Submitted:
15 August 2023
Posted:
16 August 2023
Read the latest preprint version here
Abstract
Keywords:
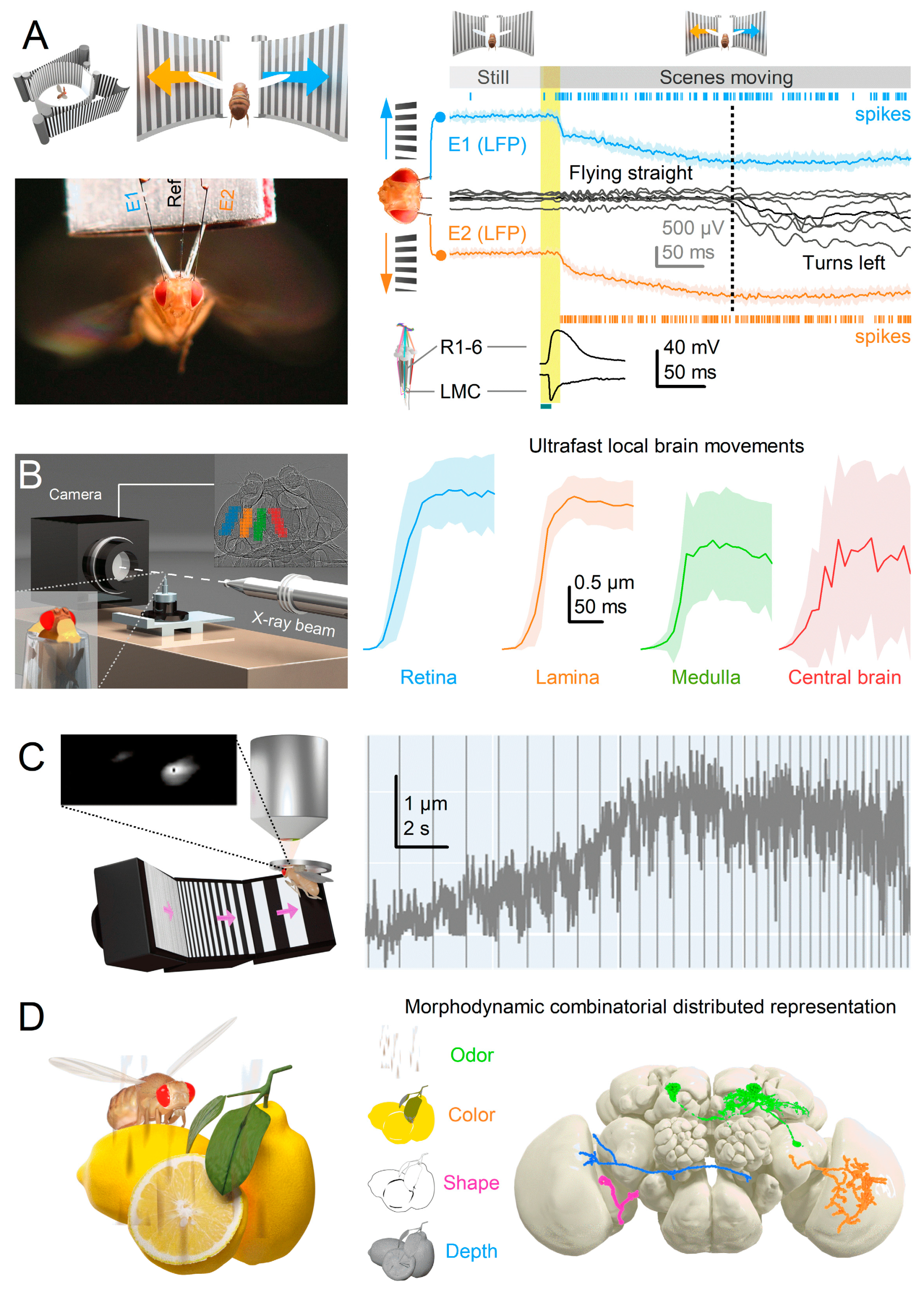
Introduction
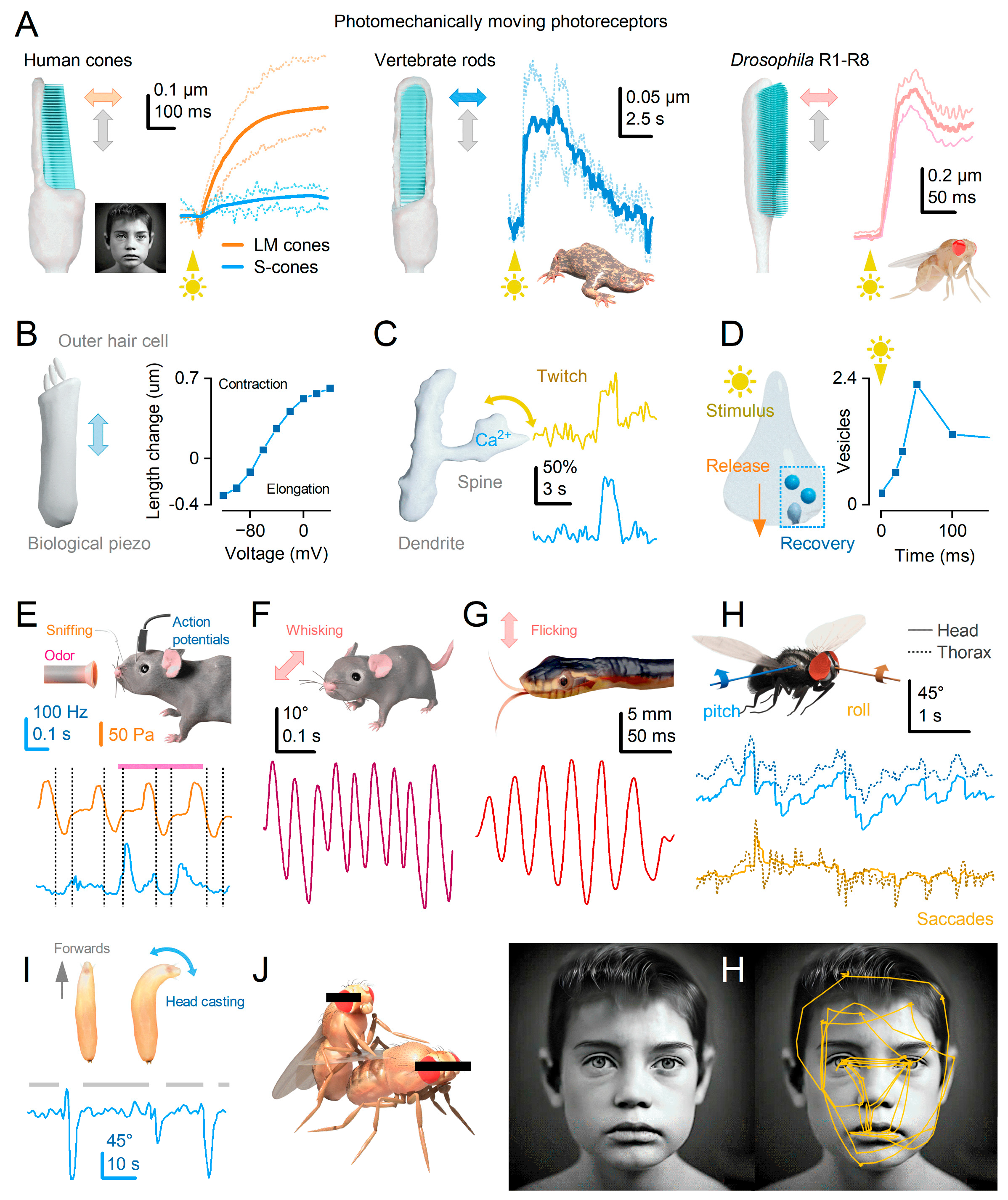
Photoreceptor movements enhance vision
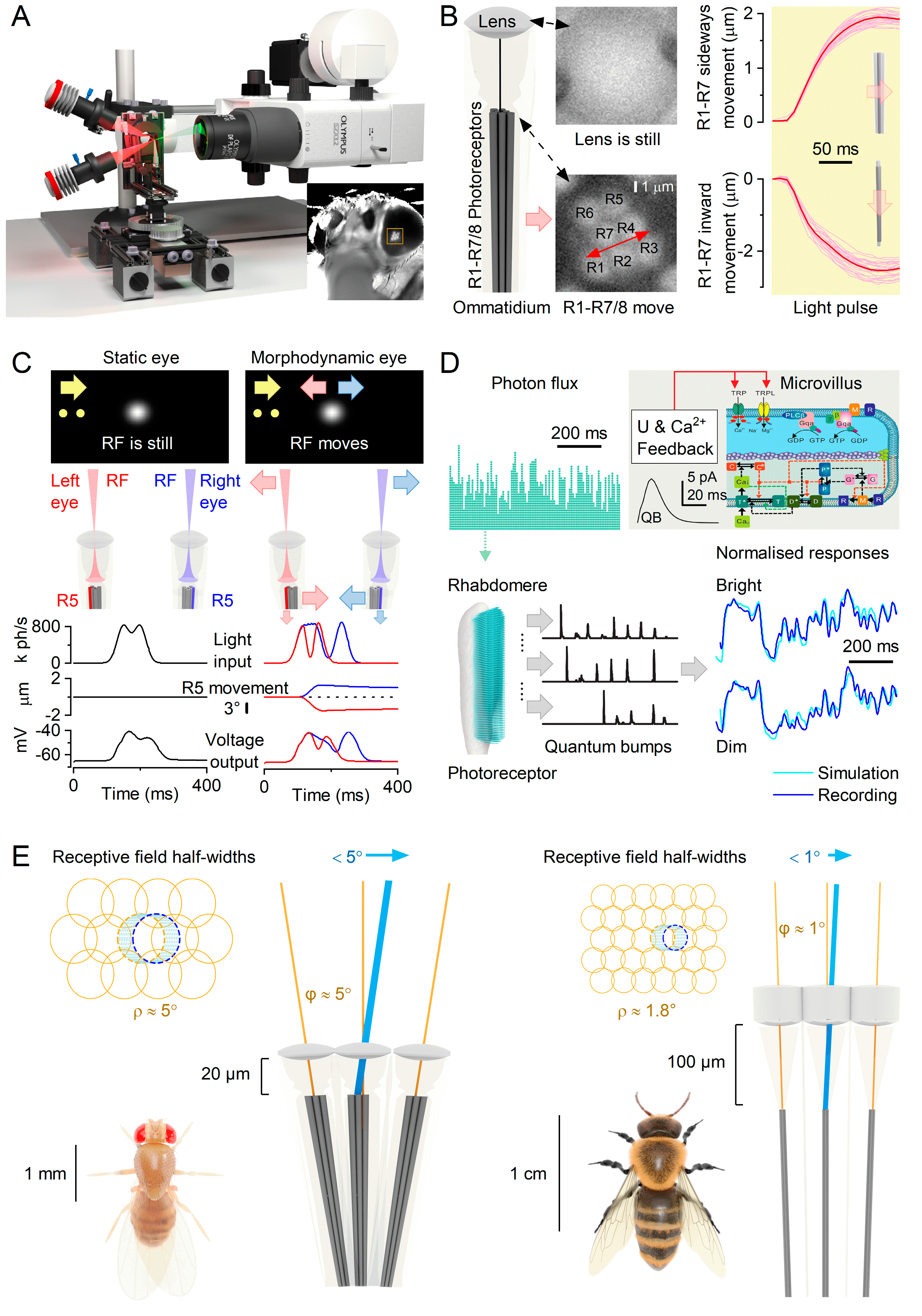
Microsaccades are photomechanical adaptations in phototransduction
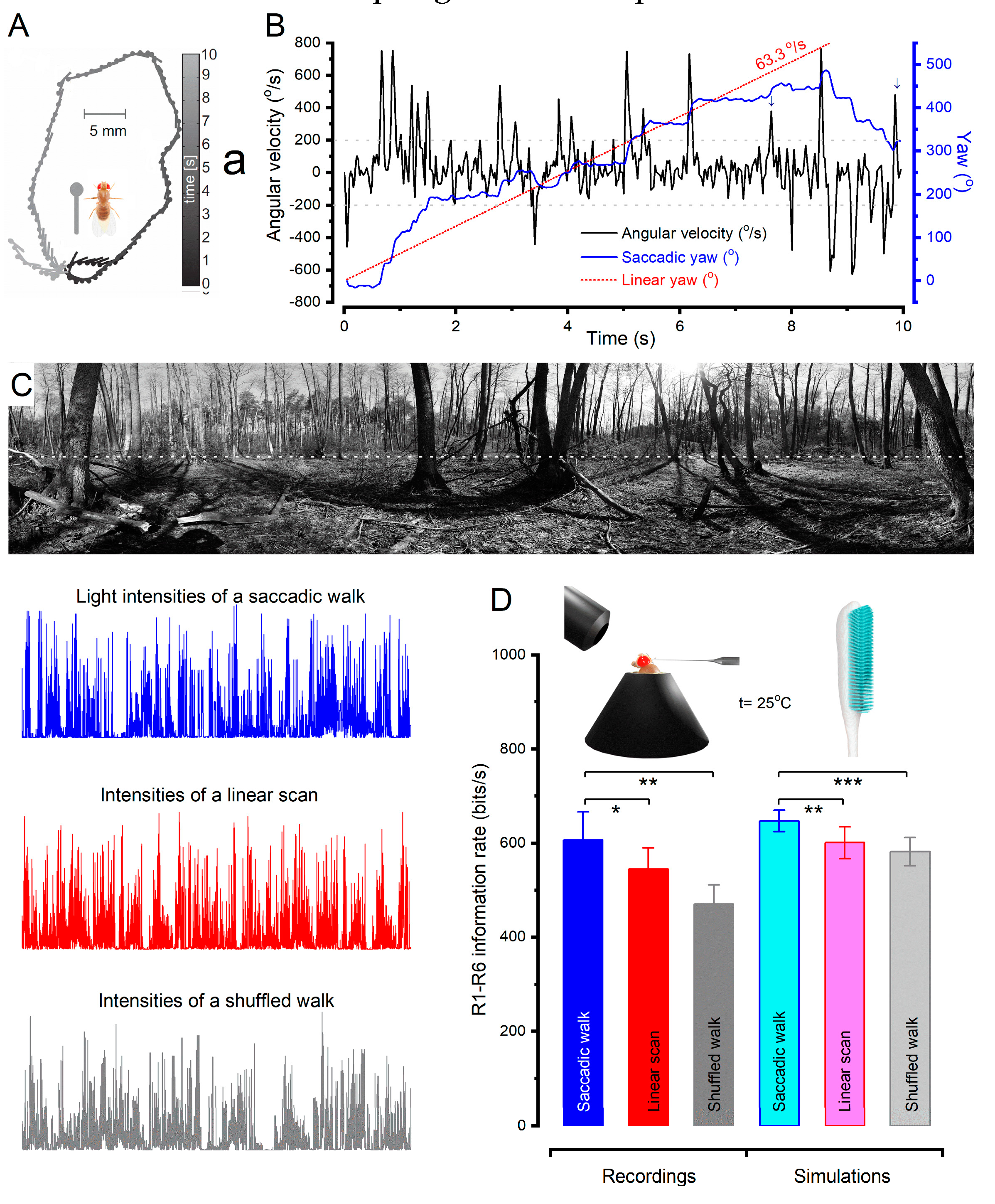
Matching saccadic behaviours to microsaccadic sampling
Mirror-symmetric microsaccades enable 3d vision
Reliable neural estimates of the variable world
Multiple layers of active sampling vs simple motion detection models
Benefits of neural morphodynamics
Efficient Neural Encoding of Reliable Representations across Wide Dynamic Range
Predictive Coding and Minimal Neural Delays
Anti-aliasing and Robust Communication
The Efficiency of Encoding Space in Time
Optimal Utilisation of Genetic Information in DNA
Future avenues of research
Genetically Enhancing Signalling Performance and Speed
Neural Activity Synchronisation
Perception Enhancement
Conclusion and future outlook
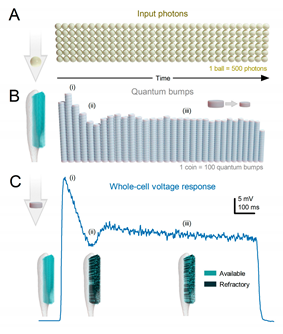
- Each microvillus can produce only one quantum bump at a time53,120-122.
- After producing a quantum bump, a microvillus becomes refractory for up to 300 ms (in Drosophila R1-6 photoreceptors at 25°C) and cannot respond to other photons120,123,124.
- Quantum bumps from all microvilli sum up the macroscopic response53,120-122,125.
- Microvilli availability sets a photoreceptor’s maximum sample rate (quantum bump production rate), adapting its macroscopic response to a light stimulus53,122.
- Global Ca2+ accumulation and membrane voltage affect samples of all microvilli. These global feedbacks strengthen with brightening light to reduce the size and duration of quantum bumps, adapting the macroscopic response55,73,126,127.
Author Contributions
Acknowledgments
Declaration of Interests
References
- Eichler, K.; Li, F.; Litwin-Kumar, A.; Park, Y.; Andrade, I.; Schneider-Mizell, C.M.; Saumweber, T.; Huser, A.; Eschbach, C.; Gerber, B.; et al. The complete connectome of a learning and memory centre in an insect brain. Nature 2017, 548, 175–182. [Google Scholar] [CrossRef]
- Oh, S.W.; Harris, J.A.; Ng, L.; Winslow, B.; Cain, N.; Mihalas, S.; Wang, Q.; Lau, C.; Kuan, L.; Henry, A.M.; et al. A mesoscale connectome of the mouse brain. Nature 2014, 508, 207–214. [Google Scholar] [CrossRef]
- Rivera-Alba, M.; Vitaladevuni, S.N.; Mishchenko, Y.; Lu, Z.; Takemura, S.-Y.; Scheffer, L.; Meinertzhagen, I.A.; Chklovskii, D.B.; de Polavieja, G.G. Wiring Economy and Volume Exclusion Determine Neuronal Placement in the Drosophila Brain. Curr. Biol. 2011, 21, 2000–2005. [Google Scholar] [CrossRef]
- Winding, M.; Pedigo, B.D.; Barnes, C.L.; Patsolic, H.G.; Park, Y.; Kazimiers, T.; Fushiki, A.; Andrade, I.V.; Khandelwal, A.; Valdes-Aleman, J.; et al. The connectome of an insect brain. Science 2023, 379, eadd9330–eadd9330. [Google Scholar] [CrossRef]
- Hudspeth, A. Making an Effort to Listen: Mechanical Amplification in the Ear. Neuron 2008, 59, 530–545. [Google Scholar] [CrossRef]
- Pandiyan, V.P.; Maloney-Bertelli, A.; Kuchenbecker, J.A.; Boyle, K.C.; Ling, T.; Chen, Z.C.; Park, B.H.; Roorda, A.; Palanker, D.; Sabesan, R. The optoretinogram reveals the primary steps of phototransduction in the living human eye. Sci. Adv. 2020, 6, eabc1124. [Google Scholar] [CrossRef]
- Hardie, R.C.; Franze, K. Photomechanical Responses in Drosophila Photoreceptors. Science 2012, 338, 260–263. [Google Scholar] [CrossRef]
- Bocchero, U.; Falleroni, F.; Mortal, S.; Li, Y.; Cojoc, D.; Lamb, T.; Torre, V. Mechanosensitivity is an essential component of phototransduction in vertebrate rods. PLOS Biol. 2020, 18, e3000750. [Google Scholar] [CrossRef]
- Korkotian, E.; Segal, M. Spike-Associated Fast Contraction of Dendritic Spines in Cultured Hippocampal Neurons. Neuron 2001, 30, 751–758. [Google Scholar] [CrossRef]
- Majewska, A.; Sur, M. Motility of dendritic spines in visual cortex in vivo : Changes during the critical period and effects of visual deprivation. Proc. Natl. Acad. Sci. 2003, 100, 16024–16029. [Google Scholar] [CrossRef]
- Crick, F. (1982). Do dendritic spines twitch? Trends in Neurosciences 5, 44-46.
- Juusola, M.; Dau, A.; Song, Z.; Solanki, N.; Rien, D.; Jaciuch, D.; Dongre, S.A.; Blanchard, F.; de Polavieja, G.G.; Hardie, R.C.; et al. Microsaccadic sampling of moving image information provides Drosophila hyperacute vision. eLife 2017, 6. [Google Scholar] [CrossRef]
- Kemppainen, J.; Mansour, N.; Takalo, J.; Juusola, M. High-speed imaging of light-induced photoreceptor microsaccades in compound eyes. Commun. Biol. 2022, 5, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Kemppainen, J.; Scales, B.; Haghighi, K.R.; Takalo, J.; Mansour, N.; McManus, J.; Leko, G.; Saari, P.; Hurcomb, J.; Antohi, A.; et al. Binocular mirror–symmetric microsaccadic sampling enables Drosophila hyperacute 3D vision. Proc. Natl. Acad. Sci. 2022, 119. [Google Scholar] [CrossRef]
- Senthilan, P.R.; Piepenbrock, D.; Ovezmyradov, G.; Nadrowski, B.; Bechstedt, S.; Pauls, S.; Winkler, M.; Möbius, W.; Howard, J.; Göpfert, M.C. Drosophila Auditory Organ Genes and Genetic Hearing Defects. Cell 2012, 150, 1042–1054. [Google Scholar] [CrossRef]
- Reshetniak, S.; Rizzoli, S.O. The vesicle cluster as a major organizer of synaptic composition in the short-term and long-term. Curr. Opin. Cell Biol. 2021, 71, 63–68. [Google Scholar] [CrossRef]
- Reshetniak, S.; Ußling, J.; Perego, E.; Rammner, B.; Schikorski, T.; Fornasiero, E.F.; Truckenbrodt, S.; Köster, S.; O Rizzoli, S. A comparative analysis of the mobility of 45 proteins in the synaptic bouton. EMBO J. 2020, 39, e104596. [Google Scholar] [CrossRef]
- Rusakov, D.A.; Savtchenko, L.P.; Zheng, K.; Henley, J.M. Shaping the synaptic signal: molecular mobility inside and outside the cleft. Trends Neurosci. 2011, 34, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Juusola, M.; O Uusitalo, R.; Weckström, M. Transfer of graded potentials at the photoreceptor-interneuron synapse. J. Gen. Physiol. 1995, 105, 117–148. [Google Scholar] [CrossRef] [PubMed]
- Fettiplace, R.; Crawford, A.C.; Kennedy, H.J. SIGNAL TRANSFORMATION BY MECHANOTRANSDUCER CHANNELS OF MAMMALIAN OUTER HAIR CELLS. 2006. [CrossRef]
- Kennedy, H.J.; Crawford, A.C.; Fettiplace, R. Force generation by mammalian hair bundles supports a role in cochlear amplification. Nature 2005, 433, 880–883. [Google Scholar] [CrossRef]
- Juusola, M.; French, A.S.; O Uusitalo, R.; Weckström, M. Information processing by graded-potential transmission through tonically active synapses. Trends Neurosci. 1996, 19, 292–297. [Google Scholar] [CrossRef]
- Watanabe, S.; Rost, B.R.; Camacho-Pérez, M.; Davis, M.W.; Söhl-Kielczynski, B.; Rosenmund, C.; Jorgensen, E.M. Ultrafast endocytosis at mouse hippocampal synapses. Nature 2013, 504, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; de Polavieja, G.G.; Wolfram, V.; Asyali, M.H.; Hardie, R.C.; Juusola, M. Feedback Network Controls Photoreceptor Output at the Layer of First Visual Synapses in Drosophila . J. Gen. Physiol. 2006, 127, 495–510. [Google Scholar] [CrossRef] [PubMed]
- Shusterman, R.; Smear, M.C.; A Koulakov, A.; Rinberg, D. Precise olfactory responses tile the sniff cycle. Nat. Neurosci. 2011, 14, 1039–1044. [Google Scholar] [CrossRef] [PubMed]
- Smear, M.; Shusterman, R.; O’connor, R.; Bozza, T.; Rinberg, D. Perception of sniff phase in mouse olfaction. Nature 2011, 479, 397–400. [Google Scholar] [CrossRef]
- E Bush, N.; A Solla, S.; Hartmann, M.J. Whisking mechanics and active sensing. Curr. Opin. Neurobiol. 2016, 40, 178–188. [Google Scholar] [CrossRef]
- Daghfous, G.; Smargiassi, M.; Libourel, P.-A.; Wattiez, R.; Bels, V. The Function of Oscillatory Tongue-Flicks in Snakes: Insights from Kinematics of Tongue-Flicking in the Banded Water Snake (Nerodia fasciata). Chem. Senses 2012, 37, 883–896. [Google Scholar] [CrossRef]
- van Hateren, J.H. , and Schilstra, C. ( 1999). Blowfly flight and optic flow II. Head movements during flight. J Exp Biol 202, 1491–1500.
- Schütz, A.C.; Braun, D.I.; Gegenfurtner, K.R. Eye movements and perception: A selective review. J. Vis. 2011, 11, 9–9. [Google Scholar] [CrossRef]
- Land, M. Eye movements in man and other animals. Vis. Res. 2019, 162, 1–7. [Google Scholar] [CrossRef]
- Davies, A.; Louis, M.; Webb, B. A Model of Drosophila Larva Chemotaxis. PLOS Comput. Biol. 2015, 11, e1004606. [Google Scholar] [CrossRef]
- Gomez-Marin, A.; Stephens, G.J.; Louis, M. Active sampling and decision making in Drosophila chemotaxis. Nat. Commun. 2011, 2, 441. [Google Scholar] [CrossRef] [PubMed]
- Schöneich, S.; Hedwig, B. Hyperacute Directional Hearing and Phonotactic Steering in the Cricket (Gryllus bimaculatus deGeer). PLOS ONE 2010, 5, e15141. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Nikolaev, A.; Wardill, T.J.; O'Kane, C.J.; de Polavieja, G.G.; Juusola, M. Network Adaptation Improves Temporal Representation of Naturalistic Stimuli in Drosophila Eye: I Dynamics. PLOS ONE 2009, 4, e4307. [Google Scholar] [CrossRef] [PubMed]
- Barlow, H.B. (1961). Possible principles underlying the transformations of sensory messages. In Sensory Communication, W. Rosenblith, ed. (M.I.T. Press), pp. 217-234.
- Darwin, C. (1859). On the origin of species by means of natural selection, or the preservation of favoured races in the struggle for life (John Murray).
- Sheehan, M.J.; Tibbetts, E.A. Specialized Face Learning Is Associated with Individual Recognition in Paper Wasps. Science 2011, 334, 1272–1275. [Google Scholar] [CrossRef]
- Miller, S.E.; Legan, A.W.; Henshaw, M.T.; Ostevik, K.L.; Samuk, K.; Uy, F.M.K.; Sheehan, M.J. Evolutionary dynamics of recent selection on cognitive abilities. Proc. Natl. Acad. Sci. 2020, 117, 3045–3052. [Google Scholar] [CrossRef]
- Kacsoh, B.Z.; Lynch, Z.R.; Mortimer, N.T.; Schlenke, T.A. Fruit Flies Medicate Offspring After Seeing Parasites. Science 2013, 339, 947–950. [Google Scholar] [CrossRef]
- Land, M.F. VISUAL ACUITY IN INSECTS. Annu. Rev. Èntomol. 1997, 42, 147–177. [Google Scholar] [CrossRef]
- Laughlin, S.B. The role of sensory adaptation in the retina. J. Exp. Biol. 1989, 146, 39–62. [Google Scholar] [CrossRef]
- Laughlin, S.B.; de Ruyter van Steveninck, R.R.; Anderson, J.C. The metabolic cost of neural information. Nat. Neurosci. 1998, 1, 36–41. [Google Scholar] [CrossRef]
- Schilstra, C.; Van Hateren, J.H. Blowfly flight and optic flow : I. Thorax kinematics and flight dynamics. J. Exp. Biol. 1999, 202, 1481–1490. [Google Scholar] [CrossRef]
- Fenk, L.M.; Avritzer, S.C.; Weisman, J.L.; Nair, A.; Randt, L.D.; Mohren, T.L.; Siwanowicz, I.; Maimon, G. Muscles that move the retina augment compound eye vision in Drosophila. Nature 2022, 612, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Guiraud, M.; Roper, M.; Chittka, L. High-Speed Videography Reveals How Honeybees Can Turn a Spatial Concept Learning Task Into a Simple Discrimination Task by Stereotyped Flight Movements and Sequential Inspection of Pattern Elements. Front. Psychol. 2018, 9, 1347. [Google Scholar] [CrossRef] [PubMed]
- Nityananda, V.; Chittka, L.; Skorupski, P. Can Bees See at a Glance? J. Exp. Biol. 2014, 217, 1933–9. [Google Scholar] [CrossRef] [PubMed]
- Vasas, V.; Chittka, L. Insect-Inspired Sequential Inspection Strategy Enables an Artificial Network of Four Neurons to Estimate Numerosity. iScience 2018, 11, 85–92. [Google Scholar] [CrossRef]
- Chittka, L. , and Skorupski, P. (2017). Active vision: A broader comparative perspective is needed. Constr Found 13, 128-129.
- Sorribes, A.; Armendariz, B.G.; Lopez-Pigozzi, D.; Murga, C.; de Polavieja, G.G. The Origin of Behavioral Bursts in Decision-Making Circuitry. PLOS Comput. Biol. 2011, 7, e1002075. [Google Scholar] [CrossRef]
- Exner, S. (1891). Die Physiologie der facettierten Augen von Krebsen und Insecten.
- Hardie, R.C.; Juusola, M. Phototransduction in Drosophila. Curr. Opin. Neurobiol. 2015, 34, 37–45. [Google Scholar] [CrossRef]
- Song, Z.; Postma, M.; Billings, S.A.; Coca, D.; Hardie, R.C.; Juusola, M. Stochastic, Adaptive Sampling of Information by Microvilli in Fly Photoreceptors. Curr. Biol. 2012, 22, 1371–1380. [Google Scholar] [CrossRef]
- Stavenga, D.G. Angular and spectral sensitivity of fly photoreceptors. II. Dependence on facet lens F-number and rhabdomere type in Drosophila. Journal of Comparative Physiology A 2003, 189, 189–202. [Google Scholar] [CrossRef]
- Juusola, M.; Hardie, R.C. Light Adaptation in Drosophila Photoreceptors. J. Gen. Physiol. 2000, 117, 27–42. [Google Scholar] [CrossRef]
- Juusola, M.; Song, Z. How a fly photoreceptor samples light information in time. J. Physiol. 2017, 595, 5427–5437. [Google Scholar] [CrossRef]
- Song, Z.; Juusola, M. Refractory Sampling Links Efficiency and Costs of Sensory Encoding to Stimulus Statistics. J. Neurosci. 2014, 34, 7216–7237. [Google Scholar] [CrossRef]
- Song, Z.; Zhou, Y.; Feng, J.; Juusola, M. Multiscale ‘whole-cell’ models to study neural information processing – New insights from fly photoreceptor studies. J. Neurosci. Methods 2021, 357, 109156. [Google Scholar] [CrossRef]
- Gonzalez-Bellido, P.T.; Wardill, T.J.; Juusola, M. Compound eyes and retinal information processing in miniature dipteran species match their specific ecological demands. Proc. Natl. Acad. Sci. 2011, 108, 4224–4229. [Google Scholar] [CrossRef]
- Juusola, M. Linear and non-linear contrast coding in light-adapted blowfly photoreceptors. Journal of Comparative Physiology A 1993, 172, 511–521. [Google Scholar] [CrossRef]
- Ditchburn, R.W.; Ginsborg, B.L. Vision with a Stabilized Retinal Image. Nature 1952, 170, 36–37. [Google Scholar] [CrossRef] [PubMed]
- Riggs, L.A. , and Ratliff, F. ( 1952). The effects of counteracting the normal movements of the eye. J Opt Soc Am 42, 872–873.
- Geurten, B.R.H.; Jã¤Hde, P.; Corthals, K.; Gã¶Pfert, M.C. Saccadic body turns in walking Drosophila. Front. Behav. Neurosci. 2014, 8, 365–365. [Google Scholar] [CrossRef]
- Borst, A. Drosophila's View on Insect Vision. Curr. Biol. 2009, 19, R36–R47. [Google Scholar] [CrossRef] [PubMed]
- Franceschini, N.; Kirschfeld, K. Les phénomènes de pseudopupille dans l'œil composé deDrosophila. Kybernetik 1971, 9, 159–182. [Google Scholar] [CrossRef] [PubMed]
- Woodgate, J.L.; Makinson, J.C.; Rossi, N.; Lim, K.S.; Reynolds, A.M.; Rawlings, C.J.; Chittka, L. Harmonic radar tracking reveals that honeybee drones navigate between multiple aerial leks. iScience 2021, 24, 102499. [Google Scholar] [CrossRef]
- Schulte, P.; Zeil, J.; Stürzl, W. An insect-inspired model for acquiring views for homing. Biol. Cybern. 2019, 113, 439–451. [Google Scholar] [CrossRef] [PubMed]
- Boeddeker, N.; Dittmar, L.; Stürzl, W.; Egelhaaf, M. The fine structure of honeybee head and body yaw movements in a homing task. Proc. R. Soc. B: Boil. Sci. 2010, 277, 1899–1906. [Google Scholar] [CrossRef] [PubMed]
- Kirschfeld, K. [Neural superposition eye]. Fortschritte der Zoöl. 1973, 21, 229–57. [Google Scholar]
- Wardill, T.J.; List, O.; Li, X.; Dongre, S.; McCulloch, M.; Ting, C.-Y.; O’kane, C.J.; Tang, S.; Lee, C.-H.; Hardie, R.C.; et al. Multiple Spectral Inputs Improve Motion Discrimination in the Drosophila Visual System. Science 2012, 336, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Song, B.-M.; Lee, C.-H. Toward a Mechanistic Understanding of Color Vision in Insects. Front. Neural Circuits 2018, 12, 16. [Google Scholar] [CrossRef]
- Johnston, R.J.; Desplan, C. Stochastic Mechanisms of Cell Fate Specification that Yield Random or Robust Outcomes. Annu. Rev. Cell Dev. Biol. 2010, 26, 689–719. [Google Scholar] [CrossRef]
- Juusola, M. , and Hardie, R.C. (2001). Light adaptation in Drosophila photoreceptors: II. Rising temperature increases the bandwidth of reliable signaling J Gen Physiol 117, 27–42.
- Vasiliauskas, D.; Mazzoni, E.O.; Sprecher, S.G.; Brodetskiy, K.; Jr, R.J.J.; Lidder, P.; Vogt, N.; Celik, A.; Desplan, C. Feedback from rhodopsin controls rhodopsin exclusion in Drosophila photoreceptors. Nature 2011, 479, 108–112. [Google Scholar] [CrossRef]
- Dippé, M.A.Z.; Wold, E.H. Antialiasing through stochastic sampling. ACM SIGGRAPH Comput. Graph. 1985, 19, 69–78. [Google Scholar] [CrossRef]
- Field, G.D.; Gauthier, J.L.; Sher, A.; Greschner, M.; Machado, T.A.; Jepson, L.H.; Shlens, J.; Gunning, D.E.; Mathieson, K.; Dabrowski, W.; et al. Functional connectivity in the retina at the resolution of photoreceptors. Nature 2010, 467, 673–677. [Google Scholar] [CrossRef]
- Götz, K.G. Flight control in Drosophila by visual perception of motion. Kybernetik 1968, 4, 199–208. [Google Scholar] [CrossRef]
- Hengstenberg, R. Das augenmuskelsystem der stubenfliege musca domestica. Kybernetik 1971, 9, 56–77. [Google Scholar] [CrossRef] [PubMed]
- Colonnier, F.; Manecy, A.; Juston, R.; Mallot, H.; Leitel, R.; Floreano, D.; Viollet, S. A small-scale hyperacute compound eye featuring active eye tremor: application to visual stabilization, target tracking, and short-range odometry. Bioinspiration Biomimetics 2015, 10, 026002–026002. [Google Scholar] [CrossRef] [PubMed]
- Viollet, S.; Godiot, S.; Leitel, R.; Buss, W.; Breugnon, P.; Menouni, M.; Juston, R.; Expert, F.; Colonnier, F.; L'Eplattenier, G.; et al. Hardware Architecture and Cutting-Edge Assembly Process of a Tiny Curved Compound Eye. Sensors 2014, 14, 21702–21721. [Google Scholar] [CrossRef] [PubMed]
- Talley, J.; Pusdekar, S.; Feltenberger, A.; Ketner, N.; Evers, J.; Liu, M.; Gosh, A.; Palmer, S.E.; Wardill, T.J.; Gonzalez-Bellido, P.T. Predictive saccades and decision making in the beetle-predating saffron robber fly. Curr. Biol. 2023, 33, 2912–2924. [Google Scholar] [CrossRef] [PubMed]
- de Polavieja, G.G. (2006). Neuronal algorithms that detect the temporal order of events. Neural Comp 18, 2102-2121.
- Hassenstein, B. , and Reichardt, W. (1956). Systemtheoretische Analyse der Zeit-, Reihenfolgen- und Vorzeichenauswertung bei der Bewegungsperzeption des Rüsselkäfers Chlorophanus. Z Naturforsch 11b, 513-524.
- Barlow, H.B.; Levick, W.R. The mechanism of directionally selective units in rabbit's retina. PEDIATRICS 1965, 178, 477–504. [Google Scholar] [CrossRef]
- Grabowska, M.J.; Jeans, R.; Steeves, J.; van Swinderen, B. Oscillations in the central brain of Drosophila are phase locked to attended visual features. Proc. Natl. Acad. Sci. 2020, 117, 29925–29936. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Juusola, M. Intrinsic Activity in the Fly Brain Gates Visual Information during Behavioral Choices. PLOS ONE 2010, 5, e14455. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.; Cohen, D.; van Swinderen, B.; Tsuchiya, N. Integrated information structure collapses with anesthetic loss of conscious arousal in Drosophila melanogaster. PLOS Comput. Biol. 2021, 17, e1008722. [Google Scholar] [CrossRef]
- Chiappe, M.E.; Seelig, J.D.; Reiser, M.B.; Jayaraman, V. Walking Modulates Speed Sensitivity in Drosophila Motion Vision. Curr. Biol. 2010, 20, 1470–1475. [Google Scholar] [CrossRef]
- Maimon, G.; Straw, A.D.; Dickinson, M.H. Active flight increases the gain of visual motion processing in Drosophila. Nat. Neurosci. 2010, 13, 393–399. [Google Scholar] [CrossRef]
- Maye, A.; Hsieh, C.-H.; Sugihara, G.; Brembs, B. Order in Spontaneous Behavior. PLOS ONE 2007, 2, e443–e443. [Google Scholar] [CrossRef] [PubMed]
- van Hateren, J.H. A Unifying Theory of Biological Function. Biol. Theory 2017, 12, 112–126. [Google Scholar] [CrossRef] [PubMed]
- van Hateren, J.H. A theory of consciousness: computation, algorithm, and neurobiological realization. Biol. Cybern. 2019, 113, 357–372. [Google Scholar] [CrossRef] [PubMed]
- Okray, Z.; Jacob, P.F.; Stern, C.; Desmond, K.; Otto, N.; Talbot, C.B.; Vargas-Gutierrez, P.; Waddell, S. Multisensory learning binds neurons into a cross-modal memory engram. Nature 2023, 617, 777–784. [Google Scholar] [CrossRef]
- Juusola, M.; de Polavieja, G.G. The Rate of Information Transfer of Naturalistic Stimulation by Graded Potentials. J. Gen. Physiol. 2003, 122, 191–206. [Google Scholar] [CrossRef]
- de Polavieja, G.G. (2002). Errors drive the evolution of biological signalling to costly codes. J Theor Biol 214, 657-664.
- de Polavieja, G.G. (2004). Reliable biological communication with realistic constraints. Phys Rev E 70, 061910.
- Li, X.; Tayoun, A.A.; Song, Z.; Dau, A.; Rien, D.; Jaciuch, D.; Dongre, S.; Blanchard, F.; Nikolaev, A.; Zheng, L.; et al. Ca2+-Activated K+ Channels Reduce Network Excitability, Improving Adaptability and Energetics for Transmitting and Perceiving Sensory Information. J. Neurosci. 2019, 39, 7132–7154. [Google Scholar] [CrossRef]
- Shannon, C.E. (1948). A mathematical theory of communication. Bell Syst Technic J 27, 379–423, 623–656.
- Brenner, N.; Bialek, W.; Steveninck, R.d.R.v. Adaptive Rescaling Maximizes Information Transmission. Neuron 2000, 26, 695–702. [Google Scholar] [CrossRef]
- Maravall, M.; Petersen, R.S.; Fairhall, A.L.; Arabzadeh, E.; E Diamond, M. Shifts in Coding Properties and Maintenance of Information Transmission during Adaptation in Barrel Cortex. PLOS Biol. 2007, 5, e19. [Google Scholar] [CrossRef]
- Arganda, S.; Guantes, R.; de Polavieja, G.G. Sodium pumps adapt spike bursting to stimulus statistics. Nat. Neurosci. 2007, 10, 1467–1473. [Google Scholar] [CrossRef]
- Laughlin, S. A Simple Coding Procedure Enhances a Neuron's Information Capacity. 1981, 36, 910–912. [CrossRef]
- van Hateren, J.H. A theory of maximizing sensory information. Biol. Cybern. 1992, 68, 23–29. [Google Scholar] [CrossRef]
- Hopfield, J.J.; Brody, C.D. What is a moment? Transient synchrony as a collective mechanism for spatiotemporal integration. Proc. Natl. Acad. Sci. 2001, 98, 1282–1287. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, M.V.; Laughlin, S.B.; Dubs, A. Predictive coding: a fresh view of inhibition in the retina. Proc. R. Soc. London. Ser. B: Boil. Sci. 1982, 216, 427–459. [Google Scholar] [CrossRef]
- Poulet, J.F.A.; Hedwig, B. The Cellular Basis of a Corollary Discharge. Science 2006, 311, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Poulet, J.F.; Hedwig, B. New insights into corollary discharges mediated by identified neural pathways. Trends Neurosci. 2007, 30, 14–21. [Google Scholar] [CrossRef]
- Fujiwara, T.; Cruz, T.L.; Bohnslav, J.P.; Chiappe, M.E. A faithful internal representation of walking movements in the Drosophila visual system. Nat. Neurosci. 2017, 20, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Solvi, C.; Al-Khudhairy, S.G.; Chittka, L. Bumble bees display cross-modal object recognition between visual and tactile senses. Science 2020, 367, 910–912. [Google Scholar] [CrossRef]
- Badre, D.; Bhandari, A.; Keglovits, H.; Kikumoto, A. The dimensionality of neural representations for control. Curr. Opin. Behav. Sci. 2021, 38, 20–28. [Google Scholar] [CrossRef]
- Yellott, J.I. Spectral analysis of spatial sampling by photoreceptors: Topological disorder prevents aliasing. Vis. Res. 1982, 22, 1205–1210. [Google Scholar] [CrossRef]
- Wikler, K.; Rakic, P. Distribution of photoreceptor subtypes in the retina of diurnal and nocturnal primates. J. Neurosci. 1990, 10, 3390–3401. [Google Scholar] [CrossRef]
- Kim, Y.J.; Peterson, B.B.; Crook, J.D.; Joo, H.R.; Wu, J.; Puller, C.; Robinson, F.R.; Gamlin, P.D.; Yau, K.-W.; Viana, F.; et al. Origins of direction selectivity in the primate retina. Nat. Commun. 2022, 13, 1–20. [Google Scholar] [CrossRef]
- Yu, W.-Q.; Swanstrom, R.; Sigulinsky, C.L.; Ahlquist, R.M.; Knecht, S.; Jones, B.W.; Berson, D.M.; Wong, R.O. Distinctive synaptic structural motifs link excitatory retinal interneurons to diverse postsynaptic partner types. Cell Rep. 2023, 42, 112006. [Google Scholar] [CrossRef] [PubMed]
- Razban Haghighi, K. (2023). The Drosophila visual system: a super-efficient encoder. PhD (University of Sheffield).
- Kapustjansky, A.; Chittka, L.; Spaethe, J. Bees use three-dimensional information to improve target detection. Sci. Nat. 2009, 97, 229–233. [Google Scholar] [CrossRef] [PubMed]
- van Swinderen, B. Attention in Drosophila. 2011, 99, 51–85. [CrossRef]
- Chittka, L.; Spaethe, J. Visual search and the importance of time in complex decision making by bees. Arthropod-Plant Interactions 2007, 1, 37–44. [Google Scholar] [CrossRef]
- Juusola, M.; Song, Z.; Hardie, R. Phototransduction Biophysics. 2022, 2758–2776. [CrossRef]
- Hochstrate, P.; Hamdorf, K. Microvillar components of light adaptation in blowflies. J. Gen. Physiol. 1990, 95, 891–910. [Google Scholar] [CrossRef]
- Pumir, A.; Graves, J.; Ranganathan, R.; Shraiman, B.I. Systems analysis of the single photon response in invertebrate photoreceptors. Proc. Natl. Acad. Sci. 2008, 105, 10354–10359. [Google Scholar] [CrossRef] [PubMed]
- Parker, I.; Miledi, R. The intracellular pupil mechanism and photoreceptor signal: noise ratios in the fly Lucilia cuprina. Proc. R. Soc. London. Ser. B: Boil. Sci. 1987, 231, 415–435. [Google Scholar] [CrossRef]
- Mishra, P.; Socolich, M.; Wall, M.A.; Graves, J.; Wang, Z.; Ranganathan, R. Dynamic Scaffolding in a G Protein-Coupled Signaling System. Cell 2007, 131, 80–92. [Google Scholar] [CrossRef]
- Scott, K.; Sun, Y.; Beckingham, K.; Zuker, C.S. Calmodulin Regulation of Drosophila Light-Activated Channels and Receptor Function Mediates Termination of the Light Response In Vivo. Cell 1997, 91, 375–383. [Google Scholar] [CrossRef]
- Liu, C.-H.; Satoh, A.K.; Postma, M.; Huang, J.; Ready, D.F.; Hardie, R.C. Ca2+-Dependent Metarhodopsin Inactivation Mediated by Calmodulin and NINAC Myosin III. Neuron 2008, 59, 778–789. [Google Scholar] [CrossRef]
- Wong, F.; Knight, B.W. Adapting-bump model for eccentric cells of Limulus. J. Gen. Physiol. 1980, 76, 539–557. [Google Scholar] [CrossRef]
- Wong, F.; Knight, B.W.; A Dodge, F. Dispersion of latencies in photoreceptors of Limulus and the adapting-bump model. J. Gen. Physiol. 1980, 76, 517–537. [Google Scholar] [CrossRef] [PubMed]
- Hardie, R.C. , and Postma, M. (2008). Phototransduction in microvillar photoreceptors of Drosophila and other invertebrates. In The senses: a comprehensive reference. Vision, A.I. Basbaum, A. Kaneko, G.M. Shepherd, and G. Westheimer, eds. (Academic), pp. 77-130.
- Postma, M.; Oberwinkler, J.; Stavenga, D.G. Does Ca2+ Reach Millimolar Concentrations after Single Photon Absorption in Drosophila Photoreceptor Microvilli? Biophys. J. 1999, 77, 1811–1823. [Google Scholar] [CrossRef] [PubMed]
- Hardie, R.C. INDO-1 Measurements of Absolute Resting and Light-Induced Ca2+Concentration inDrosophilaPhotoreceptors. J. Neurosci. 1996, 16, 2924–2933. [Google Scholar] [CrossRef] [PubMed]
- Faivre, O.; Juusola, M. Visual Coding in Locust Photoreceptors. PLOS ONE 2008, 3, e2173. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



