1. Introduction
Among the wide variety of human genes dedicated to membrane transport processes, up to half of them encode members of the SLC solute carrier superfamily. The proton-coupled oligopeptide transporters (POTs) make up the SLC15 family (reviewed in [
1]). The SLC15 family is also part of the major facilitator superfamily, and transporters encoded by this family have 12 transmembrane spanning regions with both amino and car-boxy-termini facing the intracellular side, and pseudosymmetry between the structural motifs [
2]. To date, five different membrane transporters encoded by the SLC15 family have been identified. The oligopeptide transporters PepT1 (
SLC15A1) and PepT2 (
SLC15A2) are mainly expressed in the apical membranes of the small intestine and kidney proximal tubules, respectively, and their physiological roles have been well characterized [
3]. In contrast, peptide/histidine transporters PHT2 (
SLC15A3) and PHT1 (
SLC15A4) are expressed in endosomal and lysosomal membranes of immune cells, where they play essential roles in metabolic and inflammatory signaling events [
4]. The recently found member of this family,
SLC15A5, was identified by bioinformatic approaches and its expression profile and functional properties are still unclear [
5].
Members of the SLC15 family are known for their substrate promiscuity, as they can transport a broad range of oligopeptides and a variety of peptide-like drugs. PepT1 and PepT2 substrates include di- and tripeptides, beta-lactam antibiotics and angiotensin converting enzyme (ACE) inhibitors, among many others [
6]. Interestingly, in addition to di- and tripeptides, PHT2 and PHT1 have been shown to transport free AAs, such as histidine [
6]. Concerning PHT1, it has been shown to transport carnosine, valacyclovir, and bacterial-derived peptides such as muramyl dipeptide (MDP) or L-Ala-γ-D-Glu-meso-diaminopimelic acid (Tri-DAP), among other nucleotide-binding oligomerization domain-like receptor (NOD) ligands [
1,
2,
4,
7,
8,
9]). Unfortunately, experimental validation and detailed kinetic analysis for these substrates is still incomplete and has been hampered by the localization of PHT1 in the endosomal-lysosomal organelles. Various expression systems have been used to generate plasma membrane expression for functional analysis. However, the published work has given rise to significant inconsistencies [
2,
7,
8,
10,
11].
PHT1 (
SLC15A4) was cloned from a rat brain cDNA library [
7], while the human homologue was cloned from an intestinal cDNA library and Caco-2 cells [
12]. The human
SLC15A4 gene encodes a 577-amino acid membrane protein whose expression has been found primarily in brain, intestine, some tumor cells and a variety of immune cells, including monocytes/macrophages and dendritic cells [
3]. PHT1 has been shown by immunofluorescence to be localized in lysosomes and late endosomes [
13], where it plays a pivotal role in lysosomal activity by controlling the levels of certain amino acids (AAs) and oligopeptides and/or by scaffolding various signaling molecules involved in different metabolic and inflammatory events [
4]. Due to the pH buffering ability of histidine, H
+-coupled transport of this amino acid by PHT1 contributes to the regulation of lysosomal acidic levels, which is key for the activity of lysosomal enzymes, such as cathepsins, or integrity of transport proteins, such as the V-ATPase. In this context, dysregulation of lysosomal pH due to PHT1 dysfunction has been associated with impaired Toll-like receptors (TLRs) mediated type I interferon (IFN-I) production [
13,
14], and also, with a failure of the IFN regulatory factor 7 (IRF7)-IFN-I regulatory circuit due to disruption of the mammalian target of rapamycin (mTOR) pathway [
15]. In addition, PHT1 mediated transport of bacterial oligopeptides, such as MDP or Tri-DAP, triggers the activation of nuclear factor-kappa-B (NF-ΚƁ), which modulates the NOD-dependent immune response [
3]. In terms of scaffolding activity, it has been shown that PHT1 interacts with Ragulator components such as LAMTOR1 and LAMTOR2, suggesting that it may be a component of the mTORC1 supercomplex [
16]. In addition, PHT1 was demonstrated to interact with the “TLR adaptor interacting with SLC15A4 on the lysosome” (TASL), which modulates TLR7, TLR8 and TLR9 signaling and mediates IFN-I production by recruiting the transcription factor IRF5 [
17]. Taken together, these findings demonstrate that PHT1 (
SLC15A4) plays a key role in the immune response, so it is not surprising that several studies have implicated PHT1 in the development of diseases associated with immune dysfunction, such as systemic lupus erythematosus (SLE) [
18,
19,
20,
21,
22] and inflammatory bowel disease (IBD) [
19]. In addition, PHT1 has been implicated in other human diseases such as type 2 diabetes [
23], combined pituitary hormone deficiency [
24] and lung adenocarcinoma [
25].
As just described, PTH1 transport and scaffolding functions may affect the lysosome environment [
4,
26], enable NOD and TLR signaling [
3,
13,
14], and modulate mTOR activity in human immune cells [
4,
15]. Furthermore, it appears to play a pivotal role in the pathogenesis of SLE [
18,
19,
20,
21,
22]. Therefore, molecules capable of modulating PHT1 functions have great potential for therapeutic interventions aimed at modulating the immune response, such as required for SLE treatment. Despite the obvious therapeutic interest of PHT1, its pharmacological development has unfortunately been hampered by the lack of appropriate functional assays and limited structural information. With the aim to provide appropriate tools for the pharmacological development of PHT1, and also, address the inconsistencies surrounding the current understanding of its functional properties, the present work presents a new transport assay, based on solid supported membrane electrophysiology (SSME) [
27]. Using this novel SSME method, a characterization of some interesting functional properties of PHT1 is presented, including substrate selectivity among a variety of AAs and peptides, pH-dependence, and transport kinetics of the identified substrates L-histidine, L-arginine, L-lysine, His-Leu and Leu-Leu. Notably, in contrast to previous functional studies developed for PHT1 [
7,
8,
10,
11], this work has was carried out with isolated lysosomal membranes, allowing the protein to be studied in its native environment.
2. Materials and Methods
All chemicals and reagents were purchased from Merck KGaA unless otherwise stated.
2.1. Cell Culture
HEK293 cells were obtained from American Type Culture Collection (ATCC) cultured in Dulbecco’s modified Eagle medium (DMEM) supplemented with 10% foetal bovine serum (FBS), 10mM HEPES, 1mM sodium pyruvate, 100μM minimal essential medium (MEM) non-essential AAs and 100 units/ml penicillin/streptomycin and maintained under standard cell culture conditions (37°C; 5% CO2). To generate a cell line stably overexpressing (OE) human SLC15A4 (PHT1), HEK293 cells were transfected with the DNA construct encoding the PHT1-GFP C-term fusion protein, using Lipofectamine 2000 (Thermo Fisher Scientific) transfection reagent according to the manufacturer’s protocol. GFP-PTH1 OE cell line was obtained by a selection strategy based on resistance to G418 antibiotic (Thermo Fisher Scientific). The culture media used to maintain HEK293 GFP-PHT1 OE cell line were also supplemented with 500μg/mL G418.
2.2. Isolation of Lysosomal Fraction
HEK293 GFP-PHT1 OE cells or HEK293 wild-type cells were washed with PBS, trypsinized and centrifuged at 500 X g for 5 min to produce cell pellets of approximately 2g. Cell pellets were then resuspended in 5mL sucrose homogenization buffer (250mM sucrose, 20mM HEPES and cOmpleteTM protease inhibitor cocktail; pH 7.4 adjusted with NMDG) and homogenized by sonication (3X 10 sonication pulses, 0.5 sec, 20% potency). Homogenates were centrifuged at 500 X g for 5 min at 4°C to discard cell debris (P1). The resulting supernatant (S1) was centrifuged at 6,800 X g for 10 min at 4°C. The pellet (P2) containing the mitochondrial fraction was discarded and the remaining supernatant (S2) was centrifuged again at 20,000 X g for 30 min to generate a pellet (P3) containing the lysosomal fraction. S3 was discarded and P3 was resuspended in 5ml ice-cold sucrose homogenization buffer and centrifuged at 20,000 X g at 4°C for 15min. The supernatant (S4) was discarded and the remaining pellet (P4) containing a clean lysosomal fraction, was resuspended in 10mM citric acid buffer at pH 6.0 supplemented with the cOmpleteTM protease inhibitor cocktail and stored until use at -80°C.
2.3. Western Blot Analyses
Protein concentration was determined by Bradford colorimetric assay (Bio Rad). Equal amounts of lysosomal fraction samples (10µg) were resolved in 8% polyacrylamide gels and transferred onto PVDF membranes (Amersham). Immunodetection was achieved using the following antibodies: mouse monoclonal anti-GFP (SC-9996, Santa Cruz Biotechnology) (1/1000 dilution), mouse anti-LAMP1 (ab25630, Abcam) (1/100 dilution), mouse monoclonal anti β-actin (SC-47778, Santa Cruz Biotechnology) (1/2000 dilution) primary antibodies and goat anti-mouse IgG (#1721011, Bio Rad) (1/3000 dilution) secondary antibody. Antibody recognition signal was revealed by enhanced chemiluminescence detection reagent (Amersham) and blot images were acquired with the ChemiDoc Imaging System (Bio Rad).
2.4. Microscale Thermophoresis (MST)
MST measurements were performed with the Monolith NT.115 instrument (NanoTemper Technologies GmbH). Membrane preparation of HEK293 cells overexpressing PHT1-GFP were obtained as described elsewhere [
28]. Equal amounts of the membrane preparation were loaded into Monolith capillaries (NanoTemper; MO-K022) together with the indicated concentrations of L-histidine; both dissolved in 10mM citric acid buffer at pH 5.0. To measure the MST, a temperature gradient was generated by infrared (IR) laser (80% of IR laser power) and fluorescence (60% of Blue LED power) was recorded continuously for 35sec. Data were collected with MO. Control v2 and analyzed with MO. Affinity v2.3 software (NanoTemper).
2.5. Solid Supported Membrane-Based Electrophysiology (SSME)
The initial protein concentration of the lysosomal fraction preparations used for the SSME experiments was between 1.5 and 3.5 mg/ml. Prior to sensor preparation, to load the same amount of total protein, the protein concentration was adjusted in all experimental groups and then the samples were diluted 1:10 in 10 mM citric acid buffer at pH 5.0. SSME experiments were conducted using the SURFE2R N1 device (Nanion Technologies GmbH) and 3mm diameter gold sensors were prepared according to the standard protocol recommended by the manufacturer. Current traces were recorded using the SURFE2R N1 Control V1 1.6.0.1 software (Nanion Technologies GmbH). Currents were recorded for the entire duration of the solution exchange protocol and amplified with a current amplifier set to a gain of 109 V/A. All current traces shown in the same graph were recorded on the same sensor. For data analysis, magnitude of the peak of the current observed during the perfusion of the activating solution (Peakon) were used. To average data obtained from different sensors, the recorded Peakon currents were normalized to the control condition, which was the Peakon current magnitude recorded for L-histidine 10 mM in citric acid buffer at pH 5.0.
2.6. Molecular Docking
Receptor coordinate files, 8P6A and Q8N697, were downloaded from the PDB (
http://www.rcsb.org/) and AlphaFold (
https://alphafold.com/) databases, respectively. The ligand coordinate files were downloaded from Pubchem (
https://pubchem.ncbi.nlm.nih.gov/). The Pubchem IDs are as follows: 6274 (L-histidine), 5962 (L-lysine), 5950 (L-alanine), 6106 (L-Leucine), 6322 (L-Arginine), 6137 (L-Methionine), 9313 (Gly-Sar), 7687 (Leu-Leu), 189008 (His-Leu), 11163 (Gly-Gly), 11161 (Gly-Gly-Gly), 44093 (Captopril) and 5362119 (Lisinopril). The individual docking experiments were performed using the AutoDock Vina tool of the UCSF Chimera software version 1.17.3. Prior to molecular docking, the energy of the protein structures was minimized using standard parameters. A docking box containing the putative PHT1 substrate binding site was defined using the following parameters: 8P6A (center: 120, 120, 121; size: 13, 23, 20) and Q8N697 (center: 0.6, 0.13, 0.8: size: 25, 26, 16). The standard AutoDock Vina protocol was run, and the best scores were annotated. The H-bonds and residues involved were identified by visual inspection of the docking results showing the best scores.
2.7. Statistics
Comparison of means between experimental groups was performed using the unpaired Student’s t-test or the Mann-Whitney U test. The test was selected for each case according to the fit of the data to the normal distribution as assessed by the Shapiro-Wilk test. All statistical tests were performed with IBM SPSS Statistics 20 software. The significance level was set at p < 0.05.
3. Results
A new functional approach based on microscale thermophoresis (MST) technology has recently been established in our laboratory to study the ligand binding properties of SLC15 family members. In the initial work focusing on PepT1 (SLC15A1), a bacterial PepT1 orthologue from Physcomitrella patens was overexpressed in yeast, membrane preparations were isolated, and MST was used to assess the binding of PepT1 to some of its known substrates, such as the dipeptide glycylsarcosine (Gly-Sar) and the antiviral prodrug valacyclovir [
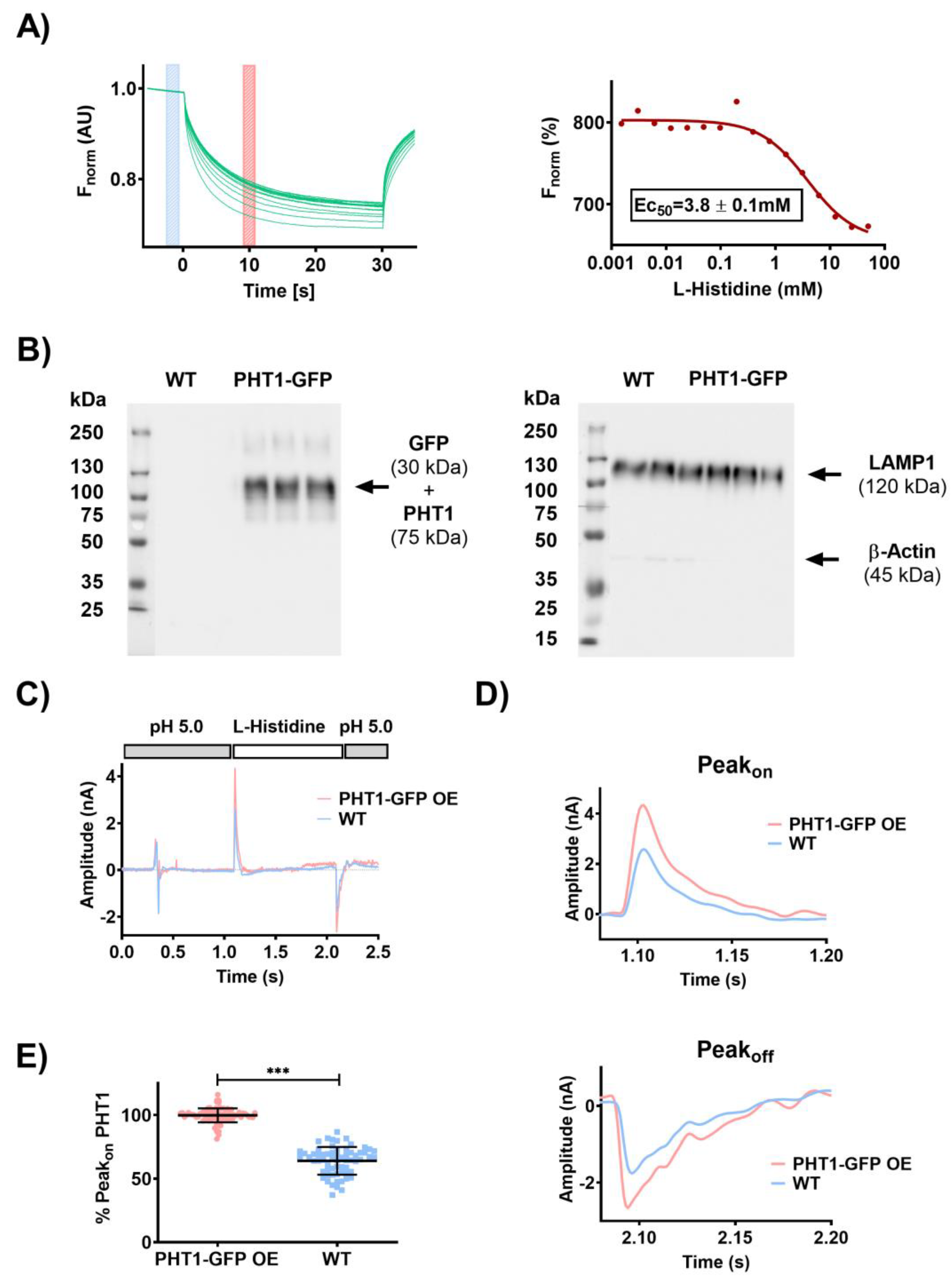
29]. Following this work, MST was used to study the functional properties of human PHT1 (SLC15A4). Briefly, a HEK293 cell line stably overexpressing PHT1 tagged with C-terminal GFP (hereafter referred to as PHT1-GFP OE) was generated and membrane preparations were isolated. The MST of PHT1-GFP was monitored by applying a temperature gradient in the presence of different substrate concentrations and/or buffer conditions. Representative MST traces recorded for the binding of a serial dilution of L-histidine to HEK293 PHT1-GFP OE membrane preparations are shown (
Figure 1A). MST traces were used to calculate the dissociation constant (K
D) for L-histidine, which was 4.1±0.5mM. In addition, binding properties were determined for a broad range of other potential PHT1 ligands, which will be published elsewhere, upon completion. These MST studies were the starting point of the present work; therefore, the same expression system has been used.
The main objective of the present work is to develop a transport assay that can be used to study the functional properties of PHT1, and moreover, serve as a tool for its pharmacological development. Secondly, it is expected that this assay will be instrumental in assessing whether the ligand binding properties observed for PHT1 by MST or other binding methodologies represent a reflection of its actual transport activities. And therefore, allowing follow-up findings to emerge from such methodologies, including the validation of new substrates and potential modulators of PHT1. Since PHT1 has been reasonable well established as a H
+-coupled symporter [
7,
8,
10,
11], its transport of substrates is expected to be electrogenic, i.e., a net translocation of positive charge should be associated with each transport cycle. Therefore, it was expected that SSME would provide an excellent method to study the transport function of PHT1, analogous to what has been successfully done for PepT1 [
27].
3.1. SSME—A New Approach to Study the Functional Activity of PHT1 (SLC15A4)
SSME uses a coated gold sensor to physically absorb membrane fractions or vesicles that should contain large amounts of the protein under study. To obtain such membrane preparations, since PHT1 is found in intracellular compartments, a protocol to isolate lysosomal membranes was followed. Briefly, large amounts of HEK293 cells, wild type (hereinafter referred to as WT) or PHT1-GFP OE, were disaggregated by sonication and the corresponding lysosomal fractions were isolated by a series of ultracentrifugation steps, as described elsewhere [
30]. PHT1-GFP expression in those fractions was validated by Western blot analysis using an anti-GFP antibody (
Figure 1B, left panel). In addition, expression levels of the cytosolic marker β-actin and the lysosomal marker LAMP1 were determined as negative and positive controls, respectively, to validate the purity of the isolated lysosomal membrane preparation (
Figure 1B, right panel).
To perform SSME recordings, PHT1-GFP OE lysosomal membranes were placed on coated gold sensors and mounted into the SURFER
2R N1 SSME device. Activation of the PHT1-GFP transport activity is achieved by rapid solution exchange of its substrate L-histidine in a buffer that provides the H
+ required for H
+-coupled histidine transport (
Figure 1C). The H
+ influx into the lysosomal membrane vesicles charges the gold sensor by capacitive coupling, which generates an on-peak (Peak
on) and, at the same time, a membrane potential in the lysosomal membrane vesicles. Due to the voltage dependence of the transport process, when the lysosomal membrane voltage reaches the equilibrium potential, H
+ transport is arrested, and the current is 0. The observed current decay is the result of the reduction of the transport rate while getting closer to the equilibrium potential. Washout of L-histidine by perfusion of L-histidine-free buffer through the gold sensor leads to H
+ efflux and generates the off-peak (Peak
off). The observed asymmetry between the Peak
on and Peak
off could reflect different transport kinetics for influx and efflux transport processes (
Figure 1D). Alternatively, it could be a consequence of differences in the driving force for each reaction. Peak
on is induced by the concentration gradient in the absence of a membrane potential, whereas Peak
off is induced by a different concentration gradient and the membrane potential that was generated through H
+ / L-histidine influx. The measurements were performed at pH 5.0, so the net charge of L-histidine was +1. Thus, the charge transfer to the sensors due to PHT1 mediated transport is the result of the contribution of both H
+ and L-histidine. Likewise, non-specific currents might arise from direct interaction of L-histidine with the membrane surface or from transport via other endogenous transporters present in the lysosomal membranes.
To determine the contribution of non-specific currents to the electrical signal induced by PHT1 transport, the same recordings were performed with the lysosomal membranes isolated from WT cells. On-peaks recorded with WT lysosomal membranes upon L-histidine perfusion were on average ~40% lower than those observed for PHT1-GFP OE lysosomal membranes (
Figure 1E). Overall, these results show for the first time electrophysiological recordings for PHT1-mediated transport, and although the PHT1 signal-to-background ratio was not optimal, there is a statistically significant and consistent difference between PHT1-induced and background signals supporting the suitability of the described SSME method to study the functional activity of PHT1.
3.2. Functional Characterization of PHT1 (SLC15A4) by SSME
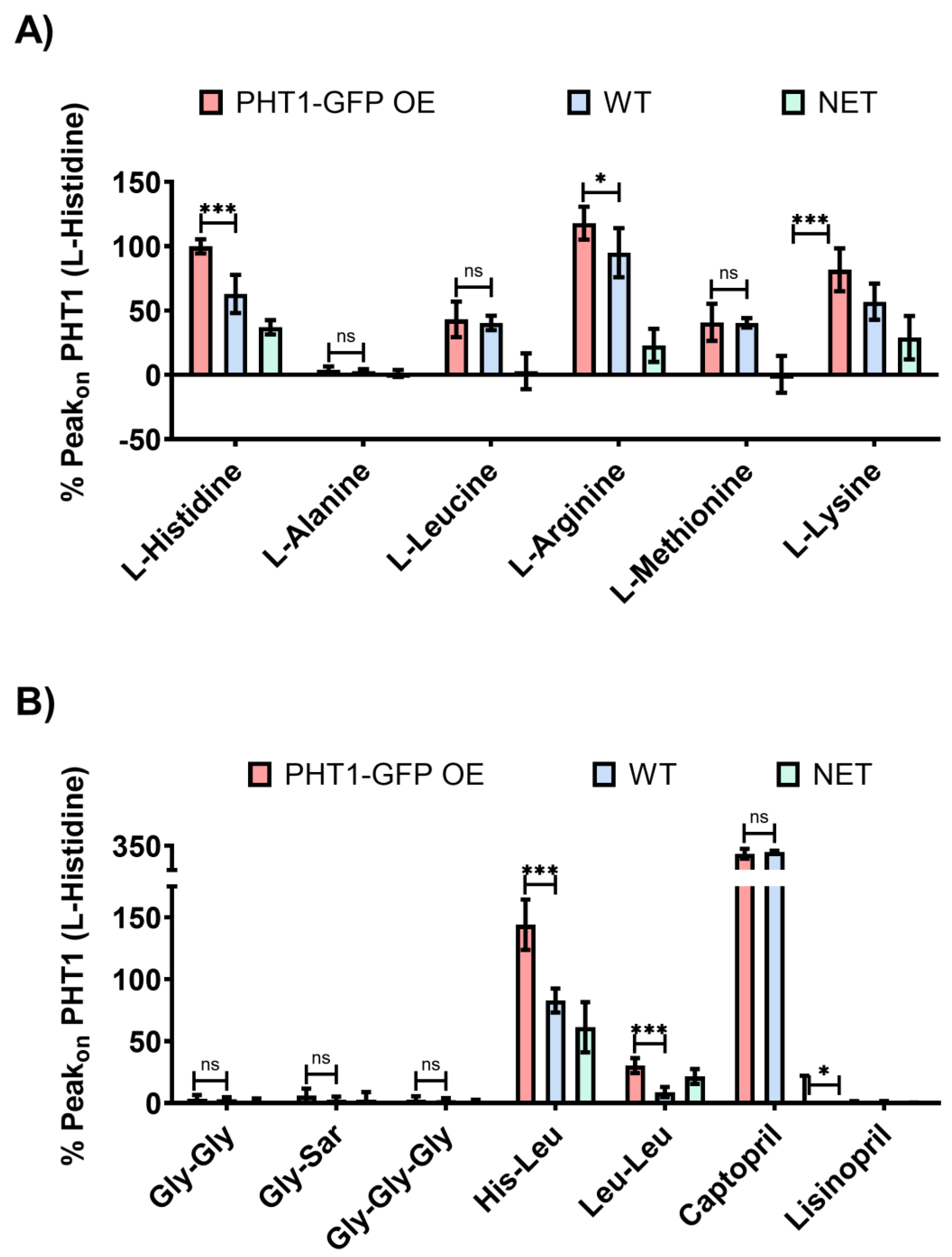
To assess the substrate selectivity of PHT1, sensors loaded with PHT1-GFP OE or WT lysosomal membrane preparations were perfused with a series of AAs (L-histidine, L-alanine, L-leucine, L-arginine, L-methionine and L-lysine), di- and tripeptides (Gly-Gly, Gly-Sar, Gly-Gly-Gly, His-leu and Leu-Leu) and peptidomimetic drugs (captopril and lisinopril), some of which have been previously proposed as PHT1 substrates [
3,
7,
8], and Peak
on currents were recorded with the SURFER
2R N1 device. Interestingly, when comparing the PHT1-GFP OE and WT Peak
on currents induced by AAs (
Figure 2A), only the positively charged AAs (L-histidine, L-arginine, L-lysine) induced significant differences, while the uncharged AAs did not induce any current (L-alanine) or were the same for both lysosomal membrane preparations (L-leucine and L-methionine). The presence of large non-specific currents (WT Peak
on) for some of these AAs suggests that there was an endogenous AAs transport system in the isolated lysosomal membrane preparations contributing to the recorded currents.
Regarding to the Peak
on currents induced by perfusion of di- and tripeptides (
Figure 2B), glycine-containing peptides (Gly-Gly, Gly-Sar and Gly-Gly-Gly) did not elicit a significant response, whereas leucine-containing peptides (His-Leu and Leu-Leu) were electrogenic and induced currents with significant differences between both PHT1-GFP OE and WT experimental groups. Interestingly, the di-peptide Leu-Leu, despite being an uncharged substrate, was able to induce specific currents, a consequence of H
+-coupled transport. Given that the Leu-Leu induced currents were exclusively due to H
+ influx, this could explain why the Leu-Leu induced currents were lower than those observed for the charged substrates (L-histidine, L-arginine, L-lysine and His-Leu), and furthermore provided the first direct evidence for H
+ cotransport by PHT1. In contrast to the results obtained with the AAs, the di- and tripeptide-induced currents had much lower background signal levels. This probably reflects the lack of an endogenous transport system for these substrates in the isolated lysosomal membrane preparations. Surprisingly, peptidomimetic drugs (captopril and lisinopril) did not induce a clear specific activity (
Figure 2B). Lisinopril induced very small currents that were statistically different between PHT1-GFP OE and WT; however, due to the reduced PHT1 signal-to-background ratio, it was not possible to investigate further its transport properties. Conversely, captopril induced very large and unspecific currents.
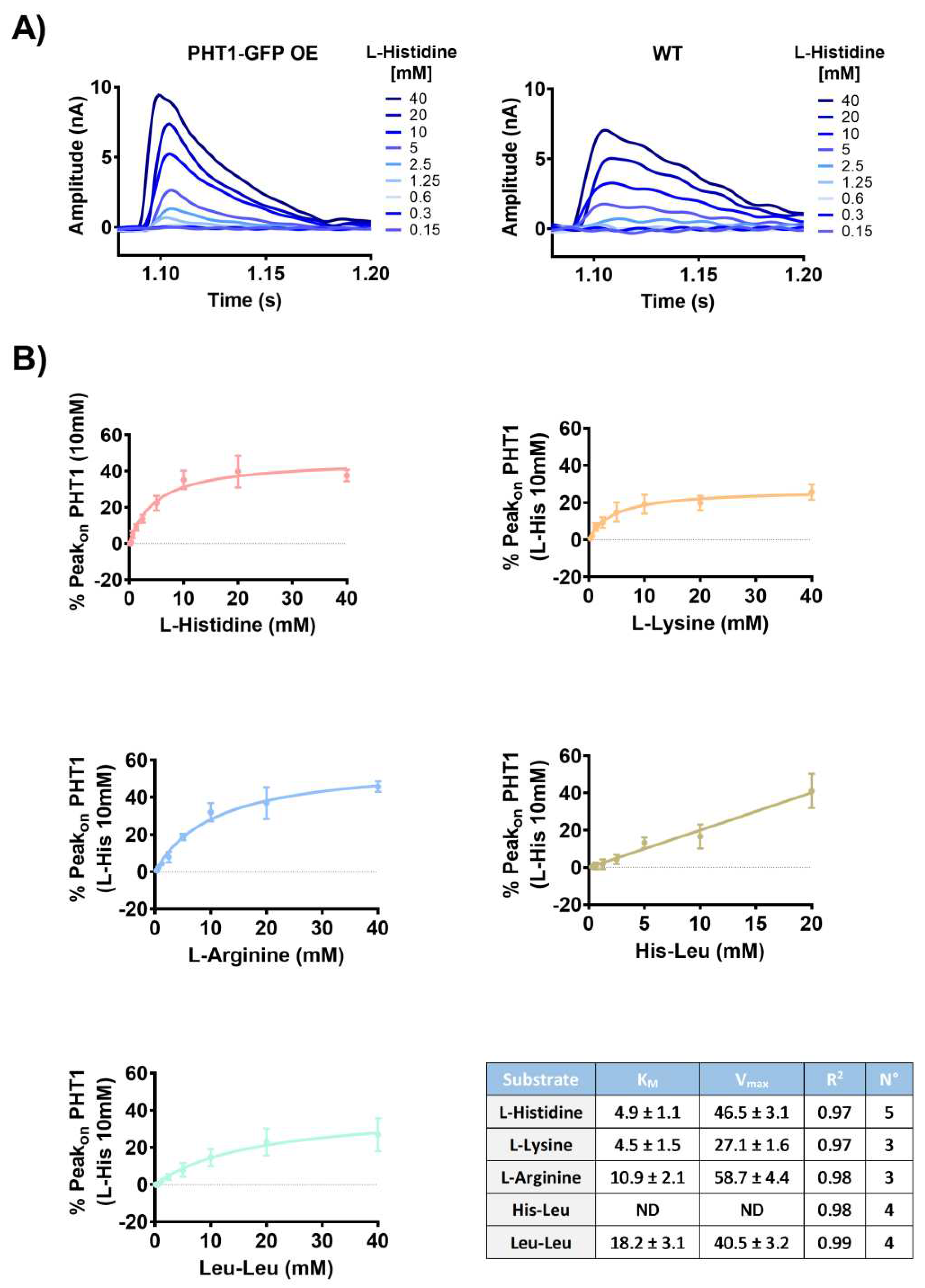
For kinetic studies, AAs and dipeptides that showed significantly different Peak
on currents between PHT1-GFP OE and WT lysosomal membrane preparations (
Figure 2A and B) were selected. Peak
on currents were induced by concentration jumps of a serial dilution of the indicated substrates (0.15-40 mM) and recorded on the same gold sensors loaded with PHT1-GFP OE or WT lysosomal membrane preparations (
Figure 3A). To eliminate the contribution of non-specific background currents (Peak
on WT) to the PHT1-GFP OE Peak
on recordings, Net Peak
on currents were calculated for each of the substrates (L-histidine, L-arginine, L-lysine, His-Leu and Leu-leu) and fitted to the Michalis-Menten equation (
Figure 3B), and the obtained kinetic parameters are shown (
Figure 3C). It is noteworthy that the calculated K
M value for L-histidine, 4.9±1.1mM, is close to the E
C50 value determined by MST (
Figure 1A), which is 3.8±0.1mM. Regarding the other substrates, K
M values for L-lysine and L-arginine were also in the low millimolar range, whereas the K
M for Leu-Leu was 3-4 times higher. The K
M for His-Leu could not be determined because the calculated Net Peak
on currents did not saturate in the range of concentrations tested. Higher concentrations of His-Leu could not be recorded because the magnitude of the currents was outside the sensitivity range of the gain settings used in these experiments.
The electrophysiological properties observed during SSME experiments are consistent with the H
+-coupled symport mechanism previously proposed for PHT1 [
7,
8,
10,
11]. To better understand the impact of H
+ electrochemical gradients on the functional activity of PHT1, sensors loaded with PHT1-GFP OE or WT lysosomal membrane preparations were used to record Peak
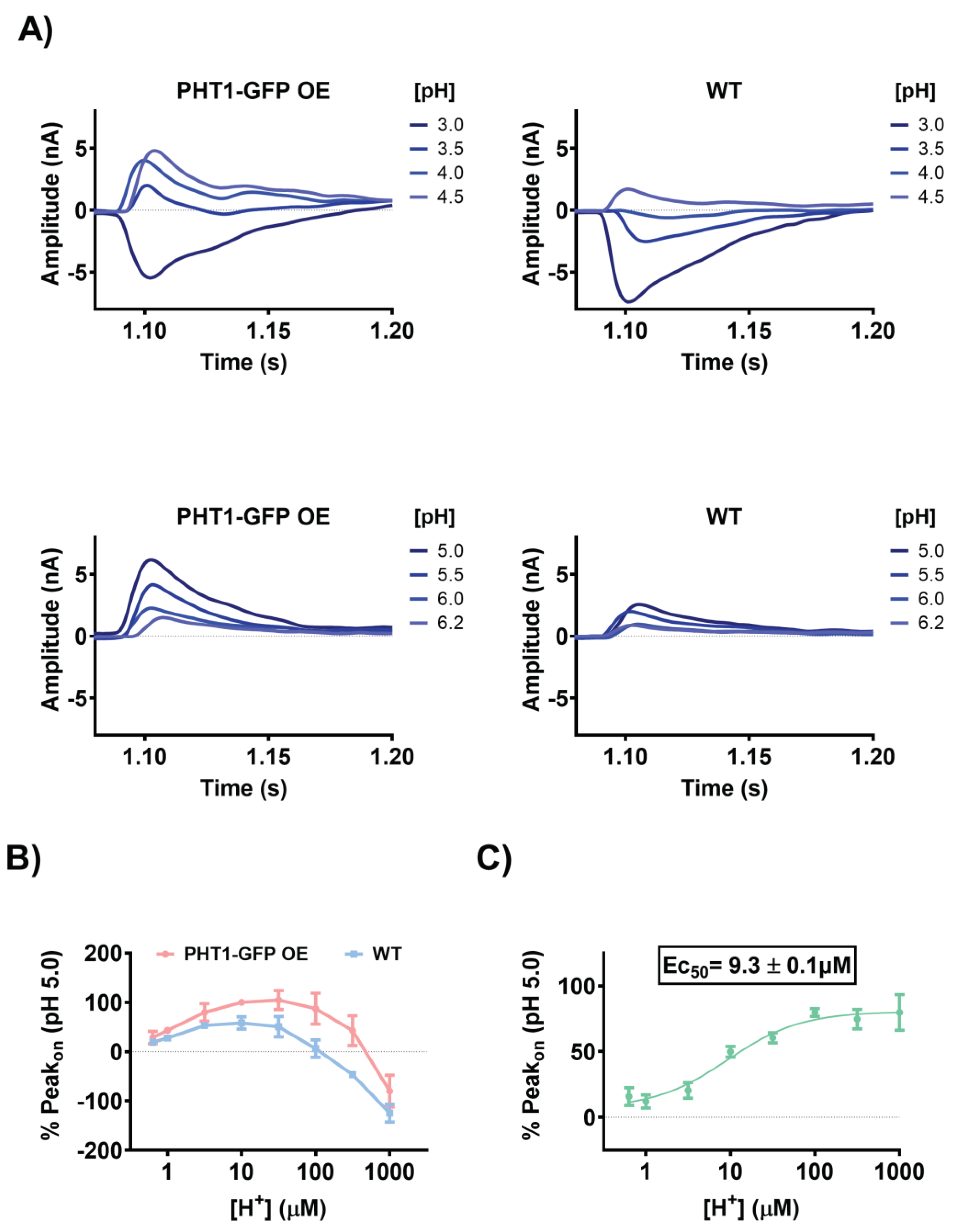
on currents upon perfusion of L-histidine in the presence of different extracellular H
+ concentrations (
Figure 4A). The direction and magnitude of the currents were strongly pH dependent for both experimental groups. Maximum current magnitudes for the PHT1-GFP OE sensors were reached in the pH range of 4.5 to 5.0, while for the WT sensors they were reached in the pH range of 4.5 to 5.5. Overall, the maximum current magnitudes were 40 to 50% higher with the PHT1-GFP OE sensors, in agreement with the previous experiments. Interestingly, at more acidic and alkaline extracellular pH conditions, the current magnitudes decreased in both experimental groups, showing a bell-shaped pH-dependence profile (
Figure 4B). Surprisingly, under highly acidic extracellular conditions, the direction of the Peak
on current direction was reversed. The reverse Peak
on currents were larger and were observed for a wider range of acidic pH conditions for WT sensors (i.e., 3.0 to 3.5). These reverse currents are likely a consequence of non-specific currents mediated by transport systems present in the lysosomal membrane preparations that mask PHT1-GFP related currents. To avoid the bias introduced by the non-specific currents (Peak
on WT), net Peak
on currents were calculated and used to determine the concentration of H
+ required to achieve half maximal activity (Ec
50) of PHT1-GFP (
Figure 4C). The calculated Ec
50, 9.3±0.13µM, corresponds to a pH 5.03.
3.3. Molecular Docking - PHT1 (SLC15A4) cryo-EM and AlphaFold 3D Structures
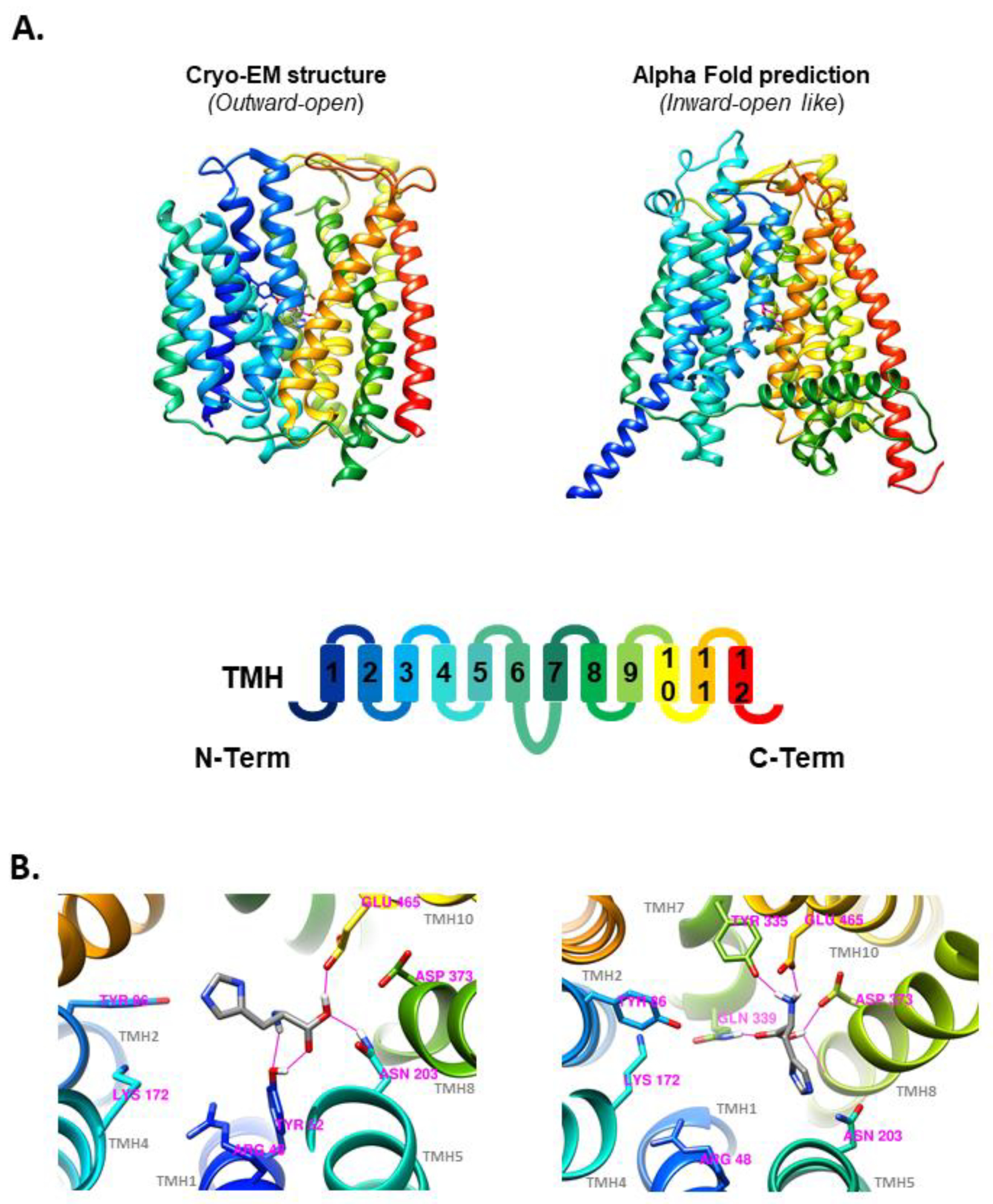
To confront the substrate selectivity results (
Figure 2) with an orthogonal method, the potential PHT1 substrates studied by SSME were docked into the human PHT1 3D structure obtained by cryo-EM (
Figure 5A; left) or predicted by AlphaFold (
Figure 5A; right). Structure energy minimization after molecular docking revealed the formation of several H-bonds between L-histidine and various AA side chains present in the docking box defined within each of the PHT1 structures. In the case of the outward-open cryo-EM structure, L-histidine formed H-bonds with residues Y52, N203 and E465 (
Figure 5B; left). In contrast, L-histidine docking with the inward-open like structure predicted by Alpha-Fold revealed the formation of H-bonds with residues Y335, N339, E465 and D373 (
Figure 5B; right). Members of the SLC15 family have several highly conserved residues proposed to be involved in peptide binding. In the case of human PHT1, R48, Y86, K172, N203, D373 and E465, are potentially required for interactions with both C- and N-termini of their substrate peptides [
2]. While residues W332, Y335, F492 and S496 could be relevant for interactions with the substrate peptide side chains [
2]. Interestingly, the molecular docking experiments showed direct interaction of L-histidine with several of these residues, supporting the reliability of the best-scoring docking conformations presented here (
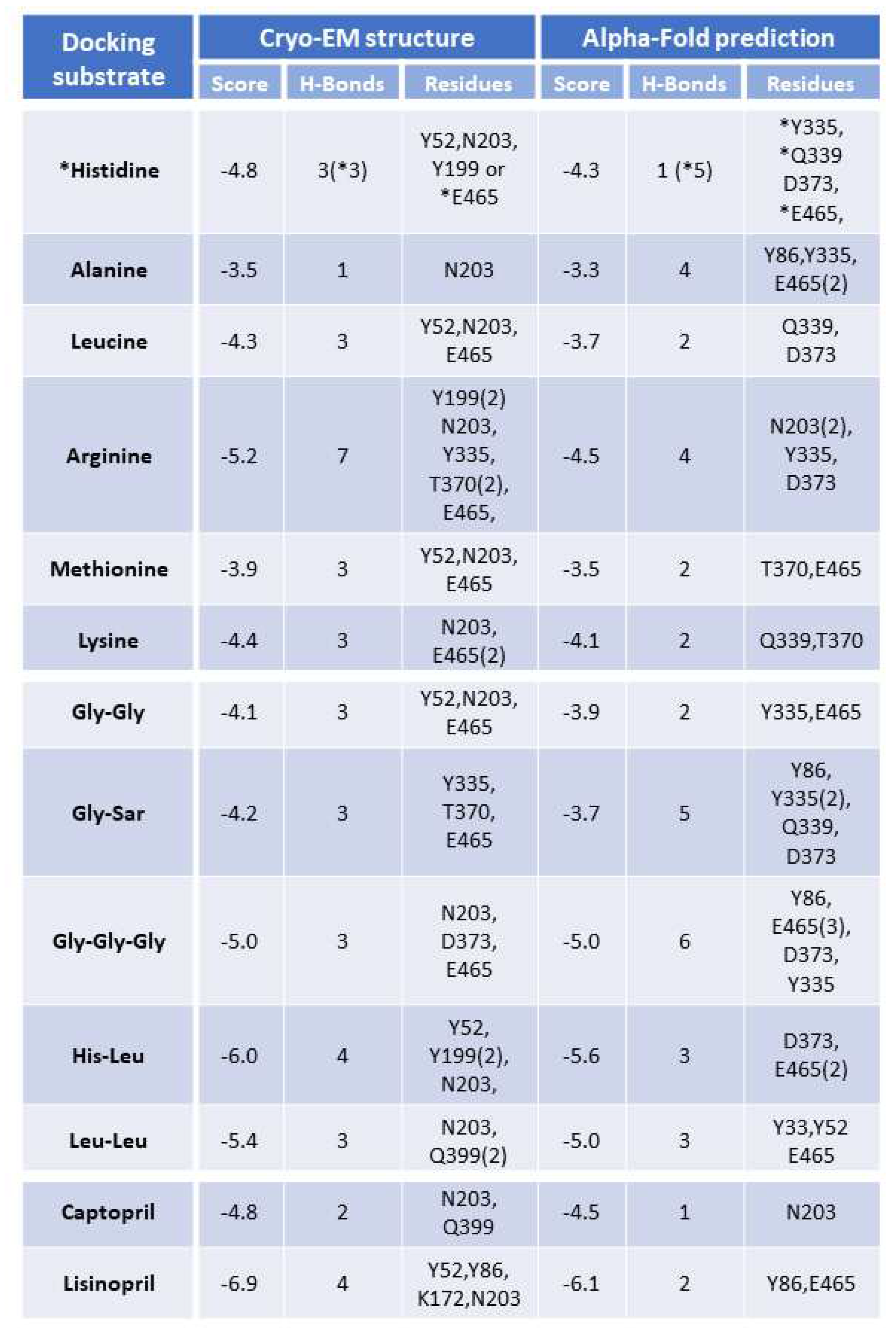
Figure 5B). Subsequently, molecular docking experiments were conducted under the same conditions with the other potential PHT1 substrates previously evaluated by SSME. The best docking scores, the number of H-Bonds and the residues involved in H-Bond formation are summarized in
Table 1. Among the single AAs, L-histidine, L-arginine and L-lysine showed the best docking scores. For di- and tripeptides, the best scores were obtained for His-Leu and Leu-Leu. For peptidomimetic drugs, lisinopril had the best score. Overall, the best scoring substrates within each substrate type, lisinopril, His-Leu, Leu-Leu, L-arginine, L-histidine and L-lysine were the substrates identified in the SSME experiments.
Table 1.
Table summarizing the docking results, including the best score, number of H-bonds and residues taking part in H-bonding in the best-scoring conformation.
Table 1.
Table summarizing the docking results, including the best score, number of H-bonds and residues taking part in H-bonding in the best-scoring conformation.
4. Discussion
Previous studies have attempted to characterize the functional properties of PHT1 (
SLC15A4) but the results are inconsistent [
2,
3,
7,
8,
10,
11]. One of the reasons for this variability is the intracellular location of PHT1, which has hindered the interpretation of the results obtained with standard cell-based transport assays, designed for transporters expressed in the plasma membranes [
10,
11]. To overcome this problem, several strategies have been employed, including the generation of genetic variants of PHT1with the goal of targeting protein expression to the plasma membrane [
8], the use of
Xenopus laevis oocytes as expression system [
7], or the direct use of the purified protein [
2]. However, the functional characterization of PHT1 was still inconsistent among the different studies, making it difficult to decide which one is the most reliable. In the present work, we introduce a new transport assay, which allowed to study the functional activity of PHT1 directly from isolated lysosomal membranes (
Figure 1B). This is a major advantage because it avoids harsh protein purification protocols that could compromise protein integrity, as well as the use of non-physiologically relevant overexpression systems. In addition, it provides the correct membrane lipid composition and possible protein expression partners, factors that likely influence the functional behavior of PHT1.
Regarding the methods used to study the functional activity of PHT1, previous studies used radiolabeled substrate transport assays as a readout of PHT1 functional activity [
3,
7,
8,
10,
11]. This classic transport assay is highly sensitive and very well-established; however, it requires specific radiolabeled substrates and the expression of the target protein on the plasma membrane. In contrast, a recent study assessed the binding of a of potential substrates to PHT1 by a thermal shift assay (TSA) using purified chicken PHT1. TSA experiments do not necessarily require expression of the target on the plasma membrane but do require validation by a direct transport assay of the observed target-ligand interactions. Moreover, while TSA experiments showed interactions with lysine- and arginine-containing peptides and even TASL, they failed to show with free AAs, including the major PHT1 substrate L-histidine, nor with histidine-containing peptides, nor with other PHT1 substrates such as carnosine or MDP [
2]. In contrast to all these previous works, in the present study, the functional activity of PHT1 is measured in its native lysosomal environment by SSME. This method allowed for the first time to measure the electrogenic properties of PHT1, which are related to the H
+-driven cotransport mechanism characteristic of SLC15 family members [
1]. Using SSME, the substrate selectivity among a series of AAs, di- and tripeptides and peptidomimetic drugs, the kinetic properties of the identified substrates and the pH-dependence were determined. As expected, our results present certain discrepancies with previous studies, which are presented and discussed below.
4.1. Substrate Selectivity
SSME recordings revealed a binding affinity for L-histidine in the low millimolar range, K
M=4.9±1.1mM (
Figure 3A-C), which was close the affinity determined by MST, K
D= 4.1±0.5mM (
Figure 1A). In contrast, previous work using the
Xenopus laevis oocytes microinjected with WT rat PHT1 [
7] or MDCK cells overexpressing a mutant variant of human PHT1, which shifted expression from the subcellular to the cell membrane [
8], showed affinities for L-histidine in the low micromolar range, K
M ≈ 16-17µM. The reason for this difference is unclear, however, in the latter case, both studies implied unnatural overexpression at the plasma membrane, where this transporter may behave differently compared to the endosomal/lysosomal environment. Surprisingly, studies using
Xenopus laevis oocytes did not assess the membrane expression of PHT1 or show the endogenous levels of L-histidine transport in non-injected oocytes. Similarly, studies using MDCK cells overexpressing a mutant variant of PHT1 did not account for the contribution of the non-specific signal to their K
M calculations. In any case, consistent with the results presented here, other lysosomal AA transporters, such as cystinosin (
SLC66A4), PAT1
(SLC36A1) and SNAT9 (
SLC38A9) have affinities for their respective substrate AAs, L-cysteine (K
M= 1.5mM) [
31], L-proline (K
M= 1.8mM) [
32] and L-arginine (K
M= 2.7mM) [
33] in the low millimolar range, supporting the results presented here using both SSME and MST.
Substrate selectivity experiments using SSME led to the identification of several novel PHT1 substrates such as L-arginine, L-lysine, His-Leu and Leu-Leu, and defined for the first time their transport kinetics (
Figure 3B and C). Among these substrates, the His-Leu and Leu-Leu dipeptides have previously been proposed as PHT1 substrates by competition experiments with radiolabeled L-histidine [
7] and direct binding by thermal shift assay [
2], respectively. L-arginine and L-lysine have been identified for the first time as PHT1 substrates in the present study. Moreover, consistent with our findings, competition experiments with radiolabeled L-histidine also showed that L-leucine and L-methionine are not PHT1 substrates [
7]. In contrast, substrates such as Gly-Sar, Gly-Gly, Gly-Gly-Gly and lisinopril, previously reported to be PHT1 substrates [
7,
8], did not show relevant electrogenic activity in the SSME recordings.
Notably, for some of the tested substrates, especially the single AAs and captopril, large unspecific currents were recorded. In this regard, several solute carriers (SLCs) have been reported to be involved in the transport of AAs across lysosomal membranes, some of which could potentially contribute to these non-specific currents. Possible candidates include, SLC38A9 [
34,
35], LYAAT1 (
SLC36A1) [
34,
36] PQLC2/LAAT1 (
SLC66A1) [
34,
37] and SNAT7 (
SLC38A7) [
34,
38], and less likely, as it is mainly expressed in immune cells, PHT2 (
SLC15A3) [
39]. Regarding captopril-induced non-specific currents, it has been shown that certain drug efflux transporters, such as the ABC transporter TAPL (
ABCB9), may be involved in peptide transport across lysosomal membranes [
40,
41]. However, it is unknown whether TAPL-mediated peptide transport is electrogenic and whether captopril is a substrate of TAPL or whether other lysosomal drug efflux transporters are involved. Thus, the origin of such non-specific currents is not clear.
4.2. pH-Dependence
In agreement with previous studies [
7,
8,
10], PHT1-mediated transport was highly dependent on the driving force provided by H
+ electrochemical gradients (
Figure 4). Furthermore, pH-dependency studies by SSME revealed that half-maximal PHT1 activity is reached at pH~5.03, which is close to the optimal lysosomal pH for hydrolase activity (pH≈4.6) [
42]. Its worth to be mentioned that the pK
a of L-histidine is ~ 6, thus, the difference in one pH unit between L-histidine pK
a and the calculated E
C50 for PHT1 supports the specificity of the recorded currents. Conversely, in the case of the WT sensors, the observed pH dependence could be only a reflection of the protonation state of L-histidine interacting with the membrane surface or the activity of other H
+-transport systems present in the lysosomal membranes.
The pH-dependence of PHT1 determined by SSME exhibited a bell-shaped profile, similar to the profiles observed for other H
+-coupled symporters, such as the bacterial sugar transporters, lactose permease (LacY), fucose permease (FucP) and xylose permease (XylE) [
43]. However, it was surprising that under highly acidic extracellular conditions, the current direction was reversed for both PHT1-GFP OE and WT sensors. This suggests that under these pH conditions, charged elements such as H
+, L-histidine, reorientation of charged elements embedded in the membrane, or other unanticipated charged elements present in the buffer solution move across the lysosomal membranes resulting in opposite membrane polarization. The origin of these reverse currents remains unclear, the presence of PHT1-GFP in the lysosomal membrane reduced the magnitude of the reverse polarization effect. This was probably a consequence of PHT1-GFP-mediated transport of H
+ and L-histidine into the lysosomal membranes, which generates a current in the opposite direction to that recorded.
Many ionic channels and SLCs are expressed in lysosomal membranes [
42], some of which could be the source origin of the non-specific currents recorded. Possible candidates to contribute to the non-specific currents induced by H
+ electrochemical gradients induced currents include the lysosomal H
+ pump [
44] and the voltage-gated H
+ channel [
45].
Finally, it is worth mentioning that SSME recordings with the uncharged substrate Leu-Leu provided the first direct evidence of H+ transport through PHT1.
4.3. Molecular Docking
To contrast the substrate selectivity results obtained by SSME (
Figure 2), potential PHT1 substrates tested by SSME were docked into the cryo-EM and AlphaFold predicted human PHT1 structures. The cryo-EM structure was obtained in an outward-open conformation (
Figure 5A; left), while the structure predicted by AlphaFold (
Figure 5A; right) resembles an inward-open like conformation, as the binding site is accessible exclusively from the cytosolic side. Therefore, molecular docking provided insight into the differences between the atomic interactions of L-histidine and the amino acid sidechains involved in substrate binding in both outward-open (
Figure 5B; left) and inward-open (
Figure 5B; right) conformations.
Overall, the molecular docking experiments with both PHT1 structures showed direct interaction of L-histidine with several of the amino acid residues described as key for transport in PHT1 [
2], supporting the reliability of the best scoring docking conformations shown. However, the orientation, H-bond, and precise location of the L-histidine docking show differences between the two structural models. Interestingly, in both cases, there is a direct interaction of the L-histidine with E465, a residue that has been shown to be essential for the binding of TASL to PHT1 [
17]. Moreover, in the inward-open-like conformation of the AlphaFold structure, the proximity of TMH7 to the binding site leads to the formation of additional H-Bonds, which may partially explain the observed conformational differences. In this regard, the reorientation of the substrate towards the exit pathway, observed in the inward-open-like docked conformation, could be a necessary step for substrate release.
Interestingly, the scores obtained during the molecular docking of the other potential PHT1 substrates support the substrate selectivity determined by SSME. However, the docking scores did not correlate with the calculated KM or substrate preferences determined by SSME. This could be consequence of the size of the substrates, as a larger molecule can potentially establish more interactions, and thus receive higher scores, as most scoring functions are additive [
46].
4.4. PHT1 SSME Assay Limitations
Although several important aspects of the functional properties of PHT1 were revealed by SSME, the significant contribution of various non-specific signal sources to the recorded PHT1-GFP-OE currents remained the main limitation during this study. To overcome this problem, Net Peakon currents were calculated prior to kinetic analysis. However, for some substrates, such as lisinopril, the net Peakon currents calculated for PHT1-GFP-OE were not sufficient to perform more detailed analyses. It is noteworthy that background signal levels were variable depending on the type of substrate and its charge. In general, charged substrates, such as the positively charged AAs, induced larger no-specific currents, suggesting a direct interaction of their charges with the membrane surface as main source of background signal. This seemed to be reflected in the higher degree of symmetry between Peakon and Peakoff currents observed for the charged substrates in the WT sensors. Interestingly, non-charged AAs, di- and tripeptides and peptidomimetic drugs showed different levels of non-specific signal. Given that different substrates were often tested on the same sensor and always at the same concentration, this is a clear indication of the presence of endogenous transport systems present in the isolated lysosomal membranes that contribute to the non-specific signal according to their own substrate preferences. In this context, some examples of lysosomal AA transporters, drug efflux transporters and H+ channels that might contribute to such signals have already been discussed.
Overall, it seems more appropriate to perform the described SSME experiments in the native lysosomal environment of PHT1. However, this has the disadvantage of introducing a background signal due to the activity of other SLCs and ion channels that mask the actual transport activity of PHT1. In this regard, future studies following the SSME approach described here would benefit from improving PHT1 signal-to-background ratio. This would require increasing the density of PHT1 in the lysosomal membranes on the gold sensors. This could probably be achieved by improving lysosomal membrane isolation protocols or PHT1 overexpression strategies. Alternatively, purified PHT1 reconstituted into proteoliposomes could be used. However, protein purification procedures are generally complex and may compromise the structural integrity of membrane proteins. Furthermore, they may also remove important accessory elements of PHT1 such as lipids or interacting proteins.
5. Conclusions
This work presents a new functional assay based on the SSME methodology, which allows the study of human PHT1 in its native lysosomal environment. Using this new approach, a characterization of several functional properties of PHT1 was performed. Major findings include the first recordings of electrophysiological properties and direct evidence for H+-cotransport by PHT1, a detailed pH-dependency analysis, an assessment of PHT1 substrate selectivity among a variety of AAs and peptides, as well as transport kinetics for L-histidine and the PHT1 substrates L-arginine, L-lysine, His-Leu and Leu-Leu identified herein. Moreover, some of these observations were validated by orthogonal methods such as MST or molecular docking.
Transport and scaffolding functions of PHT1 can influence the lysosomal environment [
4,
26], enable NOD and TLR signaling [
3,
13,
14], and modulate mTOR activity in human immune cells [
4,
15]. In addition, PHT1 plays a central role in SLE pathogenesis [
18,
19,
20,
21,
22]. Thus, major efforts are currently underway to identify molecules capable of modulating PHT1 function as strategy for the treatment of diseases associated with altered innate immune responses [
47,
48]. Despite the obvious interest of PHT1 as drug target, pharmacological development of PHT1 has been limited by the lack of appropriate functional assays and scarce structural information. Therefore, it is expected that the SSME-based assay introduced in the present work could serve as a screening platform to identify novel PHT1 modulators or to evaluate the pharmacological activity of newly found modulators [
47,
48]. Furthermore, this SSME assay can also be used to further characterize the functional properties of PHT1 or to study the impact of medically relevant SNPs [
21] on normal PHT1 function, all of which would significantly contribute to a better understanding of its physiological role and therapeutic value as drug target.