Submitted:
20 November 2023
Posted:
21 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Planarian
2.2. Contamination
2.3. Swimming Speed
2.4. Body Measurements
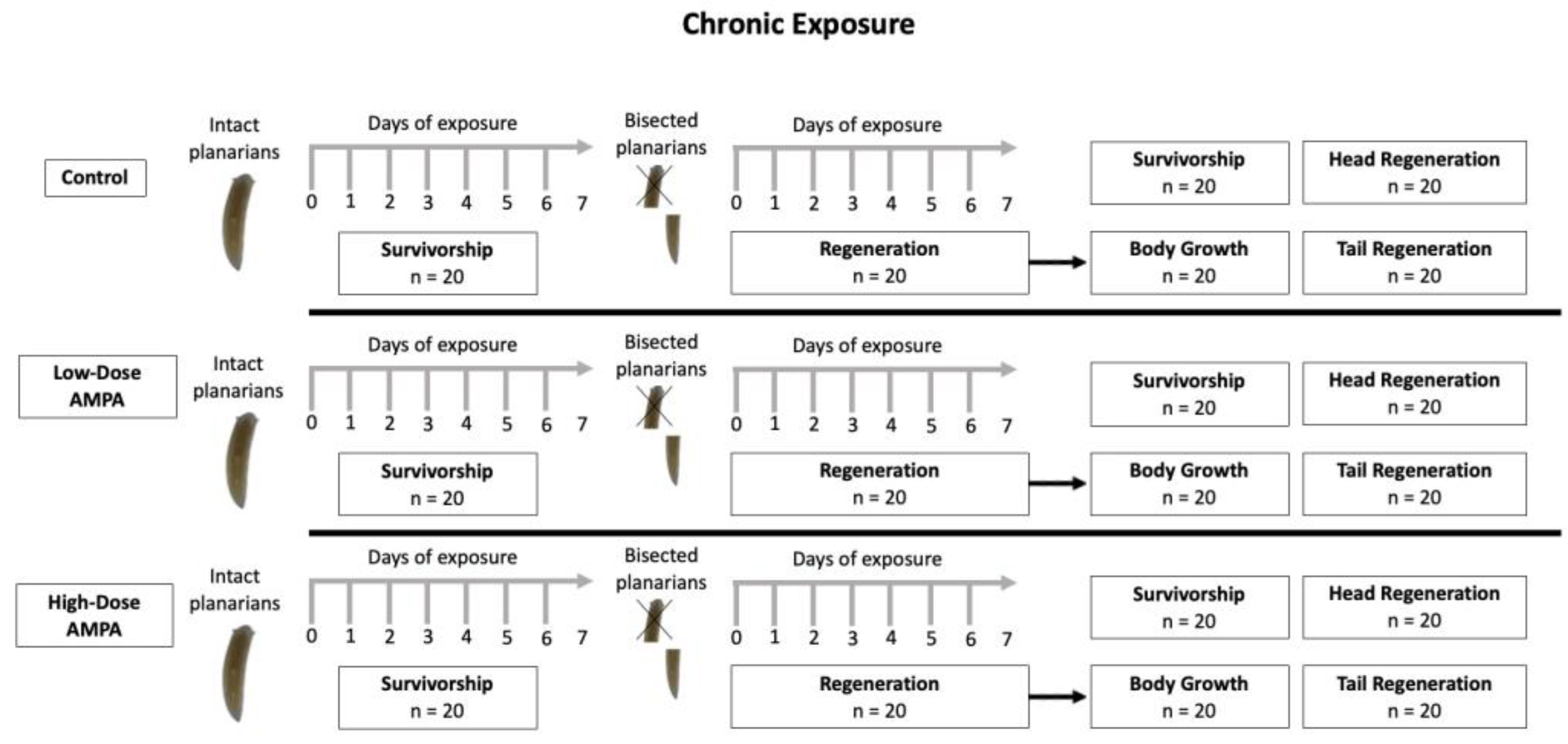
2.5. Chronic Exposure
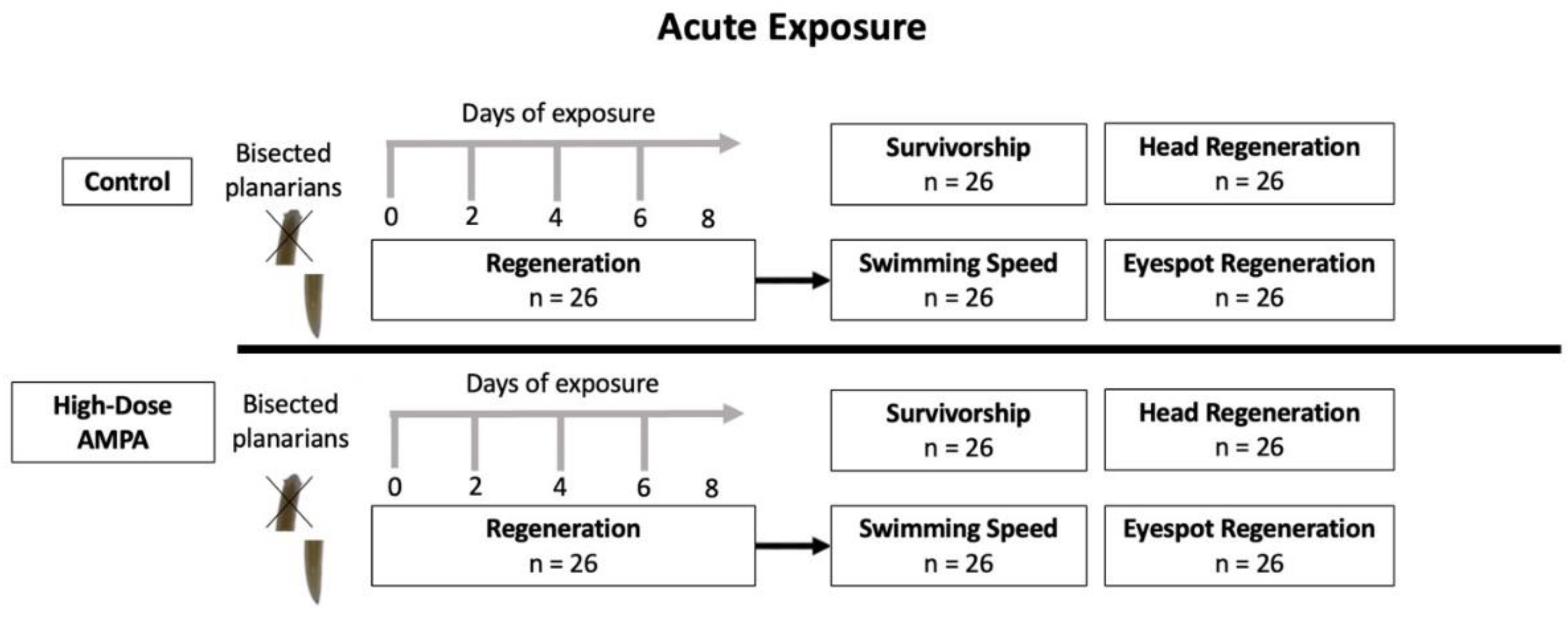
2.6. Acute AMPA Exposure
2.7. Data Analysis
3. Results
3.1. Survivorship while Regenerating during Chronic Exposure
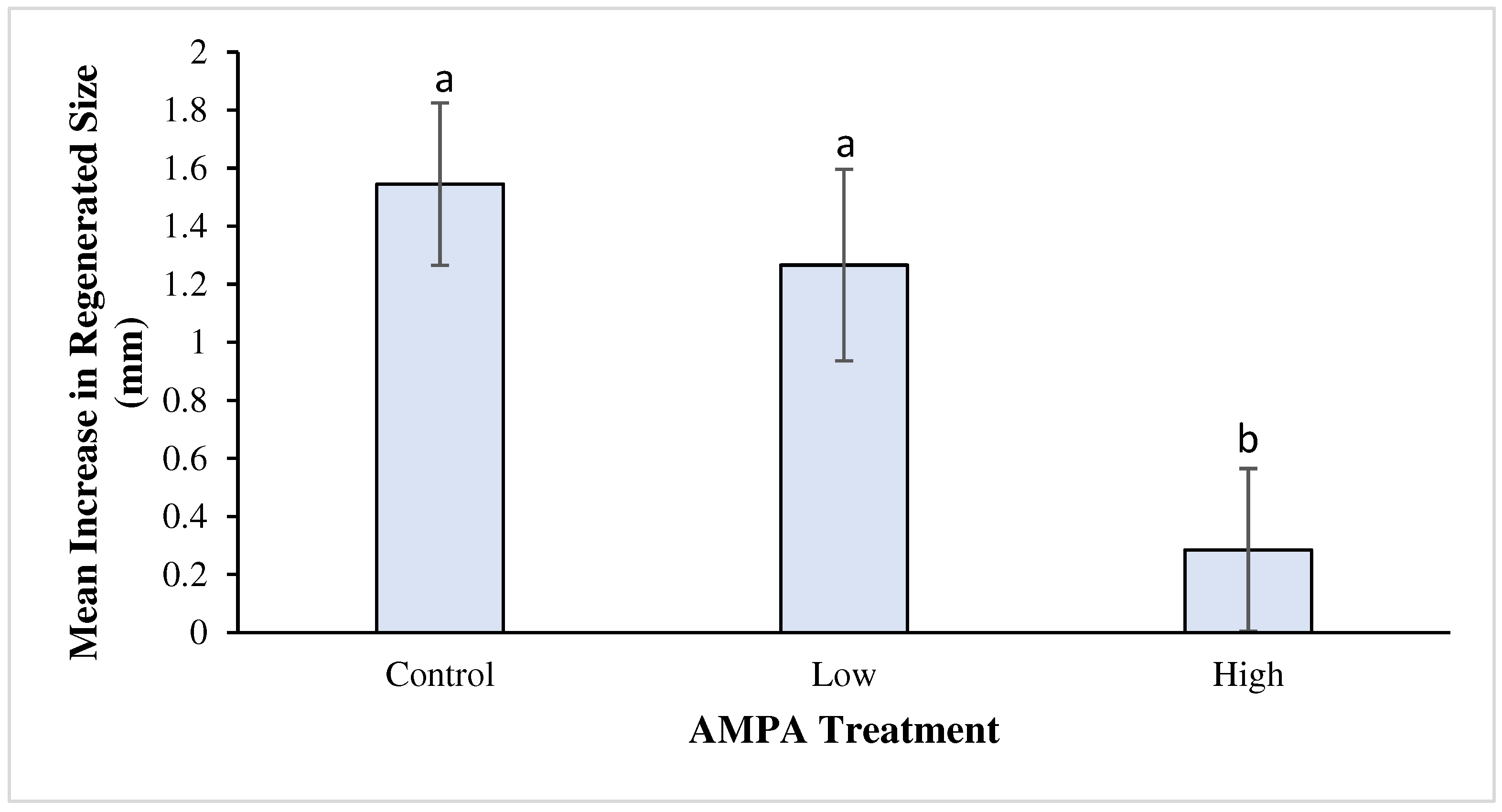
3.2. Growth during Chronic Exposure
3.3. Head Regeneration after Chronic Exposure
3.4. Tail Regeneration after Chronic Exposure
3.5. Survivorship after Acute Exposure
3.6. Body Regeneration after Acute Exposure
3.7. Eye Spot Regeneration after Acute Exposure
3.8. Swimming Speed after Acute Exposure
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Myers, J.P.; Antoniou, M.N.; Blumberg, B.; Carroll, L.; Colborn, T.; Everett, L.G.; Hansen, M.; Landrigan, P.J.; Lanphear, B.P.; Mesnage, R.; Vandenberg, L.N. Concerns over use of glyphosate-based herbicides and risks associated with exposures: a consensus statement. Environ Health 2016, 15, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bach, N.C.; Marino, D.J.; Natale, G.S.; Somoza, G.M. Effects of glyphosate and its commercial formulation, Roundup® Ultramax, on liver histology of tadpoles of the neotropical frog, Leptodactylus latrans (amphibia: Anura). Chemosphere 2018, 202, 289–297. [Google Scholar] [CrossRef]
- Rudell, E.C.; Zanrosso, B.A.; Frandaloso, D.; Giacomini, A.J.; Spadotto, D.V.; Vargas, L.; Nunes, A.L.; Santos, F.M. Integrated weed management strategies in a long-term crop rotation system. Weed Science 2023, 41, e020220053. [Google Scholar] [CrossRef] [PubMed]
- Battaglin, W.A.; Meyer, M.T.; Kuivila, K.M.; Dietze, J.E. Glyphosate and its degradation product AMPA occur frequently and widely in US soils, surface water, groundwater, and precipitation. JAWRA 2014, 50, 275–290. [Google Scholar] [CrossRef]
- Baek, Y.; Bobadilla, L.K.; Giacomini, D.A.; Montgomery, J.S.; Murphy, B.P.; Tranel, P.J. Evolution of glyphosate-resistant weeds. Rev Environ Contam T 2021, 255, 93–128. [Google Scholar] [CrossRef]
- Gill, J.P.K.; Sethi, N.; Mohan, A.; Datta, S.; Girdhar, M. Glyphosate toxicity for animals. Environ Chem Lett 2018, 16, 401–426. [Google Scholar] [CrossRef]
- Pochron, S.; Choudhury, M.; Gomez, R.; Hussaini, S.; Illuzzi, K.; Mann, M.; Mezic, M.; Nikakis, J.; Tucker, C. Temperature and body mass drive earthworm (Eisenia fetida) sensitivity to a popular glyphosate-based herbicide. Appl Soil Ecol 2019, 139, 32–39. [Google Scholar] [CrossRef]
- Pochron, S.; Simon, L.; Mirza, A.; Littleton, A.; Sahebzada, F.; Yudell, M. 2020. Glyphosate but not Roundup® harms earthworms (Eisenia fetida). Chemosphere 2020, 241, 125017. [Google Scholar] [CrossRef] [PubMed]
- Pochron, S.T.; Mirza, A.; Mezic, M.; Chung, E.; Ezedum, Z.; Geraci, G.; Mari, J.; Meiselbach, C.; Shamberger, O.; Smith, R.; Tucker, W.J. Earthworms Eisenia fetida recover from Roundup exposure. Appl Soil Ecol 2021, 158, 103793. [Google Scholar] [CrossRef]
- Kanabar, M.; Bauer, S.; Ezedum, Z.M.; Dwyer, I.P.; Moore, W.S.; Rodriguez, G.; Mall, A.; Littleton, A.T.; Yudell, M.; Kanabar, J.; Tucker, W.J. Roundup negatively impacts the behavior and nerve function of the Madagascar hissing cockroach (Gromphadorhina portentosa). Environ Sci Pollut Res 2021, 28, 32933–32944. [Google Scholar] [CrossRef]
- Pochron, S.T.; Mezic, M.; Byrne, S.; Sasoun, S.; Casamassima, A.; Kilic, M.; Nuzzo, A.; Beaudet, C.E. Exposure to Roundup increases movement speed and decreases body mass in earthworms. Front Environ Sci 2021, 1832. [Google Scholar] [CrossRef]
- Levine, S.L.; von Mérey, G.; Minderhout, T.; Manson, P.; Sutton, P. Aminomethylphosphonic acid has low chronic toxicity to Daphnia magna and Pimephales promelas. Environ Toxicol Chem 2015, 34, 1382–1389. [Google Scholar] [CrossRef] [PubMed]
- Tresnakova, N.; Stara, A.; Velisek, J. Effects of glyphosate and its metabolite AMPA on aquatic organisms. Appl Sci-Basel 2021, 11, 9004–9011. [Google Scholar] [CrossRef]
- Medalie, L.; Baker, N.T.; Shoda, M.E.; Stone, W.W.; Meyer, M.T.; Stets, E.G.; Wilson, M. Influence of land use and region on glyphosate and aminomethylphosphonic acid in streams in the USA. Sci Tot Environ 2020, 707, 136008. [Google Scholar] [CrossRef]
- Carretta, L.; Masin, R.; Zanin, G. Review of studies analysing glyphosate and aminomethylphosphonic acid (AMPA) occurrence in groundwater. Environ Rev 2022, 30, 88–109. [Google Scholar] [CrossRef]
- Giesy, J.P.; Dobson, S.; Solomon, K.R. Ecotoxicological risk assessment for Roundup® herbicide; Springer: New York, USA, 2000; pp. 35–120. [Google Scholar]
- Bergström, L.; Börjesson, E.; Stenström, J. Laboratory and lysimeter studies of glyphosate and aminomethylphosphonic acid in a sand and a clay soil. J Environ Qual 2011, 40, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Coupe, R.H.; Kalkhoff, S.J.; Capel, P.D.; Gregoire, C. Fate and transport of glyphosate and aminomethylphosphonic acid in surface waters of agricultural basins. Pest Manag Sci 2012, 68, 16–30. [Google Scholar] [CrossRef]
- Manenti, R.; Barzaghi, B.; Lana, E.; Stocchino, G.A.; Manconi, R.; Lunghi, E. The stenoendemic cave-dwelling planarians (Platyhelminthes, Tricladida) of the Italian Alps and Apennines. J Nat Conserv 2018, 45, 90–97. [Google Scholar] [CrossRef]
- Reynoldson, T.B.; Young, J. A key to the freshwater triclads of Britain and Ireland with notes on their ecology; FBA Scientific Publishing: Cumbria, England, 2000; pp. 28–33. [Google Scholar]
- Melo, A.S.; Andrade, C.F.S. Differential predation of the planarian Dugesia tigrina on two mosquito species under laboratory conditions. J Am Mosquito Contr 2001, 17, 81–83. [Google Scholar]
- Tranchida, M.C.; Pelizza, S.A.; Micieli, M.V.; Maciá, A. Consequences of the introduction of the planarian Girardia anceps (Tricladida: Dugesiidae) in artificial containers with larvae of the mosquitoes Aedes aegypti and Culex pipiens (Diptera: Culicidae) from Argentina. Biol Contr 2014, 71, 49–55. [Google Scholar] [CrossRef]
- Culver, D.C.; Pipan, T. Shallow Subterranean Habitats: Ecology, Evolution, and Conservation; Oxford University Press: Oxford, United Kingdom, 2014; pp. 101–112. [Google Scholar]
- Pestana, J.L.T.; Ofoegbu, P.U. Ecotoxicity Assays Using Freshwater Planarians. In C. M. M. Palmeira, D. P. DeOliveira, & D. J. Dorta (Eds.), TOXICITY ASSESSMENT: Methods and Protocols 2021, (Vol. 2240, pp. 125–137).
- Sheiman, I.M.; Kreshchenko, N.D. Regeneration of planarians: Experimental object. Russ J Dev Biol 2015, 46, 1–9. [Google Scholar] [CrossRef]
- Mangel, M.; Bonsall, M.B.; Aboobaker, A. Feedback control in planarian stem cell systems. BMC Syst Biol 2016, 10, 1–18. [Google Scholar] [CrossRef]
- Byrne, T. Effects of ethanol on negative phototaxis and motility in brown planarians (Dugesia tigrina). Neurosci Lett 2018, 685, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Gomes, M.P.; dos Santos, M.P.; de Freitas, P.L.; Schafaschek, A.M.; de Barros, E.N.; Kitamura, R.S.A.; Navarro-Silva, M.A. The aquatic macrophyte Salvinia molesta mitigates herbicides (glyphosate and aminomethylphosphonic acid) effects to aquatic invertebrates. Environ Sci Pollut R 2023, 30, 12348–12361. [Google Scholar] [CrossRef]
- de Brito Rodrigues, L., Costa, G.G., Thá, E.L., da Silva, L.R., de Oliveira, R., Leme, D.M., Cestari, M.M., Grisolia, C.K., Valadares, M.C. and de Oliveira, G.A.R., 2019. Impact of the glyphosate-based commercial herbicide, its components and its metabolite AMPA on non-target aquatic organisms. Mutation Research/Genetic Toxicology and Environmental Mutagenesis, 842, pp.94-101. [CrossRef]
- da Silva Santos, J.; da Silva Pontes, M.; Grillo, R.; Fiorucci, A.R.; de Arruda, G.J.; Santiago, E.F. Physiological mechanisms and phytoremediation potential of the macrophyte Salvinia biloba towards a commercial formulation and an analytical standard of glyphosate. Chemosphere 2020, 259, 127417. [Google Scholar] [CrossRef]
- Struger, J.; Thompson, D.; Staznik, B.; Martin, P.; McDaniel, T.; Marvin, C. Occurrence of glyphosate in surface waters of southern Ontario. B Environ Contamin Tox 2008, 80, 378–384. [Google Scholar] [CrossRef]
- Domínguez, A.; Brown, G. G.; Sautter, K. D.; Ribas de Oliveira, C. M.; de Vasconcelos, E. C.; Niva, C. C.; Bedano, J.C. Toxicity of AMPA to the earthworm Eisenia andrei Bouché, 1972 in tropical artificial soil. Sci Rep-UK 2019, 6, 19731. [Google Scholar] [CrossRef] [PubMed]
- Rawls, S.M.; Thomas, T.; Adeola, M.; Patil, T.; Raymondi, N.; Poles, A.; Raffa, R.B. Topiramate antagonizes NMDA- and AMPA-induced seizure-like activity in planarians. Pharmacol Biochem Be 2009, 93, 363–367. [Google Scholar] [CrossRef]
- Raffa, R.B.; Finno, K.E.; Tallarida, C.S.; Rawls, S.M. Topiramate-antagonism of L-glutamate-induced paroxysms in planarians. Eur J Pharmacol 2010, 649, 150–153. [Google Scholar] [CrossRef]
- Gotti, R., Fiori, J., Furlanetto, S., Orlandini, S., Candela, M. and Franzellitti, S., 2022. Assessment of bioaccumulation of glyphosate and aminomethylphosphonic acid in marine mussels using capillary electrophoresis with light-emitting diode-induced fluorescence detection. Journal of Chromatography A, 1681, p.463452. [CrossRef]
- Yan, B., Sun, Y., Fu, K., Zhang, Y., Lei, L., Men, J., Guo, Y., Wu, S., Han, J. and Zhou, B., 2023. Effects of glyphosate exposure on gut–liver axis: Metabolomic and mechanistic analysis in grass carp (Ctenopharyngodon idellus). Science of The Total Environment, 902, p.166062. [CrossRef]
- Druart, C., Millet, M., Scheifler, R., Delhomme, O., Raeppel, C. and De Vaufleury, A., 2011. Snails as indicators of pesticide drift, deposit, transfer and effects in the vineyard. Science of the total environment, 409(20), pp.4280-4288. [CrossRef]
- Pelosi, C., Bertrand, C., Bretagnolle, V., Coeurdassier, M., Delhomme, O., Deschamps, M., Gaba, S., Millet, M., Nélieu, S. and Fritsch, C., 2022. Glyphosate, AMPA and glufosinate in soils and earthworms in a French arable landscape. Chemosphere, 301, p.134672. [CrossRef]
- Witchley, J.N.; Mayer, M.; Wagner, D.E.; Owen, J.H.; Reddien, P.W. Muscle cells provide instructions for planarian regeneration. Cell Rep 2013, 4, 633–641. [Google Scholar] [CrossRef]
- Reddien, P.W. The cellular and molecular basis for planarian regeneration. Cell 2018, 175, 327–345. [Google Scholar] [CrossRef] [PubMed]
- Petersen, C.P.; Reddien, P.W. A wound-induced Wnt expression program controls planarian regeneration polarity. PNAS 2009, 106, 17061–17066. [Google Scholar] [CrossRef] [PubMed]
- Gurley, K.A.; Elliott, S.A.; Simakov, O.; Schmidt, H.A.; Holstein, T.W.; Alvarado, A.S. Expression of secreted Wnt pathway components reveals unexpected complexity of the planarian amputation response. Dev Biol 2010, 24–39. [Google Scholar] [CrossRef]
- Zhang, X.; Cheong, S.M.; Amado, N.G.; Reis, A.H.; MacDonald, B.T.; Zebisch, M.; Jones, E.Y.; Abreu, J.G.; He, X. Notum is required for neural and head induction via Wnt deacylation, oxidation, and inactivation. Dev Cell 2015, 32, 719–730. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



