Submitted:
30 July 2023
Posted:
01 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Quantum dot synthesis and solubilization
2.2. Quantum dot characterization
2.3. Animals
2.4. Estimation of the acute toxicity of quantum dots in vivo
2.5. Quantum dot immunotoxicity
2.5.1. Assessment of alterations in the spleen, thymus, and bone marrow
2.5.2. Assessment of changes in the serum cytokine concentrations
3. Results
3.1. Quantum dot synthesis, solubilization, and characterization
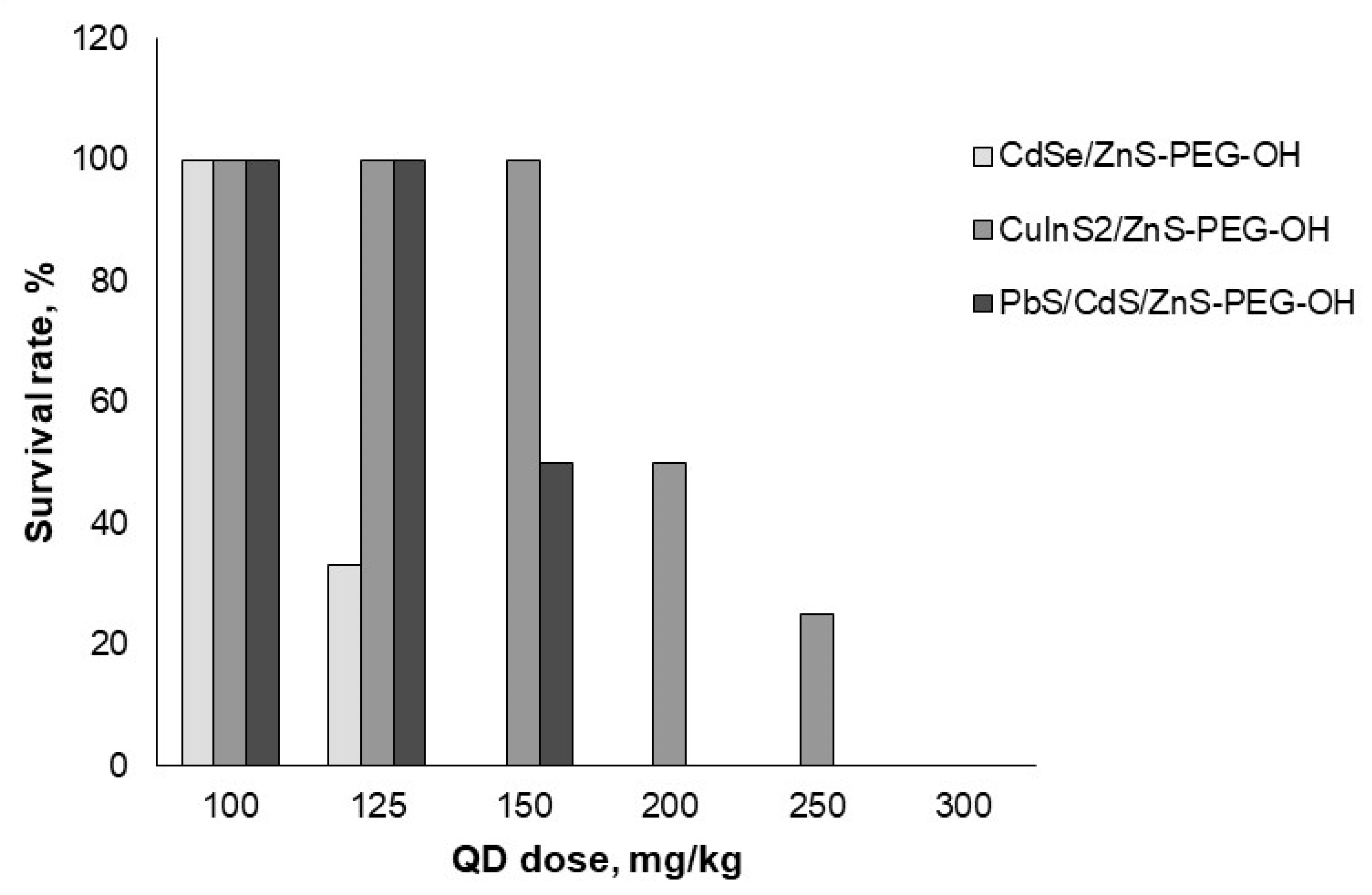
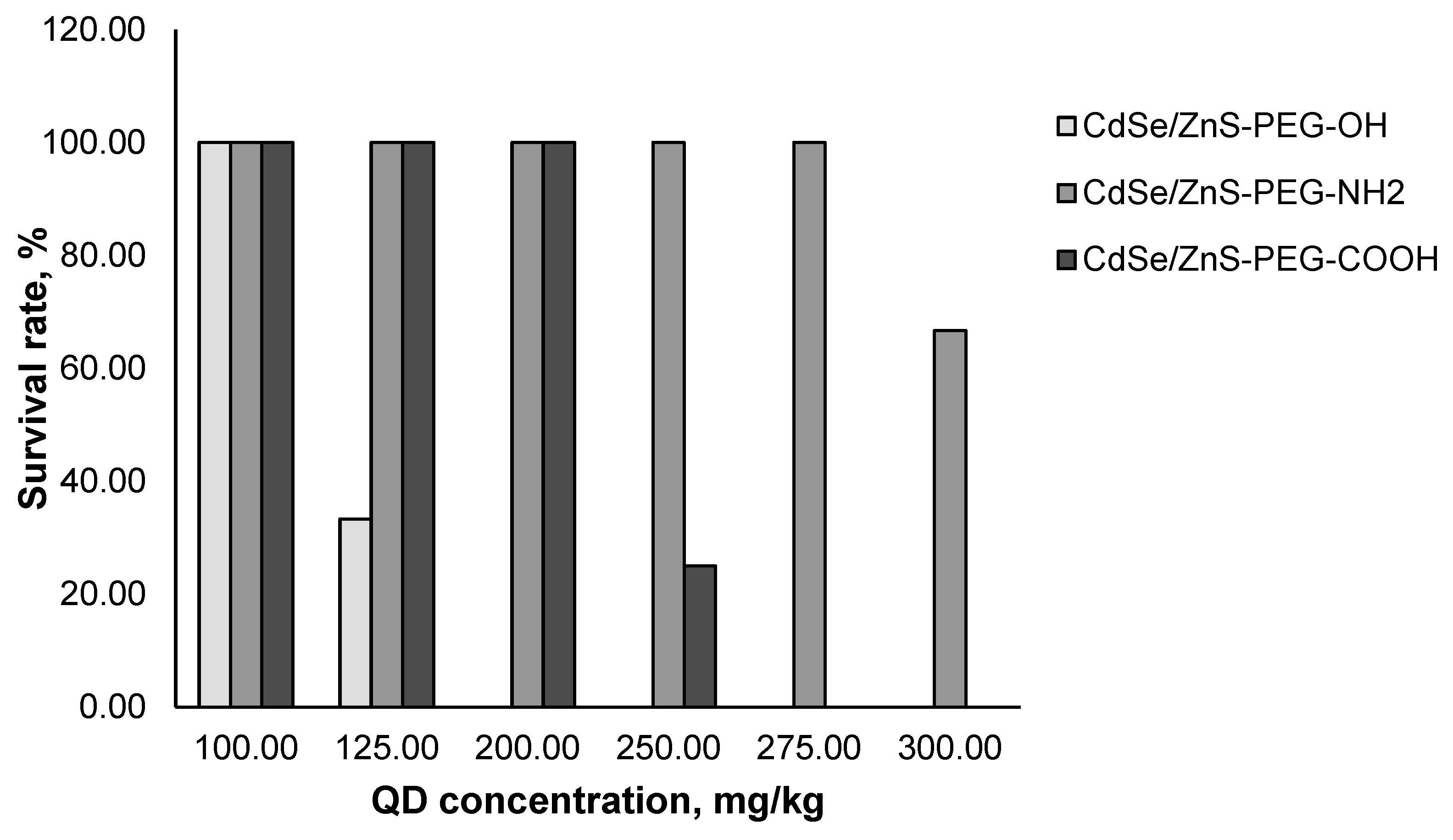
3.2. In vivo acute toxicity of quantum dots
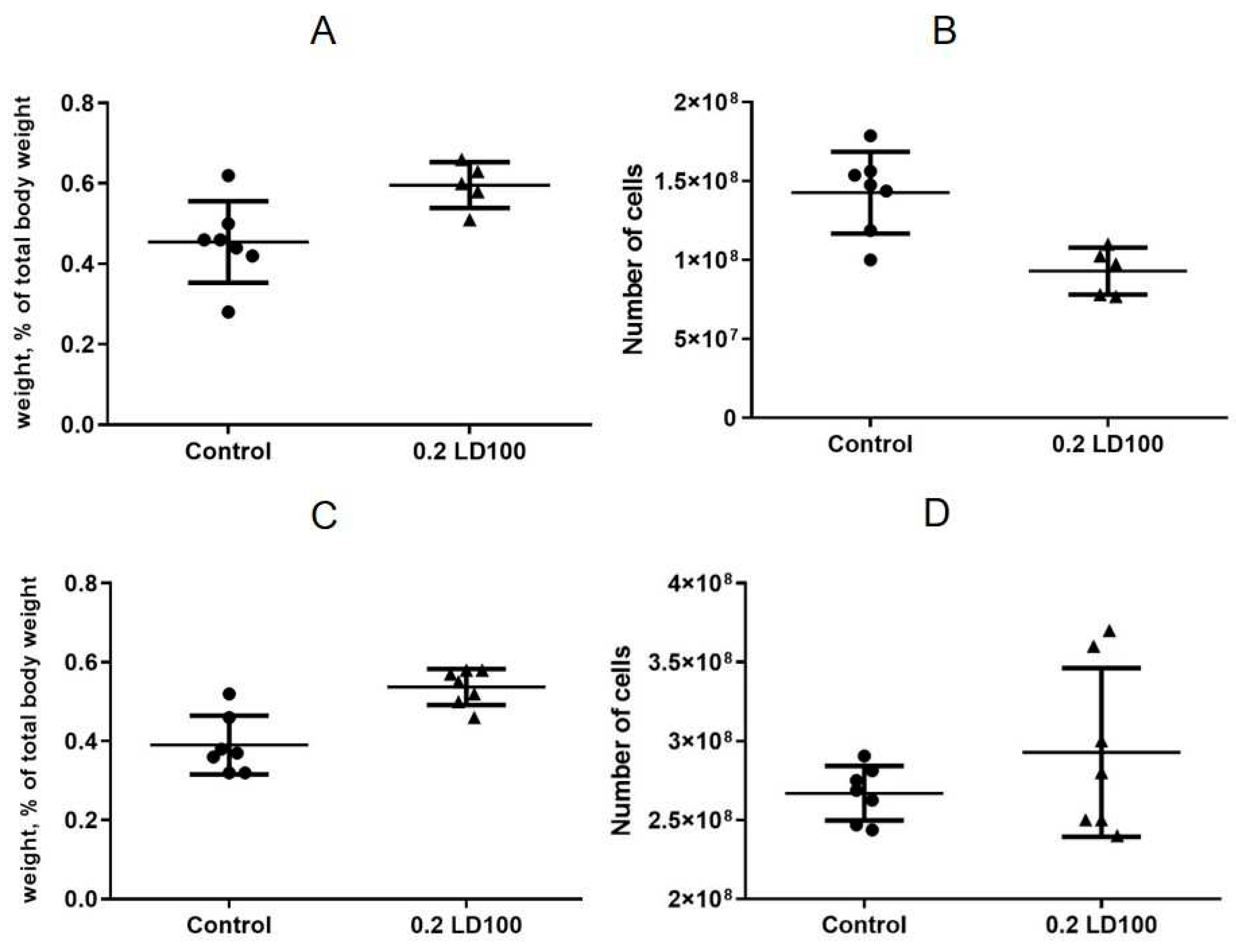
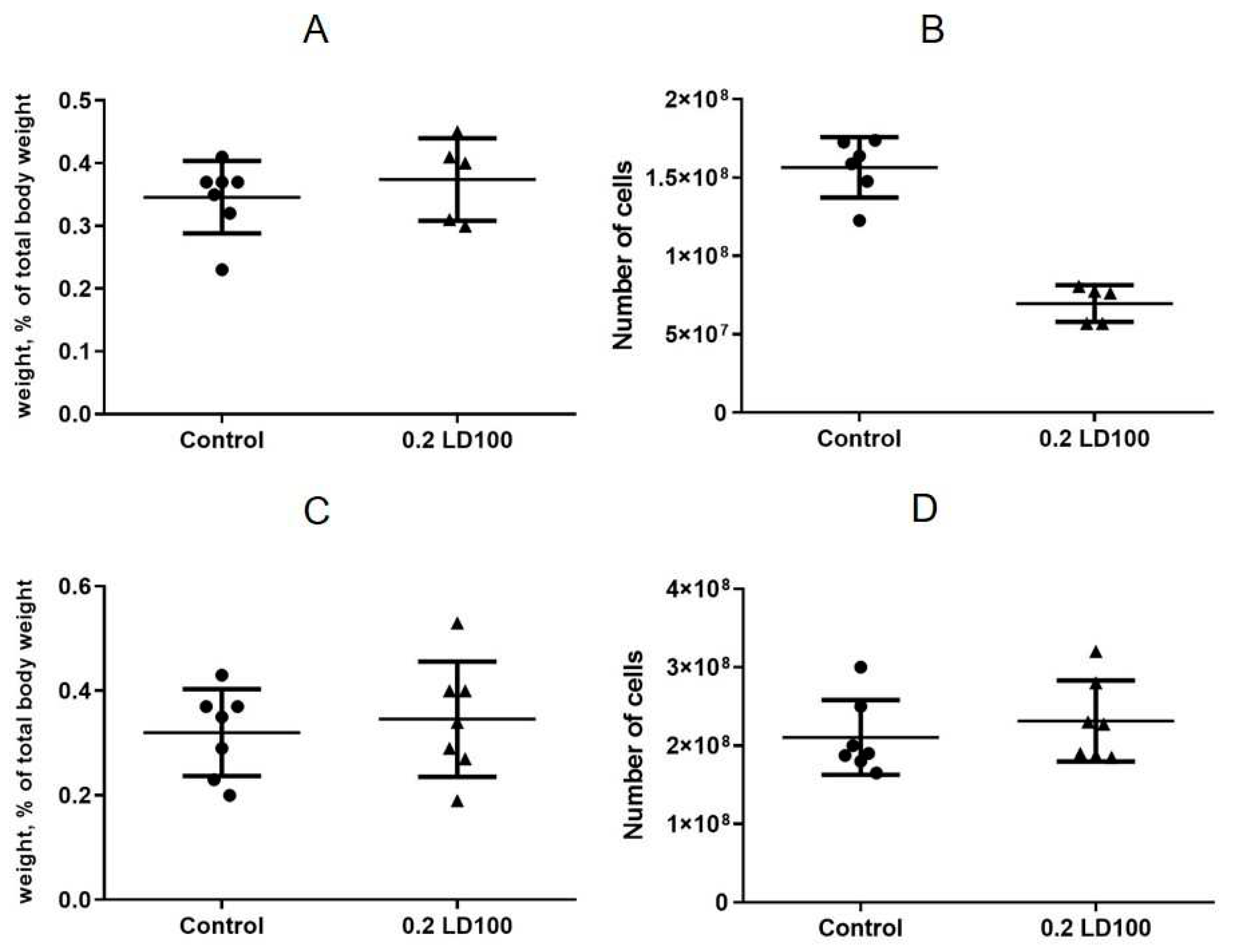
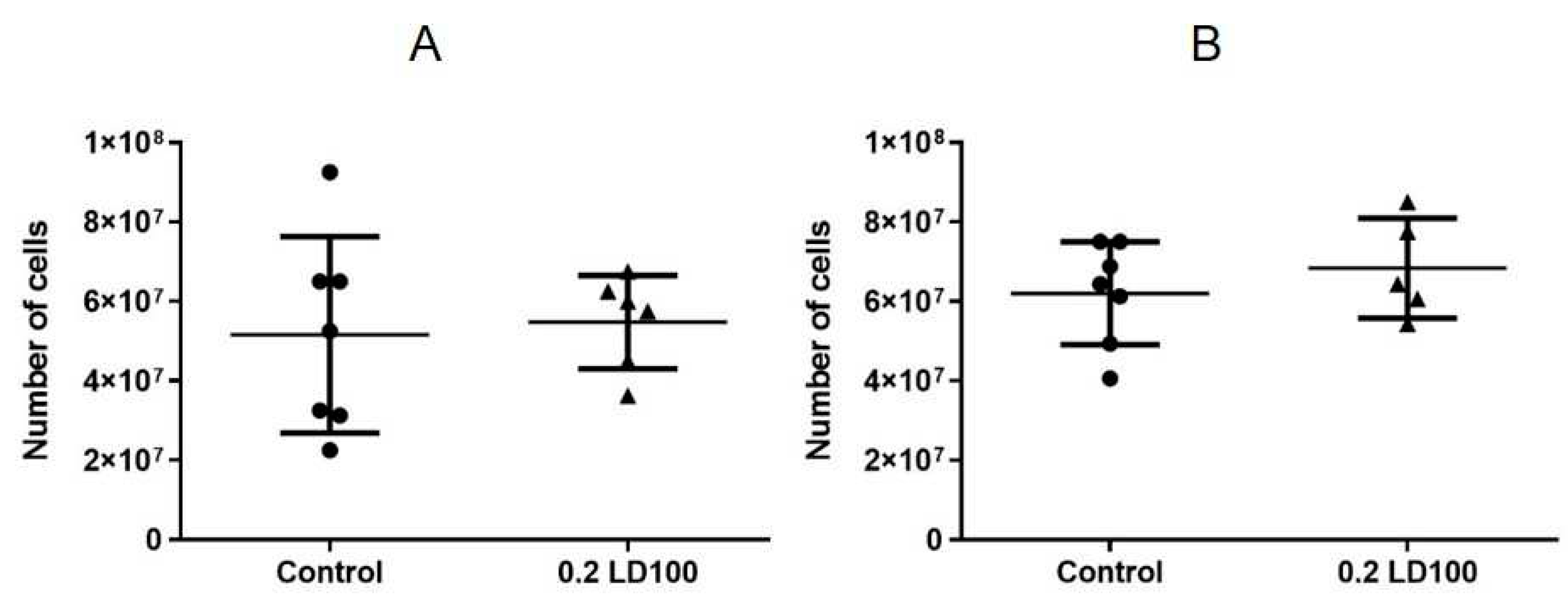
3.3. Effects of quantum dots on the mouse spleen, thymus, and bone marrow
3.4. Effects of quantum dots on the cytokine profile
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Medintz, I.L.; Uyeda, H.T.; Goldman, E.R.; Mattoussi, H. Quantum Dot Bioconjugates for Imaging, Labelling and Sensing. Nat. Mater. 2005, 4, 435–446. [Google Scholar] [CrossRef]
- Matea, C.T.; Mocan, T.; Tabaran, F.; Pop, T.; Mosteanu, O.; Puia, C.; Iancu, C.; Mocan, L. Quantum Dots in Imaging, Drug Delivery and Sensor Applications. Int. J. Nanomedicine 2017, 12, 5421–5431. [Google Scholar] [CrossRef] [PubMed]
- Bilan, R.; Nabiev, I.; Sukhanova, A. Quantum Dot-Based Nanotools for Bioimaging, Diagnostics, and Drug Delivery. ChemBioChem 2016, 17, 2103–2114. [Google Scholar] [CrossRef] [PubMed]
- Brazhnik, K.; Sokolova, Z.; Baryshnikova, M.; Bilan, R.; Efimov, A.; Nabiev, I.; Sukhanova, A. Quantum Dot-Based Lab-on-a-Bead System for Multiplexed Detection of Free and Total Prostate-Specific Antigens in Clinical Human Serum Samples. Nanomedicine Nanotechnology, Biol. Med. 2015, 11, 1065–1075. [Google Scholar] [CrossRef]
- Wagner, A.M.; Knipe, J.M.; Orive, G.; Peppas, N.A. Quantum Dots in Biomedical Applications. Acta Bioomaterialia 2019, 94, 44–63. [Google Scholar] [CrossRef]
- Lu, J.; Tang, M.; Zhang, T. Review of Toxicological Effect of Quantum Dots on the Liver. J. Appl. Toxicol. 2019, 39, 72–86. [Google Scholar] [CrossRef]
- Lu, Y.; Xu, S.; Chen, H.; He, M.; Deng, Y.; Cao, Z.; Pi, H.; Chen, C.; Li, M.; Ma, Q.; et al. CdSe/ZnS Quantum Dots Induce Hepatocyte Pyroptosis and Liver Inflammation via NLRP3 Inflammasome Activation. Biomaterials 2016, 90, 27–39. [Google Scholar] [CrossRef]
- Lin, C.H.; Yang, M.H.; Chang, L.W.; Yang, C.S.; Chang, H.; Chang, W.H.; Tsai, M.H.; Wang, C.J.; Lin, P. Cd/Se/Te-Based Quantum Dot 705 Modulated Redox Homeostasis with Hepatotoxicity in Mice. Nanotoxicology 2011, 5, 650–663. [Google Scholar] [CrossRef]
- Su, Y.; Peng, F.; Jiang, Z.; Zhong, Y.; Lu, Y.; Jiang, X.; Huang, Q.; Fan, C.; Lee, S.T.; He, Y. In Vivo Distribution, Pharmacokinetics, and Toxicity of Aqueous Synthesized Cadmium-Containing Quantum Dots. Biomaterials 2011, 32, 5855–5862. [Google Scholar] [CrossRef]
- Zhao, L.; Zong, W.; Zhang, H.; Liu, R. Kidney Toxicity and Response of Selenium Containing Protein-Glutathione Peroxidase (Gpx3) to CdTe QDs on Different Levels. Toxicol. Sci. 2019, 168, 201–208. [Google Scholar] [CrossRef]
- Liang, Y.; Zhang, T.; Tang, M. Toxicity of Quantum Dots on Target Organs and Immune System. J. Appl. Toxicol. 2021, 1–24. [Google Scholar] [CrossRef]
- Fitzpatrick, J.A.J.; Andreko, S.K.; Ernst, L.A.; Waggoner, A.S.; Ballou, B.; Bruchez, M.P. Long Term Persistence and Spectral Blue Shifting of Quantum Dots in Vivo. Nano Lett. 2009, 9, 2736–2741. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Tian, J.; Yong, K.T.; Zhu, X.; Lin, M.C.M.; Jiang, W.; Li, J.; Huang, Q.; Lin, G. Immunotoxicity Assessment of CdSe/ZnS Quantum Dots in Macrophages, Lymphocytes and BALB/c Mice. J. Nanobiotechnology 2016, 14, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Haque, M.M.; Im, H.Y.; Seo, J.E.; Hasan, M.; Woo, K.; Kwon, O.S. Acute Toxicity and Tissue Distribution of CdSe/CdS-MPA Quantum Dots after Repeated Intraperitoneal Injection to Mice. J. Appl. Toxicol. 2013, 33, 940–950. [Google Scholar] [CrossRef]
- Chen, S.; Chen, Y.; Chen, Y.; Yao, Z. InP/ZnS Quantum Dots Cause Inflammatory Response in Macrophages through Endoplasmic Reticulum Stress and Oxidative Stress. Int. J. Nanomedicine 2019, 14, 9577–9586. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.C.; Zhang, Y.; Todd, J.; Kittle, K.; Patry, D.; Caldwell, D.; Lalande, M.; Smith, S.; Parks, D.; Navarro, M.; et al. Biodistribution and Systemic Effects in Mice Following Intravenous Administration of Cadmium Telluride Quantum Dot Nanoparticles. Chem. Res. Toxicol. 2019, 32, 1491–1503. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Li, L.; Lin, X.; Yang, Z.; Zou, W.; Chen, Y.; Xu, J.; Liu, D.; Wang, X.; Lin, G. In Vitro and in Vivo Immunotoxicity of PEGylated Cd-Free CuInS2/ZnS Quantum Dots. Nanotoxicology 2020, 14, 372–387. [Google Scholar] [CrossRef]
- Dai, T.; Li, N.; Liu, L.; Liu, Q.; Zhang, Y. AMP-Conjugated Quantum Dots: Low Immunotoxicity Both In Vitro and In Vivo. Nanoscale Res. Lett. 2015, 10, 1–9. [Google Scholar] [CrossRef]
- Corsini, E.; House, R. V. Evaluating Cytokines in Immunotoxicity Testing. Methods Mol. Biol. 2018, 1803, 297–314. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in Inflammation, Immunity, and Disease. Cold Spring Harb. Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef] [PubMed]
- Zelová, H.; Hošek, J. TNF-α Signalling and Inflammation: Interactions between Old Acquaintances. Inflamm. Res. 2013, 62, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Vignali, D.A. .; Kuchroo, V.K. IL-12 Family Cytokines: Immunological Playmakers. Nat. Immunol. 2012, 13, 722–728. [Google Scholar] [CrossRef]
- Schroder, K.; Hertzog, P.J.; Ravasi, T.; Hume, D.A. Interferon-γ: An Overview of Signals, Mechanisms and Functions. J. Leukoc. Biol. 2004, 75, 163–189. [Google Scholar] [CrossRef]
- Deshmane, S.L.; Kremlev, S.; Amini, S.; Sawaya, B.E. Monocyte Chemoattractant Protein-1 (MCP-1): An Overview. J. Interf. Cytokine Res. 2009, 29, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Nabiev, I.; Sukhanova, A.; Even-Desrumeaux, K.; Chames, P.; Baty, D.; Artemyev, M.; Oleinikov, V.; Nabiev, I. Engineering of Ultra-Small Diagnostic Nanoprobes through Oriented Conjugation of Single-Domain Antibodies and Quantum Dots. Protoc. Exch. 2012. [Google Scholar] [CrossRef]
- Vokhmintcev, K. V; Linkov, P.A.; Samokhvalov, P.S.; Nabiev, I.R. Two-Stage ZnS Shell Coating on the CuInS2 Quantum Dots for Their Effective Solubilization. KnE Energy 2018, 3, 535. [Google Scholar] [CrossRef]
- Ren, F.; del Rosal, B.; An, S.Y.; Yang, F.; Carrasco, E.; Benayas, A.; Kwon Oh, J.; Jaque, D.; de la Fuente, Á.J.; Vetrone, F.; Ma, D. Development and Investigation of Ultrastable PbS/CdS/ZnS Quantum Dots for Near-Infrared Tumor Imaging. Part. Part. Syst. Charact. 2017, 34, 1600242. [Google Scholar] [CrossRef]
- Lee, H.; Park, K. Acute Toxicity of Benzalkonium Chloride in BALB/c Mice Following Intratracheal Instillation and Oral Administration. Environ. Anal. Health Toxicol. 2019, 34, e2019009. [Google Scholar] [CrossRef]
- Zhao, Q.; Yang, Z.S.; Cao, S.J.; Chang, Y.F.; Cao, Y.Q.; Li, J.B.; Yao, Z.X.; Wen, Y.P.; Huang, X.B.; Wu, R.; Yan, Q.G. Acute Oral Toxicity Test and Assessment of Combined Toxicity of Cadmium and Aflatoxin B1 in Kunming Mice. Food Chem. Toxicol. 2019, 131, 110577. [Google Scholar] [CrossRef] [PubMed]
- Zou, W.; Li, L.; Chen, Y.; Chen, T.; Yang, Z.; Wang, J.; Liu, D.; Lin, G.; Wang, X. In Vivo Toxicity Evaluation of PEGylated CuInS2/ZnS Quantum Dots in BALB/c Mice. Front. Pharmacol. 2019, 10, 437. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Chang, T.; Marcq, G.; Liu, C.; Kiss, B.; Rouse, R.; Mach, K.E.; Cheng, Z.; Liao, J.C. In Vivo Biodistribution and Toxicity of Intravesical Administration of Quantum Dots for Optical Molecular Imaging of Bladder Cancer. Sci. Reps. 2017, 7, 9309. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.; Ouyang, Q.; Hu, R.; Ding, Z.; Tian, J.; Yin, F.; Xu, G.; Chen, Q.; Wang, X.; Yong, K.T. In Vivo Toxicity Assessment of Non-Cadmium Quantum Dots in BALB/c Mice. Nanomedicine: NBM. 2015, 11, 341–350. [Google Scholar] [CrossRef]
- Sukhanova, A.; Bozrova, S.; Sokolov, P.; Berestovoy, M.; Karaulov, A.; Nabiev, I. Dependence of Nanoparticle Toxicity on their Physical and Chemical Properties. Nanoscale Res. Lett. 2018, 13, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Robe, A.; Pic, E.; Lassalle, H.P.; Bezdetnaya, L.; Guillemin, F.; Marchal, F. Quantum Dots in Axillary Lymph Node Mapping: Biodistribution Study in Healthy Mice. BMC Cancer 2008, 8, 1–9. [Google Scholar] [CrossRef]
- Liu, W.; Choi, H.S.; Zimmer, J.P.; Tanaka, E.; Frangioni, J. V; Bawendi, M.G. Compact Cystein-Coated CdSe(ZnCdS) QDs for in Vivo Applications. J. Am. Chem. Soc. 2008, 129, 14530–14531. [Google Scholar] [CrossRef] [PubMed]
- Bozrova, S.; Gerasimovich, E.; Baryshnikova, M.; Sokolova, Z.; Samokhvalov, P.; Guhrenz, C.; Gaponik, N.; Karaulov, A.; Nabiev, I. Dependence of Quantum Dot Toxicity in Vitro on Their Size, Chemical Composition, and Surface Charge. Nanomaterials 2022, 12, 2734. [Google Scholar] [CrossRef]
- Su, Y.; Peng, F.; Jiang, Z.; Zhong, Y.; Lu, Y.; Jiang, X.; Huang, Q.; Fan, C.; Lee, S.T.; He, Y. In Vivo Distribution, Pharmacokinetics, and Toxicity of Aqueous Synthesized Cadmium-Containing Quantum Dots. Biomaterials 2011, 32, 5855–5862. [Google Scholar] [CrossRef]
| QD composition | Size, nm | ζ-potential, mV |
|---|---|---|
| PbS/CdS/ZnS-PEG-OH1 | 32.04±0.87 | –10.60±2.92 |
| CuInS2/ZnS-PEG-OH | 16.08±0.51 | –6.12±1.81 |
| CdSe/ZnS-PEG-OH | 16.74±0.28 | –4.72±0.38 |
| CdSe/ZnS-PEG-COOH2 | 15.37±0.14 | –17.80±3.01 |
| CdSe/ZnS-PEG-NH23 | 22.77±0.36 | 6.43±1.12 |
| QD type | LD50, mg/kg |
|---|---|
| CdSe/ZnS-PEG-OH | 112.5 |
| CuInS2/ZnS-PEG-OH | 200 |
| PbS/CdS/ZnS-PEG-OH | 150 |
| CdSe/ZnS-PEG-COOH | >300 |
| CdSe/ZnS-PEG-NH2 | 240 |
| Cytokine concentration, pg/ml | Time | Control | Sodium phosphate buffer | CdSe/ZnS-PEG-OH QDs |
|---|---|---|---|---|
| IL-12p70 | 0 h | 10.26±1.04 | 10.17±0.93 | 10.31±1.21 |
| 6 h | 10.22±1.62 | 10.36±1.06 | 7.38±0.81 | |
| 24 h | 10.56±0.30 | 10.29±0.74 | 10.27±1.25 | |
| TNF | 0 h | 21.71±0.74 | 22.25±0.91 | 21.09±0.85 |
| 6 h | 21.57±0.68 | 22.39±0.64 | 17.14±1.45 | |
| 24 h | 21.53±0.27 | 22.94±0.35 | 24.87±1.24 | |
| IFN-γ | 0 h | 2.68±0.17 | 2.27±0.21 | 2.42±0.34 |
| 6 h | 2.47±0.25 | 2.76±0.51 | 2.60±0.31 | |
| 24 h | 2.35±0.33 | 2.97±0.37 | 2.84±0.44 | |
| MCP-1 | 0 h | 35.47±3.21 | 35.15±2.79 | 34.97±4.09 |
| 6 h | 35.74±1.76 | 43.34±5.71 | 77.72±5.59 | |
| 24 h | 35.54±0.28 | 31.28±2.25 | 32.49±3.46 | |
| IL-6 | 0 h | 1.54±0.13 | 1,49±0.15 | 1.63±0.23 |
| 6 h | 1.59±0.19 | 3.68±0.74 | 8,56±0.79 | |
| 24 h | 1.51±0.32 | 1.81±0.17 | 2.73±0.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




