Submitted:
28 August 2023
Posted:
31 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods and materials
2.1. Chemicals
2.2. Synthesis of oak-nZVI
2.3. Experimental procedure
2.4. Definitive screening design (DSD)
3. Results and Discussion
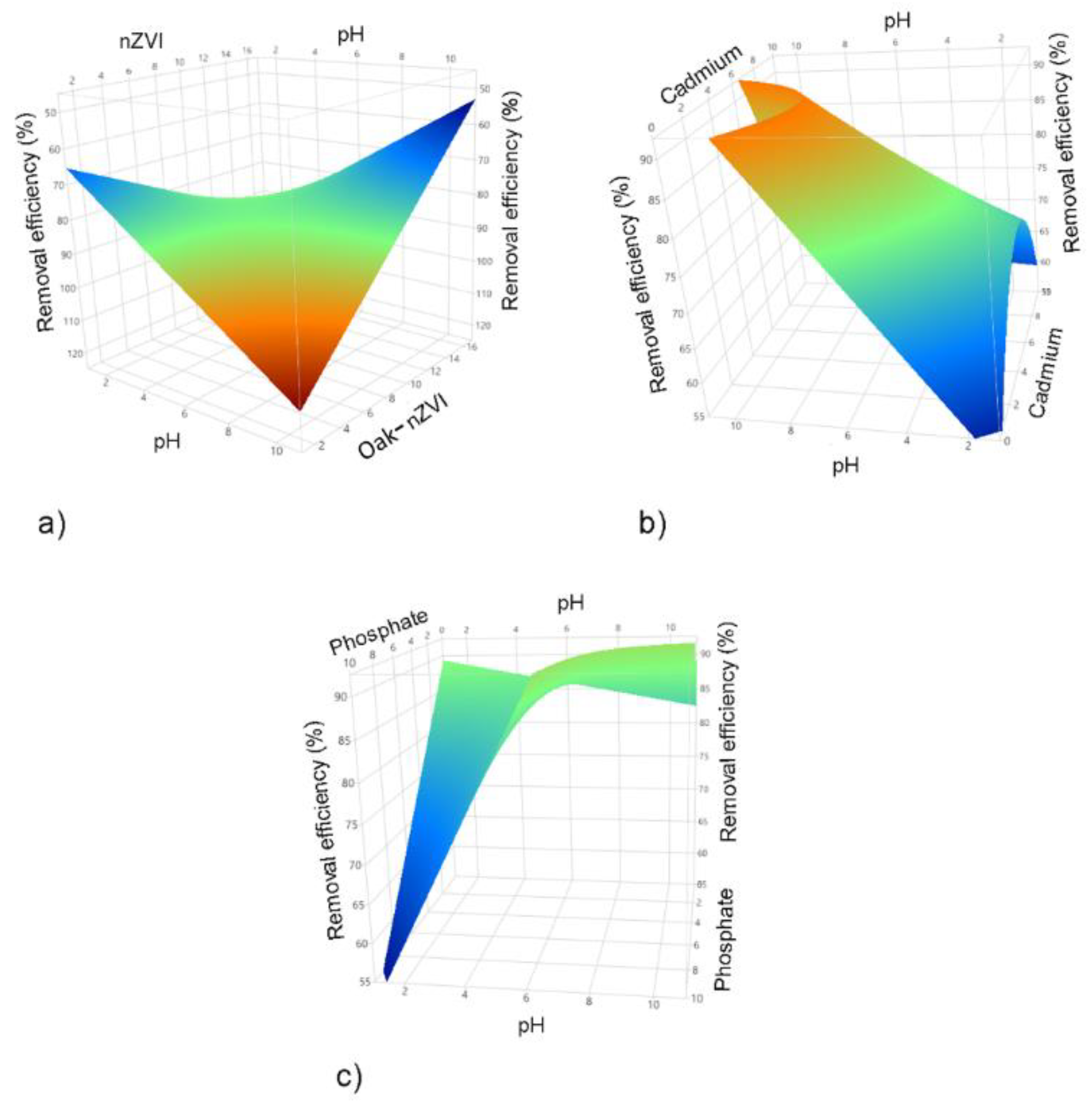
3.1. Statistical analysis for the simultaneous cadmium and phosphate removal
3.1.1. DSD model evaluation - cadmium/phosphate
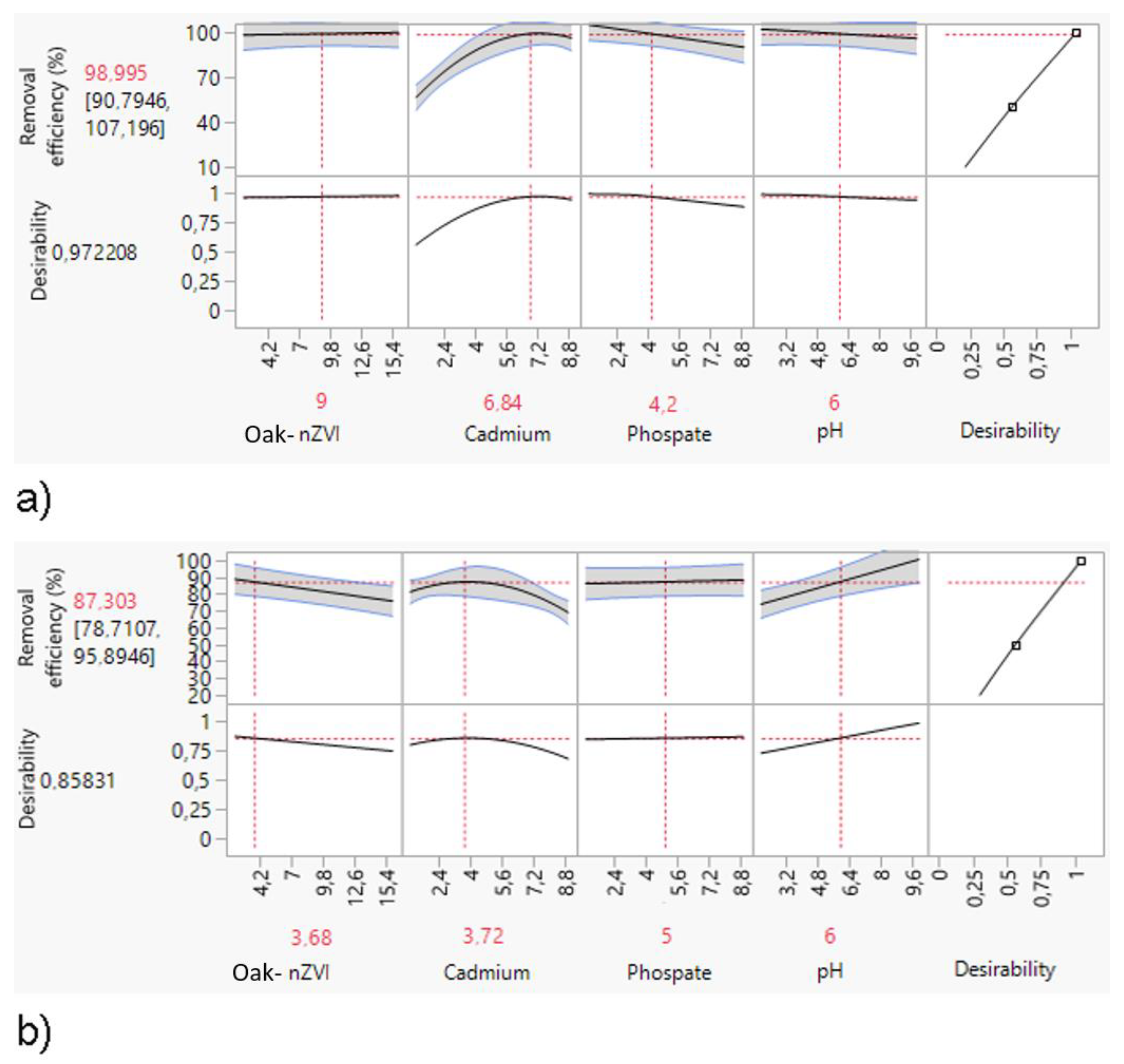
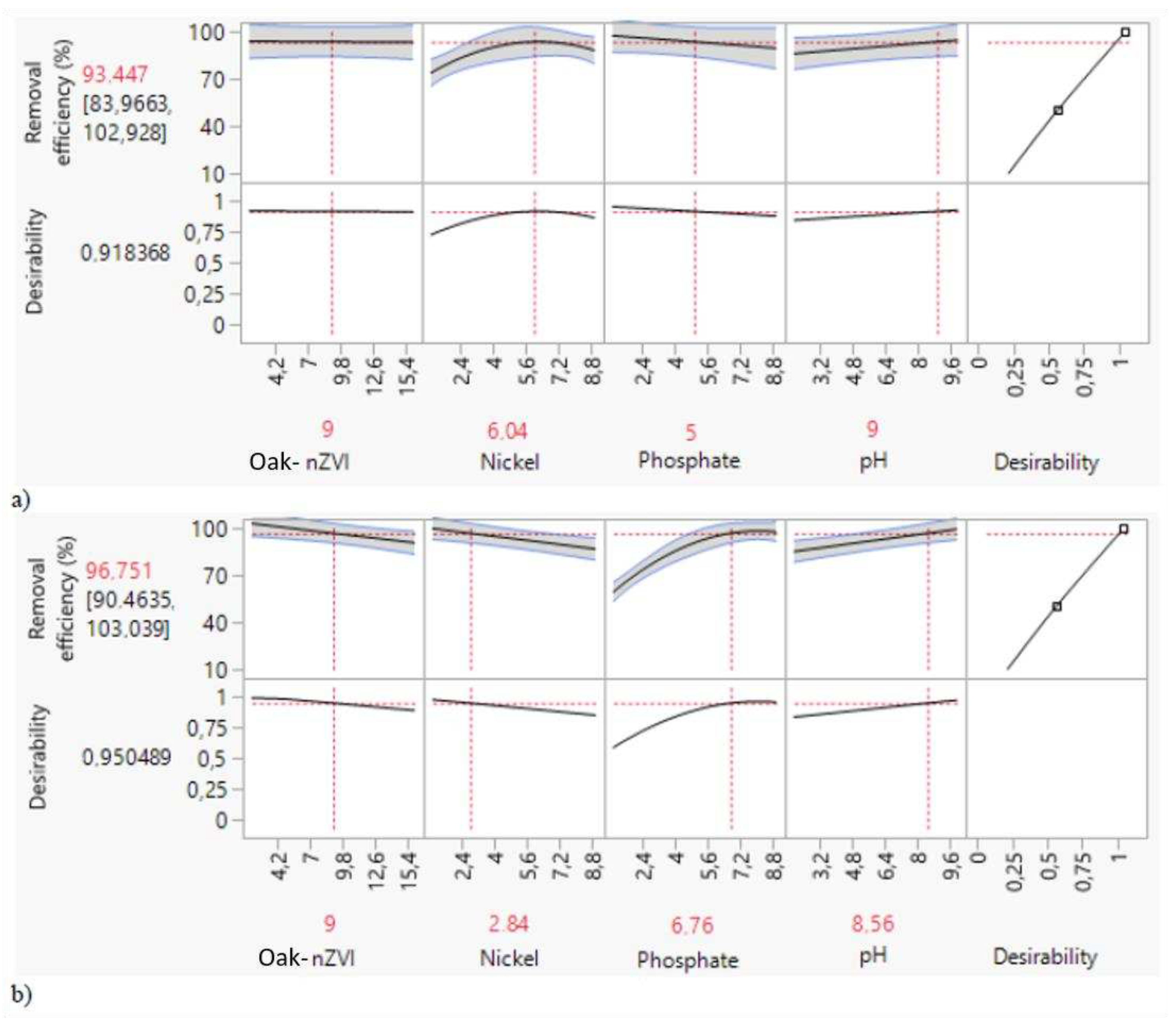
3.1.2. Process optimization of cadmium and phosphate adsorption
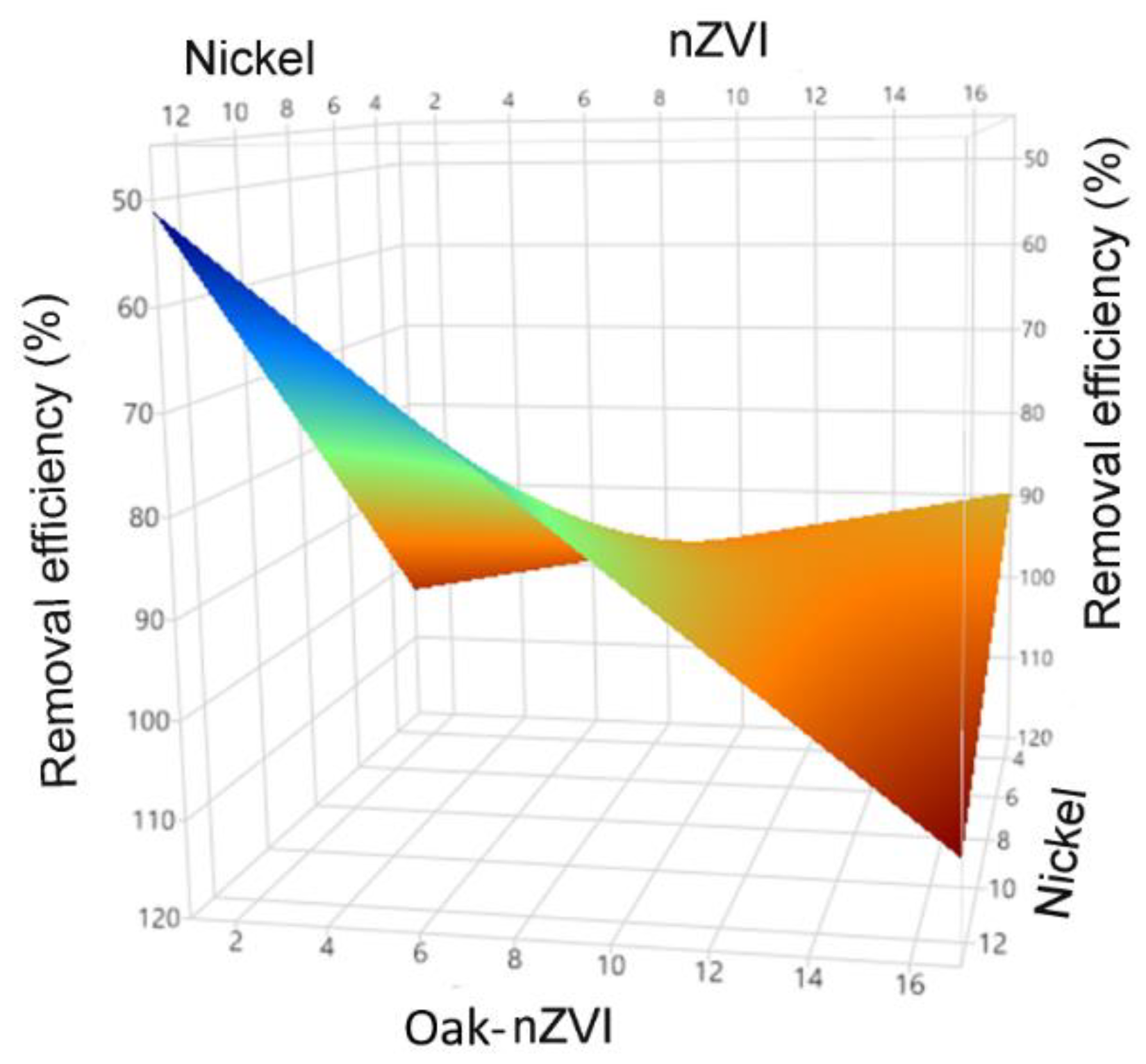
3.2. Statistical analysis for the simultaneous removal of nickel and phosphate
3.2.1. DSD model evaluation - nickel/phosphate
3.2.2. Process optimization of nickel and phosphate adsorption
3.3. Adsorption kinetics
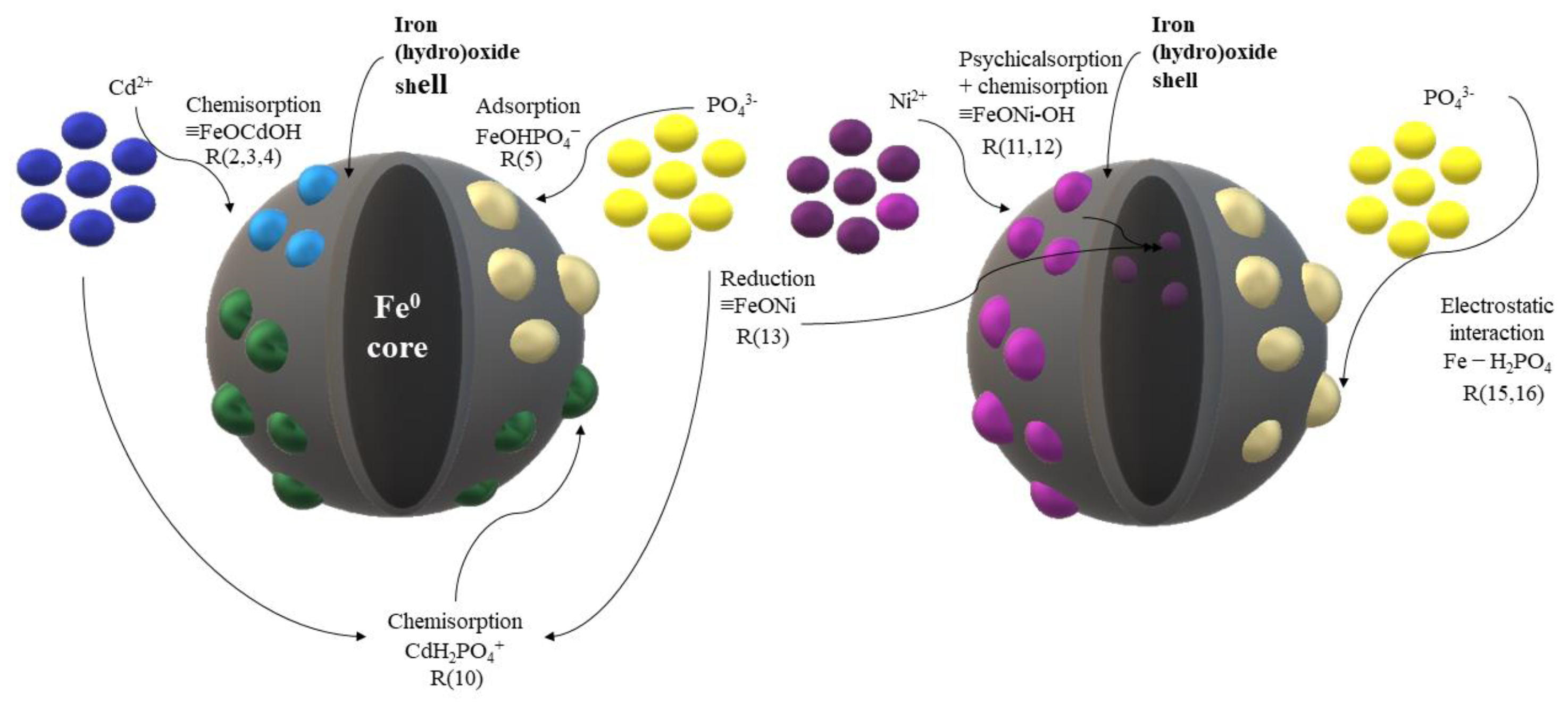
3.4. Interaction mechanisms summary
3.5. Consideration for environmental applicability
4. Conclusion
Supplementary Materials
Acknowledgments
References
- Abdelwaheb, M. , Jebali, K., Dhaouadi, H., Dridi-Dhaouadi, S., 2019. Adsorption of nitrate, phosphate, nickel, and lead on soils: Risk of groundwater contamination. Ecotoxicol. Environ. Saf. 179,182-187. [CrossRef]
- Afridi, M.N. , Lee, W.H., Kim, J.O., 2019. Effect of phosphate concentration, anions, heavy metals, and organic matter on phosphate adsorption from wastewater using anodized iron oxide nanoflakes. Environ. Res. 171, 428–436. [CrossRef]
- Alimohammadi, N. , Shadizadeh, S.R., Kazeminezhad, I., 2013. Removal of cadmium from drilling fluid using nano-adsorbent. Fuel. 111, 505-509. [CrossRef]
- Ayati, M. , Madsen, H.E.L., 2001 Solubility Product of the Cadmium Phosphate Cd5H2(PO4)4·4H2O at 37 °C. J. Chem. Eng. Data. 46,113-116. [CrossRef]
- Bai, L. , Hu, C., Liu H., Qu, J., 2019. Selective adsorption of fluoride from drinking water using NiAl-layered metal oxide film electrode. J. Colloid. Interface Sci. 539, 146-151. [CrossRef]
- Baohong, H. , Lei, S., Li, H., Hongwei, S., 2020. Immobilization of Cd and phosphorus utilization in eutrophic river sediments by biochar-supported nanoscale zero-valent iron, Environ. Technol. [CrossRef]
- Boparai, H.K. , Joseph, M., O'Carroll, D.M., 2011. Kinetics and thermodynamics of cadmium ion removal by adsorption onto nano zerovalent iron particles. J. Hazard. Mater. 186, 458–465. [CrossRef]
- Borja, J.Q. , Ngo, M.A.S., Saranglao, C.C., Tiongco, R.P.M., Roque, E.C., Dugos, N.P., 2015. Synthesis of green zero-valent iron using polyphenols from dried green tea extract, School of Engineering, Taylor’s University.
- Ebrahimi, R. , Hayati, B., Shahmoradi, B., Rezaee, R., Safari, M., Maleki, A., Yetilmezsoy, K., 2018. Adsorptive removal of nickel and lead ions from aqueous solutions by poly(amidoamine) (PAMAM) dendrimers (G4). Environ. Technol. & Inn. 12, 261–272. [CrossRef]
- Efecan, N. , Shahwan, T., Eroğlu, A.E., Lieberwirth, I., 2009. Characterization of the uptake of aqueous Ni2+ ions on nanoparticles of zero-valent iron (nZVI). Desalination. 249, 1048-1054. [CrossRef]
- Fidaledo, M. , Lavecchia, R., Petrucci, E., Zuorro, A., 2016. Application of a novel definitive screening design to decolorization of an azo dye on boron-doped diamond electrodes. Int. J. Environ. Sci. Technol. 13, 835-842. [CrossRef]
- Gasemloo, S. , Khosravi, M., Sohrabi, M.R., Dastmalchi, S., Gharbani, P., 2019. Response surface methodology (RSM) modeling to improve removal of Cr(VI) ions from tannery wastewater using sulfated carboxymethyl cellulose nanofiller. J. Clean. Prod. 208, 736-742. [CrossRef]
- Husson, J.S. , Graber, E.O., Zwieten, L., Taherymoosavi, S., Thomas, T., Nielsen, S., Ye, J., Pan, G., Chia, C., Munroe, P., Allen, J., Lin, Y., Fan, X., Donne, S., 2015. The Electrochemical Properties of Biochars and How They Affect Soil Redox Properties and Processes. Agronomy. 5, 322-340. [CrossRef]
- Ihsanullah, F.A. , Al-Khaldi, B., Abusharkh, B., Khaledet, M., Atieh, M.A., Nasser, M.S., Tahar, I., Tawfik A.S., Agarwal, S., Vinod, T., Gupta, K., 2015. Adsorptive removal of cadmium(II) ions from liquid phase using acid modified carbon-based adsorbents. J. Mol. Liq. 204, 255–263. [CrossRef]
- Jafaryan, A. , Sadjadi, S., Gharib, A., Ahmadi, S.J., 2019. Optimization of cadmium adsorption from aqueous solutions by functionalized graphene and the reversible magnetic recovery of the adsorbent using response surface methodology, Appl. Organomet. Chem. 33, e5085. [CrossRef]
- Jie, L. , Changlun, C., Kairuo, Z., Xiangke, W., 2016. Nanoscale zero-valent iron particles modified on reduced graphene oxides using a plasma technique for Cd(II) removal. J. Taiwan Inst. of Chem. Eng. 59, 384-394. [CrossRef]
- Jokić Govedarica, J. , Tomašević Pilipović, D., Gvojić, V., Kerkez, Đ., Leovac Maćerak, A., Slijepčević, N., Bečelić-Tomin, M., 2022. The application of „green„ synthesized nulavalant iron for removal of Cd (II) from water. Water and sanitary technology L XII(2) 13-20. UDK: 502.51:504.5:546.48 546.72:502.174.
- Jones, B. , Nachtsheim, C.J., 2013. Definitive screening designs with added two-level categorical factors. J. Qual. Technol. 45,121-129. [CrossRef]
- Kong, L. , Tian, Y., Li, N., Liu, Y., Zhang, J., Zuo, W., 2018. Highly-effective phosphate removal from aqueous solutions by calcined nano-porous palygorskite matrix with embedded lanthanum hydroxide. App. Clay Sci. 162, 507–517. [CrossRef]
- Kecić, V. , Kerkez, Đ., Prica, M., Lužanin, O., Bečelić-Tomin, M., Tomašević Pilipović, D., Dalmacija, B., 2018. Optimization of azo printing dye removal with oak leaves-nZVI/H2O2 system using statistically designed experiment. J. Clean. Prod. 202, 65-80. [CrossRef]
- Krüger, O. , Adam, C., 2015. Phosphorus in recycling fertilizers-analytical challenges. Environ. Res. 155, 353–358. [CrossRef]
- Lee, W.H. , Kim, J.O., 2017. Effect of coexisting components on phosphate adsorption using magnetite particles in water, Environ. Sci. Pollut. Res. 26, 1054–1060. [CrossRef]
- Li, X.Q. , Zhang, W.X., 2007. Sequestration of metal cations with zero-valent iron nanoparticles – a study with high-resolution X-ray photoelectron spectroscopy (HR-XPS). J. Phys. Chem. C. 111, 6939–6946. [CrossRef]
- Li, Q. , Chen, Z., Wang, H., Yang, H., Wen, T., Wang, S., Hu, B., Wang X. (2021) Removal of organic compounds by nanoscale zero-valent iron and its composites. Science of the Total Environment 792,148546. [CrossRef]
- Machado, S. , Pinto, S., Grosso, J., Nouws, H., Albergaria, J., Deleruee Matos, C., 2013. Green production of zero-valent iron nanoparticles using tree leaf extracts. Sci. Total Environ. 445, 1-8. [CrossRef]
- Mohamed, O.A. , Masood, S.H., Bhowmik, J.L., 2017. Investigation on the flexural creep stiffness behavior of PC-ABS material processed by fused deposition modeling using response surface definitive screening design. J. Min. Met. Mat. Soc. 69, 498-505. [CrossRef]
- Moharami, S. , Jalal, M., 2013. Removal of phosphorus from aqueous solution by Iranian natural adsorbents. Chem. Eng. J. 223, 328–339. [CrossRef]
- Nagoya, S. , Nakamichi, S., Kawase, Y., 2019. Mechanisms of phosphate removal from aqueous solution by zero-valent iron: A novel kinetic model for electrostatic adsorption, surface complexation and precipitation of phosphate under oxic conditions. Sep. Purif. Technol. 218, 120–129. [CrossRef]
- Nguyen, T. , Ngo, H.H., Guo, W.S., 2015. Adsorption of phosphate from aqueous solutions and sewage using zirconium loaded okara (ZLO): fixed-bed column study. Sci. Total Environ. 523, 40–49. [CrossRef]
- O’Carroll, D. , Sleep, B., Krol, M., Boparai, H., Kocur, C. 2013. Nanoscale zero valent iron and bimetallic particles for contaminated site remediation. Adv. Water Resour. 51, 104-122. [CrossRef]
- Pasinszki, T. , Krebsz, M., 2020. Synthesis and Application of Zero-Valent Iron Nanoparticles in Water Treatment, Environmental Remediation, Catalysis, and Their Biological Effects. A review, Nanomaterials. 10. [CrossRef]
- Poguberović, S. , Krčmar, D., Maletić, S., Dalmacija, B., Tomašević Pilipović, D., Kerkez, Ð., Rončević, S, 2016. Removal of Ni(II) and Cu(II) from aqueous solutions using ‘green’ zero-valent iron nanoparticles produced by oak and mulberry leaf extracts. Water Sci. Technol. 74, 2115-2123. [CrossRef]
- Rana, A. , Kumari, N., Tyagi, M., Jagadevan, S., 2018. Leaf-extract mediated zero-valent iron for oxidation of Arsenic (III): Preparation, characterization and kinetics. Chem. Eng. J. 347, 91–100. [CrossRef]
- Sleiman, N. , Deluchat, V., Wazne, M., Mallet, M., Courtin-Nomade, A., Kazpard, V., Baudu, M., 2017. Phosphate removal from aqueous solutions using zero valent iron(ZVI): Influence of solution composition and ZVI aging, Colloid. Surf. A-Physicochem. Eng. Asp. 514, 1–10. [CrossRef]
- Stietiya, M.H. , Wang, J.J., 2014. Zinc and cadmium adsorption to aluminum oxide nanoparticles is affected by naturally occurring ligands. J. Environ. Qual. 43, 498-506. [CrossRef]
- Tang, H. , Zhang, S, Pang, H., Wang, J., Wang, X, Song, G., Yu, S. (2021) Insights into enhanced removal of U(VI) by melamine sponge supported sulfurized nanoscale zero-valent iron. Journal of Cleaner Production 329,129662. [CrossRef]
- Tosco, T. , Papini, P.M., Cruz. C.V., Sethi, R., 2014. Nanoscale zerovalent iron particles for groundwater remediation: a review. J. Clean. Prod. 77, 10–21. [CrossRef]
- Tu, Y.J. , You, C.F., Chang, C.K., 2012. Kinetics and thermodynamics of adsorption for Cd on green manufactured nano-particles. J. Hazard. Mater. 235, 116-122. [CrossRef]
- Veljković, N. , Vidojević, D., Jovičić, M., 2010. Environmental and health impact of pollutants from urban wastewater. “Wastewater, municipal solid waste and hazardous waste, Association for Water Technology and Sanitary Engineering, 29. March - 1. April 2010, Subotica, Serbia.
- Wang, K. , Xing, B., 2004. Mutual effects of cadmium and phosphate on their adsorption and desorption by goethite, Environ. Pollut. 127, 13-20. [CrossRef]
- Wang, C. , Luo, H., Zhang, Z., Wu, Y., Zhang, J., Chen, S., 2014. Removal of As(III) and As(V) from aqueous solutions using nanoscale zero-valent iron-reduced graphite oxide modified composites. J. Hazard. Mater. 268, 124–131. [CrossRef]
- Yu, S., Pang H., Huang, S., Tang, H., Wang S., Qiu, M., Chen Z., Yang, H., Song, G., Fu D., Hu B., Wang X. (2021) Recent advances in metal-organic framework membranes for water treatment: A review. Science of the Total Environment 800 149662. [CrossRef]
- Zhang, Y.H. , Liu, F.Q., Zhu, C.Q., Zhang, X.P., Wei, M.M., Wang, F.H., Ling, C., Li, A.M., 2017 Multifold enhanced synergistic removal of nickel and phosphate by a(N.Fe)-dual-functional bio-sorbent: Mechanism and application. J. Hazard. Mater. 329, 290–298. [CrossRef]
| Parameter | Estimate | Std Error | t ratio | Prob> |t| |
|---|---|---|---|---|
| Cadmium | ||||
| Oak-nZVI | 0.814 | 2.881 | 0.28 | 0.781 |
| Cadmium | 21.335 | 2.881 | 7.40 | <0.0001 |
| Phosphate | -10.642 | 2.881 | -3.69 | 0.0014 |
| pH | -5.913 | 2.881 | -2.05 | 0.0535 |
| Cadmium * Phosphate | 6.886 | 3.327 | 2.07 | 0.0516 |
| Cadmium * pH | 6.185 | 3.327 | 1.86 | 0.0778 |
| Cadmium * Cadmium | -17.185 | 5.391 | -3.19 | 0.0046 |
| Phosphate | ||||
| Oak-nZVI | -6.507 | 1.963 | -3.31 | 0.0036 |
| Cadmium | -6.110 | 1.963 | -3.11 | 0.0057 |
| Phosphate | 1.133 | 1.963 | 0.58 | 0.571 |
| pH | -1.499 | 1.963 | -0.76 | 0.455 |
| Oak-nZVI * pH | -16.260 | 3.269 | -4.97 | <0.0001 |
| Cadmium * pH | -7.561 | 2.647 | -2.86 | 0.010 |
| Phosphate * pH | 12.803 | 3.269 | 3.92 | 0.0009 |
| Cadmium * Cadmium | -11.334 | 5.537 | -2.05 | 0.055 |
| Parameter | Estimate | Std Error | t ratio | Prob> |t| |
|---|---|---|---|---|
| Nickel | ||||
| Oak-nZVI | -0.330 | 2.524 | -0.13 | 0.898 |
| Nickel | 7.150 | 2.524 | 2.83 | 0.012 |
| Phosphate | -0.256 | 2.524 | -0.10 | 0.921 |
| pH | 4.254 | 2.524 | 1.69 | 0.110 |
| Nickel * Nickel | -11.396 | 5.239 | -2.18 | 0.044 |
| Phosphate * pH | -4.926 | 3.111 | -1.58 | 0.132 |
| Phosphate | ||||
| Oak-nZVI | 2.646 | 1.791 | 1.48 | 0.156 |
| Nickel | -6.516 | 1.791 | -3.64 | 0.0017 |
| Phosphate | 19.201 | 1.791 | 10.72 | <0.0001 |
| pH | 7.105 | 1.791 | 3.97 | 0.0008 |
| Oak-nZVI * Nickel | 12.730 | 2.296 | 5.54 | <0.0001 |
| Oak-nZVI * Phosphates | -4.289 | 2.083 | -2.06 | 0.053 |
| Phosphate * Phosphate | -12.194 | 3.744 | -3.26 | 0.0041 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




