Submitted:
30 November 2023
Posted:
01 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Chemical composition, preparation, and acid-base properties of LDHs
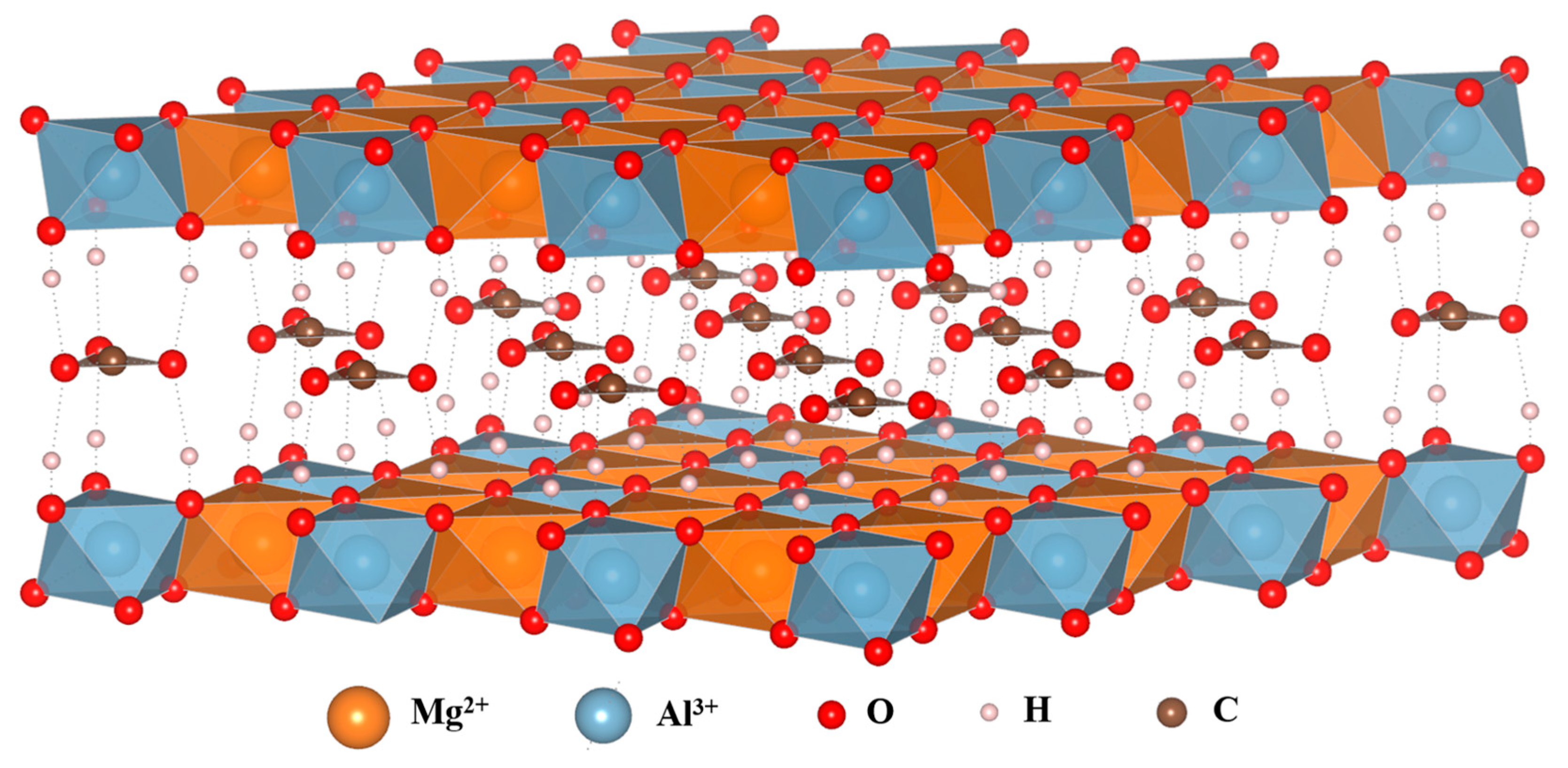
2.1. Chemical composition
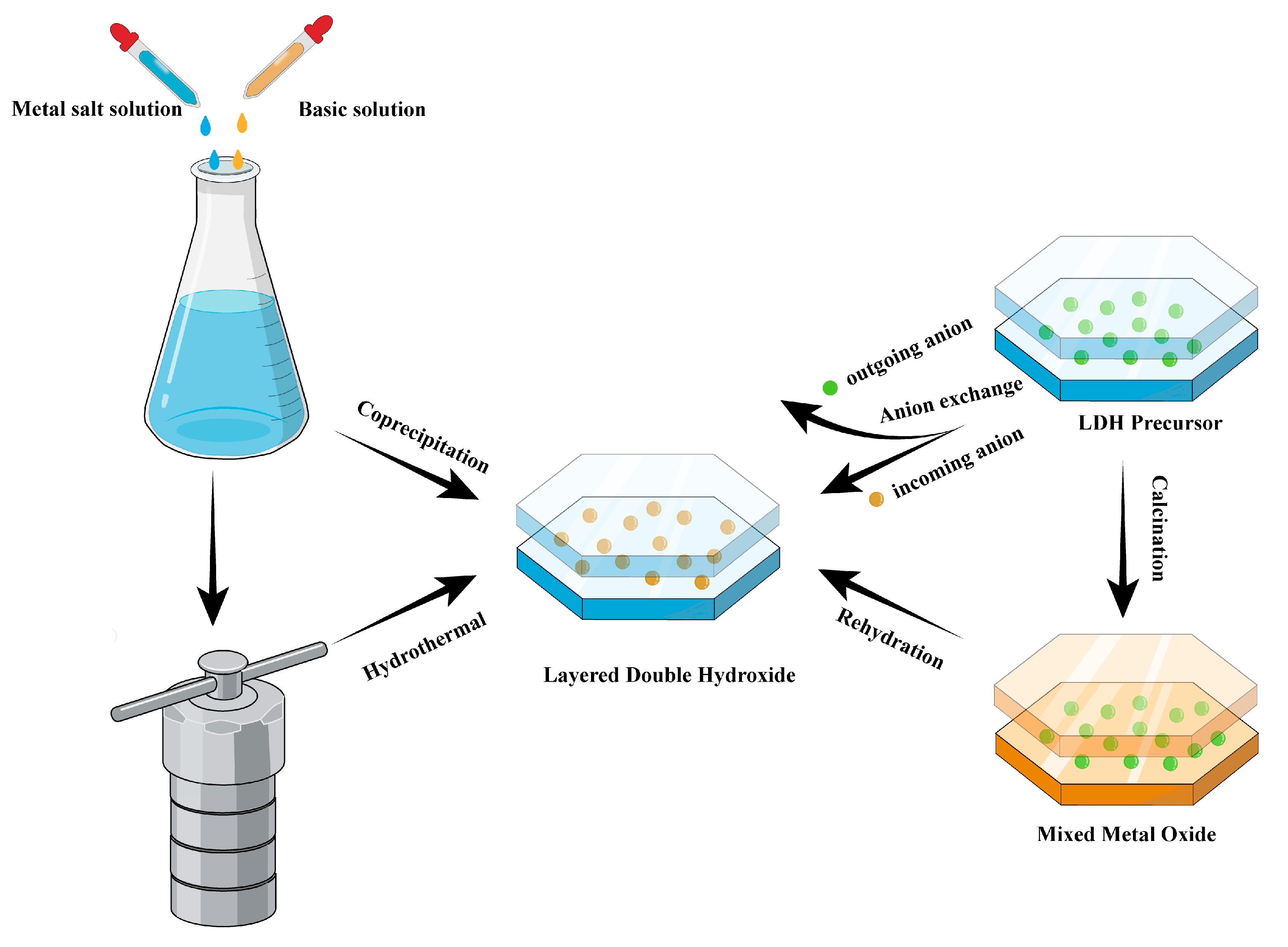
2.2. Preparation methods
2.3. Acid-base properties
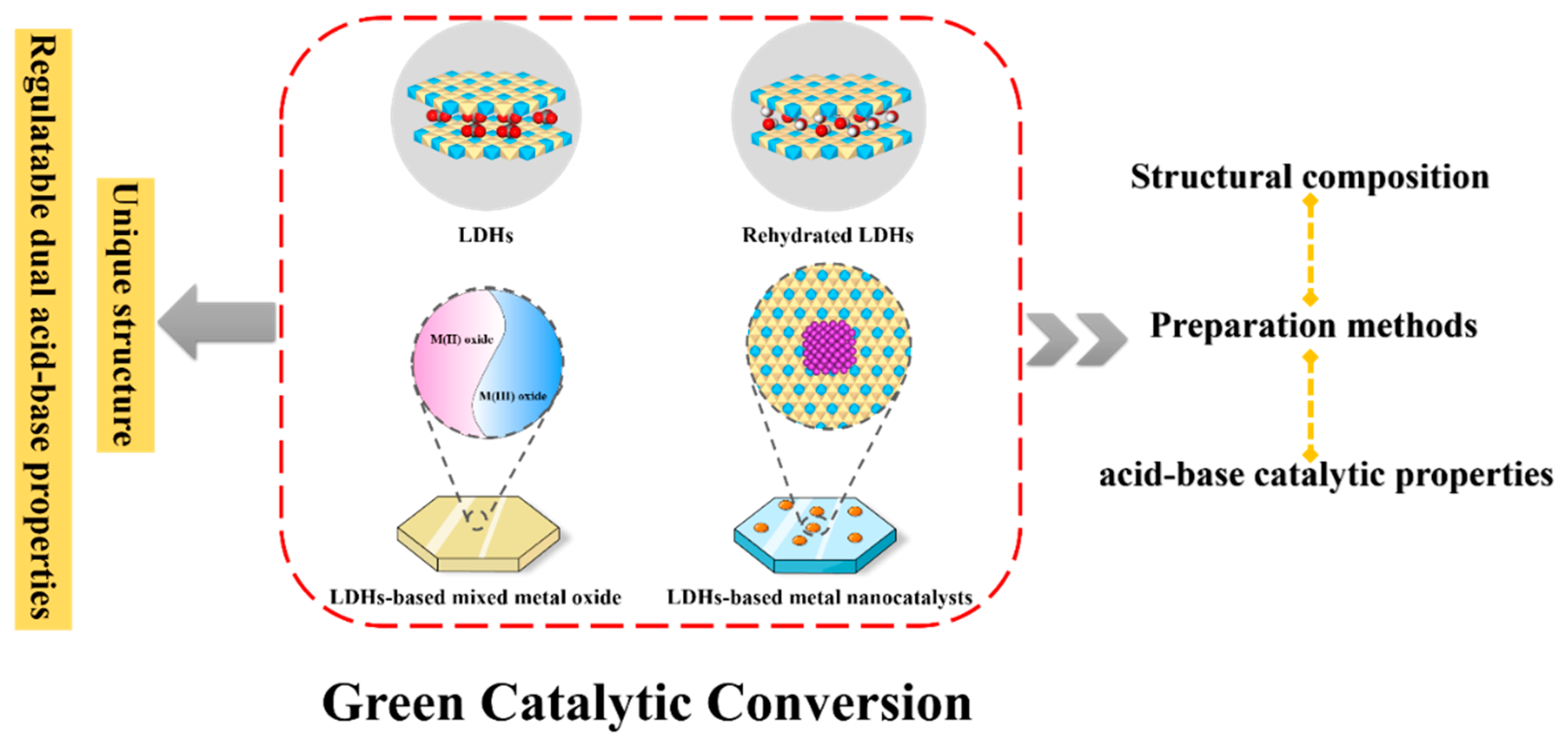
3. Application of LDH-based catalysts in the green acid-base catalytic transformation
3.1. LDH catalysts
3.1.1. Original LDH catalysts
3.1.2. Rehydrated LDH catalysts
3.2. LDHs-based metal nanocatalysts
3.3. LDHs-based mixed metal oxide catalysts
4. Conclusions and perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Vafaeezadeh, M.; Alinezhad, H. Brønsted acidic ionic liquids: Green catalysts for essential organic reactions. Journal of Molecular Liquids 2016, 218, 95–105. [Google Scholar] [CrossRef]
- Vafaeezadeh, M.; Fattahi, A. A study on the catalytic activity and theoretical modeling of a novel dual acidic mesoporous silica. RSC Advances 2014, 4. [Google Scholar] [CrossRef]
- Manríquez-Ramírez, M.; Gómez, R.; Hernández-Cortez, J.G.; Zúñiga-Moreno, A.; Reza-San Germán, C.M.; Flores-Valle, S.O. Advances in the transesterification of triglycerides to biodiesel using MgO–NaOH, MgO–KOH and MgO–CeO2 as solid basic catalysts. Catalysis Today 2013, 212, 23–30. [Google Scholar] [CrossRef]
- Suwannakarn, K.; Lotero, E.; Goodwin, J.G. Solid bronsted acid catalysis in the gas-phase esterification of acetic acid. INDUSTRIAL & ENGINEERING CHEMISTRY RESEARCH 2007, 46, 7050–7056. [Google Scholar] [CrossRef]
- Wen, C.; Yin, A.; Dai, W.-L. Recent advances in silver-based heterogeneous catalysts for green chemistry processes. Applied Catalysis B: Environmental 2014, 160-161, 730–741. [Google Scholar] [CrossRef]
- Patial, S.; Raizada, P.; Aslam Parwaz Khan, A.; Singh, A.; Van Le, Q.; Huy Nguyen, V.; Selvasembian, R.; Mustansar Hussain, C.; Singh, P. Emerging new-generation covalent organic frameworks composites as green catalysts: design, synthesis and solar to fuel production. Chemical Engineering Journal 2022, 433. [Google Scholar] [CrossRef]
- Liu, K.-G.; Sharifzadeh, Z.; Rouhani, F.; Ghorbanloo, M.; Morsali, A. Metal-organic framework composites as green/sustainable catalysts. Coordination Chemistry Reviews 2021, 436. [Google Scholar] [CrossRef]
- Yadav, G.; Yadav, N.; Ahmaruzzaman, M. Microwave-assisted synthesis of biodiesel by a green carbon-based heterogeneous catalyst derived from areca nut husk by one-pot hydrothermal carbonization. Sci Rep 2022, 12, 21455. [Google Scholar] [CrossRef]
- Suganuma, S.; Nakajima, K.; Kitano, M.; Yamaguchi, D.; Kato, H.; Hayashi, S.; Hara, M. Synthesis and acid catalysis of cellulose-derived carbon-based solid acid. Solid State Sciences 2010, 12, 1029–1034. [Google Scholar] [CrossRef]
- Elmekawy, A.A.; Shiju, N.R.; Rothenberg, G.; Brown, D.R. Environmentally Benign Bifunctional Solid Acid and Base Catalysts. Industrial & Engineering Chemistry Research 2014, 53, 18722–18728. [Google Scholar] [CrossRef]
- Yan, Q.; Hou, X.; Liu, G.; Li, Y.; Zhu, T.; Xin, Y.; Wang, Q. Recent advances in layered double hydroxides (LDHs) derived catalysts for selective catalytic reduction of NO(x) with NH(3). J Hazard Mater 2020, 400, 123260. [Google Scholar] [CrossRef]
- Hu, S.-L.; Liang, S.; Mo, L.-Z.; Su, H.-H.; Huang, J.-S.; Zhang, P.-J.; Qin, J.-N. Conversion of biomass-derived monosaccharide into furfural over Cr–Mg-LDO@bagasse catalysts. Sustainable Chemistry and Pharmacy 2023, 32. [Google Scholar] [CrossRef]
- Zhang, S.; Zou, K.; Li, B.; Shim, H.; Huang, Y. Key Considerations on the Industrial Application of Lignocellulosic Biomass Pyrolysis toward Carbon Neutrality. Engineering 2023. [Google Scholar] [CrossRef]
- Xiong, J.; Zhang, S.; Ke, L.; Wu, Q.; Zhang, Q.; Cui, X.; Dai, A.; Xu, C.; Cobb, K.; Liu, Y.; et al. Research progress on pyrolysis of nitrogen-containing biomass for fuels, materials, and chemicals production. Sci Total Environ 2023, 872, 162214. [Google Scholar] [CrossRef] [PubMed]
- Thakur, A.K.; Kumar, R.; Shukla, P.; Sharma, P.; Kulabhi, A. Biochar from agricultural biomass: Current status and future scope. Materials Today: Proceedings 2023. [Google Scholar] [CrossRef]
- Deng, W.; Feng, Y.; Fu, J.; Guo, H.; Guo, Y.; Han, B.; Jiang, Z.; Kong, L.; Li, C.; Liu, H.; et al. Catalytic conversion of lignocellulosic biomass into chemicals and fuels. Green Energy & Environment 2023, 8, 10–114. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, W.; Li, C.; Zhang, C. Recent Progress of Hydrogenation and Hydrogenolysis Catalysts Derived from Layered Double Hydroxides. Catalysts 2022, 12. [Google Scholar] [CrossRef]
- Zhu, J.; Li, T.; Wang, S.; Chen, Y.; Ge, F.; Xu, Y. Lattice-distortion active sites of Ni-doped CuMgFe LDH for benzotraizole degradation. Journal of Environmental Chemical Engineering 2022, 10. [Google Scholar] [CrossRef]
- Theiss, F.L.; Ayoko, G.A.; Frost, R.L. Synthesis of layered double hydroxides containing Mg2+, Zn2+, Ca2+ and Al3+ layer cations by co-precipitation methods—A review. Applied Surface Science 2016, 383, 200–213. [Google Scholar] [CrossRef]
- Mohapatra, L.; Parida, K. A review on the recent progress, challenges and perspective of layered double hydroxides as promising photocatalysts. Journal of Materials Chemistry A 2016, 4, 10744–10766. [Google Scholar] [CrossRef]
- Panda, H.S.; Srivastava, R.; Bahadur, D. Synthesis and <i>in situ</i> mechanism of nuclei growth of layered double hydroxides. BULLETIN OF MATERIALS SCIENCE 2011, 34, 1599–1604. [Google Scholar] [CrossRef]
- Seftel, E.M.; Popovici, E.; Mertens, M.; Witte, K.; Tendeloo, G.; Cool, P.; Vansant, E.F. Zn-Al layered double hydroxides: Synthesis, characterization and photocatalytic application. MICROPOROUS AND MESOPOROUS MATERIALS 2008, 113, 296–304. [Google Scholar] [CrossRef]
- Morel-Desrosiers, N.; Pisson, J.; Israëli, Y.; Taviot-Guého, C.; Besse, J.-P.; Morel, J.-P. Intercalation of dicarboxylate anions into a Zn–Al–Cl layered double hydroxide: microcalorimetric determination of the enthalpies of anion exchange. J. Mater. Chem. 2003, 13, 2582–2585. [Google Scholar] [CrossRef]
- Zou, W.; Guo, W.; Liu, X.; Luo, Y.; Ye, Q.; Xu, X.; Wang, F. Anion Exchange of Ni-Co Layered Double Hydroxide (LDH) Nanoarrays for a High-Capacitance Supercapacitor Electrode: A Comparison of Alkali Anion Exchange and Sulfuration. Chemistry 2018, 24, 19309–19316. [Google Scholar] [CrossRef]
- Das, J.; Parida, K.M. Heteropoly acid intercalated Zn/Al HTlc as efficient catalyst for esterification of acetic acid using n-butanol. Journal of Molecular Catalysis A: Chemical 2007, 264, 248–254. [Google Scholar] [CrossRef]
- Wei, Y.; Li, F.; Liu, L. Liquid exfoliation of Zn–Al layered double hydroxide using NaOH/urea aqueous solution at low temperature. RSC Advances 2014, 4. [Google Scholar] [CrossRef]
- Liu, H.; Yu, T.; Su, D.; Tang, Z.; Zhang, J.; Liu, Y.; Yuan, A.; Kong, Q. Ultrathin Ni-Al layered double hydroxide nanosheets with enhanced supercapacitor performance. Ceramics International 2017, 43, 14395–14400. [Google Scholar] [CrossRef]
- Jiang, M.; Zhang, X. Carbon fiber/Ni-Co layered double hydroxide@NiMoO4/graphene oxide sandwich structure flexible electrode materials: Facile synthesis and high supercapacitor performance. Journal of Alloys and Compounds 2019, 794, 13–20. [Google Scholar] [CrossRef]
- Gao, Z.; Sasaki, K.; Qiu, X. Structural Memory Effect of Mg-Al and Zn-Al layered Double Hydroxides in the Presence of Different Natural Humic Acids: Process and Mechanism. Langmuir 2018, 34, 5386–5395. [Google Scholar] [CrossRef]
- Dubnova, L.; Danhel, R.; Meinhardova, V.; Korolova, V.; Smolakova, L.; Kondratowicz, T.; Kikhtyanin, O.; Capek, L. Reconstruction of the ZnAl Mixed Oxides Into the Layered Double Hydroxide Catalysts Active in the Aldol Condensation of Furfural: The Role of ZnO Particles. Front Chem 2021, 9, 803764. [Google Scholar] [CrossRef]
- Timofeeva, M.N.; Kapustin, A.E.; Panchenko, V.N.; Butenko, E.O.; Krupskaya, V.V.; Gil, A.; Vicente, M.A. Synthetic and natural materials with the brucite-like layers as high active catalyst for synthesis of 1-methoxy-2-propanol from methanol and propylene oxide. Journal of Molecular Catalysis A: Chemical 2016, 423, 22–30. [Google Scholar] [CrossRef]
- Terzi, C.M.; dos Santos, E.H.; Carvalho, C.; Prevot, V.; Wypych, F.; Forano, C.; Nakagaki, S. MgAl and ZnAl layered double hydroxides modified with molybdate and tungstate anions as catalysts for oxidation of cyclohexane. Catalysis Today 2023, 422. [Google Scholar] [CrossRef]
- Wang, Z.; Lu, G.; Guo, Y.; Guo, Y.; Gong, X.-Q. Effect of One-Pot Rehydration Process on Surface Basicity and Catalytic Activity of MgyAl1-aREEaOx Catalyst for Aldol Condensation of Citral and Acetone. ACS Sustainable Chemistry & Engineering 2016, 4, 1591–1601. [Google Scholar] [CrossRef]
- Pithakratanayothin, S.; Tongsri, R.; Chaisuwan, T.; Wongkasemjit, S. Influences of M–Sn intermetallics (M = Ni, Cu) prepared by mechanical alloying on phenol hydroxylation. Catalysis Science & Technology 2017, 7, 5413–5421. [Google Scholar] [CrossRef]
- Huang, Y.-Y.; Konnerth, H.; Yeh, J.-Y.; Prechtl, M.H.G.; Wen, C.-Y.; Wu, K.C.W. De novo synthesis of Cr-embedded MOF-199 and derived porous CuO/CuCr2O4 composites for enhanced phenol hydroxylation. Green Chemistry 2019, 21, 1889–1894. [Google Scholar] [CrossRef]
- Jiang, Y.; Xu, W.; Liang, J.; Shen, J.; Fu, X.; He, H.; Yan, S.; Ren, X. Enhanced catalytic phenol hydroxylation by CuZnFeAl layered double hydroxides: synergistic effects of Cu+ and oxygen vacancies. New Journal of Chemistry 2021, 45, 179–188. [Google Scholar] [CrossRef]
- Liu, B.; Zheng, Z.; Liu, Y.; Zhang, M.; Wang, Y.; Wan, Y.; Yan, K. Efficient electrooxidation of biomass-derived aldehydes over ultrathin NiV-layered double hydroxides films. Journal of Energy Chemistry 2023, 78, 412–421. [Google Scholar] [CrossRef]
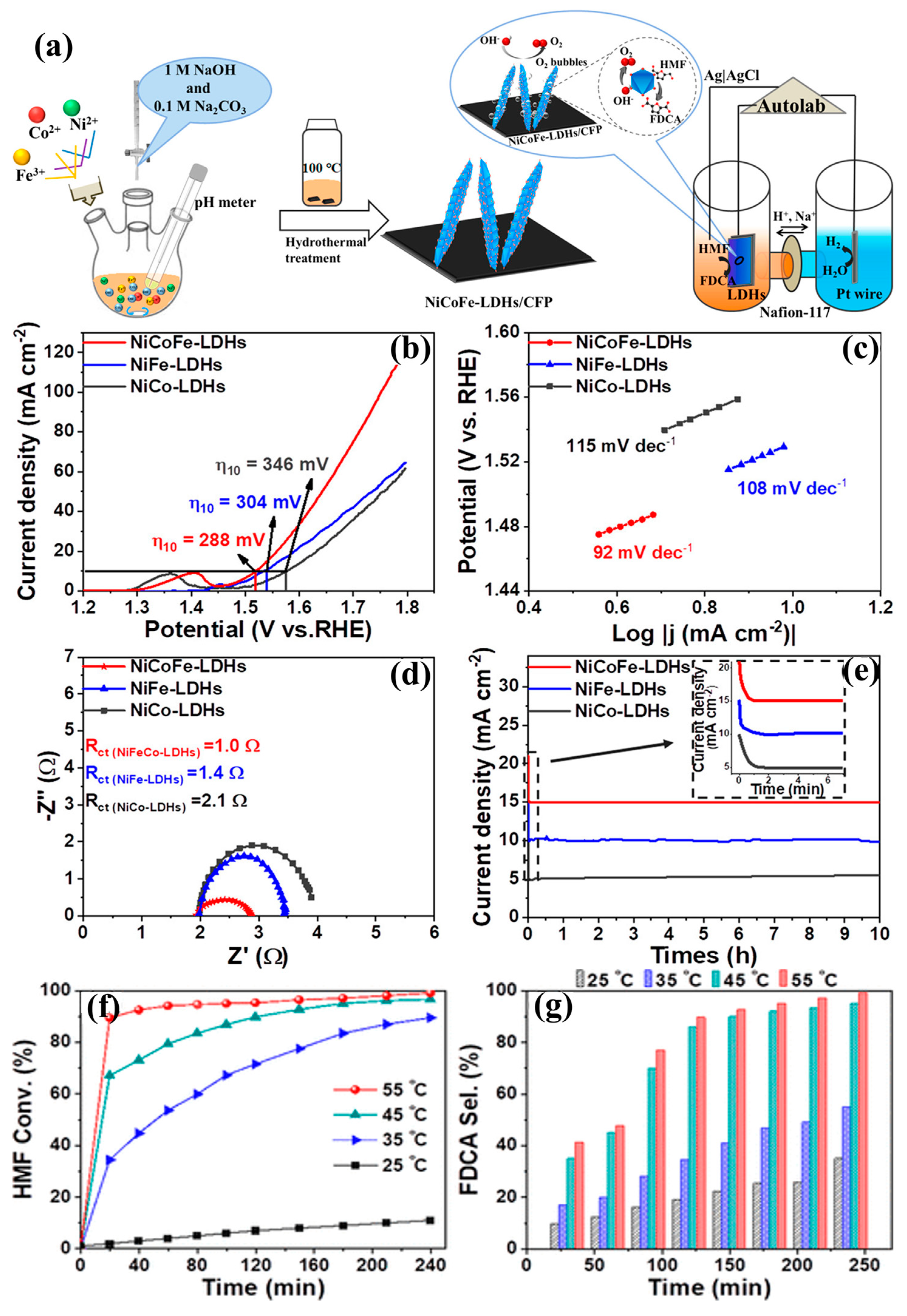
- Zhang, M.; Liu, Y.; Liu, B.; Chen, Z.; Xu, H.; Yan, K. Trimetallic NiCoFe-Layered Double Hydroxides Nanosheets Efficient for Oxygen Evolution and Highly Selective Oxidation of Biomass-Derived 5-Hydroxymethylfurfural. ACS Catalysis 2020, 10, 5179–5189. [Google Scholar] [CrossRef]
- Wang, T.; Hu, A.; Wang, H.; Xia, Y. Catalytic transfer hydrogenation of furfural into furfuryl alcohol over Ni–Fe-layered double hydroxide catalysts. Journal of the Chinese Chemical Society 2019, 66, 1610–1618. [Google Scholar] [CrossRef]
- Ye, X.; Shi, X.; Xu, H.; Feng, Y.; Jin, B.; Duan, P. Enhanced catalytic activity of layered double hydroxides via in-situ reconstruction for conversion of glucose/food waste to methyl lactate in biorefinery. Sci Total Environ 2022, 829, 154540. [Google Scholar] [CrossRef]
- Yu, I.K.M.; Hanif, A.; Tsang, D.C.W.; Shang, J.; Su, Z.; Song, H.; Ok, Y.S.; Poon, C.S. Tuneable functionalities in layered double hydroxide catalysts for thermochemical conversion of biomass-derived glucose to fructose. Chemical Engineering Journal 2020, 383. [Google Scholar] [CrossRef]
- Upare, P.P.; Chamas, A.; Lee, J.H.; Kim, J.C.; Kwak, S.K.; Hwang, Y.K.; Hwang, D.W. Highly Efficient Hydrotalcite/1-Butanol Catalytic System for the Production of the High-Yield Fructose Crystal from Glucose. ACS Catalysis 2019, 10, 1388–1396. [Google Scholar] [CrossRef]
- Delidovich, I.; Palkovits, R. Catalytic Isomerization of Biomass-Derived Aldoses: A Review. ChemSusChem 2016, 9, 547–561. [Google Scholar] [CrossRef]
- Liu, W.; Sun, J.; Zhang, X.; Wei, M. Supported Ag Catalysts on Mg–Al Oxides toward Oxidant-Free Dehydrogenation Reaction of Benzyl Alcohol. Industrial & Engineering Chemistry Research 2018, 57, 15606–15612. [Google Scholar] [CrossRef]
- Valente, J.S.; Pfeiffer, H.; Lima, E.; Prince, J.; Flores, J. Cyanoethylation of alcohols by activated Mg–Al layered double hydroxides: Influence of rehydration conditions and Mg/Al molar ratio on Brönsted basicity. Journal of Catalysis 2011, 279, 196–204. [Google Scholar] [CrossRef]
- Kikhtyanin, O.; Tišler, Z.; Velvarská, R.; Kubička, D. Reconstructed Mg-Al hydrotalcites prepared by using different rehydration and drying time: Physico-chemical properties and catalytic performance in aldol condensation. Applied Catalysis A: General 2017, 536, 85–96. [Google Scholar] [CrossRef]
- Liang, G.; Wang, A.; Zhao, X.; Lei, N.; Zhang, T. Selective aldol condensation of biomass-derived levulinic acid and furfural in aqueous-phase over MgO and ZnO. Green Chemistry 2016, 18, 3430–3438. [Google Scholar] [CrossRef]
- Lee, R.; Vanderveen, J.R.; Champagne, P.; Jessop, P.G. CO2-Catalysed aldol condensation of 5-hydroxymethylfurfural and acetone to a jet fuel precursor. Green Chemistry 2016, 18, 5118–5121. [Google Scholar] [CrossRef]
- Bing, W.; Zheng, L.; He, S.; Rao, D.; Xu, M.; Zheng, L.; Wang, B.; Wang, Y.; Wei, M. Insights on Active Sites of CaAl-Hydrotalcite as a High-Performance Solid Base Catalyst toward Aldol Condensation. ACS Catalysis 2017, 8, 656–664. [Google Scholar] [CrossRef]
- Bing, W.; Wang, H.; Zheng, L.; Rao, D.; Yang, Y.; Zheng, L.; Wang, B.; Wang, Y.; Wei, M. A CaMnAl-hydrotalcite solid basic catalyst toward the aldol condensation reaction with a comparable level to liquid alkali catalysts. Green Chemistry 2018, 20, 3071–3080. [Google Scholar] [CrossRef]
- Kwon, D.; Kang, J.Y.; An, S.; Yang, I.; Jung, J.C. Tuning the base properties of Mg–Al hydrotalcite catalysts using their memory effect. Journal of Energy Chemistry 2020, 46, 229–236. [Google Scholar] [CrossRef]
- Yabushita, M.; Shibayama, N.; Nakajima, K.; Fukuoka, A. Selective Glucose-to-Fructose Isomerization in Ethanol Catalyzed by Hydrotalcites. ACS Catalysis 2019, 9, 2101–2109. [Google Scholar] [CrossRef]
- Zhan, Y.; Song, K.; Shi, Z.; Wan, C.; Pan, J.; Li, D.; Au, C.; Jiang, L. Influence of reduction temperature on Ni particle size and catalytic performance of Ni/Mg(Al)O catalyst for CO2 reforming of CH4. International Journal of Hydrogen Energy 2020, 45, 2794–2807. [Google Scholar] [CrossRef]
- Cui, G.; Meng, X.; Zhang, X.; Wang, W.; Xu, S.; Ye, Y.; Tang, K.; Wang, W.; Zhu, J.; Wei, M.; et al. Low-temperature hydrogenation of dimethyl oxalate to ethylene glycol via ternary synergistic catalysis of Cu and acid−base sites. Applied Catalysis B: Environmental 2019, 248, 394–404. [Google Scholar] [CrossRef]
- Chen, S.; Wojcieszak, R.; Dumeignil, F.; Marceau, E.; Royer, S. How Catalysts and Experimental Conditions Determine the Selective Hydroconversion of Furfural and 5-Hydroxymethylfurfural. Chem Rev 2018, 118, 11023–11117. [Google Scholar] [CrossRef]
- Mariscal, R.; Maireles-Torres, P.; Ojeda, M.; Sádaba, I.; López Granados, M. Furfural: a renewable and versatile platform molecule for the synthesis of chemicals and fuels. Energy & Environmental Science 2016, 9, 1144–1189. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, L.; Chen, Y.; Liu, W.; Feng, H.; Wang, B.; Zhang, X.; Wei, M. The selective hydrogenation of furfural over intermetallic compounds with outstanding catalytic performance. Green Chemistry 2019, 21, 5352–5362. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhang, J.; Ma, X.; An, Z.; Guo, S.; Shu, X.; Song, H.; Xiang, X.; He, J. A gradient reduction strategy to produce defects-rich nano-twin Cu particles for targeting activation of carbon-carbon or carbon-oxygen in furfural conversion. Journal of Catalysis 2020, 389, 78–86. [Google Scholar] [CrossRef]
- Kazi, F.K.; Patel, A.D.; Serrano-Ruiz, J.C.; Dumesic, J.A.; Anex, R.P. Techno-economic analysis of dimethylfuran (DMF) and hydroxymethylfurfural (HMF) production from pure fructose in catalytic processes. Chemical Engineering Journal 2011, 169, 329–338. [Google Scholar] [CrossRef]
- Hu, S.; Zhang, Z.; Zhou, Y.; Han, B.; Fan, H.; Li, W.; Song, J.; Xie, Y. Conversion of fructose to 5-hydroxymethylfurfural using ionic liquids prepared from renewable materials. Green Chemistry 2008, 10. [Google Scholar] [CrossRef]
- Wang, Q.; Yu, Z.; Feng, J.; Fornasiero, P.; He, Y.; Li, D. Insight into the Effect of Dual Active Cu0/Cu+ Sites in a Cu/ZnO-Al2O3 Catalyst on 5-Hydroxylmethylfurfural Hydrodeoxygenation. ACS Sustainable Chemistry & Engineering 2020, 8, 15288–15298. [Google Scholar] [CrossRef]
- Wang, Q.; Feng, J.; Zheng, L.; Wang, B.; Bi, R.; He, Y.; Liu, H.; Li, D. Interfacial Structure-Determined Reaction Pathway and Selectivity for 5-(Hydroxymethyl)furfural Hydrogenation over Cu-Based Catalysts. ACS Catalysis 2019, 10, 1353–1365. [Google Scholar] [CrossRef]
- An, Z.; Wang, W.; Dong, S.; He, J. Well-distributed cobalt-based catalysts derived from layered double hydroxides for efficient selective hydrogenation of 5-hydroxymethyfurfural to 2,5-methylfuran. Catalysis Today 2019, 319, 128–138. [Google Scholar] [CrossRef]
- Gao, D.L.; Lin, W.W.; Lin, Q.J.; Dai, F.F.; Xue, Y.X.; Chen, J.H.; Liu, Y.X.; Huang, Y.; Yang, Q. Remarkable adsorption capacity of Cu2+-doped ZnAl layered double hydroxides and the calcined materials toward phosphate. Journal of Environmental Chemical Engineering 2023, 11. [Google Scholar] [CrossRef]
- Takehira, K. Recent development of layered double hydroxide-derived catalysts − Rehydration, reconstitution, and supporting, aiming at commercial application −. Applied Clay Science 2017, 136, 112–141. [Google Scholar] [CrossRef]
- Teruel, L.; Bouizi, Y.; Atienzar, P.; Fornes, V.; Garcia, H. Hydrotalcites of zinc and titanium as precursors of finely dispersed mixed oxide semiconductors for dye-sensitized solar cells. Energy Environ. Sci. 2010, 3, 154–159. [Google Scholar] [CrossRef]
- Xia, S.; Shao, M.; Zhou, X.; Pan, G.; Ni, Z. Theoretical and experimental investigation into photoelectrocatalytic oxidation and reduction property of ZnFeTi mixed metal oxides. Journal of Molecular Catalysis A: Chemical 2015, 406, 127–136. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, J.; He, Y.; Feng, J.; Wu, T.; Li, D. Highly efficient PdAg catalyst using a reducible Mg-Ti mixed oxide for selective hydrogenation of acetylene: Role of acidic and basic sites. Journal of Catalysis 2017, 348, 135–145. [Google Scholar] [CrossRef]
- Yan, T.; Bing, W.; Xu, M.; Li, Y.; Yang, Y.; Cui, G.; Yang, L.; Wei, M. Acid-base sites synergistic catalysis over Mg-Zr-Al mixed metal oxide toward synthesis of diethyl carbonate. RSC Adv 2018, 8, 4695–4702. [Google Scholar] [CrossRef]
- Kuljiraseth, J.; Wangriya, A.; Malones, J.M.C.; Klysubun, W.; Jitkarnka, S. Synthesis and characterization of AMO LDH-derived mixed oxides with various Mg/Al ratios as acid–basic catalysts for esterification of benzoic acid with 2-ethylhexanol. Applied Catalysis B: Environmental 2019, 243, 415–427. [Google Scholar] [CrossRef]
- Salehpour, S.; Dubé, M.A. Towards the Sustainable Production of Higher-Molecular-Weight Polyglycerol. Macromolecular Chemistry and Physics 2011, 212, 1284–1293. [Google Scholar] [CrossRef]
- Pérez-Barrado, E.; Pujol, M.C.; Aguiló, M.; Llorca, J.; Cesteros, Y.; Díaz, F.; Pallarès, J.; Marsal, L.F.; Salagre, P. Influence of acid–base properties of calcined MgAl and CaAl layered double hydroxides on the catalytic glycerol etherification to short-chain polyglycerols. Chemical Engineering Journal 2015, 264, 547–556. [Google Scholar] [CrossRef]
- Wei, B.; Jin, L.; Wang, D.; Shi, H.; Hu, H. Catalytic upgrading of lignite pyrolysis volatiles over modified HY zeolites. Fuel 2020, 259. [Google Scholar] [CrossRef]
- Yang, H.; Zhang, J.; Li, C.; Chen, Z.; Wang, D.; Wang, D.; Ibrahim Sharif, M.; Li, J.; Yu, J.; Gao, S. The effect of acid-base synergistic catalysis on upgrading of volatiles from coal pyrolysis over Mg-Al composite oxides. Fuel 2022, 321. [Google Scholar] [CrossRef]
- Wang, Q.; Xie, W.; Jia, X.; Chen, B.; An, S.; Xie, X.; Huang, L. Ca–Al layered double hydroxides-derived Ni-based catalysts for hydrogen production via auto-thermal reforming of acetic acid. International Journal of Hydrogen Energy 2019, 44, 20007–20016. [Google Scholar] [CrossRef]
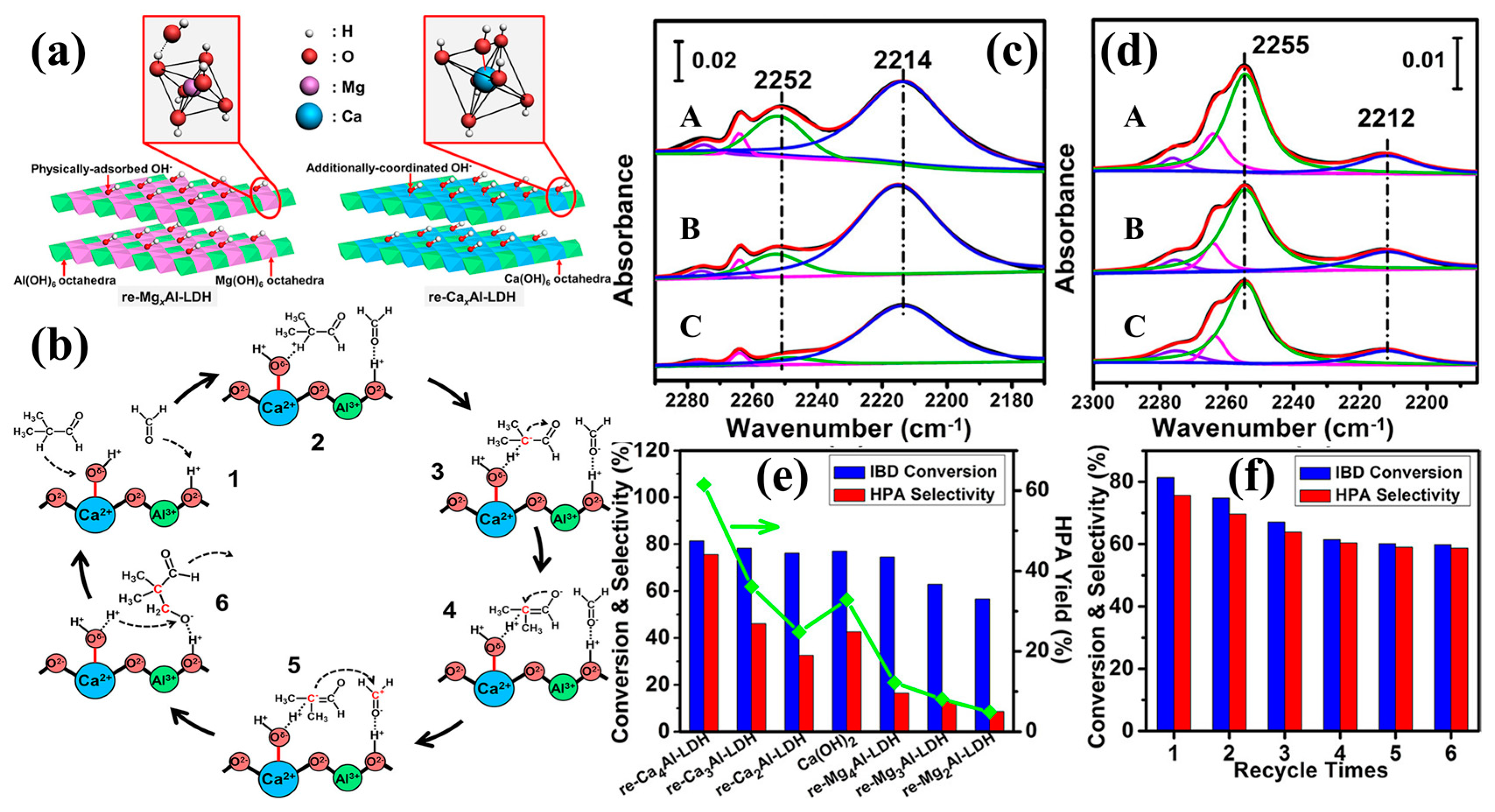
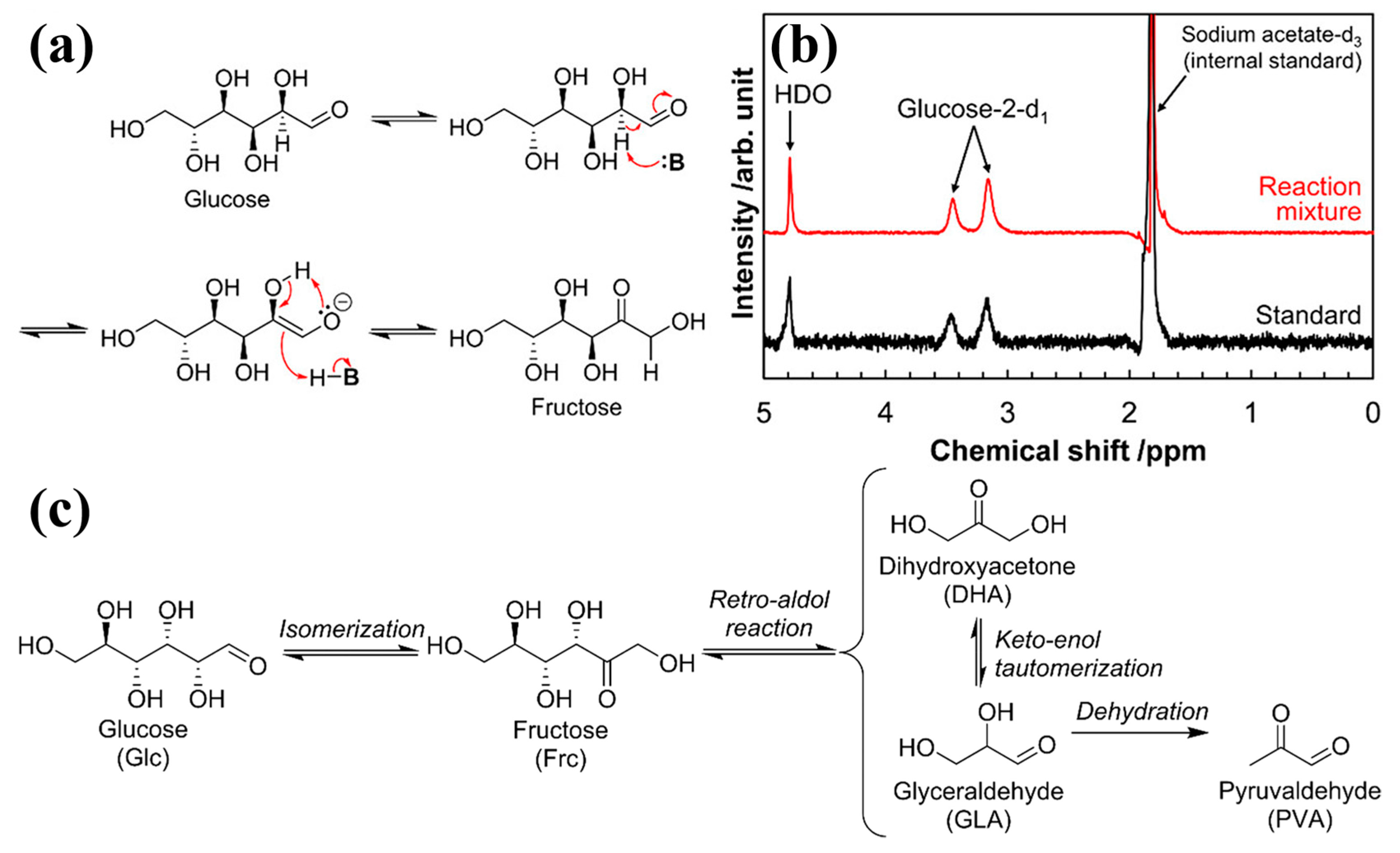
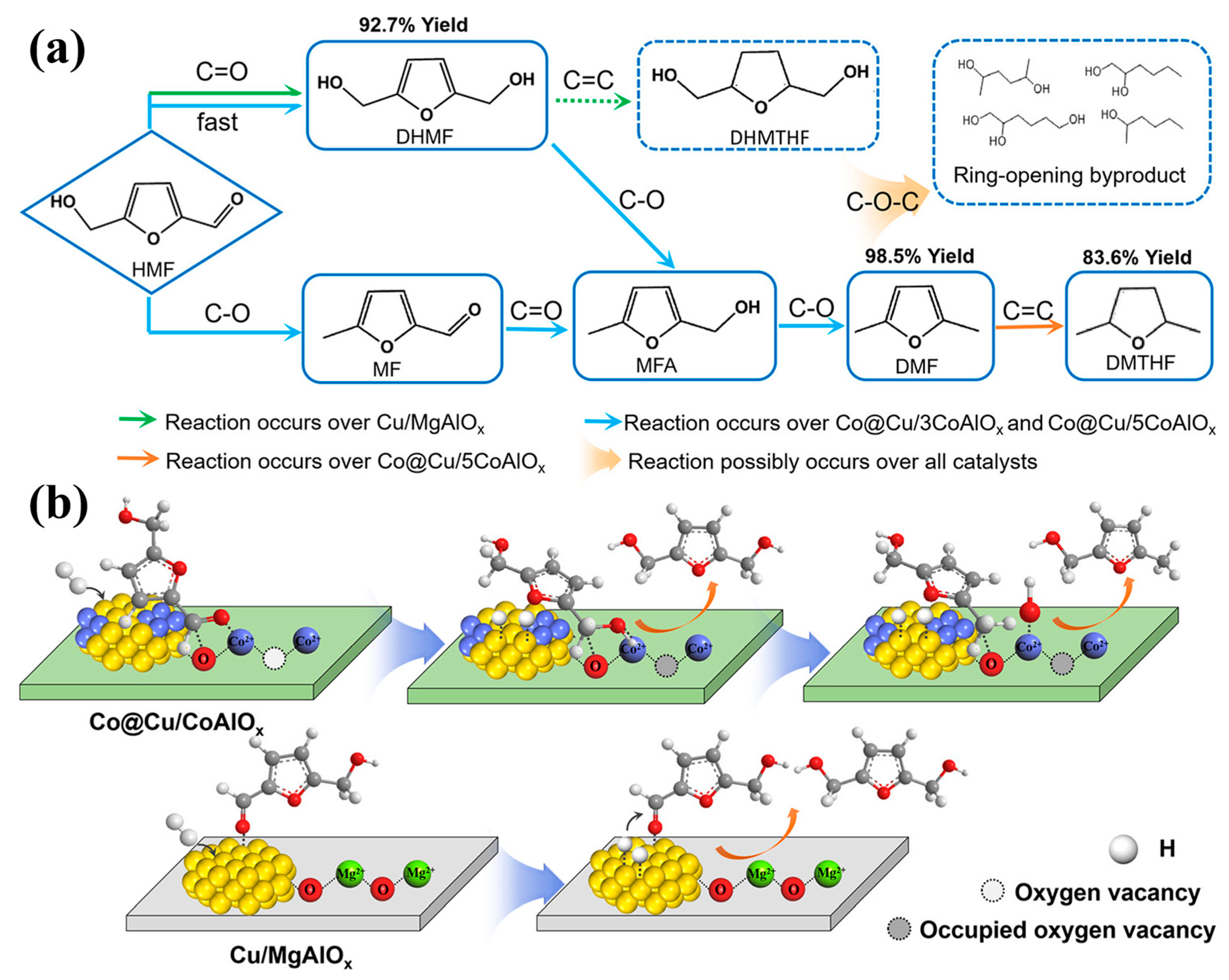
| Reaction type | Catalysts | Reaction conditions* | Yield (Y) or conversion (C) | Ref. |
|---|---|---|---|---|
| Phenol hydroxylation | CuZnFeAl LDHs | T=60 ℃, P=1 MPa | C=66.9% | [36] |
| Electrocatalytic aldehyde oxidation | NiV LDHs | ED=200 mA cm-2 | Y=77.6% | [37] |
| NiCoFe LDHs | ED=30 mA cm-2 | C=95% | [38] | |
| Furfural hydrogenation | NiFe (3:1) LDHs | T=140 ℃, RT=5 h | Y=90.2% | [39] |
| NiSn IMCs@Al2O3 | T=100 ℃, P=2 MPa H2 | C=100% | [57] | |
| Cu0-Zn (Al)(Zr)O-10 | T=160 ℃, P=2.5 MPa H2 | C=100% | [58] | |
| Hydrogenation of pentahydroxymethylfurfural | Cu/ZnAl MMO | T=180 ℃, P=1.2MPa H2 | C=100% | [61] |
| Cu/CoAlOx | T=150 ℃, P=1.5 Mpa H2, RT=5 h | C=100% | [62] | |
| Co-(ZnO-ZnAl2O4) | T=130 ℃, P=0.7 MPa H2 | Y=74.2% | [63] | |
| Conversion of glucose to methyl lactate | MgAl (5:1) LDH | T=150 ℃, RT=2 h | Y=47.6% | [40] |
| Glucose isomerization | MgAl LDH | T=120 ℃, RT=5 min | Y=25mol% | [41] |
| re-MgAl LDH | T=100 ℃, RT=5 h | C>40% | [51] | |
| MgAl (3:1) LDH | T=90 ℃, RT=2 h, in ethanol | Y=56% | [52] | |
| Aldehyde-alcohol condensation | re-CaAl LDH | T=70 ℃, RT=6 h | Y=61.5% | [49] |
| re-CaMnAl LDH | T=70 ℃, RT=8 h | Y=70.3% | [50] | |
| Dimethyl oxalate hydrogenation | CuMgAl−MMO | T=165 ℃ | 94.4% | [54] |
| Glycerol etherification | CaAl-MMO | T=235 ℃ | Y=59% | [72] |
| Ethanol vapour reconstitution | Ni/CaO-Al2O3 | T=650 ℃, RT=9 h | C=100% | [75] |
| Low-order coal pyrolysis upgrading | MgxAl-LDOs | T=450 ℃ | Y= 63.0 wt% | [74] |
| Synthesis of diethylcarbonate | MgZrAl-MMO | T=200 ℃, RT=5 h | Y=37.6% | [69] |
| Benzoate conversion | AMO-MgxAlO | T=110 ℃, RT=10 h | C=63% | [70] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




