Submitted:
22 January 2024
Posted:
24 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experiment
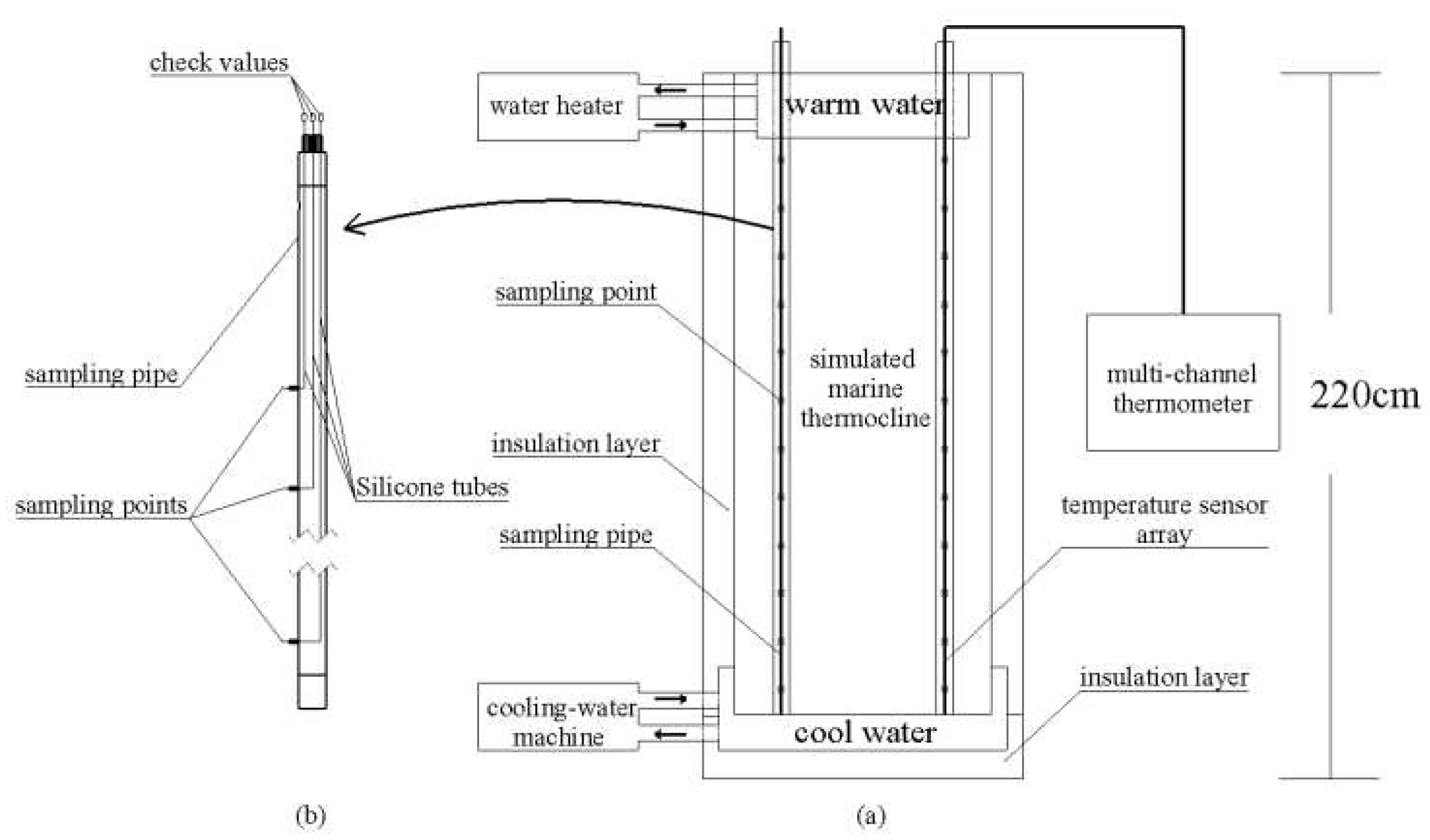
2.1. Simulated marine thermocline
2.1.1. Marine thermocline simulator
2.1.2. Measurement of marine thermocline parameters
2.2. Material
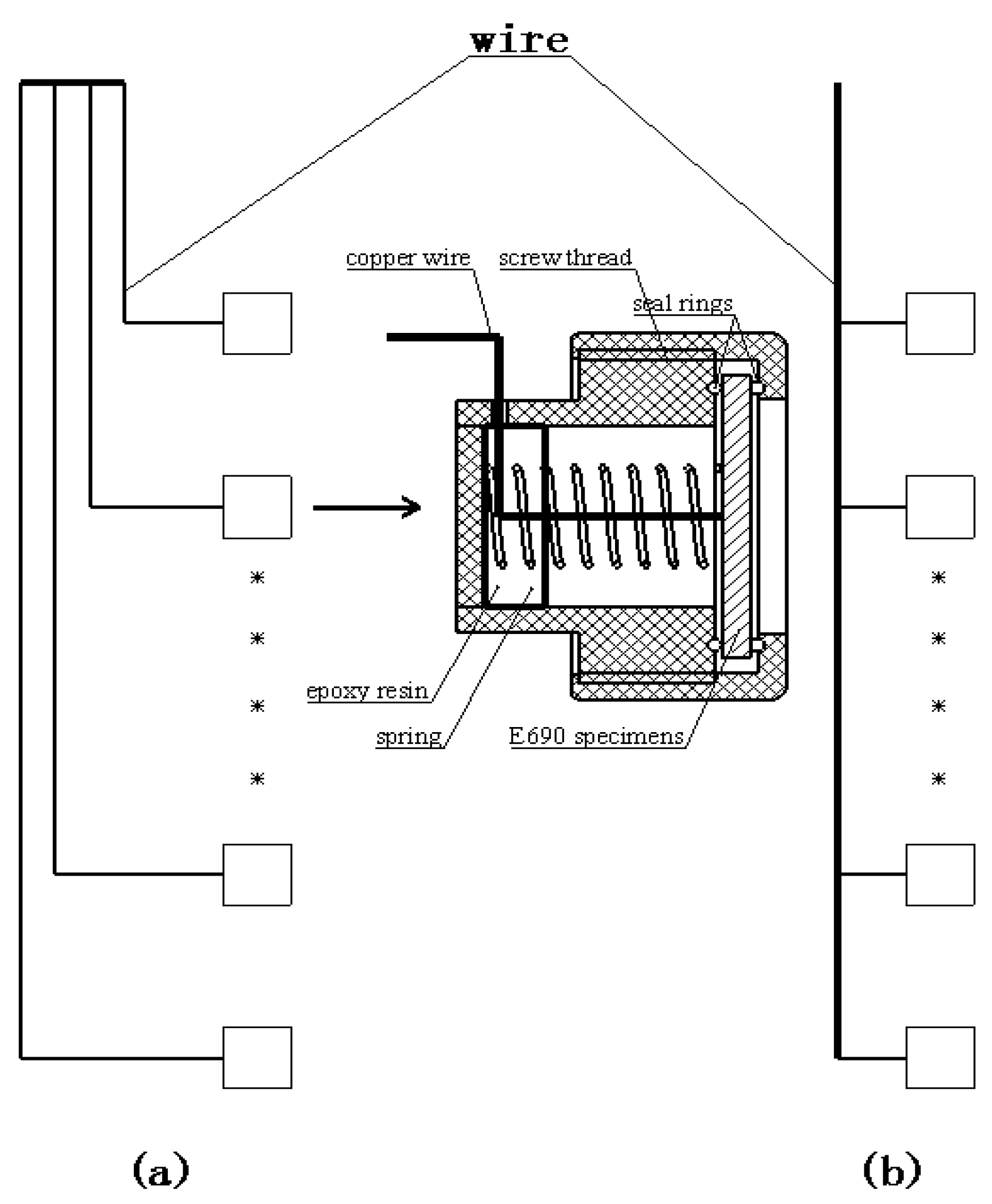
2.3. Corrosion research method
2.3.1. Marine thermocline simulator
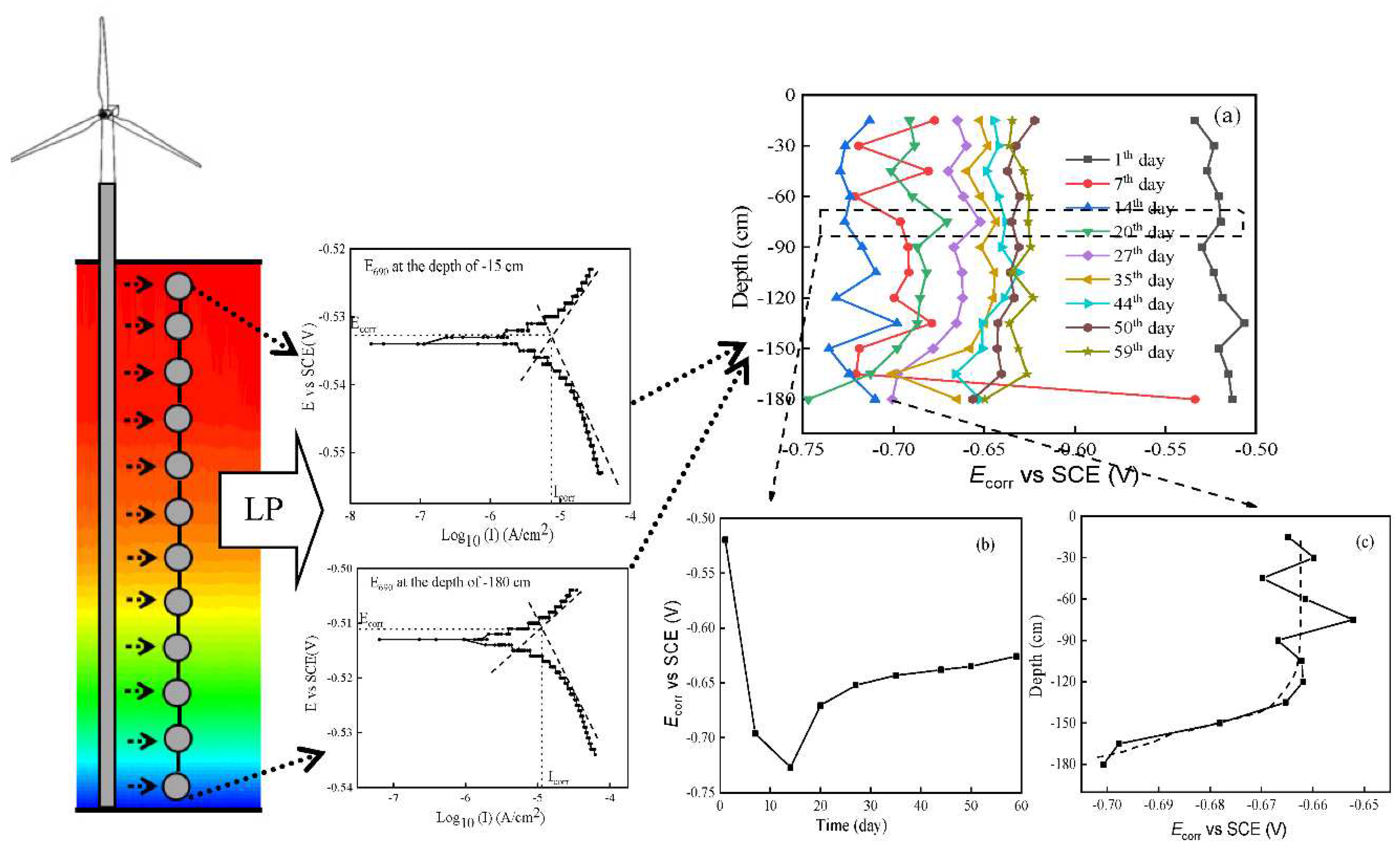
2.3.2. Measurement of instantaneous Icorr and Ecorr
2.3.3. Corrosion morphology observation
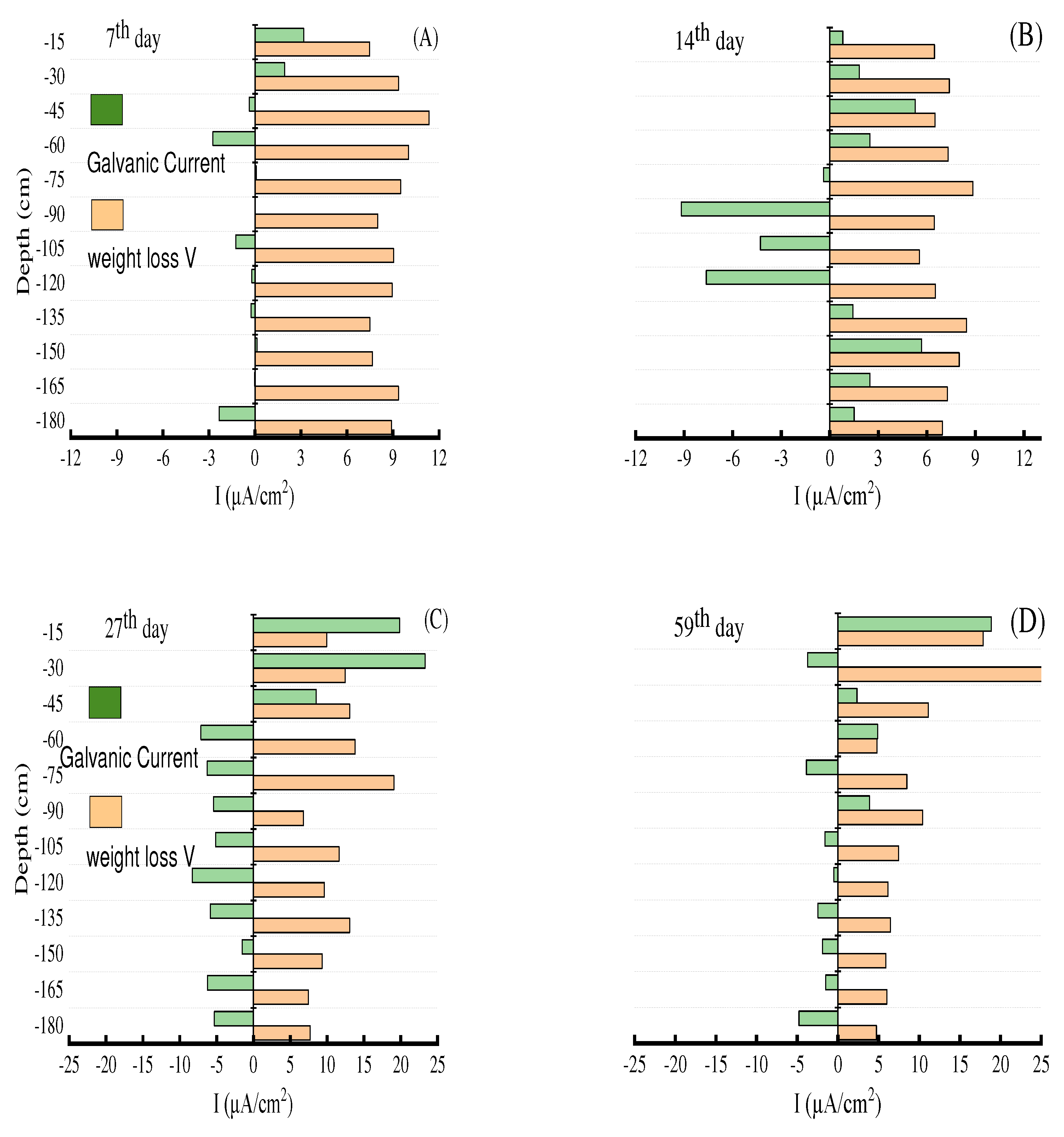
2.3.4. wt loss measurement
3. Results and discussion
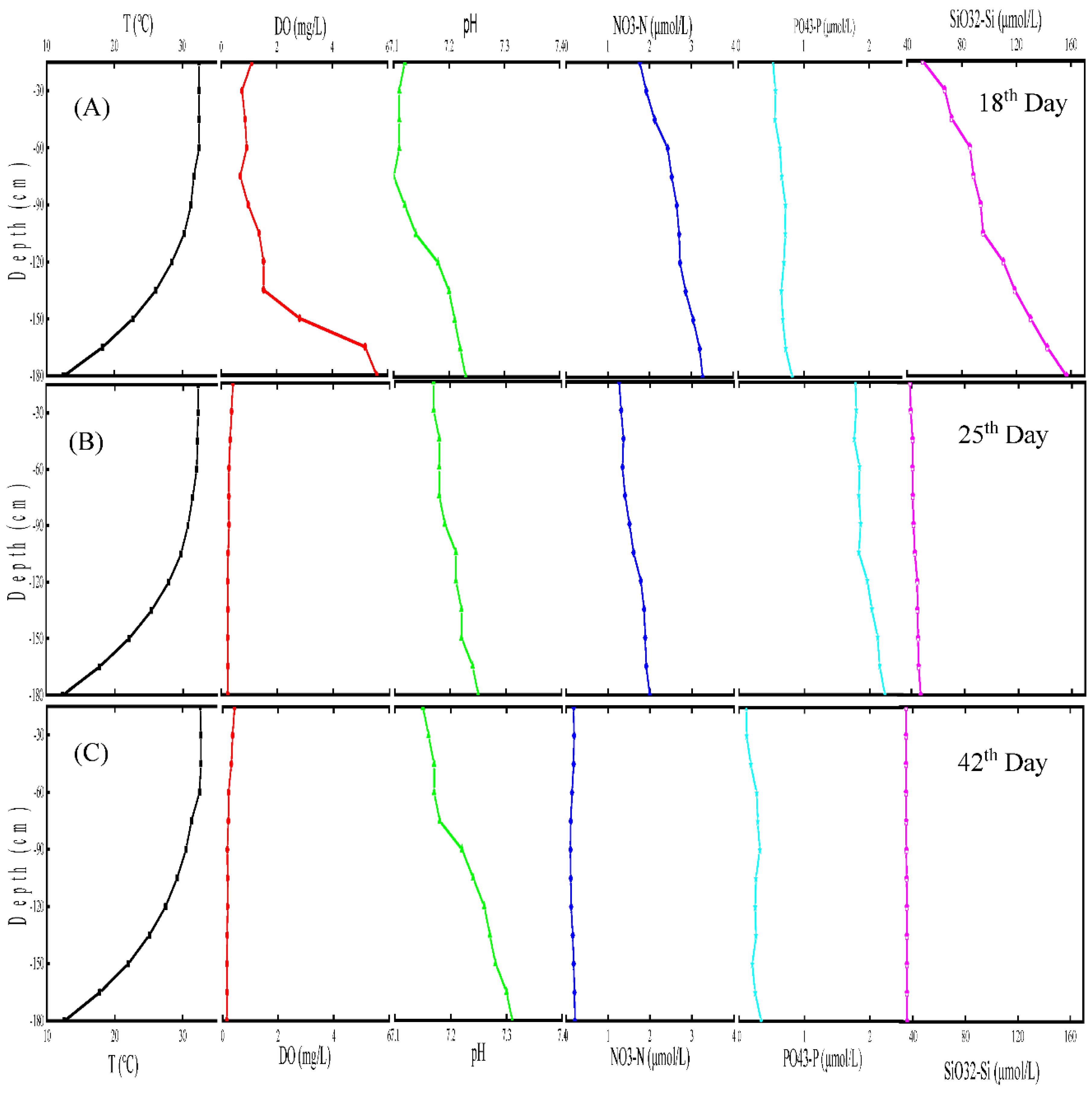
3.1. Characterization of the simulated marine thermocline
3.1.1. Temperature variation in the simulated marine thermocline
3.1.2. Components variation of simulated marine thermocline
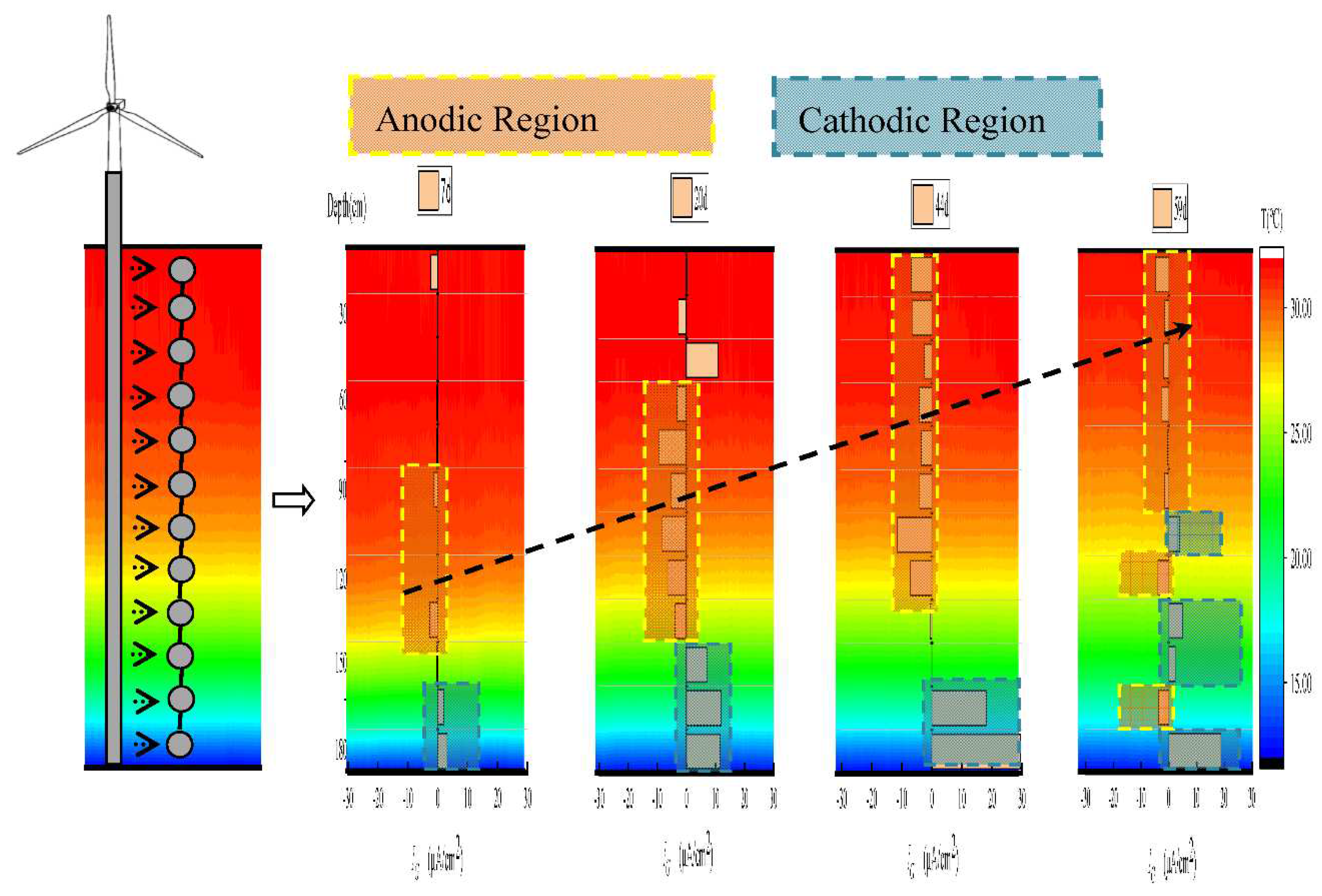
3.2. Galvanic corrosion of E690 offshore platform steel in a simulated marine thermocline
3.3. Driver of E690 offshore platform steel galvanic corrosion in SMT
3.3. Proportion of galvanic corrosion in E690 offshore platform steel corrosion
4. Conclusions
- (1)
- The SMT was a stable multilayer structure. The variations of temperature, DO, pH and nutrient concentration in the SMT were similar to those in the natural marine thermocline.
- (2)
- Galvanic corrosion occurs after the intrusion of E690 steel into the marine thermocline. Primary anodic regions were located in the area with the fastest temperature variation, and the anodic region was intermixed with the cathodic region in the lower part of the stable marine thermocline.
- (3)
- The driver of galvanic corrosion of E690 steel in the marine thermocline was the of E690 steel at various depths. The continuous reduction of Ecorr with depth contributed to the large-scale galvanic corrosion, and the oscillation variation of Ecorrwith depth was the reason for small-scale galvanic corrosion.
- (4)
- The Ecorr of E690 steel was influenced by the temperature, pH and DO in the marine thermocline, and the order was DO >> T > pH.
- (5)
- There were at least two forms of E690 steel corrosion in the marine thermocline: galvanic corrosion and see-water corrosion. The proportion of galvanic corrosion in the average corrosion rate could increase up to approximately 80% in the anodic region. There were many deep corrosion pits in the long-term and stable anodic region of galvanic corrosion.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lynne D.T.; Pickard, G.L. Typical Distributions of Water Characteristics. Descriptive physical oceanography: an introduction, 6th ed.; Elsevier, London, 2011; 67-110.
- Zhang, X.; Prange, M. Changes in equatorial Pacific thermocline depth in response to Panamanian seaway closure: Insights from a multi-model study. Earth Planet Sci. Lett. 2012, 317, 76–84. [Google Scholar] [CrossRef]
- Dehghani, A.; Aslani, F. A review on defects in steel offshore structures and developed strengthening techniques. Structures. 2019, 20, 635–657. [Google Scholar] [CrossRef]
- Saiful Islam, A.B.M.; Jameel, M. Review of offshore energy in Malaysia and floating Spar platform for sustainable exploration. Renewable Sustainable Energy Rev. 2012, 16, 6268–6284. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Z. Effect of cathodic potential on stress corrosion cracking behavior of different heat-affected zone microstructures of E690 steel in artificial seawater. J. Mater. Sci. Technol. 2021, 64, 141–152. [Google Scholar] [CrossRef]
- Liu, Z.; Hao, W. Fundamental investigation of stress corrosion cracking of E690 steel in simulated marine thin electrolyte layer. Corros. Sci. 2019, 148, 388–396. [Google Scholar] [CrossRef]
- Ma, H.C.; Fan, Y. Effect of pre-strain on the electrochemical and stress corrosion cracking behavior of E690 steel in simulated marine atmosphere. Ocean Eng. 2019, 182, 188–195. [Google Scholar] [CrossRef]
- Ma, H.C.; Liu, Z.Y. Stress corrosion cracking of E690 steel as a welded joint in a simulated marine atmosphere containing sulphur dioxide. Corros. Sci. 2015, 100, 627–641. [Google Scholar] [CrossRef]
- Lu, Q.; Wang, L. Corrosion evolution and stress corrosion cracking of E690 steel for marine construction in artificial seawater under potentiostatic anodic polarization. Constr. Build. Mater. 2020, 238, 117763. [Google Scholar] [CrossRef]
- Tian, H.; Wang, X. Electrochemical corrosion, hydrogen permeation and stress corrosion cracking behavior of E690 steel in thiosulfate-containing artificial seawater. Corros. Sci. 2018, 144, 145–162. [Google Scholar] [CrossRef]
- Ma, H.; Liu, Z. Comparative study of the SCC behavior of E690 steel and simulated HAZ microstructures in a SO2-polluted marine atmosphere. Mater. Sci. Eng., A. 2016, 650, 93–101. [Google Scholar] [CrossRef]
- Ma, H.; Du, C. Effect of SO2 content on SCC behavior of E690 high-strength steel in SO2-polluted marine atmosphere. Ocean Eng. 2018, 164, 256–262. [Google Scholar] [CrossRef]
- Ma, H.; Chen, L. Effect of prior austenite grain boundaries on corrosion fatigue behaviors of E690 high strength low alloy steel in simulated marine atmosphere. Mater. Sci. Eng., A. 2020, 773, 138884. [Google Scholar] [CrossRef]
- Ma, H.; Liu, Z. Effect of cathodic potentials on the SCC behavior of E690 steel in simulated seawater. Mater. Sci. Eng., A. 2015, 642, 22–31. [Google Scholar] [CrossRef]
- Gardiner, C.P.; Melchers, R.E. Corrosion analysis of bulk carriers, Part I: operational parameters influencing corrosion rates. Mar. Struct. 2003, 16, 547–566. [Google Scholar] [CrossRef]
- Melchers, R.E. Effect of small compositional changes on marine immersion corrosion of low alloy steels. Corros. Sci. 2004, 46, 1669–1691. [Google Scholar] [CrossRef]
- Melchers, R.E.; Jeffrey, R. Early corrosion of mild steel in seawater. Corros. Sci. 2005, 47, 1678–1693. [Google Scholar] [CrossRef]
- Robert, E. Melchers, Robert Jeffrey. 2012. Corrosion of long vertical steel strips in the marine tidal zone and implications for ALWC. Corros. Sci. 2012, 65, 26–36. [Google Scholar]
- Traverso, P.; Canepa, E. A review of studies on corrosion of metals and alloys in deep-sea environment. Ocean Eng. 2014, 87, 10–15. [Google Scholar] [CrossRef]
- Wall, H.; Wadsö, L. Corrosion rate measurements in steel sheet pile walls in a marine environment. Mar. Struct. 2013, 33, 21–32. [Google Scholar]
- Committee, C.N.S.M. GB/T 12763.4-2007, in Code for marine surveys Part 4 Investigation of seawater chemical elements. 2008, China Standard Press: Beijing.
- Deng, P.; Li, Z. Vertical galvanic corrosion of pipeline steel in simulated marine thermocline. Ocean Eng. 2020, 217, 107584. [Google Scholar]
- Hu, J.; Deng, P. The vertical Non-uniform corrosion of Reinforced concrete exposed to the marine environments. Constr. Build. Mater. 2018, 183, 180–188. [Google Scholar] [CrossRef]

| C | Si | Mn | P | S | Cr | Ni | Cu | Mo | V | Als | Fe |
| 0.15 | 0.20 | 1.00 | 0.0058 | 0.0014 | 0.99 | 1.45 | 0.0091 | 0.37 | 0.03 | 0.036 | Bal. |
| Depth (cm) |
Time | ||||
|---|---|---|---|---|---|
| 7th day | 20th day | 27th day | 44th day | 59th day | |
 | |||||
| Depth (cm) |
Time | ||||
|---|---|---|---|---|---|
| 7th day | 20th day | 27th day | 44th day | 59th day | |
 | |||||
| Depth (cm) |
Time | ||||
|---|---|---|---|---|---|
| 7th day | 20th day | 27th day | 44th day | 59th day | |
 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




