Submitted:
18 January 2024
Posted:
19 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Stress, glucocorticoids and the hippocampus
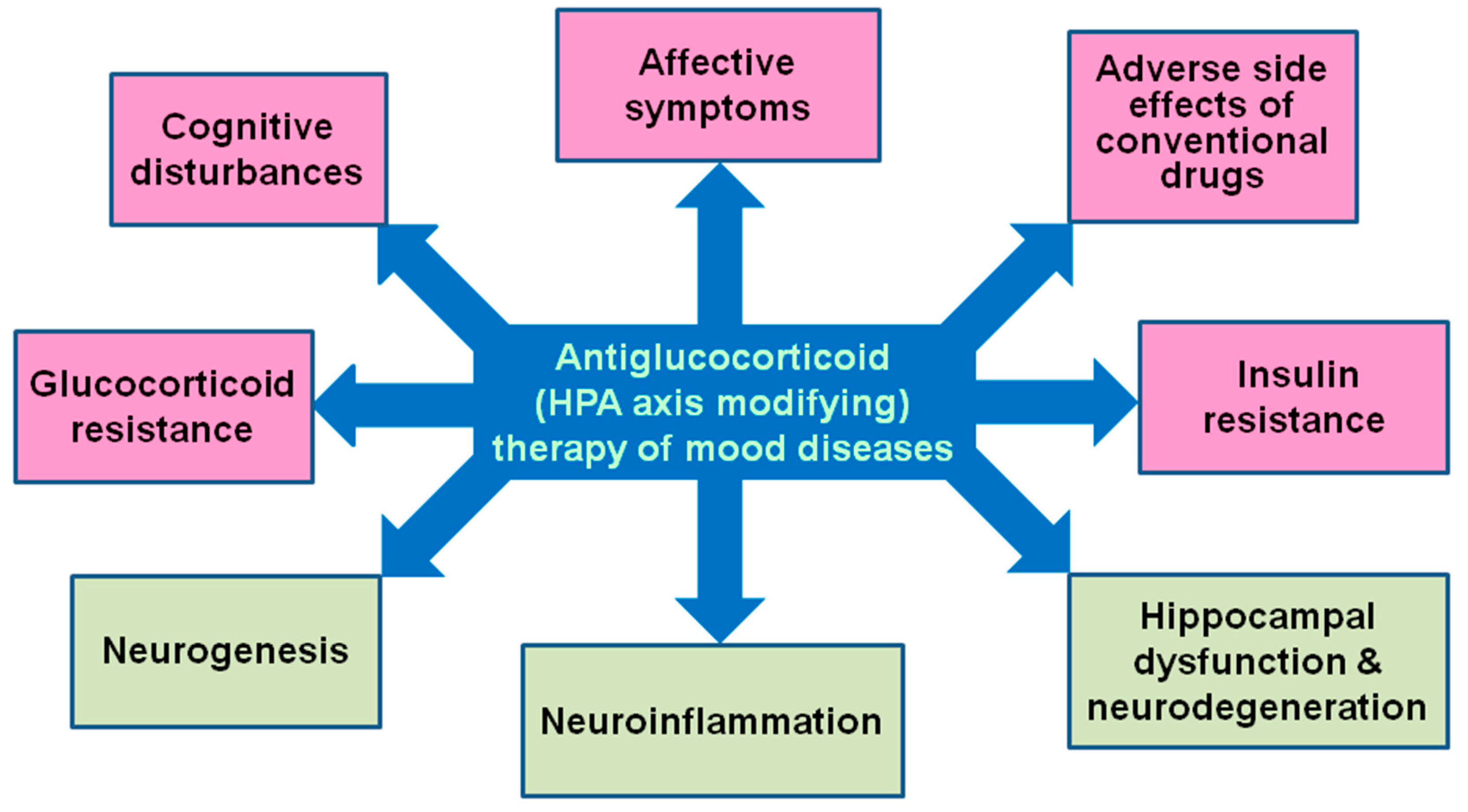
3. HPA dysfunction and mood diseases: Rationale for antiGC development
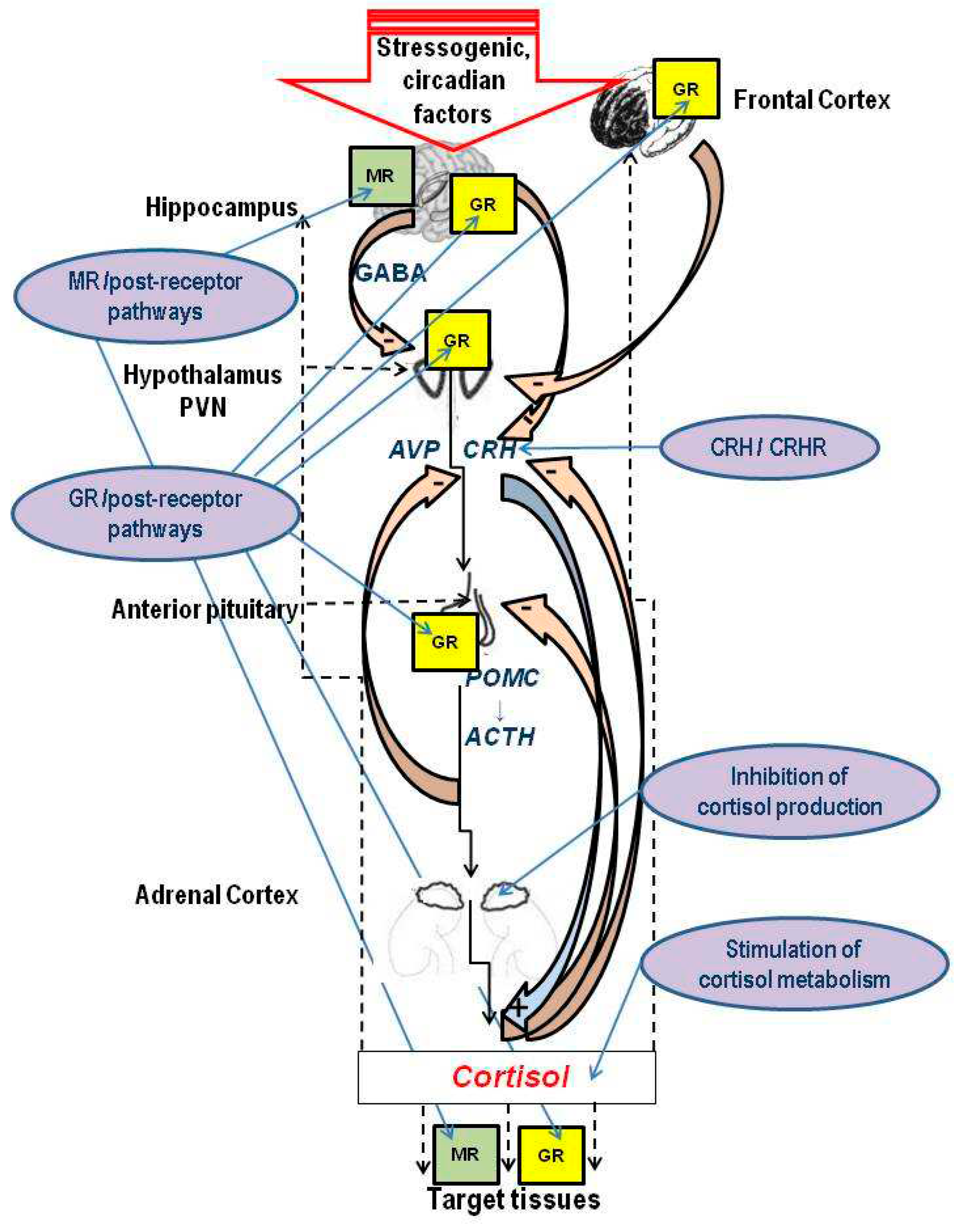
4. What cortisol level is abnormal: Cautionary notes
5. GR antagonism
5.1. Mifepristone (RU 486) as a prototypic GR antagonist
5.2. Development of GR antagonists
5.3. Endogenous antiGCs
6. AntiGC treatment of affective disorders
6.1. Major depressive disease
6.2. Treatment-resistant depression
6.3. Bipolar disorder, schizophrenia
6.4. Beyond affective symptoms: Cognitive dysfunction, metabolic disturbances, diabetes
7. Conclusions
Funding
Conflicts of Interest
Abbrevations
References
- McEwen, B.S., Akil. H. Revisiting the Stress Concept: Implications for Affective Disorders. J. Neurosci. 2020, 40,:12-21. [CrossRef]
- Dhikav, V., Anand, K.S. Is hippocampal atrophy a future drug target? Med. Hypotheses. 2007, 68, 1300-1306. [CrossRef]
- Gulyaeva, N.V. Functional Neurochemistry of the Ventral and Dorsal Hippocampus: Stress, Depression, Dementia and Remote Hippocampal Damage. Neurochem Res. 2019, 44, 1306-1322. [CrossRef]
- Gulyaeva, N V. Biochemical Mechanisms and Translational Relevance of Hippocampal Vulnerability to Distant Focal Brain Injury: The Price of Stress Response. Biochemistry (Mosc.) 2019, 84, 1306-1328. [CrossRef]
- Uchoa, E.T., Aguilera, G., Herman, J.P., Fiedler, J.L., Deak, T., de Sousa, M.B. Novel aspects of glucocorticoid actions. J. Neuroendocrinol. 2014, 26, 557-572. [CrossRef]
- de Kloet, E.R., Joëls, M. The cortisol switch between vulnerability and resilience. Mol. Psychiatry. 2023. [CrossRef]
- Gulyaeva, N.V. Glucocorticoids Orchestrate Adult Hippocampal Plasticity: Growth Points and Translational Aspects. Biohemistry (Mosc.) 2023, 88, 565-589. [CrossRef]
- Joëls, M., Sarabdjitsingh, R.A., Karst, H. Unraveling the time domains of corticosteroid hormone influences on brain activity: rapid, slow, and chronic modes. Pharmacol Rev. 2012, 64, 901-938. [CrossRef]
- de Kloet, E.R., de Kloet, S.F., de Kloet, C.S., de Kloet, A.D. Top-down and bottom-up control of stress-coping. J. Neuroendocrinol. 2019, 31, e12675. [CrossRef]
- Daskalakis, N.P., Meijer, O.C., de Kloet, E.R. Mineralocorticoid receptor and glucocorticoid receptor work alone and together in cell-type-specific manner: Implications for resilience prediction and targeted therapy. Neurobiol. Stress. 2022,18, 100455. [CrossRef]
- de Kloet, E.R. Functional profile of the binary brain corticosteroid receptor system: mediating, multitasking, coordinating, integrating. Eur J Pharmacol. 2013, 719, 53-62. [CrossRef]
- Sousa, N., Cerqueira, J.J., Almeida, O.F. Corticosteroid receptors and neuroplasticity. Brain Res. Rev. 2008, 57, 561-570. [CrossRef]
- Madalena, K.M., Lerch, J.K. The Effect of Glucocorticoid and Glucocorticoid Receptor Interactions on Brain, Spinal Cord, and Glial Cell Plasticity. Neural Plast. 2017, 8640970. [CrossRef]
- Jeanneteau, F., Borie, A., Chao, M.V., Garabedian, M.J. Bridging the Gap between Brain-Derived Neurotrophic Factor and Glucocorticoid Effects on Brain Networks. Neuroendocrinology. 2019, 109, 277-284. [CrossRef]
- Kim, J.S., Iremonger, K.J. Temporally Tuned Corticosteroid Feedback Regulation of the Stress Axis. Trends Endocrinol. Metab. 2019, 30, 783-792. [CrossRef]
- Suri, D., Vaidya, V.A. Glucocorticoid regulation of brain-derived neurotrophic factor: relevance to hippocampal structural and functional plasticity. Neuroscience. 2013, 239, 196-213. [CrossRef]
- Podgorny, O.V., Gulyaeva, N.V. Glucocorticoid-mediated mechanisms of hippocampal damage: Contribution of subgranular neurogenesis. J. Neurochem. 2021, 157, 370-392. [CrossRef]
- Reus, V.I., Wolkowitz, O.M. Antiglucocorticoid drugs in the treatment of depression. Expert Opin. Investig. Drugs. 2001, 10, 1789-1796. [CrossRef]
- Resmini, E., Santos, A., Webb, S.M. Cortisol Excess and the Brain. Front. Horm. Res. 2016, 46, 74-86. [CrossRef]
- Hill, A.R., Spencer-Segal, J.L. Glucocorticoids and the Brain after Critical Illness. Endocrinology. 2021, 162, bqaa242. [CrossRef]
- Gulyaeva, N.V. Molecular Mechanisms of Neuroplasticity: An Expanding Universe. Biochemistry (Mosc). 2017, 82, 237-242. [CrossRef]
- Bolshakov, A.P., Tret'yakova, L.V., Kvichansky, A.A., Gulyaeva, N.V. Glucocorticoids: Dr. Jekyll and Mr. Hyde of Hippocampal Neuroinflammation. Biochemistry (Mosc.). 2021, 86, 156-167. [CrossRef]
- Martocchia, A., Stefanelli, M., Falaschi, G.M., Toussan, L., Rocchietti March, M., Raja, S., Romano, G., Falaschi, P. Targets of anti-glucocorticoid therapy for stress-related diseases. Recent Pat. CNS Drug Discov. 2013,;8, 79-87. [CrossRef]
- Appendix B - Common Laboratory Tests. in: Pharmacotherapy Principles and Practice, 6th Edition. Chisholm-Burns, M. A., Schwinghammer, T. L., Malone, P. M., Kolesar, J. M., Lee, K.C., Bookstaver P. B., eds. McGraw-Hill Medical 2022.
- Wang, J., Guan, Q., Sheng, Y., Yang, Y., Guo, L., Li, W., Gu, Y., Han, C. The potential predictive value of salivary cortisol on the occurrence of secondary cognitive impairment after ischemic stroke. Neurosurg. Rev. 2021, 44, 1103–1108. [CrossRef]
- Ben Assayag, E., Tene, O., Korczyn, A. D., Shopin, L., Auriel, E., Molad, J., Hallevi, H., Kirschbaum, C., Bornstein, N. M., Shenhar-Tsarfaty, S., Kliper, E., Stalder, T. High hair cortisol concentrations predict worse cognitive outcome after stroke: Results from the TABASCO prospective cohort study. Psychoneuroendocrinology. 2017, 82, 133–139. [CrossRef]
- Tene, O., Hallevi, H., Korczyn, A. D., Shopin, L., Molad, J., Kirschbaum, C., Bornstein, N. M., Shenhar-Tsarfaty, S., ., Kliper, E., Auriel, E., Usher, S., Stalder, T., Ben Assayag, E. The Price of Stress: High Bedtime Salivary Cortisol Levels Are Associated with Brain Atrophy and Cognitive Decline in Stroke Survivors. Results from the TABASCO Prospective Cohort Study. J. Alzheimers Dis. 2018, 65, 1365–1375. [CrossRef]
- Lombardo, G., Enache, D., Gianotti, L., Schatzberg, A.F., Young, A.H., Pariante, C.M., Mondelli, V. Baseline cortisol and the efficacy of antiglucocorticoid treatment in mood disorders: A meta-analysis. Psychoneuroendocrinology. 2019, 110, 104420. https://doi.org/10.1016/j.psyneuen.2019.104420.
- Zalachoras, I., Houtman, R., Atucha, E., Devos, R., Tijssen, A.M., Hu, P., Lockey, P.M., Datson, N.A., Belanoff, J.K., Lucassen, P.J., Joëls, M., de Kloet, E.R., Roozendaal, B., Hunt, H., Meijer, O.C. Differential targeting of brain stress circuits with a selective glucocorticoid receptor modulator. Proc. Natl. Acad. Sci .U. S. A. 2013, 110, 7910-7915. [CrossRef]
- De Nicola, A.F., Meyer, M., Guennoun, R., Schumacher, M., Hunt, H., Belanoff, J., de Kloet, E.R., GonzalezDeniselle, M,C. Insights into the Therapeutic Potential of Glucocorticoid Receptor Modulators for Neurodegenerative Diseases. Int. J. Mol. Sci. 2020, 21, 2137. [CrossRef]
- de Kloet, E.R. Glucocorticoid feedback paradox: a homage to Mary Dallman. Stress. 2023, 26, 2247090. [CrossRef]
- Dalm, S., Karssen, A.M., Meijer, O.C., Belanoff, J.K., de Kloet, E.R.. Resetting the Stress System with a Mifepristone Challenge. Cell. Mol. Neurobiol. 2019, 39, :503-522. [CrossRef]
- Kakade, A.S., Kulkarni, Y.S. Mifepristone: current knowledge and emerging prospects. J. Indian Med. Assoc. 2014, 112, 36-40. PMID: 25935948.
- Fleseriu, M., Biller, B.M., Findling, J.W., Molitch, M.E., Schteingart, D.E., Gross, C.; SEISMIC Study Investigators. Mifepristone, a glucocorticoid receptor antagonist, produces clinical and metabolic benefits in patients with Cushing's syndrome. J. Clin. Endocrinol. Metab. 2012, 97, 2039-2049. [CrossRef]
- DeBattista, C., Belanoff, J., Glass, S., Khan, A., Horne, R.L., Blasey, C., Carpenter, L.L., Alva, G. Mifepristone versus placebo in the treatment of psychosis in patients with psychotic major depression. Biol. Psychiatry. 2006, 60, 1343-1349. [CrossRef]
- Cotella, E.M., Morano, R.L., Wulsin, A.C., Martelle, S.M., Lemen, P., Fitzgerald, M., Packard, B.A., Moloney, R.D., Herman, J.P. Lasting Impact of Chronic Adolescent Stress and Glucocorticoid Receptor Selective Modulation in Male and Female Rats. Psychoneuroendocrinology. 2020, 112, 104490. [CrossRef]
- Garner,B., Phassouliotis, C., Phillips, L.J., Markulev, C., Butselaar, F., Bendall, S., Yun, Y., McGorry, P.D. Cortisol and dehydroepiandrosterone-sulphate levels correlate with symptom severity in first-episode psychosis. J. Psychiat.r Res. 2011, 45, 249-255. [CrossRef]
- Young, A.H., Gallagher, P., Porter, R.J. Elevation of the cortisol-dehydroepiandrosterone ratio in drug-free depressed patients. Am. J. Psychiatry. 2002, 159, 1237-1239. [CrossRef]
- Souza-Teodoro, L.H., de Oliveira, C., Walters, K., Carvalho, L.A. Higher serum dehydroepiandrosterone sulfate protects against the onset of depression in the elderly: Findings from the English Longitudinal Study of Aging (ELSA). Psychoneuroendocrinology. 2016, 64, 40-46. [CrossRef]
- Markopoulou, K., Papadopoulos, A., Juruena, M.F., Poon, L., Pariante, C.M., Cleare, A.J. The ratio of cortisol/DHEA in treatment resistant depression. Psychoneuroendocrinology. 2009, 34, 19-26. [CrossRef]
- Dumas, T.C., Gillette, T., Ferguson, D., Hamilton, K., Sapolsky, R.M. Anti-glucocorticoid gene therapy reverses the impairing effects of elevated corticosterone on spatial memory, hippocampal neuronal excitability, and synaptic plasticity. J. Neurosci. 2010, 30, 1712-1720. [CrossRef]
- Rădulescu, I., Drăgo,i A.M., Trifu, S.C., Cristea, M.B. Neuroplasticity and depression: Rewiring the brain's networks through pharmacological therapy (Review). Exp. Ther. Med. 2021, 22, 1131. [CrossRef]
- Fitzsimons, C.P,. van Hooijdonk, L.W., Morrow, J.A., Peeters, B.W., Hamilton, N., Craighead, M., Vreugdenhil, E. Antiglucocorticoids, neurogenesis and depression. Mini Rev. Med. Chem. 2009, 9, 249-264. [CrossRef]
- Murphy, B.E. Antiglucocorticoid therapies in major depression: a review. Psychoneuroendocrinology. 1997, 22 Suppl 1, S125-S132. PMID: 9264159. [CrossRef]
- Wolkowitz ,O.M., Reus, V.I. Treatment of depression with antiglucocorticoid drugs. Psychosom. Med. 1999, 6, 698-711. [CrossRef]
- Schüle, C., Baghai, T.C., Eser, D., Rupprecht, R. Hypothalamic-pituitary-adrenocortical system dysregulation and new treatment strategies in depression. Expert Rev. Neurother. 2009, 9, 1005-1019. [CrossRef]
- Maric, N.P., Adzic, M. Pharmacological modulation of HPA axis in depression - new avenues for potential therapeutic benefits. Psychiatr. Danub. 2013, 25, 299-305. PMID: 24048401.
- Wolkowitz, O.M., Reus, V.I., Manfredi, F., Ingbar, J., Brizendine, L. Antiglucocorticoid strategies in hypercortisolemic states. Psychopharmacol. Bull. 1992, 28, 247-251. PMID: 1480727.
- Wolkowitz, O.M., Reus, V.I., Manfredi ,F., Ingbar, J., Brizendine, L., Weingartner, H. Ketoconazole administration in hypercortisolemic depression. Am. J. Psychiatry. 1993, 150, 810-812. [CrossRef]
- Anand, A., Malison, R., McDougle, C.J., Price, L.H. Antiglucocorticoid treatment of refractory depression with ketoconazole: a case report. Biol. Psychiatry. 1995, 37, 338-340. [CrossRef]
- Wolkowitz, O.M., Reus, V.I., Chan, T., Manfredi, F., Raum, W., Johnson, R., Canick, J. Antiglucocorticoid treatment of depression: double-blind ketoconazole. Biol. Psychiatry. 1999, 45,1070-1074. [CrossRef]
- Murphy, B.E., Filipini, D., Ghadirian, A.M. Possible use of glucocorticoid receptor antagonists in the treatment of major depression: preliminary results using RU 486. J. Psychiatry Neurosci. 1993, 18, 209-213. PMID: 8297920.
- Reus, V.I., Wolkowitz, O.M., Frederick, S. Antiglucocorticoid treatments in psychiatry. Psychoneuroendocrinology. 1997, 22 Suppl 1, S121- S124. [CrossRef]
- Healy, D.G., Harkin, A., Cryan, J.F., Kelly, J.P., Leonard, B.E. Metyrapone displays antidepressant-like properties in preclinical paradigms. Psychopharmacology (Berl.) 1999, 145, 303-308. [CrossRef]
- Hirtz, R., Libuda, L., Hinney, A., Föcker, M., Bühlmeier, J., Holterhus, P.M., Kulle, A., Kiewert, C., Hauffa, B.P., Hebebrand, J., Grasemann, C. The adrenal steroid profile in adolescent depression: a valuable bio-readout? Transl. Psychiatry. 2022, 12, 255. [CrossRef]
- Ding, Y., Wei, Z., Yan, H., Guo, W. Efficacy of Treatments Targeting Hypothalamic-Pituitary-Adrenal Systems for Major Depressive Disorder: A Meta-Analysis. Front. Pharmacol. 2021, 12, 732157. [CrossRef]
- McAllister-Williams, R.H., Smith, E., Anderson, I.M., Barnes, J., Gallagher, P., Grunze, H.C., Haddad, P.M., House, A.O., Hughes, T., Lloyd, A.J., McColl, E.M., Pearce, S.H., Siddiqi, N., Sinha, B., Speed, C., Steen, I.N., Wainright, J., Watson, S., Winter, F.H., Ferrier, I.N. Study protocol for the randomised controlled trial: antiglucocorticoid augmentation of anti-Depressants in Depression (The ADD Study). BMC Psychiatry. 2013, 13, 205. [CrossRef]
- Sigalas, P.D., Garg, H., Watson, S., McAllister-Williams, R.H., Ferrier, I.N. Metyrapone in treatment-resistant depression. Ther. Adv. Psychopharmacol. 2012, 2, 139-149. [CrossRef]
- Strawbridge, R., Jamieson, A., Hodsoll, J., Ferrier, I.N., McAllister-Williams, R.H., Powell, T.R., Young, A.H., Cleare, A.J., Watson, S. The Role of Inflammatory Proteins in Anti-Glucocorticoid Therapy for Treatment-Resistant Depression. J. Clin. Med. 2021, 10, 784. PMID: 33669254. [CrossRef]
- McAllister-Williams, R.H., Anderson I,M., Finkelmeyer, A., Gallagher, P., Grunze, H.C., Haddad, P.M., Hughes, T., Lloyd, A.J., Mamasoula, C., McColl, E., Pearce, S., Siddiqi, N., Sinha, B.N., Steen, N., Wainwright, J., Winter, F.H., Ferrier, I.N., Watson, S.; ADD Study Team. Antidepressant augmentation with metyrapone for treatment-resistant depression (the ADD study): a double-blind, randomised, placebo-controlled trial. Lancet Psychiatry. 2016, 3, 117-127. [CrossRef]
- Ferrier, I.N., Anderson, I.M., Barnes, J., Gallagher, P., Grunze, H.C.R., Haddad, P.M., House, A.O., Hughes, T., Lloyd, A.J., Mamasoul,a C., McColl. E., Pearce, S., Siddiqi, N., Sinha, B., Speed, C., Steen, N., Wainwright, J., Watson, S., Winter, F.H., McAllister-Williams ,R.H.; the ADD Study Team. Randomised controlled trial of Antiglucocorticoid augmentation (metyrapone) of antiDepressants in Depression (ADD Study). Southampton (UK): NIHR Journals Library; 2015, PMID: 26086063. [CrossRef]
- Young, A.H., Gallagher, P., Watson, S., Del-Estal D., Owen, B.M., Ferrier, I.N. Improvements in neurocognitive function and mood following adjunctive treatment with mifepristone (RU-486) in bipolar disorder. Neuropsychopharmacology. 2004, 29, 1538-1545. [CrossRef]
- Gallagher, P., Watson, S., Elizabeth Dye, C., Young, A.H., Nicol Ferrier, I. Persistent effects of mifepristone (RU-486) on cortisol levels in bipolar disorder and schizophrenia. J. Psychiatr. Res. 2008, 42, 1037-1041. [CrossRef]
- Marco, E.J., Wolkowitz, O.M., Vinogradov, S., Poole, J.H., Lichtmacher, J., Reus, V.I. Double-blind antiglucocorticoid treatment in schizophrenia and schizoaffective disorder: a pilot study. World J. Biol. Psychiatry. 2002, 3,156-161. [CrossRef]
- Bauer, M.E, Teixeira, A.L. Inflammation in psychiatric disorders: what comes first? Ann. N. Y. Acad .Sci. 2019, 1437, 57-67. [CrossRef]
- Jeon, S.W., Kim, Y.K. The role of neuroinflammation and neurovascular dysfunction in major depressive disorder. J. Inflamm. Res. 2018 ,11, 179-192. [CrossRef]
- Herbert, J., Lucassen, P.J. Depression as a risk factor for Alzheimer's disease: Genes, steroids, cytokines and neurogenesis - What do we need to know? Front. Neuroendocrinol. 2016, 41, 153-171. [CrossRef]
- Adzic, M., Brkic, Z., Mitic, M., Francija, E., Jovicic, M.J., Radulovic, J., Maric, N.P. Therapeutic Strategies for Treatment of Inflammation-related Depression. Curr. Neuropharmacol. 2018, 16, 176-209. [CrossRef]
- Linnemann, C., Lang, U.E. Pathways Connecting Late-Life Depression and Dementia. Front. Pharmacol. 2020,11, 279. [CrossRef]
- Hunt, H.J., Donaldson, K., Strem, M., Tudor, I.C., Sweet-Smith, S., Sidhu, S. Effect of Miricorilant, a Selective Glucocorticoid Receptor Modulator, on Olanzapine-Associated Weight Gain in Healthy Subjects: A Proof-of-Concept Study. J. Clin. Psychopharmacol. 2021, 41, 632-637. [CrossRef]
- Detka, J., Kurek, A., Basta-Kaim, A, Kubera, M., Lasoń, W., Budziszewska, B. Neuroendocrine link between stress, depression and diabetes. Pharmacol. Rep. 2013, 65, 1591-1600. [CrossRef]
- Roat-Shumway, S., Wroolie, T.E., Watson, K., Schatzberg, A.F., Rasgon N.L. Cognitive effects of mifepristone in overweight, euthymic adults with depressive disorders. J. Affect. Disord. 2018, 239,242-246. [CrossRef]
- Gray, J.D., Kogan, J.F., Marrocco, J., McEwen, B.S. Genomic and epigenomic mechanisms of glucocorticoids in the brain. Nat. Rev. Endocrinol. 2017,13, 661-673. [CrossRef]
- Gallagher, P., Malik, N., Newham, J., Young, A.H., Ferrier, I.N., Mackin, P. Antiglucocorticoid treatments for mood disorders. Cochrane Database Syst. Rev. 2008, CD005168. Update in: Cochrane Database Syst. Rev. 2015, CD005168. [CrossRef]
- Wulsin, A.C., Kraus, K.L., Gaitonde, K.D., Suru, V., Arafa, S.R., Packard, B.A., Herman, J.P., Danzer, S.C. The glucocorticoid receptor specific modulator CORT108297 reduces brain pathology following status epilepticus. Exp. Neurol. 2021, 341, 113703. [CrossRef]
- Meijer, O.C., Koorneef, L.L., Kroon, J. Glucocorticoid receptor modulators. Ann. Endocrinol (Paris). 2018, 79, 107-111. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).