Submitted:
04 March 2024
Posted:
06 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Synthesis of D8 THC
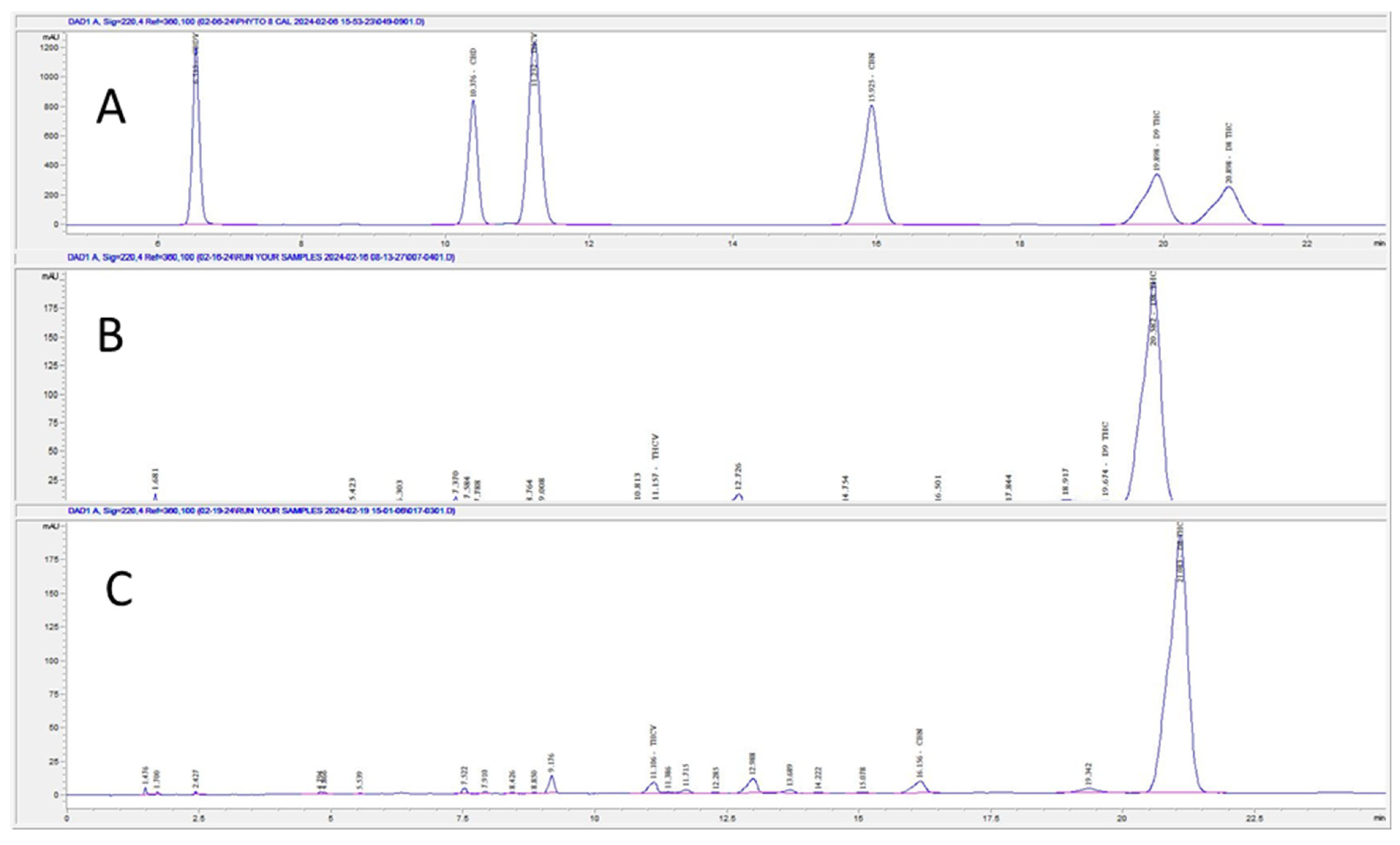
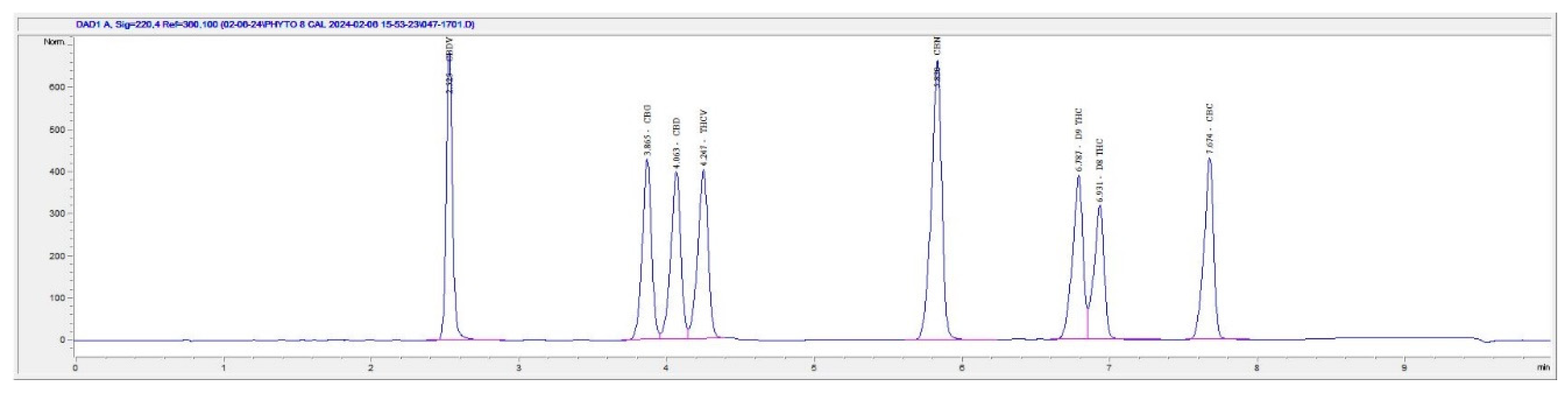
2.2. HPLC method for the Separation of THC Isomers
| Time (min) | Solvent A | Solvent B | Solvent C | Solvent D | Flow (mL/min) | Pressure (bar) |
|---|---|---|---|---|---|---|
| 0.00 | 45 | 55 | 0 | 0 | 1.5 | 375 |
| 5.50 | 43 | 57 | 0 | 0 | 1.5 | 375 |
| 6.51 | 40 | 60 | 0 | 0 | 1.5 | 375 |
| 11.00 | 40 | 60 | 0 | 0 | 1.5 | 375 |
| 25.00 | 40 | 60 | 0 | 0 | 1.5 | 375 |
| Time (min) | Solvent A | Solvent B | Solvent C | Solvent D | Flow (mL/min) | Pressure (bar) |
|---|---|---|---|---|---|---|
| 0.00 | 30 | 70 | 0 | 0 | 1.60 | 375 |
| 3.00 | 30 | 70 | 0 | 0 | 1.60 | 375 |
| 7.00 | 15 | 85 | 0 | 0 | 1.60 | 375 |
| 7.01 | 5 | 95 | 0 | 0 | 1.60 | 375 |
| 8.00 | 5 | 95 | 0 | 0 | 1.60 | 375 |
| 8.01 | 30 | 70 | 0 | 0 | 1.60 | 375 |
| 10.00 | 30 | 70 | 0 | 0 | 1.60 | 375 |
3. Results and Discussion
4. Conclusion
Supplementary Materials
Author Contributions
Funding
Acknowledgements
Author Disclosure
Abbreviations
| ACN | Acetonitrile |
| CBD | Cannabidiol |
| CPG | Consumer Product Good |
| CRM | Certified Reference Material |
| HPLC | High Performance Liquid Chromatography |
| THC | Tetrahydrocannabinol |
References
- Schilling, S.; Melzer, R.; McCabe, P.F. Quick Guide Cannabis Sativa, Current Biology 2020, 30, R1-R9.
- Bassetti, B.; Hone, C. A.; Kappe, C. O. Continuous-Flow Synthesis of Δ9-Tetrahydrocannabinol and Δ8-Tetrahydrocannabinol from Cannabidiol. J. Org. Chem. 2023, 88 (9), 6227–6231. [CrossRef]
- Giorgi, P. D.; Liautard, V.; Pucheault, M.; Antoniot-ti, S. Biomimetic Cannabinoid Synthesis Revisit-ed: Batch and Flow All-Catalytic Synthesis of (±)-Ortho -Tetrahydrocannabinols and Analogues from Natural Feedstocks: Biomimetic Canna-binoid Synthesis Revisited: Batch and Flow All-Catalytic Synthesis of (±)-Ortho-Tetrahydrocannabinols and Analogues from Natural Feeds. European J. Org. Chem. 2018, 2018 (11), 1307–1311. [CrossRef]
- Chiurchiù, E.; Sampaolesi, S.; Allegrini, P.; Ciceri, D.; Ballini, R.; Palmieri, A. A Novel and Practical Continuous Flow Chemical Synthesis of Canna-bidiol (CBD) and Its CBDV and CBDB Ana-logues. European J. Org. Chem. 2021, 2021 (8), 1286–1289. [CrossRef]
- Bloemendal, V. R. L. J.; Spierenburg, B.; Boltje, T. J.; van Hest, J. C. M.; Rutjes, F. P. J. T. One-Flow Synthesis of Tetrahydrocannabinol and Cannabidiol Using Homo- and Heterogeneous Lewis Acids. J. Flow Chem. 2021, 11 (2), 99–105. [CrossRef]
- Marzullo, P.; Foschi, F.; Coppini, D. A.; Fanchini, F.; Magnani, L.; Rusconi, S.; Luzzani, M.; Passarella, D. Cannabidiol as the Substrate in Acid-Catalyzed Intramolecular Cyclization. J. Nat. Prod. 2020, 83 (10), 2894–2901. [CrossRef]
- Collins, A.; Ramirez, G.; Tesfatsion, T.; Ray, K. P.; Caudill, S.; Cruces, W. Synthesis and Characterization of the Diastereomers of HHC and H4CBD. Nat. Prod. Commun. 2023, 18 (3), 1934578X2311589. [CrossRef]
- Ramirez, G. A., Docampo-Palacios, M. L., Tesfatsion, T. T., Cruces, I., Hellmann, A. J., Okhovat, A., Pittiglio, M., Ray, K. P., Cruces, W., Ultrasonic or Microwave modified continuous flow chemistry for the synthesis of tetrahydrocannabinol: Observing the effects of various solvents and acids, Accepted, ACS Omega, 2024, Preprint. [CrossRef]
- Tesfatsion, T. T., Ramirez, G. A., Docampo, M. L., Collins, A. C., Mzannar, Y., Khan, H. Y., Aboukameel, O., Azmi, A. S., Jagtap, P. G., Ray, K. P., Cruces, W., Antineoplastic Properties of THCV, HHC and their anti-Proliferative effects on HPAF-II, MIA-paca2, Aspc-1, and PANC-1 PDAC Pancreatic Cell Lines, 2022, Submitted to J. Cannabis Res., Preprint. [CrossRef]
- Gong X, Sun C, Abame MA, et al. Synthesis of CBD and its derivatives bearing various C4′-side chains with a late-stage diversification method. J Org Chem 2020;85(4):2704–2715. [CrossRef]
- Erickson BE. Delta-8-THC craze concerns chemists. Chem Eng News. 2021;99:25-28.
- Docampo-Palacios, M. L., Ramirez, G. A., Tesfatsion, T. T., Pittiglio, M. K., Ray, K. P., Cruces, Comprehensive Safety Assessment of Diverse Cannabinoids: A Scientific Inquiry, 2024. [CrossRef]
- Segall, B. 13News investigation finds some Delta-8 products exceed state’s THC limit, underscoring need for regulation, 13WTHR, accessed February 23, 2024 https://www.wthr.com/article/news/investigations/13-investigates/13news-investigation-shows-some-delta-8-products-exceed-legal-delta-9-thc-limit-posing-risks-consumers-retailers/531-755951a1-981c-446d-b5f5-995370a9c397.
- Geci M, Scialdone M, Tishler J. The dark side of cannabidiol: The unanticipated social and clinical implications of synthetic Δ8-THC. Cannabis Cannabinoid Res. Published online 2022. [CrossRef]
- Babalonis S, Raup-Konsavage WM, Akpunonu PD, Balla A, Vrana KE. Δ8-THC: Legal status, widespread availability, and safety concerns. Cannabis Cannabinoid Res. 2021;6(5):362-365. [CrossRef]
- Livne O, Budney A, Borodovsky J, Walsh C, Shmulewitz D, Fink DS, et al. Delta-8 THC use in US adults: Sociodemographic characteristics and correlates. Addict Behav. 2022;133(107374):107374. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




