Submitted:
28 February 2024
Posted:
29 February 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Antibodies
2.2. Plasmids
2.3. Cell culture and transfections.
2.4. Cell viability assay
2.5. Infectivity assays.
2.6. Synthetic HPV particle production and labeling.
2.7. Fusion protein purification and binding assays.
2.8. Mass spectrometry analysis.
3. Results
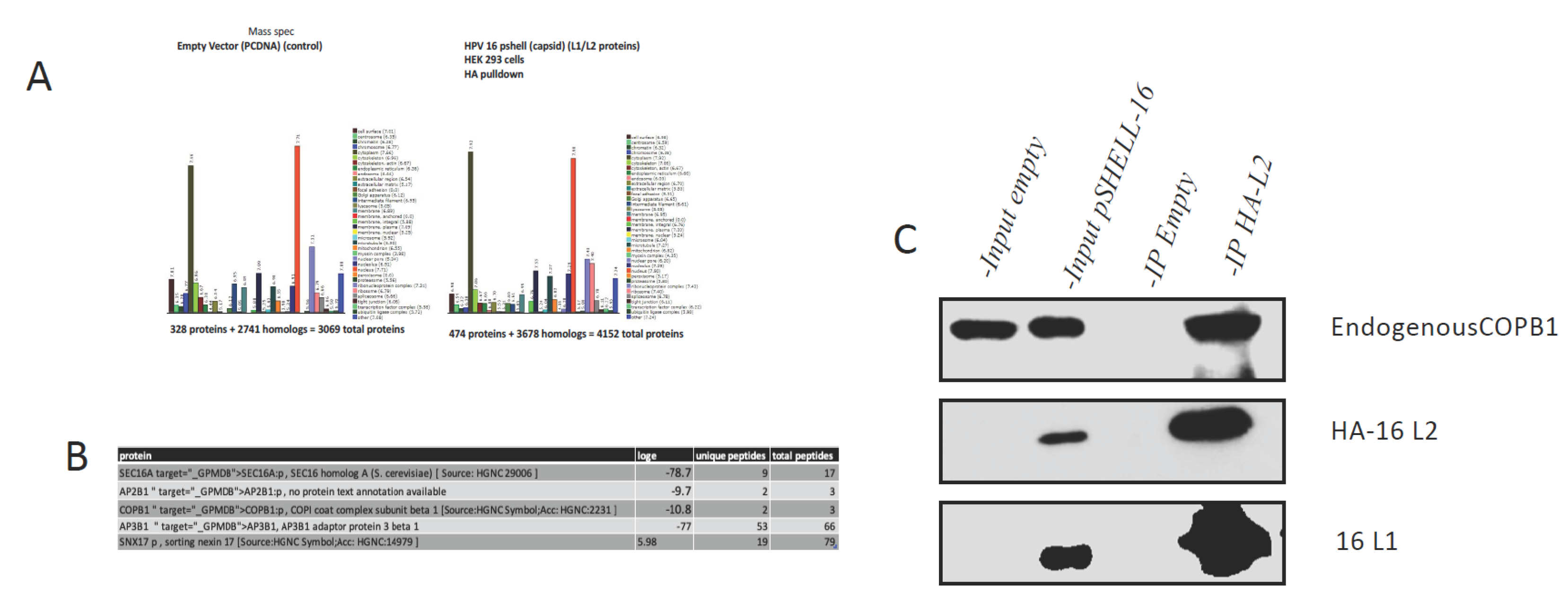
3.1. Mass spectrometry assay identifies COPB1 cargo protein as an HPV-16 interacting protein
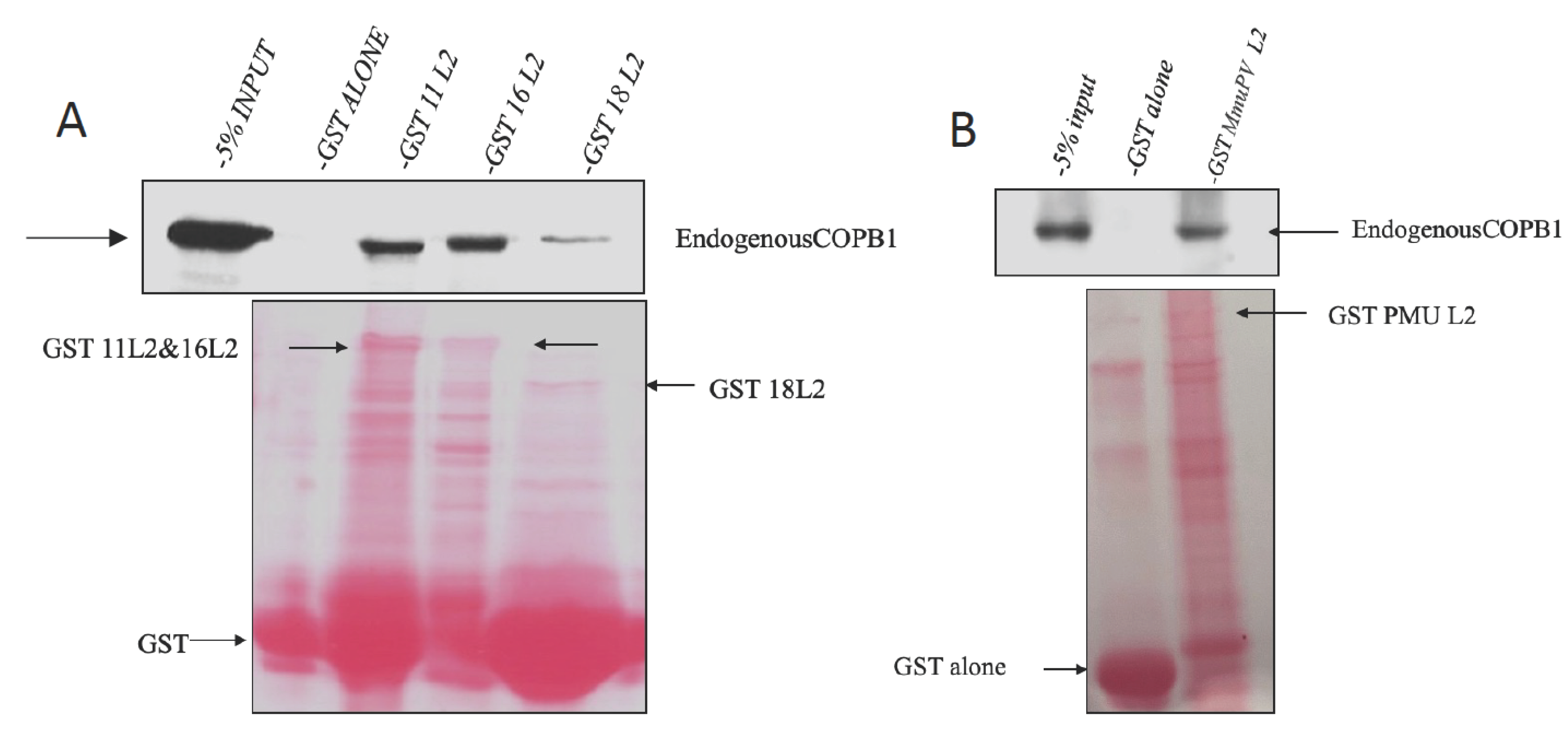
3.2. GST-L2 fusion protein binds COPB1 in vitro
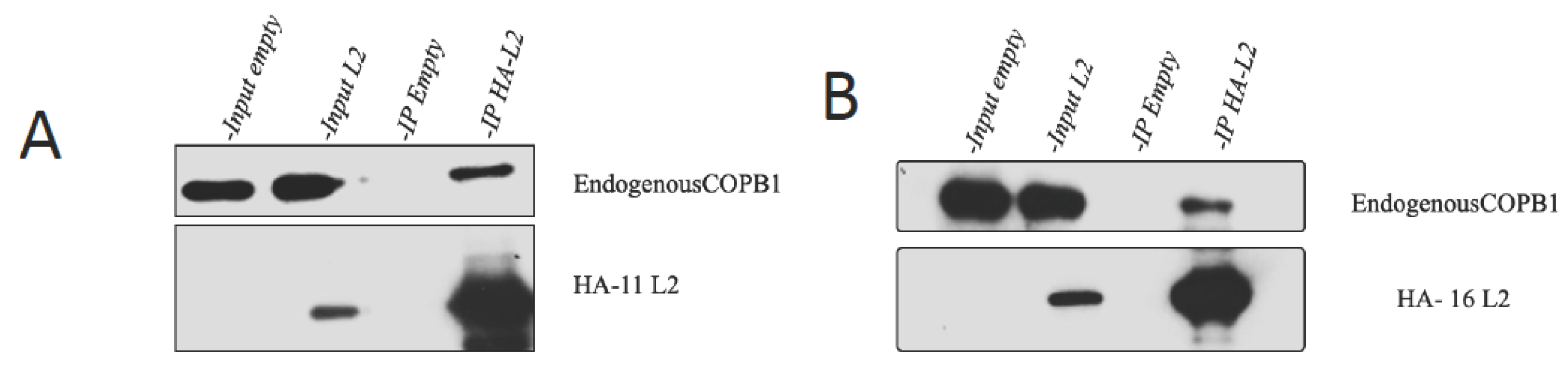
3.3. HPV-16 AND HPV-11 L2 interact with COPB1 in vivo.
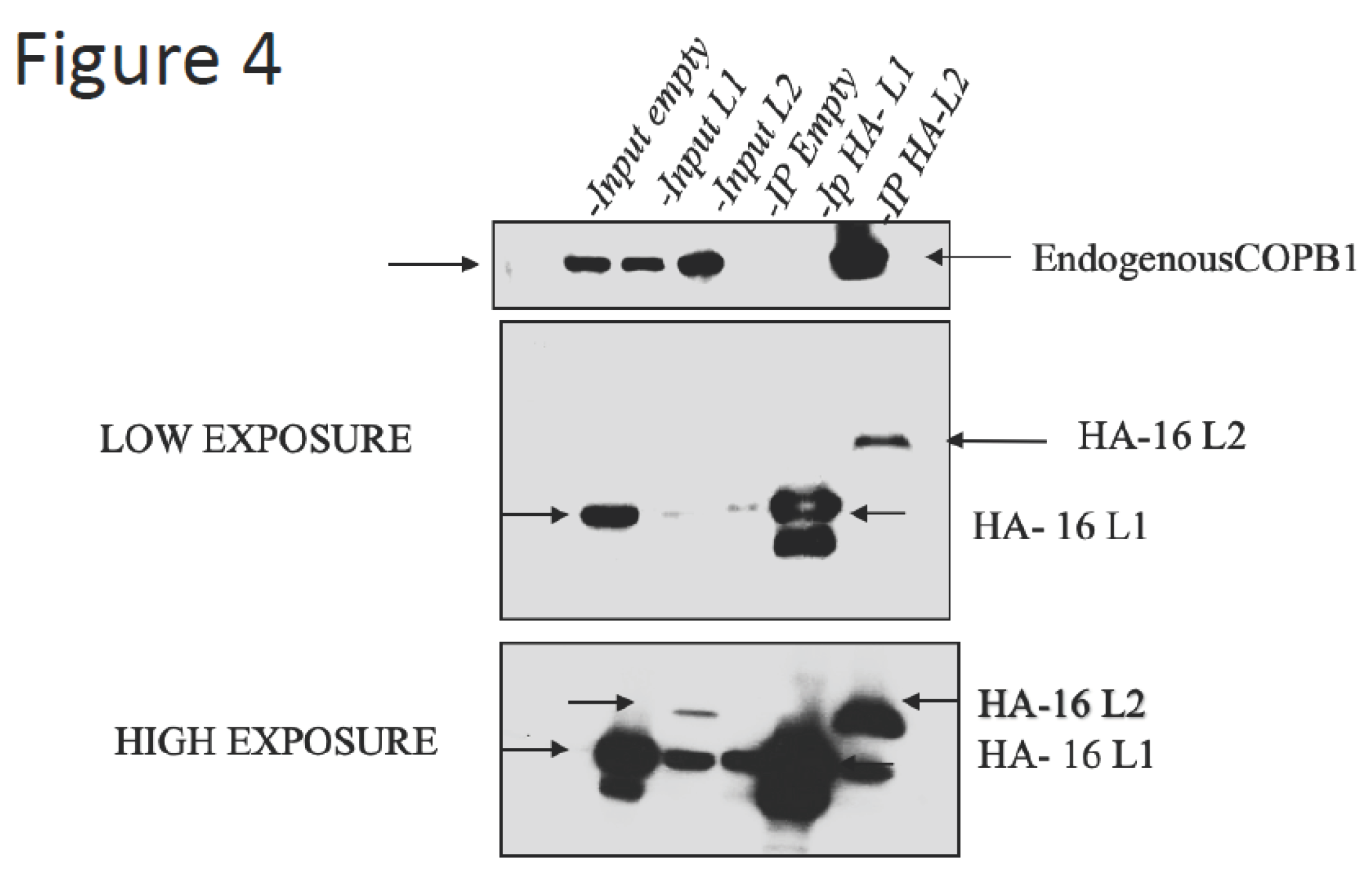
3.4. HPV-16 L2, but not L1, binds to COPB1.
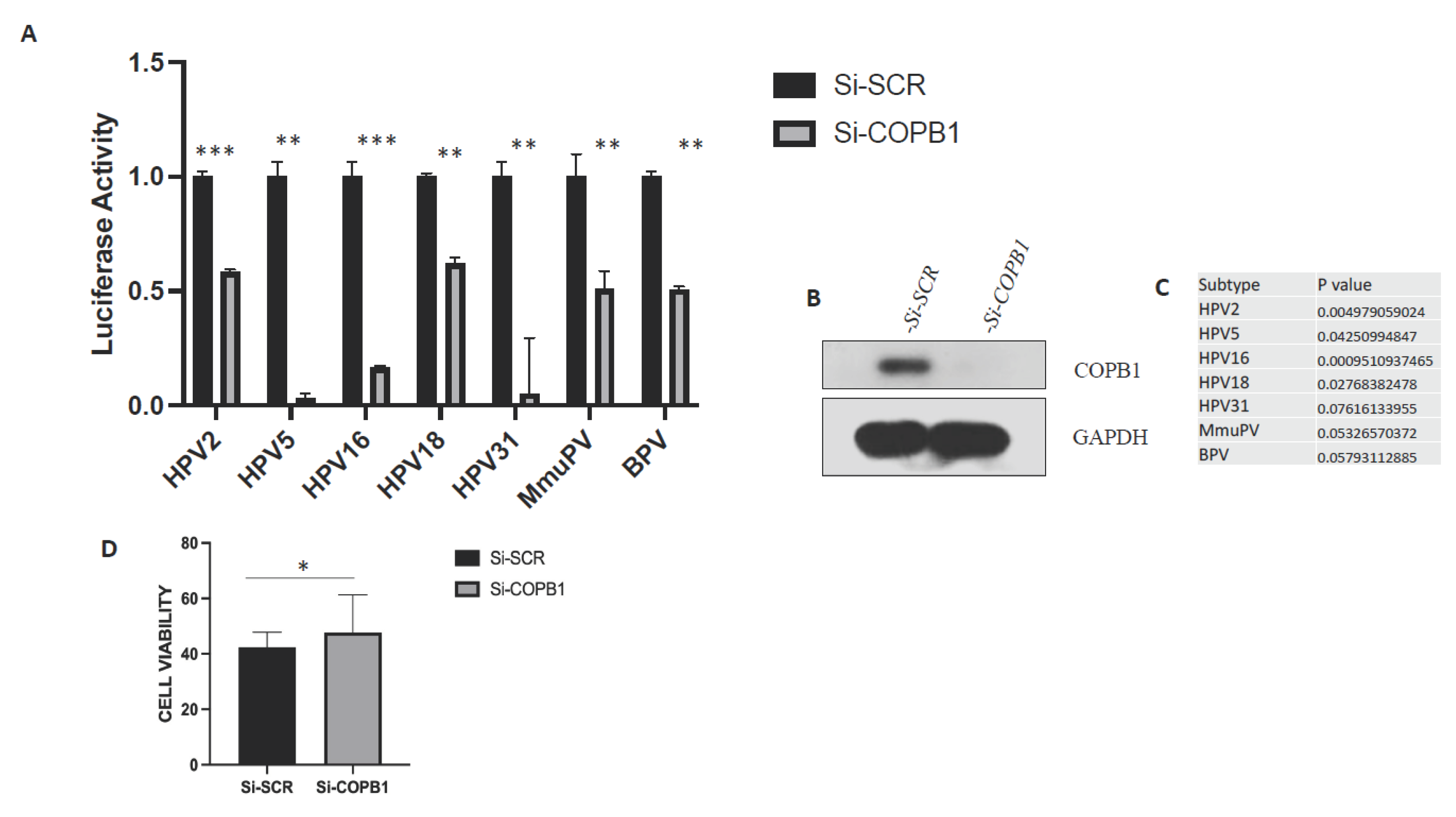
3.5. HPV infection requires COPB1.
4. Discussion
References
- Schelhaas, M.; Shah, B.; Holzer, M.; Blattmann, P.; Kühling, L.; Day, P.M.; Schiller, J.T.; Helenius, A. Entry of Human Papillomavirus Type 16 by Actin-Dependent, Clathrin- and Lipid Raft-Independent Endocytosis. PLOS Pathog. 2012, 8, e1002657. [Google Scholar] [CrossRef] [PubMed]
- Aksoy, P.; Gottschalk, E.Y.; Meneses, P.I. HPV entry into cells. Mutat. Res. Mol. Mech. Mutagen. 2017, 772, 13–22. [Google Scholar] [CrossRef]
- Day, P.M.; Thompson, C.D.; Schowalter, R.M.; Lowy, D.R.; Schiller, J.T. Identification of a Role for the trans -Golgi Network in Human Papillomavirus 16 Pseudovirus Infection. J. Virol. 2013, 87, 3862–3870. [Google Scholar] [CrossRef]
- Broniarczyk, J.; Massimi, P.; Pim, D.; Marušič, M.B.; Myers, M.P.; Garcea, R.L.; Banks, L. Phosphorylation of Human Papillomavirus Type 16 L2 Contributes to Efficient Virus Infectious Entry. J. Virol. 2019, 93, e00128-19. [Google Scholar] [CrossRef] [PubMed]
- Duden, R. ER-to-Golgi transport: COP I and COP II function (Review). Mol. Membr. Biol. 2003, 20, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Duden, R. ER-to-Golgi transport: COP I and COP II function (Review). Mol. Membr. Biol. 2003, 20, 197–207. [Google Scholar] [CrossRef]
- Gomez-Navarro, N.; Miller, E.A. COP-coated vesicles. Current Biology 2016, 26, R54–R57. [Google Scholar] [CrossRef]
- Bonifacino, J.S.; Lippincott-Schwartz, J. Coat proteins: shaping membrane transport. Nat. Rev. Mol. Cell Biol. 2003, 4, 409–414. [Google Scholar] [CrossRef]
- Basukala, O.; Trejo-Cerro, O.; Myers, M.P.; Pim, D.; Massimi, P.; Thomas, M.; Guarnaccia, C.; Owen, D.; Banks, L. HPV-16 E7 Interacts with the Endocytic Machinery via the AP2 Adaptor μ2 Subunit. mBio 2022, 13, e0230222. [Google Scholar] [CrossRef]
- Shin, J.; Nile, A.; Oh, J.-W. Role of adaptin protein complexes in intracellular trafficking and their impact on diseases. Bioengineered 2021, 12, 8259–8278. [Google Scholar] [CrossRef]
- Park, S.Y.; Guo, X. Adaptor protein complexes and intracellular transport. Biosci. Rep. 2014, 34, 381–390. [Google Scholar] [CrossRef]
- Duden, R. ER-to-Golgi transport: COP I and COP II function (Review). Mol. Membr. Biol. 2003, 20, 197–207. [Google Scholar] [CrossRef]
- Day, P.M.; Thompson, C.D.; Schowalter, R.M.; Lowy, D.R.; Schiller, J.T. Identification of a Role for the trans -Golgi Network in Human Papillomavirus 16 Pseudovirus Infection. J. Virol. 2013, 87, 3862–3870. [Google Scholar] [CrossRef]
- Valdano, M.B.; Massimi, P.; Broniarczyk, J.; Pim, D.; Myers, M.; Gardiol, D.; Banks, L. Human Papillomavirus Infection Requires the CCT Chaperonin Complex. J. Virol. 2021, 95, e01943-20. [Google Scholar] [CrossRef]
- Pim, D.; Broniarczyk, J.; Siddiqa, A.; Massimi, P.; Banks, L. Human Papillomavirus 16 L2 Recruits both Retromer and Retriever Complexes during Retrograde Trafficking of the Viral Genome to the Cell Nucleus. J. Virol. 2021, 95, e02068-20. [Google Scholar] [CrossRef] [PubMed]
- Peden, A.A.; Oorschot, V.; Hesser, B.A.; Austin, C.D.; Scheller, R.H.; Klumperman, J. Localization of the AP-3 adaptor complex defines a novel endosomal exit site for lysosomal membrane proteins. J. Cell Biol. 2004, 164, 1065–1076. [Google Scholar] [CrossRef]
- DiGiuseppe, S.; Keiffer, T.R.; Bienkowska-Haba, M.; Luszczek, W.; Guion, L.G.M.; Müller, M.; Sapp, M. Topography of the Human Papillomavirus Minor Capsid Protein L2 during Vesicular Trafficking of Infectious Entry. J. Virol. 2015, 89, 10442–10452. [Google Scholar] [CrossRef] [PubMed]
- Tsakogiannis, D.; Nikolaidis, M.; Zagouri, F.; Zografos, E.; Kottaridi, C.; Kyriakopoulou, Z.; Tzioga, L.; Markoulatos, P.; Amoutzias, G.D.; Bletsa, G. Mutation Profile of HPV16 L1 and L2 Genes in Different Geographic Areas. Viruses 2022, 15, 141. [Google Scholar] [CrossRef]
- Schoppe, J.; Schubert, E.; Apelbaum, A.; Yavavli, E.; Birkholz, O.; Stephanowitz, H.; Han, Y.; Perz, A.; Hofnagel, O.; Liu, F.; et al. Flexible open conformation of the AP-3 complex explains its role in cargo recruitment at the Golgi. J. Biol. Chem. 2021, 297, 101334. [Google Scholar] [CrossRef]
- McMahon, H.T.; Mills, I.G. COP and clathrin-coated vesicle budding: different pathways, common approaches. Curr. Opin. Cell Biol. 2004, 16, 379–391. [Google Scholar] [CrossRef]
- Beck, R.; Ravet, M.; Wieland, F.; Cassel, D. The COPI system: Molecular mechanisms and function. FEBS Lett. 2009, 583, 2701–2709. [Google Scholar] [CrossRef]
- Drake, M.T.; Zhu, Y.; Kornfeld, S. The Assembly of AP-3 Adaptor Complex-containing Clathrin-coated Vesicles on Synthetic Liposomes. Mol. Biol. Cell 2000, 11, 3723–3736. [Google Scholar] [CrossRef]
- Darsow, T.; Burd, C.G.; Emr, S.D. Acidic Di-leucine Motif Essential for AP-3–dependent Sorting and Restriction of the Functional Specificity of the Vam3p Vacuolar t-SNARE. J. Cell Biol. 1998, 142, 913–922. [Google Scholar] [CrossRef]
- Broniarczyk, J.; Massimi, P.; Bergant, M.; Banks, L. Human Papillomavirus Infectious Entry and Trafficking Is a Rapid Process. J. Virol. 2015, 89, 8727–8732. [Google Scholar] [CrossRef]
- Buck, C.B.; Pastrana, D.V.; Lowy, D.R.; Schiller, J.T. Generation of HPV pseudovirions using transfection and their use in neutralization assays. Methods Mol. Med. 2005, 119, 445–462. [Google Scholar]
- Graham, F.L.; Van Der Eb, A.J. A new technique for the assay of infectivity of human adenovirus 5 DNA. Virology 1973, 52, 456–467. [Google Scholar] [CrossRef]
- Marušič, M.B.; Ozbun, M.A.; Campos, S.K.; Myers, M.P.; Banks, L. Human Papillomavirus L2 Facilitates Viral Escape from Late Endosomes via Sorting Nexin 17. Traffic 2011, 13, 455–467. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Kazakov, T.; Popa, A.; DiMaio, D. Vesicular Trafficking of Incoming Human Papillomavirus 16 to the Golgi Apparatus and Endoplasmic Reticulum Requires γ-Secretase Activity. mBio 2014, 5, e01777–14. [Google Scholar] [CrossRef] [PubMed]
- Siddiqa, A.; Massimi, P.; Pim, D.; Broniarczyk, J.; Banks, L. Human Papillomavirus 16 Infection Induces VAP-Dependent Endosomal Tubulation. J. Virol. 2018, 92. [Google Scholar] [CrossRef] [PubMed]
- Marušič, M.B.; Mencin, N.; Ličen, M.; Banks, L.; Grm, H.S. Modification of Human Papillomavirus Minor Capsid Protein L2 by Sumoylation. J. Virol. 2010, 84, 11585–11589. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.; Massimi, P.; Jenkins, J.; Banks, L. HPV-18 E6 mediated inhibition of p53 DNA binding activity is independent of E6 induced degradation. Oncogene 1995, 10, 261–8. [Google Scholar]
- Buck, C.B.; Pastrana, D.V.; Lowy, D.R.; Schiller, J.T. Generation of HPV pseudovirions using transfection and their use in neutralization assays. Methods Mol. Med. 2005, 119, 445–462. [Google Scholar]
- Valdano, M.B.; Massimi, P.; Broniarczyk, J.; Pim, D.; Myers, M.; Gardiol, D.; Banks, L. Human Papillomavirus Infection Requires the CCT Chaperonin Complex. J. Virol. 2021, 95, e01943-20. [Google Scholar] [CrossRef]
- Bergant, M.; Banks, L. SNX17 Facilitates Infection with Diverse Papillomavirus Types. J. Virol. 2013, 87, 1270–1273. [Google Scholar] [CrossRef]
- Matsuoka, K.; Morimitsu, Y.; Uchida, K.; Schekman, R. Coat Assembly Directs v-SNARE Concentration into Synthetic COPII Vesicles. Mol. Cell 1998, 2, 703–708. [Google Scholar] [CrossRef]
- Xie, J.; Zhang, P.; Crite, M.; Lindsay, C.V.; DiMaio, D. Retromer stabilizes transient membrane insertion of L2 capsid protein during retrograde entry of human papillomavirus. Sci. Adv. 2021, 7. [Google Scholar] [CrossRef]
- Duden, R. ER-to-Golgi transport: COP I and COP II function (Review). Mol. Membr. Biol. 2003, 20, 197–207. [Google Scholar] [CrossRef]
- Gomez-Navarro, N.; Miller, E.A. COP-coated vesicles. Curr. Biol. 2016, 26, R54–R57. [Google Scholar] [CrossRef] [PubMed]
- Szymonowicz, K.A.; Chen, J. Biological and clinical aspects of HPV-related cancers. Cancer Biol. Med. 2020, 17, 864–878. [Google Scholar] [CrossRef] [PubMed]
- Marur, S.; D'Souza, G.; Westra, W.H.; Forastiere, A.A. HPV-associated head and neck cancer: a virus-related cancer epidemic. Lancet Oncol. 2010, 11, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.K.; Aimagambetova, G.; Ukybassova, T.; Kongrtay, K.; Azizan, A. Human Papillomavirus Infection and Cervical Cancer: Epidemiology, Screening, and Vaccination—Review of Current Perspectives. J. Oncol. 2019, 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Skoulakis, A.; Fountas, S.; Mantzana-Peteinelli, M.; Pantelidi, K.; Petinaki, E. Prevalence of human papillomavirus and subtype distribution in male partners of women with cervical intraepithelial neoplasia (CIN): a systematic review. BMC Infect. Dis. 2019, 19, 192. [Google Scholar] [CrossRef] [PubMed]
- Horvath, C.A.; Boulet, G.A.; Renoux, V.M.; O Delvenne, P.; Bogers, J.-P.J. Mechanisms of cell entry by human papillomaviruses: an overview. Virol. J. 2010, 7, 11–11. [Google Scholar] [CrossRef] [PubMed]
- Siddiqa, A.; Broniarczyk, J.; Banks, L. Papillomaviruses and Endocytic Trafficking. Int. J. Mol. Sci. 2018, 19, 2619. [Google Scholar] [CrossRef] [PubMed]
- Morante, A.V.; Baboolal, D.D.; Simon, X.; Pan, E.C.-Y.; Meneses, P.I. Human Papillomavirus Minor Capsid Protein L2 Mediates Intracellular Trafficking into and Passage beyond the Endoplasmic Reticulum. Microbiol. Spectr. 2022, 10, e0150522. [Google Scholar] [CrossRef]
- Calton, C.M.; Bronnimann, M.P.; Manson, A.R.; Li, S.; Chapman, J.A.; Suarez-Berumen, M.; Williamson, T.R.; Molugu, S.K.; Bernal, R.A.; Campos, S.K. Translocation of the papillomavirus L2/vDNA complex across the limiting membrane requires the onset of mitosis. PLOS Pathog. 2017, 13, e1006200. [Google Scholar] [CrossRef]
- Broniarczyk, J.; Ring, N.; Massimi, P.; Giacca, M.; Banks, L. HPV-16 virions can remain infectious for 2 weeks on senescent cells but require cell cycle re-activation to allow virus entry. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Siddiqa, A.; Broniarczyk, J.; Banks, L. Papillomaviruses and Endocytic Trafficking. Int. J. Mol. Sci. 2018, 19, 2619. [Google Scholar] [CrossRef] [PubMed]
- DiGiuseppe, S.; Bienkowska-Haba, M.; Guion, L.G.M.; Keiffer, T.R.; Sapp, M. Human Papillomavirus Major Capsid Protein L1 Remains Associated with the Incoming Viral Genome throughout the Entry Process. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Day, P.M.; Weisberg, A.S.; Thompson, C.D.; Hughes, M.M.; Pang, Y.Y.; Lowy, D.R.; Schiller, J.T. Human Papillomavirus 16 Capsids Mediate Nuclear Entry during Infection. J. Virol. 2019, 93. [Google Scholar] [CrossRef]
- Peotter, J.; Kasberg, W.; Pustova, I.; Audhya, A. COPII-mediated trafficking at the ER/ERGIC interface. Traffic 2019, 20, 491–503. [Google Scholar] [CrossRef]
- Sohn, K.; Orci, L.; Ravazzola, M.; Amherdt, M.; Bremser, M.; Lottspeich, F.; Fiedler, K.; Helms, J.B.; Wieland, F.T. A major transmembrane protein of Golgi-derived COPI-coated vesicles involved in coatomer binding. J. Cell Biol. 1996, 135, 1239–1248. [Google Scholar] [CrossRef] [PubMed]
- Szul, T.; Sztul, E. COPII and COPI Traffic at the ER-Golgi Interface. Physiology 2011, 26, 348–364. [Google Scholar] [CrossRef] [PubMed]
- A Ozbun, M.; Campos, S.K. The long and winding road: human papillomavirus entry and subcellular trafficking. Curr. Opin. Virol. 2021, 50, 76–86. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



