Submitted:
11 April 2024
Posted:
14 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods and Materials
- -
- Non-polar molecules such as the n-alkanes () such as n-pentane (), n-hexane (), n-heptane (), n-octane (), and n-nonane ()
- -
-
Polar molecules, divided into three groups:
- ➢
- Lewis’s acid solvents such as dichloromethane, chloroform, and carbon tetrachloride
- ➢
- Basic solvent such as ethyl acetate, diethyl ether, tetrahydrofuran (THF)
- ➢
- Amphoteric molecule such as toluene
3. Experimental Results
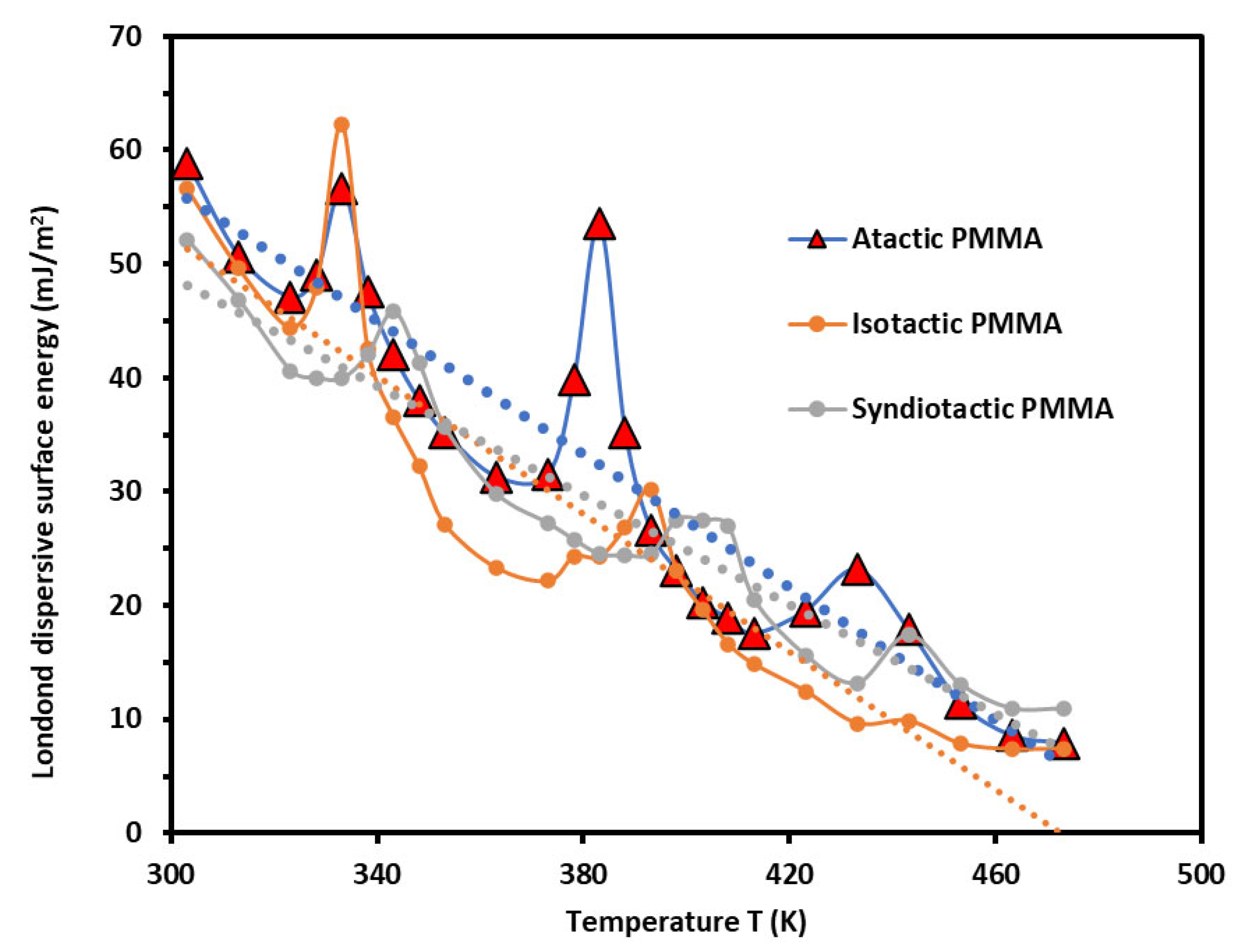
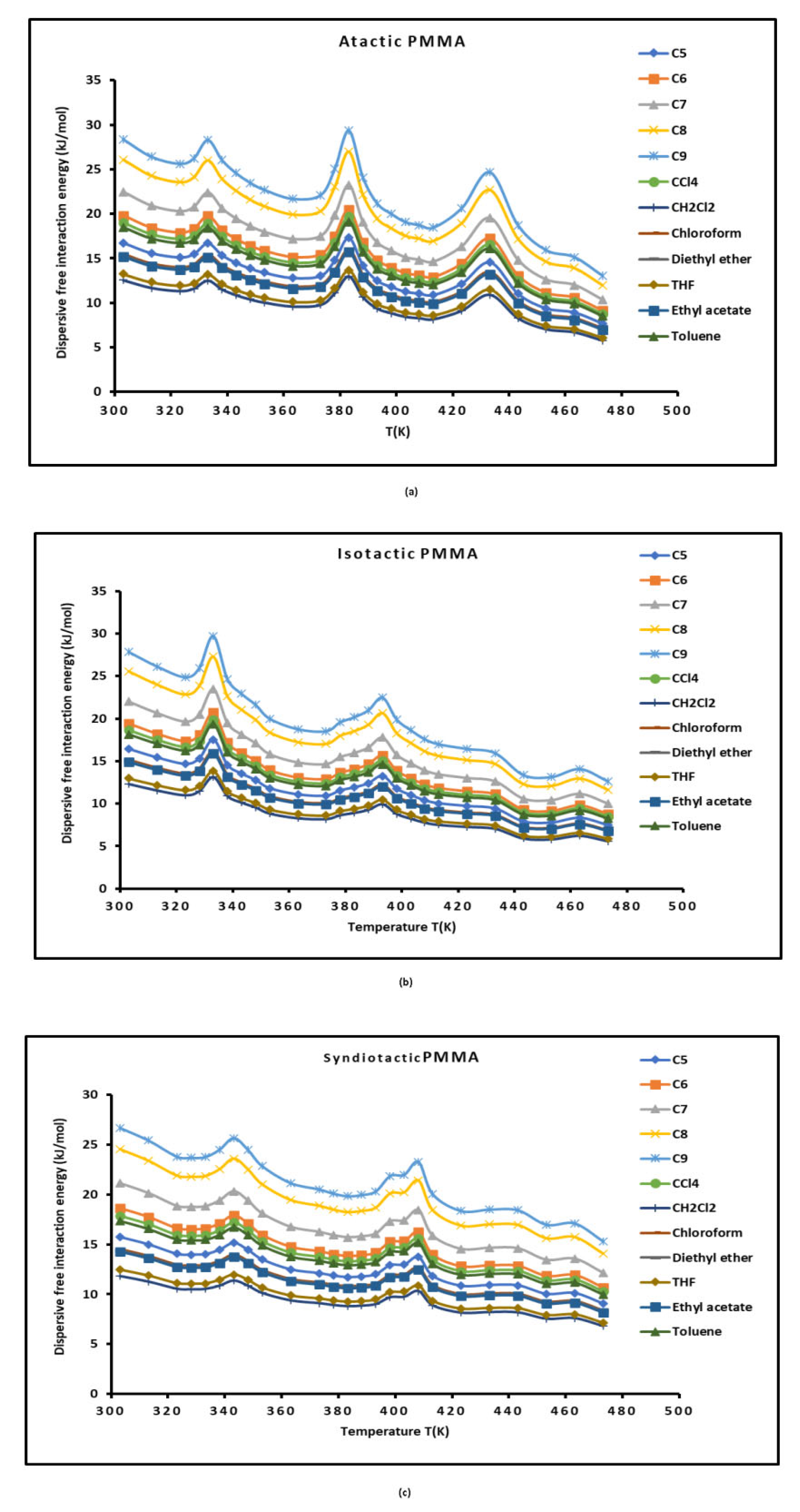
3.1. London Dispersive Component of Surface Energy of PMMA Polymers
- -
- The beta-relaxation ,
- -
- The glass transition ,
- -
- The liquid–liquid transitions .
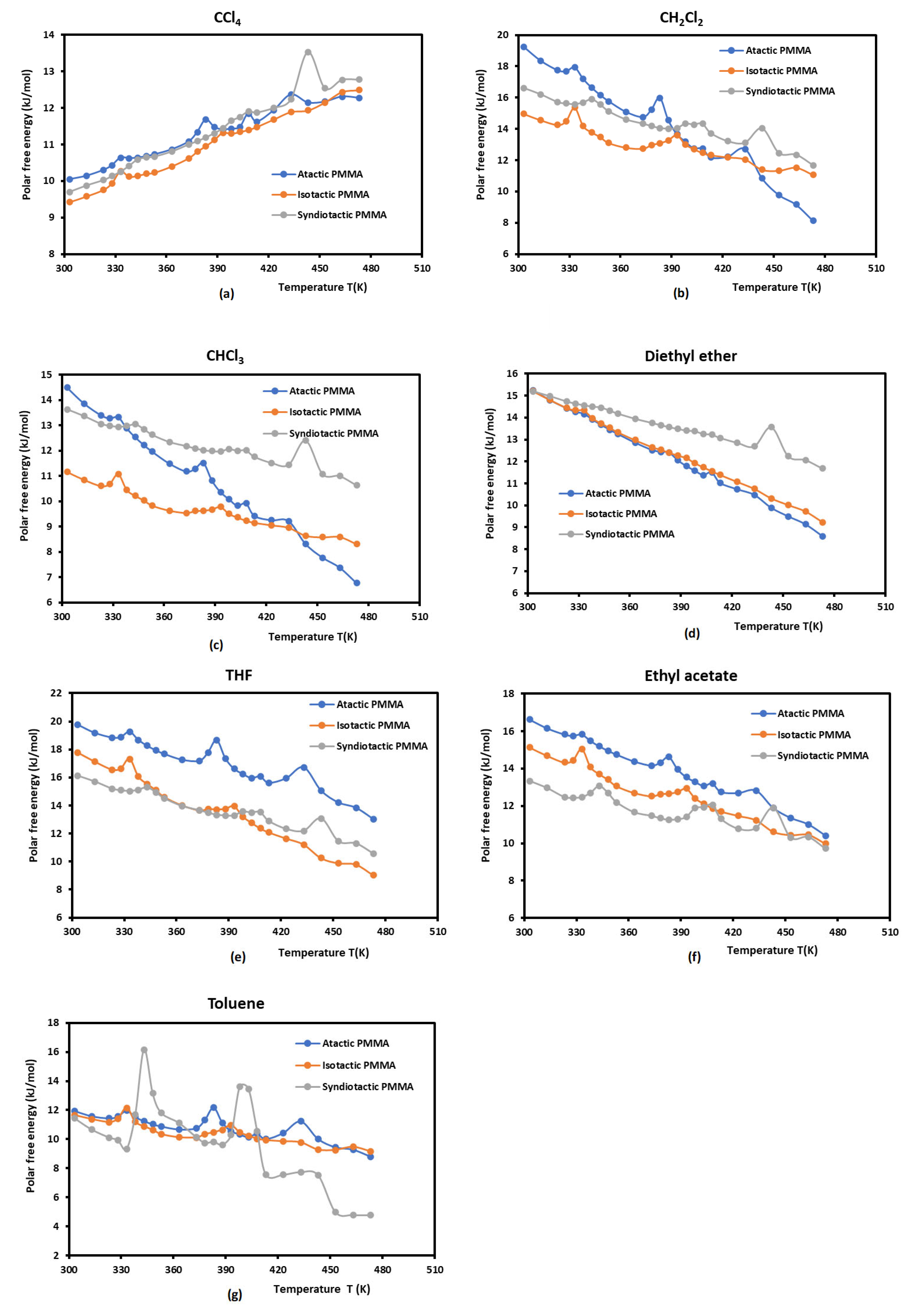
3.1. Polar Free Surface Energy of Atactic, Isotactic and Syndiotactic PMMAs
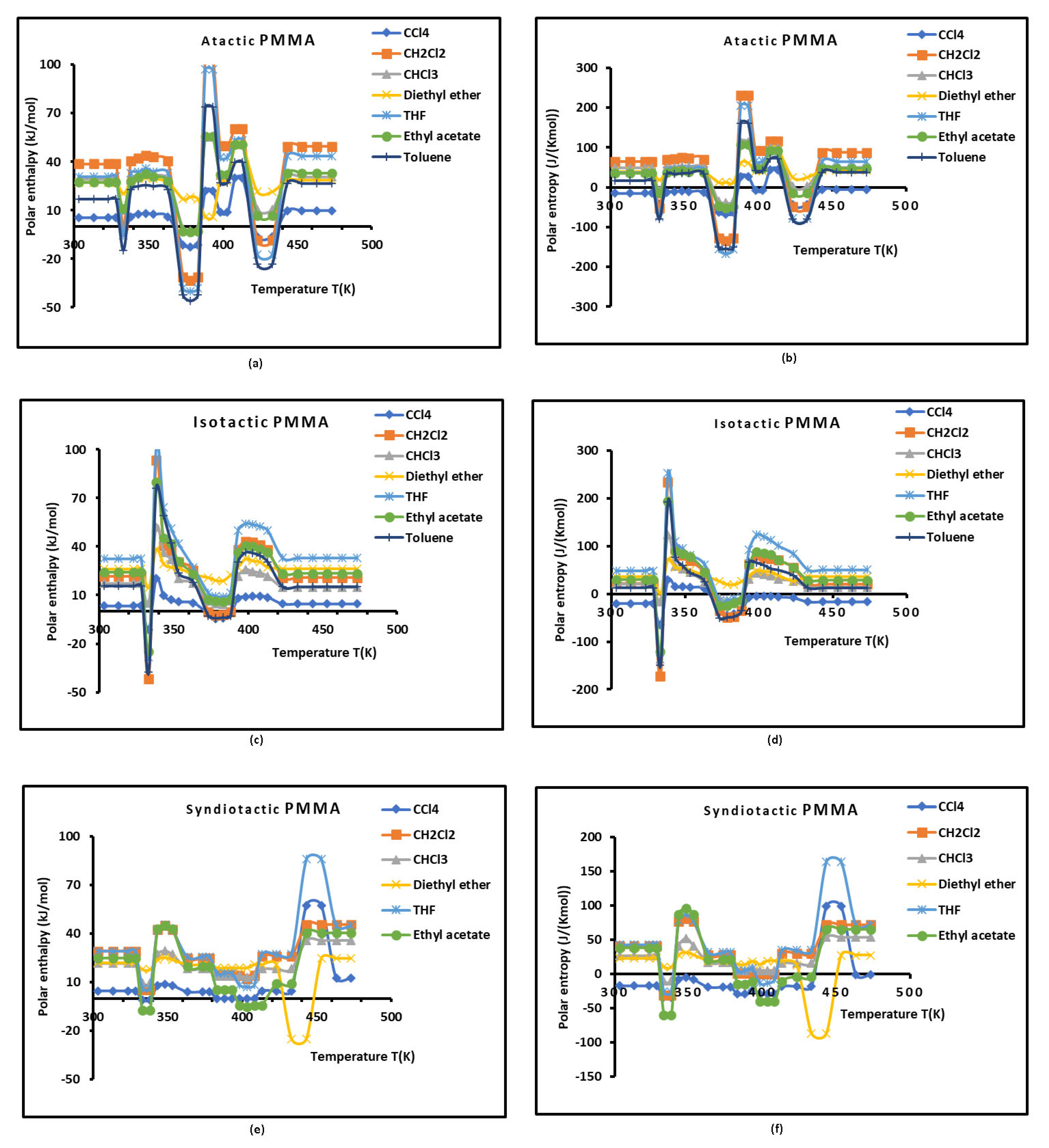
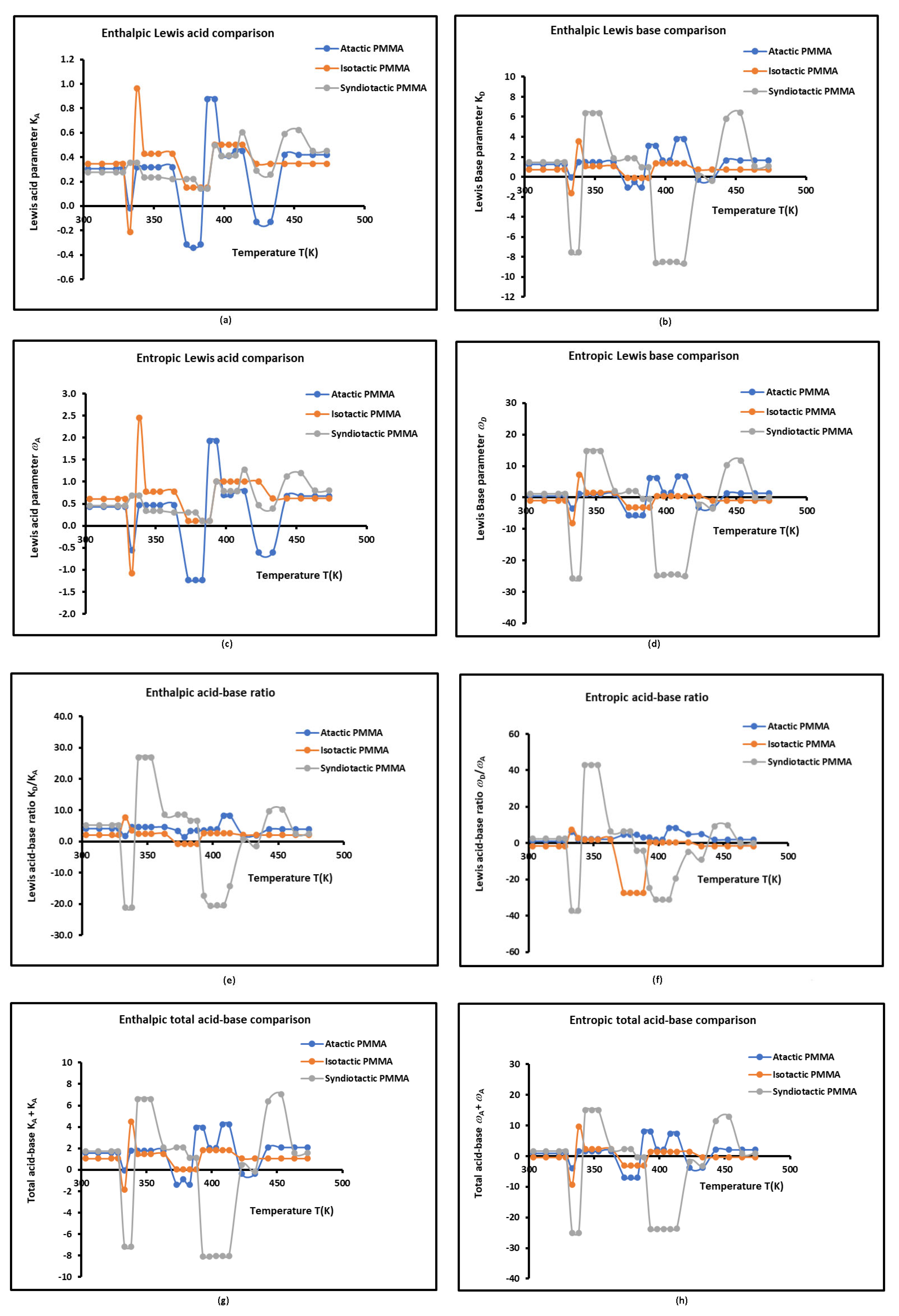
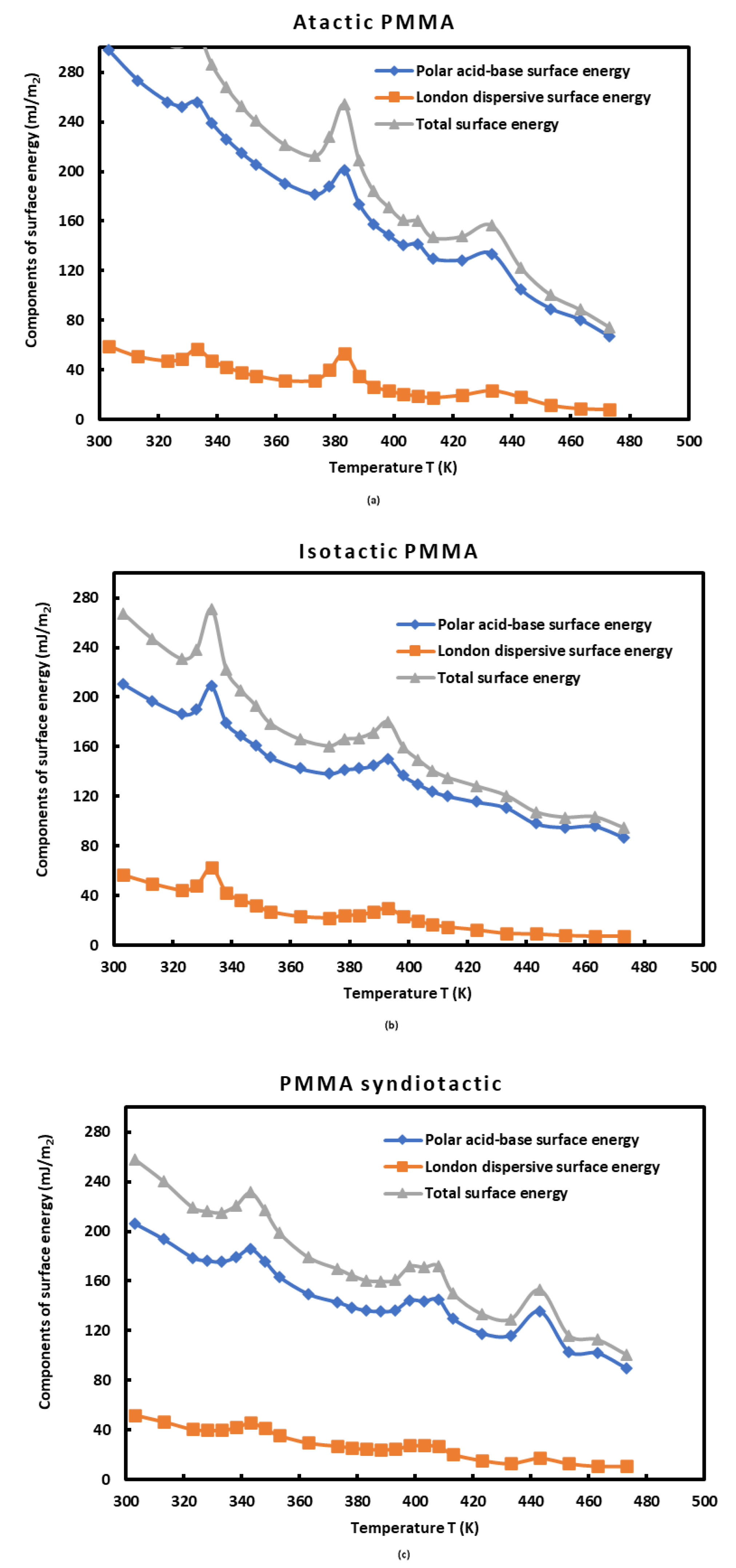
3.3. Lewis’s Acid-Base Properties
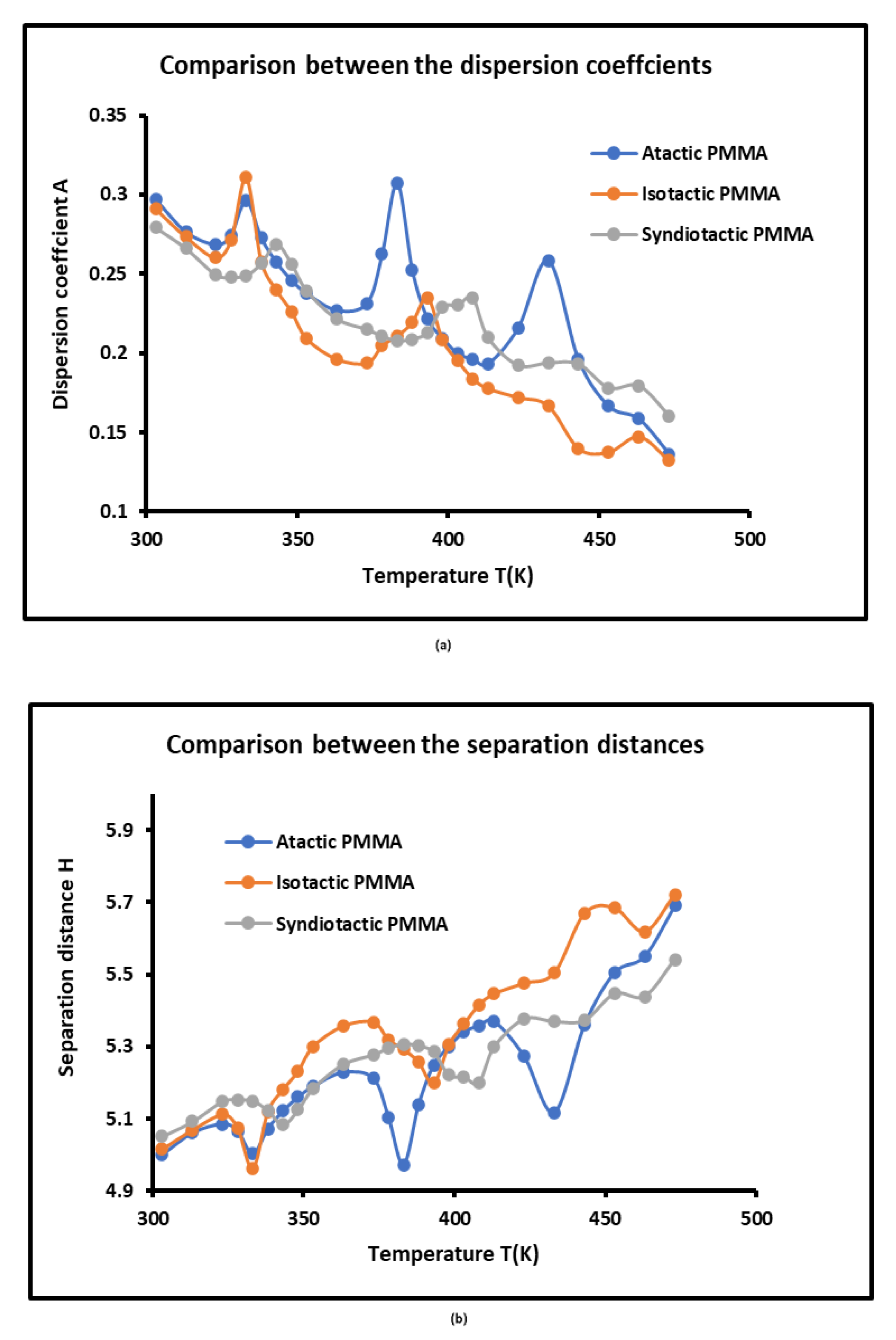
3.4. London Dispersive Free Energies of PMMAs and Dispersion Factor
3.5. Lewis Acid-Base Surface Energies of PMMAs and Polar Component of the Surface Energy of Polar Molecules
4. Conclusions
Supplementary Materials
Funding
Data Availability Statement
Conflicts of Interest
References
- Miri, M.J.; Pritchard, B.P.; Cheng, H.N. A versatile approach for modeling and simulating the tacticity of polymers. J Mol Model, 2011, 17, 1767–1780. [CrossRef]
- Worch, J.C.; Prydderch, H.; Jimaja, S. et al. Stereochemical enhancement of polymer properties. Nat Rev Chem, 2019, 3, 514–535. [CrossRef]
- Teiji Tsuruta, Yuhsuke Kawakami, 33 - Anionic Ring-opening Polymerization: Stereospecificity for Epoxides, Episulfides and Lactones, Editor(s): Geoffrey Allen, John C. Bevington, Comprehensive Polymer Science and Supplements, Pergamon, 1989, Pages 489-500, . [CrossRef]
- Izzo, L.; Griffiths, P.C.; Nilmini, R.; King, S.M.; Wallom, K.L.; Ferguson, E.L. Ruth Duncan, Impact of polymer tacticity on the physico-chemical behaviour of polymers proposed as therapeutics, International Journal of Pharmaceutics, 2011, 408 (1–2), 213-222, . [CrossRef]
- Grigoriadi, K.; Westrik, J. B. H. M.; Vogiatzis, G.; van Breemen, L.; Anderson, P.; Hütter, M. Physical ageing of polystyrene: does tacticity play a role? Macromolecules, 2019, 52(15), 5948-5954. [CrossRef]
- Chat, K.; Tu, W.; Unni, A.B.; and Adrjanowicz, K.; Influence of Tacticity on the Glass-Transition Dynamics of Poly(methyl methacrylate) (PMMA) under Elevated Pressure and Geometrical Nanoconfinement, Macromolecules, 2021, 54 (18), 8526-8537. [CrossRef]
- Cheng, G.; Barriere, T. Effect of viscoplasticity on microfluidic cavity filling efficiency of a thermoplastic polymer in hot-embossing process. Int J Adv Manuf Technol, 2019,103, 549–565. [CrossRef]
- Adrjanowicz, K.; Paluch, M. Discharge of the Nanopore Confinement Effect on the Glass Transition Dynamics via Viscous Flow. Phys. Rev. Lett., 2019, 122, No. 176101. [CrossRef]
- Chat, K.; Adrjanowicz, K. The Impact of the Molecular Weight on the Nonequilibrium Glass Transition Dynamics of Poly(Phenylmethyl Siloxane) in Cylindrical Nanopores. J. Phys. Chem. C, 2020, 124, 22321−22330. [CrossRef]
- Floudas, G.; Paluch, M.; Grzybowski, A.; Ngai, K. Molecular Dynamics of Glass-Forming Systems; Advances in Dielectrics; Springer: Berlin, Heidelberg, 2011; Vol. 1.
- Karasz, F. E.; Bair, H. E.; O’Reilly, J. M. Thermal Properties of Atactic and Isotactic Polystyrene. J. Phys. Chem. A, 1965, 69, 2657− 2667. [CrossRef]
- Chang, L.; Woo, E. M. Tacticity Effects on Glass Transition and Phase Behavior in Binary Blends of Poly(Methyl Methacrylate)s of Three Different Configurations. Polym. Chem. 2010, 1, 198−202. [CrossRef]
- Karasz, F. E.; MacKnight, W. J. The Influence of Stereoregularity on the Glass Transition Temperatures of Vinyl Polymers. Macromolecules, 1968, 1, 537−540. [CrossRef]
- Napolitano, S.; Glynos, E.; Tito, N. B. Glass Transition of Polymers in Bulk, Confined Geometries, and near Interfaces. Rep. Prog. Phys. 2017, 80, No. 036602. [CrossRef]
- Keddie, J. L.; Jones, R. A. L.; Cory, R. A. Interface and Surface Effects on the Glass-Transition Temperature in Thin Polymer Films. Faraday Discuss. 1994, 98, No. 219.
- Geng, K.; Tsui, O. K. C. Effects of Polymer Tacticity and Molecular Weight on the Glass Transition Temperature of Poly(Methyl Methacrylate) Films on Silica. Macromolecules, 2016, 49, 2671−2678. [CrossRef]
- Ngai, K. L.; Paluch, M. Classification of Secondary Relaxation in Glass-Formers Based on Dynamic Properties. J. Chem. Phys. 2004, 120, 857−873. [CrossRef]
- Cowie, J. M. G. Glass Transition Temperatures of Stereoblock, Isotactic and Atactic Polypropylenes of Various Chain Lengths. Eur. Polym. J. 1973, 9, 1041−1049. [CrossRef]
- Duarte, D. M.; Tu, W.; Dzienia, A.; Adrjanowicz, K. Study on the Effect of Side-Chain Group on the Segmental Dynamics of Selected Methacrylate Polymers at Ambient and High Pressures. Polymer, 2019, 183, No. 121860. [CrossRef]
- Adrjanowicz, K.; Winkler, R.; Chat, K.; Duarte, D. M.; Tu, W.; Unni, A. B.; Paluch, M.; Ngai, K. L. Study of Increasing Pressure and Nanopore Confinement Effect on the Segmental, Chain, and Secondary Dynamics of Poly(Methylphenylsiloxane). Macromolecules, 2019, 52, 3763−3774. [CrossRef]
- Adrjanowicz, K.; Winkler, R.; Dzienia, A.; Paluch, M.; Napolitano, S. Connecting 1D and 2D Confined Polymer Dynamics to Its Bulk Behavior via Density Scaling. ACS Macro Lett. 2019, 8, 304−309. [CrossRef]
- Kunal, K.; Robertson, C. G.; Pawlus, S.; Hahn, S. F.; Sokolov, A. P. Role of Chemical Structure in Fragility of Polymers: A Qualitative Picture. Macromolecules, 2008, 41, 7232−7238. [CrossRef]
- Casalini, R.; Roland, C. M.; Capaccioli, S. Effect of Chain Length on Fragility and Thermodynamic Scaling of the Local Segmental Dynamics in Poly(Methylmethacrylate). J. Chem. Phys. 2007, 126, No. 184903. [CrossRef]
- Wang, Y.; Lu, Y.; Jiang, Z.; Men, Y. Molecular Weight Dependency of Crystallization Line, Recrystallization Line, and Melting Line of Polybutene-1. Macromolecules, 2014, 47, 6401−6407. [CrossRef]
- Beena Unni, A.; Chat, K.; Duarte, D. M.; Wojtyniak, M.; Geppert-Rybczynska, M.; Kubacki, J.; Wrzalik, R.; Richert, R.; Adrjanowicz, K. Experimental Evidence on the Effect of Substrate Roughness on Segmental Dynamics of Confined Polymer Films. Polymer, 2020, 199, No. 122501.
- (46) Winkler, R.; Tu, W.; Laskowski, L.; Adrjanowicz, K. Effect of Surface Chemistry on the Glass-Transition Dynamics of Poly(Phenyl Methyl Siloxane) Confined in Alumina Nanopores. Langmuir 2020, 36, 7553−7565. [CrossRef]
- Beiner, M. Relaxation in Poly(Alkyl Methacrylate)s: Crossover Region and Nanophase Separation. Macromol. Rapid Commun. 2001, 22, 869−895.
- Ngai, K. L. L.; Gopalakrishnan, T. R. R.; Beiner, M. Relaxation in Poly(Alkyl Methacrylate)s: Change of Intermolecular Coupling with Molecular Structure, Tacticity, Molecular Weight, Copolymerization, Crosslinking, and Nanoconfinement. Polymer, 2006, 47, 7222−7230. [CrossRef]
- Chen, K.; Harris, K.; Vyazovkin, S. Tacticity as a Factor Contributing to the Thermal Stability of Polystyrene. Macromol. Chem. Phys. 2007, 208, 2525−2532.
- Shi, Y.; He, X. Effect of Tacticity Sequence of the Poly(N-isopropylacrylamide) Oligomer on Phase Transition Behavior in Aqueous Solution, The Journal of Physical Chemistry B, 2023, 127 (40), 8660-8668, https://doi.org/10.1021/acs.jpcb.3c03765. [CrossRef]
- Hasan, N.; Nguyen, T. M. H.; Busse, K.; Kressler, J. Influence of Tacticity on the Structure Formation of Poly(methacrylic acid) in Langmuir/Langmuir–Blodgett and Thin Films. Macromol. Chem. Phys. 2023, 224, 2200428. [CrossRef]
- Zhang, X.; Lin, F.; Cao, M.; Zhong, M. Rare earth–cobalt bimetallic catalysis mediates stereocontrolled living radical polymerization of acrylamides. Nat. Synth, 2023, 2, 855–863. [CrossRef]
- Biswas, C.S., Mitra, K., Singh, S. et al. Study of the effect of isotacticity on some physical properties of poly(N-isopropylacrylamide). Colloid Polym Sci, 2015, 293, 1749–1757. [CrossRef]
- Hamieh, T.; Rezzaki, M.; Schultz J. Study of the second order transitions and acid-base properties of polymers adsorbed on oxides, by using inverse gas chromatography at infinite dilution, I and II, J. Colloid Interface Sci., 2001, 233 (2), 339-347. [CrossRef]
- Hamieh, T. Study of the Specific Entropy of Poly (α-n-alkyl) Methacrylates Adsorbed on Alumina or Silica by Inverse Gas Chromatography (IGC), 2011, Soft Materials, 9 (1), 15 – 31. [CrossRef]
- Hamieh, T. New Approach for the Determination of Acid Base Properties of Poly(α-n-alkyl) Methacrylates Adsorbed on Silica by Inverse Gas Chromatography (IGC), Chromatographia, 2011, 73 (7-8), 709-719. [CrossRef]
- Hamieh, T.; Toufaily, J.; Mouneimné, A.B. Effect of the Tacticity of PMMA Adsorbed on Alumina and Silica on the Specific Entropy Change of Polymer by Inverse GC, Chromatographia, 2011, 73 (1-2), 99-107. [CrossRef]
- Hamieh T., Schultz J., New approach to characterise physicochemical properties of solid substrates by inverse gas chromatography at infinite dilution. I. Some new methods to determine the surface areas of some molecules adsorbed on solid surfaces, J. Chromatogr. A, 2002, 969(1-2), 17-36. [CrossRef]
- Hamieh, T.; Fadlallah, M.B.; Schultz, J. New approach to characterise physicochemical properties of solid substrates by inverse gas chromatography at infinite dilution: III. Determination of the acid–base properties of some solid substrates (polymers, oxides and carbon fibres): a new model, J Chromatogr A. 2002, 969 (1-2), 37-47. [CrossRef]
- Ito M; Ishizone T. Living anionic polymerization of N-methoxymethyl-N-isopropylacrylamide: Synthesis of well-defined poly(N-isopropylacrylamide) having various stereoregularity, J. Polym. Sci. A Polym. Chem., 2006, 44 , 4832–4845, . [CrossRef]
- Hirano, T.; Okumura, Y.; Kitajim,a H.; Seno, M.; Sato, T. Dual roles of alkyl alcohols as syndiotactic-specificity inducers and accelerators in the radical polymerization of N-isopropylacrylamide and some properties of syndiotactic poly(N-isopropylacrylamide), J. Polym. Sci. A Polym. Chem., 2006, 44 , 4450–4460, . [CrossRef]
- Nuopponen M, Kalliomäki K, Laukkanen A, Hietala S, Tenhu H. A–B–A stereoblock copolymers of N-isopropylacrylamide, J. Polym. Sci. A Polym. Chem., 2008, 46, 38–46. [CrossRef]
- Hirano, T.; Miki, H.; Seno, M.; Sato, T. Effect of polymerization conditions on the syndiotactic-specificity in radical polymerization of N-isopropylacrylamide and fractionation of the obtained polymer according to the stereoregularity, Polymer, 2005, 46 (15), 5501-5505, . [CrossRef]
- Papadopoulou, S.K.; Panayiotou, C. Assessment of the thermodynamic properties of poly(2,2,2-trifluoroethyl methacrylate) by inverse gas chromatography, J. Chromatogr. A, 2014, 1324, 207– 214. [CrossRef]
- Saint-Flour, C.; Papirer, E.; Gas-solid chromatography. A method of measuring surface free energy characteristics of short carbon fibers. 1. Through adsorption isotherms, Ind. Eng. Chem. Prod. Res. Dev., 1982, 21, 337-341, . [CrossRef]
- Saint-Flour, C.; Papirer, E.; Gas-solid chromatography: method of measuring surface free energy characteristics of short fibers. 2. Through retention volumes measured near zero surface coverage, Ind. Eng. Chem. Prod. Res. Dev., 1982, 21, 666-669, . [CrossRef]
- Donnet, J.-B.; Park, S.; Balard, H. Evaluation of specific interactions of solid surfaces by inverse gas chromatography, Chromatographia, 1991, 31, 434–440. [CrossRef]
- Brendlé, E.; Papirer, E. A new topological index for molecular probes used in inverse gas chromatography for the surface nanorugosity evaluation, 2. Application for the Evaluation of the Solid Surface Specific Interaction Potential, J. Colloid Interface Sci., 1997, 194, 217–2224. [CrossRef]
- Brendlé, E.; Papirer, E. A new topological index for molecular probes used in inverse gas chromatography for the surface nanorugosity evaluation, 1. Method of Evaluation, J. Colloid Interface Sci. 1997, 194, 207–216. [CrossRef]
- Sawyer, D.T.; Brookman. D.J. Thermodynamically based gas chromatographic retention index for organic molecules using salt-modified aluminas and porous silica beads, Anal. Chem. 1968, 40, 1847-1850. [CrossRef]
- Hamieh, T. New methodology to study the dispersive component of the surface energy and acid–base properties of silica particles by inverse gas chromatography at infinite dilution, Journal of Chromatographic Science, 2022, 60 (2), 126-142, . [CrossRef]
- Voelkel, A. Inverse gas chromatography: characterization of polymers, fibers, modified silicas, and surfactants. Crit Rev Anal Chem. 1991, 22, 411-439. [CrossRef]
- Hamieh, T.; Ahmad, A.A.; Roques-Carmes, T.; Toufaily, J. New approach to determine the surface and interface thermodynamic properties of H-β-zeolite/rhodium catalysts by inverse gas chromatography at infinite dilution. Sci. Rep. 2020, 10, 20894. [CrossRef]
- Hamieh, T. The Effect of Temperature on the Surface Energetic Properties of Carbon Fibers Using Inverse Gas Chromatography, Crystals, 2024, 14(1), 28; . [CrossRef]
- Hamieh, T. New physicochemical methodology for the determination of the surface thermodynamic properties of solid particles. Appliedchem, 2023, 3, 229–255. [CrossRef]
- Hamieh, T. New Progress on London Dispersive Energy, Polar Surface Interactions, and Lewis’s Acid–Base Properties of Solid Surfaces, Molecules, 2024, 29 (5), 949, . [CrossRef]
- Hamieh, T. London Dispersive and Lewis Acid-Base Surface Energy of 2D Single-Crystalline and Polycrystalline Covalent Organic Frameworks, Crystals, 2024, 14(2), 148; [CrossRef]
- Hamieh, T. Inverse Gas Chromatography to Characterize the Surface Properties of Solid Materials, Chem. Mater. 2024, 2024, . [CrossRef]
- Hamieh, T. Study of the temperature effect on the surface area of model organic molecules, the dispersive surface energy and the surface properties of solids by inverse gas chromatography, J. Chromatogr. A, 2020, 1627, 461372.
- Dritsas, G.S.; Karatasos, K.; Panayiotou, C. Investigation of thermodynamic properties of hyperbranched aliphatic polyesters by inverse gas chromatography, J. Chromatogr. A, 2009, 1216 (51), 8979-8985. [CrossRef]
- Papadopoulou, S.K.; Panayiotou, C. Thermodynamic characterization of poly(1,1,1,3,3,3-hexafluoroisopropyl methacrylate) by inverse gas chromatography, J. Chromatogr. A, 2012, 1229, 230-236. [CrossRef]
- Kołodziejek, J.; Voelkel, A.; K. Heberger, Characterization of hybrid materials by means of inverse gas chromatography and chemometrics, J. Pharm. Sci., 2013, 102, 15241531. [CrossRef]
- Belgacem, M.N.; Czeremuszkin, G.; Sapieha, S.; Gandini, A. Surface by XPS characterization and inverse gas of cellulose fibres chromatography, Cellulose, 1995, 2, 145157, . [CrossRef]
- Papadopoulou, S. K.; Tsioptsias, .; Pavlou, A. K.; Kaderides, K.; Sotiriou S.; Panayiotou, C. “Superhydrophobic surfaces from hydrophobic or hydrophilic polymers via nanophase separation or electrospinning/electrospraying. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2011, 387, 71-78. [CrossRef]
- Papirer E.; Perrin J.-M.; Siffert B.; Philipponneau G. Surface characteristics of aluminas in relation with polymer adsorption, J. Colloid Interface Sci., 1991, 144, 263–270, . [CrossRef]
- Gutmann, V. The Donor-acceptor Approach to Molecular Interactions, Plenum. New York, 1978.
- Riddle, F. L.; Fowkes, F. M. Spectral shifts in acid-base chemistry. Van der Waals contributions to acceptor numbers, Spectral shifts in acid-base chemistry. 1. van der Waals contributions to acceptor numbers. J. Am. Chem. Soc., 1990, 112 (9), 3259-3264. [CrossRef]
- Hamieh, T.; Rezzaki M.; Schultz, J. Study of the transition temperatures and acid-base properties of poly (methyl methacrylate) adsorbed on alumina and silica, by using inverse gas chromatography technique, Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2001, 189 (1-3), 279-291. [CrossRef]
- Hamieh, T. Some Irregularities in the Evaluation of Surface Parameters of Solid Materials by Inverse Gas Chromatography, Langmuir, 2023, 39, 17059-17070, . [CrossRef]
- David R. Lide, ed., CRC Handbook of Chemistry and Physics, Internet Version 2007, (87th Edition), <http:/www.hbcpnetbase.com>, Taylor and Francis, Boca Raton, FL, 2007.
| Transition temperature | |||
|---|---|---|---|
| Atactic PMMA | 60°C | 110°C | 160°C |
| Isotactic PMMA | 60°C | 120°C | 170°C |
| Syndiotactic PMMA | 70°C | 130°C | 170°C |
| Atactic PMMA | ||
| Equation of | R² | Temperature interval |
| 0.935 | 303.15K - 333.15K | |
| 0.9722 | 333.15K - 378.15K | |
| 0.9477 | 378.15K - 423.15K | |
| 0.9902 | 423.15K - 473.15K | |
| Isotactic PMMA | ||
| Equation of | R² | Temperature interval |
| 0.9398 | 303.15K - 333.15K | |
| 0.9557 | 333.15K - 393.15K | |
| 0.9925 | 393.15K - 443.15K | |
| 0.9896 | 443.15K - 473.15K | |
| Syndiotactic PMMA | ||
| Equation of | R² | Temperature interval |
| 0.9485 | 303.15K - 343.15K | |
| 0.9900 | 343.15K - 403.15K | |
| 0.9569 | 403.15K - 443.15K | |
| 1.000 | 443.15K - 473.15K | |
| Equation of | R² | Temperature interval | |
| Atactic PMMA | 0.8368 | 303.15K - 473.15K | |
| Isotactic PMMA | 0.8491 | 303.15K - 473.15K | |
| Syndiotactic PMMA | 0.8368 | 303.15K - 473.15K |
| Atactic PMMA | 144.37 | 494.1 | |
| Isotactic PMMA | 143.16 | 472.6 | |
| Syndiotactic PMMA | 120.8 | 503.8 |
| Polar free energy of atactic PMMA | |||||||
| Temperature T(K) | CCl4 | CH2Cl2 | CHCl3 | Diethyl ether | THF | Ethyl acetate | Toluene |
| 303.15 | 10.047 | 19.251 | 14.493 | 15.248 | 19.777 | 16.627 | 11.951 |
| 313.15 | 10.142 | 18.357 | 13.862 | 14.794 | 19.179 | 16.162 | 11.585 |
| 323.15 | 10.303 | 17.750 | 13.399 | 14.416 | 18.845 | 15.830 | 11.442 |
| 328.15 | 10.433 | 17.669 | 13.278 | 14.259 | 18.889 | 15.755 | 11.561 |
| 333.15 | 10.642 | 17.926 | 13.323 | 14.166 | 19.257 | 15.828 | 11.961 |
| 338.15 | 10.621 | 17.193 | 12.884 | 13.904 | 18.679 | 15.479 | 11.526 |
| 343.15 | 10.638 | 16.636 | 12.532 | 13.665 | 18.266 | 15.198 | 11.244 |
| 348.15 | 10.676 | 16.168 | 12.229 | 13.447 | 17.938 | 14.959 | 11.033 |
| 353.15 | 10.729 | 15.768 | 11.962 | 13.242 | 17.673 | 14.749 | 10.878 |
| 363.15 | 10.865 | 15.098 | 11.494 | 12.854 | 17.267 | 14.384 | 10.679 |
| 373.15 | 11.081 | 14.759 | 11.178 | 12.521 | 17.185 | 14.163 | 10.758 |
| 378.15 | 11.342 | 15.218 | 11.285 | 12.442 | 17.767 | 14.323 | 11.335 |
| 383.15 | 11.682 | 15.988 | 11.514 | 12.403 | 18.664 | 14.617 | 12.180 |
| 388.15 | 11.479 | 14.558 | 10.824 | 12.058 | 17.371 | 13.963 | 11.140 |
| 393.15 | 11.408 | 13.673 | 10.371 | 11.792 | 16.616 | 13.543 | 10.566 |
| 398.15 | 11.435 | 13.186 | 10.086 | 11.582 | 16.256 | 13.295 | 10.331 |
| 403.15 | 11.476 | 12.754 | 9.827 | 11.382 | 15.950 | 13.070 | 10.144 |
| 408.15 | 11.850 | 12.756 | 9.922 | 11.500 | 16.078 | 13.204 | 10.372 |
| 413.15 | 11.627 | 12.174 | 9.424 | 11.019 | 15.621 | 12.744 | 10.013 |
| 423.15 | 11.940 | 12.230 | 9.255 | 10.729 | 15.944 | 12.693 | 10.431 |
| 433.15 | 12.367 | 12.723 | 9.224 | 10.476 | 16.727 | 12.831 | 11.230 |
| 443.15 | 12.147 | 10.846 | 8.317 | 9.886 | 15.066 | 11.893 | 10.015 |
| 453.15 | 12.172 | 9.778 | 7.776 | 9.494 | 14.219 | 11.360 | 9.451 |
| 463.15 | 12.310 | 9.164 | 7.383 | 9.138 | 13.845 | 11.019 | 9.285 |
| 473.15 | 12.276 | 8.131 | 6.767 | 8.597 | 13.044 | 10.414 | 8.814 |
| Polar free energy of isotactic PMMA | |||||||
| Temperature T(K) | CCl4 | CH2Cl2 | CHCl3 | Diethyl ether | THF | Ethyl acetate | Toluene |
| 303.15 | 9.422 | 14.973 | 11.160 | 15.214 | 17.783 | 15.139 | 11.652 |
| 313.15 | 9.579 | 14.564 | 10.848 | 14.812 | 17.121 | 14.694 | 11.374 |
| 323.15 | 9.760 | 14.259 | 10.605 | 14.441 | 16.553 | 14.331 | 11.174 |
| 328.15 | 9.943 | 14.504 | 10.689 | 14.334 | 16.646 | 14.457 | 11.401 |
| 333.15 | 10.269 | 15.363 | 11.071 | 14.330 | 17.328 | 15.059 | 12.148 |
| 338.15 | 10.120 | 14.192 | 10.454 | 13.969 | 16.068 | 14.088 | 11.188 |
| 343.15 | 10.147 | 13.788 | 10.220 | 13.741 | 15.539 | 13.712 | 10.874 |
| 348.15 | 10.198 | 13.485 | 10.041 | 13.539 | 15.103 | 13.414 | 10.639 |
| 353.15 | 10.231 | 13.105 | 9.833 | 13.329 | 14.592 | 13.058 | 10.338 |
| 363.15 | 10.401 | 12.806 | 9.635 | 12.975 | 14.011 | 12.701 | 10.129 |
| 373.15 | 10.625 | 12.735 | 9.544 | 12.655 | 13.652 | 12.521 | 10.117 |
| 378.15 | 10.804 | 12.970 | 9.615 | 12.537 | 13.740 | 12.637 | 10.338 |
| 383.15 | 10.955 | 13.096 | 9.636 | 12.400 | 13.722 | 12.671 | 10.471 |
| 388.15 | 11.121 | 13.277 | 9.678 | 12.268 | 13.759 | 12.745 | 10.649 |
| 393.15 | 11.326 | 13.611 | 9.781 | 12.161 | 13.952 | 12.936 | 10.956 |
| 398.15 | 11.297 | 13.014 | 9.509 | 11.928 | 13.210 | 12.416 | 10.465 |
| 403.15 | 11.344 | 12.718 | 9.363 | 11.737 | 12.768 | 12.127 | 10.232 |
| 408.15 | 11.400 | 12.466 | 9.238 | 11.556 | 12.367 | 11.872 | 10.034 |
| 413.15 | 11.482 | 12.320 | 9.155 | 11.384 | 12.074 | 11.698 | 9.930 |
| 423.15 | 11.687 | 12.183 | 9.055 | 11.068 | 11.642 | 11.467 | 9.850 |
| 433.15 | 11.891 | 12.051 | 8.958 | 10.751 | 11.213 | 11.241 | 9.776 |
| 443.15 | 11.933 | 11.393 | 8.645 | 10.315 | 10.245 | 10.615 | 9.277 |
| 453.15 | 12.152 | 11.328 | 8.582 | 10.009 | 9.880 | 10.440 | 9.259 |
| 463.15 | 12.437 | 11.521 | 8.595 | 9.719 | 9.789 | 10.457 | 9.468 |
| 473.15 | 12.492 | 11.059 | 8.311 | 9.232 | 9.025 | 9.972 | 9.163 |
| Polar free energy of syndiotactic PMMA | |||||||
| Temperature T(K) | CCl4 | CH2Cl2 | CHCl3 | Diethyl ether | THF | Ethyl acetate | Toluene |
| 303.15 | 9.704 | 16.617 | 13.631 | 15.200 | 16.129 | 13.339 | 11.448 |
| 313.15 | 9.879 | 16.212 | 13.363 | 14.978 | 15.709 | 12.965 | 10.664 |
| 323.15 | 10.028 | 15.714 | 13.055 | 14.733 | 15.199 | 12.476 | 10.121 |
| 328.15 | 10.138 | 15.625 | 12.986 | 14.642 | 15.093 | 12.442 | 9.935 |
| 333.15 | 10.259 | 15.581 | 12.939 | 14.559 | 15.031 | 12.470 | 9.343 |
| 338.15 | 10.416 | 15.691 | 12.969 | 14.506 | 15.116 | 12.701 | 11.685 |
| 343.15 | 10.594 | 15.893 | 13.038 | 14.458 | 15.293 | 13.068 | 16.158 |
| 348.15 | 10.646 | 15.560 | 12.853 | 14.326 | 14.953 | 12.704 | 13.145 |
| 353.15 | 10.673 | 15.123 | 12.624 | 14.180 | 14.510 | 12.198 | 11.800 |
| 363.15 | 10.809 | 14.608 | 12.344 | 13.950 | 13.968 | 11.670 | 11.126 |
| 373.15 | 11.001 | 14.337 | 12.182 | 13.759 | 13.661 | 11.473 | 10.155 |
| 378.15 | 11.091 | 14.178 | 12.092 | 13.660 | 13.483 | 11.342 | 9.749 |
| 383.15 | 11.190 | 14.056 | 12.021 | 13.570 | 13.341 | 11.259 | 9.829 |
| 388.15 | 11.309 | 14.017 | 11.982 | 13.488 | 13.283 | 11.293 | 9.622 |
| 393.15 | 11.444 | 14.041 | 11.971 | 13.417 | 13.287 | 11.408 | 10.293 |
| 398.15 | 11.646 | 14.333 | 12.065 | 13.380 | 13.561 | 11.891 | 13.616 |
| 403.15 | 11.755 | 14.278 | 11.999 | 13.262 | 13.493 | 11.937 | 13.468 |
| 408.15 | 11.905 | 14.333 | 12.013 | 13.226 | 13.527 | 12.062 | 10.553 |
| 413.15 | 11.879 | 13.723 | 11.757 | 13.076 | 12.890 | 11.309 | 7.576 |
| 423.15 | 12.004 | 13.210 | 11.520 | 12.858 | 12.328 | 10.781 | 7.563 |
| 433.15 | 12.235 | 13.114 | 11.441 | 12.694 | 12.192 | 10.819 | 7.746 |
| 443.15 | 13.534 | 14.036 | 12.398 | 13.561 | 13.081 | 11.903 | 7.539 |
| 453.15 | 12.543 | 12.452 | 11.078 | 12.240 | 11.439 | 10.311 | 4.992 |
| 463.15 | 12.772 | 12.352 | 10.999 | 12.073 | 11.299 | 10.345 | 4.800 |
| 473.15 | 12.779 | 11.679 | 10.632 | 11.688 | 10.574 | 9.720 | 4.785 |
| Atactic PMMA | ||||||||
| T(K) | / | / | ||||||
| 303.15 | 0.304 | 1.260 | 1.565 | 4.142 | 0.43 | 0.52 | 0.95 | 1.22 |
| 313.15 | 0.304 | 1.260 | 1.565 | 4.142 | 0.43 | 0.52 | 0.95 | 1.22 |
| 323.15 | 0.304 | 1.260 | 1.565 | 4.142 | 0.43 | 0.52 | 0.95 | 1.22 |
| 328.15 | 0.304 | 1.260 | 1.565 | 4.142 | 0.43 | 0.52 | 0.95 | 1.22 |
| 333.15 | -0.018 | -0.033 | -0.051 | 1.847 | -0.56 | -3.43 | -3.99 | 6.16 |
| 338.15 | 0.317 | 1.459 | 1.775 | 4.602 | 0.46 | 1.09 | 1.56 | 2.36 |
| 343.15 | 0.317 | 1.459 | 1.775 | 4.602 | 0.46 | 1.09 | 1.56 | 2.36 |
| 348.15 | 0.317 | 1.459 | 1.775 | 4.602 | 0.46 | 1.09 | 1.56 | 2.36 |
| 353.15 | 0.317 | 1.459 | 1.775 | 4.602 | 0.46 | 1.09 | 1.56 | 2.36 |
| 363.15 | 0.317 | 1.459 | 1.775 | 4.602 | 0.46 | 1.09 | 1.56 | 2.36 |
| 373.15 | -0.313 | -1.063 | -1.376 | 3.397 | -1.23 | -5.71 | -6.93 | 4.65 |
| 378.15 | -0.343 | -0.508 | -0.851 | 1.478 | -1.23 | -5.71 | -6.93 | 4.65 |
| 383.15 | -0.313 | -1.063 | -1.376 | 3.397 | -1.23 | -5.71 | -6.93 | 4.65 |
| 388.15 | 0.876 | 3.081 | 3.957 | 3.518 | 1.93 | 6.11 | 8.04 | 3.17 |
| 393.15 | 0.876 | 3.081 | 3.957 | 3.518 | 1.93 | 6.11 | 8.04 | 3.17 |
| 398.15 | 0.411 | 1.630 | 2.042 | 3.966 | 0.69 | 1.44 | 2.13 | 2.07 |
| 403.15 | 0.411 | 1.630 | 2.042 | 3.966 | 0.69 | 1.44 | 2.13 | 2.07 |
| 408.15 | 0.453 | 3.784 | 4.237 | 8.350 | 0.79 | 6.62 | 7.41 | 8.40 |
| 413.15 | 0.453 | 3.784 | 4.237 | 8.350 | 0.79 | 6.62 | 7.41 | 8.40 |
| 423.15 | -0.128 | -0.259 | -0.388 | 2.016 | -0.61 | -3.12 | -3.73 | 5.13 |
| 433.15 | -0.128 | -0.259 | -0.388 | 2.016 | -0.61 | -3.12 | -3.73 | 5.13 |
| 443.15 | 0.418 | 1.667 | 2.085 | 3.989 | 0.67 | 1.40 | 2.08 | 2.08 |
| 453.15 | 0.418 | 1.667 | 2.085 | 3.989 | 0.67 | 1.40 | 2.08 | 2.08 |
| 463.15 | 0.418 | 1.667 | 2.085 | 3.989 | 0.67 | 1.40 | 2.08 | 2.08 |
| 473.15 | 0.418 | 1.667 | 2.085 | 3.989 | 0.67 | 1.40 | 2.08 | 2.08 |
| Isotactic PMMA | ||||||||
| T(K) | / | / | ||||||
| 303.15 | 0.344 | 0.717 | 1.060 | 2.084 | 0.61 | -1.03 | -0.42 | -1.68 |
| 313.15 | 0.344 | 0.717 | 1.060 | 2.084 | 0.61 | -1.03 | -0.42 | -1.68 |
| 323.15 | 0.344 | 0.717 | 1.060 | 2.084 | 0.61 | -1.03 | -0.42 | -1.68 |
| 328.15 | 0.344 | 0.717 | 1.060 | 2.084 | 0.61 | -1.03 | -0.42 | -1.68 |
| 333.15 | -0.211 | -1.628 | -1.838 | 7.730 | -1.08 | -8.20 | -9.28 | 7.59 |
| 338.15 | 0.964 | 3.531 | 4.494 | 3.664 | 2.45 | 7.28 | 9.72 | 2.98 |
| 343.15 | 0.427 | 1.066 | 1.493 | 2.498 | 0.78 | 1.52 | 2.30 | 1.95 |
| 348.15 | 0.427 | 1.066 | 1.493 | 2.498 | 0.78 | 1.52 | 2.30 | 1.95 |
| 353.15 | 0.427 | 1.066 | 1.493 | 2.498 | 0.78 | 1.52 | 2.30 | 1.95 |
| 363.15 | 0.427 | 1.066 | 1.493 | 2.498 | 0.78 | 1.52 | 2.30 | 1.95 |
| 373.15 | 0.153 | -0.119 | 0.034 | -0.778 | 0.12 | -3.21 | -3.10 | -27.41 |
| 378.15 | 0.153 | -0.119 | 0.034 | -0.778 | 0.12 | -3.21 | -3.10 | -27.41 |
| 383.15 | 0.153 | -0.119 | 0.034 | -0.778 | 0.12 | -3.21 | -3.10 | -27.41 |
| 388.15 | 0.153 | -0.119 | 0.034 | -0.778 | 0.12 | -3.21 | -3.10 | -27.41 |
| 393.15 | 0.501 | 1.338 | 1.839 | 2.668 | 1.00 | 0.49 | 1.50 | 0.49 |
| 398.15 | 0.501 | 1.338 | 1.839 | 2.668 | 1.00 | 0.49 | 1.50 | 0.49 |
| 403.15 | 0.501 | 1.338 | 1.839 | 2.668 | 1.00 | 0.49 | 1.50 | 0.49 |
| 408.15 | 0.501 | 1.338 | 1.839 | 2.668 | 1.00 | 0.49 | 1.50 | 0.49 |
| 413.15 | 0.501 | 1.338 | 1.839 | 2.668 | 1.00 | 0.49 | 1.50 | 0.49 |
| 423.15 | 0.347 | 0.728 | 1.076 | 2.097 | 1.00 | 0.49 | 1.50 | 0.49 |
| 433.15 | 0.347 | 0.728 | 1.076 | 2.097 | 0.63 | -0.99 | -0.37 | -1.58 |
| 443.15 | 0.347 | 0.728 | 1.076 | 2.097 | 0.63 | -0.99 | -0.37 | -1.58 |
| 453.15 | 0.347 | 0.728 | 1.076 | 2.097 | 0.63 | -0.99 | -0.37 | -1.58 |
| 463.15 | 0.347 | 0.728 | 1.076 | 2.097 | 0.63 | -0.99 | -0.37 | -1.58 |
| 473.15 | 0.347 | 0.728 | 1.076 | 2.097 | 0.63 | -0.99 | -0.37 | -1.58 |
| Syndiotactic PMMA | ||||||||
| T(K) | / | / | ||||||
| 303.15 | 0.276 | 1.452 | 1.728 | 5.262 | 0.46 | 1.14 | 1.60 | 2.47 |
| 313.15 | 0.276 | 1.452 | 1.728 | 5.262 | 0.46 | 1.14 | 1.60 | 2.47 |
| 323.15 | 0.276 | 1.452 | 1.728 | 5.262 | 0.46 | 1.14 | 1.60 | 2.47 |
| 328.15 | 0.276 | 1.452 | 1.728 | 5.262 | 0.46 | 1.14 | 1.60 | 2.47 |
| 333.15 | 0.355 | -7.527 | -7.172 | -21.189 | 0.69 | -25.75 | -25.06 | -37.30 |
| 338.15 | 0.355 | -7.527 | -7.172 | -21.189 | 0.69 | -25.75 | -25.06 | -37.30 |
| 343.15 | 0.237 | 6.367 | 6.603 | 26.907 | 0.34 | 14.74 | 15.08 | 42.88 |
| 348.15 | 0.237 | 6.367 | 6.603 | 26.907 | 0.34 | 14.74 | 15.08 | 42.88 |
| 353.15 | 0.237 | 6.367 | 6.603 | 26.907 | 0.34 | 14.74 | 15.08 | 42.88 |
| 363.15 | 0.220 | 1.888 | 2.108 | 8.591 | 0.30 | 2.01 | 2.32 | 6.60 |
| 373.15 | 0.220 | 1.888 | 2.108 | 8.591 | 0.30 | 2.01 | 2.32 | 6.60 |
| 378.15 | 0.220 | 1.888 | 2.108 | 8.591 | 0.30 | 2.01 | 2.32 | 6.60 |
| 383.15 | 0.143 | 0.961 | 1.104 | 6.703 | 0.11 | -0.46 | -0.35 | -4.25 |
| 388.15 | 0.143 | 0.961 | 1.104 | 6.703 | 0.11 | -0.46 | -0.35 | -4.25 |
| 393.15 | 0.496 | -8.578 | -8.082 | -17.310 | 1.01 | -24.79 | -23.79 | -24.65 |
| 398.15 | 0.411 | -8.490 | -8.079 | -20.645 | 0.79 | -24.58 | -23.79 | -31.17 |
| 403.15 | 0.413 | -8.464 | -8.052 | -20.514 | 0.79 | -24.51 | -23.72 | -30.94 |
| 408.15 | 0.413 | -8.464 | -8.052 | -20.514 | 0.79 | -24.51 | -23.72 | -30.94 |
| 413.15 | 0.605 | -8.652 | -8.047 | -14.291 | 1.28 | -24.99 | -23.71 | -19.53 |
| 423.15 | 0.290 | 0.185 | 0.475 | 0.639 | 0.47 | -2.19 | -1.72 | -4.69 |
| 433.15 | 0.259 | -0.401 | -0.143 | -1.552 | 0.39 | -3.54 | -3.15 | -8.98 |
| 443.15 | 0.590 | 5.780 | 6.369 | 9.796 | 1.12 | 10.32 | 11.44 | 9.24 |
| 453.15 | 0.624 | 6.415 | 7.039 | 10.283 | 1.19 | 11.79 | 12.98 | 9.86 |
| 463.15 | 0.451 | 1.105 | 1.556 | 2.453 | 0.80 | 0.10 | 0.90 | 0.12 |
| 473.15 | 0.451 | 1.105 | 1.556 | 2.453 | 0.80 | 0.10 | 0.90 | 0.12 |
| Atactic PMMA | Isotactic PMMA | Syndiotactic PMMA | |||||||
|---|---|---|---|---|---|---|---|---|---|
| T(K) | |||||||||
| 303.15 | 203.75 | 108.73 | 297.68 | 123.27 | 90.13 | 210.82 | 151.82 | 69.97 | 206.14 |
| 313.15 | 183.42 | 101.70 | 273.16 | 115.44 | 84.06 | 197.02 | 143.04 | 65.44 | 193.50 |
| 323.15 | 169.77 | 96.59 | 256.11 | 109.56 | 79.16 | 186.25 | 133.06 | 59.99 | 178.69 |
| 328.15 | 167.38 | 95.20 | 252.46 | 112.78 | 80.16 | 190.16 | 130.89 | 59.37 | 176.30 |
| 333.15 | 171.41 | 95.61 | 256.03 | 125.91 | 86.53 | 208.76 | 129.50 | 59.34 | 175.32 |
| 338.15 | 156.91 | 90.97 | 238.96 | 106.91 | 75.36 | 179.52 | 130.68 | 61.25 | 178.94 |
| 343.15 | 146.17 | 87.28 | 225.89 | 100.41 | 71.04 | 168.91 | 133.41 | 64.53 | 185.56 |
| 348.15 | 137.38 | 84.13 | 215.02 | 95.56 | 67.65 | 160.81 | 127.25 | 60.68 | 175.74 |
| 353.15 | 130.01 | 81.38 | 205.72 | 89.82 | 63.79 | 151.39 | 119.60 | 55.66 | 163.18 |
| 363.15 | 118.03 | 76.64 | 190.22 | 84.91 | 59.76 | 142.47 | 110.49 | 50.45 | 149.32 |
| 373.15 | 111.70 | 73.58 | 181.32 | 83.16 | 57.51 | 138.31 | 105.40 | 48.28 | 142.68 |
| 378.15 | 118.18 | 74.88 | 188.14 | 85.83 | 58.30 | 141.48 | 102.57 | 46.95 | 138.79 |
| 383.15 | 129.80 | 77.62 | 200.74 | 87.09 | 58.32 | 142.53 | 100.32 | 46.05 | 135.93 |
| 388.15 | 107.09 | 70.48 | 173.76 | 89.07 | 58.72 | 144.64 | 99.29 | 46.10 | 135.31 |
| 393.15 | 94.01 | 65.98 | 157.52 | 93.16 | 60.20 | 149.77 | 99.14 | 46.82 | 136.26 |
| 398.15 | 87.01 | 63.28 | 148.40 | 84.75 | 55.19 | 136.79 | 102.80 | 50.62 | 144.27 |
| 403.15 | 81.01 | 60.87 | 140.44 | 80.55 | 52.40 | 129.94 | 101.53 | 50.77 | 143.59 |
| 408.15 | 80.65 | 61.82 | 141.22 | 77.02 | 49.97 | 124.08 | 101.82 | 51.59 | 144.95 |
| 413.15 | 73.10 | 57.31 | 129.46 | 74.87 | 48.29 | 120.25 | 92.89 | 45.13 | 129.50 |
| 423.15 | 73.07 | 56.31 | 128.29 | 72.51 | 45.96 | 115.46 | 85.25 | 40.62 | 117.70 |
| 433.15 | 78.33 | 57.00 | 133.64 | 70.27 | 43.74 | 110.89 | 83.23 | 40.53 | 116.15 |
| 443.15 | 56.38 | 48.50 | 104.59 | 62.22 | 38.64 | 98.07 | 94.44 | 48.59 | 135.48 |
| 453.15 | 45.40 | 43.84 | 89.22 | 60.93 | 37.02 | 94.99 | 73.62 | 36.11 | 103.13 |
| 463.15 | 39.51 | 40.86 | 80.36 | 62.44 | 36.80 | 95.87 | 71.77 | 36.02 | 101.68 |
| 473.15 | 30.81 | 36.16 | 66.75 | 56.99 | 33.15 | 86.93 | 63.57 | 31.50 | 89.50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




