Submitted:
05 March 2024
Posted:
06 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
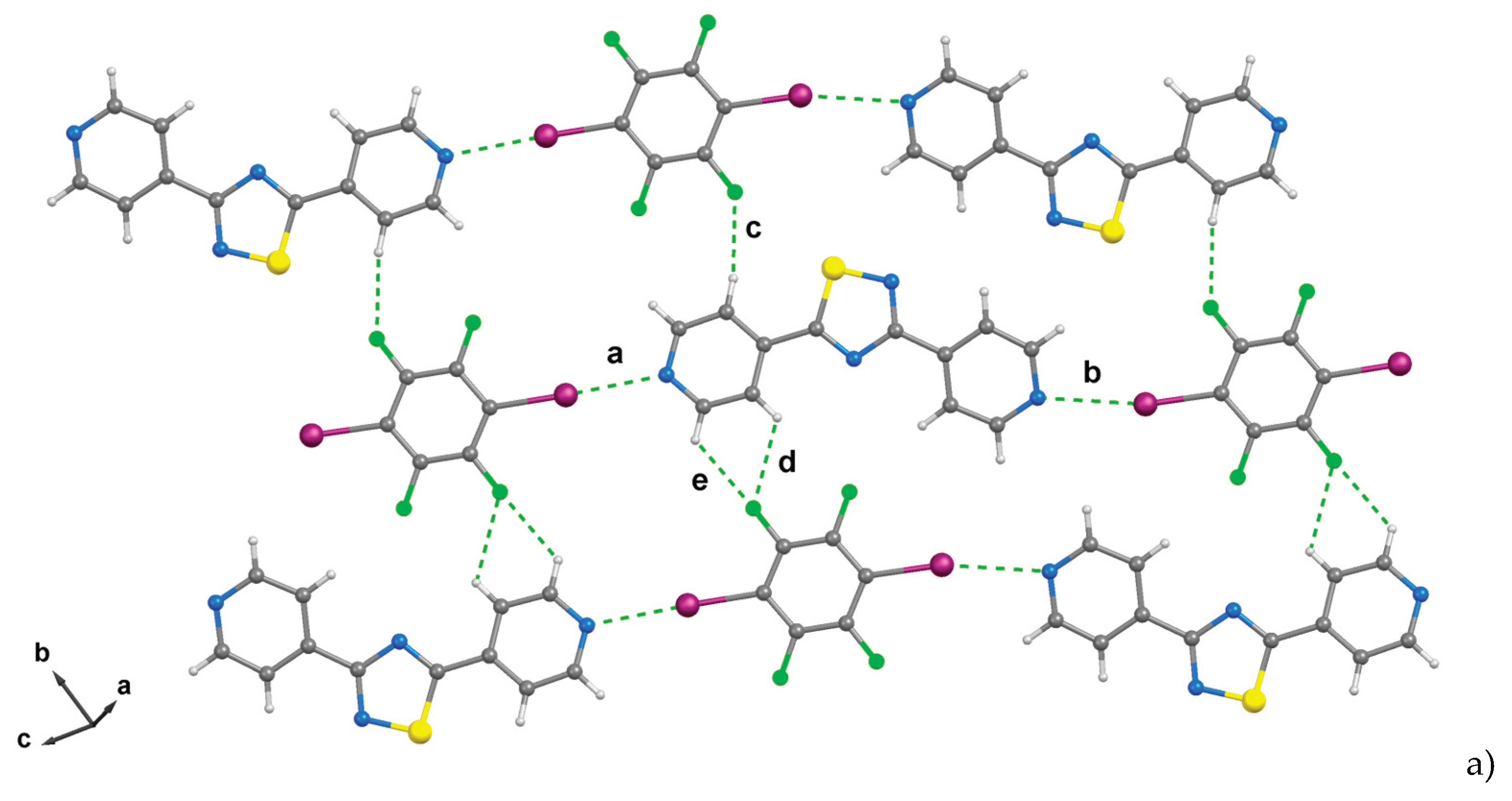
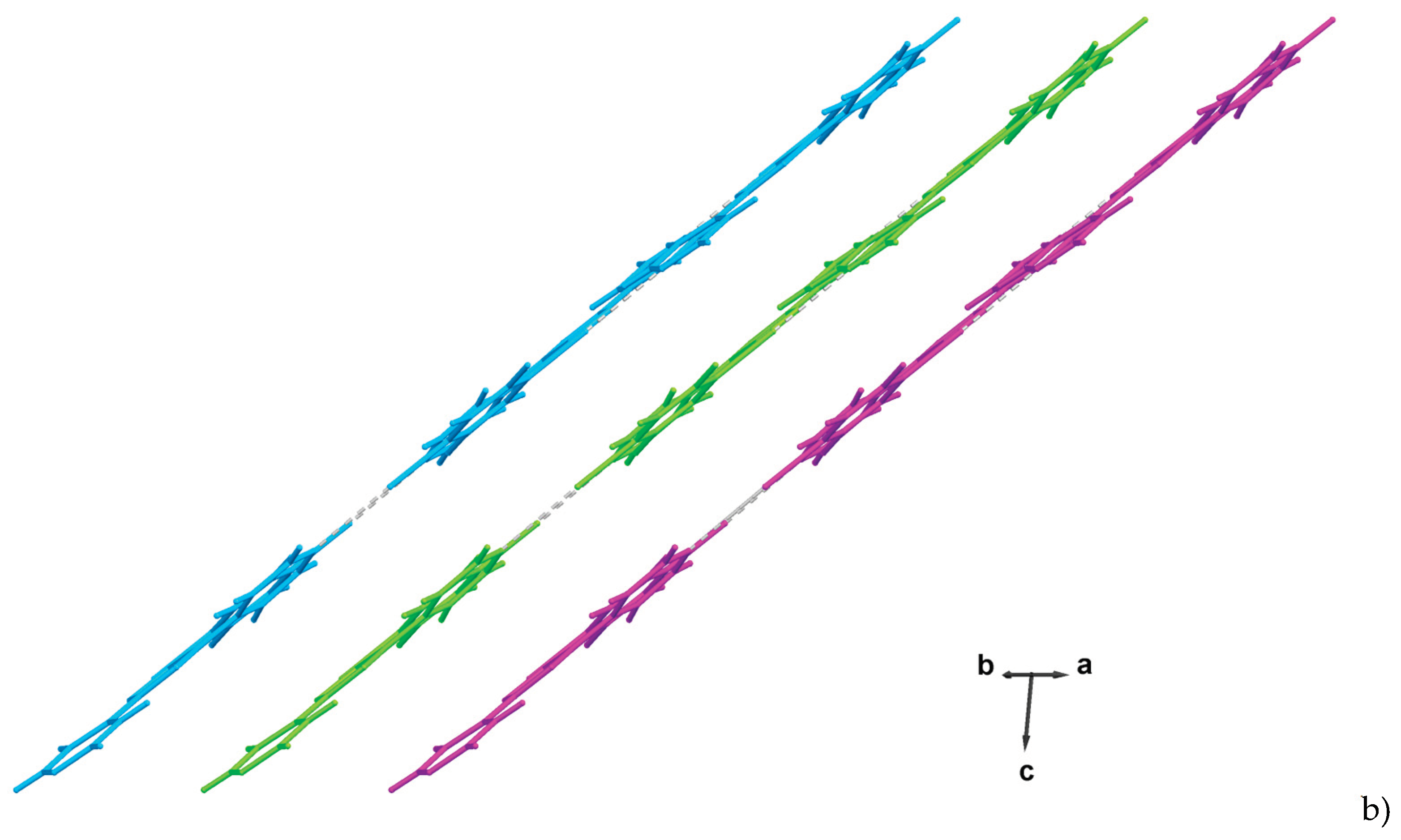
2. Results
3. Materials and Methods
3.1. General
3.2. Synthesis of (L1)(1,4-DITFB) (1)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Frija, L.M.T.; Pombeiro, A.J.L.; Kopylovich, M.N. Building 1,2,4-Thiadiazole: Ten Years of Progress. European J Org Chem 2017, 2017 (19), 2670–2682. [CrossRef]
- Iizawa, Y.; Okonogi, K.; Hayashi, R.; Iwahi, T.; Yamazaki, T.; Imada, A. Therapeutic Effect of Cefozopran (SCE-2787), a New Parenteral Cephalosporin, against Experimental Infections in Mice. Antimicrob Agents Chemother 1993, 37 (1), 100–105. [CrossRef]
- Romagnoli, R.; Baraldi, P.G.; Carrion, M.D.; Cruz-Lopez, O.; Preti, D.; Tabrizi, M.A.; Fruttarolo, F.; Heilmann, J.; Bermejo, J.; Estévez, F. Hybrid Molecules Containing Benzo[4,5]Imidazo[1,2-d][1,2,4]Thiadiazole and α-Bromoacryloyl Moieties as Potent Apoptosis Inducers on Human Myeloid Leukaemia Cells. Bioorg Med Chem Lett 2007, 17 (10), 2844–2848. [CrossRef]
- Leung-Toung, R.; Tam, T.F.; Wodzinska, J.M.; Zhao, Y.; Lowrie, J.; Simpson, C.D.; Karimian, K.; Spino, M. 3-Substituted Imidazo[1,2-d][1,2,4]-Thiadiazoles: A Novel Class of Factor XIIIa Inhibitors. J Med Chem 2005, 48 (7), 2266–2269. [CrossRef]
- Perlovich, G.L.; Proshin, A.N.; Volkova, T.V.; Petrova, L.N.; Bachurin, S.O. Novel 1,2,4-Thiadiazole Derivatives as Potent Neuroprotectors: Approach to Creation of Bioavailable Drugs. Mol Pharm 2012, 9 (8), 2156–2167. [CrossRef]
- Martinez, A.; Alonso, M.; Castro, A.; Pérez, C.; Moreno, F.J. First Non-ATP Competitive Glycogen Synthase Kinase 3 β (GSK-3β) Inhibitors: Thiadiazolidinones (TDZD) as Potential Drugs for the Treatment of Alzheimer’s Disease. J Med Chem 2002, 45 (6), 1292–1299. [CrossRef]
- Cashman, J.R.; Hanzlik, R.P. Oxidation and Other Reactions of Thiobenzamide Derivatives of Relevance to Their Hepatotoxicity. J.Org. Chem. 1982, 47 (24), 4645–4650. [CrossRef]
- Castro, A.; Castaño, T.; Encinas, A.; Porcal, W.; Gil, C. Advances in the Synthesis and Recent Therapeutic Applications of 1,2,4-Thiadiazole Heterocycles. Bioorg Med Chem 2006, 14 (5), 1644–1652. [CrossRef]
- Meltzer, R.I.; Lewis, A.D.; King, J.A. Antitubercular Substances. IV. Thioamides. J Am Chem Soc 1955, 77 (15), 4062–4066. [CrossRef]
- Aragoni, M.C.; Arca, M.; Coles, S.J.; Crespo Alonso, M.; Coles, S.L.; Davies, R.P.; Hursthouse, M.B.; Isaia, F.; Lai, R.; Lippolis, V. Coordination Polymers and Polygons Using Di-Pyridyl-Thiadiazole Spacers and Substituted Phosphorodithioato Ni II Complexes: Potential and Limitations for Inorganic Crystal Engineering. CrystEngComm 2016, 18 (30), 5620–5629. [CrossRef]
- Podda, E.; Arca, M.; Coles, S.J.; Crespo Alonso, M.; Isaia, F.; Pintus, A.; Lippolis, V.; Aragoni, M.C. Supramolecular Assemblies Tailored by Dipyridyl-1,2-4-Thiadiazoles: Influence of the Building Blocks in the Predictability of the Final Network. Supramol Chem 2020, 32 (4). [CrossRef]
- Aragoni, M.C.; Arca, M.; Caltagirone, C.; Castellano, C.; Demartin, F.; Garau, A.; Isaia, F.; Lippolis, V.; Montis, R.; Pintus, A. Cationic and Anionic 1D Chains Based on NH+⋯N Charge-Assisted Hydrogen Bonds in Bipyridyl Derivatives and Polyiodides. CrystEngComm 2012, 14 (18), 5809–5823. [CrossRef]
- Peloquin, A.J.; McMillen, C.D.; Pennington, W.T. Isolation of Unique Heterocycles Formed from Pyridine-Thiocarboxamides as Diiodine, Iodide, or Polyiodide Salts. CrystEngComm 2022, 24 (35), 6251–6261. [CrossRef]
- Wang, H.; Jin, W.J. Cocrystal Assembled by 1,4-Diiodotetrafluorobenzene and Phenothiazine Based on C - I...π/N/S Halogen Bond and Other Assisting Interactions. Acta Cryst. B 2017, 73 (2), 210–216. [CrossRef]
- Aragoni, M.C.; Podda, E.; Chaudhary, S.; Bhasin, A.K.K.; Bhasin, K.K.; Coles, S.J.; Orton, J.B.; Isaia, F.; Lippolis, V.; Pintus, A.; Slawin, A.M.Z.; Woollins, J.D.; Arca, M. An Experimental and Theoretical Insight into I2/Br2 Oxidation of Bis(Pyridin-2-Yl)Diselane and Ditellane. Chem Asian J 2023, 18 (23), e202300836. [CrossRef]
- Aragoni, M.C.; Podda, E.; Arca, M.; Pintus, A.; Lippolis, V.; Caltagirone, C.; Bartz, R.H.; Lenardão, E.J.; Perin, G.; Schumacher, R.F.; Coles, S.J.; Orton, J.B. An Unprecedented Non-Classical Polyinterhalogen Anion Made of [I2Cl]− and I2 at the 2-(p-Tolyl)Selenopheno[2,3-b]Pyridinium Cation Template. New J. Chem. 2022, 46 (45), 21921–21929. [CrossRef]
- Thalladi, V. R.; Weiss, H.C.; Bläser, D.; Boese, R.; Nangia, A.; Desiraju, G.R. C−H···F Interactions in the Crystal Structures of Some Fluorobenzenes. J. Am. Chem. Soc. 1998, 120, 34, 8702–8710. [CrossRef]
- Rigaku Oxford Diffraction (2015), CrysAlisPro. version 1.171.39.8d. Rigaku Corporation, Tokyo, Japan.
- Sheldrick, G.M. SHELXT - Integrated Space-Group and Crystal-Structure Determination. Acta Cryst. A 2015, 71 (1), 3–8. [CrossRef]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Cryst. C 2015, 71 (1), 3–8. [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Cryst. 2009, 42 (2), 339–341. [CrossRef]
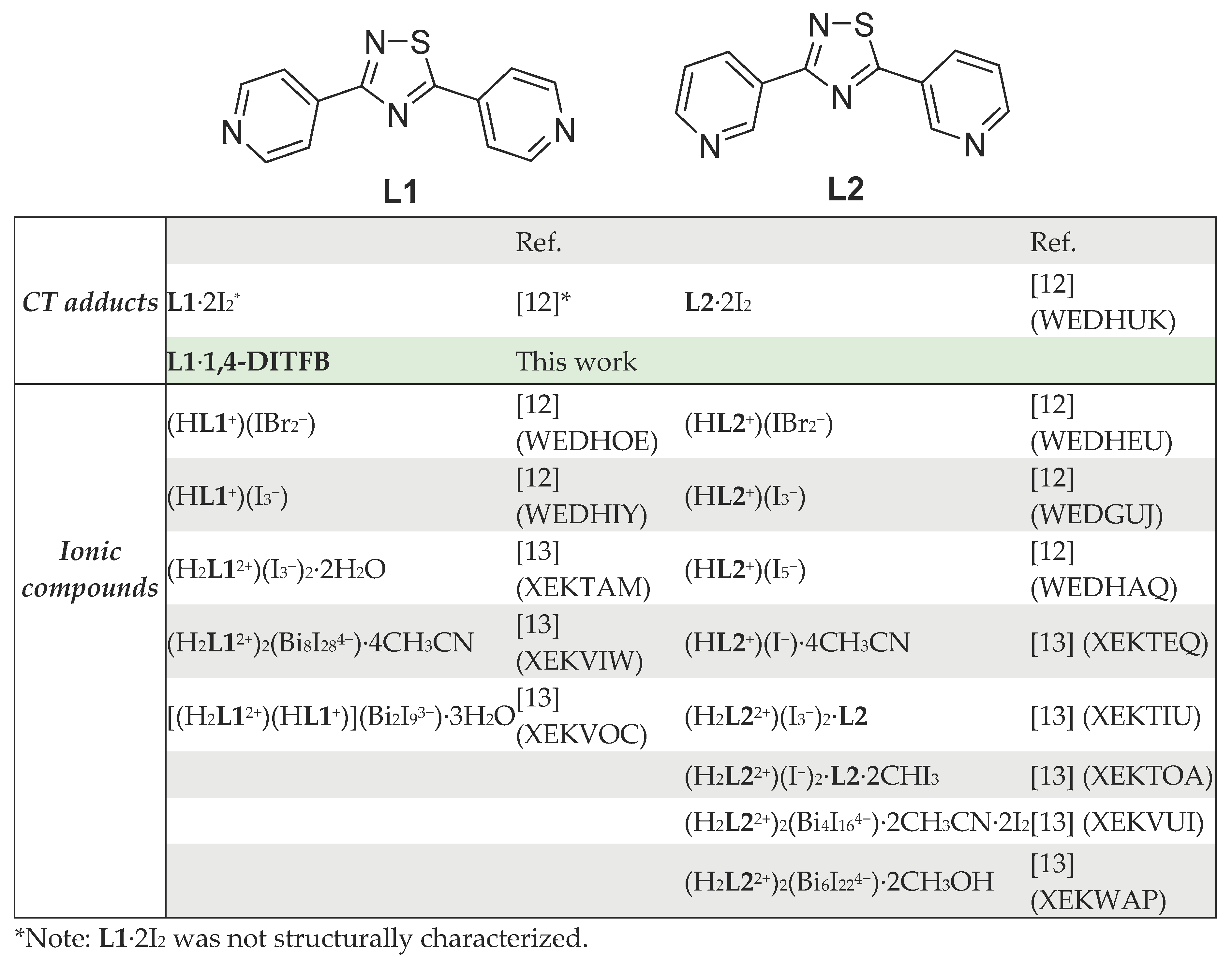
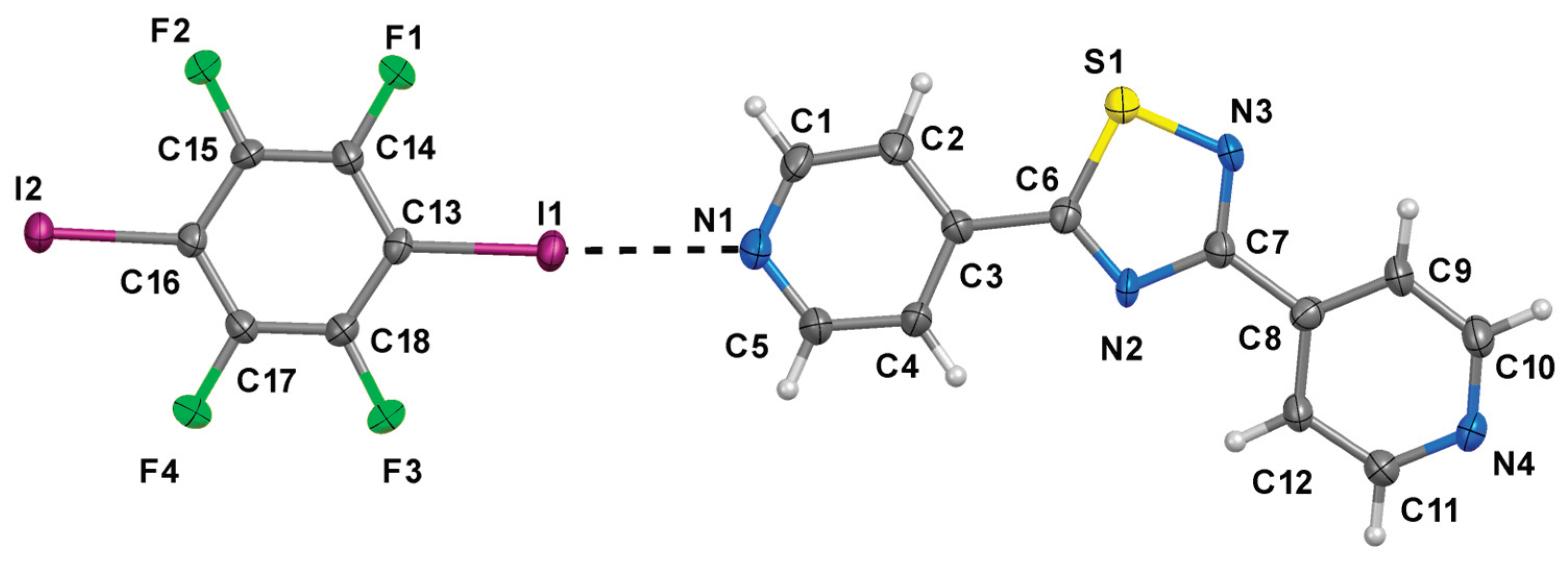
| C‒I∙∙∙N | dC–I (Å) | dI∙∙∙N (Å) | αC–I···N (°) | ||
| a | C13‒I1∙∙∙N1 | 2.101(5) | 2.801(5) | 177.4(2) | |
| b | C16i‒I2i∙∙∙N4 | 2.092(5) | 2.947(4) | 168.3(2) | |
| C‒H∙∙∙F | dC–H (Å) | dH∙∙∙F (Å) | dC∙∙∙F (Å) | αC–H···F (°) | |
| c | C2‒H2∙∙∙F2ii | 0.95 | 2.450 | 3.307(6) | 150 |
| d | C4‒H4∙∙∙F3iii | 0.95 | 2.607 | 3.142(6) | 122 |
| e | C5‒H5∙∙∙F3iii | 0.95 | 2.505 | 3.111(6) | 116 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




