Submitted:
16 April 2025
Posted:
21 April 2025
You are already at the latest version
Abstract
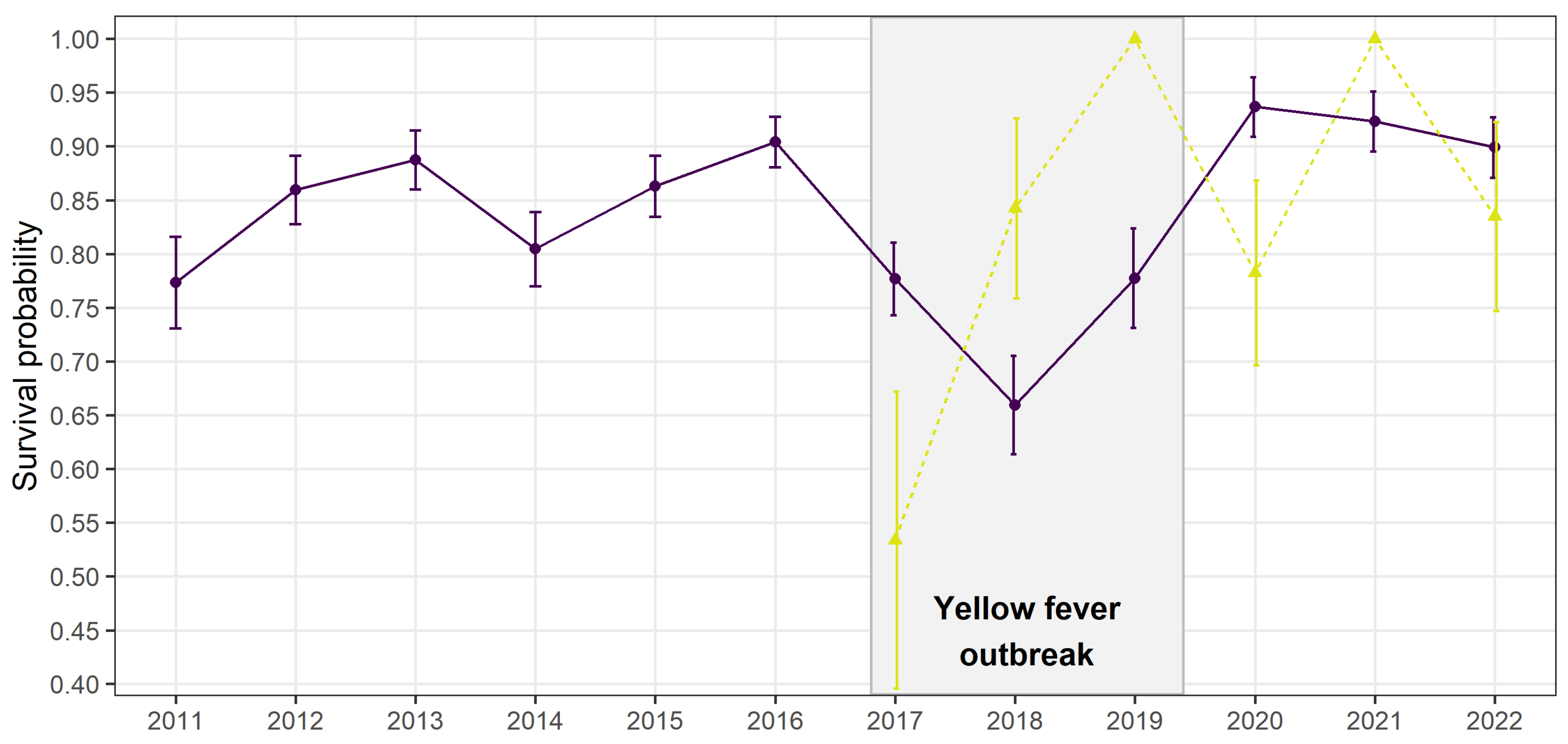
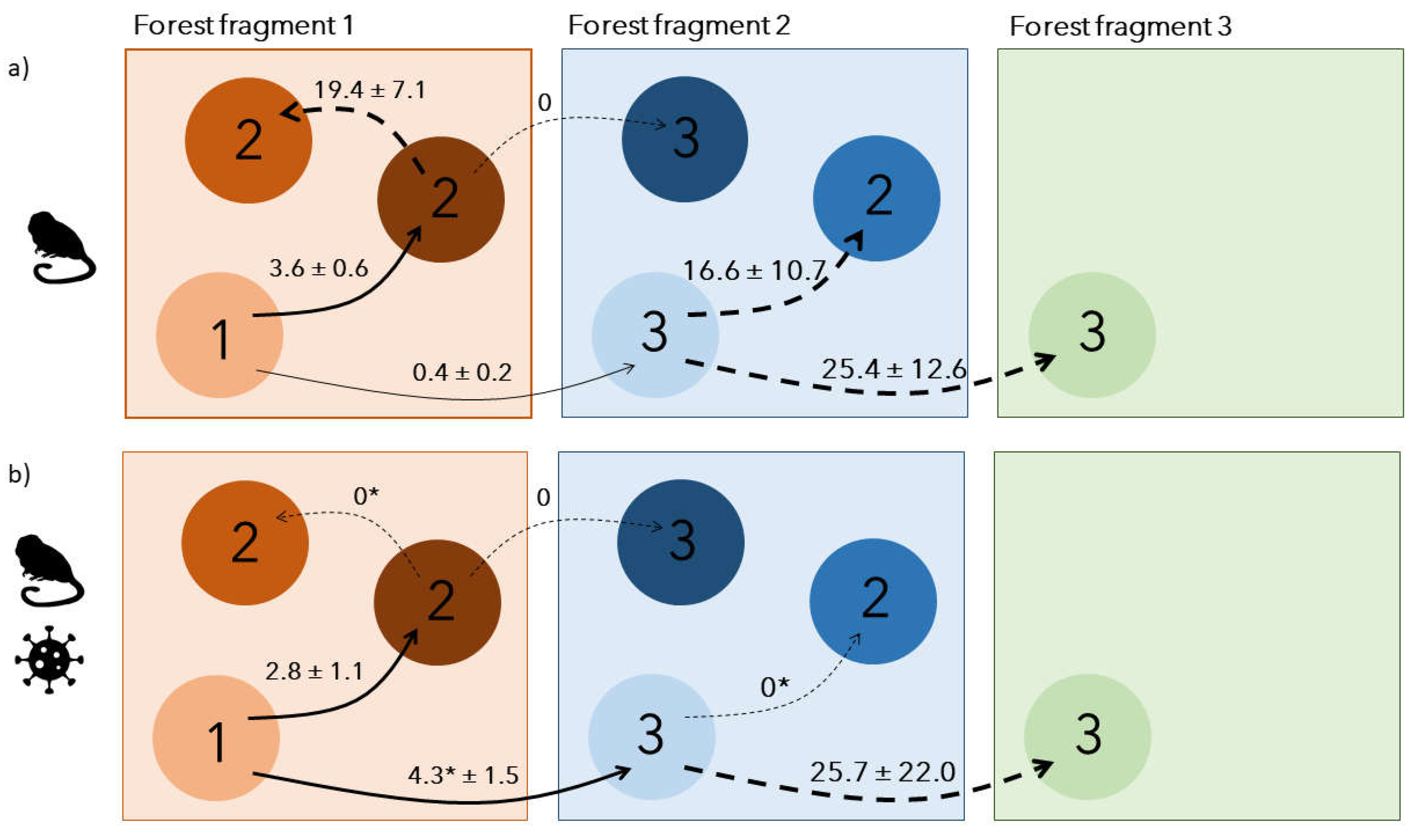
Disease outbreaks can severely affect populations in the wild. However, their consequences on individual social behaviour and population demography are poorly understood. We used a multi-site capture-recapture model to investigate the impacts of a yellow fever outbreak on the endangered golden lion tamarin Leontopithecus rosalia, in the highly fragmented Atlantic forest, Brazil. Annual adult survival rate severely declined in 2017-2018, coinciding with the outbreak period. Simultaneously, dispersal patterns changed temporarily, with a reduction of settlement time for individuals dispersing within the same forest fragments, and a significant increase of dispersal between forest fragments, from 0.4±0.2 to 4.3±1.5%. Our results indicate a spatial rearrangement of individuals during the outbreak potentially due to non-random mortality leading to changes in social group structure at a local and regional scale. They advocate for a better integration of host movements, host social behaviour and habitat connectivity when evaluating species response to infectious diseases.
Keywords:
Introduction
Material and Methods
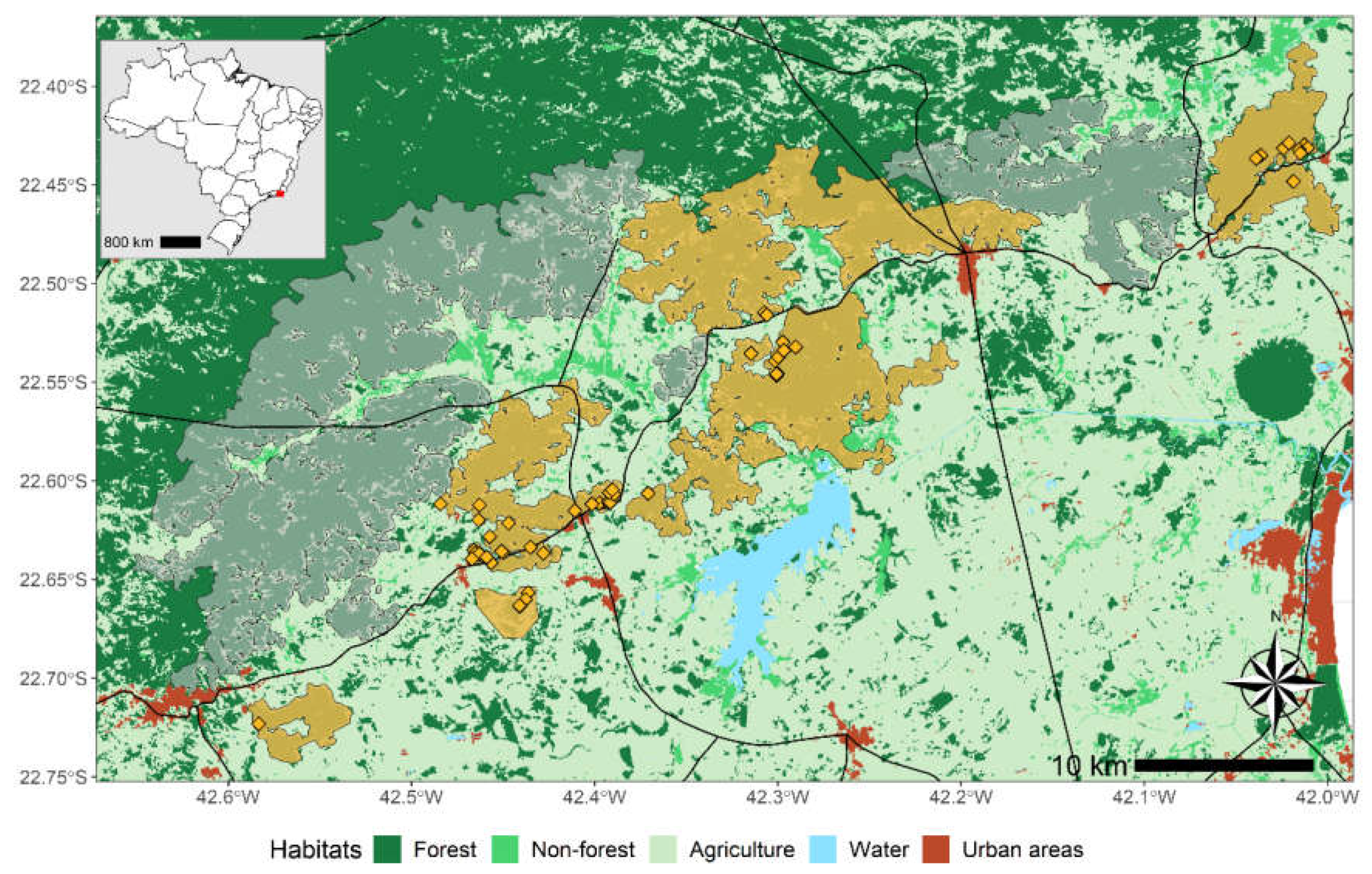
Study Area and Monitoring Survey
Data Selection
Capture-Recapture Analysis
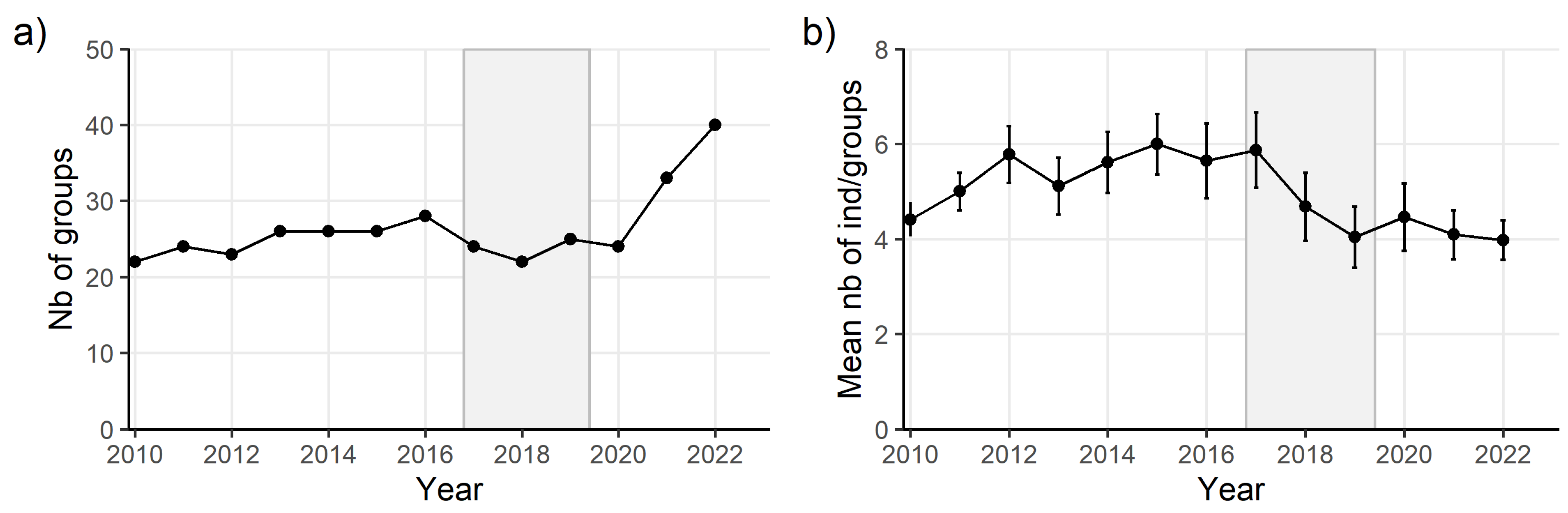
Results
Discussion
Supplementary Materials
References
- Abreu, F. V., Ferreira-de-Brito, A., Azevedo, A. D., Linhares, J. H., de Oliveira Santos, V., Hime Miranda, E., Neves, M. S., Yousfi, L., Ribeiro, I. P., Santos, A. A., dos Santos, E., Santos, T. P., Teixeira, D. S., Gomes, M. Q., Fernandes, C. B., Silva, A. M., Lima, M. D., Paupy, C., Romano, A. P., … Lourenço-de-Oliveira, R. (2020). Survey on Non-Human Primates and Mosquitoes Does not Provide Evidences of Spillover/Spillback between the Urban and Sylvatic Cycles of Yellow Fever and Zika Viruses Following Severe Outbreaks in Southeast Brazil. Viruses, 12(4). [CrossRef]
- Arce-Peña, N. P., Arroyo-Rodríguez, V., Dias, P. A. D., Franch-Pardo, I., & Andresen, E. (2019). Linking changes in landscape structure to population changes of an endangered primate. Landscape Ecology, 34(11), Article 11. [CrossRef]
- Arnold, T. W. (2010). Uninformative Parameters and Model Selection Using Akaike’s Information Criterion. The Journal of Wildlife Management, 74(6), 1175–1178. [CrossRef]
- Baker, A. J., & Dietz, J. M. (1996). Immigration in wild groups of golden lion tamarins (Leontopithecus rosalia). American Journal of Primatology, 38(1), Article 1. [CrossRef]
- Bermejo, M., Rodríguez-Teijeiro, J. D., Illera, G., Barroso, A., Vilà, C., & Walsh, P. D. (2006). Ebola Outbreak Killed 5000 Gorillas. Science, 314(5805), 1564–1564. [CrossRef]
- Berthet, M., Mesbahi, G., Duvot, G., Zuberbühler, K., Cäsar, C., & Bicca-Marques, J. C. (2021). Dramatic decline in a titi monkey population after the 2016–2018 sylvatic yellow fever outbreak in Brazil. American Journal of Primatology, 83(12), e23335. [CrossRef]
- Bicca-Marques, J. C., & de Freitas, D. S. (2010). The Role of Monkeys, Mosquitoes, and Humans in the Occurrence of a Yellow Fever Outbreak in a Fragmented Landscape in South Brazil: Protecting Howler Monkeys is a Matter of Public Health. Tropical Conservation Science, 3(1), 78–89. [CrossRef]
- Binning, S. A., Shaw, A. K., & Roche, D. G. (2017). Parasites and Host Performance: Incorporating Infection into Our Understanding of Animal Movement. Integrative and Comparative Biology, 57(2), 267–280. [CrossRef]
- Boulinier, T., Kada, S., Ponchon, A., Dupraz, M., Dietrich, M., Gamble, A., Bourret, V., Duriez, O., Bazire, R., Tornos, J., Tveraa, T., Chambert, T., Garnier, R., & McCoy, K. D. (2016). Migration, Prospecting, Dispersal? What Host Movement Matters for Infectious Agent Circulation? Integrative and Comparative Biology, 56, 330–342. [CrossRef]
- Burnham, K. P., & Anderson, D. R. (2002). Model selection and multimodel inference: A practical information-theoretic approach (2nd Edition). Springer.
- Caillaud, D., Levréro, F., Cristescu, R., Gatti, S., Dewas, M., Douadi, M., Gautier-Hion, A., Raymond, M., & Ménard, N. (2006). Gorilla susceptibility to Ebola virus: The cost of sociality. Current Biology, 16(13), R489-91. PubMed. [CrossRef]
- Chala, B., & Hamde, F. (2021). Emerging and Re-emerging Vector-Borne Infectious Diseases and the Challenges for Control: A Review. Frontiers in Public Health, 9. https://www.frontiersin.org/journals/public-health/articles/10.3389/fpubh.2021.715759.
- Choquet, R., Lebreton, J.-D., Gimenez, O., Reboulet, A.-M., & Pradel, R. (2009). U-CARE: Utilities for performing goodness of fit tests and manipulating CApture–REcapture data. Ecography, 32(6), 1071–1074. [CrossRef]
- Choquet, R., Rouan, L., & Pradel, R. (2009). Program E-Surge: Software application for fitting multievent models. In D. Thomson, E. Cooch, & M. Conroy (Eds.), Modeling demographic processes in marked populations (Vol. 3, pp. 845–865). Springer US.
- Coelho Couto de Azevedo Fernandes, N., Sequetin Cuhna, M., Mariotti Guerra, J., Albergaria Réssio, R., dos Santos Cirqueira, C., D’Andretta Iglezias, S., de Carvalho, J., Araujo, E. L. L., Catão-Dias, J. L., & Dias-Delagdo, J. (2017). Outbreak of Yellow Fever among Nonhuman Primates, Espirito Santo, Brazil, 2017. Emerging Infectious Disease, 2038–2041.
- Dekelaita, D. J., Epps, C. W., German, D. W., Powers, J. G., Gonzales, B. J., Abella-Vu, R. K., Darby, N. W., Hughson, D. L., & Stewart, K. M. (2023). Animal movement and associated infectious disease risk in a metapopulation. Royal Society Open Science, 10(2), 220390. [CrossRef]
- Dietz, J. M., Hankerson, S. J., Alexandre, B. R., Henry, M. D., Martins, A. F., Ferraz, L. P., & Ruiz-Miranda, C. R. (2019). Yellow fever in Brazil threatens successful recovery of endangered golden lion tamarins. Scientific Reports, 9(1), 12926. [CrossRef]
- Dietz, J. M., Mickelberg, J., Traylor-Holzer, K., Martins, A. F., Souza, M. N., & Hankerson, S. J. (2024). Golden lion tamarin metapopulation dynamics five years after heavy losses to yellow fever. American Journal of Primatology, 86(7), e23635. [CrossRef]
- Dietz, J. M., Peres, C. A., & Pinder, L. (1997). Foraging ecology and use of space in wild golden lion tamarins (Leontopithecus rosalia). American Journal of Primatology, 41(4), Article 4. [CrossRef]
- Dougherty, E. R., Seidel, D. P., Carlson, C. J., Spiegel, O., & Getz, W. M. (2018). Going through the motions: Incorporating movement analyses into disease research. Ecology Letters, 21(4), 588–604. [CrossRef]
- Duriez, O., Sassi, Y., Le Gall-Ladevèze, C., Giraud, L., Straughan, R., Dauverné, L., Terras, A., Boulinier, T., Choquet, R., Van De Wiele, A., Hirschinger, J., Guérin, J.-L., & Le Loc’h, G. (2023). Highly pathogenic avian influenza affects vultures’ movements and breeding output. Current Biology, 33(17), 3766-3774.e3. [CrossRef]
- Estrada, A., Garber, P. A., Rylands, A. B., Roos, C., Fernandez-Duque, E., Di Fiore, A., Nekaris, K. A.-I., Nijman, V., Heymann, E. W., Lambert, J. E., Rovero, F., Barelli, C., Setchell, J. M., Gillespie, T. R., Mittermeier, R. A., Arregoitia, L. V., de Guinea, M., Gouveia, S., Dobrovolski, R., … Li, B. (2017). Impending extinction crisis of the world’s primates: Why primates matter. Science Advances, 3(1), e1600946. [CrossRef]
- Gaitan, J., & Millien, V. (2016). Stress level, parasite load, and movement pattern in a small-mammal reservoir host for Lyme disease. Canadian Journal of Zoology, 94(8), 565–573. [CrossRef]
- Genton, C., Pierre, A., Cristescu, R., Lévréro, F., Gatti, S., Pierre, J.-S., Ménard, N., & Le Gouar, P. (2015). How Ebola impacts social dynamics in gorillas: A multistate modelling approach. Journal of Animal Ecology, 84(1), 166–176. [CrossRef]
- Giovanetti Marta, de Mendonça Marcos Cesar Lima, Fonseca Vagner, Mares-Guia Maria Angélica, Fabri Allison, Xavier Joilson, de Jesus Jaqueline Goes, Gräf Tiago, dos Santos Rodrigues Cintia Damasceno, dos Santos Carolina Cardoso, Sampaio Simone Alves, Chalhoub Flavia Lowen Levy, de Bruycker Nogueira Fernanda, Theze Julien, Romano Alessandro Pecego Martins, Ramos Daniel Garkauskas, de Abreu Andre Luiz, Oliveira Wanderson Kleber, do Carmo Said Rodrigo Fabiano, … de Filippis Ana Maria Bispo. (2019). Yellow Fever Virus Reemergence and Spread in Southeast Brazil, 2016–2019. Journal of Virology, 94(1), 10.1128/jvi.01623-19. [CrossRef]
- Höner, O. P., Wachter, B., Goller, K. V., Hofer, H., Runyoro, V., Thierer, D., Fyumagwa, R. D., Müller, T., & East, M. L. (2012). The impact of a pathogenic bacterium on a social carnivore population. Journal of Animal Ecology, 81(1), 36–46. [CrossRef]
- Jeglinski, J. W. E., Lane, J. V., Votier, S. C., Furness, R. W., Hamer, K. C., McCafferty, D. J., Nager, R. G., Sheddan, M., Wanless, S., & Matthiopoulos, J. (2024). HPAIV outbreak triggers short-term colony connectivity in a seabird metapopulation. Scientific Reports, 14(1), 3126. [CrossRef]
- Jones, K. E., Patel, N. G., Levy, M. A., Storeygard, A., Balk, D., Gittleman, J. L., & Daszak, P. (2008). Global trends in emerging infectious diseases. Nature, 451(7181), 990–993. [CrossRef]
- Kierulff, M. C. M., Ruiz-Miranda, C. R., de Oliveira, P. P., Beck, B. B., Martins, A., Dietz, J. M., Rambaldi, D. M., & Baker, A. J. (2012). The Golden lion tamarin Leontopithecus rosalia: A conservation success story. International Zoo Yearbook, 46(1), 36–45. [CrossRef]
- Lachish, S., Miller, K. J., Storfer, A., Goldizen, A. W., & Jones, M. E. (2011). Evidence that disease-induced population decline changes genetic structure and alters dispersal patterns in the Tasmanian devil. Heredity, 106(1), 172–182. [CrossRef]
- Lagrange, P., Pradel, R., Bélisle, M., & Gimenez, O. (2014). Estimating dispersal among numerous sites using capture–recapture data. Ecology, 95(8), Article 8.
- Langwig, K. E., Frick, W. F., Bried, J. T., Hicks, A. C., Kunz, T. H., & Marm Kilpatrick, A. (2012). Sociality, density-dependence and microclimates determine the persistence of populations suffering from a novel fungal disease, white-nose syndrome. Ecology Letters, 15(9), 1050–1057. [CrossRef]
- Leendertz, F. H., Ellerbrok, H., Boesch, C., Couacy-Hymann, E., Mätz-Rensing, K., Hakenbeck, R., Bergmann, C., Abaza, P., Junglen, S., Moebius, Y., Vigilant, L., Formenty, P., & Pauli, G. (2004). Anthrax kills wild chimpanzees in a tropical rainforest. Nature, 430(6998), Article 6998. [CrossRef]
- Lopes, P. C., Block, P., & König, B. (2016). Infection-induced behavioural changes reduce connectivity and the potential for disease spread in wild mice contact networks. Scientific Reports, 6(1), 31790. [CrossRef]
- Loveridge, A. J., & Macdonald, D. W. (2001). Seasonality in spatial organization and dispersal of sympatric jackals (Canis mesomelas and C. adustus): Implications for rabies management. Journal of Zoology, 253(1), 101–111. Cambridge Core. [CrossRef]
- McCallum, H., & Dobson, A. (2002). Disease, habitat fragmentation and conservation. Proceedings of the Royal Society of London. Series B: Biological Sciences, 269(1504), 2041–2049. [CrossRef]
- Moraes, A. M., Ruiz-Miranda, C. R., Galetti Jr., P. M., Niebuhr, B. B., Alexandre, B. R., Muylaert, R. L., Grativol, A. D., Ribeiro, J. W., Ferreira, A. N., & Ribeiro, M. C. (2018). Landscape resistance influences effective dispersal of endangered golden lion tamarins within the Atlantic Forest. Biological Conservation, 224, 178–187. [CrossRef]
- Morrison, R. E., Mushimiyimana, Y., Stoinski, T. S., & Eckardt, W. (2021). Rapid transmission of respiratory infections within but not between mountain gorilla groups. Scientific Reports, 11(1), Article 1. [CrossRef]
- Motes-Rodrigo, A., Albery, G. F., Negron-Del Valle, J. E., Philips, D., Cayo Biobank Research Unit, Platt, M. L., Brent, L. J. N., & Testard, C. (2025). A Natural Disaster Exacerbates and Redistributes Disease Risk Among Free-Ranging Macaques by Altering Social Structure. Ecology Letters, 28(1), e70000. [CrossRef]
- Muths, E., Scherer, R. D., & Pilliod, D. S. (2011). Compensatory effects of recruitment and survival when amphibian populations are perturbed by disease. Journal of Applied Ecology, 48(4), 873–879. [CrossRef]
- Nunn, C. L., Thrall, P. H., Stewart, K., & Harcourt, A. H. (2008). Emerging infectious diseases and animal social systems. Evolutionary Ecology, 22(4), 519–543. [CrossRef]
- Pfenning-Butterworth, A., Buckley, L. B., Drake, J. M., Farner, J. E., Farrell, M. J., Gehman, A.-L. M., Mordecai, E. A., Stephens, P. R., Gittleman, J. L., & Davies, T. J. (2024). Interconnecting global threats: Climate change, biodiversity loss, and infectious diseases. The Lancet Planetary Health, 8(4), e270–e283. [CrossRef]
- Ponchon, A. (2024). Prospecting for informed dispersal: Reappraisal of a widespread but overlooked ecological process. Preprints. [CrossRef]
- Possamai, C. B., Rodrigues de Melo, F., Mendes, S. L., & Strier, K. B. (2022). Demographic changes in an Atlantic Forest primate community following a yellow fever outbreak. American Journal of Primatology, 84(9), e23425. [CrossRef]
- Possas, C., Lourenço-de-Oliveira, R., Tauil, P. L., Pinheiro, F. de P., Pissinatti, A., Cunha, R. V. da, Freire, M., Martins, R. M., & Homma, A. (2018). Yellow fever outbreak in Brazil: The puzzle of rapid viral spread and challenges for immunisation. Memórias Do Instituto Oswaldo Cruz, 113.
- Ribeiro Prist, P., Reverberi Tambosi, L., Filipe Mucci, L., Pinter, A., Pereira de Souza, R., de Lara Muylaert, R., Roger Rhodes, J., Henrique Comin, C., da Fontoura Costa, L., Lang D’Agostini, T., Telles de Deus, J., Pavão, M., Port-Carvalho, M., Del Castillo Saad, L., Mureb Sallum, M. A., Fernandes Spinola, R. M., & Metzger, J. P. (2022). Roads and forest edges facilitate yellow fever virus dispersion. Journal of Applied Ecology, 59(1), 4–17. [CrossRef]
- Romano, V., MacIntosh, A. J. J., & Sueur, C. (2020). Stemming the Flow: Information, Infection, and Social Evolution. Trends in Ecology & Evolution, 35(10), 849–853. [CrossRef]
- Romano, V., Martins, A. F., & Ruiz-Miranda, C. R. (2019). Unraveling the dispersal patterns and the social drivers of natal emigration of a cooperative breeding mammal, the golden lion tamarin. American Journal of Primatology, 81(3), e22959. [CrossRef]
- Ruiz-Miranda, C. R., de Morais, M. M., Jr., Dietz, L. A., Rocha Alexandre, B., Martins, A. F., Ferraz, L. P., Mickelberg, J., Hankerson, S. J., & Dietz, J. M. (2019). Estimating population sizes to evaluate progress in conservation of endangered golden lion tamarins (Leontopithecus rosalia). PLOS ONE, 14(6), Article 6. [CrossRef]
- Sacchetto, L., Drumond, B. P., Han, B. A., Nogueira, M. L., & Vasilakis, N. (2020). Re-emergence of yellow fever in the neotropics—Quo vadis? Emerging Topics in Life Sciences, 4(4), 411–422. [CrossRef]
- Schradin, C., Pillay, N., & Bertelsmeier, C. (2019). Social flexibility and environmental unpredictability in African striped mice. Behavioral Ecology and Sociobiology, 73(7), 94. [CrossRef]
- Smith, K. F., Acevedo-Whitehouse, K., & Pedersen, A. B. (2009). The role of infectious diseases in biological conservation. Animal Conservation, 12(1), 1–12. [CrossRef]
- Stockmaier, S., Stroeymeyt, N., Shattuck, E. C., Hawley, D. M., Meyers, L. A., & Bolnick, D. I. (2021). Infectious diseases and social distancing in nature. Science, 371(6533), eabc8881. [CrossRef]
- Stoddard, S. T., Morrison, A. C., Vazquez-Prokopec, G. M., Paz Soldan, V., Kochel, T. J., Kitron, U., Elder, J. P., & Scott, T. W. (2009). The Role of Human Movement in the Transmission of Vector-Borne Pathogens. PLOS Neglected Tropical Diseases, 3(7), e481. [CrossRef]
- Strier, K. B., Tabacow, F. P., de Possamai, C. B., Ferreira, A. I. G., Nery, M. S., de Melo, F. R., & Mendes, S. L. (2019). Status of the northern muriqui (Brachyteles hypoxanthus) in the time of yellow fever. Primates, 60(1), 21–28. [CrossRef]
- Sumner, T., Orton, R. J., Green, D. M., Kao, R. R., & Gubbins, S. (2017). Quantifying the roles of host movement and vector dispersal in the transmission of vector-borne diseases of livestock. PLOS Computational Biology, 13(4), Article 4. [CrossRef]
- Teichroeb, J. A., Wikberg, E. C., & Sicotte, P. (2011). Dispersal in male ursine colobus monkeys (Colobus vellerosus): Influence of age, rank and contact with other groups on dispersal decisions. Behaviour, 148(7), Article 7.
- Tracey, J. A., Bevins, S. N., VandeWoude, S., & Crooks, K. R. (2014). An agent-based movement model to assess the impact of landscape fragmentation on disease transmission. Ecosphere, 5(9), art119. [CrossRef]
- Walsh, P. D., Abernethy, K. A., Bermejo, M., Beyers, R., De Wachter, P., Akou, M. E., Huijbregts, B., Mambounga, D. I., Toham, A. K., Kilbourn, A. M., Lahm, S. A., Latour, S., Maisels, F., Mbina, C., Mihindou, Y., Ndong Obiang, S., Effa, E. N., Starkey, M. P., Telfer, P., … Wilkie, D. S. (2003). Catastrophic ape decline in western equatorial Africa. Nature, 422(6932), 611–614. [CrossRef]
- Watts, A. G., Saura, S., Jardine, C., Leighton, P., Werden, L., & Fortin, M.-J. (2018). Host functional connectivity and the spread potential of Lyme disease. Landscape Ecology, 33(11), 1925–1938. [CrossRef]
| Transience | Trap-dependence | |||
| Test WBWA | Test 3G.SR | Test 3G.Sm | Test M-ITEC | |
| Χ² | 4.53 | 17.17 | 41.56 | 6.01 |
| df | 9 | 11 | 21 | 2 |
| p-value | 0.87 | 0.10 | 0.005 | 0.05 |
| Number of groups | Corresponding years | Disease outbreak? | |
| Survival probabilities | |||
| t | 1 cohort | 2011-2022 | Same annual survival for all individuals |
| c2 | 2 cohorts | 2011-2016; 2019-2022 |
Second observation before or after the outbreak |
| 2016-2017 | Second observation during the outbreak | ||
| c3 | 3 cohorts | 2011-2015 | Second observation before the outbreak |
| 2016-2017 | Second observation during the outbreak | ||
| 2018-2022 | Second observation after the outbreak | ||
| Dispersal probabilities | |||
| 1T | 1 period | 2011-2022 | Same dispersal rates for the whole period |
| 2T | 2 periods | 2011-2016;2019-2022 | Before or the after outbreak |
| 2017-2018 | During outbreak | ||
| 3T | 3 periods | 2011-2016 | Before outbreak |
| 2017-2018 | During outbreak | ||
| 2019-2022 | After outbreak |
| Model | # Id. Par. | Deviance | QAIC | QAICc | ΔAIC |
| s(c2.t)psi(2T)p(2f) | 31 | 1846.381 | 1908.381 | 1909.5783 | 0 |
| s(c2.t)psi(1T)p(2f) | 25 | 1863.6626 | 1913.6626 | 1914.4443 | 4.9 |
| s(c3.t)psi(2T)p(2f) | 35 | 1845.2729 | 1915.2729 | 1916.7974 | 7.2 |
| s(c2.t)psi(3T)p(2f) | 37 | 1842.902 | 1916.902 | 1918.6052 | 9.0 |
| s(t)psi(2T)p(2f) | 25 | 1867.5549 | 1917.5549 | 1918.3366 | 8.8 |
| s(c3+t)psi(2T)p(2f) | 27 | 1864.3688 | 1918.3688 | 1919.2791 | 9.7 |
| s(c3.t)psi(1T)p(2f) | 29 | 1862.4505 | 1920.4505 | 1921.4993 | 11.9 |
| s(t)psi(1T)p(2f) | 19 | 1884.6895 | 1922.6895 | 1923.1449 | 13.6 |
| s(c3+t)psi(1T)p(2f) | 21 | 1881.5787 | 1923.5787 | 1924.133 | 14.6 |
| s(c2.t)psi(2T)p(i) | 29 | 1868.5175 | 1926.5175 | 1927.5663 | 18.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




