Submitted:
15 August 2023
Posted:
16 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Voltammetry
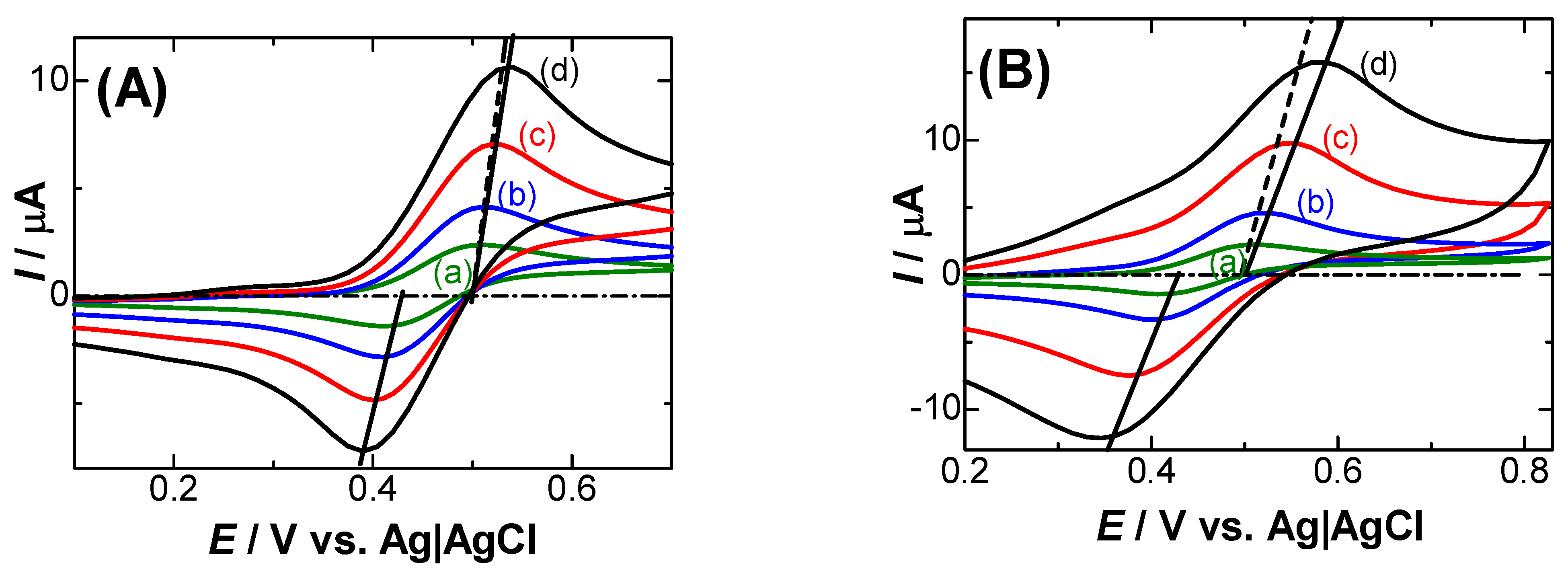
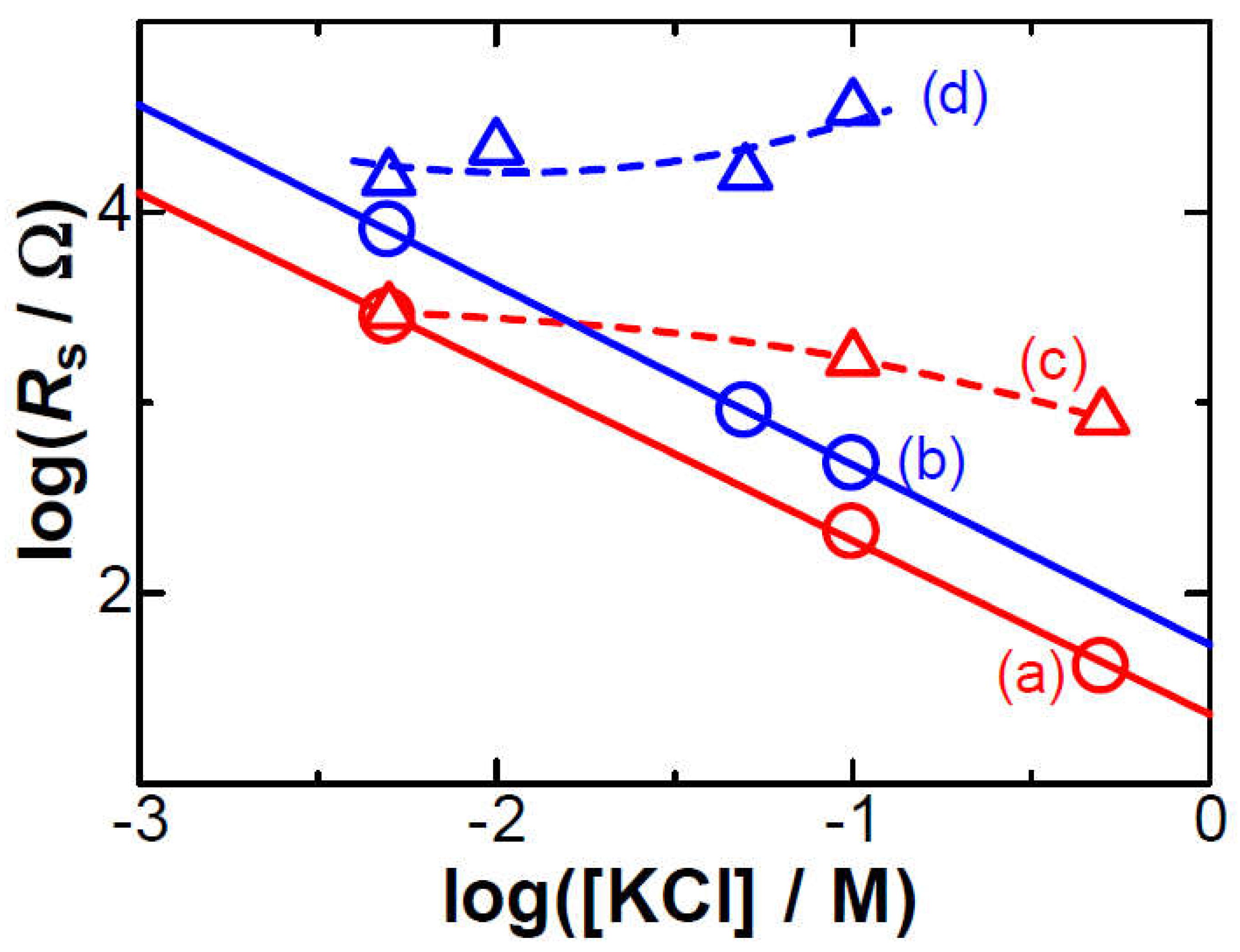
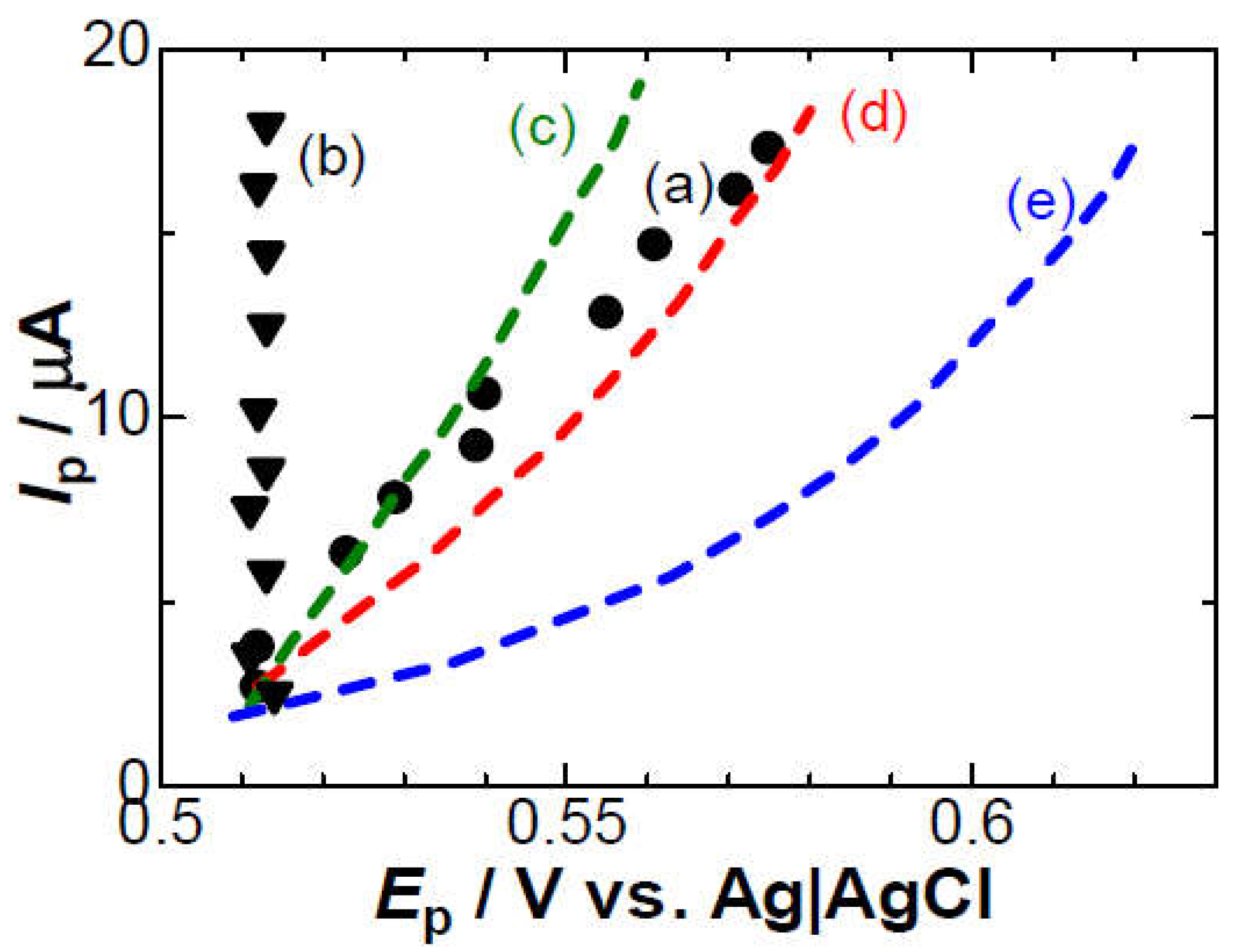
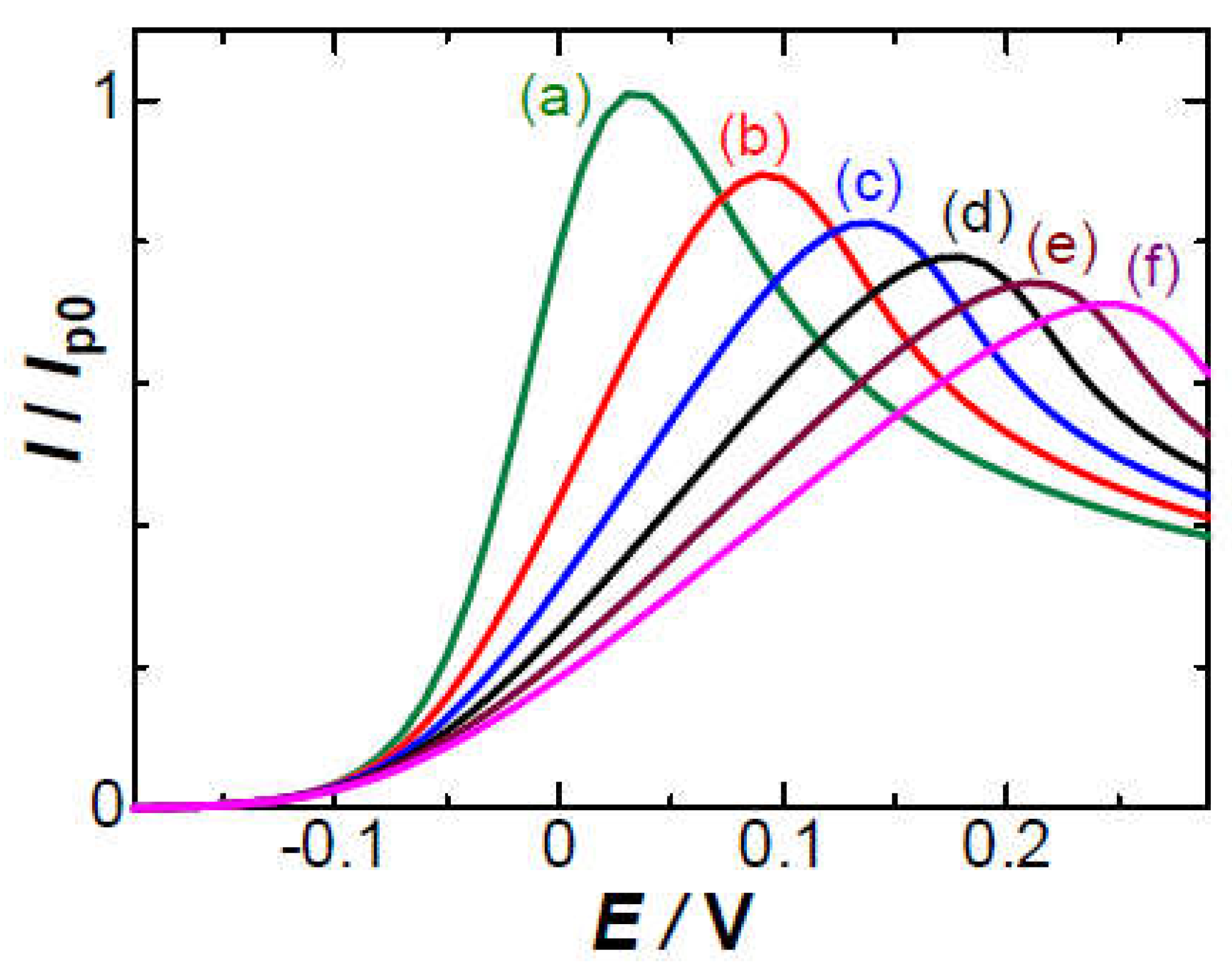
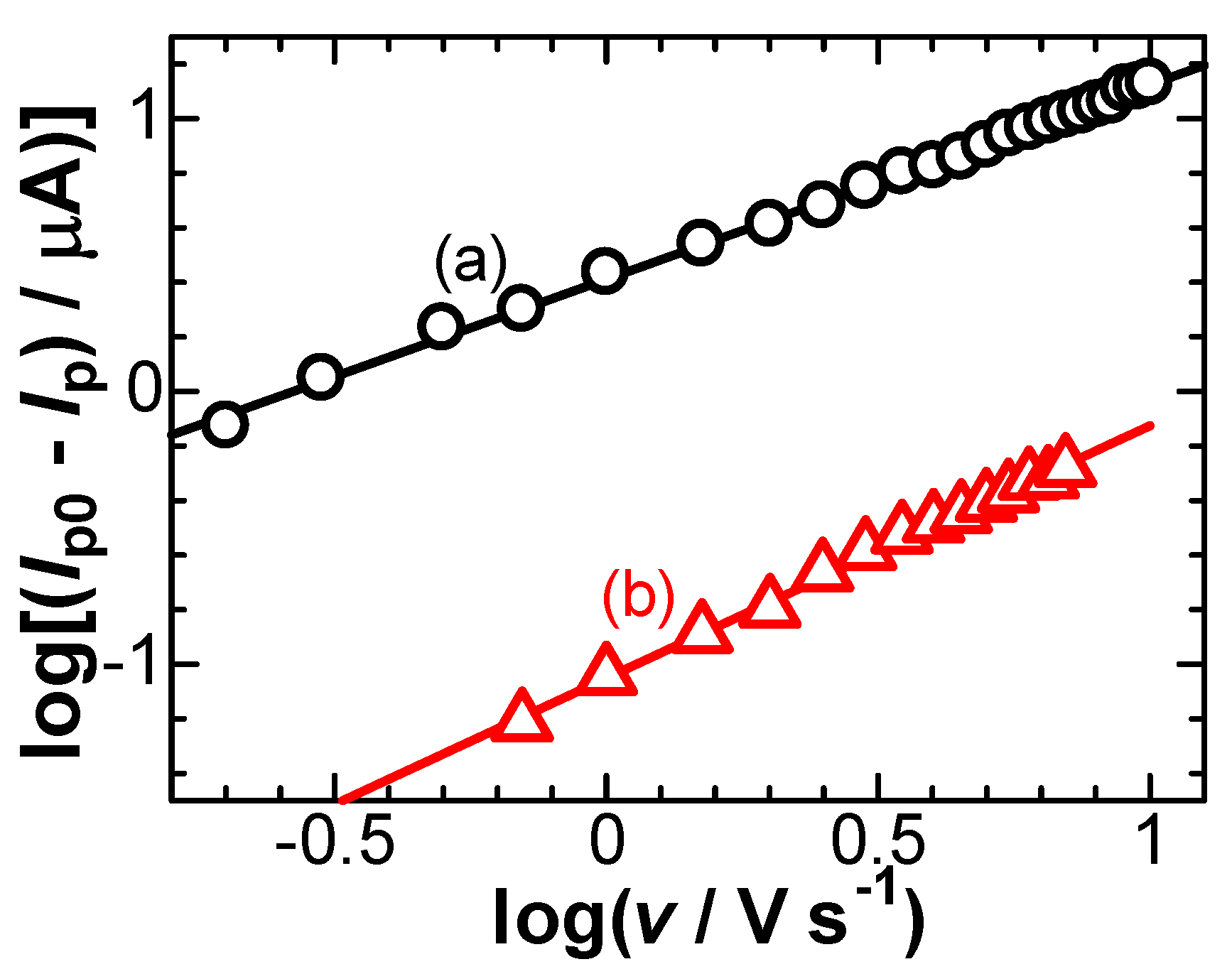
3. Results
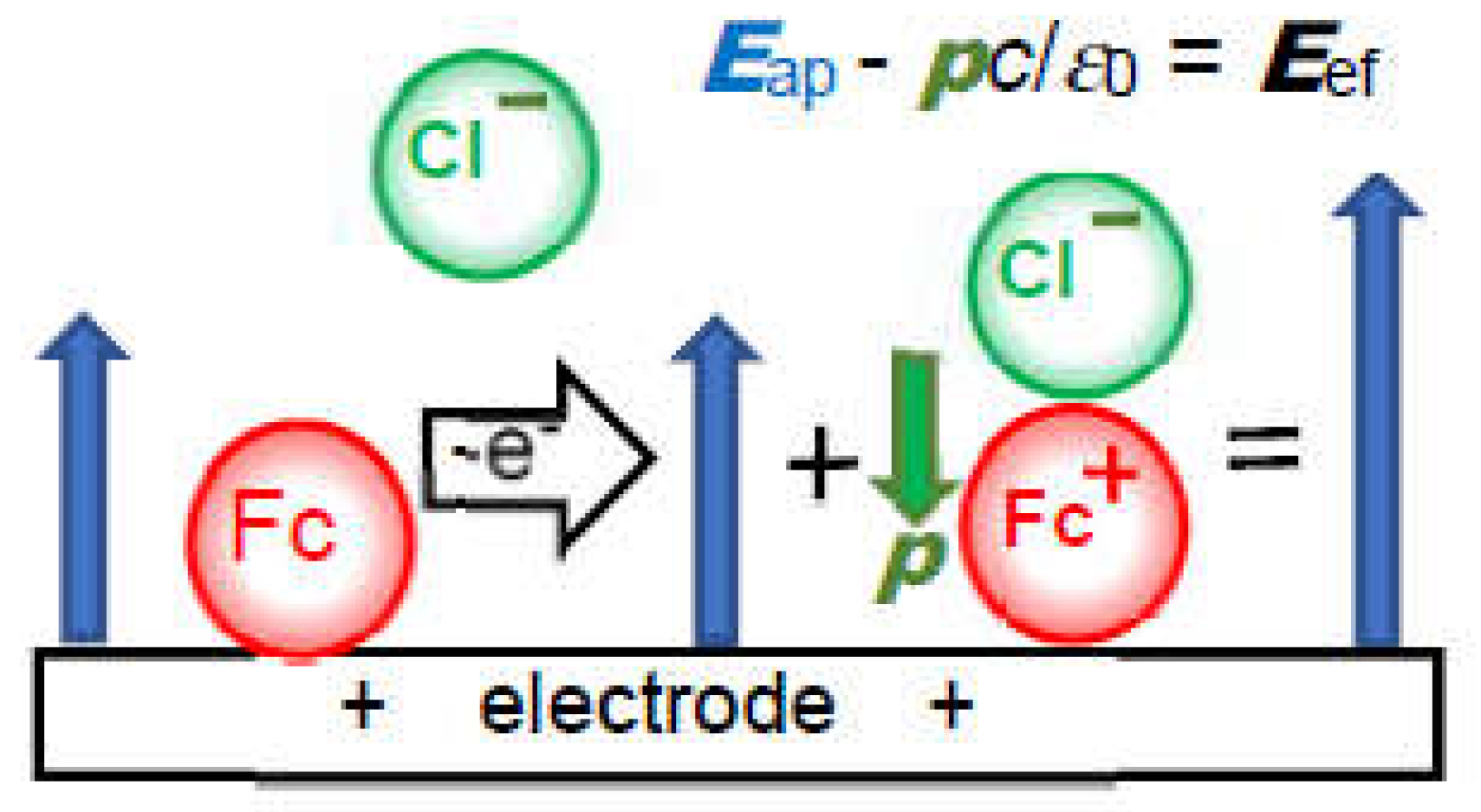
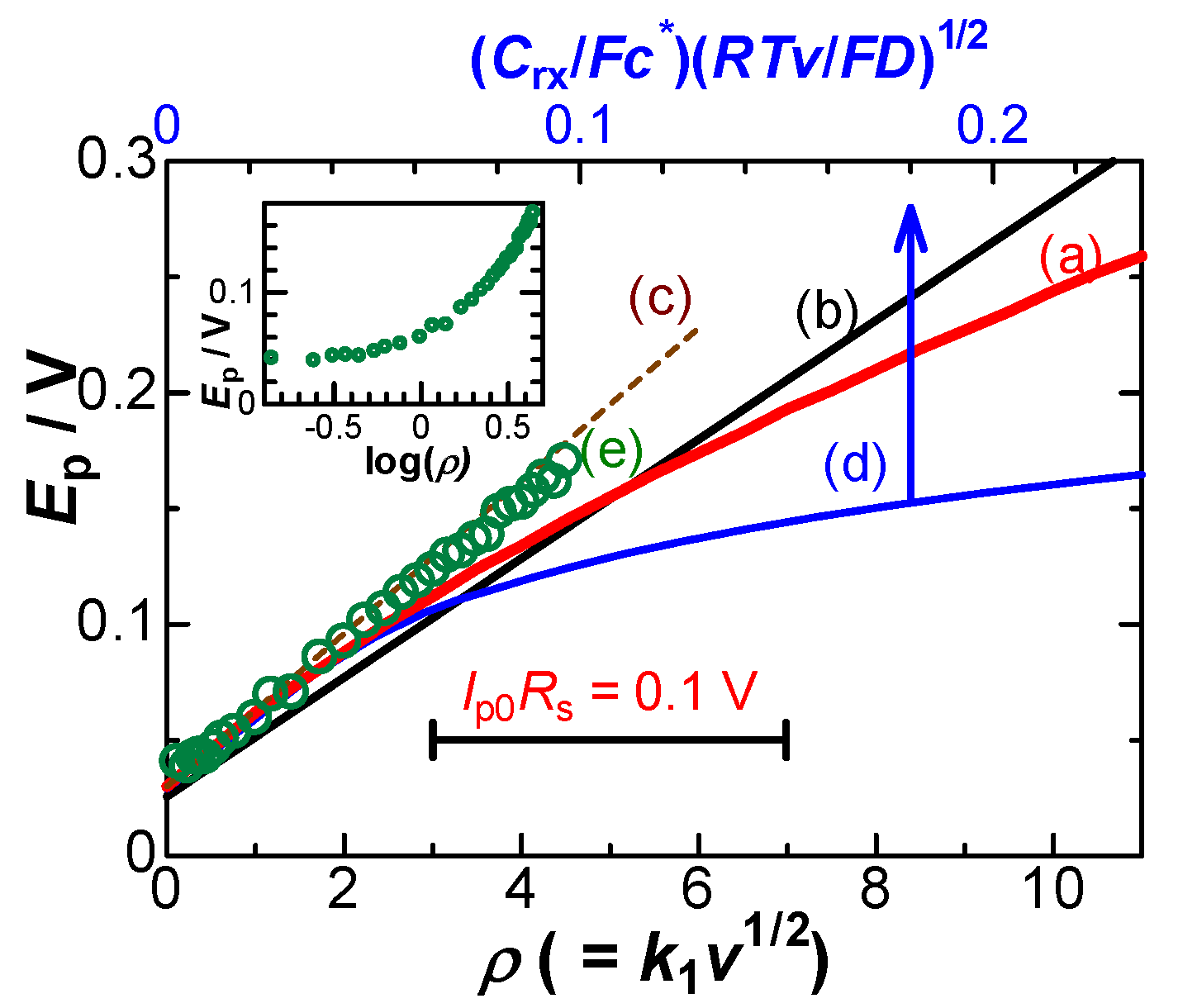
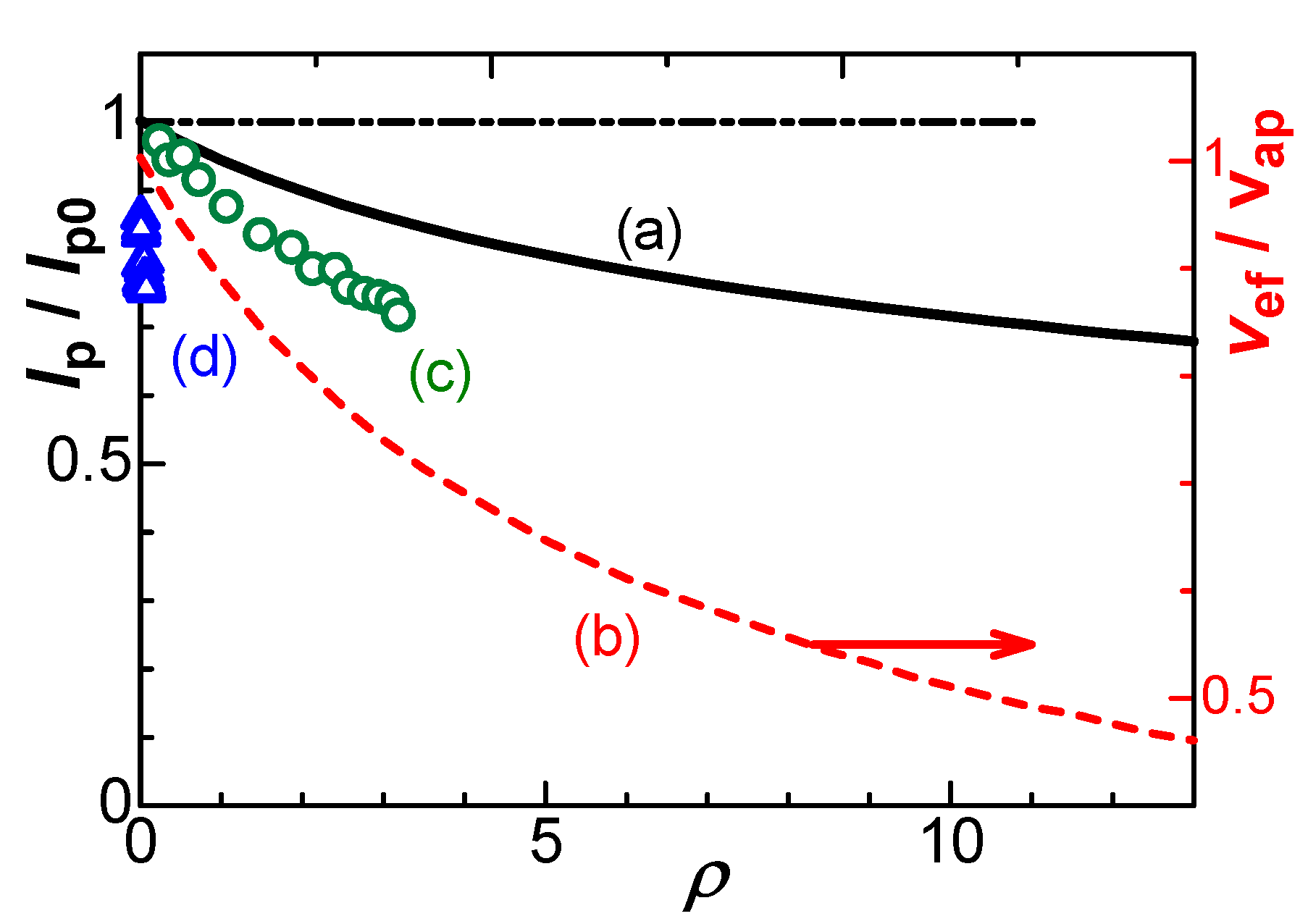
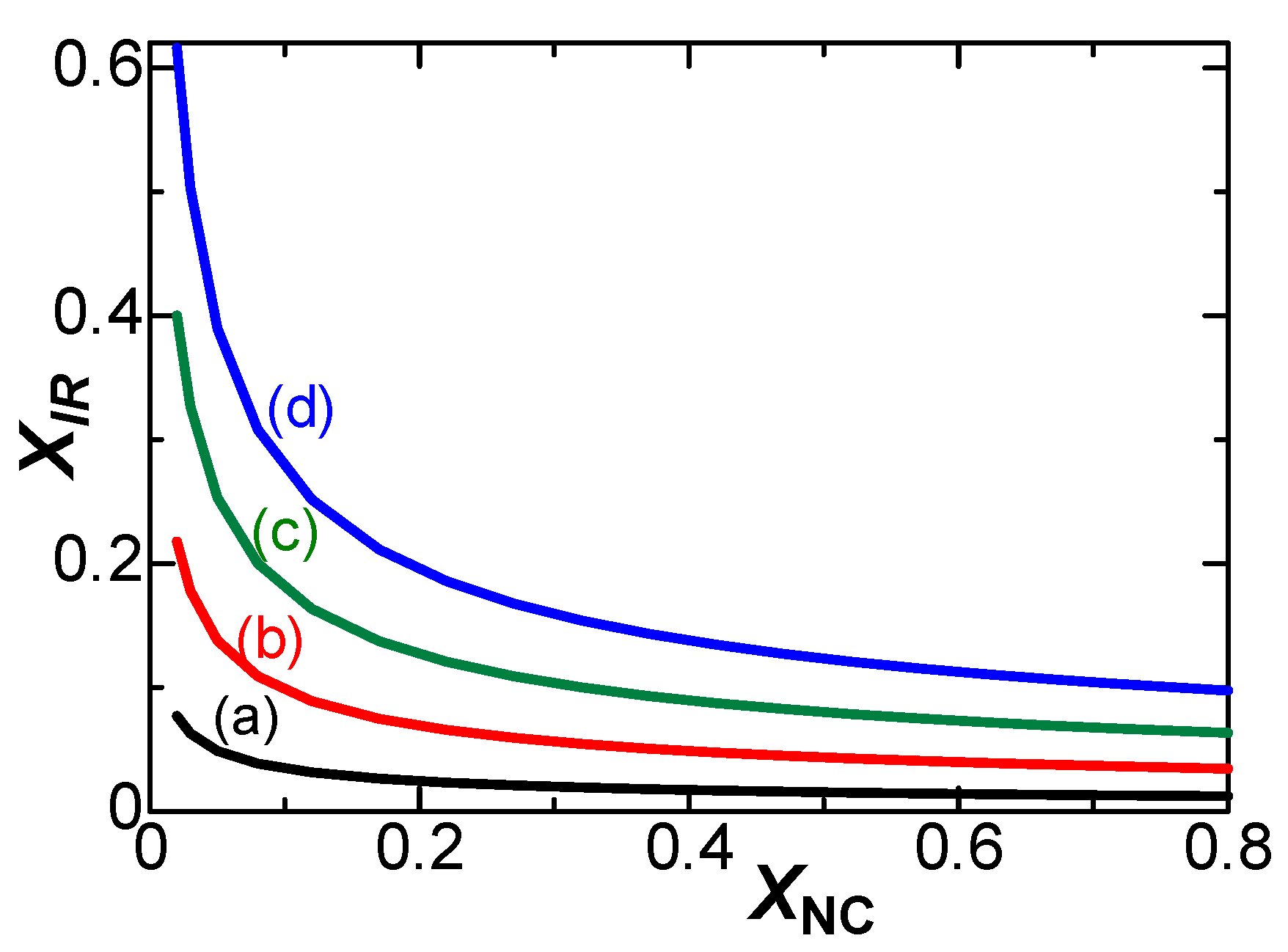
4. Theory of effects of IR-drop
5. Discussion
6. Conclusion
6. Supporting information
References
- A.J. Bard. L.R. Faulkner, Electrochemical Methods; Fundamentals and Applications, John Wiley & Sons, New York, 2nd. Ed., 2001, pp. 331–332.
- A.J. Bard. L.R. Faulkner, Electrochemical Methods; Fundamentals and Applications, John Wiley & Sons, New York, 2nd. Ed., 2001, pp. 170–176.
- J.O. Howell, J.M. Goncalves, C. Amatore, L. Klasinc, R.M. Wightman, J.K. Kochi, J. Am. Chem. Sot., 1984, 106, 3968.
- C. Amatore, C. Lefrou, J. Electroanal. Chem., 1990, 296, 335–358.
- M.I. Montenegro, D. Pletcher, J. Electroanal. Chem., 1986, 200, 371.
- A. Fitch, D.H. Evans, J. ElectroanaI. Chem., 1986, 202, 83.
- C. Amatore, E. Maisonhaute, G. Simonneau, Electrochem. Commun., 2000, 2, 81–84.
- C.P. Andrieux, D. Garreau, P. Hapiot, J. Pinson, J.M. Saveant, J. Eleetroanal. Chem., 1988, 243, 321–335.
- G. Wosiak, D. Coelho, E. B. Carneiro-Neto, E. C. Pereira, M. C. Lopes, Anal. Chem., 2020, 92, 15412-15419.
- C. Amatore, A. Oleinick, I. Svir, Anal. Chem., 2008, 80, 7947-7956.
- J.G. Roberts, L.A. Sombers, Anal. Chem. 2018, 90, 490–504.
- J.E. Baur, E.W. Kristensen, L.J. May, D.J. Wiedemann, R.M. Wightman, Anal. Chem., l988, 60, 1268–1272.
- B.J. Venton, Q. Cao, Analyst, 2020, 145, 1158.
- P. Puthongkham, B.J. Venton, Analyst, 2020, 145, 1087–1102.
- BioLogic, EC-Lab – Application Note #41, 07/2012, CV Sim: Simulation of the simple redox reaction (E) Part II: The effect of ohmic drop and double layer capacitance.
- K.J. Aoki, J. Chen, Tips of Voltammetry, IntehOpen, 2018, pp. 1–19.
- K.J. Aoki, J. Chen, Y. Liu, B.Jia, J. Electroanal. Chem., 2020, 856, 113609.
- H. Matsuda, Y. Ayabe, Z. Elektrochmie. 1955, 59, 494–503.
- K.J Aoki, J. Chen, X. Zeng, Z. Wang, RSC Adv. 2017, 7, 22501–22501.
- K.J. Aoki, J. Chen. P. Tang, J. Phys. Chem. C, 2018, 122, 16727–16732.
- K.J. Aoki, J. Chen, R. Wang, Electroanalysis, 2019, 31, 1–9.
- K.J. Aoki, T. Peng, J. Chen, Am. J. Anal. Chem., 2019, 10, 286–295.
- R. Wang, K.J. Aoki, J. Chen, Electrochem, 2022, 3, 397–406.
- K.J. Aoki, C. Zhang, J. Chen, T. Nishiumi, J. Electroanal. Chem., 2013, 706, 40–47.
- P. Sun, M.V. Mirkin, Anal. Chem., 2006, 78, 6526–6534.
- J. Velmurugan, P. Sun, M. V. Mirkin, J. Phys. Chem. C, 2009, 113, 459–464.
- J. F. Smalley, S. W. Feldberg, C. E. D. Chidsey, M. R. Linford, M. D. Newton, Y.-P. Liu, J. Phys. Chem., 1995, 99, 13141–13149.
- N. Nioradze, J. Kim, S. Amemiya, Anal. Chem., 2011, 83, 828–835.
- Y. Li, D. Bergman, B. Zhang, Anal. Chem., 2009, 81, 5496–5502.
- Y. Zhang, J. Zhou, L. Lin, Z. Lin, Electroanal., 2008, 20, 1490–1494.
- P. Atkins, J.de Paula, Atkin's Physical Chemistry, 10th ed., Oxford University Press, pp, 799–802.
- K. J. Aoki, R. He, J. Chen, Electrochem, 2021, 2, 71–82.
- K.J. Aoki, R. He, J. Chen, Electrochem, 2021, 2, 631–642.
- Y. Zhou, P. Yang, C. Yuan, Y. Huo, Chem. Eng. Trans., 2013, 33, 559–564.
- R.S. Sorbello, Mater. Res. Soc. Conference Proceedings 1996, 427, 73–81.
- B.R. Putra, K.J. Aoki, J. Chen, F. Marken, Langmuir, 2019, 35, 2055–2065.
- P. Song, A.C. Fisher, J.D. Wadhawan, J.J. Cooper, H.J. Ward, N.S. Lawrence, RSC Adv., 2016, 6, 70237–70242.
- H. M. Nassef, A.-E. Radi, C.K. O'Sullivan, Electrochem. Commun., 2006, 8, 1719–1725.
- A.J. Bard. L.R. Faulkner, Electrochemical Methods; Fundamentals and Applications, John Wiley & Sons, New York, 2nd. Ed., 2001, pp. 29.
- A.Molina, J. Gonzalez, E. Laborda, R.G. Compton, Phys. Chem. Chem. Phys., 2013, 15, 2381–2388.
- K.J Aoki, S. Taniguchi, J. Chen, Omega ACS, 2020, 5, 29447–29452.
- K. Aoki, Electroanalysis, 2005, 17, 1379–1383.
- M. Abramowitz, I.A. Stegun, Handbook of Mathematical Functions, National Bureau of Standards, 1964, pp. 11, 3.3.7.
- J.S. Newman, Electrochemical Systems, Prentice-Hall Inc. Englewood Cliff, N. J. Chapter 18, pp. 340–345.
| Range of v | Advantages | Risks |
|---|---|---|
| (I) < 0.2 V s-1 |
Small background currents Easy extraction of diffusion currents Usage of low-cost potentiostats Possibility of theoretical analysis |
Misleading kinetic reaction mechanisms Time consumption |
| (II) < 5 V s-1 |
Possibility of subtraction of IR-drop Possibility of determining reaction mechanisms Evaluation of heterogeneous kinetics Comparison of the results with those by other rapid electrochemical methods Commercially available potentiostats |
Discussion required for peak shifts Deformation of waveform Limitation to microelectrodes in order to prevent large currents |
| (III) < 500 V s-1 |
Detection of kinetics with milli-second orders such as neurotransmitters | Empirical search for detecting conditions A loss of theoretical support |
| variables | Ep | Ip0- Ip | ||
|---|---|---|---|---|
| IR | NC | IR | NC | |
| c* | c* | 1 | c*3/2 | c* |
| A(disk) | A1/2 | 1 | A5/4 | A |
| v | v1/2 | v1/2 | v3/4 | v |
| Rs | Rs | 1 | Rs1/2 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




