Submitted:
05 September 2023
Posted:
07 September 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Mechanisms
2.1. Macrophage phagocytosis
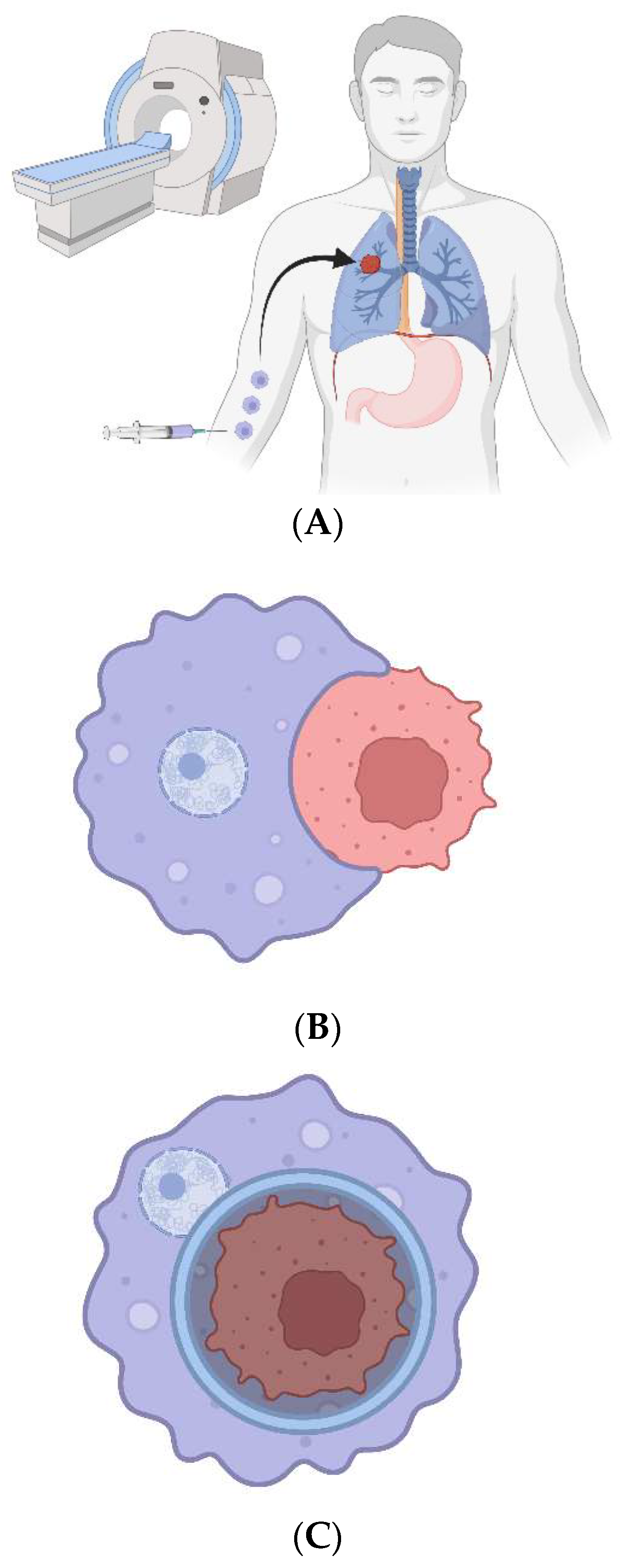
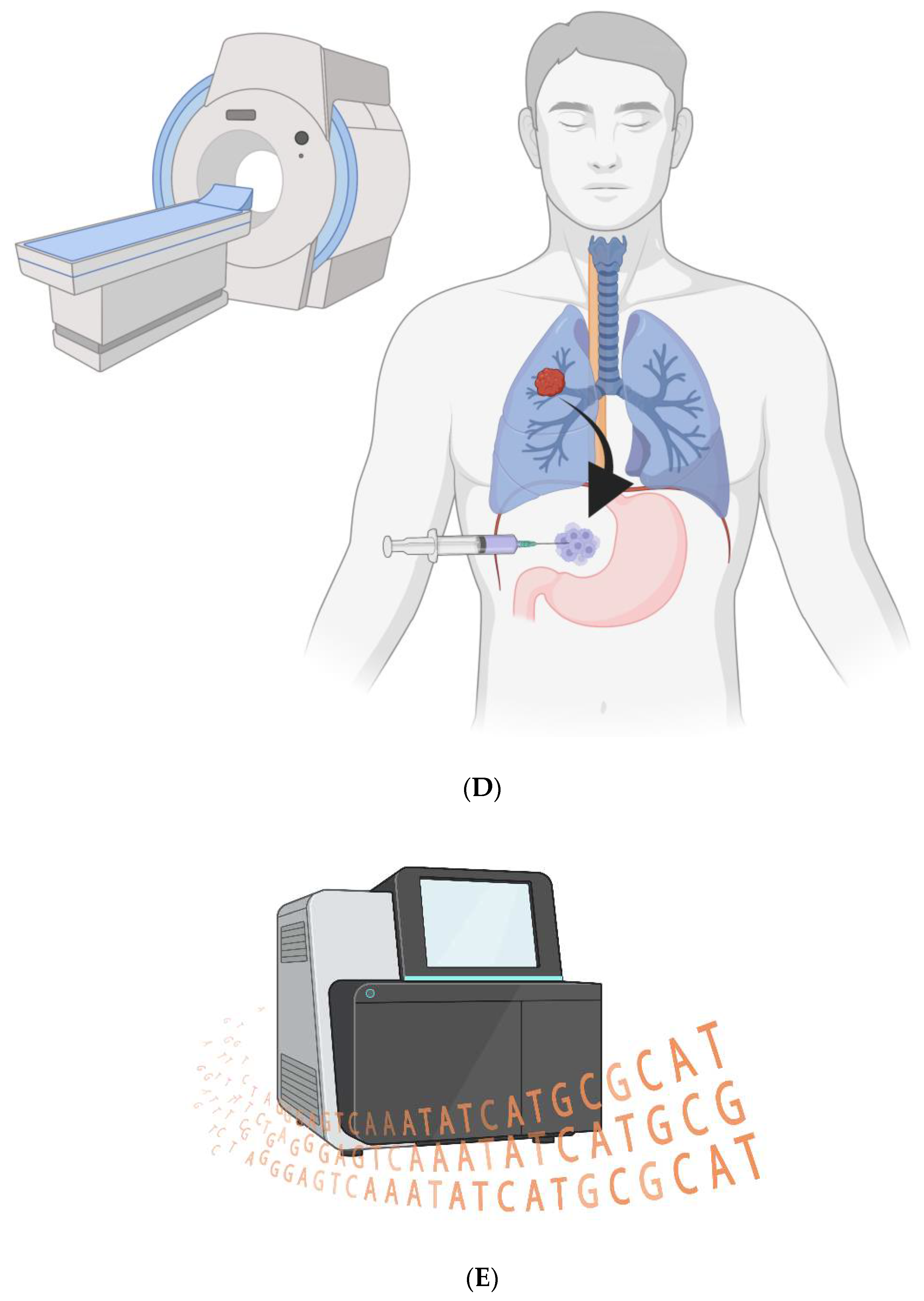
2.2. Bacterial phagocytosis
2.3. Remaining strategies
2.4. Brain and spinal cord tumors:
3. Conclusion
Acknowledgments
References
- Renteln, M. Conditional replication of oncolytic viruses based on detection of oncogenic mRNA. Gene Ther. 2018, 25, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Renteln, M. Correction: Conditional replication of oncolytic viruses based on detection of oncogenic mRNA. Gene Ther. 2021, 28, 469–469. [Google Scholar] [CrossRef] [PubMed]
- Renteln, M.A. Promoting oncolytic vector replication with switches that detect ubiquitous mutations. Curr. Cancer Ther. Rev. 2023. [Google Scholar] [CrossRef]
- Muthana, M.; Kennerley, A.J.; Hughes, R.; Fagnano, E.; Richardson, J.; Paul, M.; Murdoch, C.; Wright, F.; Payne, C.; Lythgoe, M.F.; et al. Directing cell therapy to anatomic target sites in vivo with magnetic resonance targeting. Nat. Commun. 2015, 6, 8009. [Google Scholar] [CrossRef]
- Dowdell, A.; Paschke, P.I.; Thomason, P.A.; Tweedy, L.; Insall, R.H. Competition between chemoattractants causes unexpected complexity and can explain negative chemotaxis. Curr. Biol. 2023, 33, 1704–1715. [Google Scholar] [CrossRef]
- Zhang, X.; Ren, L.; Wu, J.; Feng, R.; Chen, Y.; Li, R.; Wu, M.; Zheng, M.; Wu, X.G.; Luo, W.; et al. ARHGEF37 overexpression promotes extravasation and metastasis of hepatocellular carcinoma via directly activating Cdc42. J. Exp. Clin. Cancer Res. 2022, 41, 1–16. [Google Scholar] [CrossRef]
- Morrissey, M.A.; Williamson, A.P.; Steinbach, A.M.; Roberts, E.W.; Kern, N.; Headley, M.B.; Vale, R.D. Chimeric antigen receptors that trigger phagocytosis. eLife 2018, 7, e36688. [Google Scholar] [CrossRef]
- Ito, A.; Teranishi, R.; Kamei, K.; Yamaguchi, M.; Ono, A.; Masumoto, S.; Sonoda, Y.; Horie, M.; Kawabe, Y.; Kamihira, M. Magnetically triggered transgene expression in mammalian cells by localized cellular heating of magnetic nanoparticles. J. Biosci. Bioeng. 2019, 128, 355–364. [Google Scholar] [CrossRef]
- Bausch-Fluck, D.; Hofmann, A.; Bock, T.; Frei, A.P.; Cerciello, F.; Jacobs, A.; Moest, H.; Omasits, U.; Gundry, R.L.; Yoon, C.; et al. A Mass Spectrometric-Derived Cell Surface Protein Atlas. PLOS ONE 2015, 10, e0121314–e0121314. [Google Scholar] [CrossRef]
- Bausch-Fluck, D.; Goldmann, U.; Müller, S.; van Oostrum, M.; Müller, M.; Schubert, O.T.; Wollscheid, B. The in silico human surfaceome. Proc. Natl. Acad. Sci. 2018, 115, E10988–E10997. [Google Scholar] [CrossRef]
- Yang, H.; Shao, R.; Huang, H.; Wang, X.; Rong, Z.; Lin, Y. Engineering macrophages to phagocytose cancer cells by blocking the CD47/SIRPɑ axis. Cancer Med. 2019, 8, 4245–4253. [Google Scholar] [CrossRef] [PubMed]
- Jungbluth, M.; Renicke, C.; Taxis, C. Targeted protein depletion in Saccharomyces cerevisiae by activation of a bidirectional degron. BMC Syst. Biol. 2010, 4, 176–176. [Google Scholar] [CrossRef] [PubMed]
- Kinchen, J.M.; Ravichandran, K.S. Phagosome maturation: going through the acid test. Nat. Rev. Mol. Cell Biol. 2008, 9, 781–795. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Maxwell, K.G.; Wang, K.; Bowers, D.T.; Flanders, J.A.; Liu, W.; Wang, L.-H.; Liu, Q.; Liu, C.; Naji, A.; et al. A nanofibrous encapsulation device for safe delivery of insulin-producing cells to treat type 1 diabetes. Sci. Transl. Med. 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- A Gilmartin, A.; Ralston, K.S.; A Petri, W. Inhibition of Amebic Cysteine Proteases Blocks Amebic Trogocytosis but Not Phagocytosis. J. Infect. Dis. 2020, 221, 1734–1739. [Google Scholar] [CrossRef]
- Underhill, D.M.; Goodridge, H.S. Information processing during phagocytosis. Nat. Rev. Immunol. 2012, 12, 492–502. [Google Scholar] [CrossRef]
- Milde, R.; Ritter, J.; Tennent, G.A.; Loesch, A.; Martinez, F.O.; Gordon, S.; Pepys, M.B.; Verschoor, A.; Helming, L. Multinucleated Giant Cells Are Specialized for Complement-Mediated Phagocytosis and Large Target Destruction. Cell Rep. 2015, 13, 1937–1948. [Google Scholar] [CrossRef]
- Li, H.; Pohler, U.; Strehlow, I.; Hertig, S.; Baccarini, M.; Emmendörffer, A.; Tschopp, J.; Lohmann-Matthes, M.-L. Macrophage precursor cells produce perforin and perform Yac-1 lytic activity in response to stimulation with interleukin-2. J. Leukoc. Biol. 1994, 56, 117–123. [Google Scholar] [CrossRef]
- Tamang, D.L.; Alves, B.N.; Elliott, V.; Redelman, D.; Wadhwa, R.; Fraser, S.A.; Hudig, D. Regulation of perforin lysis: Implications for protein disulfide isomerase proteins. Cell. Immunol. 2009, 255, 82–92. [Google Scholar] [CrossRef]
- Heusel, J.W.; Wesselschmidt, R.L.; Shresta, S.; Russell, J.H.; Ley, T.J. Cytotoxic lymphocytes require granzyme B for the rapid induction of DNA fragmentation and apoptosis in allogeneic target cells. Cell 1994, 76, 977–987. [Google Scholar] [CrossRef]
- Voskoboinik, I.; Smyth, M.J.; Trapani, J.A. Perforin-mediated target-cell death and immune homeostasis. Nat. Rev. Immunol. 2006, 6, 940–952. [Google Scholar] [CrossRef]
- Gupta, M.; Shin, D.-M.; Ramakrishna, L.; Goussetis, D.J.; Platanias, L.C.; Xiong, H.; Iii, H.C.M.; Ozato, K. IRF8 directs stress-induced autophagy in macrophages and promotes clearance of Listeria monocytogenes. Nat. Commun. 2015, 6, 1–14. [Google Scholar] [CrossRef]
- Yoon, Y.G.; Koob, M.D. Nonreplicating Intracellular Bacterial Vector for Conjugative DNA Transfer into Mitochondria. Pharm. Res. 2012, 29, 1040–1045. [Google Scholar] [CrossRef]
- Sánchez-Gorostiaga, A.; Palacios, P.; Martínez-Arteaga, R.; Sánchez, M.; Casanova, M.; Vicente, M. Life without Division: Physiology of Escherichia coli FtsZ-Deprived Filaments. mBio 2016, 7. [Google Scholar] [CrossRef]
- Shiratori, T.; Suzuki, S.; Kakizawa, Y.; Ishida, K.-I. Phagocytosis-like cell engulfment by a planctomycete bacterium. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef]
- Kolinko, I.; Lohße, A.; Borg, S.; Raschdorf, O.; Jogler, C.; Tu, Q.; Pósfai, M.; Tompa, É.; Plitzko, J.M.; Brachmann, A.; et al. Biosynthesis of magnetic nanostructures in a foreign organism by transfer of bacterial magnetosome gene clusters. Nat. Nanotechnol. 2014, 9, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Grillot-Courvalin, C.; Goussard, S.; Huetz, F.; Ojcius, D.M.; Courvalin, P. Functional gene transfer from intracellular bacteria to mammalian cells. Nat. Biotechnol. 1998, 16, 862–866. [Google Scholar] [CrossRef]
- Shiomi, D.; Sakai, M.; Niki, H. Determination of bacterial rod shape by a novel cytoskeletal membrane protein. EMBO J. 2008, 27, 3081–3091. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, G.; Metzger, S.; Aizenman, E.; Roza, S.; Cashel, M.; Glaser, G. Overexpression of the relA gene in Escherichia coli. J. Biol. Chem. 1991, 266, 3760–3767. [Google Scholar] [CrossRef] [PubMed]
- Büke, F.; Grilli, J.; Lagomarsino, M.C.; Bokinsky, G.; Tans, S.J. ppGpp is a bacterial cell size regulator. Curr. Biol. 2022, 32, 870–877. [Google Scholar] [CrossRef]
- Reniere, M.L.; Whiteley, A.T.; Hamilton, K.L.; John, S.M.; Lauer, P.; Brennan, R.G.; Portnoy, D.A. Glutathione activates virulence gene expression of an intracellular pathogen. Nature 2015, 517, 170–173. [Google Scholar] [CrossRef] [PubMed]
- Basan, M.; Zhu, M.; Dai, X.; Warren, M.; Sévin, D.; Wang, Y.; Hwa, T. Inflating bacterial cells by increased protein synthesis. Mol. Syst. Biol. 2015, 11, 836. [Google Scholar] [CrossRef]
- Taylor, C.M.; Beresford, M.; Epton, H.A.S.; Sigee, D.C.; Shama, G.; Andrew, P.W.; Roberts, I.S. Listeria monocytogenes relA and hpt Mutants Are Impaired in Surface-Attached Growth and Virulence. J. Bacteriol. 2002, 184, 621–628. [Google Scholar] [CrossRef]
- Kume, K.; Cantwell, H.; Burrell, A.; Nurse, P. Nuclear membrane protein Lem2 regulates nuclear size through membrane flow. Nat. Commun. 2019, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, M.B.; A Theriot, J. Shigella flexneri surface protein IcsA is sufficient to direct actin-based motility. . 1995, 92, 6572–6576. [Google Scholar] [CrossRef]
- Xu, C.; Ren, X.-H.; Han, D.; Peng, Y.; Lei, J.-J.; Yu, L.-X.; Liu, L.-J.; Xu, W.-C.; Cheng, S.-X. Precise Detection on Cell–Cell Fusion by a Facile Molecular Beacon-Based Method. Anal. Chem. 2022, 94, 17334–17340. [Google Scholar] [CrossRef] [PubMed]
- Magnin, R.; Rabusseau, F.; Salabartan, F.; Mériaux, S.; Aubry, J.-F.; Le Bihan, D.; Dumont, E.; Larrat, B. Magnetic resonance-guided motorized transcranial ultrasound system for blood-brain barrier permeabilization along arbitrary trajectories in rodents. J. Ther. Ultrasound 2015, 3, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Weber-Adrian, D.; Thévenot, E.; O'Reilly, M.A.; Oakden, W.; Akens, M.K.; Ellens, N.; Markham-Coultes, K.; Burgess, A.; Finkelstein, J.; Yee, A.J.; et al. Gene delivery to the spinal cord using MRI-guided focused ultrasound. Gene Ther. 2015, 22, 568–577. [Google Scholar] [CrossRef]
- Nagai, Y.; Miyakawa, N.; Takuwa, H.; Hori, Y.; Oyama, K.; Ji, B.; Takahashi, M.; Huang, X.-P.; Slocum, S.T.; DiBerto, J.F.; et al. Deschloroclozapine, a potent and selective chemogenetic actuator enables rapid neuronal and behavioral modulations in mice and monkeys. Nat. Neurosci. 2020, 23, 1157–1167. [Google Scholar] [CrossRef]
- Yevtodiyenko, A.; Bazhin, A.; Khodakivskyi, P.; Godinat, A.; Budin, G.; Maric, T.; Pietramaggiori, G.; Scherer, S.S.; Kunchulia, M.; Eppeldauer, G.; et al. Portable bioluminescent platform for in vivo monitoring of biological processes in non-transgenic animals. Nat. Commun. 2021, 12, 1–12. [Google Scholar] [CrossRef]
- Park, J.S.; Rhau, B.; Hermann, A.; McNally, K.A.; Zhou, C.; Gong, D.; Weiner, O.D.; Conklin, B.R.; Onuffer, J.; Lim, W.A. Synthetic control of mammalian-cell motility by engineering chemotaxis to an orthogonal bioinert chemical signal. Proc Natl Acad Sci USA 2014, 11, 5896–5901. [Google Scholar] [CrossRef]
- Lu, H.-B.; Cao, Y.; Hu, J.-Z.; Xu, J.-Q.; Liu, Q.-Q.; Huang, S.-Y.; Duan, C.-Y. The lymphatic system: a therapeutic target for central nervous system disorders. Neural Regen. Res. 2023, 18, 1249–1256. [Google Scholar] [CrossRef] [PubMed]
- Beltran-Huarac, J.; Yamaleyeva, D.N.; Dotti, G.; Hingtgen, S.; Sokolsky-Papkov, M.; Kabanov, A.V. Magnetic Control of Protein Expression via Magneto-mechanical Actuation of ND-PEGylated Iron Oxide Nanocubes for Cell Therapy. ACS Appl. Mater. Interfaces 2023, 15, 19877–19891. [Google Scholar] [CrossRef] [PubMed]
- Mazuel, F.; Espinosa, A.; Luciani, N.; Reffay, M.; Le Borgne, R.; Motte, L.; Desboeufs, K.; Michel, A.; Pellegrino, T.; Lalatonne, Y.; et al. Massive Intracellular Biodegradation of Iron Oxide Nanoparticles Evidenced Magnetically at Single-Endosome and Tissue Levels. ACS Nano 2016, 10, 7627–7638. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



