Submitted:
21 March 2024
Posted:
26 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
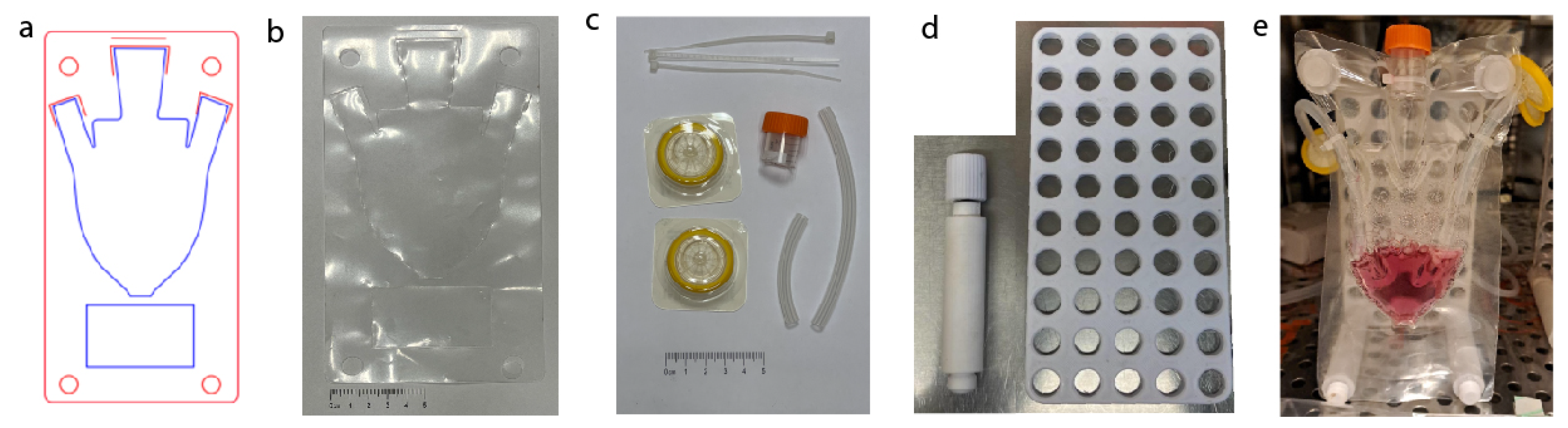
2.1. Fabrication of Macrofluidic Devices
2.1.1. Laser Fabrication of Macrofluidic Devices from Thermoplastic Film
2.1.2. Fabrication Back Board
2.1.3. Air Pump
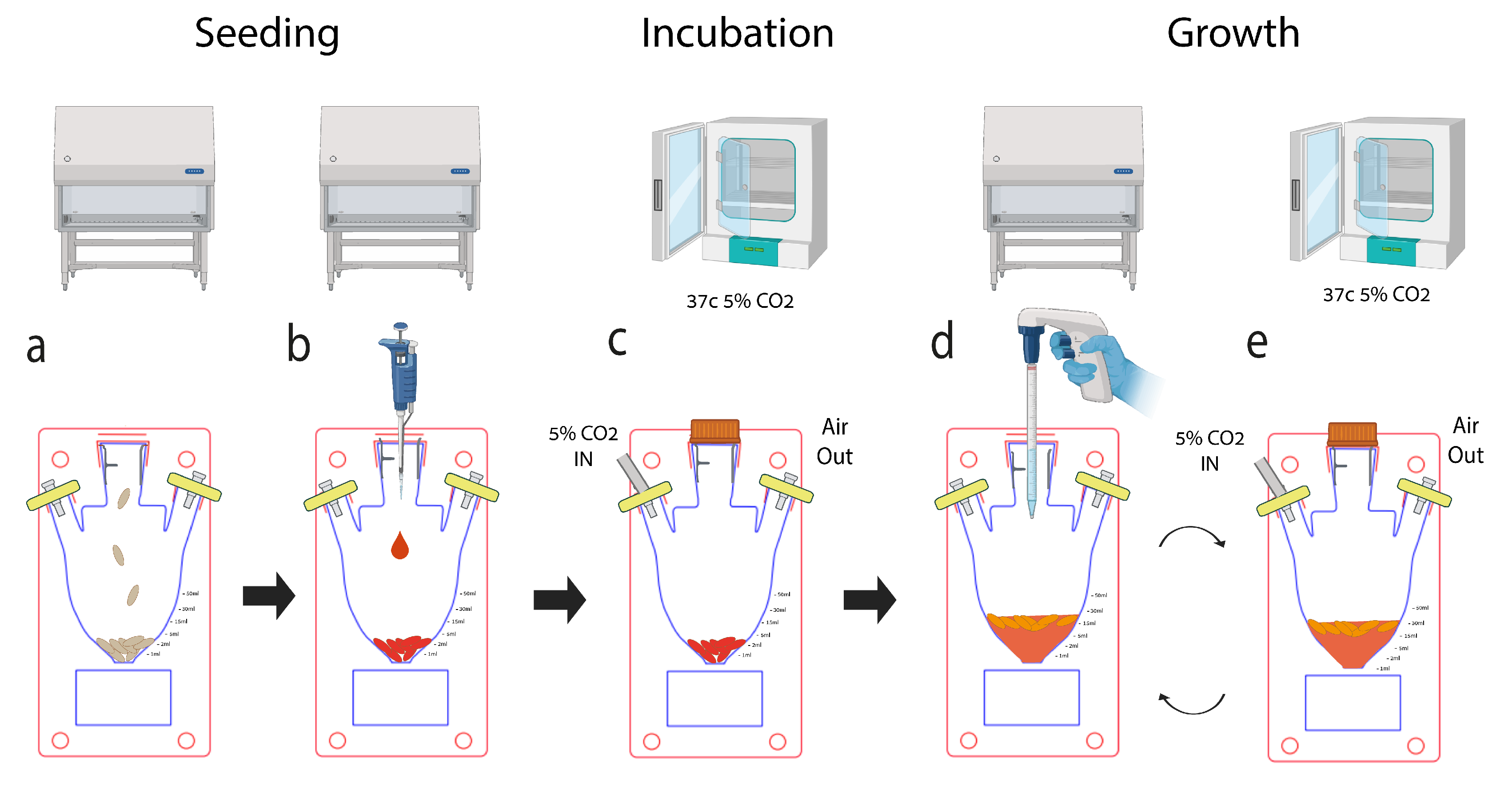
2.1.4. Macrofluidic Prototyping for Plant Based Scaffolds
2.1.5. Cell Culture and Immortalization
2.1.6. Experimental Setup
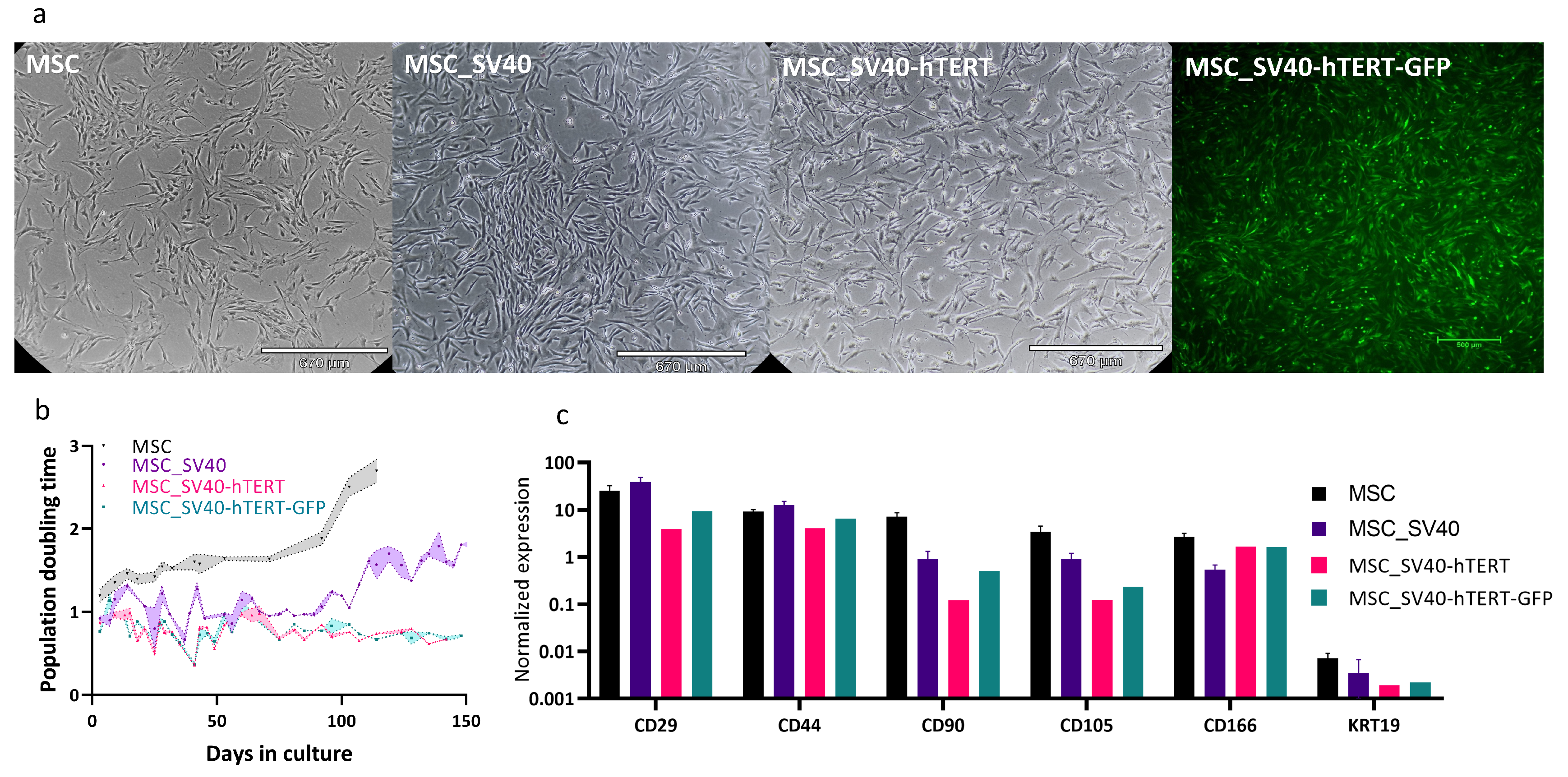
2.1.7. Expression of MSC Surface Markers
2.1.8. Microscopy
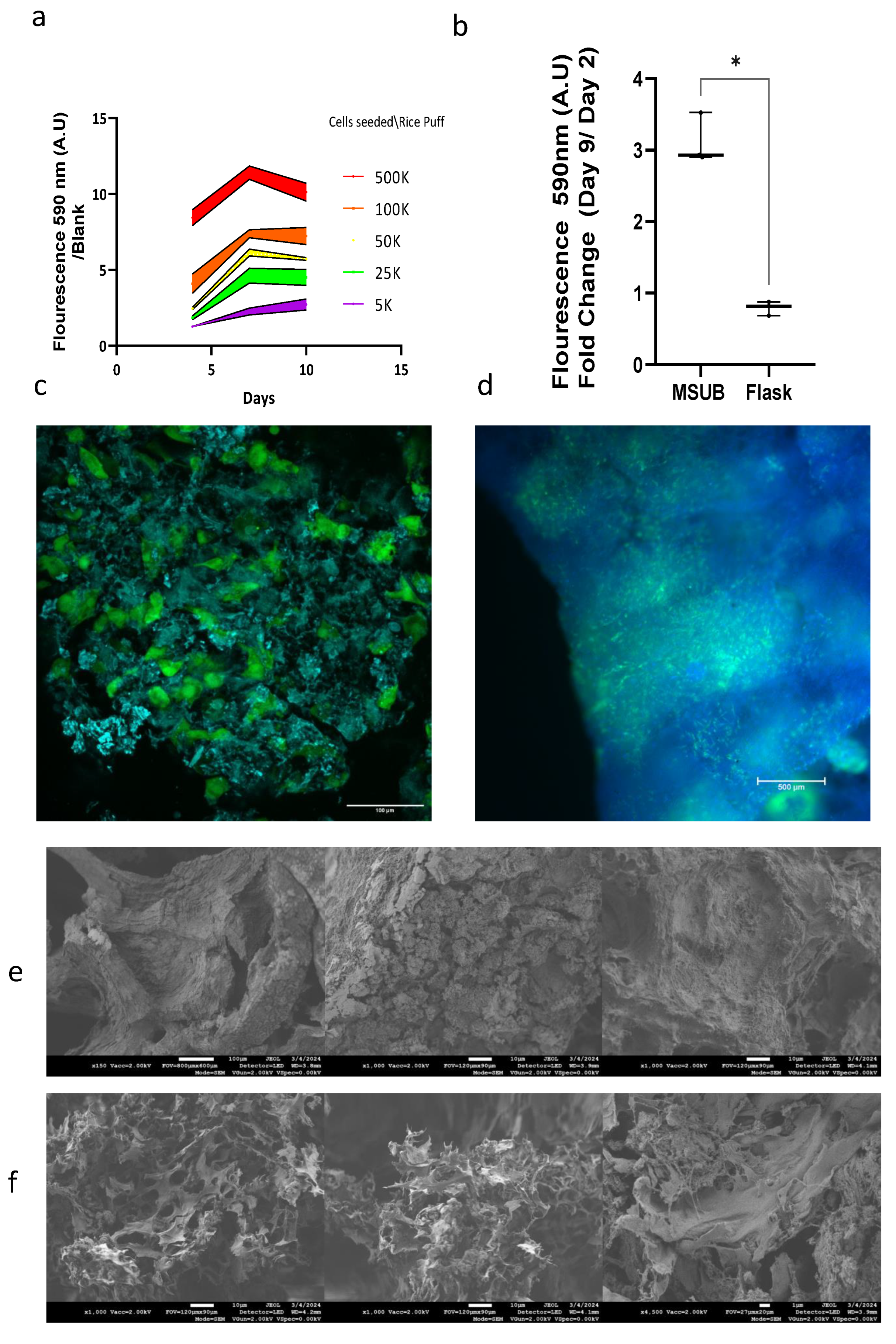
3. Results
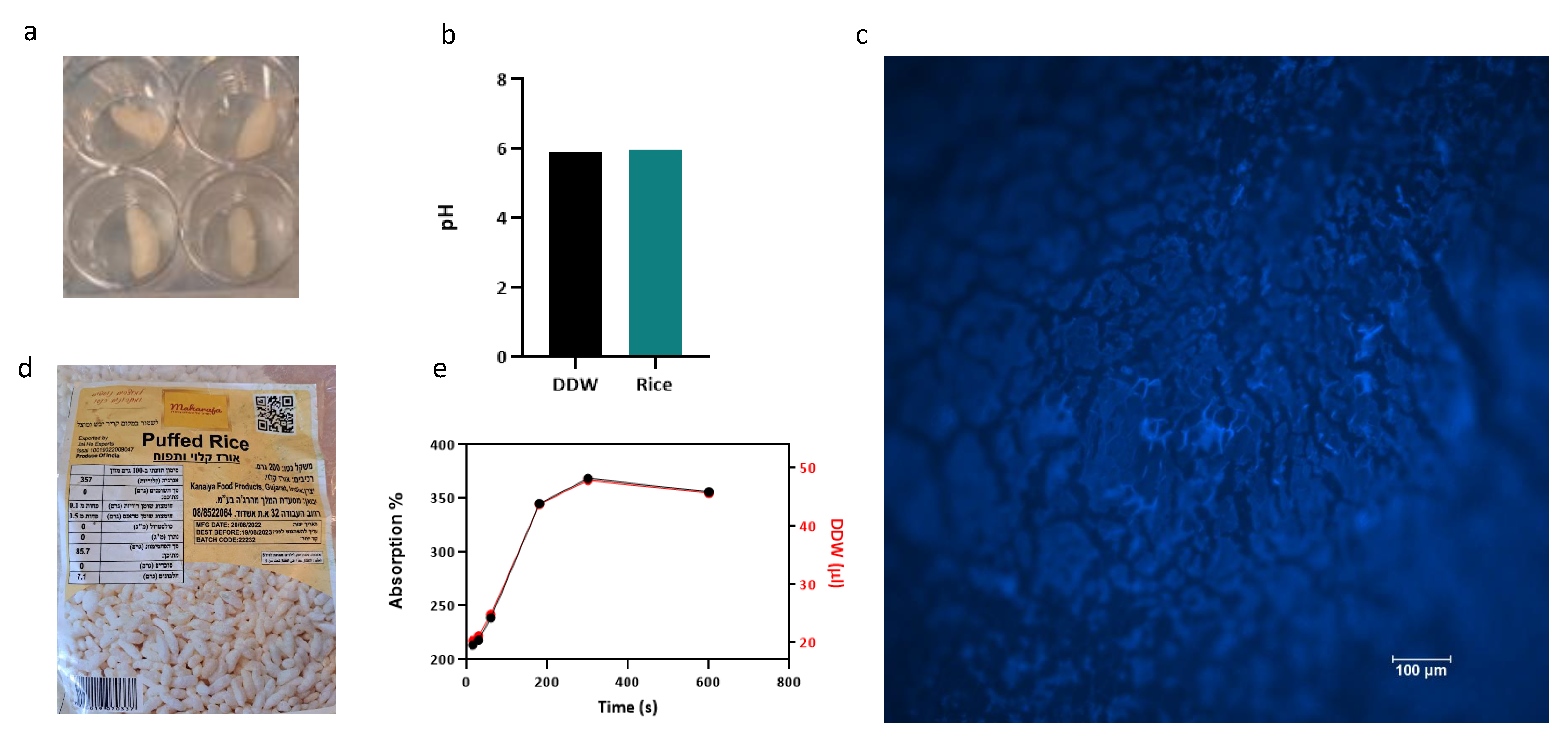
3.1. Rice-Based Scaffold Microscopy, Water Absorption and pH Assay
3.2. Scanning Electron Microscopy (SEM)
3.3. Cell Line Properties in 2D
3.3.1. Proliferation on Assay in Wells and in MSUB
4. Discussion
5. Conclusions
6. Patents
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PAPE | Polyethylene-Polyamide |
| MSUB | Macrofluidic Single-use Bioreactor |
References
- Levi, S.; Yen, F.C.; Baruch, L.; Machluf, M. Scaffolding technologies for the engineering of cultured meat: Towards a safe, sustainable, and scalable production. Trends in Food Science & Technology 2022, 126, 13–25. [Google Scholar]
- Lee, S.H.; Choi, J. Three-dimensional scaffolds, materials, and fabrication for cultured meat applications: A scoping review and future direction. Food Hydrocolloids, 2024; 109881. [Google Scholar]
- Bomkamp, C.; Skaalure, S.C.; Fernando, G.F.; Ben-Arye, T.; Swartz, E.W.; Specht, E.A. Scaffolding biomaterials for 3D cultivated meat: prospects and challenges. Advanced Science 2022, 9, 2102908. [Google Scholar] [CrossRef]
- Seah, J.S.H.; Singh, S.; Tan, L.P.; Choudhury, D. Scaffolds for the manufacture of cultured meat. Critical Reviews in Biotechnology 2022, 42, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Sood, A.; Han, S.S. Technological and structural aspects of scaffold manufacturing for cultured meat: recent advances, challenges, and opportunities. Critical Reviews in Food Science and Nutrition 2023, 63, 585–612. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, X.; Li, X.; Du, G.; Zhou, J.; Chen, J. Challenges and possibilities for bio-manufacturing cultured meat. Trends in Food Science & Technology 2020, 97, 443–450. [Google Scholar]
- Modulevsky, D.J.; Lefebvre, C.; Haase, K.; Al-Rekabi, Z.; Pelling, A.E. Apple derived cellulose scaffolds for 3D mammalian cell culture. PloS one 2014, 9, e97835. [Google Scholar] [CrossRef] [PubMed]
- Ben-Arye, T.; Shandalov, Y.; Ben-Shaul, S.; Landau, S.; Zagury, Y.; Ianovici, I.; Lavon, N.; Levenberg, S. Textured soy protein scaffolds enable the generation of three-dimensional bovine skeletal muscle tissue for cell-based meat. Nature Food 2020, 1, 210–220. [Google Scholar] [CrossRef]
- Xiang, N.; Yuen Jr, J.S.; Stout, A.J.; Rubio, N.R.; Chen, Y.; Kaplan, D.L. 3D porous scaffolds from wheat glutenin for cultured meat applications. Biomaterials 2022, 285, 121543. [Google Scholar] [CrossRef] [PubMed]
- Bar-Shai, N.; Sharabani-Yosef, O.; Zollmann, M.; Lesman, A.; Golberg, A. Seaweed cellulose scaffolds derived from green macroalgae for tissue engineering. Scientific reports 2021, 11, 11843. [Google Scholar] [CrossRef]
- Park, S.; Lee, M.; Jung, S.; Lee, H.; Choi, B.; Choi, M.; Lee, J.M.; Yoo, K.H.; Han, D.; Lee, S.T. ; others. Rice grains integrated with animal cells: A shortcut to a sustainable food system. Matter, 2024. [Google Scholar]
- De Wilde, D.; Dreher, T.; Zahnow, C.; Husemann, U.; Greller, G.; Adams, T.; Fenge, C. Superior scalability of single-use bioreactors. Innovations in Cell Culture 2014, 14, 14–19. [Google Scholar]
- Kurt, T.; Höing, T.; Oosterhuis, N. The Potential Application of Single-Use Bioreactors in Cultured Meat Production. Chemie Ingenieur Technik 2022, 94, 2026–2030. [Google Scholar] [CrossRef]
- Bodiou, V.; Moutsatsou, P.; Post, M.J. Microcarriers for upscaling cultured meat production. Frontiers in nutrition 2020, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Roy, A. Selecting high amylose rice variety for puffing: A correlation between physicochemical parameters and sensory preferences. Measurement: Food 2022, 5, 100021. [Google Scholar] [CrossRef]
- Jia, L.; Huang, R.; Wang, S.; Dong, Y.; Lv, J.; Zhong, W.; Yan, F. Effects of Explosion Puffing on the Composition, Structure, and Functional Characteristics of Starch and Protein in Grains. ACS Food Science & Technology 2021, 1, 1869–1879. [Google Scholar]
- Bagchi, T.; Chattopadhyay, K.; Sarkar, S.; Sanghamitra, P.; Kumar, A.; Basak, N.; Sivashankari, M.; Priyadarsini, S.; Pathak, H. Rice Products and their Nutritional Status. Research Bulletin 2020. [Google Scholar]
- Pashkuleva, I.; López-Pérez, P.M.; Azevedo, H.S.; Reis, R.L. Highly porous and interconnected starch-based scaffolds: production, characterization and surface modification. Materials Science and Engineering: C 2010, 30, 981–989. [Google Scholar] [CrossRef]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Tian, L.; Shamirzaei-Jeshvaghani, E.; Dehghani, L.; Ramakrishna, S. Structural properties of scaffolds: crucial parameters towards stem cells differentiation. World journal of stem cells 2015, 7, 728. [Google Scholar] [CrossRef] [PubMed]
- Okano, T.; Yamada, N.; Okuhara, M.; Sakai, H.; Sakurai, Y. Mechanism of cell detachment from temperature-modulated, hydrophilic-hydrophobic polymer surfaces. Biomaterials 1995, 16, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H.; Khil, M.S.; Kim, H.Y.; Lee, H.U.; Jahng, K.Y. An improved hydrophilicity via electrospinning for enhanced cell attachment and proliferation. Journal of Biomedical Materials Research Part B: Applied Biomaterials: An Official Journal of The Society for Biomaterials, The Japanese Society for Biomaterials, and The Australian Society for Biomaterials and the Korean Society for Biomaterials 2006, 78, 283–290. [Google Scholar] [CrossRef]
- Horbett, T.A.; Schway, M.B.; Ratner, B.D. Hydrophilic-hydrophobic copolymers as cell substrates: Effect on 3T3 cell growth rates. Journal of colloid and interface science 1985, 104, 28–39. [Google Scholar] [CrossRef]
- Sbardelotto, P.R.; Balbinot-Alfaro, E.; da Rocha, M.; Alfaro, A.T. Natural alternatives for processed meat: Legislation, markets, consumers, opportunities and challenges. Critical Reviews in Food Science and Nutrition 2023, 63, 10303–10318. [Google Scholar] [CrossRef] [PubMed]
- India, E. Puffed Rice Suppliers in India. https://www.exportersindia.com/indian-suppliers/puffed-rice.htm, year = 2024. Date Accessed: February 11, 2024.
- IndiaMART. Puffed Rice Suppliers on IndiaMART. https://dir.indiamart.com/impcat/puffed-rice.htm, year = 2024. Date Accessed: February 11, 2024.
- Chen, L.; Guttieres, D.; Koenigsberg, A.; Barone, P.W.; Sinskey, A.J.; Springs, S.L. Large-scale cultured meat production: Trends, challenges and promising biomanufacturing technologies. Biomaterials 2022, 280, 121274. [Google Scholar] [CrossRef] [PubMed]
- Djisalov, M.; Knežić, T.; Podunavac, I.; Živojević, K.; Radonic, V.; Knežević, N.Ž.; Bobrinetskiy, I.; Gadjanski, I. Cultivating multidisciplinarity: Manufacturing and sensing challenges in cultured meat production. Biology 2021, 10, 204. [Google Scholar] [CrossRef]
- Garrison, G.L.; Biermacher, J.T.; Brorsen, B.W. How much will large-scale production of cell-cultured meat cost? Journal of Agriculture and Food Research 2022, 10, 100358. [Google Scholar] [CrossRef]
- Tuomisto, H.L.; Allan, S.J.; Ellis, M.J. Prospective life cycle assessment of a bioprocess design for cultured meat production in hollow fiber bioreactors. Science of the Total Environment 2022, 851, 158051. [Google Scholar] [CrossRef] [PubMed]
- Ge, C.; Selvaganapathy, P.R.; Geng, F. Advancing our understanding of bioreactors for industrial-sized cell culture: health care and cellular agriculture implications. American Journal of Physiology-Cell Physiology 2023, 325, C580–C591. [Google Scholar] [CrossRef]
- Allan, S.J.; De Bank, P.A.; Ellis, M.J. Bioprocess design considerations for cultured meat production with a focus on the expansion bioreactor. Frontiers in Sustainable Food Systems 2019, 3, 44. [Google Scholar] [CrossRef]
- Thangadurai, M.; Srinivasan, S.S.; Sekar, M.P.; Sethuraman, S.; Sundaramurthi, D. Emerging perspectives on 3D printed bioreactors for clinical translation of engineered and bioprinted tissue constructs. Journal of Materials Chemistry B 2023. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, H.K.; Doeven, E.H.; Merenda, A.; Dumée, L.F.; Guijt, R.M. 3D printing for the integration of porous materials into miniaturised fluidic devices: A review. Analytica chimica acta 2021, 1185, 338796. [Google Scholar] [CrossRef]
- Bader, C.; Patrick, W.G.; Kolb, D.; Hays, S.G.; Keating, S.; Sharma, S.; Dikovsky, D.; Belocon, B.; Weaver, J.C.; Silver, P.A.; others. Grown, printed, and biologically augmented: An additively manufactured microfluidic wearable, functionally templated for synthetic microbes. 3D Printing and Additive Manufacturing 2016, 3, 79–89. [Google Scholar] [CrossRef]
- Priyadarshini, B.M.; Dikshit, V.; Zhang, Y. 3D-printed bioreactors for in vitro modeling and analysis. International Journal of Bioprinting 2020, 6. [Google Scholar] [CrossRef]
- Merkel, M.; Noll, P.; Lilge, L.; Hausmann, R.; Henkel, M. Design and evaluation of a 3D-printed, lab-scale perfusion bioreactor for novel biotechnological applications. Biotechnology Journal 2023, 18, 2200554. [Google Scholar] [CrossRef] [PubMed]
- Linz, G.; Rauer, S.B.; Kuhn, Y.; Wennemaring, S.; Siedler, L.; Singh, S.; Wessling, M. 3D-Printed Bioreactor with Integrated Impedance Spectroscopy for Cell Barrier Monitoring. Advanced Materials Technologies 2021, 6, 2100009. [Google Scholar] [CrossRef]
- Ng, S.; Kurisawa, M. Integrating biomaterials and food biopolymers for cultured meat production. Acta Biomaterialia 2021, 124, 108–129. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Wu, J.Y.; Kennedy, K.M.; Yeager, K.; Bernhard, J.C.; Ng, J.J.; Zimmerman, B.K.; Robinson, S.; Durney, K.M.; Shaeffer, C.; others. Tissue engineered autologous cartilage-bone grafts for temporomandibular joint regeneration. Science translational medicine 2020, 12, eabb6683. [Google Scholar] [CrossRef] [PubMed]
- Borisov, V.; Gili Sole, L.; Reid, G.; Milan, G.; Hutter, G.; Grapow, M.; Eckstein, F.S.; Isu, G.; Marsano, A. Upscaled Skeletal Muscle Engineered Tissue with In Vivo Vascularization and Innervation Potential. Bioengineering 2023, 10, 800. [Google Scholar] [CrossRef] [PubMed]
- Samandari, M.; Saeedinejad, F.; Quint, J.; Chuah, S.X.Y.; Farzad, R.; Tamayol, A. Repurposing biomedical muscle tissue engineering for cellular agriculture: challenges and opportunities. Trends in Biotechnology 2023. [Google Scholar] [CrossRef] [PubMed]
- Humbird, D. Scale-up economics for cultured meat. Biotechnology and Bioengineering 2021, 118, 3239–3250. [Google Scholar] [CrossRef]
- Tsao, C.W. Polymer microfluidics: Simple, low-cost fabrication process bridging academic lab research to commercialized production. Micromachines 2016, 7, 225. [Google Scholar] [CrossRef]
- Lu, Q.; Ou, J.; Wilbert, J.; Haben, A.; Mi, H.; Ishii, H. milliMorph–Fluid-Driven Thin Film Shape-Change Materials for Interaction Design. In Proceedings of the 32nd Annual ACM Symposium on User Interface Software and Technology; 2019; pp. 663–672. [Google Scholar]
- Abdelgawad, M.; Wheeler, A.R. Low-cost, rapid-prototyping of digital microfluidics devices. Microfluidics and nanofluidics 2008, 4, 349–355. [Google Scholar] [CrossRef]
- Perrone, E.; Cesaria, M.; Zizzari, A.; Bianco, M.; Ferrara, F.; Raia, L.; Guarino, V.; Cuscunà, M.; Mazzeo, M.; Gigli, G.; others. Potential of CO2-laser processing of quartz for fast prototyping of microfluidic reactors and templates for 3D cell assembly over large scale. Materials Today Bio 2021, 12, 100163. [Google Scholar] [CrossRef] [PubMed]
- Saadat, M.; Taylor, M.; Hughes, A.; Hajiyavand, A.M. Rapid prototyping method for 3D PDMS microfluidic devices using a red femtosecond laser. Advances in Mechanical Engineering 2020, 12, 1687814020982713. [Google Scholar] [CrossRef]
- Hosic, S.; Bindas, A.J.; Puzan, M.L.; Lake, W.; Soucy, J.R.; Zhou, F.; Koppes, R.A.; Breault, D.T.; Murthy, S.K.; Koppes, A.N. Rapid prototyping of multilayer microphysiological systems. ACS biomaterials science & engineering 2020, 7, 2949–2963. [Google Scholar]
- Hanga, M.P.; Ali, J.; Moutsatsou, P.; de la Raga, F.A.; Hewitt, C.J.; Nienow, A.; Wall, I. Bioprocess development for scalable production of cultivated meat. Biotechnology and Bioengineering 2020, 117, 3029–3039. [Google Scholar] [CrossRef] [PubMed]
- Kirsch, M.; Morales-Dalmau, J.; Lavrentieva, A. Cultivated meat manufacturing: Technology, trends, and challenges. Engineering in Life Sciences 2023, 23, e2300227. [Google Scholar] [CrossRef]
- Klebe, R.J.; Lyn, S.; Magnuson, V.L.; Zardeneta, G. Cultivation of mammalian cells in heat-sealable pouches that are permeable to carbon dioxide. Experimental Cell Research 1990, 188, 316–319. [Google Scholar] [CrossRef]
- Shaegh, S.A.M.; Pourmand, A.; Nabavinia, M.; Avci, H.; Tamayol, A.; Mostafalu, P.; Ghavifekr, H.B.; Aghdam, E.N.; Dokmeci, M.R.; Khademhosseini, A.; others. Rapid prototyping of whole-thermoplastic microfluidics with built-in microvalves using laser ablation and thermal fusion bonding. Sensors and Actuators B: Chemical 2018, 255, 100–109. [Google Scholar] [CrossRef]
- Truckenmüller, R.; Giselbrecht, S.; van Blitterswijk, C.; Dambrowsky, N.; Gottwald, E.; Mappes, T.; Rolletschek, A.; Saile, V.; Trautmann, C.; Weibezahn, K.F.; others. Flexible fluidic microchips based on thermoformed and locally modified thin polymer films. Lab on a Chip 2008, 8, 1570–1579. [Google Scholar] [CrossRef]
- Lee, J.; Kim, H.; Lim, H.R.; Kim, Y.S.; Hoang, T.T.T.; Choi, J.; Jeong, G.J.; Kim, H.; Herbert, R.; Soltis, I.; others. Large-scale smart bioreactor with fully integrated wireless multivariate sensors and electronics for long-term in situ monitoring of stem cell culture. Science Advances 2024, 10, eadk6714. [Google Scholar] [CrossRef]
- Zhao, J.; Griffin, M.; Cai, J.; Li, S.; Bulter, P.E.; Kalaskar, D.M. Bioreactors for tissue engineering: An update. Biochemical Engineering Journal 2016, 109, 268–281. [Google Scholar] [CrossRef]
- Suryawanshi, P.L.; Gumfekar, S.P.; Bhanvase, B.A.; Sonawane, S.H.; Pimplapure, M.S. A review on microreactors: Reactor fabrication, design, and cutting-edge applications. Chemical Engineering Science 2018, 189, 431–448. [Google Scholar] [CrossRef]
- Gome, G.; Fein, Y.; Waksberg, J.; Maayan, Y.; Grishko, A.; Wald, I.Y.; Zuckerman, O. My First Biolab: a System for Hands-On Biology Experiments. Extended Abstracts of the 2019 CHI Conference on Human Factors in Computing Systems, 2019, pp. 1–6.
- Baksh, D.; Song, L.; Tuan, R.S. Adult mesenchymal stem cells: characterization, differentiation, and application in cell and gene therapy. Journal of cellular and molecular medicine 2004, 8, 301–316. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.B.T.; Bressan, F.F.; Murphy, B.D.; Garcia, J.M. Applications of mesenchymal stem cell technology in bovine species. Stem Cell Research & Therapy 2019, 10, 1–13. [Google Scholar]
- Shimoni, C.; Goldstein, M.; Ribarski-Chorev, I.; Schauten, I.; Nir, D.; Strauss, C.; Schlesinger, S. Heat shock alters mesenchymal stem cell identity and induces premature senescence. Frontiers in Cell and Developmental Biology, 2020; 956. [Google Scholar]
- Addgene. Human telomerase gene hTERT.
- Wongsa, J.; Uttapap, D.; Lamsal, B.P.; Rungsardthong, V. Effect of puffing conditions on physical properties and rehydration characteristic of instant rice product. International Journal of Food Science & Technology 2016, 51, 672–680. [Google Scholar]
- Shinde, R.; Vinokur, Y.; Fallik, E.; Rodov, V. Effects of Genotype and Modified Atmosphere Packaging on the Quality of Fresh-Cut Melons. Foods 2024, 13, 256. [Google Scholar] [CrossRef]
- Huang, H.D.; Ren, P.G.; Zhong, G.J.; Olah, A.; Li, Z.M.; Baer, E.; Zhu, L. Promising strategies and new opportunities for high barrier polymer packaging films. Progress in Polymer Science, 2023; 101722. [Google Scholar]
- Horsthuis, E. The future of plastic packaging for the fresh food industry. Master’s thesis, University of Twente, 2023.
- Caldwell, D.G. Automation in Food Manufacturing and Processing. In Springer Handbook of Automation; Springer, 2023; pp. 949–971.



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



