Submitted:
15 November 2023
Posted:
15 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
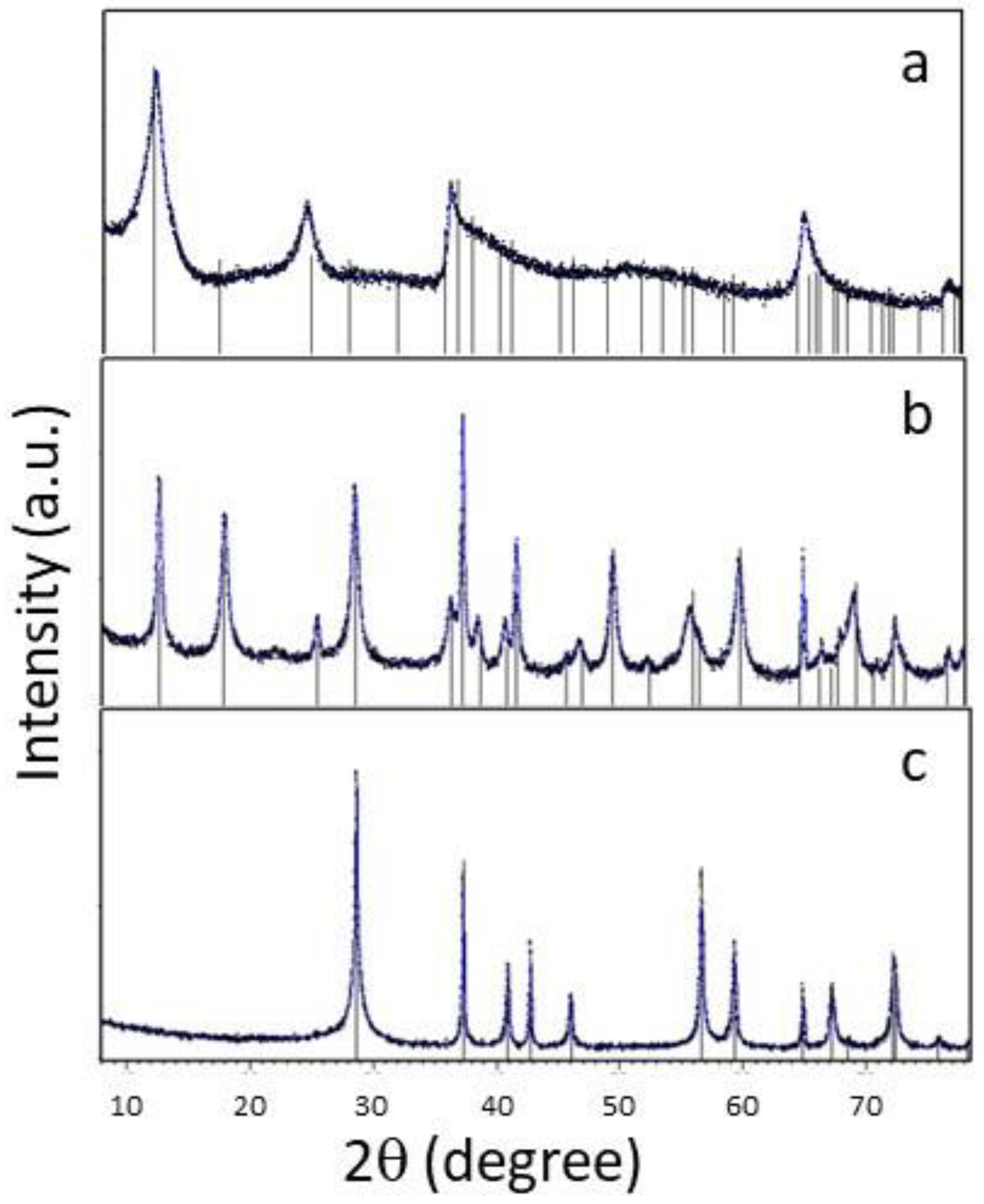
2.1. Synthesis and characterization of MnO2
- 0.627 g of KMnO4 were dissolved in 56 mL of deionized water with continuous magnetic stirring. Once the dissolution is homogeneous, 1.4 mL of HCl was added. After 15 min the solution was transferred to a 100 mL autoclave and place it in an oven maintained at 80 °C for 12 h. This sample is referred to as S80 [18].
- The following synthesis was conducted by dissolving 0.363 g of KMnO4 in 80 mL of deionized water. After achieving homogeneity in the solution, 0.8 mL of HCl was added. Subsequently, the mixture was subjected to continuous magnetic stirring for 1 hour, following which it was transferred to an autoclave and maintained at 140°C for 12 h. This sample was designated as 'S140' [19].
- The synthesis of the sample named S210 was carried out by adding 45 mL of a 0.60 mol/L solution of MnSO4 dropwise, using a burette, to a beaker containing 28.2 mL of a 0.60 mol/L solution of KMnO4, while maintaining constant magnetic stirring. The mixture was stirred continuously for 30 min, after which it was transferred to an autoclave and kept in the oven at 210°C for 12 h [20].
2.2. Synthesis of polystyrene
2.2. Experimental setup and conditions for the recovery of microplastics
3. Results and discussion
3.1. Characterization of the MnO2 synthesized
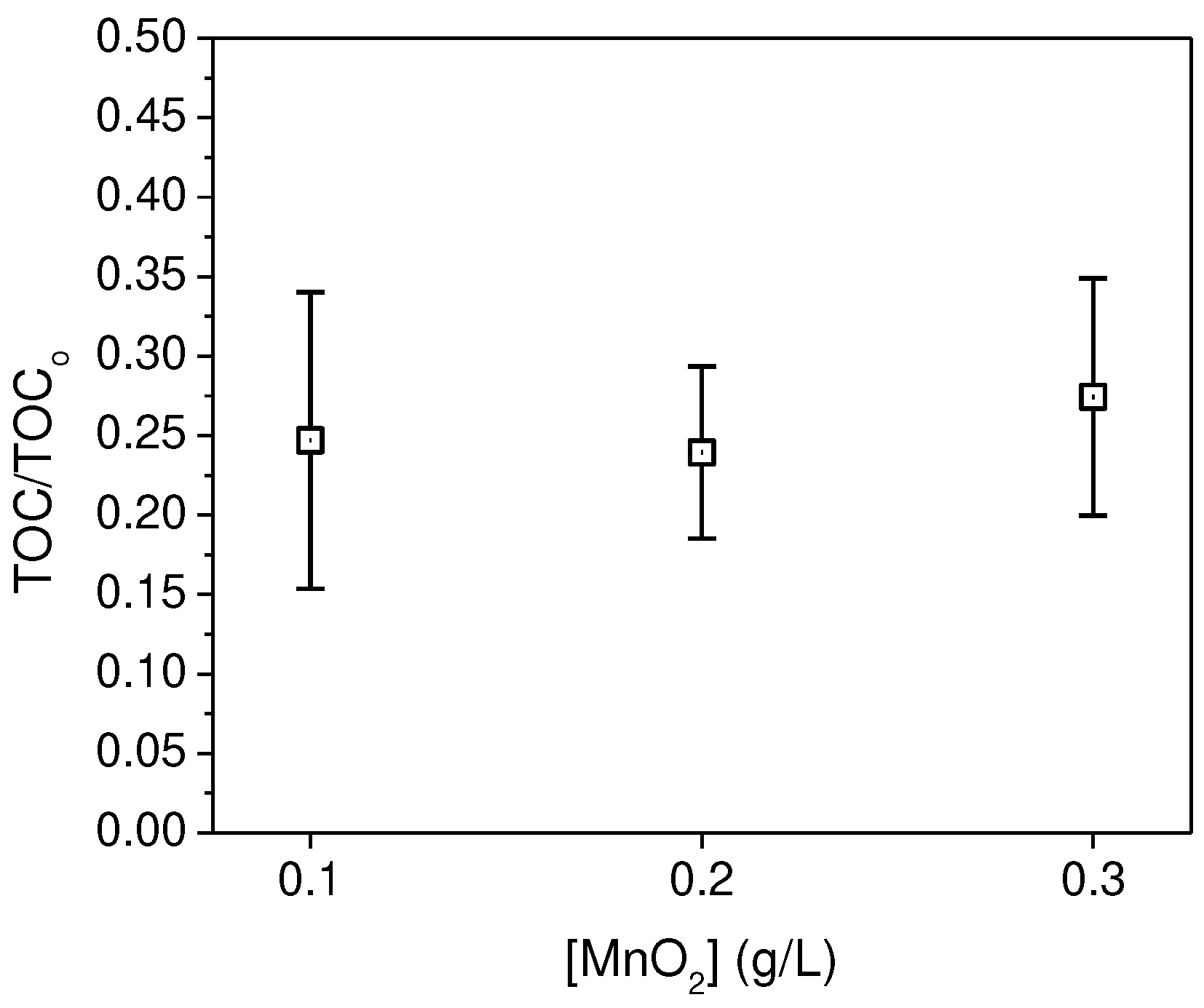
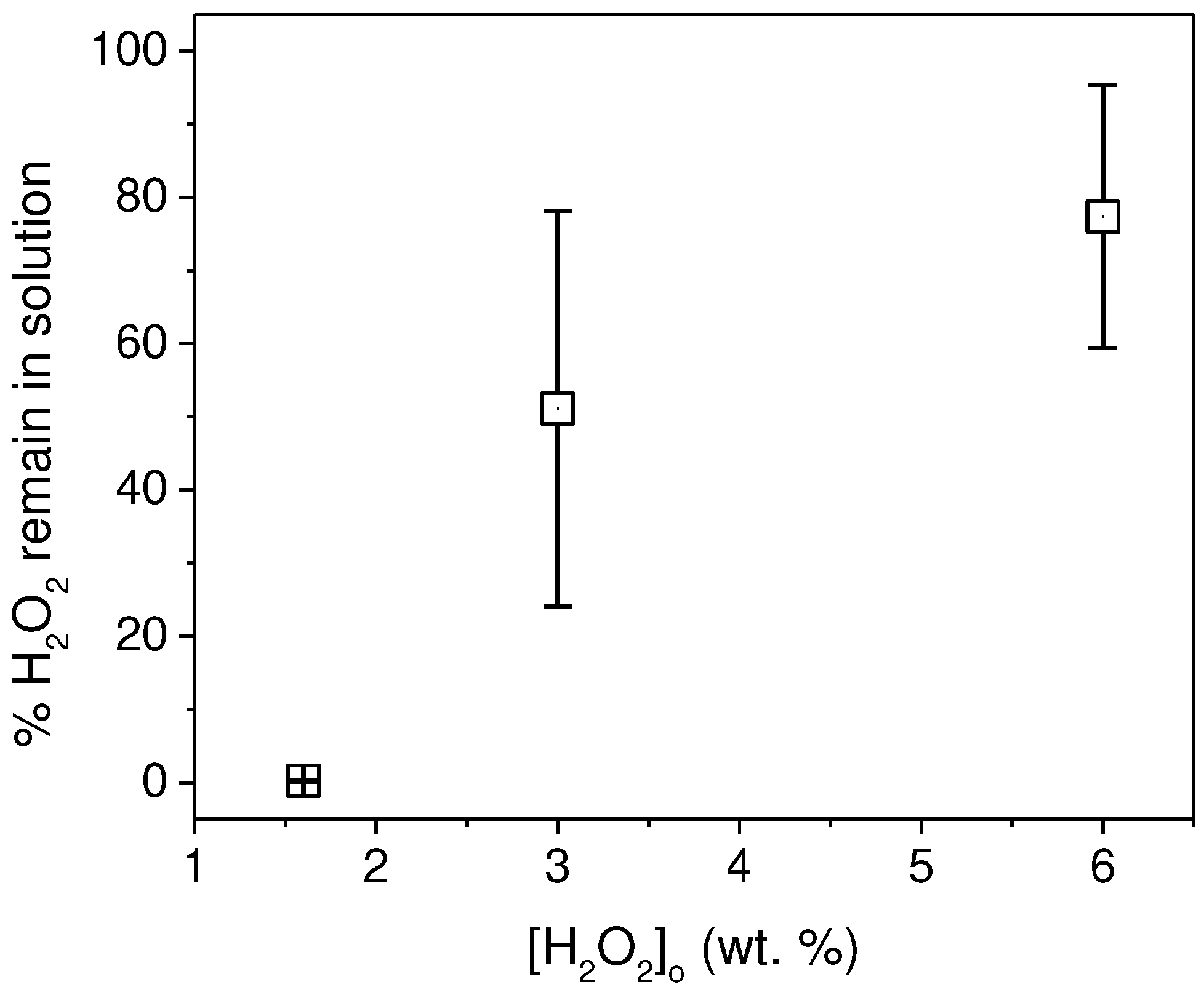
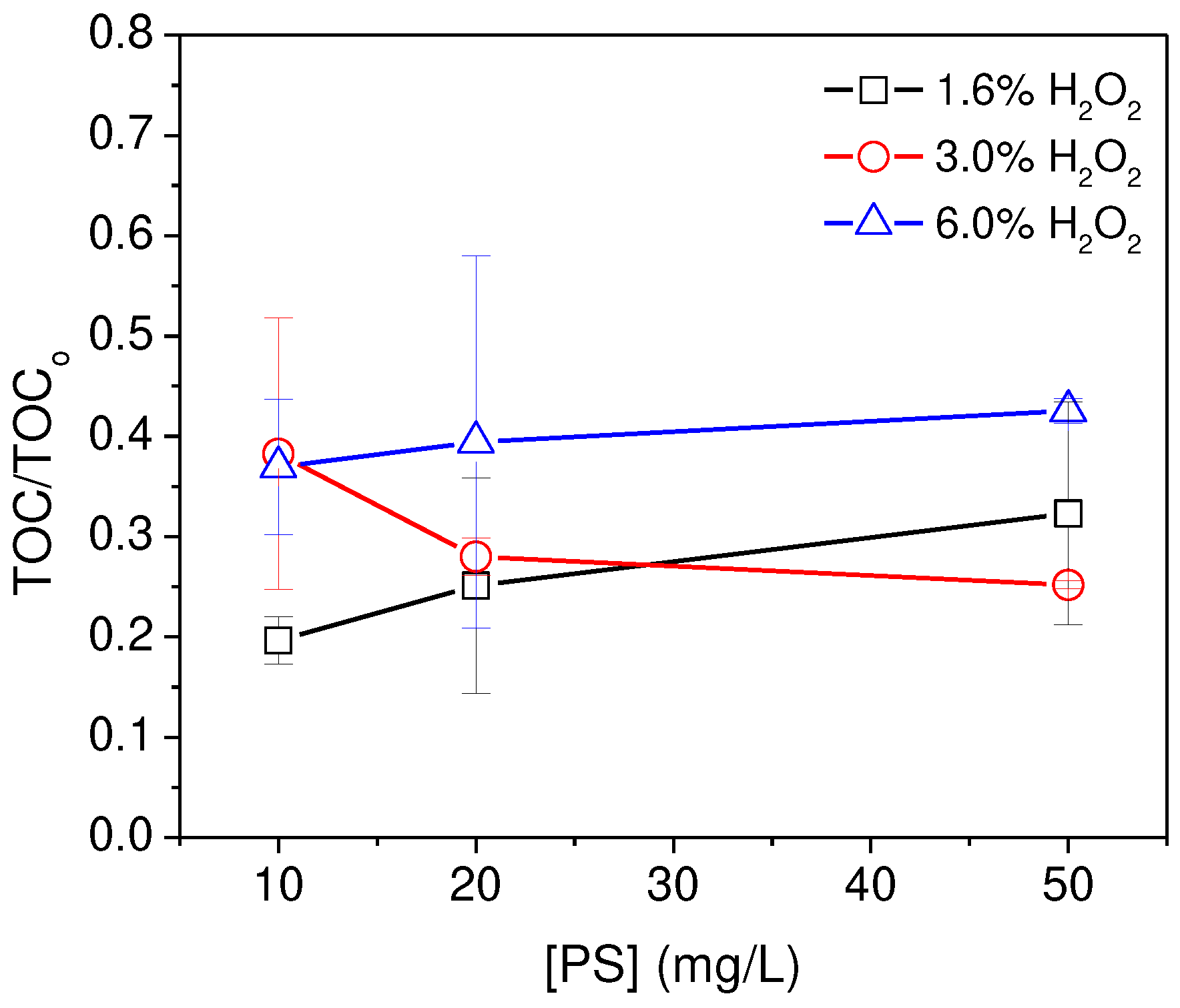
3.2. Recovery of microplastics
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Loganathan, Y.; Kizhakedathil, M.P.J. A review on microplastics-an indelible ubiquitous pollutant. Biointerface Res. Appl. Chem. 2023, 13, 126. [Google Scholar]
- Plastics – the Facts 2022. Available online: https://plasticseurope.org/knowledge-hub/plastics-the-facts-2022/ (accessed on 1 November 2023).
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; et al. Plastic waste inputs from land into the ocean. Science. 2015, 347, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Li, W.C.; Tse, H.F.; Fok, L. Plastic Waste in the Marine Environment: A Review of Sources, Occurrence and Effects. Sci. Total Environ. 2016, 566, 333–349. [Google Scholar] [CrossRef]
- Guo, X.; Wang, J. The Chemical Behaviors of Microplastics in Marine Environment: A Review. Mar. Pollut. Bull. 2019, 142, 1–14. [Google Scholar] [CrossRef]
- Tiwari, R.; Azad, N.; Dutta, D.; Yadav, B.R.; Kumar, S. A critical review and future perspective of plastic waste recycling. Sci. Total Environ. 2023, 163433. [Google Scholar] [CrossRef] [PubMed]
- Padervand, M.; Lichtfouse, E.; Robert, D.; Wang, C. Removal of Microplastics from the Environment. A Review. Environmental Chem. Lett. 2020, 18, 807–828. [Google Scholar] [CrossRef]
- Liu, Q.; Duan, X.; Sun, H.; Wang, Y.; et al. Size-tailored porous spheres of manganese oxides for catalytic oxidation via peroxymonosulfate activation. J. Phys. Chem. C. 2016, 120, 16871–16878. [Google Scholar] [CrossRef]
- Gunatilake, U.B.; Morales, R.; Basabe-Desmonts, L.; Benito-Lopez, F. Magneto Twister: Magneto Deformation of the Water–Air Interface by a Superhydrophobic Magnetic Nanoparticle Layer. Langmuir. 2022, 38, 3360–3369. [Google Scholar] [CrossRef]
- Ye, H.; Wang, Y.; Liu, X.; Xu, D.; et al. Magnetically steerable iron oxides-manganese dioxide core–shell micromotors for organic and microplastic removals. J. Colloid Interface Sci. 2021, 588, 510–521. [Google Scholar] [CrossRef] [PubMed]
- Schwaminger, S.P.; Schwarzenberger, K.; Gatzemeier, J.; Lei, Z.; Eckert, K. Magnetically induced aggregation of iron oxide nanoparticles for carrier flotation strategies. ACS Appl. Mater. Interfaces. 2021, 13, 20830–20844. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, J.; Liu, Z.; Tian, S.; et al. Coagulation removal of microplastics from wastewater by magnetic magnesium hydroxide and PAM. J. Water Proc. Eng. 2021, 43, 102250. [Google Scholar] [CrossRef]
- Villa, K.; Děkanovský, L.; Plutnar, J.; Kosina, J.; Pumera, M. Swarming of perovskite-like Bi2WO6 microrobots destroy textile fibers under visible light. Adv. Funct. Mater. 2020, 30, 2007073. [Google Scholar] [CrossRef]
- Misra, A. , Zambrzycki, C., Kloker, G., Kotyrba, A., et al. Water purification and microplastics removal using magnetic polyoxometalate-supported ionic liquid phases (magPOM-SILPs). Angew. Chem. Int. Ed. 2020, 59, 1601–1605. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Chen, W.; Fan, X.; Tian, C.; et al. Cooperative recyclable magnetic microsubmarines for oil and microplastics removal from water. Appl. Mater. Today. 2020, 20, 100682. [Google Scholar] [CrossRef]
- Kang, J.; Zhou, L.; Duan, X.; Sun, H.; et al. Degradation of cosmetic microplastics via functionalized carbon nanosprings. Matter. 2019, 1, 745–758. [Google Scholar] [CrossRef]
- Wang, L.; Kaeppler, A.; Fischer, D.; Simmchen, J. Photocatalytic TiO2 micromotors for removal of microplastics and suspended matter. ACS Appl. Mater. Interfaces. 2019, 11, 32937–32944. [Google Scholar] [CrossRef] [PubMed]
- Safdar, M.; Minh, T.D.; Kinnunen, N.; Jänis, J. Manganese Oxide Based Catalytic Micromotors: Effect of Polymorphism on Motion. ACS Appl. Mater. Interfaces. 2016, 8, 32624–32629. [Google Scholar] [CrossRef] [PubMed]
- Van Nguyen, T.; Phan, N.M.; Kim, K. J.; Huy, N.N.; Dung, N.T. Orange G degradation by heterogeneous peroxymonosulfate activation based on magnetic MnFe2O4/α-MnO2 hybrid. J. Environ. Sci. 2023, 124, 379–396. [Google Scholar]
- Wan, J.; Zhou, L.; Deng, H.; Zhan, F.; Zhang, R. Oxidative degradation of sulfamethoxazole by different MnO2 nanocrystals in aqueous solution. J. Mol. Catal. A Chem. 2015, 407, 67–74. [Google Scholar] [CrossRef]
- Lu, S.; Zhu, K.; Song, W.; Song, G.; et al. Impact of water chemistry on surface charge and aggregation of polystyrene microspheres suspensions Sci. Total Environ. 2018, 630, 951–959. [Google Scholar] [CrossRef]
- Eisenberg, G. Colorimetric determination of hydrogen peroxide. Ind. Eng. Chem. Anal. Ed. 1943, 15, 327–328. [Google Scholar] [CrossRef]
- Devaraj, S.; Munichandraiah, N. Effect of crystallographic structure of MnO2 on its electrochemical capacitance properties. J. Phys. Chem. C. 2008, 112, 4406–4417. [Google Scholar] [CrossRef]

| MPs characteristics | Material | Mechanism | Operating conditions | Results | Reference |
| PS with a diameter 0.5 – 1.0 mm | Hydrophobic Fe3O4 particles | Magnetic guiding forming hydrophobic twister | ● 4 mg of Fe3O4 in a petri dish with height 8.0 ± 0.5 mm ● Twister speed 47.8 mm/s ● 105 mT of magnetic field |
● Efficient qualitative removal for the capture of floating PS particles | [9] |
| MPs extracted from Nivea brand facial cleanser using a 0.8 μm filter. | Fe2O3-MnO2 with 4 μm in diameter c.a. with a spherical shape | Adsorptive bubble separation (ABS) | ● [Catalyst] = 0.3 g/L ● [MPs] = 5 g/L ● 0.01 % Triton-X-100 as surfactant ● 5 % H2O2 ● t = 0.5–6 h |
● Removal rate of 10 % after 2 h of reaction | [10] |
| PMMA sphere particles (diameter between 20 and 50 μm) | Oleic acid coated Fe3O4 particles with 10 nm in diameter c.a. | Carrier flotation magnetically induced | ● [Fe3O4] = 2 g/L ● [NaCl] = 0–10 mol/L ● pH = 7 |
● Magnet-induced aggregation that can be collected with the assistance of permanent magnets | [11] |
| PE ≤ 270 μm | Magnetic magnesium hydroxide (Mg(OH)2 with Fe3O4 57 – 147 μm) and non-ionic polyacrylamide (PAM) | Coagulation | ● [Mg(OH)2] = 50–250 mg/L ● [Fe3O4] = 40–200 mg/L ● [PAM] = 0–5 mg/L ● [MPs] = 0.05 g/L |
● Removal efficiency of 87.1% when Mg2+:OH– was 1:1 ● Removal efficiency of 87.1% when accompanied by PAM |
[12] |
| Textile fibers obtained from commercial wet wipes with a diameter ≈ 13 μm | Sphere-like Bi2WO6 particles with 6.9 μm in diameter c.a. | Degradation | ● [MNMs] = 1 g/L ● A piece of wipe was added to the essays ● 300 W high-pressure UV-vis lamp ● t = 50 h |
● Partial degradation (unquantified) | [13] |
| PS beads (diameter 1 mm and 10 mm) | Polyoxometalate ionic liquid adsorbed onto magnetic microporous core-shell Fe2O3/ SiO2 particles (magPOM-SILP) |
Removing by surface-binding | ● [magPOM-SILP] = 10 g/L ● [MPs] = 1 g/L ● t = 24 h |
● Removal efficiencies over 90% | [14] |
| PS spheres with 100 μm or 40 μm in diameter, and MP extracted from face cleansing cream sample | Magnetic sunflower pollen grains with 30 μm in diameter c.a. | Shoveling | ● [MNMs] Unspecified ● [MPs] = 40 – 80 pieces / 11 – 15 μL. ● Conteo de piezas con ayuda de un microscopio de luz |
● Removal effectiveness of 75% for the microplastics obtained from the facial cream and 70% for the PS microplastics | [15] |
| Cosmetic microplastics obtained by commercial facial cleansers (0.01 – 1.5 mm) | Magnetic N-doped nanocarbon springs with 3–5 mm in length and 20–40 nm in diameter | Degradation | ● [Catalyst] = 0.2 g/L ● [MPs] = 5 g/L ● [PMS] = 6.5 mM ● T = 100–160 °C ● t = 0 – 8 h |
● Activation of peroxymonosulfate (PMS) to evolve reactive radicals reaching 50 % weight loss | [16] |
| Carboxylated PS Bead with 3 μm in diameter | Photocatalytic Au@Ni@TiO2 (< 1 μm in diameter) chains | Phoretic interaction | ● [MNMs] = 2.5 g/L ● [MPs] = 1 g/L ● 0.10 – 1.67 % H2O2 |
● Removal of 77% after 120 s of reaction with 0.10% H2O2 and 63 mW UV ligh | [17] |
| Sample | SBET (m2/g) | SMP (m2/g) | SEXT (m2/g) | VMP (cm3/g) | VT (cm3/g) | Mesoporous size (nm) | Microporous size (nm) |
| δ-MnO2 S80 | 57 | 23 | 34 | 0.011 | 0.160 | 9.3 | 1.1 |
| α-MnO2 S150 | 17 | 4 | 13 | 0.005 | 0.049 | - | 1.1 |
| β-MnO2 S210 | 13 | 4 | 9 | 0.003 | 0.011 | 11.7 | 1.1, 1.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




