Submitted:
21 November 2023
Posted:
22 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Currently established RNA labeling tools
2.1. RNA category and functions
2.2. RNA Tracing methods
2.2.1. GFP fused to targeting RNA
2.2.2. Aptamer-based RNA labeling
3. Aggregation-Induced Emission
3.1. ACQ effect limits the application of traditional fluorescent materials
3.2. The development of AIEgens
3.3. The luminescence mechanism of AIEgens
4. Application of AIEgens as a fluorescent probe and possibilities for RNA labeling in plants
4.1. Application of AIEgens in animal and medical
4.1.1. Probes for Drug Screening
4.1.2. Probes for Micromolecular Biomarkers
4.1.3. Cell Imaging
4.2. Application of AIEgens in plants
4.2.1. AIEgens enhance the efficiency of photosynthesis in plants
4.2.2. The application of AIEgens as fluorescent labeling in plant science.
5. Outlook
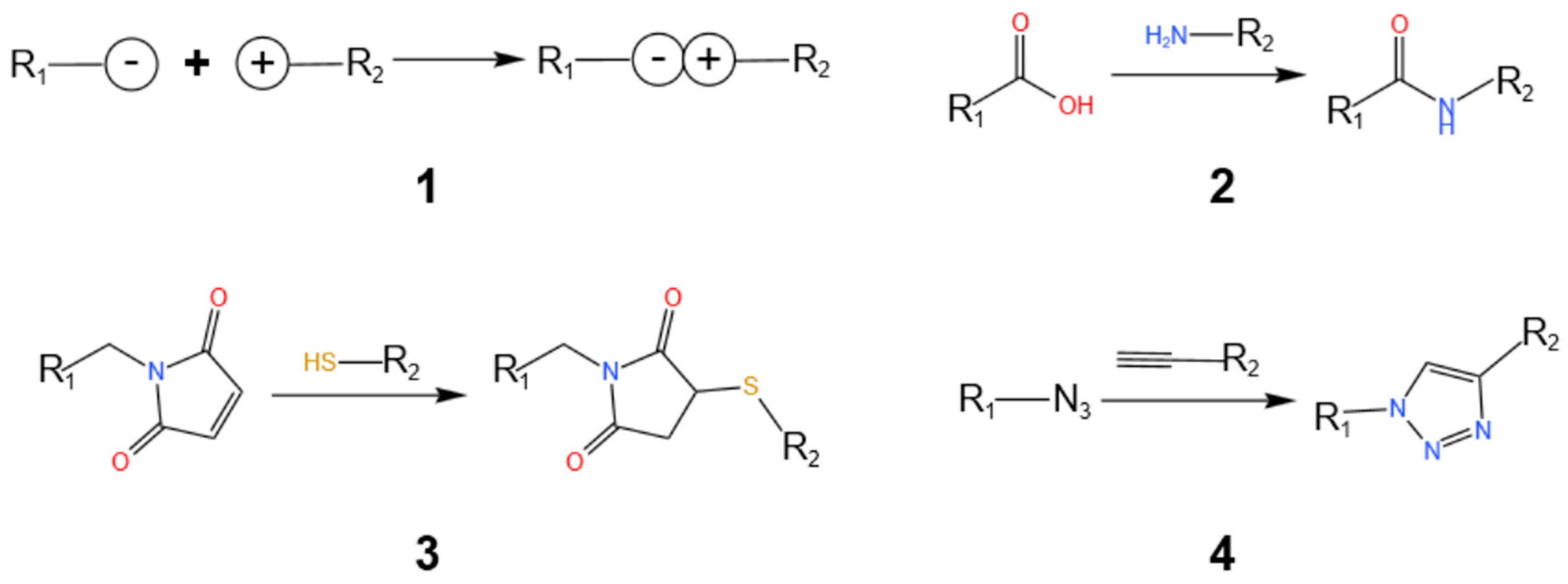
5.1. Click chemistry for AIE labeling RNA
5.2. For AIE, CRISPR may be on the way
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Shimomura, O.; Johnson, F.H.; Saiga, Y. Extraction, purification and properties of aequorin, a bioluminescent protein from the luminous hydromedusan, Aequorea. J. Cell. Comp. Physiol. 1962, 59, 223–239. [Google Scholar] [CrossRef] [PubMed]
- Paige, J. S.; Wu, K. Y.; Jaffrey, S. R. RNA Mimics of Green Fluorescent Protein. Science 2011, 333, 642–646. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Leung, N.L.; Kwok, R.T.; Lam, J.W.; Tang, B.Z. Aggregation-Induced Emission: Together We Shine, United We Soar! Chem Rev. 2015, 115, 11718-940. [Google Scholar] [CrossRef] [PubMed]
- Crick, F.H. On protein synthesis. Symp. Soc. Exp. Biol. 1958, 12, 138–163. [Google Scholar]
- Sprinzl, M.; Cramer, F. The -C-C-A end of tRNA and its role in protein biosynthesis. Prog. Nucleic. Acid. Res. Mol. Biol. 1979, 22, 1–69. [Google Scholar]
- Hoagland, M.B.; Stephenson, M.L.; Scott, J.F.; Hecht, L.I.; Zamecnik, P.C. A soluble ribonucleic acid intermediate in protein synthesis. J. Biol. Chem. 1958, 231, 241–257. [Google Scholar] [CrossRef] [PubMed]
- Jahn, M.; Rogers, M.J.; Söll, D. Anticodon and acceptor stem nucleotides in tRNA(Gln) are major recognition elements for E. coli glutaminyl-tRNA synthetase. Nature 1991, 352, 258–260. [Google Scholar] [CrossRef]
- Tamura, K. Origins and Early Evolution of the tRNA Molecule. Life (Basel). 2015, 5, 1687–1699. [Google Scholar] [CrossRef]
- Morceau, F.; Chateauvieux, S.; Gaigneaux, A.; Dicato, M.; Diederich, M. Long and short non-coding RNAs as regulators of hematopoietic differentiation. Int. J. Mol. Sci. 2013, 14, 14744–14770. [Google Scholar] [CrossRef]
- Lee, R.C.; Feinbaum, R.L.; Ambros, V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 1993, 75, 843–854. [Google Scholar] [CrossRef]
- O'Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front Endocrinol (Lausanne) 2018, 9, 402. [Google Scholar] [CrossRef] [PubMed]
- Wightman, B.; Ha, I.; Ruvkun, G. Posttranscriptional regulation of the heterochronic gene lin-14 by lin-4 mediates temporal pattern formation in C. elegans. Cell 1993, 75, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Rana, T.M. Illuminating the silence: understanding the structure and function of small RNAs. Nat. Rev. Mol. Cell. Biol. 2007, 8, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Truesdell, S.S.; Mortensen, R.D.; Seo, M.; Schroeder J., C.; Lee J., H.; LeTonqueze, O.; Vasudevan, S. MicroRNA-mediated mRNA translation activation in quiescent cells and oocytes involves recruitment of a nuclear microRNP. Sci. Rep. 2012, 2, 842. [Google Scholar] [CrossRef] [PubMed]
- Vasudevan, S.; Steitz, J.A. AU-rich-element-mediated upregulation of translation by FXR1 and Argonaute 2. Cell 2007, 128, 1105–1118. [Google Scholar] [CrossRef] [PubMed]
- Kung, J.T.; Colognori, D.; Lee, J.T. Long noncoding RNAs: past, present, and future. Genetics 2013, 193, 651–669. [Google Scholar] [CrossRef] [PubMed]
- Fire, A.; Xu, S.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E.; Mello, C.C. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 1998, 391, 806–811. [Google Scholar] [CrossRef]
- Elbashir, S.M.; Harborth, J.; Lendeckel, W.; Yalcin, A.; Weber, K.; Tuschl, T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 2001, 411, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Yisraeli, J. K.; Sokol, S.; Melton, D. A. A two-step model for the localization of maternal mRNA in Xenopus oocytes: involvement of microtubules and microfilaments in the translocation and anchoring of Vg1 mRNA. Development (Cambridge, England) 1990, 108, 289–298. [Google Scholar] [CrossRef]
- Bashirullah, A.; Cooperstock, R. L.; Lipshitz, H. D. RNA localization in development. Annu. Rev. Biochem. 1998, 67, 335–394. [Google Scholar] [CrossRef]
- Jose A., M. Movement of regulatory RNA between animal cells. Genesis. 2015, 53, 395–416. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Chen, X. . Intercellular and systemic trafficking of RNAs in plants. Nat. Plants 2018, 4, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Peabody, D.S. The RNA binding site of bacteriophage MS2 coat protein. EMBO. J. 1993, 12, 595–600. [Google Scholar] [CrossRef]
- Bertrand, E.; Chartrand, P.; Schaefer, M.; Shenoy, S.M.; Singer, R.H.; Long, R.M. Localization of ASH1 mRNA particles in living yeast. Mol. Cell 1998, 2, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Chubb, J.R.; Trcek, T.; Shenoy, S.M.; Singer, R.H. Transcriptional pulsing of a developmental gene. Curr. Biol. 2006, 16, 1018–1025. [Google Scholar] [CrossRef]
- Golding, I.; Paulsson, J.; Zawilski, S.M.; Cox, E.C. Real-time kinetics of gene activity in individual bacteria. Cell 2005, 123, 1025–1036. [Google Scholar] [CrossRef] [PubMed]
- Luo, K.R.; Huang, N.C.; Yu, T.S. Selective Targeting of Mobile mRNAs to Plasmodesmata for Cell-to-Cell Movement. Plant Physiol. 2018, 177, 604–614. [Google Scholar] [CrossRef]
- Shimomura, O. Structure of the chromophore of Aequorea green fluorescent protein. FEBS Lett. 1979, 104, 220–222. [Google Scholar] [CrossRef]
- Cody, C.W.; Prasher, D.C.; Westler, W.M.; Prendergast, F.G.; Ward, W.W. Chemical structure of the hexapeptide chromophore of the Aequorea green-fluorescent protein. Biochemistry 1993, 32, 1212–1218. [Google Scholar] [CrossRef]
- Ormö, M.; Cubitt, A.B.; Kallio, K.; Gross, L.A.; Tsien, R.Y.; Remington, S.J. Crystal structure of the Aequorea victoria green fluorescent protein. Science 1996, 273, 1392–1395. [Google Scholar] [CrossRef]
- Yang., F.; Moss, L.G.; Phillips, G.N. Jr. The molecular structure of green fluorescent protein. Nat. Biotechnol. 1996, 14, 1246–1251. [Google Scholar] [CrossRef]
- Kinghorn, A.B.; Fraser, L.A.; Lang, S.; Shiu, S.C.C.; Tanner, J.A. Aptamer Bioinformatics. Int J Mol Sci. 2017, 18, 2516. [Google Scholar] [CrossRef] [PubMed]
- Ellington, A.D.; Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 1990, 346, 818–822. [Google Scholar] [CrossRef]
- Tuerk, C.; Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Strack, R. L.; Disney, M. D.; Jaffrey, S. R. A superfolding Spinach2 reveals the dynamic nature of trinucleotide repeat-containing RNA. Nat. Methods 2013, 10, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Filonov, G. S.; Moon, J.D.; Svensen, N.; Jaffrey, S. R. Broccoli: rapid selection of an RNA mimic of green fluorescent protein by fluorescence-based selection and directed evolution. J. Am. Chem. Soc. 2014, 136, 16299–16308. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, D.; Su, N.; Bao, B.; Xie, X.; Zuo, F.; Yang, L.; Wang, H.; Jiang, L.; Lin, Q.; et al. Visualizing RNA dynamics in live cells with bright and stable fluorescent RNAs. Nat Biotechnol. 2019, 37, 1287–1293. [Google Scholar] [CrossRef]
- Bai, J.; Luo, Y.; Wang, X.; Li, S.; Luo, M.; Yin, M.; Zuo, Y.; Li, G.; Yao, J.; Yang, H.; Zhang, M.; Wei, W.; Wang, M.; Wang, R.; Fan, C.; Zhao, Y. A protein-independent fluorescent RNA aptamer reporter system for plant genetic engineering. Nat. Commun. 2020, 11, 3847. [Google Scholar] [CrossRef]
- Dolgosheina, E. V.; Jeng, S. C.; Panchapakesan, S. S.; Cojocaru, R.; Chen, P. S.; Wilson, P. D.; Hawkins, N.; Wiggins, P. A.; Unrau, P. J. RNA mango aptamer-fluorophore: a bright, high-affinity complex for RNA labeling and tracking. ACS Chem. Biol. 2014, 9, 2412–2420. [Google Scholar] [CrossRef]
- Song, W.; Filonov, G. S.; Kim, H.; Hirsch, M.; Li, X.; Moon, J. D.; Jaffrey, S. R. Imaging RNA polymerase III transcription using a photostable RNA-fluorophore complex. Nat. Chem. Biol. 2017, 13, 1187–1194. [Google Scholar] [CrossRef]
- Yu, Z.; Wang, Y.; Mei, F.; Yan, H.; Jin, Z.; Zhang, P.; Zhang, X.; Tör, M.; Jackson, S.; Shi, N.; Hong, Y. Spinach-based RNA mimicking GFP in plant cells. Funct Integr Genomics 2022, 22, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Autour, A.; Jeng, S. C. Y.; Cawte, A. D.; Abdolahzadeh, A.; Galli, A.; Panchapakesan, S. S. S.; Rueda, D.; Ryckelynck, M.; Unrau, P. J. Fluorogenic RNA Mango aptamers for imaging small non-coding RNAs in mammalian cells. Nat. Commun. 2018, 9, 656. [Google Scholar] [CrossRef] [PubMed]
- Han, P.B.; Xu, H.; An, Z.F.; Cai, Z.Y.; Cai, Z.X.; Chao, H.; Chen, B.; Chen, M.; Chen, Y.; Chi, Z.G.; et al. Aggregation-Induced Emission. Prog. Chem. 2022, 34, 1–130. [Google Scholar]
- Hong, Y.; Lam, J.W.; Tang, B.Z. Aggregation-induced emission. Chem Soc Rev. 2011, 40, 5361-88. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; W, Y.; Lam, Jacky.; Tang, B.Z. Aggregation-Induced Emission of Tetraarylethene Luminogens. Current Organic Chemistry. 2010, 14, 2109-2132.
- Chen, J et al. Silole-containing polyacetylenes. Synthesis, thermal stability, light emission, nanodimensional aggregation, and restricted intramolecular rotation. Macromolecules 2003, 36, 1108-1117.
- Hong, Y.; Lam, JW.; Tang, BZ. Aggregation-induced emission: phenomenon, mechanism and applications. Chem Commun (Camb). 2009, 29, 4332-53.
- Xu, L.; Liang, X.; Zhang, S.; Wang, B.; Zhong, S.; Wang, M.; Cui, X. Riboflavin: A natural aggregation-induced emission luminogen (AIEgen) with excited-state proton transfer process for bioimaging. Dyes and Pigments 2020, 18, 108642.
- Yu, X.; Gao, YC.; Li, HW.; Wu, Y. Fluorescent Properties of Morin in Aqueous Solution: A Conversion from Aggregation Causing Quenching (ACQ) to Aggregation Induced Emission Enhancement (AIEE) by Polyethyleneimine Assembly. Macromol Rapid Commun. 2020, 14, e2000198.
- He, T.; Wang, H.; Chen, Z.; Liu, S.; Li, J.; Li, S. Natural Quercetin AIEgen Composite Film with Antibacterial and Antioxidant Properties for in Situ Sensing of Al3+ Residues in Food, Detecting Food Spoilage, and Extending Food Storage Times. ACS Appl Bio Mater. 2018, 1, 636-642.
- Lei, Y.; Liu, L.; Tang, X.; Yang, D.; Yang, X.; He, F. Sanguinarine and chelerythrine: two natural products for mitochondria-imaging with aggregation-induced emission enhancement and pH-sensitive characteristics. RSC 2018, 8, 3919-3927.
- Zhao, Q.; Li, L.; Li, F.; Yu, M.; Liu, Z.; Yi, T.; Huang, C. Aggregation-induced phosphorescent emission (AIPE) of iridium(III) complexes. Chem Commun (Camb). 2008, 6, 685-687.
- Dong, Y.; Lam, J.W.; Qin, A.; Sun, J.; Liu, J.; Li, Z.; Sun, J.; Sung, H.H.; Williams, I.D.; Kwok, H.S.; Tang, B.Z. Aggregation-induced and crystallization-enhanced emissions of 1,2-diphenyl-3,4-bis(diphenylmethylene)-1-cyclobutene. Chem Commun (Camb). 2007, 31, 3255-7.
- Yuan, W.Z.; Shen, X.Y.; Zhao, H.; Lam, J.W.; Tang, L.; Lu, P.; Wang, C.; Liu, Y.; Wang, Z.; Zheng, Q.; Sun, J.Z.; Ma, Y.; Tang, B.Z. Crystallization-Induced Phosphorescence of Pure Organic Luminogens at Room Temperature. Journal of Physical Chemistry C. 2010, 114, 6090-6099.
- Bolton, O.; Lee, K.; Kim, H.J.; Lin, KY.; Kim, J. Activating efficient phosphorescence from purely organic materials by crystal design. Nat Chem. 2011, 3, 205-210.
- Leung, N.L.; Xie, N.; Yuan, W.; Liu Y.; Wu, Q.; Peng, Q.; Miao, Q.; Lam, J.W.; Tang, B.Z. Restriction of intramolecular motions: the general mechanism behind aggregation-induced emission. Chemistry. 2014, 20, 15349-53.
- Tu, Y.; Zhao, Z.; Lam, J.W.Y.; Tang, B.Z. Mechanistic connotations of restriction of intramolecular motions (RIM). Natl Sci Rev. 2021, 8, nwaa260.
- Liu, D.; Zhao, Z.; Tang, B.Z. Natural products with aggregation-induced emission properties: from discovery to their multifunctional applications. Sci. Sin. Chim. 2022, 52, 1524–1546. [Google Scholar] [CrossRef]
- Jeong, H.; Shin, H.; Hong, S.; Kim, Y. Physiological Roles of Monomeric Amyloid-beta and Implications for Alzheimer's Disease Therapeutics. Exp. Neurobiol. 2022, 31, 65–88. [Google Scholar] [CrossRef] [PubMed]
- Mihaescu, A.S.; Valli, M.; Uribe, C.; Diez-Cirarda, M.; Masellis, M.; Graff-Guerrero, A.; Strafella, A. P. Beta amyloid deposition and cognitive decline in Parkinson's disease: a study of the PPMI cohort. Mol. Brain 2022, 15, 79. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Jiang, X.; Ma, L.; Wei, W.; Li, Z.; Chang, S.; Wen, J.; Sun, J.; Li, H. Role of Abeta in Alzheimer's-related synaptic dysfunction. Front. Cell Dev. Biol. 2022, 10, 964075. [Google Scholar]
- Lee, J.S.; Lee, S.-J. Mechanism of Anti-alpha-Synuclein Immunotherapy. J. Mov. Disord. 2016, 9, 14–19. [Google Scholar] [CrossRef]
- Liu, Y.; Jiao, C.; Lu, W.; Zhang, P.; Wang, Y. Research progress in the development of organic small molecule fluorescent probes for detecting H2O2. RSC Adv. 2019, 9, 18027–18041. [Google Scholar] [CrossRef]
- Shah, K.; DeSilva, S.; Abbruscato, T. The Role of Glucose Transporters in Brain Disease: Diabetes and Alzheimer's Disease. Int. J. Mol. Sci. 2012, 13, 12629–12655. [Google Scholar] [CrossRef] [PubMed]
- Sobotka, L. Is glucosis only basic energy substrate? Vnitr. Lek. 2016, 62, S100–102. [Google Scholar]
- Yuan, J.; Cen, Y.; Kong, X.J.; Wu, S.; Liu, C.L.W.; Yu, R.Q.; Chu, X. MnO2-Nanosheet-Modified Upconversion Nanosystem for Sensitive Turn-On Fluorescence Detection of H2O2 and Glucose in Blood. ACS Appl. Mater. Interfaces 2015, 7, 10548–10555. [Google Scholar] [CrossRef]
- Wang, X.W.; Huang, Y.Z.; Lv, W.W.; Li, C.M.; Zeng, W.; Zhang, Y.; Feng, X.P. A novel fluorescent probe based on ESIPT and AIE processes for the detection of hydrogen peroxide and glucose and its application in nasopharyngeal carcinoma imaging. Anal. Methods 2017, 9, 1872–1875. [Google Scholar] [CrossRef]
- Gorodzanskaya, E. G.; Larionova, V. B.; Zubrikhina, G. N.; Kormosh, N. G.; Davydova, T. V.; Laktionov, K. P. Role of glutathione-dependent peroxidase in regulation of lipoperoxide utilization in malignant tumors. Biochemistry (Mosc). 2001, 66, 221–224. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Zhan, C. Y.; Wang, J.; Zeng, F.; Wu, S.Z. An Activatable Nano-Prodrug for Treating Tyrosine-Kinase-Inhibitor-Resistant Non-Small Cell Lung Cancer and for Optoacoustic and Fluorescent Imaging. Small 2020, 16, e2003451. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Liu, B. Organelle-specific bioprobes based on fluorogens with aggregation-induced emission (AIE) characteristics. Org. Biomol. Chem. 2016, 14, 9931–9944. [Google Scholar] [CrossRef]
- Ueno, T.; Nagano, T. Fluorescent probes for sensing and imaging. Nat. Methods 2011, 8, 642–645. [Google Scholar] [CrossRef]
- Shim, E. B. Mitochondria Medicine and its Research Trend. J. Biomed. Eng. Res. 2009, 30, 355–361. [Google Scholar]
- Li, N.; Liu, L. Y.; Luo, H. Q.; Wang, H.Q.; Yang, D.P.; He, F. Flavanone-Based Fluorophores with Aggregation-Induced Emission Enhancement Characteristics for Mitochondria-Imaging and Zebrafish-Imaging. Molecules 2020, 25, 3298. [Google Scholar] [CrossRef]
- Pu, J. Targeting the lysosome: Mechanisms and treatments for nonalcoholic fatty liver disease. J. Cell. Biochem. 2022; 10, 1624–1633. [Google Scholar]
- Lou, X.D.; Zhang, M.S.; Zhao, Z.J.; Min, X.H.; Hakeem, A.; Huang, F.J.; Gao, P.C.; Xia, F.; Tang, B.Z. A photostable AIE fluorogen for lysosome-targetable imaging of living cells. J. Mater. Chem. B. 2016, 4, 7168–7168. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.; Liu, H.; Chen, X.; Hu, R.; Li, M.; He, W.; Du, J.; Liu, Z.; Qin, A.; Lam, J.W.Y. ; Kwok, RTK.; Tang, B.Z. Augmenting photosynthesis through facile AIEgen-chloroplast conjugation and efficient solar energy utilization. Mater Horiz. 2021; 8, 1433–1438. [Google Scholar]
- Daming, X.; Mingyue, J.; Xiongfei, L.; Shouxin, L.; Jian, L.; Zhijun, Chen.; and Shujun, L. Sustainable Carbon Dot-Based AIEgens: Promising Light-Harvesting Materials for Enhancing Photosynthesis.ACS Sustainable Chemistry & Engineering 2021, 9, 4139-4145.
- Zhao, Y.; Zhang, L.; Liu, Y.; Deng, Z.; Zhang, R.; Zhang, S.; He, W.; Qiu, Z.; Zhao, Z.; Tang, B.Z. AIEgens in Solar Energy Utilization: Advances and Opportunities. Langmuir. 2022, 38, 8719-8732.
- Liu, H.; Yan, N.; Bai, H.; Kwok, R. T. K.; Tang, B. Z. Aggregation-induced emission luminogens for augmented photosynthesis. Exploration. 2022, 2, 20210053.
- Nicol, A.; Wang, K.; Wong, K.; Kwok, R.T.K.; Song, Z.; Li, N.; Tang, B.Z. Uptake, Distribution, and Bioimaging Applications of Aggregation-Induced Emission Saponin Nanoparticles in Arabidopsis thaliana. ACS Appl Mater Interfaces. 2017, 30, 28298-28304liana.
- Sun, L.; Wang, X.; Shi, J.; Yang, S.; Xu, L. Kaempferol as an AIE-active natural product probe for selective Al3+ detection in Arabidopsis thaliana. Spectrochim Acta A Mol Biomol Spectrosc. 2021, 249, 119303.
- Wu, M.; Yin, C.; Jiang, X.; Sun, Q.; Xu, X.; Ma, Y.; Liu, X.; Niu, N.; Chen, L. Biocompatible Abscisic Acid-Sensing Supramolecular Hybridization Probe for Spatiotemporal Fluorescence Imaging in Plant Tissues. Anal Chem. 2022, 94, 8999-9008.
- Himo, F.; Lovell, T.; Hilgraf, R.; Rostovtsev, V.V.; Noodleman, L.; Sharpless, K.B.; Fokin, V.V. Copper(I)-catalyzed synthesis of azoles. DFT study predicts unprecedented reactivity and intermediates. J. Am. Chem. Soc. 2005, 127, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Rostovtsev, V, V.; Green, L.G.; Fokin, V.V.; Sharpless, K, B. A stepwise huisgen cycloaddition process: copper(I)-catalyzed regioselective "ligation" of azides and terminal alkynes. Angew. Chem. Int. Ed. Engl. 2002, 14, 2596–9. [Google Scholar] [CrossRef]
- Agard, N.J.; Prescher, J.; Bertozzi, C.R. A strain-promoted [3 + 2] azide-alkyne cycloaddition for covalent modification of biomolecules in living systems. J. Am. Chem. Soc. 2004, 126, 15046–15047. [Google Scholar] [CrossRef] [PubMed]
- Jao, C.Y.; Salic, A. Exploring RNA transcription and turnover in vivo by using click chemistry. Proc. Natl. Acad. Sci. USA. 2008, 105, 15779–15784. [Google Scholar] [CrossRef]
- Sheet, S.K.; Sen, B.; Patra, S. .;, Rabha, M.; Aguan, K.; Khatua, S. Aggregation-Induced Emission-Active Ruthenium(II) Complex of 4,7-Dichloro Phenanthroline for Selective Luminescent Detection and Ribosomal RNA Imaging. ACS Appl. Mater. Interfaces 2018, 10, 14356–14366. [Google Scholar] [CrossRef] [PubMed]
- Jinek, M.; Chylinski, K.; Fonfara, I.; Hauer, M.; Doudna, J.A.; Charpentier, E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 2012, 337, 816–821. [Google Scholar] [CrossRef]
- Cong, L.; Ran, F.A.; Cox, D.; Lin, S.; Barretto, R.; Habib, N.; Hsu, P.D.; Wu, X.; Jiang, W.; Marraffini, L.A.; Zhang, F. Multiplex genome engineering using CRISPR/Cas systems. Science 2013, 339, 819–823. [Google Scholar] [CrossRef]
- Anzalone, A.V.; Koblan, L.W.; Liu, D.R. Genome editing with CRISPR-Cas nucleases, base editors, transposases and prime editors. Nat Biotechnol. 2020, 38, 824–844. [Google Scholar] [CrossRef]
- Lapinaite, A.; Knott, G.J.; Palumbo, C.M.; Lin-Shiao, E.; Richter, M.F.; Zhao, K.T.; Beal, P.A.; Liu, D.R.; Doudna, J.A. DNA capture by a CRISPR-Cas9-guided adenine base editor. Science 2020, 369, 566–571. [Google Scholar] [CrossRef]
- Abudayyeh, O.O.; Gootenberg, J.S.; Franklin, B.; Koob, J.; Kellner, M.J.; Ladha, A.; Joung, J.; Kirchgatterer, P.; Cox, D.B.T.; Zhang, F. A cytosine deaminase for programmable single-base RNA editing. Science 2019, 365, 382–386. [Google Scholar] [CrossRef] [PubMed]
- Cox, D.B.T.; Gootenberg, J.S.; Abudayyeh, O.O.; Franklin, B.; Kellner, M.J.; Joung, J.; Zhang, F. RNA editing with CRISPR-Cas13. Science 2017, 358, 1019-1027.
- Abudayyeh, O.O.; Gootenberg, J.S.; Essletzbichler, P.; Han, S.; Joung, J.; Belanto, J.J.; Verdine, V.; Cox, D.B.T.; Kellner, M.J.; Regev, A.; Lander, E.S.; Voytas, D.F.; Ting, A.Y.; Zhang, F. RNA targeting with CRISPR-Cas13. Nature 2017, 550, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Abudayyeh, O.O.; Gootenberg, J.S.; Konermann, S.; Joung, J.; Slaymaker, I.M.; Cox, D.B.; Shmakov, S.; Makarova, K.S.; Semenova, E.; Minakhin, L.; Severinov, K.; Regev, A.; Lander, E.S.; Koonin, E.V.; Zhang, F. C2c2 is a single-component programmable RNA-guided RNA-targeting CRISPR effector. Science 2016, 353, aaf5573. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Bu, S.; Wang, Z.; Zhou, H.; Li, X.; Wei, J.; He, X.; Wan, J. Click Chemistry Actuated Exponential Amplification Reaction Assisted CRISPR-Cas12a for the Electrochemical Detection of MicroRNAs. ACS Omega 2022, 7, 35515–35522. [Google Scholar] [CrossRef]
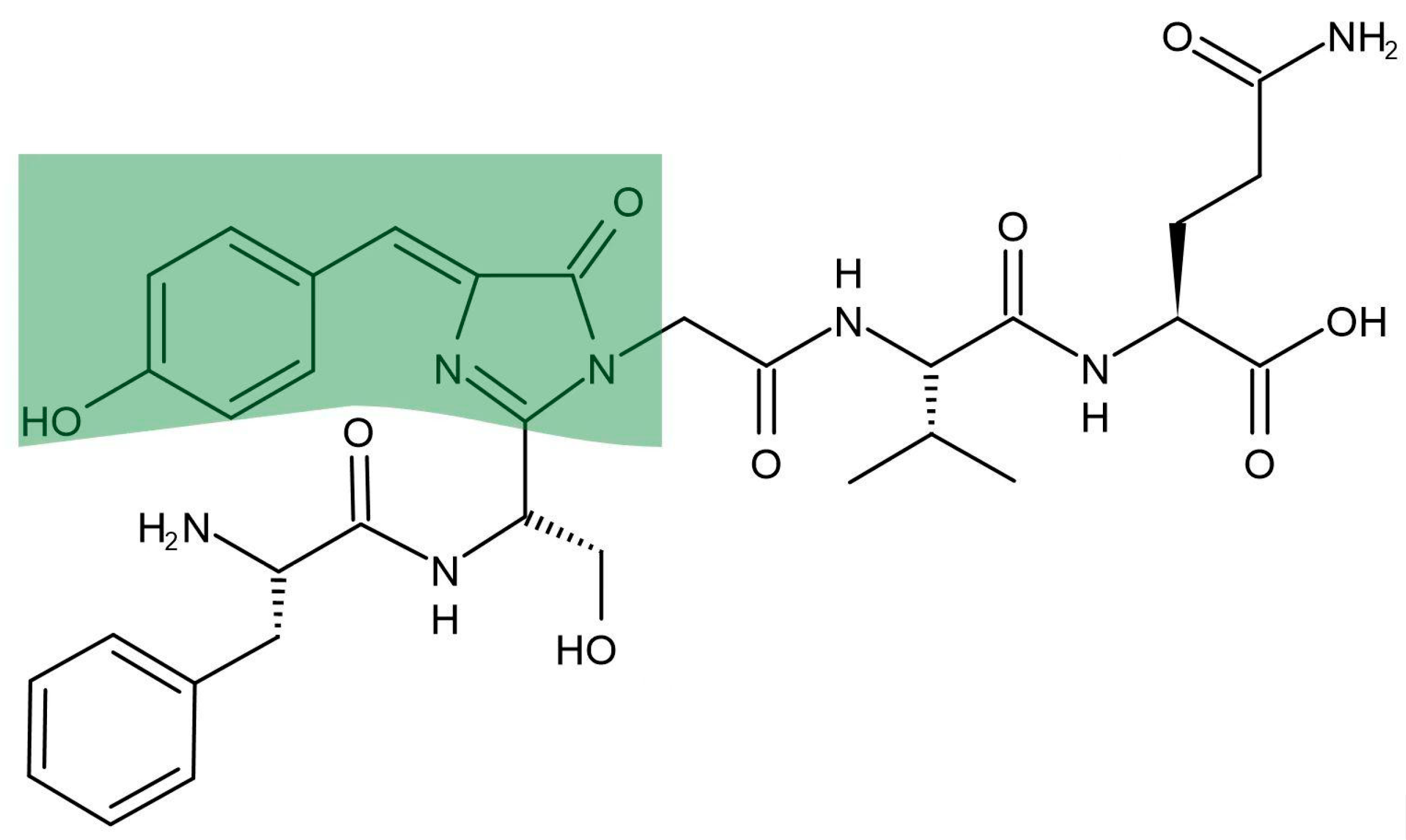
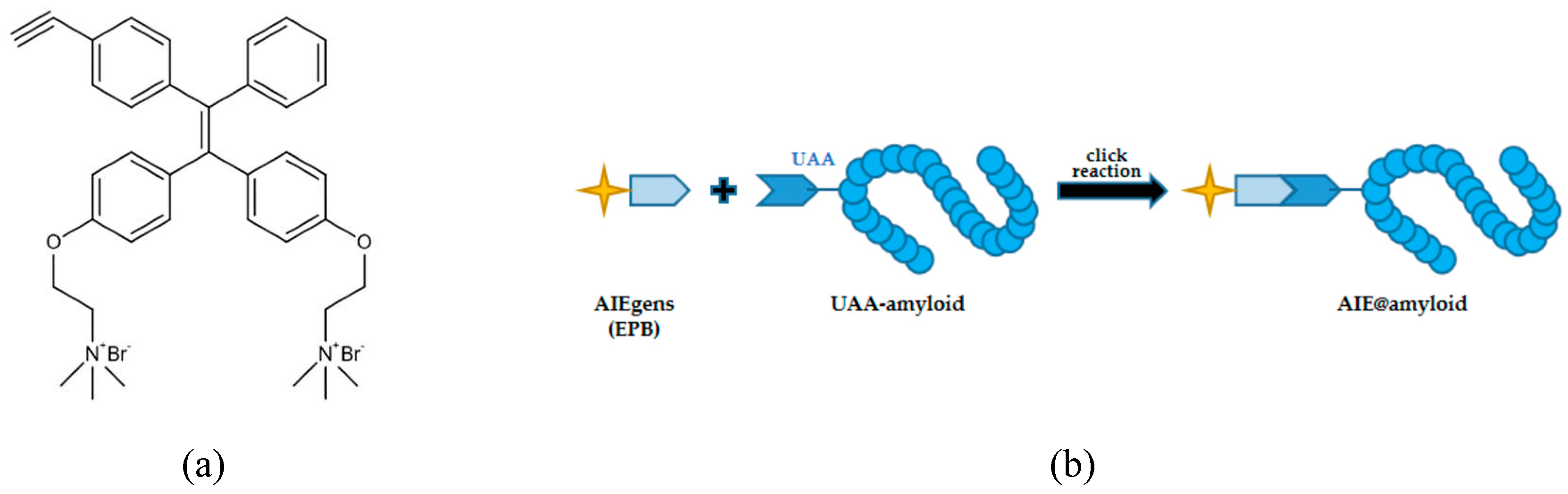
| RNA aptamer | Fluorophore | Color | Application | Length of RNA |
|---|---|---|---|---|
| Spinach [2, 41] | DFHBI | Green | HEK-293T, E. coli and Onion | 98-nt |
| Spinach2 [35] | DFHBI | Green | E. coli, HEK293T, HeLa and COS-7 | 95-nt |
| Pepper [37] | HBC | Green | E. coli, 293T/17 HeLa, COS-7, NIH/3T3, U-87, HCT 116 and MKN-45 | 43-nt |
| Broccoli [36] | DFHBI-1T | Green | E. coli and HEK293T | 49-nt |
| Corn [40] | DFHO | Yellow | E. coli and HEK293T | 28-nt |
| 3WJ-4 × Bro [38] | DFHBI-1T | Green | E. coli and Nicotiana benthamiana | 1701-nt |
| Mango [39] | Thiazole Orange(TO1) | Yellow | E. coli and C. elegans | 23-nt |
| Mango [39] | TO3 | Red | E. coli and C. elegans | 23-nt |
| Mango II [42] | Thiazole Orange(TO1) | Yellow | HEK293T | 40-nt |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




