Submitted:
12 August 2023
Posted:
15 August 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chickens
2.2. Viruses
2.3. Generation of novel fpIBD1 recombinants
2.4. Experimental design
2.5. Sample processing
2.5.1. RNA extraction
2.5.2. Frozen sections for immunohistochemical staining
2.5.3. H&E staining
2.6. Real-time quantitative RT-PCR
2.7. Construction of standard curves for quantitative PCR and RT-PCR assays
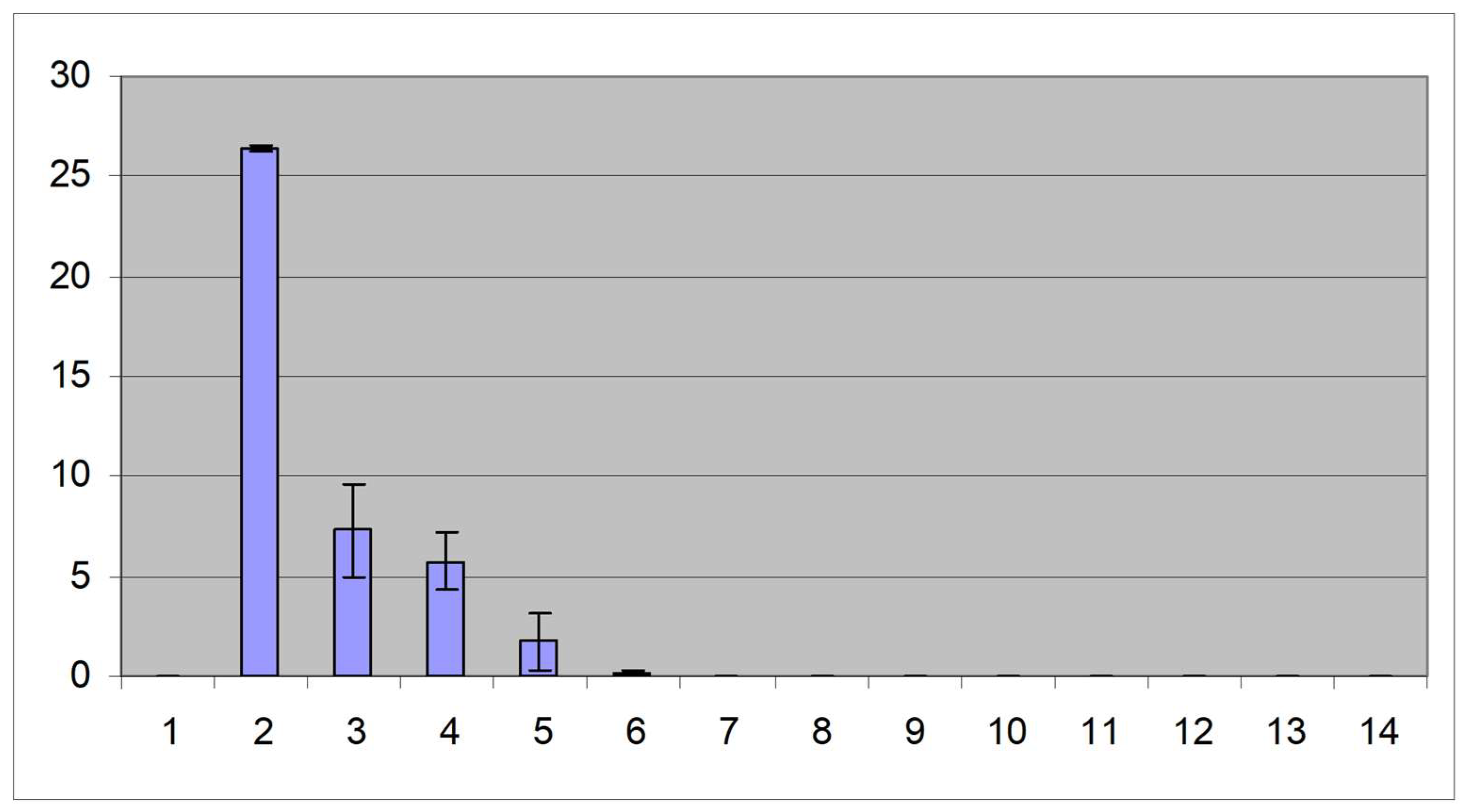
3. Results
3.1. Protection from IBDV challenge by the recombinant vaccine fpIBD1 and the new viral constructs
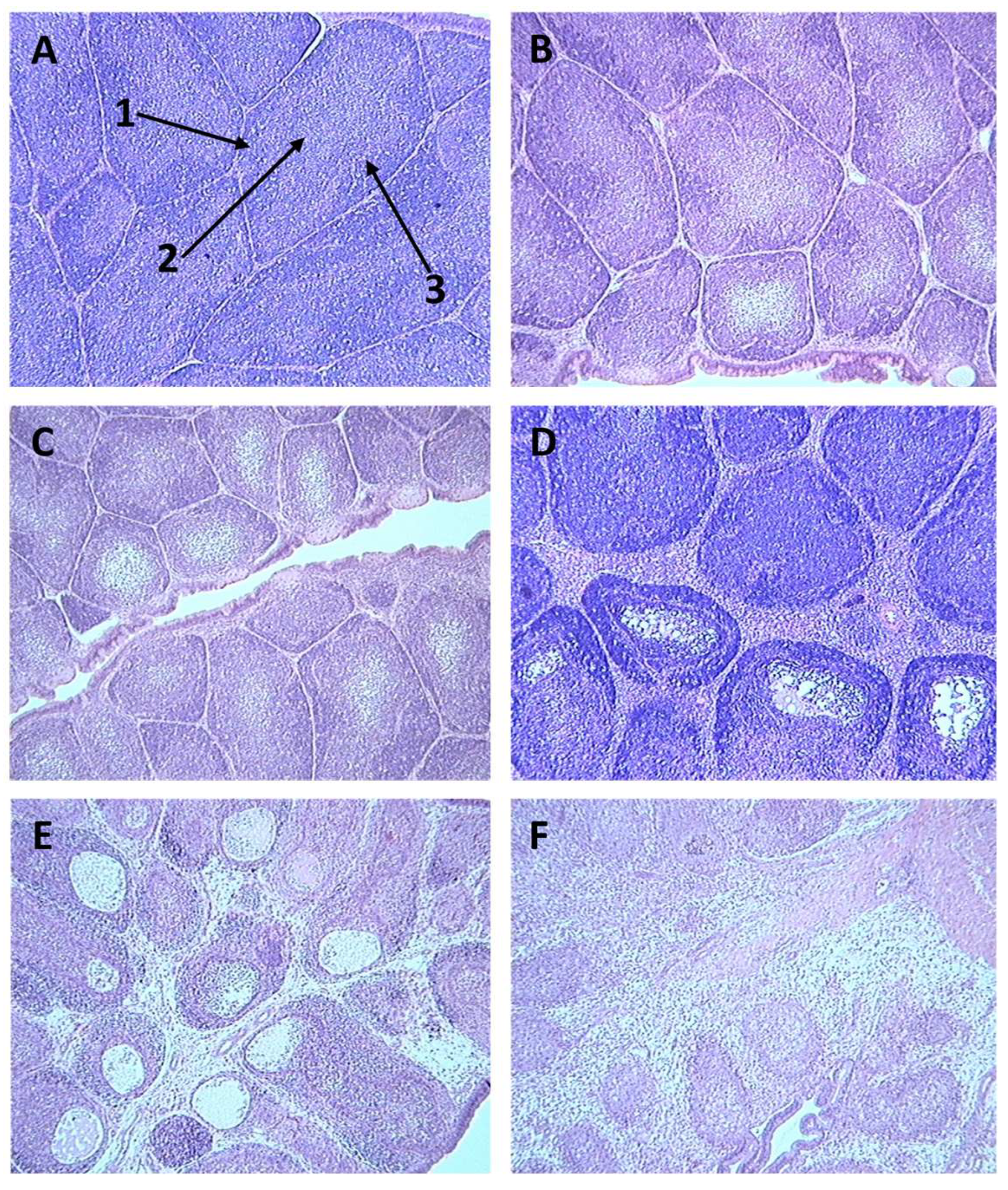
3.2. Detection of IBDV using immunohistochemistry
3.3. Detection of IBDV using real-time quantitative RT-PCR
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cosgrove, A. An apparently new disease of chickens - Avian Nephrosis. Avian Dis. 1962, 6, 385–389. [Google Scholar] [CrossRef]
- Eterradossi, N.; Saif, Y.M. Infectious bursal disease. In Diseases of Poultry, 14th ed.; David E. Swayne, Martine Boulianne, Catherine M. Logue, Larry R. McDougald, Venugopal Nair, David L. Suarez, Sjaak de Wit, Tom Grimes, Deirdre Johnson, Michelle Kromm, Teguh Yodiantara Prajitno, Ian Rubinoff, Guillermo Zavala, Ed.; John Wiley & Sons, Inc., 2020; pp. 257–283. [Google Scholar]
- Gao, H.; Wang, Y.; Gao, L.; Zheng, S.J. Genetic Insight into the Interaction of IBDV with Host-A Clue to the Development of Novel IBDV Vaccines. Int J Mol Sci. 2023, 24, 8255. [Google Scholar] [CrossRef] [PubMed]
- Van der Berg, T.P.; Gonze, M.; Meulemans, G. Acute infectious bursal disease in poultry: Isolation and characterisation of a highly virulent strain. Avian Pathol. 1991, 20, 133–143. [Google Scholar] [CrossRef]
- Hein, R.; Koopman, R.; García, M.; Armour, N.; Dunn, J.R.; Barbosa, T.; Martinez, A. Review of Poultry Recombinant Vector Vaccines. Avian Dis. 2021, 65, 438–452. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.; Edbauer, C.; Rey, S.A.; Bouquet, J.F.; Norton, E.; Goebel, S.; et al. Newcastle disease virus fusion protein expressed in a fowlpox virus recombinant confers protection in chickens. J. Virol. 1990, 64, 1441–1450. [Google Scholar] [CrossRef]
- Criado, M.F.; Bertran, K.; Lee, D.H.; Killmaster, L.; Stephens, C.B.; Spackman, E.; Sa E Silva, M.; Atkins, E.; Mebatsion, T.; Widener, J.; Pritchard, N.; King, H.; Swayne, D.E. Efficacy of novel recombinant fowlpox vaccine against recent Mexican H7N3 highly pathogenic avian influenza virus. Vaccine. 2019, 37, 2232–2243. [Google Scholar] [CrossRef]
- Qingzhong, Y.; Barrett, T.; Brown, T.D.; Cook, J.K.; Green, P.; Skinner, M.A.; et al. Protection against turkey rhinotracheitis pneumovirus (TRTV) induced by a fowlpox virus recombinant expressing the TRTV fusion glycoprotein (F). Vaccine 1994, 12, 569–573. [Google Scholar] [CrossRef]
- Lee, L.F.; Bacon, L.D.; Yoshida, S.; Yanagida, N.; Zhang, H.M.; Witter, R.L. The efficacy of recombinant fowlpox vaccine protection against Marek's disease: Its dependence on chicken line and B haplotype. Avian Dis. 2004, 48, 129–137. [Google Scholar] [CrossRef]
- Bayliss, C.D.; Peters, R.W.; Cook, J.K.; Reece, R.L.; Howes, K.; Binns, M.M.; et al. A recombinant fowlpox virus that expressed the VP2 antigen of infectious bursal disease virus induces protection against mortality caused by the virus. Arch. Virol. 1991, 120, 193–205. [Google Scholar] [CrossRef]
- Fahey, K.J.; Erny, K.; Crooks, J. A conformational immunogen on VP2 of infectious bursal disease virus that induce virus-neutralizing antibodies that passively protect chickens. J. Gen. Virol. 1989, 70, 1473–1481. [Google Scholar] [CrossRef]
- Laidlaw, S.M.; Skinner, M.A. Comparison of the genome sequence of FP9, an attenuated, tissue culture-adapted European strain of Fowlpox virus, with those of those virulent American and European viruses. J. Gen. Virol. 2004, 85, 305–322. [Google Scholar] [CrossRef] [PubMed]
- Shaw, I.; Davison, T.F. Protection from IBDV-induced bursal damage by a recombinant fowlpox vaccine, fpIBD1, is dependent on the titre of challenge virus and chicken genotype. Vaccine 2000, 18, 3230–3241. [Google Scholar] [CrossRef] [PubMed]
- Schneider, K.; Puehler, F.; Baeuerle, D.; Elvers, S.; Staeheli, P.; Kaspers, B.; et al. cDNA cloning of biologically active chicken interleukin-18. J Interferon Cyt. Res. 2000, 20, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Afonso, C.L.; Tulman, E.R.; Lu, Z.; Zsak, L.; Kutish, G.F.; Rock, D.L. The genome of fowlpox virus. J. Virol. 2000, 74, 3815–3831. [Google Scholar] [CrossRef]
- Laidlaw, S.M.; Anwar, M.A.; Thomas, W.; Green, P.; Shaw, K.; Skinner, M.A. Fowlpox virus encodes nonessential homologs of cellular alpha-SNAP, PC- 1, and an orphan human homolog of a secreted nematode protein. J. Virol. 1998, 72, 6742–6751. [Google Scholar] [CrossRef]
- Nooruzzaman, M.; Hossain, I.; Rahman, M.M.; Uddin, A.J.; Mustari, A.; Parvin, R.; Chowdhury, E.H.; Islam, M.R. Comparative pathogenicity of infectious bursal disease viruses of three different genotypes. Microb Pathog. 2022, 169, 105641. [Google Scholar] [CrossRef]
- Trapp, J.; Rautenschlein, S. Infectious bursal disease virus' interferences with host immune cells: What do we know? Avian Pathol. 2022, 51, 303–316. [Google Scholar] [CrossRef]
- Wyatt, L.S.; Earl, P.L.; Moss, B. Generation of Recombinant Vaccinia Viruses. Curr Protoc Protein Sci. 2017, 89, 5–13. [Google Scholar] [CrossRef]
- Rothwell, C.J.; Vervelde, L.; Davison, T.F. Identification of chicken Bu-1 alloantigens using the monoclonal antibody AV20. Vet Immunol Immunopathol. 1996, 55, 225–234. [Google Scholar] [CrossRef]
- Techera, C.; Tomás, G.; Panzera, Y.; Banda, A.; Perbolianachis, P.; Pérez, R.; Marandino, A. Development of real-time PCR assays for single and simultaneous detection of infectious bursal disease virus and chicken anemia virus. Mol Cell Probes. 2019, 43, 58–63. [Google Scholar] [CrossRef]
- Bayliss, C.D.; Spies, U.; Shaw, K.; Peters, R.W.; Papageorgiou, A.; Muller, H.; et al. A comparison of the sequence of segment A of four infectious bursal disease virus strains and identification of a variable region in the VP2. J. Gen. Virol. 1990, 71, 1303–1312. [Google Scholar] [CrossRef] [PubMed]
- Muskett, J.C.; Hopkins, I.G.; Edwards, K.R.; Thornton, D.H. Comparison of two infectious bursal disease strains: Efficacy and potential hazards in susceptible and maternally immune birds. Vet. Rec. 1979, 104, 332–334. [Google Scholar] [CrossRef] [PubMed]
| Virus | Final concentration (pfu/ml) |
|---|---|
| fpIBD1 | 1X109 |
| fpIBD1Δ073 | 2X109 |
| fpIBD1Δ214 | 1X109 |
| fpIBD1::IL-18 | 2X108 |
| fpIBD1Δ073::IL-18 | 2X108 |
| fpIBD1Δ214::IL-18 | 3X108 |
| RNA target | Primer/Probe* | Sequence (5’-3’) |
|---|---|---|
| 28S | F | GGC GAA GCC AGA GGA AAC T |
| R | GAC GAC CGA TTT GCA CGT C | |
| Probe | AGG ACC GCT ACG GAC CTC CAC CA | |
| IBDV (VP2) | F | GAG GTG GCC GAC CTC AAC T |
| R | AGC CCG GAT TAT GTC TTT GAA G | |
| Probe | TCC CCT GAA GAT TGC AGG AGC ATT TG |
| Vaccination | IBDV challenge | Bursal Damage Score | |||||
|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 | ||
| - | - | 5 | - | - | - | - | - |
| - | + | - | - | - | 1 | 3 | 1 |
| fpIBD1 | + | 1 | 3 | 1 | - | - | - |
| fpIBD1Δ073 | + | 2 | 2 | 1 | - | - | - |
| fpIBD1Δ214 | + | 4 | 1 | - | - | - | - |
| fpIBD1::IL-18 | + | 5 | - | - | - | - | - |
| fpIBD1Δ073::IL-18 | + | 5 | - | - | - | - | - |
| fpIBD1Δ214::IL-18 | + | 5 | - | - | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




