1. Introduction
Nowadays, fossil fuels are the major energy source for the humanity. The combustion of fossil fuels will produce carbon dioxide and lead to greenhouse effect. With the shortage of traditional fossil fuels and the aggravation of environmental problems, it is pivotal to develop clean, cheap, convenient, and sustainable alternative energy.
Hydrogen energy is considered as an ideal substitute to the conventional fuels, due to its widespread sources, high calorific value of combustion, and zero carbon emissions [
1]. Currently, approximately 95% of the world’s hydrogen is produced from the natural gas and fossil fuel through an energy-intensive process [
2]. Compared with traditional technologies, electrocatalytic water-splitting systems can realize using renewable energy (e.g., solar energy) as power for uninterrupted and efficient hydrogen production, have gained ever-rising attention in recent years [
3].
The electrocatalytic water-splitting comprises a cathodic hydrogen evolution reaction (HER) and an anodic oxygen evolution reaction (OER). The efficiency and activity of the water-splitting system rely on the electrocatalysts. To date, only less than 4% of the hydrogen is produced by the electrocatalytic water-splitting systems [
4], due to the high cost and unsatisfied economy benefits compared with conventional fossil energy production. Currently, metal-based electrocatalysts are widely applied in both HER and OER. For example, Pt group electrocatalysts are regarded as the benchmark for HER because they have excellent activity in both acid and alkaline media [
5]. However, the high price and natural scarcity of noble metals hinder their practical applications as water-splitting electrocatalysts. Reducing noble-metal content of the metal-based electrocatalysts is an effective and straightforward way to reduce the cost. When the noble-metal content is reduced, the utilization of metal atoms should be increased to maintain the intrinsic activity. Therefore, many studies focus on increasing metal centers with low coordination by reducing the size of nanoparticle catalysts. In these studies, atomic-level catalysts have become a hotspot.
Single-atom catalysts (SACs) is a kind of catalyst which have atomic active sites dispersed on the surface of supports. By merit of maximum atom utilization and adjustable coordination, it has been widely applied in the field of electrocatalysis since it was first reported by Zhang et al. in 2011 [
6]. Whereas, SACs often suffers from the predicament of low loading (<1.5wt%), which may limit the overall activity. Furthermore, some multi-electron-proton coupling reactions are limited at single-atom sites, the random dispersion of SACs leads to an unsatisfied synergy.
As an extension of SAC, heteronuclear dual-atom catalysts (DACs) with multi-metals co-doped have received ever-rising attention in recent years. It retains the advantages of SACs and overcome the limitations of SACs, which can increase the metal loading, break the linear scaling relations [
7] and adjust electronic structure of the catalysts [
8]. Meanwhile, the cooperation between the two metal elements of DACs can create a synergistic effect to enhance the intermediates adsorption and modulate reaction pathways due to the chemical/structural flexibility of active sites [
9].
Although numerous high-quality reviews have focused on DACs, including definition of the DACs, the micro-structures, and different applications of DACs in various fields, making it become the next frontier after SACs [
10,
11,
12,
13,
14], the discussion on the design of heterogeneous DACs for water-splitting is not sufficient. Therefore, it is valuable to review the progress of DACs in water-splitting reaction.
In this review, we present a comprehensive overview of the rational design and development of heterogeneous DACs in water-splitting reactions. First, we briefly introduce the typical synthetic strategies for heterogeneous DACs. Subsequently, the typical characterizations of DACs are summarized. Then, the recent experimental advances in water-splitting reactions are discussed in detail. Finally, this review provides a summary of the problems and challenges regarding the water-splitting technologies.
2. Synthetic Strategies for DACs
Efficient methods have been explored to synthesize well-defined DACs. Each technique brings its advantages and drawbacks. We will introduce several common approaches of construction DACs for water-splitting reaction.
2.1. High Temperature Pyrolysis
High temperature pyrolysis is a general strategy to synthesize atomic-level reactive sites. The pyrolysis temperature and atmosphere composition are crucial for the formation of DACs. Benefiting from the tunable pore structure and large surface area, metal-organic frameworks (MOFs) and zeolitic imidazolate frameworks (ZIFs) have been widely adopted as precursors to fabricate HER/OER electrocatalysts.
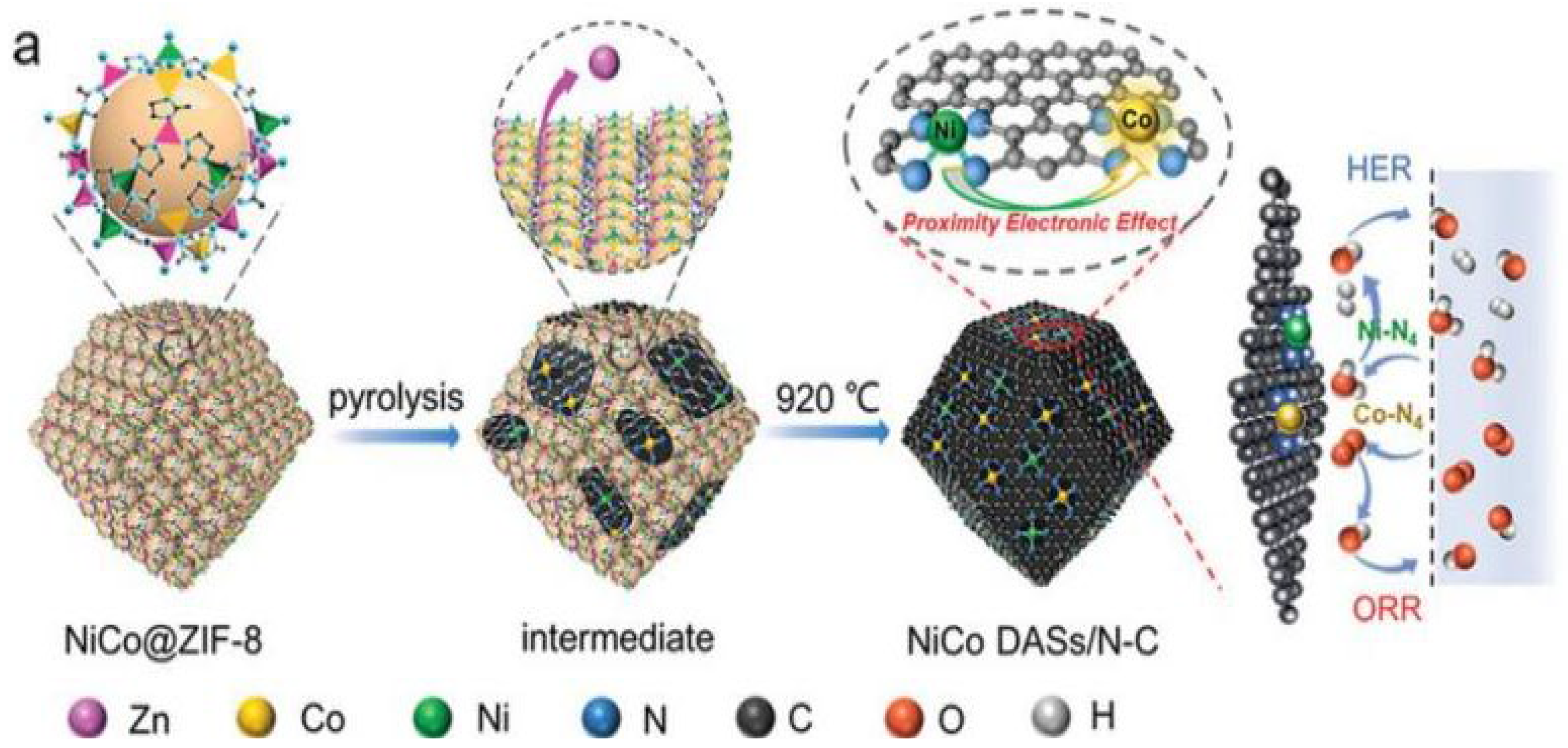
For example, Pan et al. reported a MOF-assisted host-guest strategy to prepare Ni-Co DACs, the Ni and Co atoms served as a guest were encapsulated in the cage of ZIF-8, while the ZIF-8 served as a host with suitable pore size. Then the NiCo@ZIF-8 precursor was pyrolyzed under N
2 at 920 °C to form Ni-N
4 and Co-N
4 sites embedded into 3D carbon substrate, during the process, the ZIF-8 was carbonized to form N-C substrate to provide anchor sites [
15], as shown in
Figure 1.
In another work, Yao et al. prepared ZIF-8 to encapsulate the Cu precursor, followed by co-pyrolysis with the trapped diphenyl diselenide to obtain the Cu-Se DACs, the inner metallic Zn evaporated during the high temperature pyrolysis [
16]. Thus, the metal atoms can be coordinated with N or O atoms in the supporting materials through the pyrolysis.
Besides, carbon black, graphitic g-C
3N
4, and irregular complexes have also been adopted as precursors for synthesize of DACs [
17,
18,
19,
20]. In the study of Jia et al., porous graphitic g-C
3N
4 was synthesized as precursors, which is an appropriate candidate for bonding and stabilizing metal ions efficiently and avoid the sites migration during the carbonization [
18]. Then Ni precursor, polydopamine, and phthalocyanine iron were coated on the surface of porous graphitic g-C
3N
4 step by step. After pyrolyzing under 900 °C, the porous graphitic g-C
3N
4 formed porous carbon nanosheets and the Fe-Ni DACs generated. In this process, the g-C
3N
4 was used as a sacrificial template to form the lamellar and porous morphology.
In short, high temperature pyrolysis can regulate the dispersion of metal sites easily, but some active metals may incorporate into the bulk of the substrate materials, which can hardly be accessible during the electrocatalysis. Meantime, undesirable impurities may generate during the pyrolysis, which usually requires an additional etching process.
2.2. Wet-Chemistry Impregnation
The wet-chemistry impregnation method is low cost and straightforward. The DACs were prepared through introducing the precursors to the surface of matrix and then post-treatment, such as reduction or thermal treatment.
For example, Han et al. added moderate amount Co solution and Pt solution into the prepared anatase TiO
2 nanosheets solution (TNS), then the mixture was kept stirred at 60 °C for 10 h to conduct the impregnation process. The precipitates were then collected and annealed to obtain the Co-Pt dimer loaded TNS [
21].
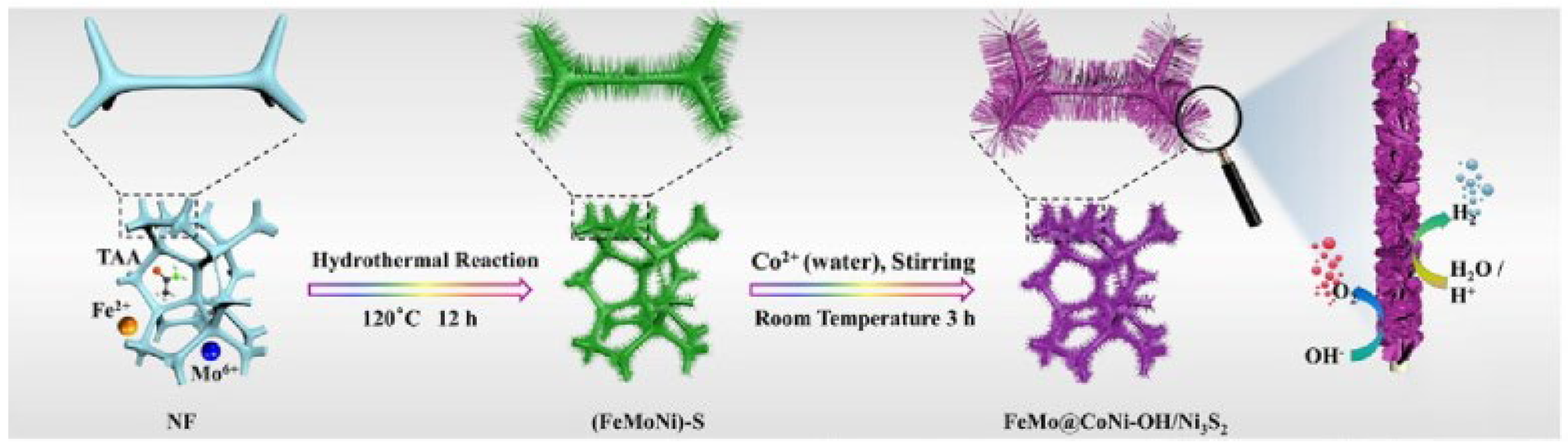
In another work, Li et al. used FeSO
4·7H
2O and Na
2MoO
4·2H
2O solutions as metal precursors, then the as-obtained solution and a piece of Ni foam were transferred to autoclave with Teflon liner [
22]. The reaction was kept at 120 °C for 24 h to synthesize FeMo dual-site catalysts, as shown in
Figure 2.
The wet-chemistry impregnation usually adopts a certain amount of precursor to produce atomically dispersed metal sites, the type and concentration of the precursor can be modulated to prepare desired DACs, however, it is difficult to precisely regulating the location of the active sites and the metal atoms tend to aggregate to form bulk sites.
2.3. Atomic Layer Deposition (ALD)
ALD generally utilizes gas-phase reaction of metal precursors with a solid substrate to synthesize DACs. It is a gas-phase deposition strategy to deposit the thin films onto the substrate accurately.
For example, Hu et al. used (methyl cyclopentadienyl)-platinum (MeCpPtMe
3) and O
3 as precursors, high-purity N
2 as purge gas and carrier gas, N-doped carbon substrate was loaded on aluminum foil and then placed into the ALD chamber. After 50 cycles of ALD, PtNi-NC were obtained [
23]. The ALD technique is beneficial for control the composition and structure of the catalyst, but it requires special equipment and harsh reaction condition.
2.4. Template Assisted
Template assisted method usually utilizes precursors act as a template or pattern to synthesize DACs. For example, SiO
2 template can be etched by NaOH solution, which is applied to construct hollow catalyst. Ma et al. synthesized Ni-N
4 and Fe-N
4 dual single-atomic sites with a template assisted strategy. Firstly, the positively charged SiO
2 nanospheres was adopted as a hard template, then electronegative [Ni(CN
4)]
2- was chosen to form Ni-N
4 single-atom sites. Next, the formed SiO
2@[Ni(CN
4)]
2- intermediate was encapsulated with graphene oxide (GO) to form SiO
2@[Ni(CN
4)]
2-@GO. The iron (II) phthalocyanine (FePc) was then adsorb on the outside of SiO
2@[Ni(CN
4)]
2-@GO to obtain SiO
2@[Ni(CN
4)]
2-@GO@FePc. Thereafter, the SiO
2@[Ni(CN
4)]
2-@GO@FePc was treated at 700 ℃ and etched with NaOH solution to remove the SiO
2 template, eventually leading to the formation of Ni-N
4 and Fe-N
4 dual single-atomic site [
24]. This work successfully synthesizes the Janus hollow spheres derived from different single-atom catalysts respectively on the inner and outer surfaces.
In other works, porous carbon [
25], metal organic framework (MOF) [
26], and salt template [
27] also have been investigated to fabricate the template. It indicates that the template assisted method can synthesize porous micro-structure, but sometimes the removal of some templates may cause damage to the nanostructure [
28].
2.5. Ball-Milling
The ball-milling techniques can mix different precursor at atomic scales homogeneously. For example, Liu et al. treated
Houttuynia cordata to remove inorganic substances and then carbonized. Thereafter, ball milling of the carbon material, copper phthalocyanine (CuPc), and manganese phthalocyanine (MnPc) were conducted under Ar atmosphere. Followed by etching in acid solution, the DACs with N, S-carbon coordinated uniform (Cu, Mn) atom pairs was prepared on a gram-scale. Compared to other methods, it is a clean and easily scaling-up method for preparing heterogeneous catalysts at high productivity of kilogram level [
29].
Except for the above-mentioned methods, ion-exchange [
30], atom trapping [
31], electrochemical method [
32], chemical vapor deposition [
33] are also employed to prepare various DACs. But there are remain some challenges, such as how to synthesize DACs with both high productivity and precise location. Only solve these problems can make the preparation of DACs become more easily and accessible.
3. Characterizations of DACs
Characterizations techniques are imperative for validate the structure-activity relationship of DACs. Tradition characterizations techniques, including scanning electron microscopy (SEM), X-ray diffraction (XRD), and low-magnification transmission electron microscopy (TEM) can provide basic crystal structure of nanomaterials. Thus, advanced characterization is crucial to identify the geometric and electronic configuration of the DACs. Various techniques were employed to confirm the existence of DACs. Here, we mainly focus on two typical techniques, high-resolution aberration corrected high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and X-ray absorption near-edge structure (XANES) spectroscopy, to recognize the nature of DACs.
3.1. High-Angle Annular Dark-Field Scanning Transmission Electron Microscopy (HAADF-STEM)
The morphology and structure of DACs can be directly observed through electron microscopy. Especially, the aberration-corrected (AC) high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) mode is widely applied for the characterization of DACs. It can scatter at high angles and particularly sensitive to heavy atoms, the DACs can be observed straightforwardly from the bright spots on the support. Usually, the heavier metal atoms with a higher Z number display bright dots in dark-field images [
34].
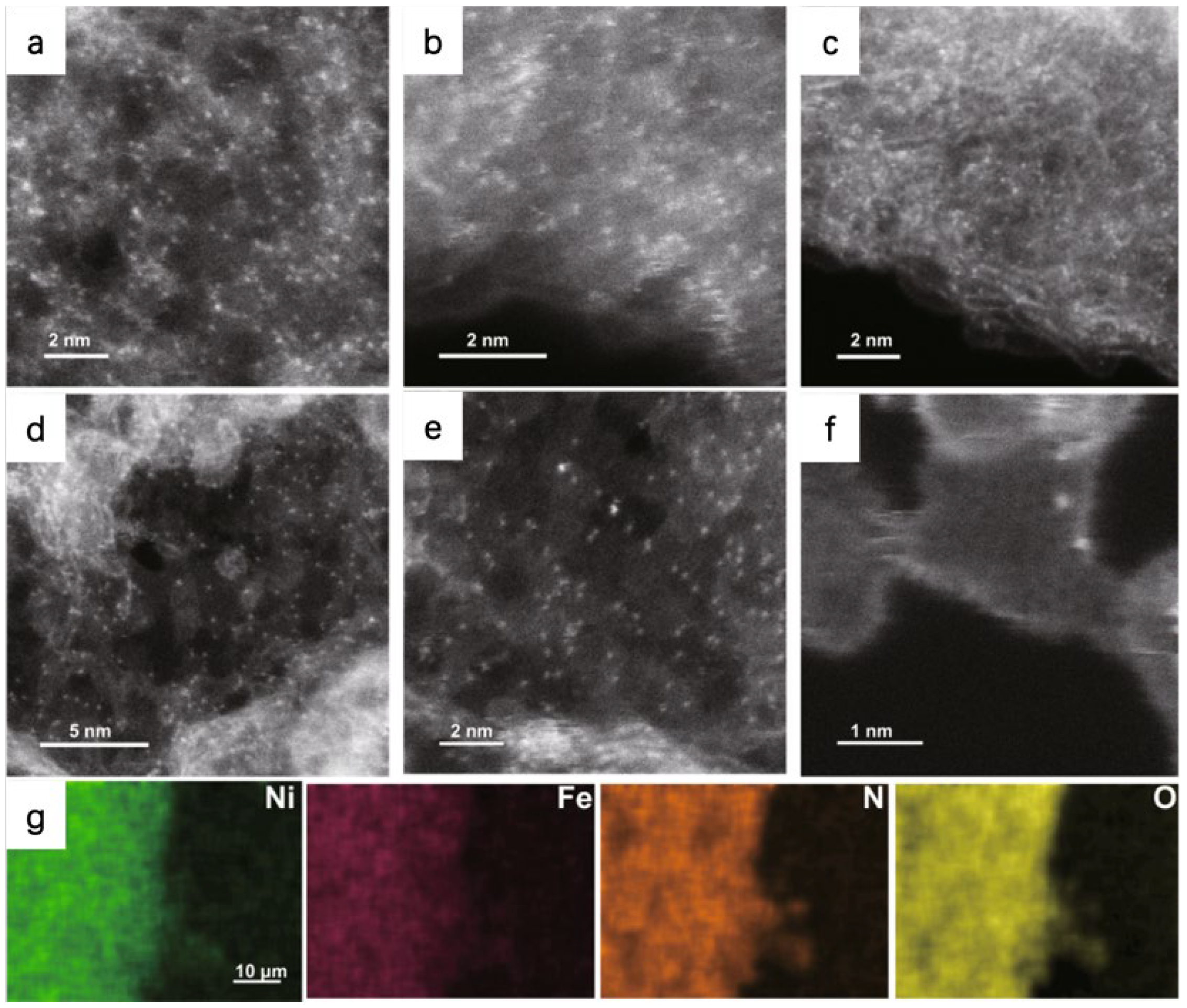
For example, Patzke et al. used the HAADF-STEM to identify the atomic dispersion of Ni and Fe atoms in the N-doped graphene nanosheets (CNG), the bright dots imply the metal atoms are homogeneously dispersed on the substrate (
Figure 3). Energy dispersive X-ray spectroscopy (EDS) mapping in HAADF-STEM is applied for further identify the atomic-resolution chemical composition and distribution. EDS confirms the uniform dispersion of metal, N, and O atoms in their as-prepared samples [
35].
The non-noble metal can also be recognized by the HAADF-STEM. In the study by Liu et al., the HAADF-STEM confirms the presence of both single atoms and dual single-atoms in FeCo DACs. The corresponding elemental mapping images show that Fe, Co, N, and C are uniform dispersed over the entire matrix.
Even though the HAADF-STEM provide a visual observation of atomic sites, but it is a challenge to identify the dual-atom sites. Electron energy loss spectroscopy (EELS) can collect the electronic signal of a certain characteristic energy loss and recognize the dispersion of element of the catalyst. Liu et al. applied EELS coupled with HAADF-STEM to recognize the chemical composition of metal-atom pairs. Two areas with coupled bright dots (marked as P1 and P2) and one background area without bright dots (marked as P3) were selected to carry out the EELS. In area P1 and P2, both Cu and Mn signals can be detected, the P3 area shows no signal for metal elements, which confirm the formation of Cu/Mn dual-atomic sites in the materials [
29].
Nowadays, the HAADF-STEM becomes an indispensable tool for the identify of DACs due to its structural visualization, but it is difficult to distinguish the atomic sites in different depths. It usually needs to be combined with other characterization techniques to recognize the DACs.
3.2. X-ray Absorption Spectroscopy
The X-ray absorption spectrum (XAS) is another useful tool to characterize the DACs. It provides a variety of information, including valences, coordination configurations, and structures. The X-ray absorption near edge structure (XANES) indicates the oxidation state of the materials, and the extended X-ray absorption fine structure (EXAFS) shows the coordination environment of catalyst through the Fourier or wavelet (FT or WT) analysis and EXAFS fitting [
36].
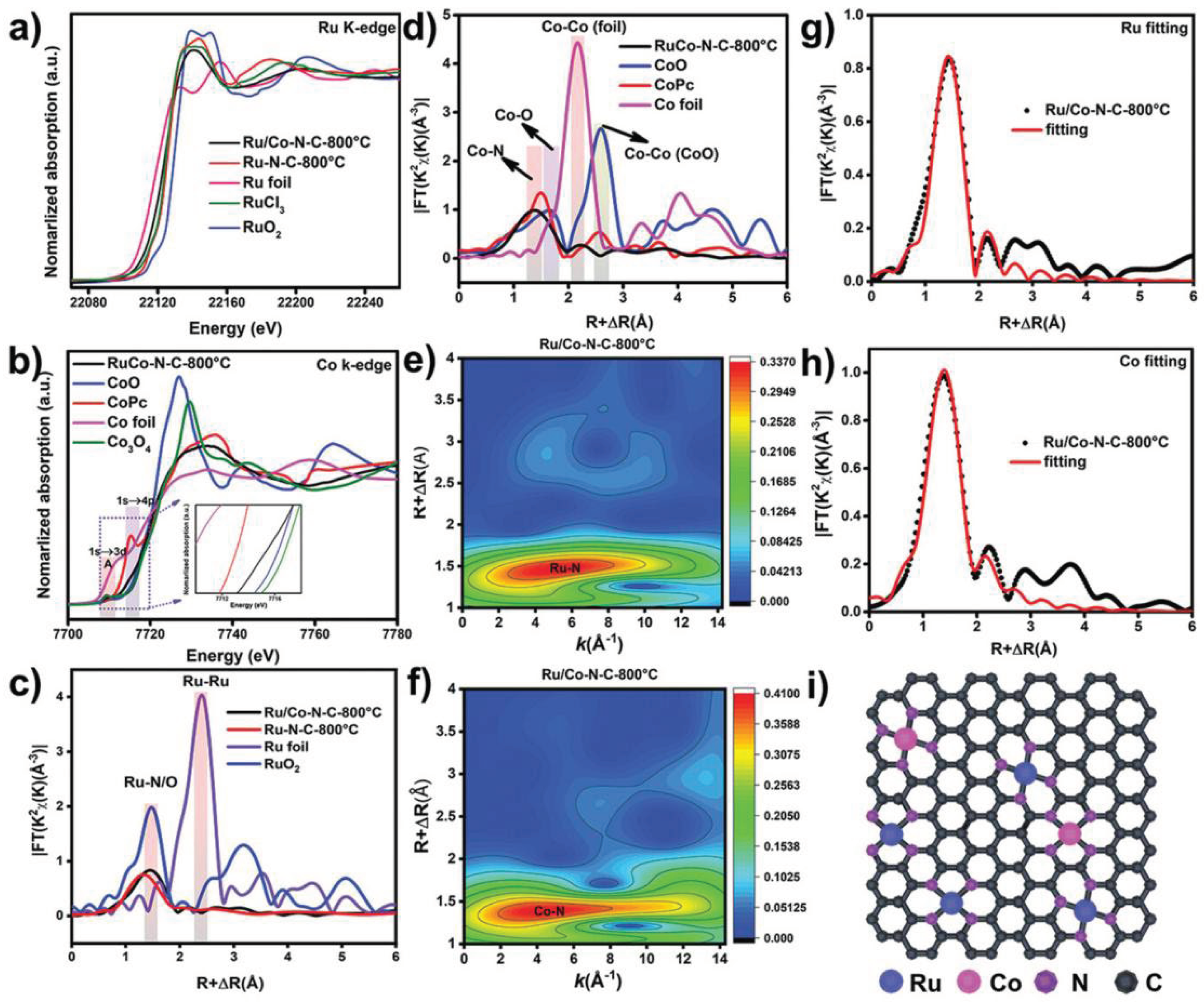
Zhao et al. carried out XAS to identify the chemical states and coordination configurations of Ru/Co dual-sites anchored on N-doped carbon (Ru/Co-N-C) [
37]. In their XANES spectra as shown in
Figure 4, the near-edge absorption energy of the Ru k-edge was positioned between those of the Ru foil and RuO
2, and close to the RuCl
3, suggesting the dominant valence state of Ru in Ru/Co-N-C-800 °C is about +3. Compared with RuO
2, a pre-edge peak of Ru in Ru/Co-N-C-800 °C near 22114 eV is related to the transition of Ru 1s to the unoccupied Ru 4d level [
38]. Besides, the Ru k-edge spectra of Ru/Co-N-C-800 °C shift to lower energy compared with the Ru-N-C-800 °C, implying the valence of Ru decreases after introducing Co sites. For Co k-edge spectra, the normalized Co adsorption spectrum in Ru/Co-N-C-800 °C is located between CoO and Co foil, indicating the oxidation of Co in Ru/Co-N-C-800 °C. A pre-edge peak (A) at ~7709.6 eV can be assigned to the dipole-forbidden but quadrupole-allowed transition, referring the 3d and 4p orbital hybridization of the Co atoms [
39]. The Fourier-transformed (FT) k
3-weigehted EXAFS (FT-EXAFS) was further conducted. The dominant peak for Ru/Co-N-C-800 °C can be assigned to the Ru-N/C bond, there is no Ru-Ru peaks can be detected in the Ru/Co-N-C-800 °C. In FT-EXAFS of Co k-edge, the peaks at ~2.2 Å exclude the presence of metal-metal bonds, confirming the isolation of Ru and Co atoms. The WT of K
3-weigehted EXAFS spectra further confirm the isolation of Ru and Co atoms. The only dominant peak for Ru at 4.3 Å in Ru/Co-N-C-800 °C is ascribed to the Ru-N structure. Similarly, only Co-N paths is detected confirms the presence of Co-N bonds.
In another work of Lee et al., the XANES and EXAFS were conducted to investigate the coordination environment of the Fe and Co species of the FeCo-N
4-hollow carbonized shell (FeCo-N
4/HCS) [
40]. Based on the Fe k edge XANES, FeCo-N
4/HCS shows the position of the absorption threshold between Fe
2O
3 and iron phthalocyanine (FePc), indicating the average valence state of Fe situates between +2 and +3 [
41], which can be caused by the drawing of Fe electrons to the adjacent N atoms [
42]. In the FT-EXAFS, the main peak in FeCo-N
4/HCS is similar to the FePc main peak, suggesting the dominant Fe-N coordination [
43]. The EXAFS fitting exhibits that the average coordination number of Fe-N was 4.7, implying that each Fe atoms is coordinated to five or four N atoms. WT-EXAFS further demonstrates the single-atom dispersion of Fe. On the other hand, the k edge XANES of Co display that the valence state of Co in FeCo-N
4/HCS is ~+2 [
44]. In the Co K edge FT-EXAFS profile, the main peak can be attributed to the coordination of Co-N, suggesting the single-atomic state of Co species. The EXAFS fitting curves further indicate the single-atom Co-N
4 model.
The XAS technique can also be applied
in situ/
operando situations, which is promising to provide insights into the mechanism. Yao et al. clarified the evolution of the local coordination environment of Cu sites under various applied voltages by
operando EXAFS [
16]. When the applied cell voltages change, the fitting curves at the Cu k-edge of Cu-Se DACs show an obvious high-R shift from 1.49 Å to 1.54 Å, suggesting a rearrangement of the coordination of Cu sites, which may be triggered by adsorption of intermediates.
Notably, the information deduced from XAS is an average result, indicating that an inaccurate structure may be obtained if the materials have various coordination configurations. Thus, the XAS should unite with other tools to confirm the real structural information.
Besides, X-ray photoelectron spectroscopy (XPS), Mössbauer spectroscopy, and Fourier-transform infrared (FTIR) spectroscopy can also be applied to research the DACs. The other unmentioned characterization methods for DACs can be seen in other reviews [
45,
46].
4. Electrocatalytic Applications
In this chapter, recent advances of DACs in water splitting applications are summarized. The relationships between the structure of DACs and electrochemical properties are discussed, which provide a new inspiration for the design of DACs.
4.1. Hydrogen Evolution Reaction
Electrochemical hydrogen evolution reaction provides a cost-efficient and sustainable method to generate high-purity H
2 effectively, which is an important component of developing green energy technologies [
47]. The widely accepted multi-step reaction pathway of HER in acid/alkaline is summarized in the
Table 1.
According to the reaction mechanism, the “Volmer step” differs from the formation of H adsorption, either from H
+ in acidic solutions or dissociated H
2O molecule in alkaline solution. The reaction rate of HER is determined by the ΔG
H* (the adsorption free energy of hydrogen on the catalyst surface). The closer the ΔG
H* to zero, the more conducive to achieving rapid HER kinetics by balancing the adsorption and desorption of H* on the surface [
48,
49,
50]. The ΔG
H* is mainly rely on the intrinsic electronic properties of the electrocatalysts. Thus, rational design of catalyst is very crucial.
So far, Pt group metal (PGM) have been widely considered as the state-of-the-art electrocatalysts for HER, due to the optimal ΔG
H* value and rapid reaction kinetics [
51]. Nevertheless, their practical applications are hindered by the high price and natural scarcity. To overcome this problem, PGM-based DACs have been widely researched in HER process, including Pt-, Pd-, and Ru- dual atom sites. We will introduce some typical DACs towards the HER.
For example, Sun et al. prepared high-quality Pt-Ru dimer structures on nitrogen-doped carbon nanotubes through an atomic layer deposition (ALD) process, which show much higher hydrogen evolution activity (more than 50 times) and excellent stability compared with the commercial Pt/C. Both the XAS spectra and first-principles calculations reveal the formation of Rt-Ru bond. The detailed study reveals that ΔG
H* becomes smaller after the electronic structures of both Pt and Ru become semiconductor. With the increase of the hydrogen adsorption, the ΔG
H* is further reduced, indicating that the Pt-Ru dimer generates a synergy effect by modulating the electronic structure, which enhances the HER activity [
52].
In another work, Yu et al. report the synergistic effect and electron redistribution effect in Pt
1Ru
1 dimer, which can simultaneously accelerate the H
2 production. The Pt
1Ru
1/ N-doped mesoporous hollow carbon sphere exhibits a quite low overpotential of 22 mV to drive a current density of 10 mA cm
-2. Meanwhile, a large mass activity of 3.49 A mg
-1 at 50 mV overpotential and a high turnover frequency (TOF) of 74.14 H
2 s
-1 at 200 mV overpotential are achieved. The spectroscopic investigations and theoretical calculations reveal the electron redistribution effect of Ru on the Pt atom, where the Ru atom promotes the electron redistribution while the protons are accumulated on the Pt for the HER process, leading to optimal HER activity [
53].
To further regulate the electronic structure and boost the HER activity, the DACs with a combination of transition metal and noble metal have been studied.
For example, Wang et al. reports dual single-atom Rh-Fe as an excellent electrocatalyst for the HER in an acidic electrolyte, the Rh-Fe perform at an ultralow overpotential of 36 mV at 10 mA cm
-2 and a Tafel slope of 26 mV dec
-1, which is superior to 20 wt% Pt/C and other single-atom electrocatalysts. First-principles calculations and experiments indicate that the binding of hydrogen on the Rh site in more favorable than on other atoms, while the Fe sites perform a weak interaction with H. After H adsorption, the electrons aggregate around Fe sites and Rh sites share the electrons with H. The excellent activity of Rh-Fe DACs for HER is attributed mainly to the synergistic effect between Rh and Fe sites [
54].
In another work, Cheng et al. prepared Pt
1Mo
1/Ni
3Se
2 dual-atom catalysts, it only needs 53 mV to deliver the current density of 10 mA cm
-2 in 1 M KOH media. Meantime, the mass activity is 4.13 times higher than that of Pt/C at 200 mV. Further density functional theory calculations indicate that the introduction of Mo atoms can effectively regulate the hydrogen-adsorption energy of Pt sites and boost the HER activity. Generally, the synergistic effect between Pt and Mo sites can realize the charge redistribution, thus optimizing the adsorption of H* and improving the HER performance [
55].
To further reduce the cost and explore more efficient DACs for HER, non-noble metal-based DACs have been synthesized. In the study of Zang et al., a pyrolysis-free Ni/Fe bimetallic electrocatalysts was synthesized as a superior HER electrocatalyst with simple method and low preparation cost. The overpotential for hydrogen evolution is 23 mV in 0.5 M H
2SO
4 at a current density of 10 mA cm
-2. The introduction of Ni atoms weakened the adsorption of intermediates, leading to optimized energy level and enhanced reaction activity [
56]. Fan et al. develop a dual-atom catalyst consisting of O-coordinated W-Mo heterodimer embedded in N-doped graphene (NG). The W
1Mo
1-NG DAC enables Pt-like activity and ultrahigh stability for HER in pH-universal electrolyte, providing a good example for the synthesis of DACs with non-noble metals, DFT results reveal that electron delocalization of W
1Mo
1-NG affords the desirable ΔG
H* and good HER kinetics [
33].
Overall, these studies pave new avenues for the development of DACs for HER, a better realization of its structure-activity relationship is beneficial to design more efficient DACs.
4.2. Oxygen Evolution Reaction
OER process is the half-reaction of electrochemical water splitting relative to HER, it also plays a crucial role in rechargeable metal-air batteries correspond with ORR [
57].
The OER comprises different four-step electron transfer in both acidic and alkaline electrolyte as summarized in
Table 2. Compared with HER, OER is more intrinsically sluggish and results in large overpotential [
58]. Meanwhile, the reaction processes are different in various electrolyte with different pH values.
To date, the Fe, Co, and Ni based DACs are the most studied electrocatalysts for OER. Among them, the synergetic effect between Ni and Fe makes it one of the most promising non-noble metal-based OER electrocatalyst.
For example, Peng et al. developed atomically dispersed Fe-Ni single atoms (SAs) embedded in a nitrogen-doped carbon matrix (NC), the as-obtained FeNi electrocatalyst display a low overpotential of 270 mV at 10 mA cm
-2 for the OER. The density functional theory calculation indicates that the rate-determining step (RDS) to be overcome in bimetallic FeNi SAs/NC is smaller than that in FeNi nanoparticles and NC, suggesting that the synergetic coupling between single atomic sites and N-doped carbon matrix can enhance the OER performance. Additionally, Fe SAs/NC was also tested, indicating that there is a synergistic effect between Fe and Ni atoms. In this work, Fe sites as the active center can facilitate the four-electron reaction process, the Ni sites can modulate the electronic structure of Fe sites and further reduce the energy barrier of the rate-determining step [
59].
The interaction between metals and supporting materials can affect the catalytic performance. In the work of Luo et al., they anchored dual metal atoms Ni and Fe via Fe-N
4 and Ni-N
4 coordination on nitrogen-doped graphene surfaces (DG). The Ni/Fe DACs exhibits high catalytic activity for OER, an overpotential of 358 mV was achieved as 10 mA cm
-2 with a lower Tafel slope of 76 mv/dec than commercial Pt/C [
60]. This study using a supporting material to anchor and stabilize two isolated metal sites (Ni/Fe), and the DFT calculations confirm that the introduction of structural defects in the supporting material may subtly tune the active sites’ reactivity.
In another work, Lu et al. fabricate Fe-Ni DACs into nitrogen-doped carbon hollow spheres (NC), the as-prepared catalyst shows outstanding catalytic activity for OER, which is even comparable to the noble metal-based benchmarks. A small potential gap of 0.73V (vs. RHE) can be reached between the potential of OER for reaching 10 mA cm
-2 (E
10) and the half-wave potential (E
1/2) of ORR in 1 M KOH electrolyte. The combined experiments and DFT calculations reveal that the OER mechanism on Fe-NiNC is different from those with single atoms. In OER process, all the intermediates preferentially adsorb on Ni site in the presence of the OH ligand bridging the Fe, the Fe-Ni pairs imposes a mutual effect for charge redistribution and enhancing the activities for both OER and ORR reaction [
61].
Except for Fe and Ni, some other elements introduced into DACs are also proved to be effective. For example, Chen et al. develop Ir-Co DACs as a bifunctional oxygen electrocatalysts. It has an exceptional OER activity with a small overpotential of 330 mV at 10 mA cm
-2 [
62]. Meantime, Noble-metal elements are also explored to fabricate DACs. Hao et al. design nitrogen-doped carbon-supported Co and Ru DACs for outstanding OER performance under alkaline conditions. The Co/Ru DACs displays a low potential of 338 mV to reach 10 mA cm
-2 current density in OER. Further DFT calculations certificate that Ru can optimize the adsorption energy of Co sites, this work provides more reasonable strategy for design DACs [
63].
In summary, OER with four-electron transfer process have a sluggish kinetics and results in large overpotential [
64]. The most widely researched electrocatalysts for OER is the noble metal oxide (e.g., RuO
2, IrO
2), but the high costs hinder their practical applications [
65]. DACs with intriguing properties are expected to realize outstanding OER performance. However, how to balance the competition between the rate-limiting steps of reversible oxygen reduction and oxygen evolution reaction remain challenges.
5. Summary and Perspective
Facing the energy crisis and environmental problems, the development of catalysts brings new opportunities. DACs have received extensive attention, due to their maximum atom-utilization, atomic active sites, adjustable electronic configuration, and outstanding catalytic performance. In this paper, we summarized the preparation strategies, characterizations, and catalytic applications of DACs in water-splitting reaction reported in recent years.
Even though DACs exhibit efficient activity during water-splitting reaction, it still faces challenges in several aspects:
- 1)
Despite the progress in the synthesis of DACs, the accurately control the atomic structure and uniform dispersion still in the initial stage. For example, impurities (e.g., SACs and nanoclusters) are inclined to generate during the high temperature pyrolysis. Meantime, the accurate amount of metal precursors is difficult to control, single atoms or metal clusters generate inevitably at the same time. How to synthesize DACs in which one metal atoms is merely bond to another, it remains a challenge. Thus, it is necessary to combine different synthetic strategies and develop new synthetic methods. Additionally, DACs consist of main group elements is worth exploring. The design of heteronuclear DACs which combines transition and main-group metals can uncover the synergistic effect between these elements.
- 2)
Different supports for DACs will bring different electronic structures and enhance the performance. MOFs, ZIFs, covalent organic frameworks, and g-C3N4 are widely applied to serve as supports for DACs, there is plenty room for optimization. An interesting aspect of metal supports, such as metallene, which has a two-dimensional nanosheet morphology, may offer cooperative electronic interactions with guest metal atoms. Meanwhile, the stability and catalytic properties of DACs supported by different supports need to be further explored.
- 3)
Different characterization techniques can identify the structure of DACs, such as HAADF-STEM and XAS. For example, the HAADF-STEM can observe the DACs with atomic level resolution, XAS can analyze the local structure of the DACs regarding the metal-metal interaction, oxidation state, bond length, and coordination environment. Whereas, it is difficult to monitor the structure change and evolution during the reaction in situ constantly. The real active sites in working conditions may be different from that of the ex-situ conditions. More advanced in situ/operando equipment should be considered, which can provide more information about the structure-activity relationship and guide the design of DACs.
- 4)
The stability timescale of electrocatalysts for water-splitting in industrial application is usually months or even years, which is far beyond the laboratory research lever, even the Pt/C catalyst with excellent performance can only be used for 40 h [
66]. DACs face the risk of agglomeration and leaching of metal atoms in actual operation (operating under high current densities), the controlled synthesis of high-quality and stable DACs remain a major obstacle.
- 5)
Due to the shortage of freshwater resources, electrolysis of seawater has become a research hotspot. Therefore, it is important to develop robust and inexpensive DACs for seawater electrolysis reaction.
Overall, this review systematically summarizes the recent advances of DACs for HER and OER electrocatalysis. DACs are promising candidates for water-splitting reactions due to their unique structures and performance. However, there is still a long way to go before they are widely applied in the practical applications, and continuous efforts should be devoted to this highly exciting research field.
Funding
The authors acknowledge financial support from the National Natural Science Foundation of China (Grant No. 22303004).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wu, H.; Huang, Q.; Shi, Y.; Chang, J.; Lu, S. Electrocatalytic water splitting: Mechanism and electrocatalyst design. Nano Res. 2023, 16, 9142–9157. [Google Scholar] [CrossRef]
- Wu, H.; Cheng, Y. J.; Wang, B. Y.; Wang, Y.; Wu, M.; Li, W. D.; Liu, B. Z.; Lu, S. Y. Carbon dots-confined CoP-CoO nanoheterostructure with strong interfacial synergy triggered the robust hydrogen evolution from ammonia borane. J. Energy Chem. 2021, 57, 198–205. [Google Scholar] [CrossRef]
- Wang, X.; Xu, L.; Li, C.; Zhang, C.; Yao, H.; Xu, R.-b.; Cui, P.; Zheng, X.; Gu, M. D.; Lee, J.; Jiang, H.; Huang, M. Developing a class of dual atom materials for multifunctional catalytic reactions. Nat. Commun. 2023, 14, 7210. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Liu, H.-x.; Yuan, P.; Jia, Y. A.; Zhuang, L.; Zhang, H.; Yan, X.; Liu, G.; Zhao, Y.; Liu, J.; Wei, S.; Song, L.; Wu, Q.; Ge, B.; Zhang, L.; Wang, K.; Wang, X.; Chang, C.-R.; Yao, X. Single Carbon Vacancy Traps Atomic Platinum for Hydrogen Evolution Catalysis. J. Am. Chem. Soc. 2022, 144, 2171–2178. [Google Scholar] [CrossRef]
- Feng, J.; Tong, S.; Tong, Y.; Li, G. R. Pt-like Hydrogen Evolution Electrocatalysis on PANI/CoP Hybrid Nanowires by Weakening the Shackles of Hydrogen Ions on the Surfaces of Catalysts. J. Am. Chem. Soc. 2018, 140, 5118–5126. [Google Scholar] [CrossRef]
- Qiao, B.; Wang, A.; Yang, X.; Allard, L. F.; Jiang, Z.; Cui, Y.; Liu, J.; Li, J.; Zhang, T. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nat. Chem. 2011, 3, 634–641. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Santiago, A. R. P.; Kong, Y.; Ahsan, M. A.; Luque, R.; Du, A.; Pan, H. Atomically Dispersed Heteronuclear Dual-Atom Catalysts: A New Rising Star in Atomic Catalysis. Small 2021, e2106091. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Wang, D. Superiority of Dual-Atom Catalysts in Electrocatalysis: One Step Further Than Single-Atom Catalysts. Adv. Energy Mater. 2022, 12, 2103564. [Google Scholar] [CrossRef]
- Ying, Y.; Luo, X.; Qiao, J.; Huang, H. “More is Different:” Synergistic Effect and Structural Engineering in Double-Atom Catalysts. Adv. Funct. Mater. 2020, 31, 2007423. [Google Scholar] [CrossRef]
- Gao, Y.; Liu, B.; Wang, D. Microenvironment Engineering of Single/Dual-Atom Catalysts for Electrocatalytic Application. Adv. Mater. 2023, 35, 2209654. [Google Scholar] [CrossRef]
- He, Y.; Zhou, X.; Jia, Y.; Li, H.; Wang, Y.; Liu, Y.; Tan, Q. Advances in Transition-Metal-Based Dual-Atom Oxygen Electrocatalysts. Small 2023, e2206477. [Google Scholar] [CrossRef]
- Choi, J.; Seo, S.; Kim, M.; Han, Y.; Shao, X.; Lee, H. Relationship between Structure and Performance of Atomic-Scale Electrocatalysts for Water Splitting. Small 2023, e2304560. [Google Scholar] [CrossRef]
- Shang, H.; Liu, D. Atomic design of carbon-based dual-metal site catalysts for energy applications. Nano Res. 2023, 16, 6477–6506. [Google Scholar] [CrossRef]
- Xu, W.-X.; Wang, Y.; Zhang, C.; Ma, X.; Wu, J.; Liu, Y.; Lu, B.; Zhang, H.; Ming, C.; Xiang, J. Insights into the Electronic Structure Coupling Effect of Dual-metal Atomic Electrocatalytic Platform for Efficient Clean Energy Conversion. Chem. Eng. J. 2023, 141911. [Google Scholar] [CrossRef]
- Li, M.; Zhu, H.; Yuan, Q.; Li, T.; Wang, M.; Zhang, P.; Zhao, Y.; Qin, D.; Guo, W.; Liu, B.; Yang, X.; Liu, Y.; Pan, Y. Proximity Electronic Effect of Ni/Co Diatomic Sites for Synergistic Promotion of Electrocatalytic Oxygen Reduction and Hydrogen Evolution. Adv. Funct. Mater. 2022, 33, 2210867. [Google Scholar] [CrossRef]
- Sun, Z.; Zhang, H.-J.; Cao, L.; Liu, X.; Wu, D.; Shen, X.; Zhang, X.; Chen, Z.; Ru, S.; Zhu, X.; Xia, Z.; Luo, Q.; Xu, F.; Yao, T. Understanding Synergistic Catalysis on Cu-Se Dual Atom Sites via Operando X-ray Absorption Spectroscopy in Oxygen Reduction Reaction. Angew. Chem. Int. Ed. 2023, 62, e202217719. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Li, R.; Zhang, J.; Wang, Y.; Ma, W.; Yue, Z.; Jin, C.; Liu, Y.; Zheng, L.; Bai, J.; Li, X.; Leng, K.; Qu, Y. N-Coordinated Iridium-Molybdenum Dual-Atom Catalysts Enabling Efficient Bifunctional Hydrogen Electrocatalysis. ACS Appl. Mater. Interfaces 2024, 16, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhao, F.; Jiao, L.; Fang, T.; Zhao, Z.; Xiao, X.; Li, D.; Yi, K.; Wang, R.; Jia, X. Atomically Dispersed Fe/N4 and Ni/N4 Sites on Separate-Sides of Porous Carbon Nanosheets with Janus Structure for Selective Oxygen Electrocatalysis. Small 2023, e2300289. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Guo, R.; Peng, X.; Wang, X.; Liu, X.; Ren, J.; He, J.; Zhuo, L.; Sun, J.; Liu, Y.; Wu, Y.; Luo, J. Highly Productive Electrosynthesis of Ammonia by Admolecule-Targeting Single Ag Sites. ACS nano 2020, 14, 6938–6946. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ji, Y.; Zhao, Y.; Chen, J.; Zheng, S.; Sang, X.; Yang, B.; Li, Z.; Lei, L.; Wen, Z.; Feng, X.; Hou, Y. Local Spin-State Tuning of Iron Single-Atom Electrocatalyst by S-Coordinated Doping for Kinetics-Boosted Ammonia Synthesis. Adv. Mater. 2022, 34, 2202240. [Google Scholar] [CrossRef]
- Wang, C.; Wang, K.; Feng, Y.; Li, C.; Zhou, X.; Gan, L.; Feng, Y.; Zhou, H.; Zhang, B.; Qu, X.; Li, H.; Li, J.; Li, A.; Sun, Y.; Zhang, S.; Yang, G.; Guo, Y.; Yang, S.-z.; Zhou, T.; Dong, F.; Zheng, K.; Wang, L.; Huang, J.; Zhang, Z.; Han, X. Co and Pt Dual-Single-Atoms with Oxygen-Coordinated Co–O–Pt Dimer Sites for Ultrahigh Photocatalytic Hydrogen Evolution Efficiency. Adv. Mater. 2021, 33, 2003327. [Google Scholar] [CrossRef]
- Fang, W.; Wu, Y.; Xin, S.; Hu, Y.; Dang, J.; Li, M.; Chen, B.; Zhao, H.; Lib, Z. Fe and Mo Dual-Site Single-Atom Catalysts for High-Efficiency Wide-pH Hydrogen Evolution and Alkaline Overall Water Splitting. Chem. Eng. J. 2023, 468, 143605. [Google Scholar] [CrossRef]
- Da, Y.; Tian, Z.; Jiang, R.; Liu, Y.; Lian, X.; Xi, S.; Shi, Y.; Wang, Y.; Lu, H.; Cui, B.; Zhang, J.; Han, X.; Chen, W.; Hopchev, P. H. Dual Pt-Ni atoms dispersed on N-doped carbon nanostructure with novel (NiPt)-N4C2 configurations for synergistic electrocatalytic hydrogen evolution reaction. Sci. China Mater. 2022, 66, 1389–1397. [Google Scholar] [CrossRef]
- Chen, J.; Li, H.; Fan, C.; Meng, Q.; Tang, Y.; Qiu, X.; Fu, G.; Ma, T. Dual Single-Atomic Ni-N4 and Fe-N4 Sites Constructing Janus Hollow Graphene for Selective Oxygen Electrocatalysis. Adv. Mater. 2020, 32, 2003134. [Google Scholar] [CrossRef]
- Zhao, J.; Zong, L.; Cui, L.; Lu, F.; Xiao, Z.; Wang, L. Synthesis of dual-metal single atom in porous carbon with efficient oxygen reduction reaction in both acidic and alkaline electrolytes. J. Colloid Interface Sci. 2022, 633, 828–835. [Google Scholar] [CrossRef]
- Han, X.; Ling, X.; Yu, D.; Xie, D.; Li, L.; Peng, S.; Zhong, C.; Zhao, N.; Deng, Y.; Hu, W. Atomically Dispersed Binary Co-Ni Sites in Nitrogen-Doped Hollow Carbon Nanocubes for Reversible Oxygen Reduction and Evolution. Adv. Mater. 2019, 31, 1905622. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; Gao, Y.; Chen, H.; Li, P.; Zhu, S.; Wang, J.; He, Q.; Chen, W. A Cu and Fe dual-atom nanozyme mimicking cytochrome c oxidase to boost the oxygen reduction reaction. J. Mater. Chem. A 2020, 8, 16994. [Google Scholar] [CrossRef]
- Yadian, X.; Kocaefe, D.; Chen, C.; Kocaefe, Y. Review of Research on Template Methods in Preparation of Nanomaterials. J. Nanomater. 2016, 2016, 2302595. [Google Scholar] [CrossRef]
- Chen, Y.; Qiao, S.; Tang, Y.; Du, Y.; Zhang, D.; Wang, W.; Xie, H.; Liu, C. Precise and scalable fabrication of metal pair-site catalysts enabled by intramolecular integrated donor atoms. J. Mater. Chem. A 2022, 10, 25307–25318. [Google Scholar] [CrossRef]
- Zheng, J.; Ye, J.; Ortuño, M. A.; Fulton, J. L.; Gutiérrez, O. Y.; Camaioni, D. M.; Motkuri, R. K.; Li, Z.; Webber, T. E.; Mehdi, B. L.; Browning, N. D.; Penn, R. L.; Farha, O. K.; Hupp, J. T.; Truhlar, D. G.; Cramer, C. J.; Lercher, J. A.; Lercher, J. A. Selective Methane Oxidation to Methanol on Cu-Oxo Dimers Stabilized by Zirconia Nodes of an NU-1000 Metal-Organic Framework. J. Am. Chem. Soc. 2019, 141, 9292–9304. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Han, K.; Li, K.; Pan, J.; Wang, X.; Shi, W.; Song, S.; Zhang, H. Dual-Site Single-Atom Catalysts with High Performance for Three-Way Catalysis. Adv. Mater. 2022, 34, 2201859. [Google Scholar] [CrossRef]
- Bai, L.; Hsu, C.-S.; Alexander, D. T. L.; Chen, H. M.; Hu, X. A Cobalt-Iron Double-Atom Catalyst for the Oxygen Evolution Reaction. J. Am. Chem. Soc. 2019, 141, 14190–14199. [Google Scholar] [CrossRef]
- Yang, Y.; Qian, Y.; Li, H.; Zhang, Z.; Mu, Y.; Do, D.; Zhou, B.; Dong, J.; Yan, W.; Qin, Y.; Fang, L.; Feng, R.; Zhou, J.; Zhang, P.; Dong, J.; Yu, G.; Liu, Y.; Zhang, X.; Fan, X. O-coordinated W-Mo dual-atom catalyst for pH-universal electrocatalytic hydrogen evolution. Sci. Adv. 2020, 6, eaba6586. [Google Scholar] [CrossRef]
- Pu, T.; Ding, J.; Zhang, F.; Wang, K.; Cao, N.; Hensen, E. J. M.; Xie, P. Dual Atom Catalysts for Energy and Environmental Applications. Angew. Chem. Int. Ed. 2023, e202305964. [Google Scholar] [CrossRef]
- Wan, W.; Zhao, Y.; Wei, S.; Triana, C. A.; Li, J.; Arcifa, A.; Allen, C. S.; Cao, R.; Patzke, G. R. Mechanistic insight into the active centers of single/dual-atom Ni/Fe-based oxygen electrocatalysts. Nat. Commun. 2021, 12, 5589. [Google Scholar] [CrossRef]
- He, Y.; Zhou, X.; Jia, Y.; Li, H.; Wang, Y.; Liu, Y.; Tan, Q. Advances in Transition-Metal-Based Dual-Atom Oxygen Electrocatalysts. Small 2023, e2206477. [Google Scholar] [CrossRef]
- Rong, C.; Shen, X.; Wang, Y.; Thomsen, L.; Zhao, T.; Li, Y.; Lu, X.; Amal, R.; Zhao, C. Electronic Structure Engineering of Single-Atom Ru Sites via Co–N4 Sites for Bifunctional pH-Universal Water Splitting. Adv. Mater. 2022, 34, 2110103. [Google Scholar] [CrossRef]
- Sardar, K.; Petrucco, E.; Hiley, C. I.; Sharman, J. D. B.; Wells, P. P.; Russell, A. E.; Kashtiban, R. J.; Sloan, J.; Walton, R. I. Water-Splitting Electrocatalysis in Acid Conditions Using Ruthenate-Iridate Pyrochlores. Angew. Chem. Int. Ed. 2014, 53, 10960–10964. [Google Scholar] [CrossRef]
- Yang, H. B.; Hung, S.-F.; Liu, S.; Yuan, K.; Miao, S.; Zhang, L.; Huang, X.; Wang, H.-Y.; Cai, W.; Chen, R.; Gao, J.; Yang, X.; Chen, W.; Huang, Y.; Chen, H. M.; Li, C. M.; Zhang, T.; Liu, B. Atomically dispersed Ni(i) as the active site for electrochemical CO2 reduction. Nat. Energy 2018, 3, 140–147. [Google Scholar] [CrossRef]
- Zhao, M.; Sun, J.; Luo, T.; Yan, Y.; Huang, W.; Lee, J. M. π-Conjugated Macrocycles Confined Dual Single-Atom Catalysts on Graphitized Bubbles for Oxygen Reduction, Evolution, and Batteries. Small 2023, e2309351. [Google Scholar] [CrossRef]
- Wan, X.; Liu, X.; Li, Y.; Yu, R.; Zheng, L.; Yan, W.; Wang, H.; Xu, M.; Shui, J. Fe–N–C electrocatalyst with dense active sites and efficient mass transport for high-performance proton exchange membrane fuel cells. Nat. Catal. 2019, 2, 259–268. [Google Scholar] [CrossRef]
- Wei, Y. S.; Sun, L.; Wang, M.; Hong, J.; Zou, L.; Liu, H.; Wang, Y.; Zhang, M.; Liu, Z.; Li, Y.; Horike, S.; Suenaga, K.; Xu, Q. Fabricating Dual-Atom Iron Catalysts for Efficient Oxygen Evolution Reaction: A Heteroatom Modulator Approach. Angew. Chem. Int. Ed. 2020, 59, 16013–16022. [Google Scholar] [CrossRef]
- Lei, C.; Chen, H.; Cao, J.; Yang, J.; Qiu, M.; Xia, Y.-q.; Yuan, C.; Yang, B.; Li, Z.; Zhang, X.; Lei, L.; Abbott, J.; Zhong, Y.; Xia, X.-h.; Wu, G.; He, Q.; Hou, Y. Fe-N4 Sites Embedded into Carbon Nanofiber Integrated with Electrochemically Exfoliated Graphene for Oxygen Evolution in Acidic Medium. Adv. Energy Mater. 2018, 8, 1801912. [Google Scholar] [CrossRef]
- Yin, P.; Yao, T.; Wu, Y.; Zheng, L.; Lin, Y.; Liu, W.; Ju, H.; Zhu, J.; Hong, X.; Deng, Z.; Zhou, G.; Wei, S.; Li, Y. Single Cobalt Atoms with Precise N-Coordination as Superior Oxygen Reduction Reaction Catalysts. Angew. Chem. Int. Ed. 2016, 55, 10800–10805. [Google Scholar] [CrossRef]
- Bhavani, P.; Praveen Kumar, D.; Suk Yoo, J.; Hussain, M.; Weon, S.; Kim, W.; Park, Y.-K. Dual-Atomic-Site-Integrated Photocatalysts for Green Energy Synthesis. Chem. Eng. J. 2023, 467, 143429. [Google Scholar] [CrossRef]
- Liu, K.; Li, J.; Liu, Y.; Wang, M.; Cui, H. Dual metal atom catalysts: advantages in electrocatalytic reactions. J. Energy Chem. 2023, 79, 515–534. [Google Scholar] [CrossRef]
- Yang, J.; Li, W.-H.; Tan, S.; Xu, K.; Wang, Y.; Wang, D.; Li, Y. The electronic metal-support interaction directing the design of single atomic site catalyst: achieving high efficiency towards hydrogen evolution. Angew. Chem. Int. Ed. 2021, 60, 19085–19091. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Tian, Y.; Luo, J.; Jin, B.; Wu, Z.; Ning, X.; Zhan, L.; Fan, X.; Zhou, T.; Zhang, S.; Zhou, X. MoC Quantum Dots@N-Doped-Carbon for Low-Cost and Efficient Hydrogen Evolution Reaction: From Electrocatalysis to Photocatalysis. Adv. Funct. Mater. 2022, 32, 2201518. [Google Scholar] [CrossRef]
- Zhao, X.; Wu, G.; Zheng, X.; Jiang, P.; Yi, J. D.; Zhou, H.; Gao, X.; Yu, Z.; Wu, Y. A Double Atomic-Tuned RuBi SAA/Bi@OG Nanostructure with Optimum Charge Redistribution for Efficient Hydrogen Evolution. Angew. Chem. Int. Ed. 2023, 62, e202300879. [Google Scholar] [CrossRef]
- Zhang, X. Y.; Xu, L. L.; Wu, X. C.; Tao, Y. R.; Xiong, W. W. Ta3N5 nanobelt-loaded Ru nanoparticle hybrids’ electrocatalysis for hydrogen evolution in alkaline media. Molecules, 2023, 28, 1100. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Liu, B.; Wang, D. Microenvironment Engineering of Single/Dual-Atom Catalysts for Electrocatalytic Application. Adv. Mater. 2023, 35, 202209654. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Si, R.; Liu, H.; Chen, N.; Wang, Q.; Adair, K.; Wang, Z.; Chen, J.; Song, Z.; Li, J.; Banis, M. N.; Li, R.; Sham, T. K.; Gu, M.; Liu, L. M.; Botton, G. A.; Sun, X. A. Atomic layer deposited Pt-Ru dual-metal dimers and identifying their active sites for hydrogen evolution reaction. Nat. Commun. 2019, 10, 5453. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Luo, C.; Lin, Y.; Wang, G.-B.; Chen, H. M.; Kuang, P.; Yu, J. Pt–Ru Dimer Electrocatalyst with Electron Redistribution for Hydrogen Evolution Reaction. ACS Catal. 2022, 12, 5540–5548. [Google Scholar] [CrossRef]
- Zhou, Y.; Song, E.; Chen, W.; Segre, C. U.; Zhou, J.; Lin, Y. C.; Zhu, C.; Ma, R.; Liu, P.; Chu, S.; Thomas, T.; Yang, M.; Liu, Q.; Suenaga, K.; Liu, Z.; Liu, J.; Wang, J. Dual-Metal Interbonding as the Chemical Facilitator for Single-Atom Dispersions. Adv. Mater. 2020, 32, 2003484. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Xia, W.; Guo, X.-W.; Liu, W.; Cao, D.; Cheng, D. Constructing Ni3Se2-Nanoisland-Confined Pt1Mo1 Dual-Atom Catalyst for Efficient Hydrogen Evolution in Basic Media. Small Struct. 2023, 5, 2300284. [Google Scholar] [CrossRef]
- Zang, Y.; Lu, D.-Q.; Wang, K.; Li, B.; Peng, P.; Lan, Y.; Zang, S. A pyrolysis-free Ni/Fe bimetallic electrocatalyst for overall water splitting. Nat. Commun. 2023, 14, 1792. [Google Scholar] [CrossRef]
- Zhang, K.; Zou, R. Advanced Transition Metal-Based OER Electrocatalysts: Current Status, Opportunities, and Challenges. Small 2021, e2100129. [Google Scholar] [CrossRef]
- Zheng, X.; Yang, J.; Xu, Z.; Wang, Q.; Wu, J.; Zhang, E.; Dou, S.; Sun, W.; Wang, D.; Li, Y. Ru-Co Pair Sites Catalyst Boosts the Energetics for Oxygen Evolution Reaction. Angew. Chem. Int. Ed. 2022, 61, e202205946. [Google Scholar] [CrossRef]
- Yu, D.; Ma, Y.; Hu, F.; Lin, C. C.; Li, L.; Chen, H.-Y.; Han, X.; Peng, S. Dual-Sites Coordination Engineering of Single Atom Catalysts for Flexible Metal–Air Batteries. Adv. Energy Mater. 2021, 11, 2101242. [Google Scholar] [CrossRef]
- Khan, K.; Yan, X.; Yu, Q.; Bae, S.-H.; White, J. J.; Liu, J.; Liu, T.; Sun, C.-J.; Kim, J.; Cheng, H. M.; Wang, Y.; Liu, B.; Amine, K.; Pan, X.; Luo, Z. Stone-Wales defect-rich carbon-supported dual-metal single atom sites for Zn-air batteries. Nano Energy 2021, 90, 106488. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, D.; Chen, C.-J.; Zhang, Q.; Liu, R. S.; Xia, Z.; Dai, L.; Amal, R.; Lu, X. Harnessing the interplay of Fe–Ni atom pairs embedded in nitrogen-doped carbon for bifunctional oxygen electrocatalysis. Nano Energy 2020, 71, 104597. [Google Scholar] [CrossRef]
- Xiao, M.; Zhu, J.; Li, S.; Li, G.; Liu, W.; Deng, Y.-P.; Bai, Z.; Ma, L.; Feng, M.; Wu, T.; Su, D.; Lu, J.; Yu, A.; Chen, Z. 3d-Orbital Occupancy Regulated Ir-Co Atomic Pair Toward Superior Bifunctional Oxygen Electrocatalysis. ACS Catal. 2021, 11, 8837–8846. [Google Scholar] [CrossRef]
- Zhang, L.; Yao, J.; Zhang, J.; He, W.; Li, Y.; Liang, L.; Liu, C.; Liu, H.; Hao, Q. Engineering Co and Ru dual-metal atoms on nitrogen-doped carbon as highly efficient bifunctional oxygen electrocatalysts. Catal. Sci. Technol. 2022, 12, 5435–5441. [Google Scholar] [CrossRef]
- Zhang, K.; Zou, R. Advanced Transition Metal-Based OER Electrocatalysts: Current Status, Opportunities, and Challenges. Small 2021, e2100129. [Google Scholar] [CrossRef]
- Suen, N. T.; Hung, S.-F.; Quan, Q.; Zhang, N.; Xu, Y. J.; Chen, H. M. Electrocatalysis for the oxygen evolution reaction: recent development and future perspectives. Chem. Soc. Rev. 2017, 46, 337–365. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Zhou, S.; Wang, Z.; Liu, J.; Pei, W.; Yang, P.; Zhao, J.; Qiu, J. Engineering Multifunctional Collaborative Catalytic Interface Enabling Efficient Hydrogen Evolution in All pH Range and Seawater. Adv. Energy Mater. 2019, 9, 1901333. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).