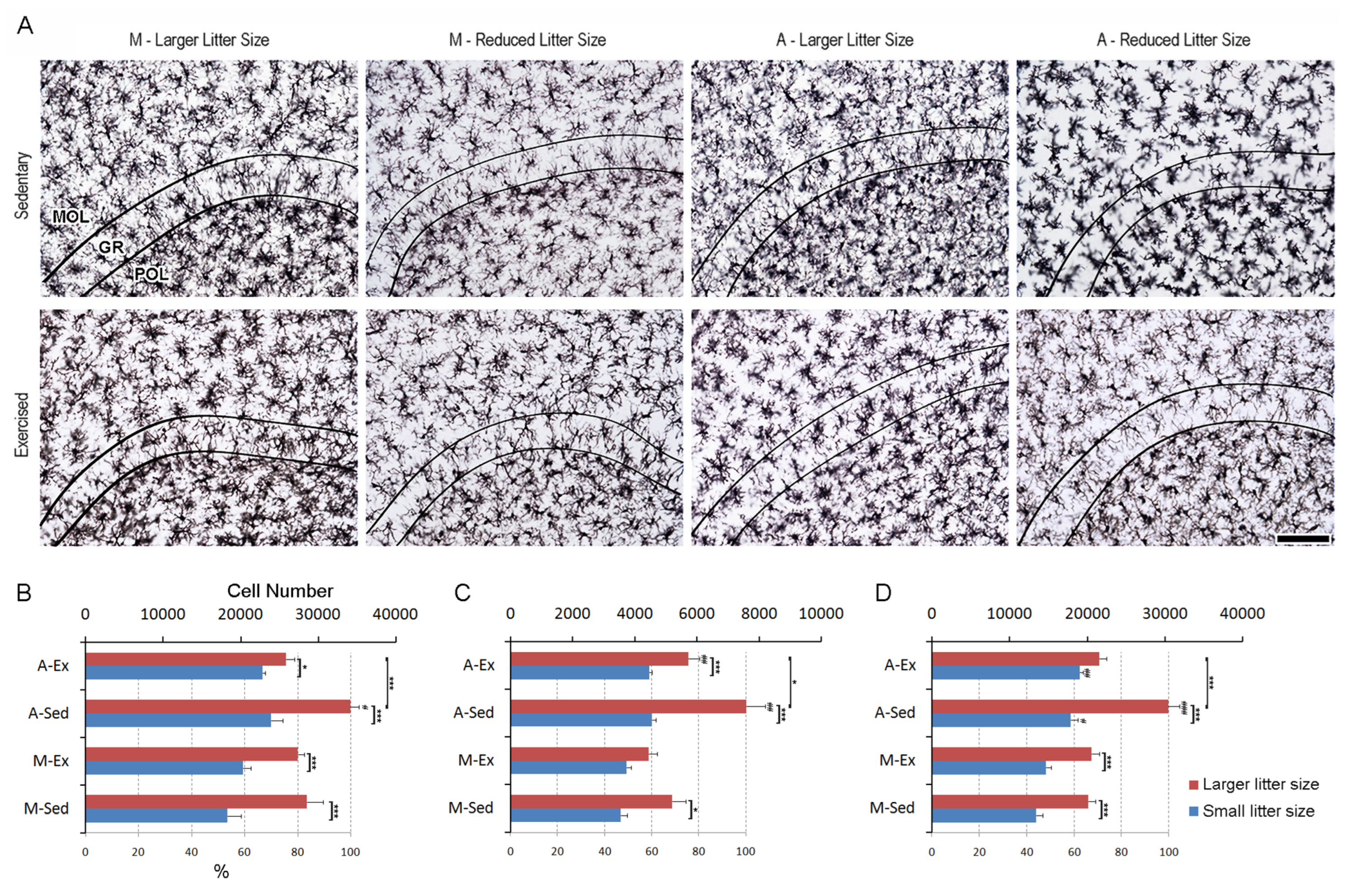
3.4. Enhanced Microglial Dynamics in the Dentate Gyrus: Insights from Litter Size, Aging, and Exercise
Figure 6A depicts IBA-1-immunolabeled sections of the dentate gyrus, illustrating the distinctive layers amidst varied experimental conditions. Within the molecular layer, a discernible increase in microglial density is evident among animals from larger litter. Notably, sedentary older individuals exhibit a significant increase in microglial presence compared to their younger counterparts. As hypothesized exercise interventions mitigate age-related microgliosis, suggesting a potential role of physical activity in attenuating heightened microglial activity associated with aging (see
Figure 6B).
In the granular layer, no notable elevation in microglial numbers is observed among exercised mature adults from larger litters. However, aging manifests in increased microglial counts compared to younger controls. Remarkably, exercise consistently diminishes microgliosis in aged animals (see
Figure 6C).
A. Photomicrographs of immunolabeled sections from mature (M) and aged (A) rats raised in small (6 pups/dam) or larger litters (12 pups/dam) subjected to either a short period of exercise (Ex) later in life or sedentary rearing (Sed). Images were chosen to represent rats with microglia counts close to the mean values of different experimental groups. Curved lines delineate the boundaries of the granular layer (GR), situated between the molecular (MOL) and polymorphic (POL) layers of the dentate gyrus. Graphical representation depicts mean values and standard error bars of unilateral dentate gyrus microglial counts in the molecular (B), granular (C), and polymorphic (D) layers. (#) p<0.05 compared to Mature; (*) indicates p<0.05, (**)p<0.01, and (***)p<0.001, denoting varying levels of statistical significance in 3-way ANOVA.
Similarly, an increase in microglial numbers is observed across all large litter groups in the granular layer. However, a noteworthy exception is noted in the exercised older group, where a reduction in this effect is evident. Additionally, older groups from smaller litters exhibit an increase in microglial numbers compared to younger groups, particularly in this layer (see
Figure 6D). Detailed stereological data can be found in the
supplementary material (Tables S1–S6).
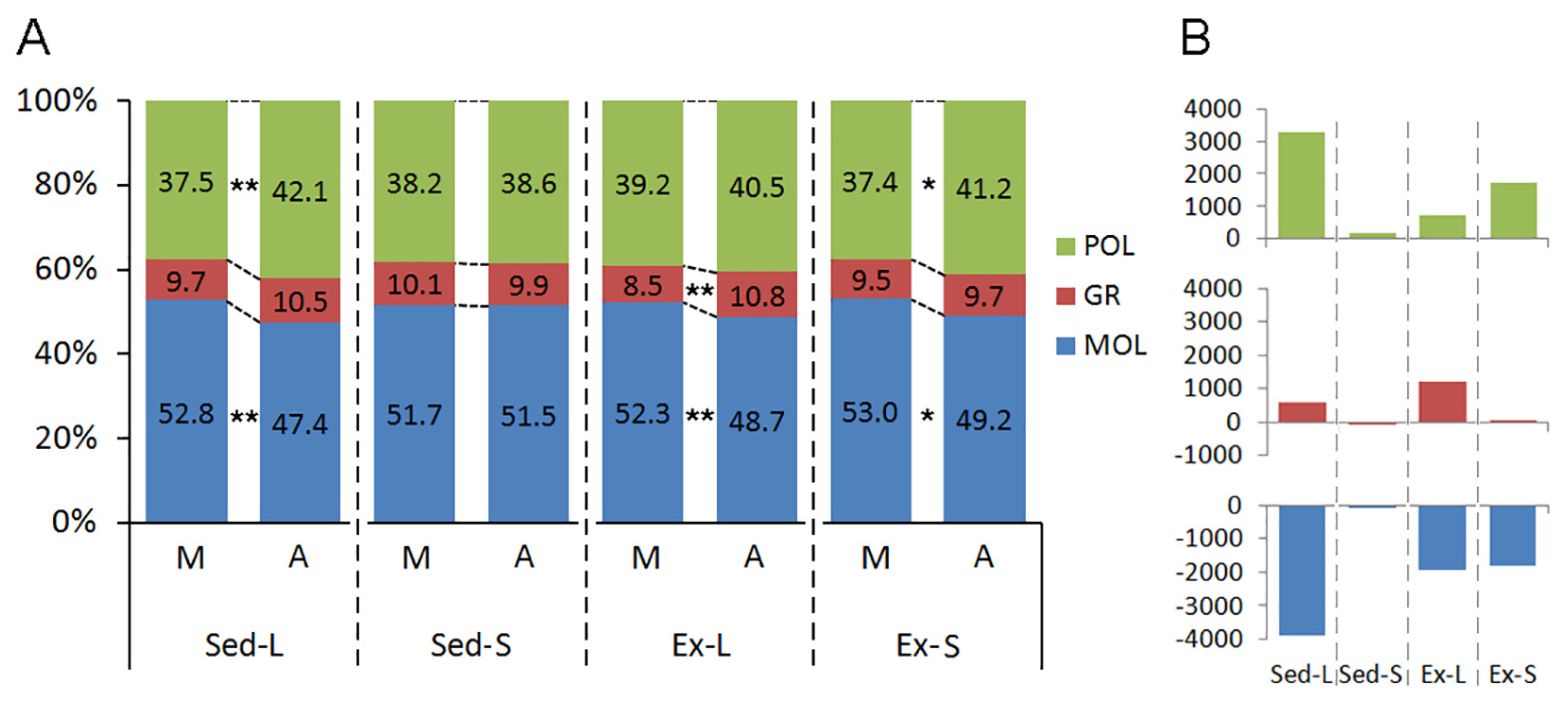
Figure 7 delineates the impact of litter size, aging, and exercise on the laminar distribution of microglia within the dentate gyrus of both mature and aged rats. Notable laminar redistributions of microglia are associated primarily with litter size and aging, while exercise interventions appear to mitigate these laminar alterations.
Aging does not alter the laminar distribution of microglia in the Sed-S groups. However, perinatal changes linked to increased litter size led to a significant shift in cell distribution, particularly elevating the proportion of cells in the granular layer compared to the molecular layer (Sed-L). Exercise interventions further accentuate this shift by increasing the proportion of microglia in the granular layer (Ex-L). Furthermore, albeit to a lesser extent, aging within the Ex-S groups also exhibits a higher proportion of microglia in the polymorphic layer compared to the molecular layer.
It's important to highlight that while these findings illuminate intriguing dynamics of microglial populations, no direct correlations were identified between the quantity of microglia and behavioral performance.
4. Discussion
We investigated the effects of litter size and late-life treadmill exercise on the laminar distribution of microglia in the dentate gyrus of mature and aged rats. Our findings revealed that exercise and a decrease in litter size reduced the number of microglia. Notably, this reduction correlated with preserving spatial and object recognition memories. Interestingly, compared to exercised animals of the same age from small litters, sedentary rats from larger litters exhibited a subtle yet enduring decline in dentate gyrus volume.
Litter Size, Growth, and Somatic Maturation
Our experimental approach drew inspiration from the protocol outlined elsewhere [
60], with a modification to limit the number of pups within the larger litters to 12 per dam, aiming to prevent undernourishment. In our model, pups from different dams were combined and redistributed among the dams, resulting in varying ratios of pups per dam. Specifically, the large litter group maintained a pup-to-dam ratio of 12:1, while the small litter group had a ratio of 6:1.
This experimental manipulation was predicated on the assumption that a greater ratio of pups to dam would result in fewer suckling per pup, a pattern observed in earlier studies. This anticipated reduction in nursing interactions directly influences maternal care behaviors [
24,
61]
The alterations observed in maternal care and the presumed differences in milk demand affecting the number of breastfeeding per pup align with the body weight curve findings, revealing significant differences from postnatal days 7 through 90. However, it is crucial to note a prior study [
62] that identified three classes of natural litter sizes in Wistar rats: they found minor distinctions in pup growth and somatic maturation within these classes: Class 1 (6, 7, and 8 pups), Class 2 (9 and 10 pups), and Class 3 (11 and 12 pups). Our experimental design pertained to classes 1 (six pups/dam) and 3 (12 pups/dam).
Moreover, existing literature has demonstrated that the typical litter size for Wistar rats ranges from 1 to 13 pups, and no undernutrition occurs with a ratio of 6 or 12 pups per dam during the weaning period [
62,
63]. Therefore, it is plausible to hypothesize that the observed behavioral and microglial alterations were triggered by changes associated with modified maternal care rather than variations in the nutritional status of the subjects.
Litter Size and Microglial Response
The impact of litter size on the microglial response has captivated research interest, particularly within investigations into the epigenetic effects of maternal behavior during early-life experiences. Pioneering studies by Michael Meaney and colleagues underscored how maternal behavior—specifically, grooming and licking—profoundly influences genes governing the hypothalamic–pituitary–adrenal axis [
64]. Their research illuminated the far-reaching epigenetic effects of early maternal care on these genetic mechanisms. Considering the pivotal role of glucocorticoids in shaping microglial function across the lifespan [
65,
66] and prior indications linking litter size with corticosterone levels [
67], it is reasonable to anticipate that changes in microglial responses due to variations in litter size may be associated to epigenetic effects of altered maternal care. Substantiating this expectation, studies examining optical density analysis of IBA-1 immunoreactivity in the dentate gyrus uncovered significant differences between adult and aged groups subjected to chronic restraint stress compared to control groups. These differences were associated with heightened corticosterone levels in stressed adult and aged groups [
68]. This suggests that diminished maternal care during early life might disrupt the innate immune response in adulthood, potentially leaving lasting imprints on the offspring’s immune system [
69]. Further studies including analysis of DNA methylation and glucocorticoid levels with microglial profile analysis may shed light on the possible mechanisms involved on the effects here observed, and possibly confirm these speculations.
In our study, manipulating the pup-to-dam ratio resulted in discernible differences in maternal care status. Notably, pups in the high ratio group received less maternal grooming and licking than those in the low ratio group. The distribution of maternal behaviors followed a normal, non-bimodal pattern, with high and low-care mothers existing as extremes on a continuum, as observed previously [
14]. By categorizing females exhibiting licking behavior one standard deviation below or above the mean, we could compare groups that experienced a 2- to 3-fold difference in received maternal care without accounting for the division of care due to the number of puppies. Studies with varying litter sizes have indicated that larger litters consistently receive reduced individual care per pup [
70].
The litter size in early life has the potential to significantly impact the social interactions among offspring, potentially leading to enduring alterations in anxiety levels, responses to new experiences, and the ability to cope with stress in adulthood [
71]. However, the precise contribution of reduced maternal care versus interactions among offspring to cognitive and emotional changes later in life and their potential interaction remains to be precisely quantified.
However, information on the long-term consequences of litter size during brain development on microglia later in life has been lacking. Our findings demonstrate a lasting impact on microglial numbers in the dentate gyrus of young and aged rats from larger litters during the suckling period. A notable increase in microglial numbers was observed in sedentary aged rats raised in large litters, and it was suggested that ad libitum access to food and water combined with late-life exercise did not suffice to reverse these changes. In larger litters, brain development is associated with enduring alterations in the brain's innate immune system, significantly impacting microglial homeostasis in aged rats. Whether a direct correlation exists between litter size, age-related plasticity in the dentate gyrus, and changes in microglial numbers remains to be established.
Moreover, we observed a laminar redistribution of microglia associated with litter size and aging, apparently mitigated by exercise. It is well known that the laminar organization of dentate gyrus plays a crucial role in processing and integrating incoming neural signals. Microglia, in this context, are strategically distributed across these layers, suggesting a potential role in modulating synaptic transmission and plasticity [
72]. Changes on the laminar distribution of microglia may suggest a spatially specific response to various stimuli, which could influence synaptic plasticity and cognitive functions differently across layers [
73].
The dentate gyrus is also known for its role in adult neurogenesis and synaptic plasticity, both of which are fundamental for learning and memory processes and known to be influenced by exercise and early environmental conditions. It has been demonstrated that cellular differentiation and the subregional distribution of microglia follow specific developmental gradients of the different parts of Ammon's horn and the dentate gyrus [
74]. As microglia have been implicated in regulating these processes through the secretion of various factors that modulate neuronal activity and synaptic connectivity, it is reasonable to consider that changes in its laminar distribution may contribute to the regional specificity of neurogenesis and synaptic remodeling within the dentate gyrus, thus influencing cognitive functions. Therefore, as mentioned before, a thorough characterization of microglial profiles within such distribution would be necessary to assume if the laminar reorganization we observed consists of a protective or detrimental effect mediated by microglia.
Litter Size, Aging, and Cognitive Decline
Under normal conditions, microglia in various CNS regions exhibit distinct branching and ramified structures, setting them apart from tissue macrophages [
75]. Despite the absence of neurological diseases, aging prompts a shift towards more reactive forms of astrocytes and microglia, contributing to an increased and sustained pro-inflammatory profile [
76,
77]. Age-related changes in microglia involve altered cytokine production [
78], modified expression of activation markers [
79], and morphological changes characterized by dystrophy [
80]. Furthermore, during aging, the bidirectional communication between neurons and microglia may become disrupted, leading to the loss of neuronal-derived factors that typically regulate microglial activation [
81].
It has been reported that while a significant rise in pro-inflammatory microglial profiles is observed in the hippocampus and dentate gyrus during aging, this does not necessarily correlate directly with cognitive decline [
82]. Additionally, a higher number of these profiles is identified in the dentate gyrus of sedentary animals [
26,
83], making it challenging to link microglial changes with memory impairments directly. However, exercise appears to curb microglial proliferation and enhance the expression of a pro neurogenic phenotype specifically in the hippocampus and dentate gyrus [
26,
83].
In alignment with these findings, our study did not detect cognitive impairments in the exercised group, except in aged animals from larger litters, where exercise failed to mitigate spatial memory decline. Notably, this group displayed a higher number of microglia in all layers of the dentate gyrus compared to age-matched exercised rats. Our observations suggest a potential presence of two distinct microglial phenotypes (pro-neurogenic and pro-inflammatory) within the dentate gyrus of Wistar rats. Furthermore, it is plausible that exercised rats primarily exhibit the dominant effects of the pro-neurogenic phenotype, as previously described in mice (Kohman et al. 2012; Mela et al. 2020). Nevertheless, it is important to note that in our work we have assessed the total number and laminar distribution of microglial cells in DG of the experimental subjects, with no further assessment of the molecular profile of these cells, therefore we can only suppose based on the abovementioned observations that the changes here reported may be related to a rise in pro-inflammatory profiles induced by aging, reduced maternal care and sedentary lifestyle, reflected into functional changes in cognitive functions. However, if the increased numbers of microglia in fact translate into increased inflammation pattern or reflect a protective mechanism to detrimental environmental conditions, is a question that demands further investigation.
Technical Limitations
Estimating object numbers in histological sections using stereological methods often exhibit variability across studies due to distinct estimation techniques, animal lineages, histological procedures, stereological protocols, and even uncertainties in defining objects and areas of interest [
84]. We standardized our procedures across all samples to mitigate potential errors when comparing animal groups. We maintained uniformity by employing identical protocols for sample processing, data collection, and analysis through consistent use of stereological methods, software, and hardware.
Furthermore, we implemented rigorous verification procedures to ascertain consistency in identifying the objects of interest. Multiple investigators independently counted the same regions using the identical anti-Iba1 antibody as a microglial marker. This meticulous approach aimed to minimize potential variations stemming from non-biological sources, ensuring reliability in our data interpretation.
Additionally, it is worth noting that corticosteroids, which can inhibit microglial activation, can influence microglial plasticity [
85]. During our experiments, treadmill exercise may have induced stress, potentially altering plasma corticosteroid levels and consequently impacting microglial numbers. Although we did not measure plasma corticosteroid levels post-exercise, it is a factor that warrants consideration as it might contribute to the observations made in our study.
Moreover, the behavioral testing protocol may have induced a bias in the cognitive results. Pre-exposure of animals to cognitive tests when mature followed by re-exposure to the same test when aged can potentially influence the results due to several behavioral factors, such as habituation effects, that may enhance performance during retesting, confounding interpretations of age-related cognitive changes. Nevertheless, the fact that all experimental groups underwent the same protocols under the same conditions, these effects may not have influenced the general outcomes of this study. Additionally, limiting the analysis to a subset of cognitive parameters, the study may not capture the full spectrum of cognitive changes induced by the experimental conditions.
The absence of sex comparisons represents another limitation of the study. Sex differences in cognitive function have been widely documented in both human and animal research, with evidence suggesting that males and females may exhibit distinct patterns of cognitive performance and response to environmental factors. By exclusively using male rats as experimental subjects, the study overlooks potential sex-specific effects that could contribute to variations in cognitive outcomes. Incorporating both male and female subjects and conducting sex-specific analyses would allow for a more nuanced understanding of how maternal care, aging, and exercise interact with biological sex to influence cognitive function. Additionally, such analyses could shed light on potential sex-specific vulnerabilities or protective factors that may have relevance for translational research in humans.
5. Conclusions
This study explores the multifaceted influence of litter size, maternal care, exercise, and aging on neurobehavioral plasticity and dentate gyrus microglia dynamics in rats. Body weight evolution revealed a progressive increase until maturity, followed by a decline during aging, with larger litter rats exhibiting lower weights initially. Notably, exercised rats from smaller litters displayed higher body weights during mature and aged stages. Dentate gyrus volumes showed no significant differences among groups, except for aged sedentary rats from smaller litters, which exhibited a reduction. Maternal care varied significantly based on litter size, with large litter dams showing lower frequencies of caregiving behaviors. Behavioral assays highlighted the detrimental impact of a sedentary lifestyle on spatial memory, mitigated by exercise in aged rats from smaller litters. Microglial dynamics in the dentate gyrus revealed age-related changes, modulated by litter size and exercise. Exercise interventions mitigated microgliosis associated with aging, particularly in aged rats. These findings underscore the complex interplay between early-life experiences, exercise, and microglial dynamics in neurobehavioral outcomes during aging.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org.
Author Contributions
CWPD, RCAG, and AP conceptualized and designed the study. LVK, CML, CRF, MAO, RPB, TTC, INFA, DGD, JBT, MBO, AACL, RFMS, RAG, AAS, and DSL conducted most of the laboratory work and performed data analysis. LVK conducted stereological measurements and wrote the first draft. JBT conducted statistical analyses and prepared the figures. CWPD, RCAG, JBT, and LVK contributed to the manuscript's final edition. All authors critically reviewed and approved the final version of the manuscript.
Acknowledgments
This study received financial support from Brazilian Government research funds. Specifically, grants were provided by the Brazilian Research Council (CNPq), with grant numbers 300203/2010-1 and 556740/2009-2 awarded to CWPD. Additional support was received from Financiadora de Estudos e Projetos (FINEP) and the Instituto Brasileiro de Neurociências (IBNnet). CWPD acknowledges support from UFPA/PROPESP-PAPQ.
Competing Interests
The authors declare that they have no competing interests.
References
- J. P. Curley, F. A. Champagne, Influence of maternal care on the developing brain: Mechanisms, temporal dynamics and sensitive periods. Front Neuroendocrinol 40, 52-66 (2016). [CrossRef]
- M. Nishi, Effects of Early-Life Stress on the Brain and Behaviors: Implications of Early Maternal Separation in Rodents. Int J Mol Sci 21, (2020). [CrossRef]
- M. Čater, G. Majdič, How early maternal deprivation changes the brain and behavior? Eur J Neurosci 55, 2058-2075 (2022).
- A. Hegde, R. Mitra, Environment and early life: Decisive factors for stress-resilience and vulnerability. International review of neurobiology 150, 155-185 (2020).
- M. Kundakovic, F. A. Champagne, Early-life experience, epigenetics, and the developing brain. Neuropsychopharmacology 40, 141-153 (2015).
- B. Seay, E. Hansen, H. F. Harlow, Mother-infant separation in monkeys. Journal of Child Psychology and Psychiatry 3, 123-132 (1962).
- D. Suchecki, Maternal regulation of the infant's hypothalamic-pituitary-adrenal axis stress response: Seymour ‘Gig’Levine's legacy to neuroendocrinology. Journal of Neuroendocrinology 30, e12610 (2018).
- R. S. Eid et al., Early and late effects of maternal experience on hippocampal neurogenesis, microglia, and the circulating cytokine milieu. Neurobiology of aging 78, 1-17 (2019). [CrossRef]
- R. Hanamsagar, S. D. Bilbo, Environment matters: microglia function and dysfunction in a changing world. Current opinion in neurobiology 47, 146-155 (2017). [CrossRef]
- J. M. M. Costa, (2018).
- A. Roque, A. Ochoa-Zarzosa, L. Torner, Maternal separation activates microglial cells and induces an inflammatory response in the hippocampus of male rat pups, independently of hypothalamic and peripheral cytokine levels. Brain, behavior, and immunity 55, 39-48 (2016). [CrossRef]
- S. Bachiller, A. Paulus, S. Vázquez-Reyes, I. García-Domínguez, T. Deierborg, Maternal separation leads to regional hippocampal microglial activation and alters the behavior in the adolescence in a sex-specific manner. Brain, Behavior, & Immunity-Health 9, 100142 (2020). [CrossRef]
- V. Reshetnikov, Y. Ryabushkina, A. Kovner, A. Lepeshko, N. Bondar, Repeated and single maternal separation specifically alter microglial morphology in the prefrontal cortex and neurogenesis in the hippocampus of 15-day-old male mice. Neuroreport 31, 1256-1264 (2020). [CrossRef]
- J. P. Curley, F. A. Champagne, Influence of maternal care on the developing brain: Mechanisms, temporal dynamics and sensitive periods. Frontiers in neuroendocrinology 40, 52-66 (2016). [CrossRef]
- M. Banqueri, M. Méndez, E. Gómez-Lázaro, J. L. Arias, Early life stress by repeated maternal separation induces long-term neuroinflammatory response in glial cells of male rats. Stress 22, 563-570 (2019). [CrossRef]
- S. Nicolas et al., Prior maternal separation stress alters the dendritic complexity of new hippocampal neurons and neuroinflammation in response to an inflammatory stressor in juvenile female rats. Brain, Behavior, and Immunity 99, 327-338 (2022). [CrossRef]
- M. Hou et al., Neonatal overfeeding induced by small litter rearing causes altered glucocorticoid metabolism in rats. PloS one 6, e25726 (2011). [CrossRef]
- A. L. O. Carvalho, B. G. Ferri, F. A. L. de Sousa, F. C. Vilela, A. Giusti-Paiva, Early life overnutrition induced by litter size manipulation decreases social play behavior in adolescent male rats. International Journal of Developmental Neuroscience 53, 75-82 (2016). [CrossRef]
- A. L. Rodrigues, E. G. De Moura, M. C. Fonseca Passos, S. C. Potente Dutra, P. C. Lisboa, Postnatal early overnutrition changes the leptin signalling pathway in the hypothalamic–pituitary–thyroid axis of young and adult rats. The Journal of physiology 587, 2647-2661 (2009). [CrossRef]
- H. G. Rödel, S. Meyer, G. Prager, V. Stefanski, R. Hudson, Litter size is negatively correlated with corticosterone levels in weanling and juvenile laboratory rats. Physiology & behavior 99, 644-650 (2010). [CrossRef]
- E. Velkoska, T. J. Cole, R. G. Dean, L. M. Burrell, M. J. Morris, Early undernutrition leads to long-lasting reductions in body weight and adiposity whereas increased intake increases cardiac fibrosis in male rats. The Journal of nutrition 138, 1622-1627 (2008). [CrossRef]
- H. Davidowa, Y. Li, A. Plagemann, Hypothalamic ventromedial and arcuate neurons of normal and postnatally overnourished rats differ in their responses to melanin-concentrating hormone. Regulatory peptides 108, 103-111 (2002). [CrossRef]
- G. Prager, V. Stefanski, R. Hudson, H. G. Rödel, Family matters: maternal and litter-size effects on immune parameters in young laboratory rats. Brain, behavior, and immunity 24, 1371-1378 (2010). [CrossRef]
- S. Enes-Marques, A. Giusti-Paiva, Litter size reduction accentuates maternal care and alters behavioral and physiological phenotypes in rat adult offspring. The journal of physiological sciences 68, 789-798 (2018). [CrossRef]
- E. Cortés-Barberena, H. González-Márquez, J. Gómez-Olivares, R. Ortiz-Muñiz, Effects of moderate and severe malnutrition in rats on splenic T lymphocyte subsets and activation assessed by flow cytometry. Clinical & Experimental Immunology 152, 585-592 (2008).
- R. A. Kohman, E. K. DeYoung, T. K. Bhattacharya, L. N. Peterson, J. S. Rhodes, Wheel running attenuates microglia proliferation and increases expression of a proneurogenic phenotype in the hippocampus of aged mice. Brain, behavior, and immunity 26, 803-810 (2012). [CrossRef]
- R. Hudson, B. Maqueda, J. V. Moctezuma, A. M. Miranda, H. G. Rödel, Individual differences in testosterone and corticosterone levels in relation to early postnatal development in the rabbit Oryctolagus cuniculus. Physiology & behavior 103, 336-341 (2011). [CrossRef]
- I. Ziko et al., Neonatal overfeeding alters hypothalamic microglial profiles and central responses to immune challenge long-term. Brain, behavior, and immunity 41, 32-43 (2014). [CrossRef]
- S. N. De Luca et al., Neonatal overfeeding by small-litter rearing sensitises hippocampal microglial responses to immune challenge: Reversal with neonatal repeated injections of saline or minocycline. Journal of Neuroendocrinology 29, e12540 (2017). [CrossRef]
- S. Tapia-González et al., Activation of microglia in specific hypothalamic nuclei and the cerebellum of adult rats exposed to neonatal overnutrition. Journal of Neuroendocrinology 23, 365-370 (2011). [CrossRef]
- L. C. Viana et al., Litter size, age-related memory impairments, and microglial changes in rat dentate gyrus: stereological analysis and three dimensional morphometry. Neuroscience 238, 280-296 (2013). [CrossRef]
- T. Bredy, R. Humpartzoomian, D. Cain, M. Meaney, Partial reversal of the effect of maternal care on cognitive function through environmental enrichment. Neuroscience 118, 571-576 (2003). [CrossRef]
- T. W. Bredy, T. Y. Zhang, R. J. Grant, J. Diorio, M. J. Meaney, Peripubertal environmental enrichment reverses the effects of maternal care on hippocampal development and glutamate receptor subunit expression. European Journal of Neuroscience 20, 1355-1362 (2004). [CrossRef]
- L. Baroncelli et al., Nurturing brain plasticity: impact of environmental enrichment. Cell Death & Differentiation 17, 1092-1103 (2010). [CrossRef]
- K. Xiao et al., Beneficial effects of running exercise on hippocampal microglia and neuroinflammation in chronic unpredictable stress-induced depression model rats. Translational psychiatry 11, 461 (2021). [CrossRef]
- A. M. Littlefield, S. E. Setti, C. Priester, R. A. Kohman, Voluntary exercise attenuates LPS-induced reductions in neurogenesis and increases microglia expression of a proneurogenic phenotype in aged mice. Journal of neuroinflammation 12, 1-12 (2015). [CrossRef]
- L. Moita et al., Moderate physical training attenuates the effects of perinatal undernutrition on the morphometry of the splenic lymphoid follicles in endotoxemic adult rats. Neuroimmunomodulation 18, 103-110 (2010). [CrossRef]
- D. G. Diniz et al., Environmental impoverishment and aging alter object recognition, spatial learning, and dentate gyrus astrocytes. European Journal of Neuroscience 32, 509-519 (2010). [CrossRef]
- D. G. Diniz et al., Age, environment, object recognition and morphological diversity of GFAP-immunolabeled astrocytes. Behavioral and Brain Functions 12, (2016). [CrossRef]
- J. E. Jans, B. Woodside, Effects of litter age, litter size, and ambient temperature on the milk ejection reflex in lactating rats. Developmental Psychobiology: The Journal of the International Society for Developmental Psychobiology 20, 333-344 (1987).
- M. Morag, F. Popliker, R. Yagil, Effect of litter size on milk yield in the rat. Laboratory animals 9, 43-47 (1975). [CrossRef]
- R. Yagil, Z. Etzion, G. Berlyne, Changes in rat milk quantity and quality due to variations in litter size and high ambient temperature. Laboratory Animal Science 26, 33-37 (1976).
- C. Caldji et al., Maternal care during infancy regulates the development of neural systems mediating the expression of fearfulness in the rat. Proceedings of the National Academy of Sciences 95, 5335-5340 (1998). [CrossRef]
- N. Uriarte, M. K. Breigeiron, F. Benetti, X. F. Rosa, A. B. Lucion, Effects of maternal care on the development, emotionality, and reproductive functions in male and female rats. Developmental Psychobiology: The Journal of the International Society for Developmental Psychobiology 49, 451-462 (2007).
- E. Dere, J. P. Huston, M. A. D. S. Silva, The pharmacology, neuroanatomy and neurogenetics of one-trial object recognition in rodents. Neuroscience & Biobehavioral Reviews 31, 673-704 (2007). [CrossRef]
- E. Tulving, Episodic memory and common sense: how far apart? Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences 356, 1505-1515 (2001).
- A. Ennaceur, S. Michalikova, A. Bradford, S. Ahmed, Detailed analysis of the behavior of Lister and Wistar rats in anxiety, object recognition and object location tasks. Behavioural brain research 159, 247-266 (2005). [CrossRef]
- E. Dere, J. P. Huston, M. A. D. S. Silva, Episodic-like memory in mice: simultaneous assessment of object, place and temporal order memory. Brain research protocols 16, 10-19 (2005). [CrossRef]
- S. L. Dix, J. P. Aggleton, Extending the spontaneous preference test of recognition: evidence of object-location and object-context recognition. Behavioural brain research 99, 191-200 (1999). [CrossRef]
- S. Shu, G. Ju, L. Fan, The glucose oxidase-DAB-nickel method in peroxidase histochemistry of the nervous system. Neuroscience letters 85, 169-171 (1988). [CrossRef]
- C. B. Saper, P. E. Sawchenko, Magic peptides, magic antibodies: guidelines for appropriate controls for immunohistochemistry. J Comp Neurol 465, 161-163 (2003). [CrossRef]
- M. J. West, Design-based stereological methods for counting neurons. Progress in brain research 135, 43-51 (2002).
- M. J. West, Stereological methods for estimating the total number of neurons and synapses: issues of precision and bias. Trends in neurosciences 22, 51-61 (1999). [CrossRef]
- D. J. Bonthius et al., Use of frozen sections to determine neuronal number in the murine hippocampus and neocortex using the optical disector and optical fractionator. Brain Research Protocols 14, 45-57 (2004). [CrossRef]
- H. Gundersen, E. Jensen, The efficiency of systematic sampling in stereology and its prediction. J Microsc 147, 229–263 (1987). [CrossRef]
- E. M. Glaser, P. D. Wilson, The coefficient of error of optical fractionator population size estimates: a computer simulation comparing three estimators. Journal of Microscopy 192, 163-171 (1998). [CrossRef]
- L. Slomianka, M. J. West, Estimators of the precision of stereological estimates: an example based on the CA1 pyramidal cell layer of rats. Neuroscience 136, 757-767 (2005). [CrossRef]
- L. Slomianka, M. West, Estimators of the precision of stereological estimates: an example based on the CA1 pyramidal cell layer of rats. Neuroscience 136, 757–767 (2005). [CrossRef]
- M. J. Meaney, Maternal care, gene expression, and the transmission of individual differences in stress reactivity across generations. Annu Rev Neurosci 24, 1161-1192 (2001). [CrossRef]
- J. M. Celedon, M. Santander, M. Colombo, Long-term effects of early undernutrition and environmental stimulation on learning performance of adult rats. J Nutr 109, 1880-1886 (1979). [CrossRef]
- M. J. Meaney, Maternal care, gene expression, and the transmission of individual differences in stress reactivity across generations. Annual review of neuroscience 24, 1161-1192 (2001). [CrossRef]
- I. Chahoud, F. J. Paumgartten, Influence of litter size on the postnatal growth of rat pups: is there a rationale for litter-size standardization in toxicity studies? Environ Res 109, 1021-1027 (2009).
- L. J. Bulfin, M. A. Clarke, K. M. Buller, S. J. Spencer, Anxiety and hypothalamic-pituitary-adrenal axis responses to psychological stress are attenuated in male rats made lean by large litter rearing. Psychoneuroendocrinology 36, 1080-1091 (2011). [CrossRef]
- M. J. Meaney, M. Szyf, Environmental programming of stress responses through DNA methylation: life at the interface between a dynamic environment and a fixed genome. Dialogues Clin Neurosci 7, 103-123 (2005). [CrossRef]
- L. van Olst, P. Bielefeld, C. P. Fitzsimons, H. E. de Vries, M. Schouten, Glucocorticoid-mediated modulation of morphological changes associated with aging in microglia. Aging Cell 17, e12790 (2018). [CrossRef]
- R. M. Barrientos et al., Greater glucocorticoid receptor activation in hippocampus of aged rats sensitizes microglia. Neurobiol Aging 36, 1483-1495 (2015). [CrossRef]
- H. G. Rodel, S. Meyer, G. Prager, V. Stefanski, R. Hudson, Litter size is negatively correlated with corticosterone levels in weanling and juvenile laboratory rats. Physiol Behav 99, 644-650 (2010). [CrossRef]
- J. H. Park et al., Comparison of glucocorticoid receptor and ionized calcium-binding adapter molecule 1 immunoreactivity in the adult and aged gerbil hippocampus following repeated restraint stress. Neurochem Res 36, 1037-1045 (2011).
- R. S. Eid et al., Early and late effects of maternal experience on hippocampal neurogenesis, microglia, and the circulating cytokine milieu. Neurobiol Aging 78, 1-17 (2019). [CrossRef]
- E. Dimitsantos, R. M. Escorihuela, S. Fuentes, A. Armario, R. Nadal, Litter size affects emotionality in adult male rats. Physiology & Behavior 92, 708-716 (2007). [CrossRef]
- M. Calisir, O. Yilmaz, H. E. Kolatan, A. K. Sezgin, Effects of litter size and caging on physical and mental development in rats. Physiol Behav 267, 114200 (2023). [CrossRef]
- J. E. Collazos-Castro, M. Nieto-Sampedro, Developmental and reactive growth of dentate gyrus afferents: cellular and molecular interactions. Restorative neurology and neuroscience 19, 169-187 (2001).
- S. Jinno, F. Fleischer, S. Eckel, V. Schmidt, T. Kosaka, Spatial arrangement of microglia in the mouse hippocampus: a stereological study in comparison with astrocytes. Glia 55, 1334-1347 (2007). [CrossRef]
- I. Dalmau, B. Finsen, J. Zimmer, B. González, B. Castellano, Development of microglia in the postnatal rat hippocampus. Hippocampus 8, 458-474 (1998).
- R. M. Ransohoff, V. H. Perry, Microglial physiology: unique stimuli, specialized responses. Annu Rev Immunol 27, 119-145 (2009). [CrossRef]
- D. S. Davies, J. Ma, T. Jegathees, C. Goldsbury, Microglia show altered morphology and reduced arborization in human brain during aging and Alzheimer's disease. Brain Pathol 27, 795-808 (2017).
- J. P. Godbout, R. W. Johnson, Age and Neuroinflammation: A Lifetime of Psychoneuroimmune Consequences. Immunology and Allergy Clinics of North America 29, 321-+ (2009). [CrossRef]
- J. Costa et al., The old guard: Age-related changes in microglia and their consequences. Mech Ageing Dev 197, 111512 (2021). [CrossRef]
- J. C. Savage, M. Carrier, M. Tremblay, Morphology of Microglia Across Contexts of Health and Disease. Methods Mol Biol 2034, 13-26 (2019). [CrossRef]
- R. K. Shahidehpour et al., Dystrophic microglia are associated with neurodegenerative disease and not healthy aging in the human brain. Neurobiol Aging 99, 19-27 (2021). [CrossRef]
- H. A. Jurgens, R. W. Johnson, Dysregulated neuronal-microglial cross-talk during aging, stress and inflammation. Exp Neurol 233, 40-48 (2012).
- H. D. VanGuilder et al., Concurrent hippocampal induction of MHC II pathway components and glial activation with advanced aging is not correlated with cognitive impairment. J Neuroinflammation 8, 138 (2011). [CrossRef]
- V. Mela et al., Exercise-induced re-programming of age-related metabolic changes in microglia is accompanied by a reduction in senescent cells. Brain, behavior, and immunity 87, 413-428 (2020). [CrossRef]
- P. R. Mouton et al., Age and gender effects on microglia and astrocyte numbers in brains of mice. Brain Res 956, 30-35 (2002). [CrossRef]
- N. R. Nichols, Glial responses to steroids as markers of brain aging. J Neurobiol 40, 585-601 (1999).
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).