Submitted:
27 September 2023
Posted:
27 September 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Subjects
2.3. Clinical data
2.4. Endpoints
2.5. Statistical analysis
3. Results
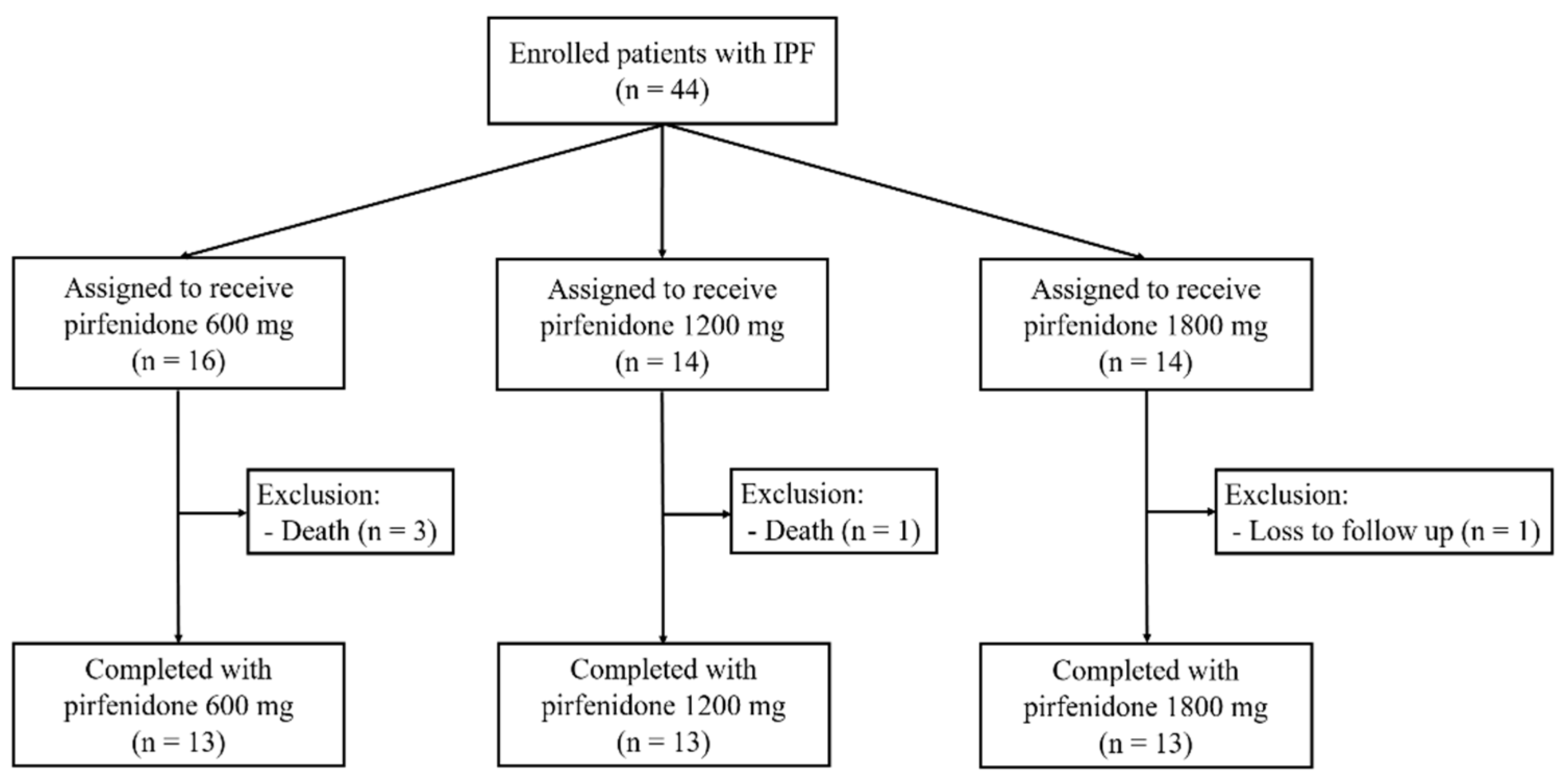
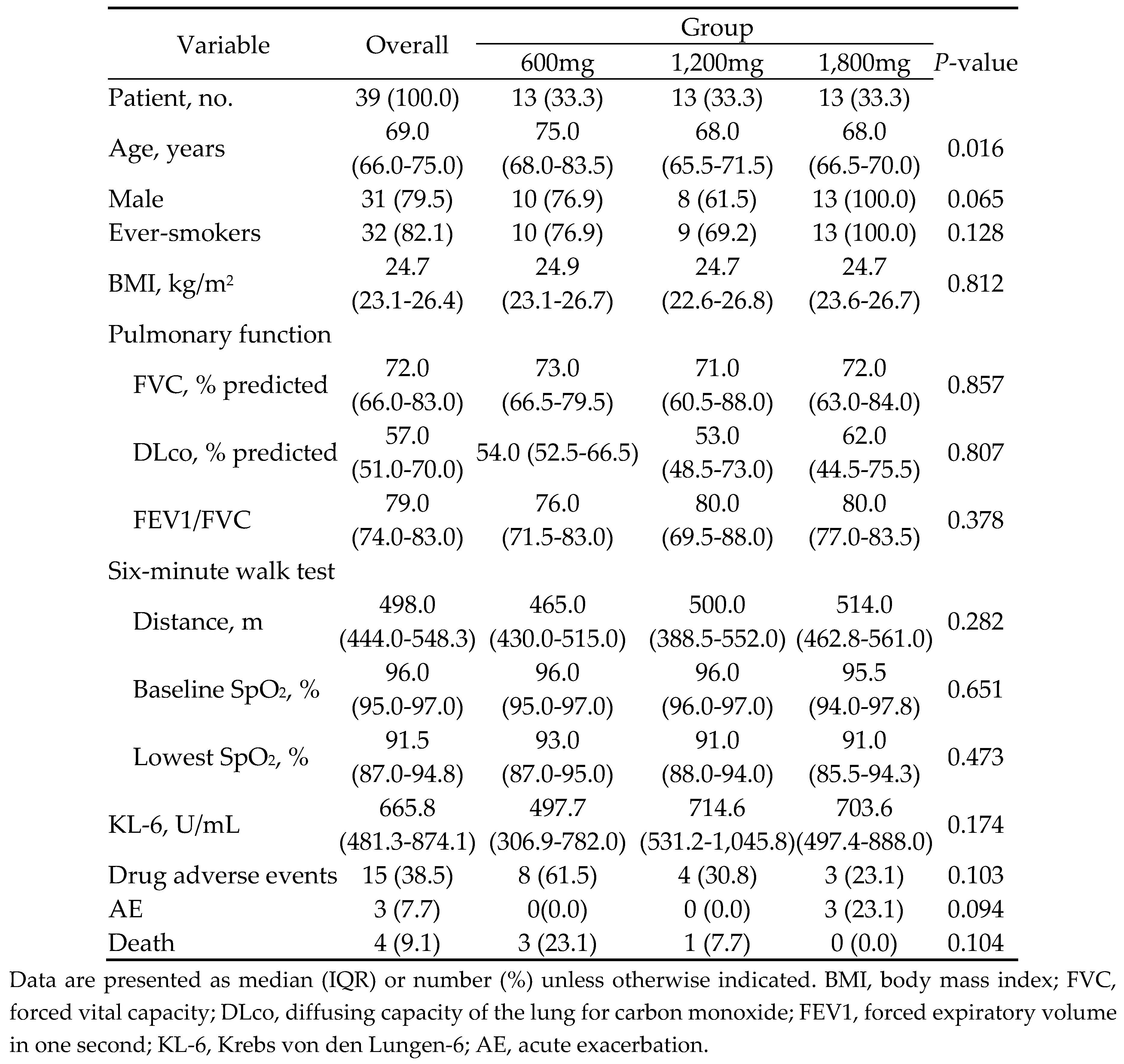
3.1. Study population and baseline characteristics
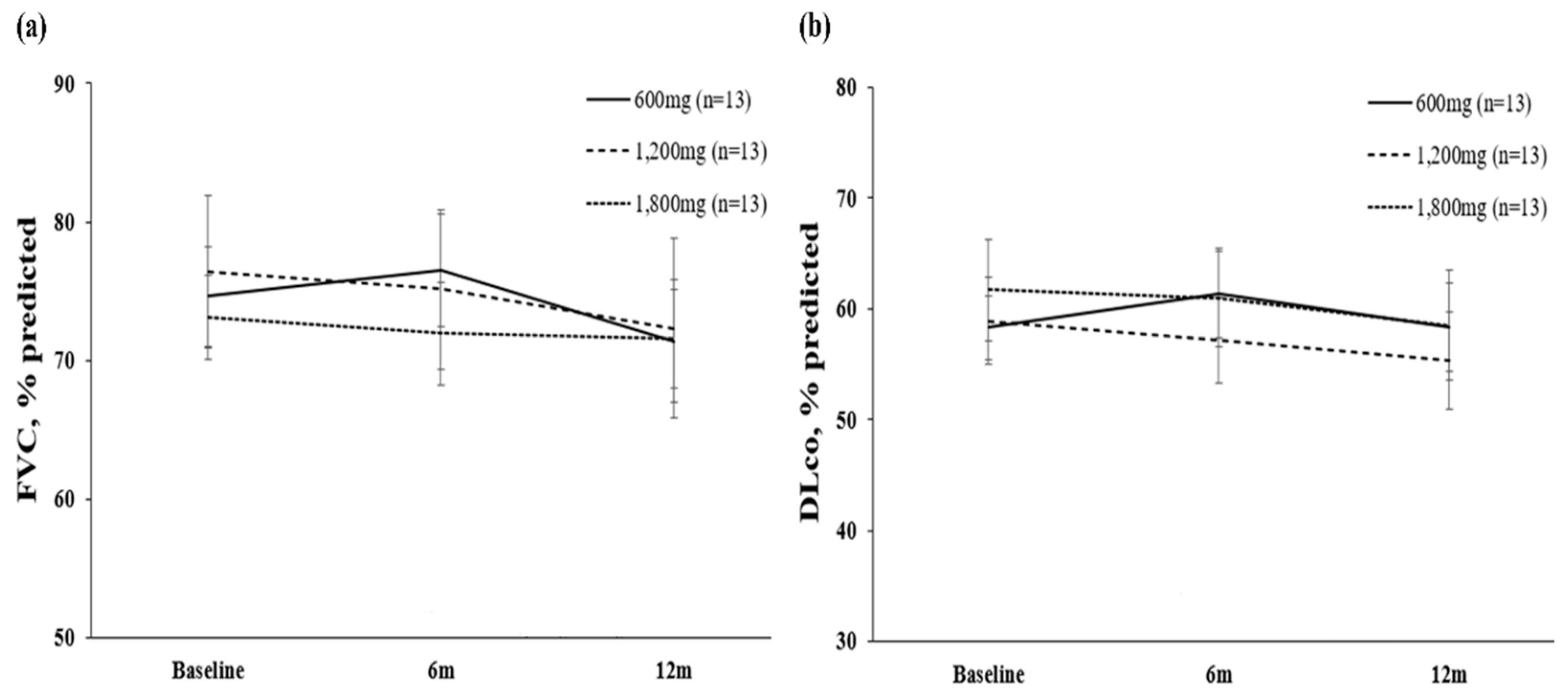
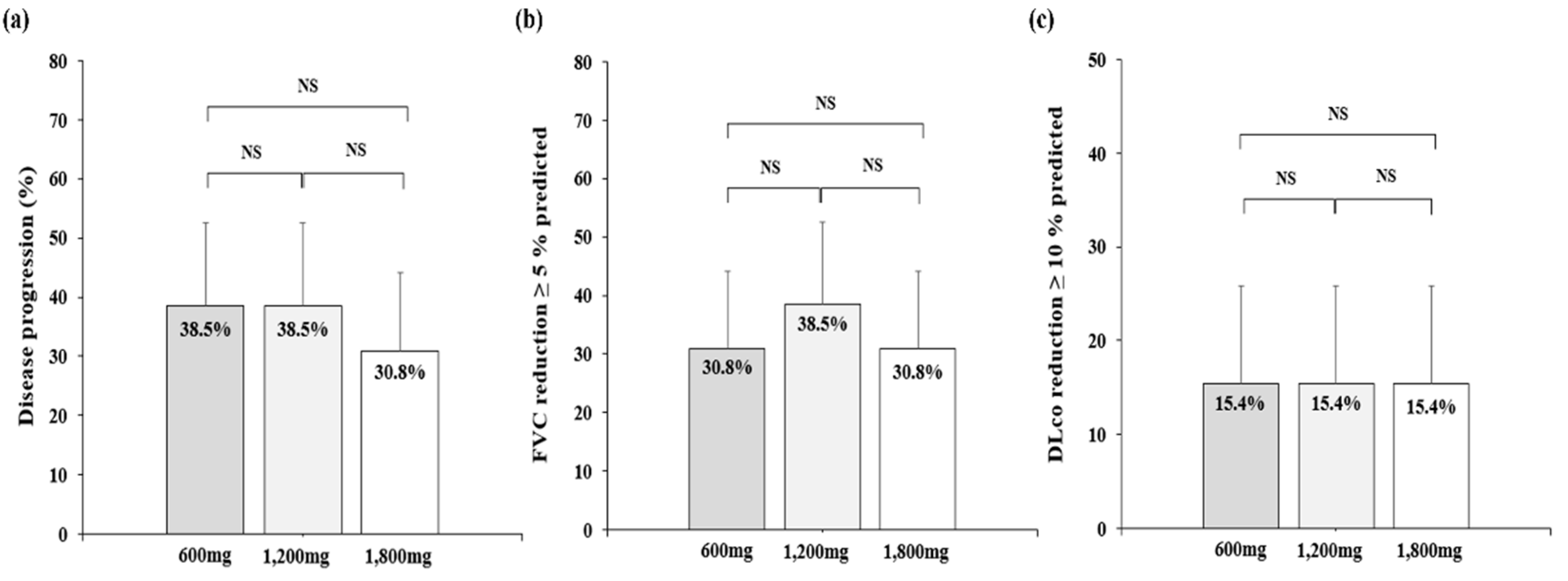
3.2. Primary endpoint
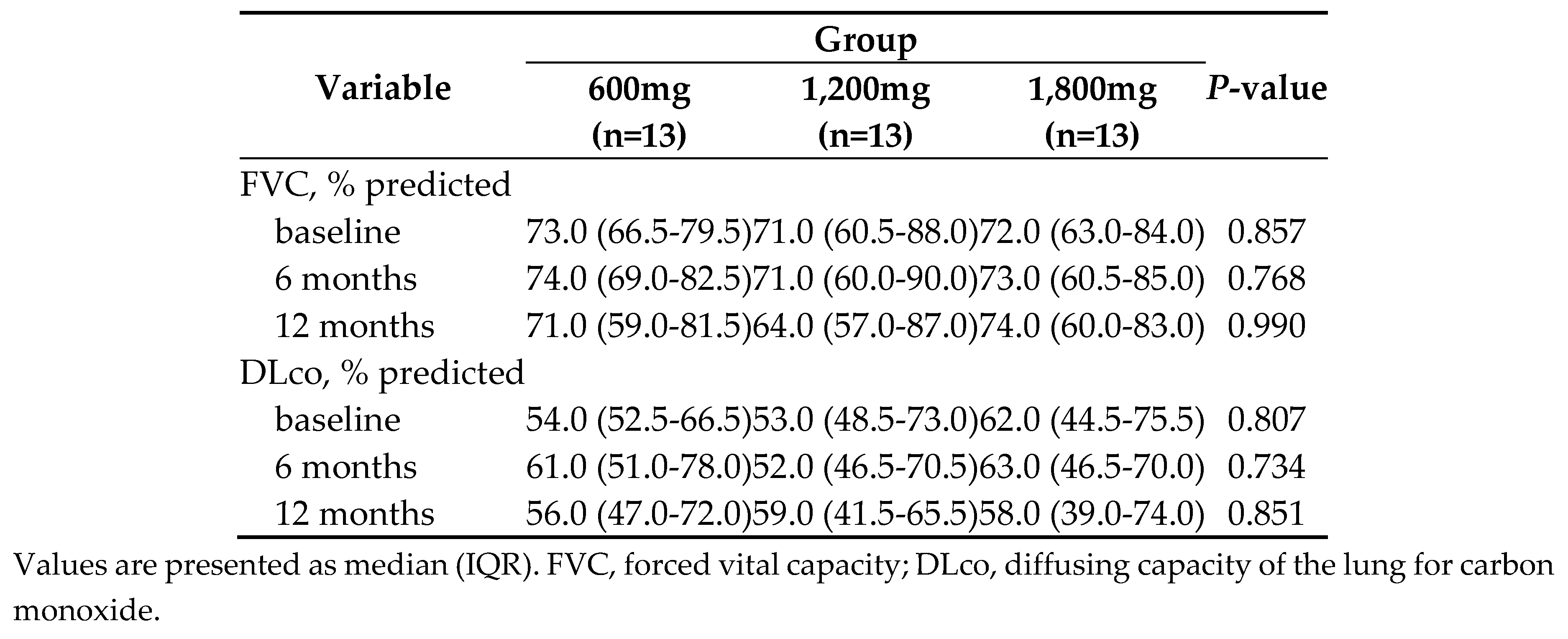
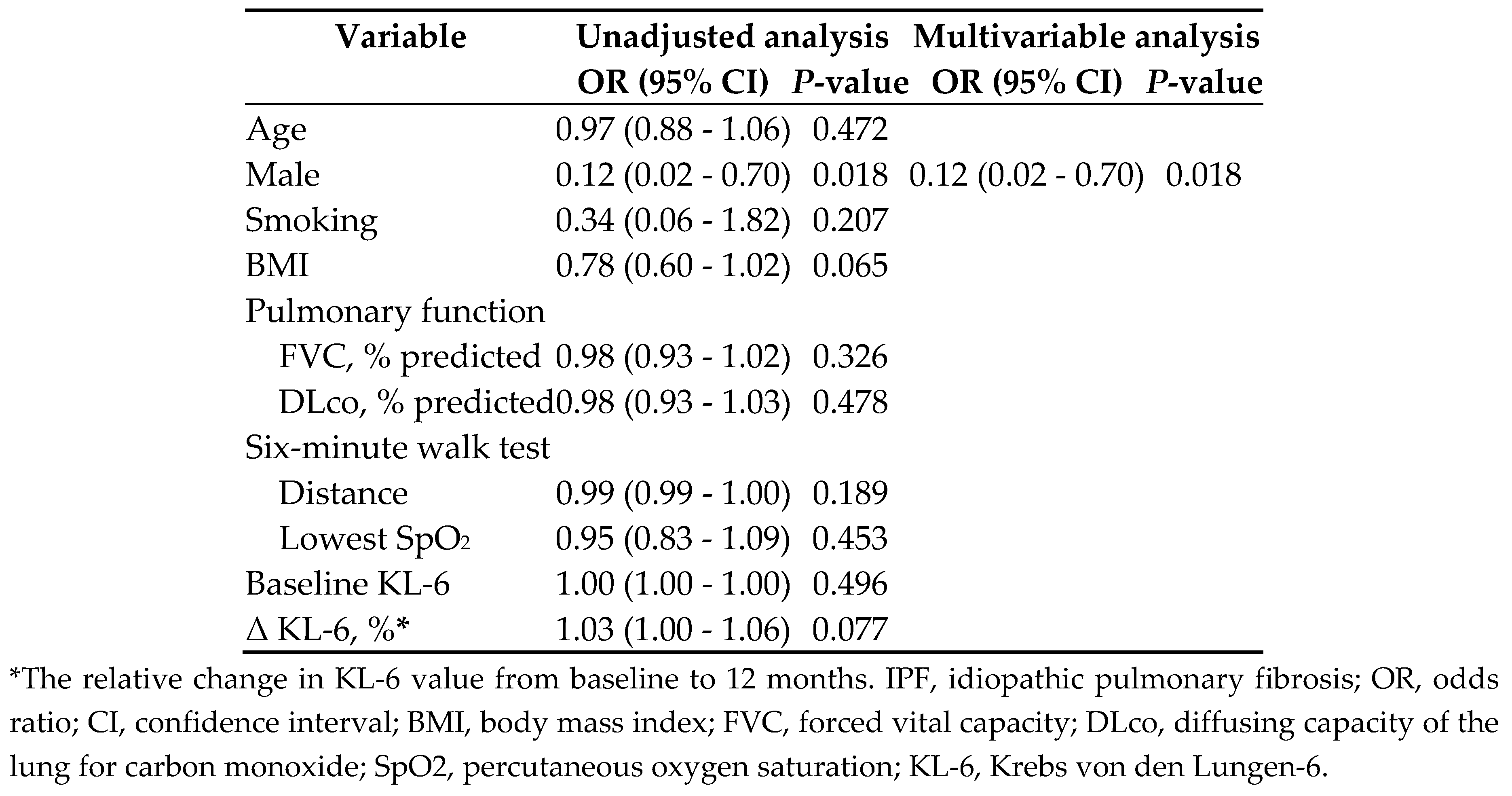
3.2. Secondary endpoint
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Travis, W.D.; Costabel, U.; Hansell, D.M.; King, T.E., Jr.; Lynch, D.A.; Nicholson, A.G.; Ryerson, C.J.; Ryu, J.H.; Selman, M.; Wells, A.U.; et al. An official American Thoracic Society/European Respiratory Society statement: Update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am J Respir Crit Care Med 2013, 188, 733–748. [Google Scholar] [CrossRef] [PubMed]
- American Thoracic Society. Idiopathic pulmonary fibrosis: diagnosis and treatment. International consensus statement. American Thoracic Society (ATS), and the European Respiratory Society (ERS). Am J Respir Crit Care Med 2000, 161, 646–664. [Google Scholar] [CrossRef] [PubMed]
- King, T.E., Jr.; Bradford, W.Z.; Castro-Bernardini, S.; Fagan, E.A.; Glaspole, I.; Glassberg, M.K.; Gorina, E.; Hopkins, P.M.; Kardatzke, D.; Lancaster, L.; et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med 2014, 370, 2083–2092. [Google Scholar] [CrossRef] [PubMed]
- Noble, P.W.; Albera, C.; Bradford, W.Z.; Costabel, U.; Glassberg, M.K.; Kardatzke, D.; King, T.E., Jr.; Lancaster, L.; Sahn, S.A.; Szwarcberg, J.; et al. Pirfenidone in patients with idiopathic pulmonary fibrosis (CAPACITY): two randomised trials. Lancet 2011, 377, 1760–1769. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.P.; Park, M.S.; Oh, I.J.; Lee, H.B.; Kim, Y.W.; Park, J.S.; Uh, S.T.; Kim, Y.S.; Jegal, Y.; Song, J.W. Safety and Efficacy of Pirfenidone in Advanced Idiopathic Pulmonary Fibrosis: A Nationwide Post-Marketing Surveillance Study in Korean Patients. Adv Ther 2020, 37, 2303–2316. [Google Scholar] [CrossRef] [PubMed]
- Ogura, T.; Azuma, A.; Inoue, Y.; Taniguchi, H.; Chida, K.; Bando, M.; Niimi, Y.; Kakutani, S.; Suga, M.; Sugiyama, Y.; et al. All-case post-marketing surveillance of 1371 patients treated with pirfenidone for idiopathic pulmonary fibrosis. Respir Investig 2015, 53, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Ito, M.; Niimi, Y.; Nakamura, A.; Kakutani, S.; Yoshida, Y.; Yoshikawa, T.; Board, P.A. Post-marketing surveillance of pirfenidone for idiopathic pulmonary fibrosis in Japan: Interim analysis of 973 patients. Eur Respir J 2012, 40. [Google Scholar]
- Hwang, H.; Lee, J.K.; Choi, S.M.; Lee, Y.J.; Cho, Y.J.; Yoon, H.I.; Lee, J.H.; Lee, C.T.; Kim, Y.W.; Park, J.S. Efficacy of lower dose pirfenidone for idiopathic pulmonary fibrosis in real practice: a retrospective cohort study. Korean J Intern Med 2022, 37, 366–376. [Google Scholar] [CrossRef]
- Lee, E.G.; Lee, T.H.; Hong, Y.; Ryoo, J.; Heo, J.W.; Gil, B.M.; Kang, H.S.; Kwon, S.S.; Kim, Y.H. Effects of low-dose pirfenidone on survival and lung function decline in patients with idiopathic pulmonary fibrosis (IPF): Results from a real-world study. PLoS One 2021, 16, e0261684. [Google Scholar] [CrossRef]
- Song, M.J.; Moon, S.W.; Choi, J.S.; Lee, S.H.; Lee, S.H.; Chung, K.S.; Jung, J.Y.; Kang, Y.A.; Park, M.S.; Kim, Y.S.; et al. Efficacy of low dose pirfenidone in idiopathic pulmonary fibrosis: real world experience from a tertiary university hospital. Sci Rep 2020, 10, 21218. [Google Scholar] [CrossRef]
- Durheim, M.T.; Judy, J.; Bender, S.; Baumer, D.; Lucas, J.; Robinson, S.B.; Mohamedaly, O.; Shah, B.R.; Leonard, T.; Conoscenti, C.S.; et al. In-Hospital Mortality in Patients with Idiopathic Pulmonary Fibrosis: A US Cohort Study. Lung 2019, 197, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Ryerson, C.J.; Vittinghoff, E.; Ley, B.; Lee, J.S.; Mooney, J.J.; Jones, K.D.; Elicker, B.M.; Wolters, P.J.; Koth, L.L.; King, T.E., Jr.; et al. Predicting survival across chronic interstitial lung disease: the ILD-GAP model. Chest 2014, 145, 723–728. [Google Scholar] [CrossRef] [PubMed]
- Raghu, G.; Remy-Jardin, M.; Richeldi, L.; Thomson, C.C.; Inoue, Y.; Johkoh, T.; Kreuter, M.; Lynch, D.A.; Maher, T.M.; Martinez, F.J.; et al. Idiopathic Pulmonary Fibrosis (an Update) and Progressive Pulmonary Fibrosis in Adults: An Official ATS/ERS/JRS/ALAT Clinical Practice Guideline. Am J Respir Crit Care Med 2022, 205, e18–e47. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Shah, B.A.; Chowdhury, B.A. Forced vital capacity in idiopathic pulmonary fibrosis--FDA review of pirfenidone and nintedanib. N Engl J Med 2015, 372, 1189–1191. [Google Scholar] [CrossRef] [PubMed]
- Clynick, B.; Corte, T.J.; Jo, H.E.; Stewart, I.; Glaspole, I.N.; Grainge, C.; Maher, T.M.; Navaratnam, V.; Hubbard, R.; Hopkins, P.M.A.; et al. Biomarker signatures for progressive idiopathic pulmonary fibrosis. Eur Respir J 2022, 59. [Google Scholar] [CrossRef]
- Stainer, A.; Faverio, P.; Busnelli, S.; Catalano, M.; Della Zoppa, M.; Marruchella, A.; Pesci, A.; Luppi, F. Molecular Biomarkers in Idiopathic Pulmonary Fibrosis: State of the Art and Future Directions. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Wakamatsu, K.; Nagata, N.; Kumazoe, H.; Oda, K.; Ishimoto, H.; Yoshimi, M.; Takata, S.; Hamada, M.; Koreeda, Y.; Takakura, K.; et al. Prognostic value of serial serum KL-6 measurements in patients with idiopathic pulmonary fibrosis. Respir Investig 2017, 55, 16–23. [Google Scholar] [CrossRef]
- Ishikawa, N.; Hattori, N.; Yokoyama, A.; Kohno, N. Utility of KL-6/MUC1 in the clinical management of interstitial lung diseases. Respir Investig 2012, 50, 3–13. [Google Scholar] [CrossRef]
- Huang, T.H.; Kuo, C.W.; Chen, C.W.; Tseng, Y.L.; Wu, C.L.; Lin, S.H. Baseline plasma KL-6 level predicts adverse outcomes in patients with idiopathic pulmonary fibrosis receiving nintedanib: a retrospective real-world cohort study. BMC Pulm Med 2021, 21, 165. [Google Scholar] [CrossRef]
- Raghu, G.; Remy-Jardin, M.; Myers, J.L.; Richeldi, L.; Ryerson, C.J.; Lederer, D.J.; Behr, J.; Cottin, V.; Danoff, S.K.; Morell, F.; et al. Diagnosis of Idiopathic Pulmonary Fibrosis. An Official ATS/ERS/JRS/ALAT Clinical Practice Guideline. Am J Respir Crit Care Med 2018, 198, e44–e68. [Google Scholar] [CrossRef]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.; Gustafsson, P.; et al. Standardisation of spirometry. Eur Respir J 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Graham, B.L.; Brusasco, V.; Burgos, F.; Cooper, B.G.; Jensen, R.; Kendrick, A.; MacIntyre, N.R.; Thompson, B.R.; Wanger, J. 2017 ERS/ATS standards for single-breath carbon monoxide uptake in the lung. Eur Respir J 2017, 49. [Google Scholar] [CrossRef]
- MacIntyre, N.; Crapo, R.O.; Viegi, G.; Johnson, D.C.; van der Grinten, C.P.M.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Enright, P.; et al. Standardisation of the single-breath determination of carbon monoxide uptake in the lung. Eur Respir J 2005, 26, 720–735. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.J.; Puhan, M.A.; Andrianopoulos, V.; Hernandes, N.A.; Mitchell, K.E.; Hill, C.J.; Lee, A.L.; Camillo, C.A.; Troosters, T.; Spruit, M.A.; et al. An official systematic review of the European Respiratory Society/American Thoracic Society: measurement properties of field walking tests in chronic respiratory disease. Eur Respir J 2014, 44, 1447–1478. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, H.; Ebina, M.; Kondoh, Y.; Ogura, T.; Azuma, A.; Suga, M.; Taguchi, Y.; Takahashi, H.; Nakata, K.; Sato, A.; et al. Pirfenidone in idiopathic pulmonary fibrosis. Eur Respir J 2010, 35, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Raghu, G.; Johnson, W.C.; Lockhart, D.; Mageto, Y. Treatment of idiopathic pulmonary fibrosis with a new antifibrotic agent, pirfenidone: results of a prospective, open-label Phase II study. Am J Respir Crit Care Med 1999, 159, 1061–1069. [Google Scholar] [CrossRef]
- Costabel, U.; Albera, C.; Lancaster, L.; Hormel, P.; Hulter, H.; Noble, P. Final analysis of RECAP, an open-label extension study of pirfenidone in patients with idiopathic pulmonary fibrosis (IPF). Eur Respir J 2016, 48. [Google Scholar]
- Jehn, L.B.; Costabel, U.; Boerner, E.; Waelscher, J.; Theegarten, D.; Taube, C.; Bonella, F. Serum KL-6 as a Biomarker of Progression at Any Time in Fibrotic Interstitial Lung Disease. J Clin Med 2023, 12. [Google Scholar] [CrossRef]
- Chung, C.; Kim, J.; Cho, H.S.; Kim, H.C. Baseline serum Krebs von den Lungen-6 as a biomarker for the disease progression in idiopathic pulmonary fibrosis. Sci Rep-Uk 2022, 12. [Google Scholar] [CrossRef]
- Jiang, Y.; Luo, Q.; Han, Q.; Huang, J.; Ou, Y.; Chen, M.; Wen, Y.; Mosha, S.S.; Deng, K.; Chen, R. Sequential changes of serum KL-6 predict the progression of interstitial lung disease. J Thorac Dis 2018, 10, 4705–4714. [Google Scholar] [CrossRef]
- Wang, C.Z.; Wang, Q.B.; Liu, T.; Zhu, J.; Zhang, B.K. Krebs von den Lungen-6 (KL-6) as a diagnostic marker for pulmonary fibrosis: A systematic review and meta-analysis. Clin Biochem 2023, 114, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.J.; Park, K.J.; Ko, D.H.; Koo, H.J.; Lee, S.M.; Song, J.W.; Lee, W.; Lee, H.K.; Do, K.H.; Chun, S.; et al. Analytical and Clinical Performance of the Nanopia Krebs von den Lungen 6 Assay in Korean Patients With Interstitial Lung Diseases. Ann Lab Med 2019, 39, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Zurkova, M.; Kriegova, E.; Kolek, V.; Lostakova, V.; Sterclova, M.; Bartos, V.; Doubkova, M.; Binkova, I.; Svoboda, M.; Strenkova, J.; et al. Effect of pirfenidone on lung function decline and survival: 5-yr experience from a real-life IPF cohort from the Czech EMPIRE registry. Respir Res 2019, 20, 16. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




