Submitted:
15 November 2024
Posted:
18 November 2024
Read the latest preprint version here
Abstract
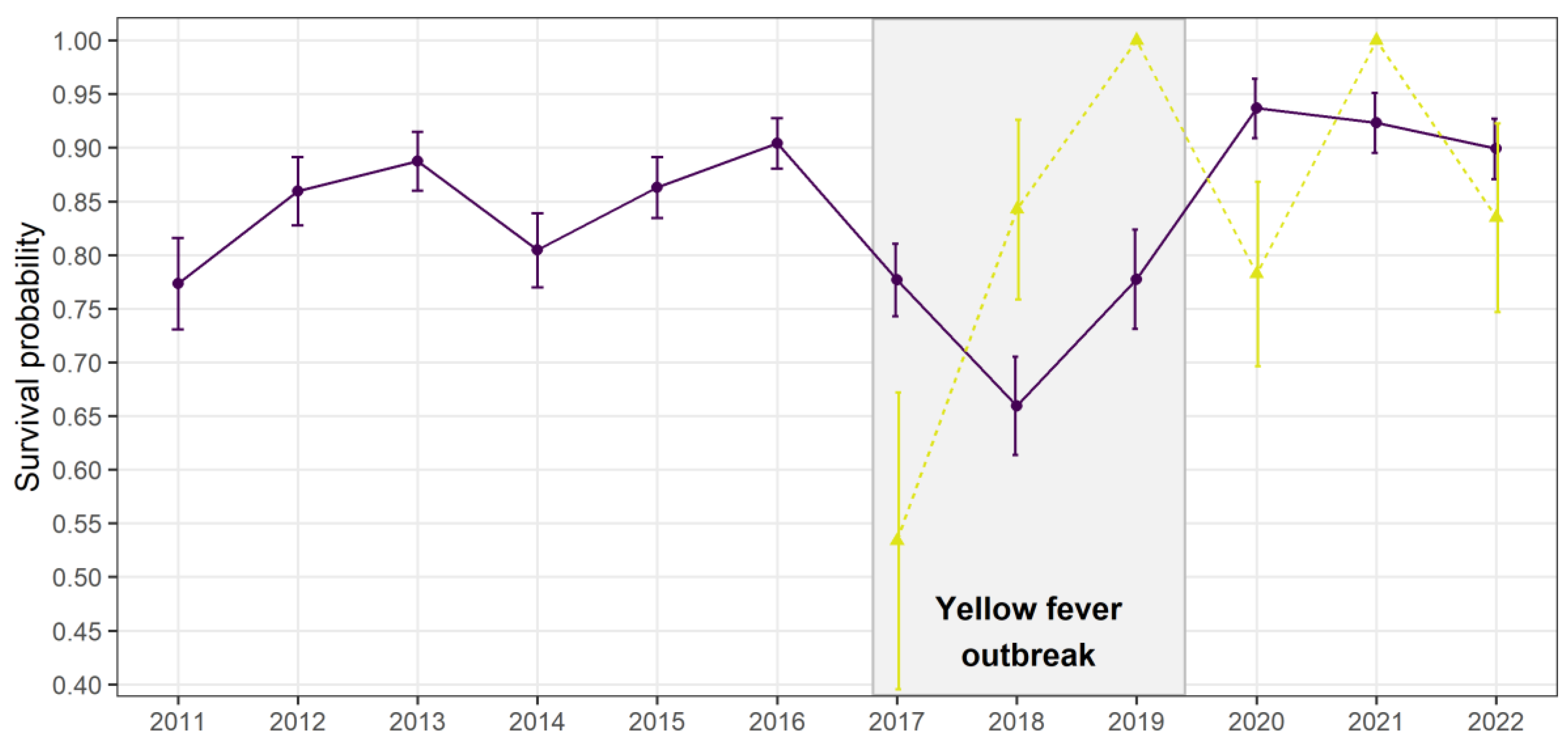
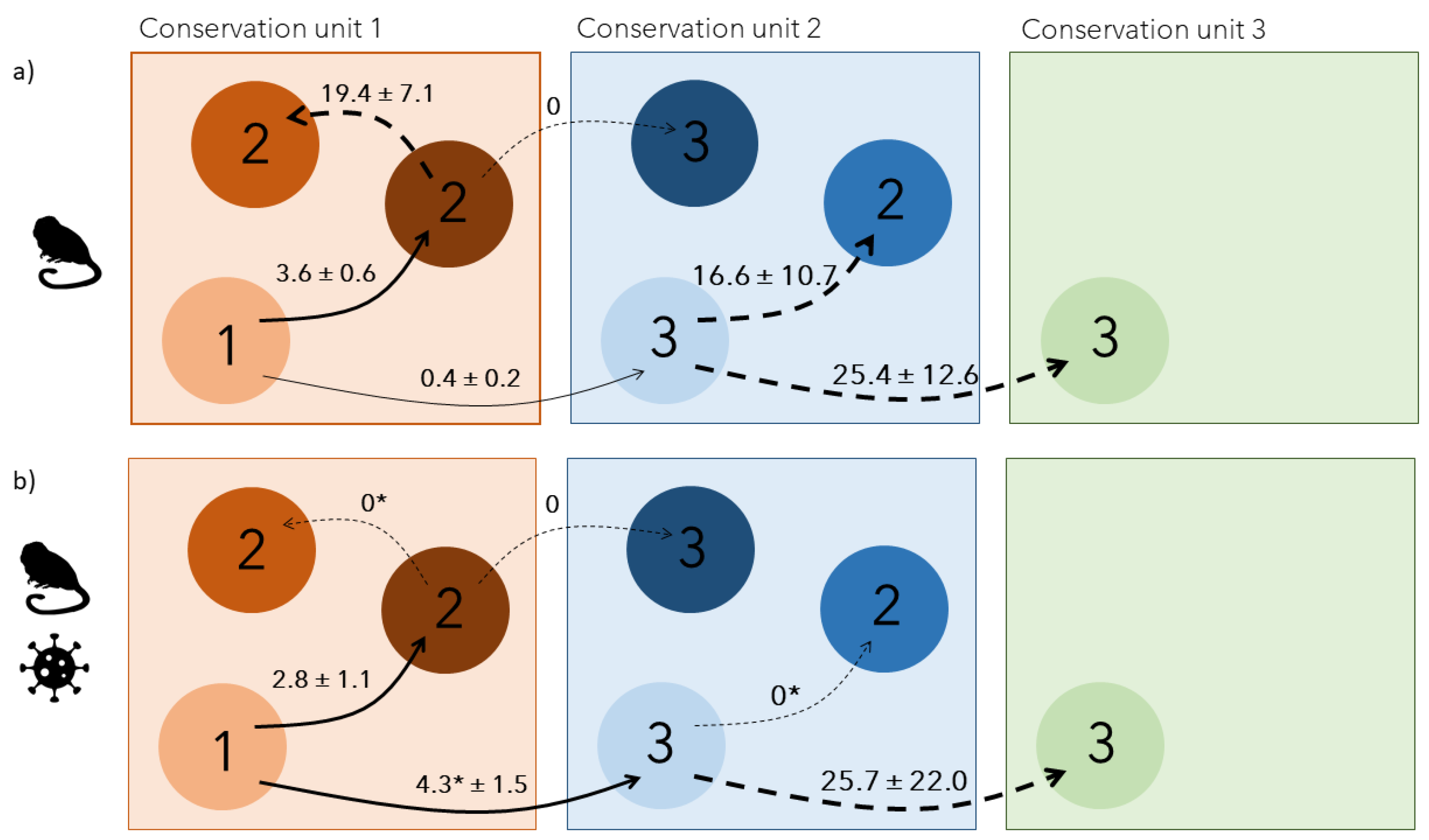
Disease outbreaks can severely affect populations in the wild. However, their consequences on individual behaviours are poorly understood. We used a multi-site capture-recapture model to investigate the impacts of a yellow fever outbreak on the endangered golden lion tamarin Leontopithecus rosalia, in the highly fragmented Atlantic forest, Brazil. Annual adult survival rate severely declined in 2017-2018, coinciding with the outbreak period. Simultaneously, dispersal patterns changed temporarily, with a reduction of settlement time for individuals dispersing within the same forest fragments, and a significant increase of dispersal between forest fragments, from 0.4±0.2 to 4.3±1.5%. Those results indicate a spatial rearrangement of individuals during the outbreak potentially due to non-random mortality leading to changes in social group structure at a local and regional scale. They advocate for a better integration of host movements, host social behaviour and habitat connectivity when evaluating species response to infectious diseases.
Keywords:
Introduction
Methods
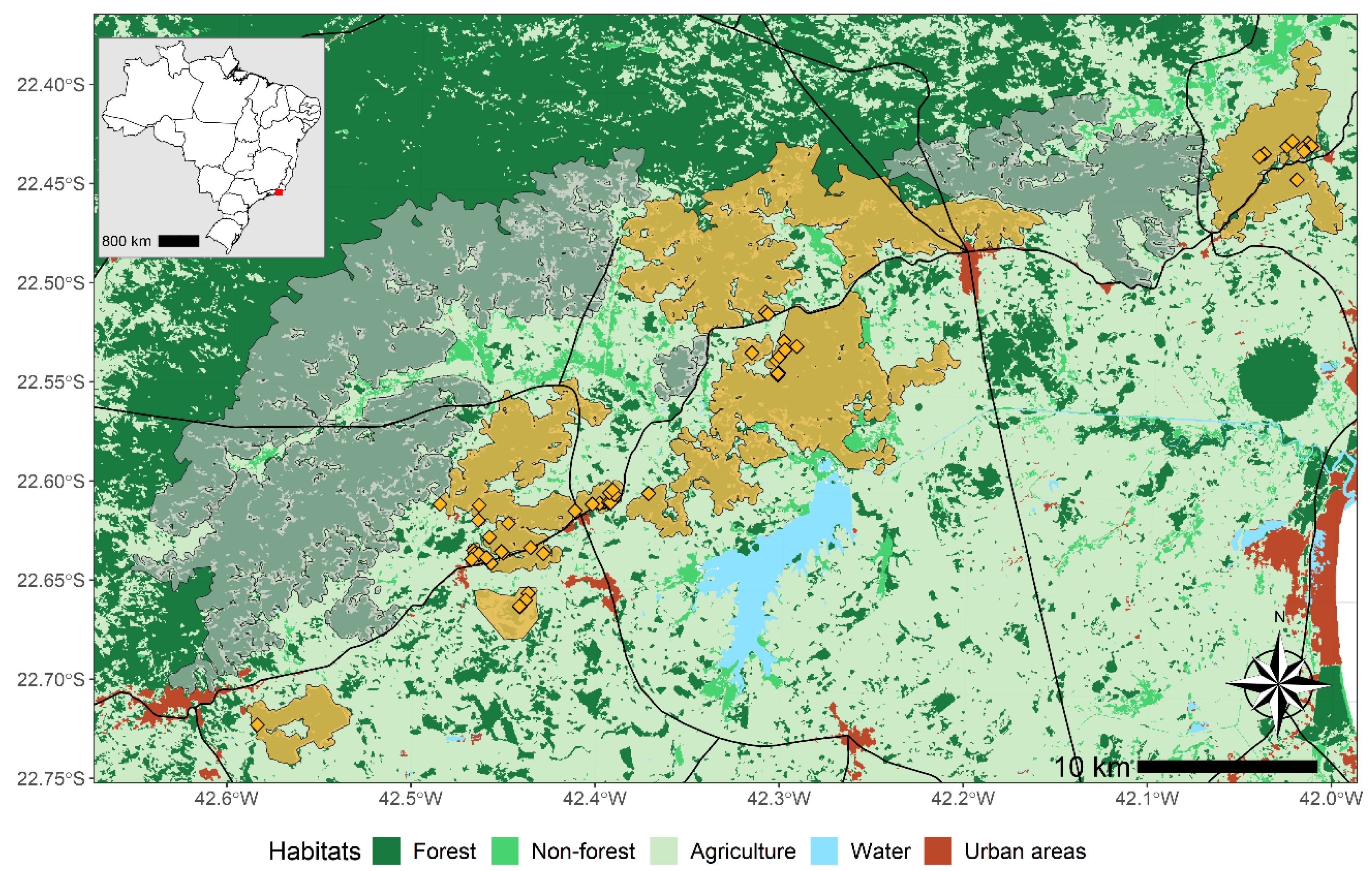
Study Site and Population
Data Selection
Capture-Recapture Analysis
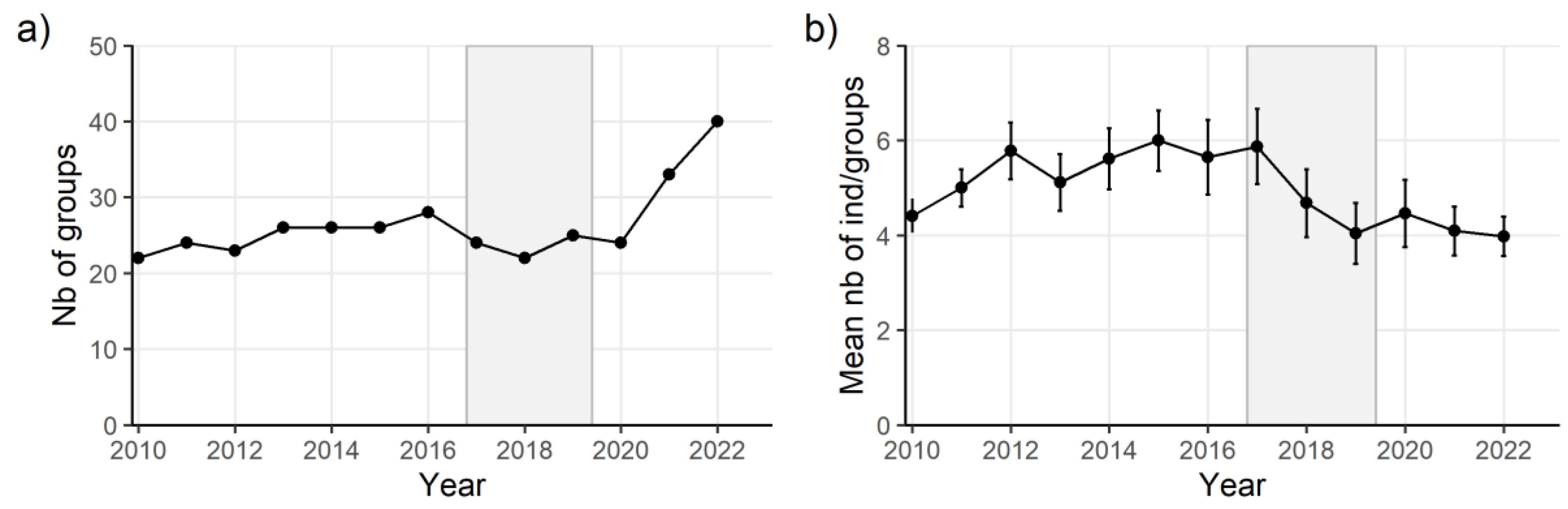
Results
Discussion
Data Availability Statement
Acknowledgments
Ethical Note
References
- Abreu, F.V.; Ferreira-de-Brito, A.; Azevedo, A.D.; Linhares, J.H.; de Oliveira Santos, V.; Hime Miranda, E.; et al. . Survey on Non-Human Primates and Mosquitoes Does not Provide Evidences of Spillover/Spillback between the Urban and Sylvatic Cycles of Yellow Fever and Zika Viruses Following Severe Outbreaks in Southeast Brazil. Viruses 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Arce-Peña, N.P.; Arroyo-Rodríguez, V.; Dias, P.A.D.; Franch-Pardo, I.; Andresen, E. Linking changes in landscape structure to population changes of an endangered primate. Landsc. Ecol. 2019, 34, 2687–2701. [Google Scholar] [CrossRef]
- Baker, A.J.; Dietz, J.M. Immigration in wild groups of golden lion tamarins (Leontopithecus rosalia). Am. J. Primatol. 1996, 38, 47–56. [Google Scholar] [CrossRef]
- Bermejo, M.; Rodríguez-Teijeiro, J.D.; Illera, G.; Barroso, A.; Vilà, C.; Walsh, P.D. Ebola Outbreak Killed 5000 Gorillas. Science 2006, 314, 1564. [Google Scholar] [CrossRef]
- Berthet, M.; Mesbahi, G.; Duvot, G.; Zuberbühler, K.; Cäsar, C.; Bicca-Marques, J.C. Dramatic decline in a titi monkey population after the 2016–2018 sylvatic yellow fever outbreak in Brazil. Am. J. Primatol. 2021, 83, e23335. [Google Scholar] [CrossRef]
- Bicca-Marques, J.C.; de Freitas, D.S. The Role of Monkeys, Mosquitoes, and Humans in the Occurrence of a Yellow Fever Outbreak in a Fragmented Landscape in South Brazil: Protecting Howler Monkeys is a Matter of Public Health. Trop. Conserv. Sci. 2010, 3, 78–89. [Google Scholar] [CrossRef]
- Binning, S.A.; Shaw, A.K.; Roche, D.G. Parasites and Host Performance: Incorporating Infection into Our Understanding of Animal Movement. Integr. Comp. Biol. 2017, 57, 267–280. [Google Scholar] [CrossRef]
- Boulinier, T.; Kada, S.; Ponchon, A.; Dupraz, M.; Dietrich, M.; Gamble, A.; et al. . Migration, Prospecting, Dispersal? What Host Movement Matters for Infectious Agent Circulation? Integr. Comp. Biol. 2016, 56, 330–342. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. (2002). Model selection and multimodel inference: A practical information-theoretic approach, 2nd Edition. Springer, New-York, USA.
- Caillaud, D.; Levréro, F.; Cristescu, R.; Gatti, S.; Dewas, M.; Douadi, M.; et al. . Gorilla susceptibility to Ebola virus: The cost of sociality. Curr. Biol. 2006, 16, R489–R491. [Google Scholar] [CrossRef]
- Chala, B.; Hamde, F. Emerging and Re-emerging Vector-Borne Infectious Diseases and the Challenges for Control: A Review. Front. Public Health 2021, 9. [Google Scholar] [CrossRef]
- Choquet, R.; Lebreton, J.-D.; Gimenez, O.; Reboulet, A.-M.; Pradel, R. U-CARE: Utilities for performing goodness of fit tests and manipulating CApture–REcapture data. Ecography 2009, 32, 1071–1074. [Google Scholar] [CrossRef]
- Choquet, R.; Rouan, L.; Pradel, R. (2009b). Program E-Surge: Software application for fitting multievent models. In: Modeling demographic processes in marked populations, Environmental and Ecological Statistics (eds. Thomson, D.; Cooch, E.; Conroy, M.). Springer US, pp. 845–865.
- Dekelaita, D.J.; Epps, C.W.; German, D.W.; Powers, J.G.; Gonzales, B.J.; Abella-Vu, R.K.; et al. . Animal movement and associated infectious disease risk in a metapopulation. R. Soc. Open Sci. 2023, 10, 220390. [Google Scholar] [CrossRef] [PubMed]
- Dietz, J.M.; Hankerson, S.J.; Alexandre, B.R.; Henry, M.D.; Martins, A.F.; Ferraz, L.P.; et al. . Yellow fever in Brazil threatens successful recovery of endangered golden lion tamarins. Sci. Rep. 2019, 9, 12926. [Google Scholar] [CrossRef] [PubMed]
- Dietz, J.M.; Mickelberg, J.; Traylor-Holzer, K.; Martins, A.F.; Souza, M.N.; Hankerson, S.J. Golden lion tamarin metapopulation dynamics five years after heavy losses to yellow fever. Am. J. Primatol. 2024, 86, e23635. [Google Scholar] [CrossRef]
- Dougherty, E.R.; Seidel, D.P.; Carlson, C.J.; Spiegel, O.; Getz, W.M. Going through the motions: Incorporating movement analyses into disease research. Ecol. Lett. 2018, 21, 588–604. [Google Scholar] [CrossRef]
- Duriez, O.; Sassi, Y.; Le Gall-Ladevèze, C.; Giraud, L.; Straughan, R.; Dauverné, L.; et al. . Highly pathogenic avian influenza affects vultures’ movements and breeding output. Curr. Biol. 2023, 33, 3766–3774.e3. [Google Scholar] [CrossRef]
- Estrada, A.; Garber, P.A.; Rylands, A.B.; Roos, C.; Fernandez-Duque, E.; Di Fiore, A.; et al. . Impending extinction crisis of the world’s primates: Why primates matter. Sci. Adv. 2017, 3, e1600946. [Google Scholar] [CrossRef]
- Gaitan, J.; Millien, V. Stress level, parasite load, and movement pattern in a small-mammal reservoir host for Lyme disease. Can. J. Zool. 2016, 94, 565–573. [Google Scholar] [CrossRef]
- Genton, C.; Pierre, A.; Cristescu, R.; Lévréro, F.; Gatti, S.; Pierre, J.-S.; et al. . How Ebola impacts social dynamics in gorillas: A multistate modelling approach. J. Anim. Ecol. 2015, 84, 166–176. [Google Scholar] [CrossRef]
- Giovanetti Marta, de Mendonça Marcos Cesar Lima, Fonseca Vagner, Mares-Guia Maria Angélica, Fabri Allison, Xavier Joilson; et al. Yellow Fever Virus Reemergence and Spread in Southeast Brazil 2019, 2016–2019. J. Virol. 94. [CrossRef]
- Gudelj, I.; White, K.A.J. Spatial heterogeneity, social structure and disease dynamics of animal populations. Theor. Popul. Biol. 2004, 66, 139–149. [Google Scholar] [CrossRef]
- Höner, O.P.; Wachter, B.; Goller, K.V.; Hofer, H.; Runyoro, V.; Thierer, D.; et al. . The impact of a pathogenic bacterium on a social carnivore population. J. Anim. Ecol. 2012, 81, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Jeglinski, J.W.E.; Lane, J.V.; Votier, S.C.; Furness, R.W.; Hamer, K.C.; McCafferty, D.J.; et al. . HPAIV outbreak triggers short-term colony connectivity in a seabird metapopulation. Sci. Rep. 2024, 14, 3126. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; et al. . Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef]
- Kierulff, M.C.M.; Ruiz-Miranda, C.R.; de Oliveira, P.P.; Beck, B.B.; Martins, A.; Dietz, J.M.; et al. . The Golden lion tamarin Leontopithecus rosalia: A conservation success story. Int. Zoo Yearb. 2012, 46, 36–45. [Google Scholar] [CrossRef]
- Lachish, S.; Miller, K.J.; Storfer, A.; Goldizen, A.W.; Jones, M.E. Evidence that disease-induced population decline changes genetic structure and alters dispersal patterns in the Tasmanian devil. Heredity 2011, 106, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Lagrange, P.; Pradel, R.; Bélisle, M.; Gimenez, O. Estimating dispersal among numerous sites using capture–recapture data. Ecology 2014, 95, 2316–2323. [Google Scholar] [CrossRef]
- Langwig, K.E.; Frick, W.F.; Bried, J.T.; Hicks, A.C.; Kunz, T.H.; Marm Kilpatrick, A. Sociality, density-dependence and microclimates determine the persistence of populations suffering from a novel fungal disease, white-nose syndrome. Ecol. Lett. 2012, 15, 1050–1057. [Google Scholar] [CrossRef]
- Leendertz, F.H.; Ellerbrok, H.; Boesch, C.; Couacy-Hymann, E.; Mätz-Rensing, K.; Hakenbeck, R.; et al. . Anthrax kills wild chimpanzees in a tropical rainforest. Nature 2004, 430, 451–452. [Google Scholar] [CrossRef]
- Loveridge, A.J.; Macdonald, D.W. Seasonality in spatial organization and dispersal of sympatric jackals (Canis mesomelas and C. adustus): Implications for rabies management. J. Zool. 2001, 253, 101–111. [Google Scholar] [CrossRef]
- McCallum, H.; Dobson, A. Disease, habitat fragmentation and conservation. Proc. R. Soc. Lond. B Biol. Sci. 2002, 269, 2041–2049. [Google Scholar] [CrossRef]
- Morrison, R.E.; Mushimiyimana, Y.; Stoinski, T.S.; Eckardt, W. Rapid transmission of respiratory infections within but not between mountain gorilla groups. Sci. Rep. 2021, 11, 19622. [Google Scholar] [CrossRef] [PubMed]
- Muths, E.; Scherer, R.D.; Pilliod, D.S. Compensatory effects of recruitment and survival when amphibian populations are perturbed by disease. J. Appl. Ecol. 2011, 48, 873–879. [Google Scholar] [CrossRef]
- Nunn, C.L.; Thrall, P.H.; Stewart, K.; Harcourt, A.H. Emerging infectious diseases and animal social systems. Evol. Ecol. 2008, 22, 519–543. [Google Scholar] [CrossRef]
- Ponchon, A. (2024). Prospecting for informed dispersal: Reappraisal of a widespread but overlooked ecological process. Preprints.
- Possamai, C.B.; Rodrigues de Melo, F.; Mendes, S.L.; Strier, K.B. Demographic changes in an Atlantic Forest primate community following a yellow fever outbreak. Am. J. Primatol. 2022, 84, e23425. [Google Scholar] [CrossRef]
- Possas, C.; Lourenço-de-Oliveira, R.; Tauil, P.L.; Pinheiro, F. de P.; Pissinatti, A.; Cunha, R.V. da; et al. Yellow fever outbreak in Brazil: The puzzle of rapid viral spread and challenges for immunisation. Mem. Inst. Oswaldo Cruz, 2018; 113. [Google Scholar]
- Ribeiro Prist, P.; Reverberi Tambosi, L.; Filipe Mucci, L.; Pinter, A.; Pereira de Souza, R.; de Lara Muylaert, R.; et al. . Roads and forest edges facilitate yellow fever virus dispersion. J. Appl. Ecol. 2022, 59, 4–17. [Google Scholar] [CrossRef]
- Romano, V.; MacIntosh, A.J.J.; Sueur, C. Stemming the Flow: Information, Infection, and Social Evolution. Trends Ecol. Evol. 2020, 35, 849–853. [Google Scholar] [CrossRef]
- Romano, V.; Martins, A.F.; Ruiz-Miranda, C.R. Unraveling the dispersal patterns and the social drivers of natal emigration of a cooperative breeding mammal, the golden lion tamarin. Am. J. Primatol. 2019, 81, e22959. [Google Scholar] [CrossRef]
- Ruiz-Miranda, C.R.; de Morais, M.M.; Jr. , Dietz, L.A.; Rocha Alexandre, B.; Martins, A.F.; Ferraz, L.P.; et al. Estimating population sizes to evaluate progress in conservation of endangered golden lion tamarins (Leontopithecus rosalia). PLoS ONE 2019, 14, e0216664. [Google Scholar] [CrossRef]
- Sacchetto, L.; Drumond, B.P.; Han, B.A.; Nogueira, M.L.; Vasilakis, N. Re-emergence of yellow fever in the neotropics — quo vadis? Emerg. Top. Life Sci. 2020, 4, 411–422. [Google Scholar]
- Schradin, C.; Pillay, N.; Bertelsmeier, C. Social flexibility and environmental unpredictability in African striped mice. Behav. Ecol. Sociobiol. 2019, 73, 94. [Google Scholar] [CrossRef]
- Smith, K.F.; Acevedo-Whitehouse, K.; Pedersen, A.B. The role of infectious diseases in biological conservation. Anim. Conserv. 2009, 12, 1–12. [Google Scholar] [CrossRef]
- Stockmaier, S.; Stroeymeyt, N.; Shattuck, E.C.; Hawley, D.M.; Meyers, L.A.; Bolnick, D.I. Infectious diseases and social distancing in nature. Science 2021, 371, eabc8881. [Google Scholar] [CrossRef] [PubMed]
- Stoddard, S.T.; Morrison, A.C.; Vazquez-Prokopec, G.M.; Paz Soldan, V.; Kochel, T.J.; Kitron, U.; et al. . The Role of Human Movement in the Transmission of Vector-Borne Pathogens. PLoS Negl. Trop. Dis. 2009, 3, e481. [Google Scholar] [CrossRef] [PubMed]
- Strier, K.B.; Tabacow, F.P.; de Possamai, C.B.; Ferreira, A.I.G.; Nery, M.S.; de Melo, F.R.; et al. . Status of the northern muriqui (Brachyteles hypoxanthus) in the time of yellow fever. Primates 2019, 60, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Sumner, T.; Orton, R.J.; Green, D.M.; Kao, R.R.; Gubbins, S. Quantifying the roles of host movement and vector dispersal in the transmission of vector-borne diseases of livestock. PLOS Comput. Biol. 2017, 13, e1005470. [Google Scholar] [CrossRef]
- Teichroeb, J.A.; Wikberg, E.C.; Sicotte, P. Dispersal in male ursine colobus monkeys (Colobus vellerosus): Influence of age, rank and contact with other groups on dispersal decisions. Behaviour 2011, 148, 765–793. [Google Scholar] [CrossRef]
- Tracey, J.A.; Bevins, S.N.; VandeWoude, S.; Crooks, K.R. An agent-based movement model to assess the impact of landscape fragmentation on disease transmission. Ecosphere 2014, 5, art119. [Google Scholar] [CrossRef]
- Walsh, P.D.; Abernethy, K.A.; Bermejo, M.; Beyers, R.; De Wachter, P.; Akou, M.E.; et al. . Catastrophic ape decline in western equatorial Africa. Nature 2003, 422, 611–614. [Google Scholar] [CrossRef]
- Watts, A.G.; Saura, S.; Jardine, C.; Leighton, P.; Werden, L.; Fortin, M.-J. Host functional connectivity and the spread potential of Lyme disease. Landsc. Ecol. 2018, 33, 1925–1938. [Google Scholar] [CrossRef]
| Transience | Trap-dependence | |||
|---|---|---|---|---|
| Test WBWA | Test 3G.SR | Test 3G.Sm | Test M-ITEC | |
| Χ² | 4.53 | 17.17 | 41.56 | 6.01 |
| df | 9 | 11 | 21 | 2 |
| p-value | 0.87 | 0.10 | 0.005 | 0.05 |
| Number of groups | Corresponding years | Disease outbreak? | |
|---|---|---|---|
| Survival probabilities | |||
| t | 1 cohort | 2011-2022 | Same survival for all individuals |
| c2 | 2 cohorts | < 2016 | Second observation before or after the outbreak |
| > 2018 | Second observation during the outbreak | ||
| c3 | 3 cohorts | < 2016 | Second observation before the outbreak |
| 2016-2017 | Second observation during the outbreak | ||
| > 2018 | Second observation after the outbreak | ||
| Dispersal probabilities | |||
| 1T | 1 period | 2011-2022 | Same dispersal rates for the whole period |
| 2T | 2 periods | 2011-2016;2019-2022 | No outbreak |
| 2017-2018 | Outbreak | ||
| 3T | 3 periods | 2011-2016 | Before outbreak |
| 2017-2018 | During outbreak | ||
| 2019-2022 | After outbreak |
| Model | # Id. Par. | Deviance | QAIC | QAICc | ΔAIC |
|---|---|---|---|---|---|
| s(c2.t)psi(2T)p(2f) | 31 | 1846.381 | 1908.381 | 1909.5783 | 0 |
| s(c2.t)psi(1T)p(2f) | 25 | 1863.6626 | 1913.6626 | 1914.4443 | 4.9 |
| s(c3.t)psi(2T)p(2f) | 35 | 1845.2729 | 1915.2729 | 1916.7974 | 7.2 |
| s(c2.t)psi(3T)p(2f) | 37 | 1842.902 | 1916.902 | 1918.6052 | 9.0 |
| s(t)psi(2T)p(2f) | 25 | 1867.5549 | 1917.5549 | 1918.3366 | 8.8 |
| s(c3+t)psi(2T)p(2f) | 27 | 1864.3688 | 1918.3688 | 1919.2791 | 9.7 |
| s(c3.t)psi(1T)p(2f) | 29 | 1862.4505 | 1920.4505 | 1921.4993 | 11.9 |
| s(t)psi(1T)p(2f) | 19 | 1884.6895 | 1922.6895 | 1923.1449 | 13.6 |
| s(c3+t)psi(1T)p(2f) | 21 | 1881.5787 | 1923.5787 | 1924.133 | 14.6 |
| s(c2.t)psi(2T)p(i) | 29 | 1868.5175 | 1926.5175 | 1927.5663 | 18.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




