Submitted:
27 July 2023
Posted:
28 July 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Aim
2.2. Eligibility criteria
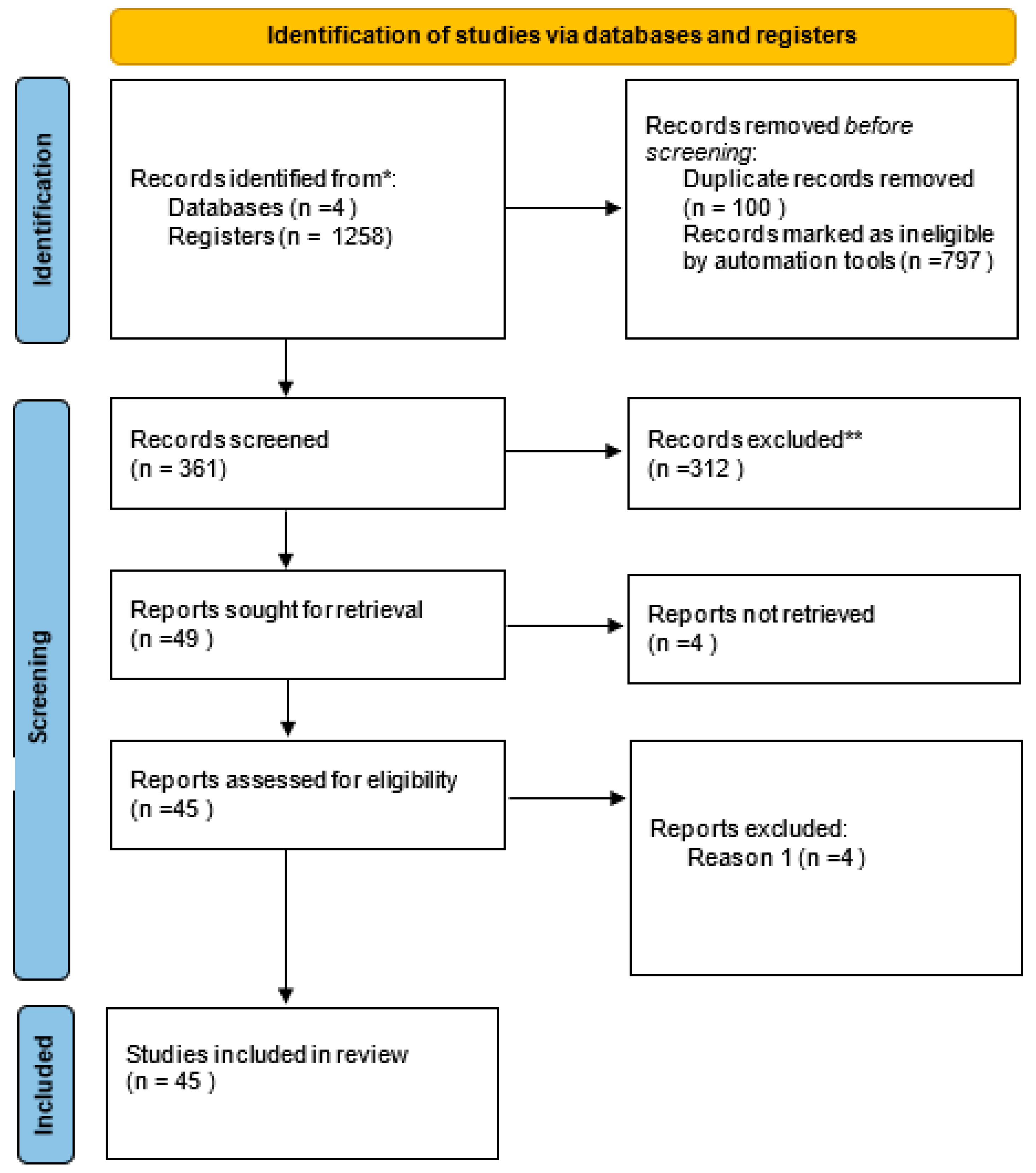
2.3. Search strategy and Data extraction
2.4. Statistical analysis plan
2.5. Role of Funding Source
3. Results
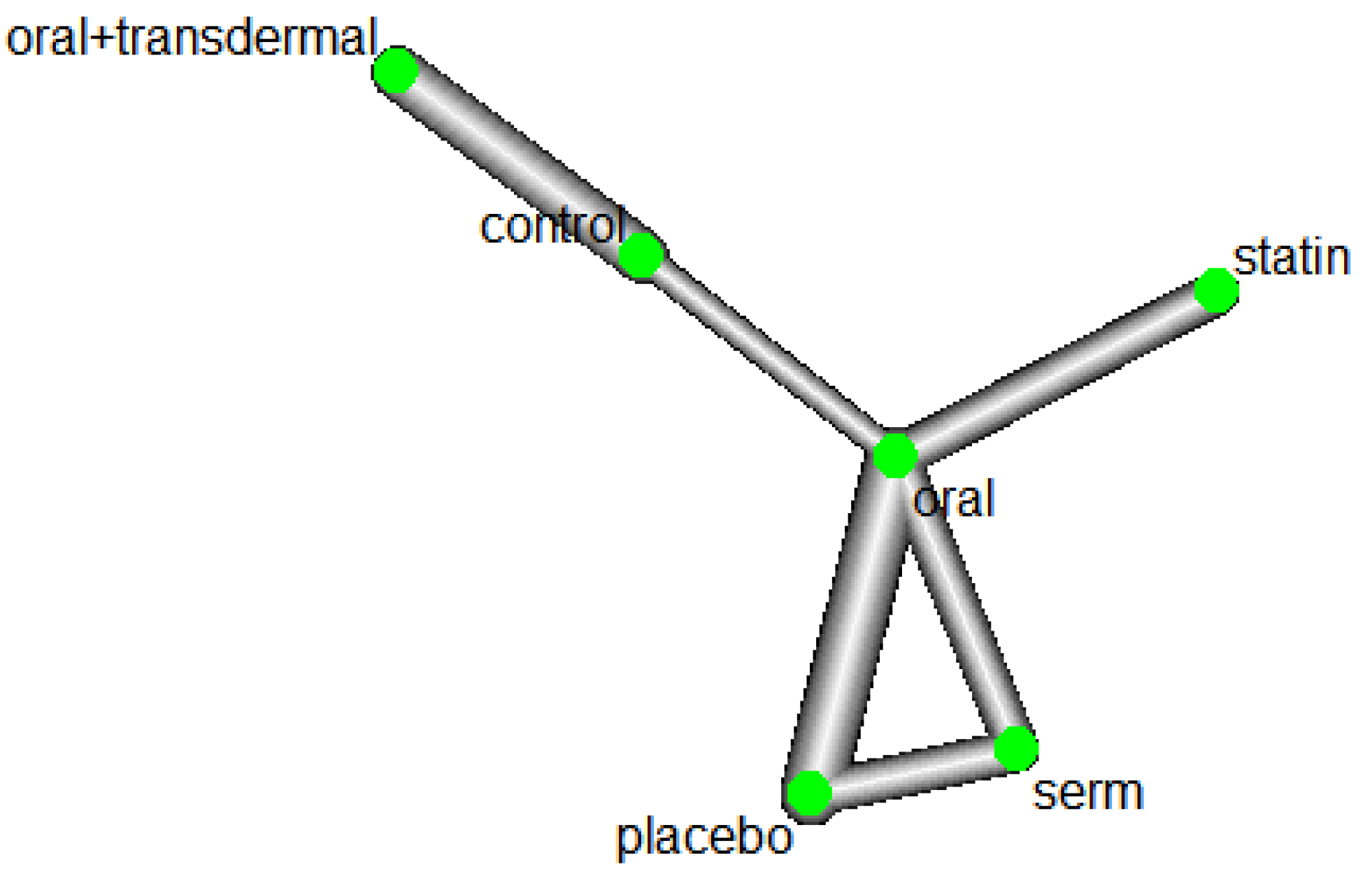
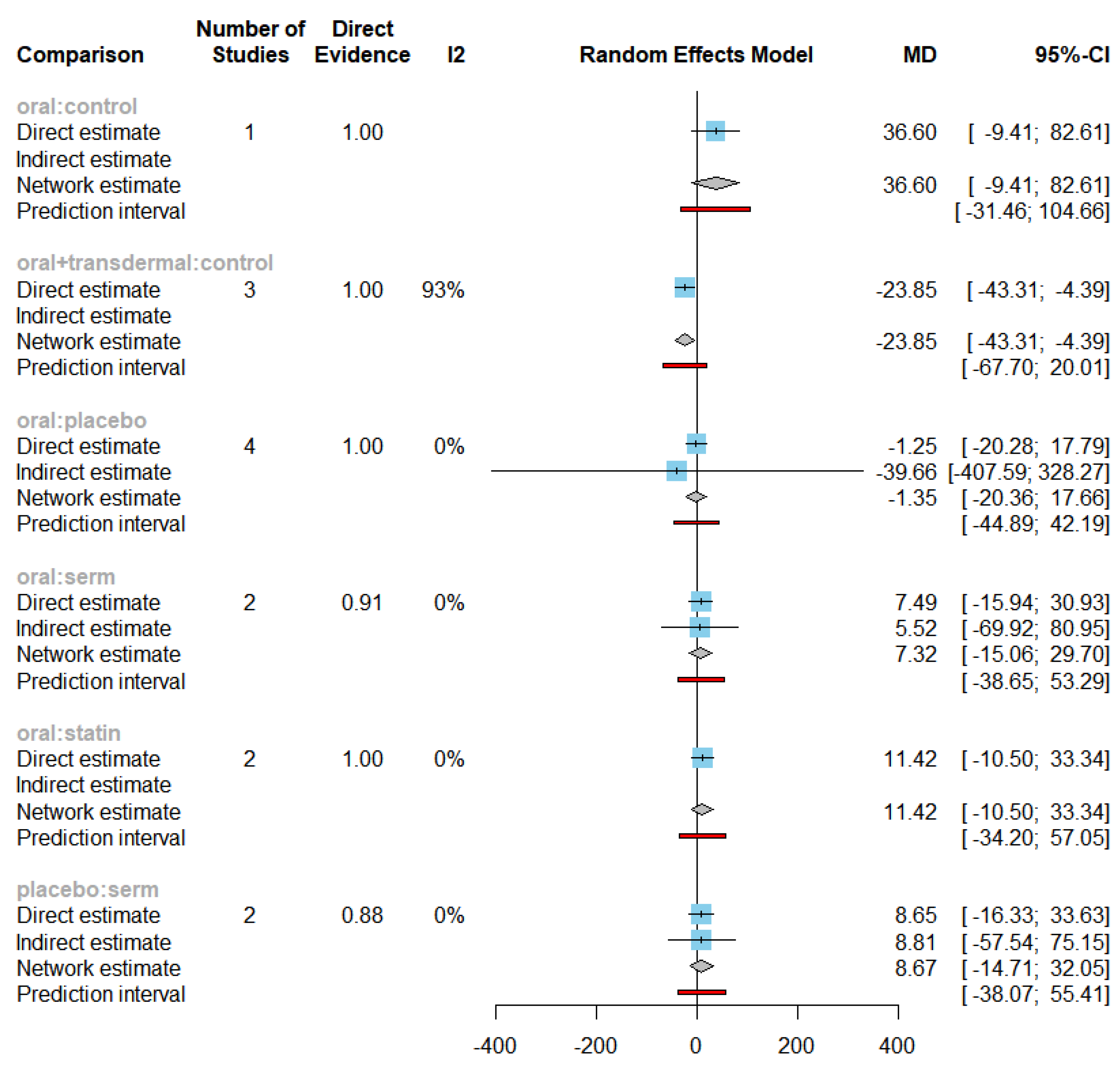
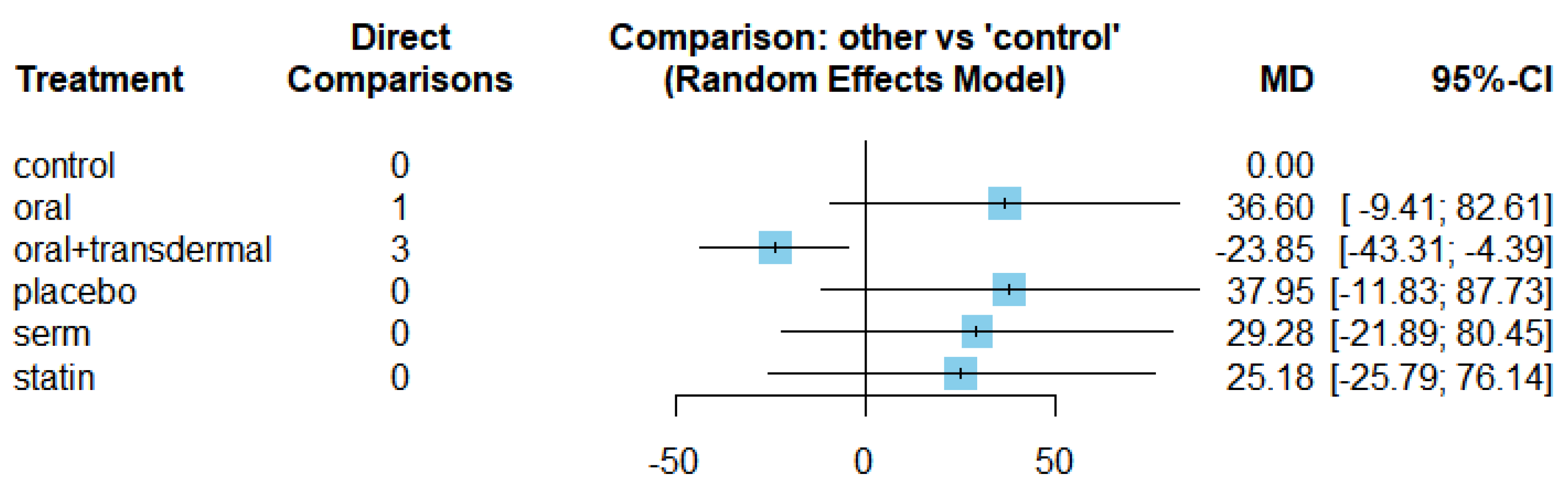
3.1. Role of HRT on Reducing Triglycerides (TG) levels
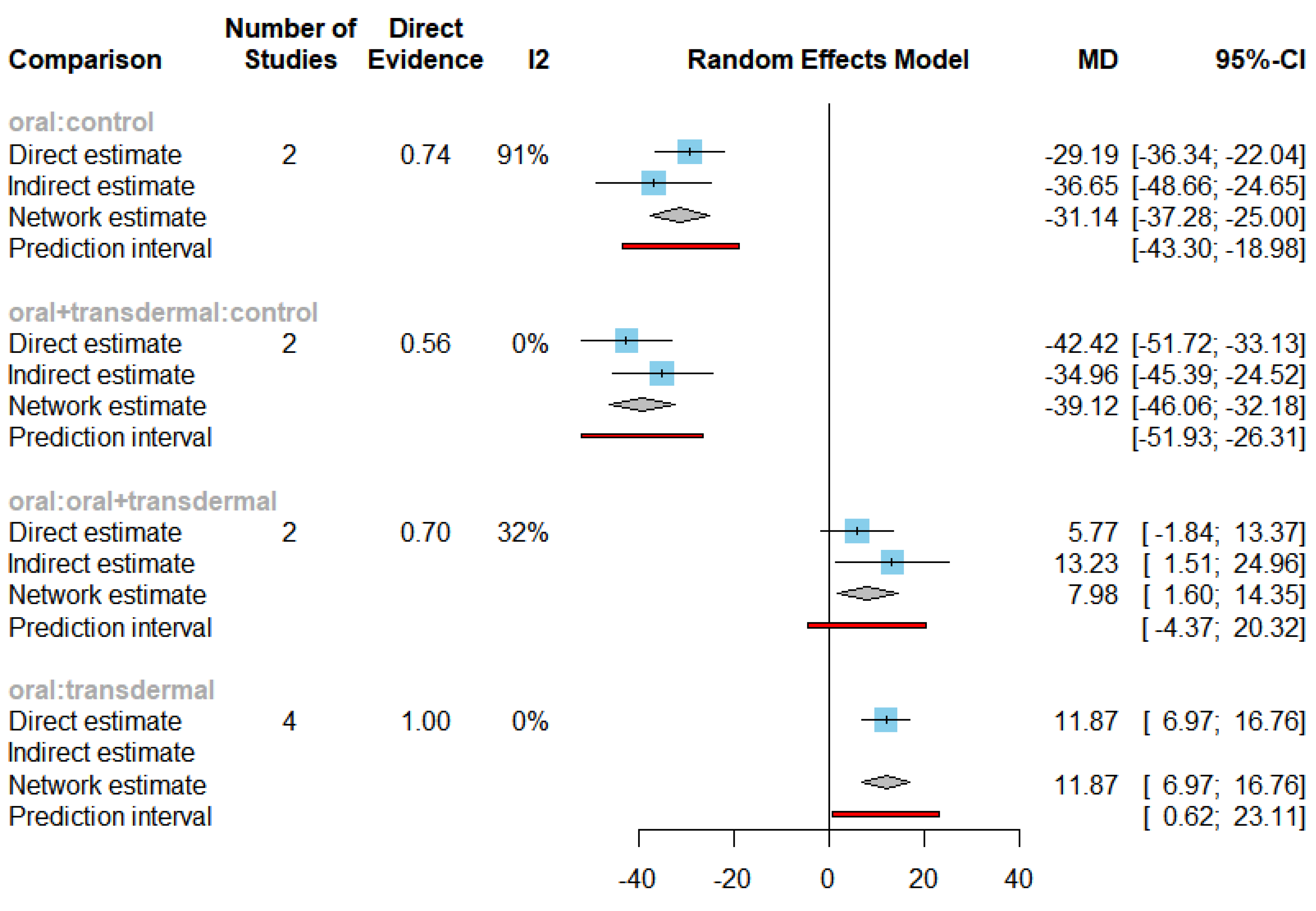
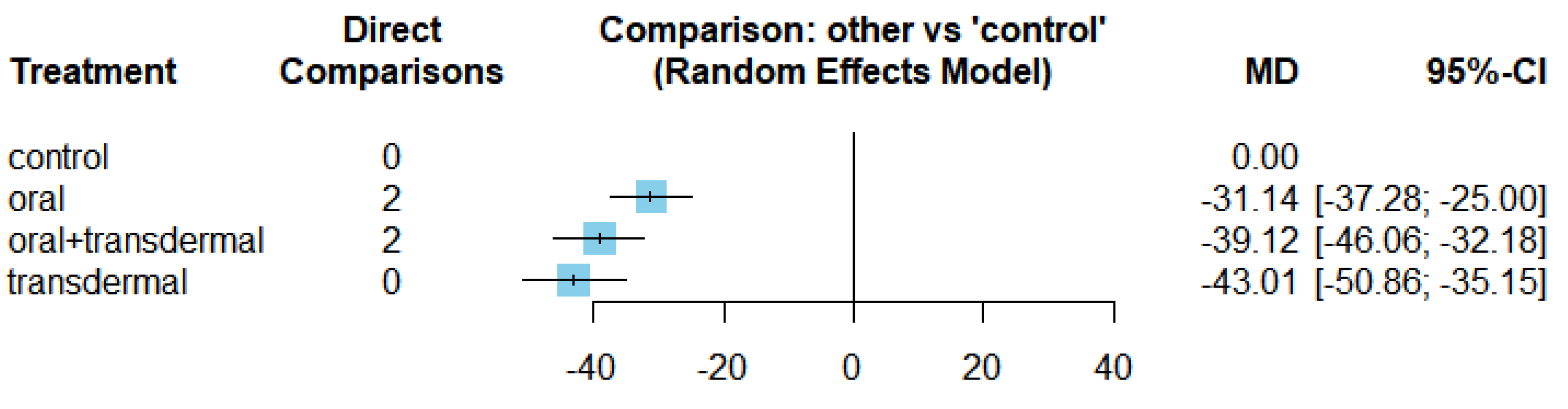
3.2. Efficacy of HRT on Reducing Follicle-Stimulating Hormone (FSH)
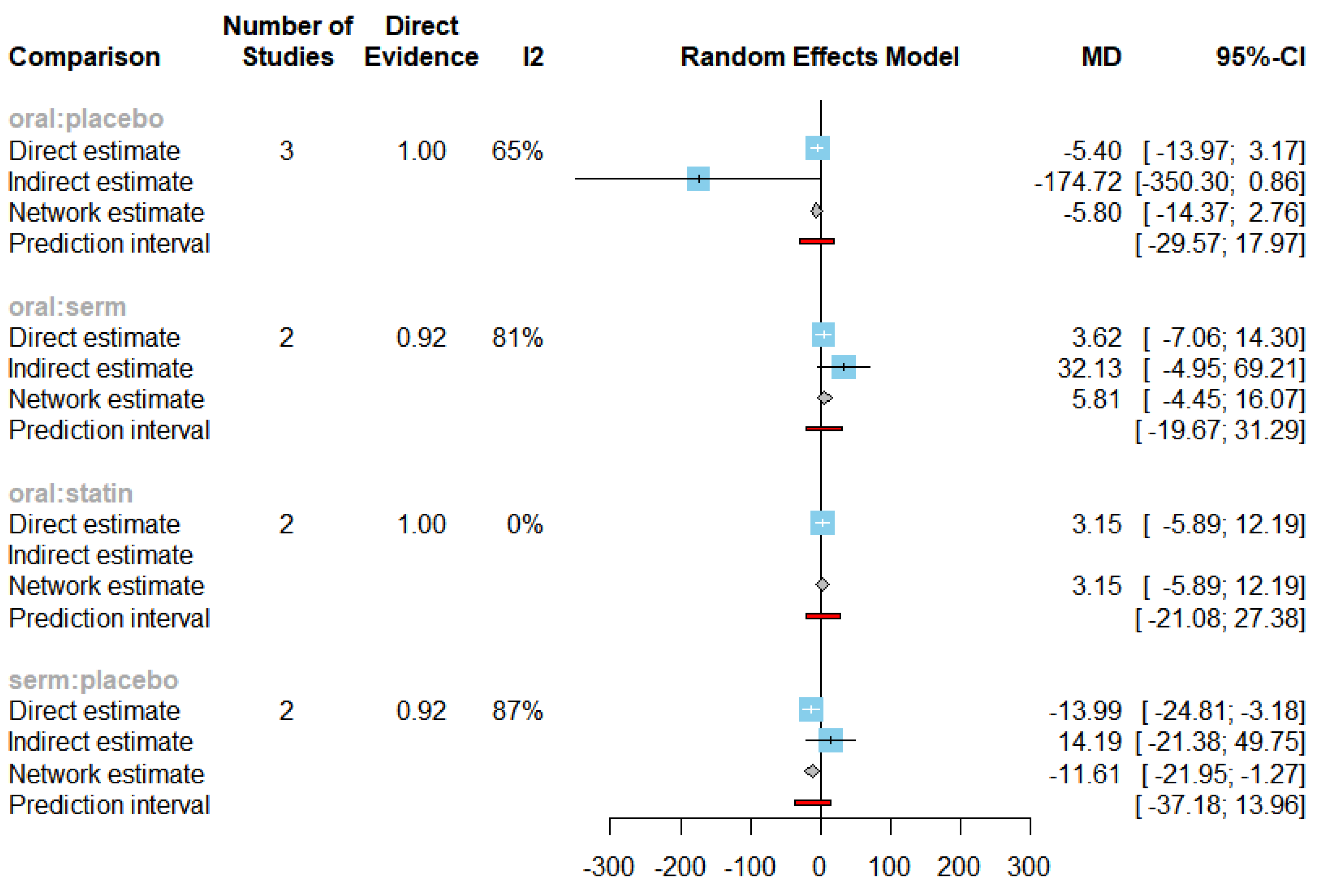
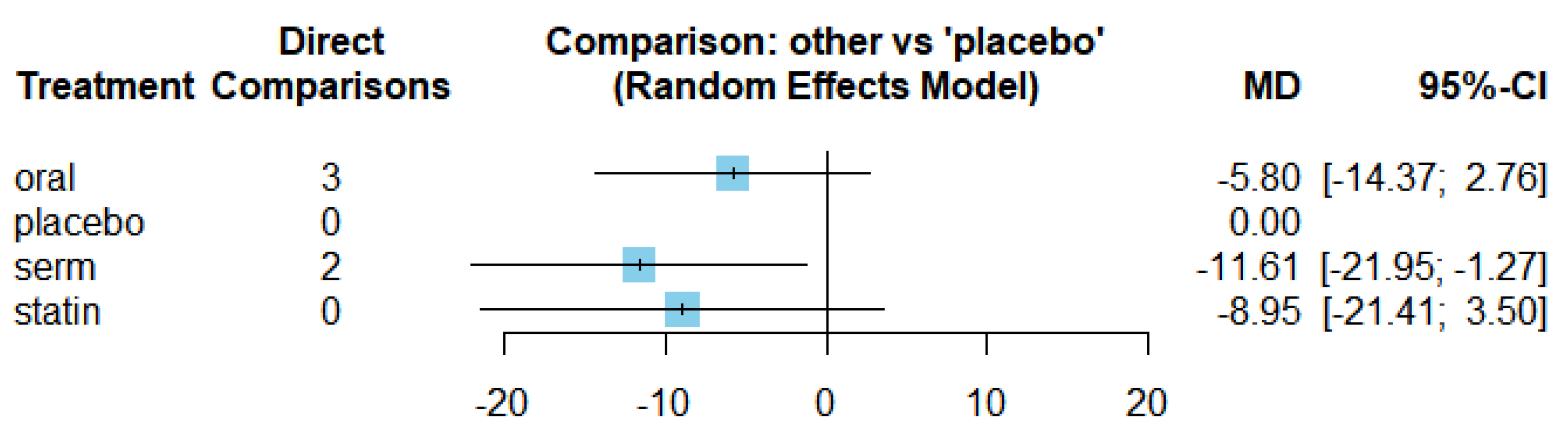
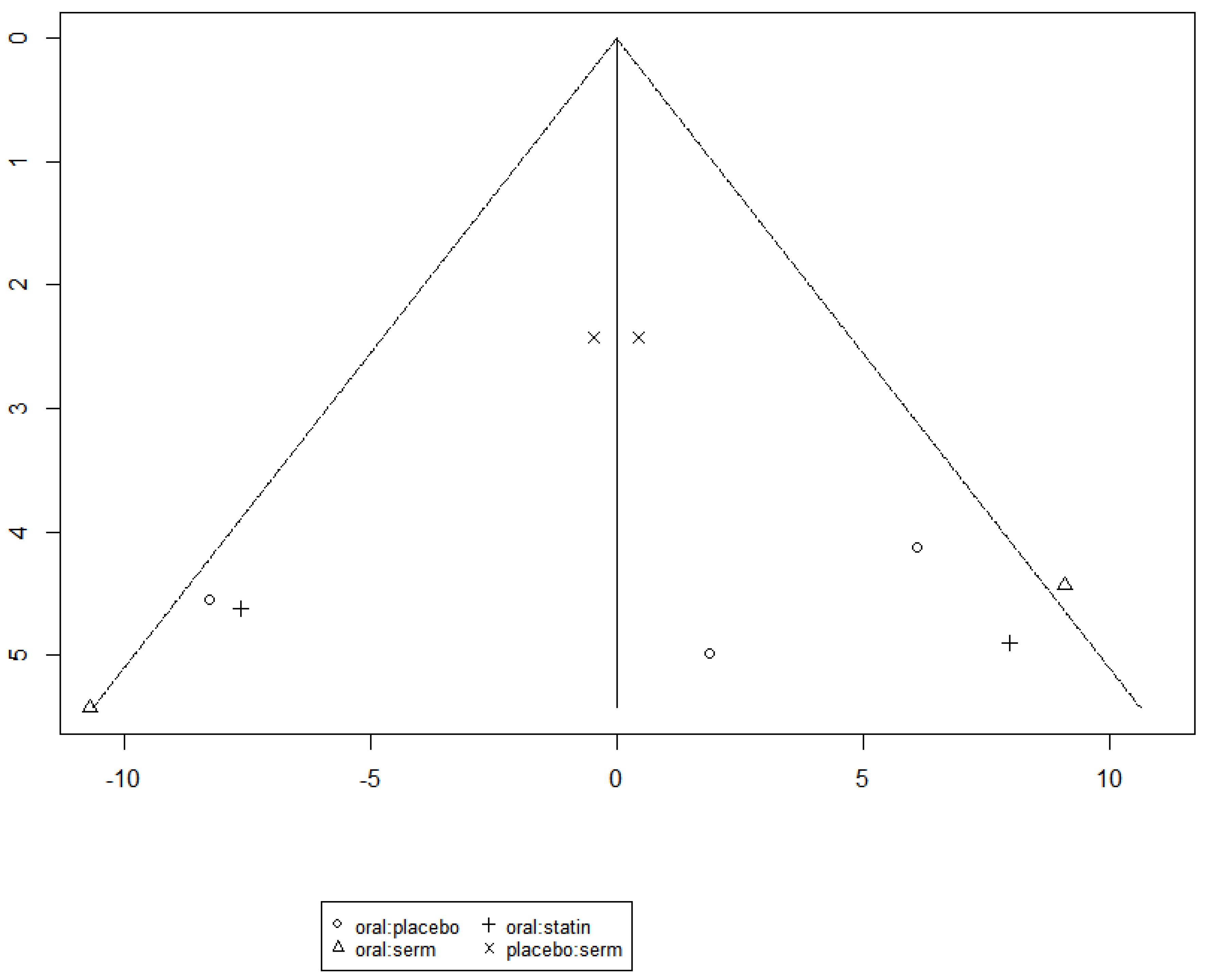
3.3. Efficacy of HRT on Reducing LDL cholesterol (LDL-C)
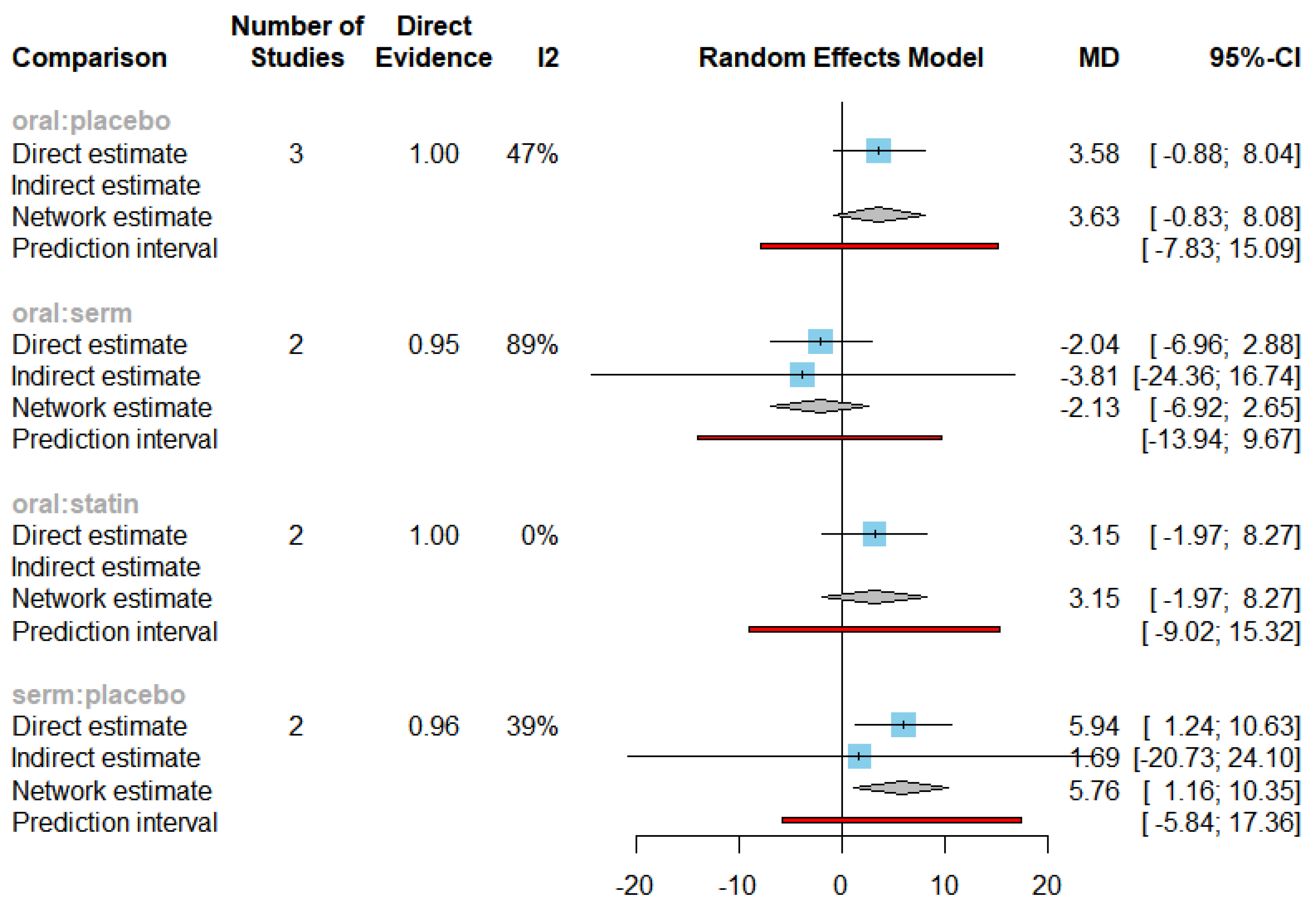
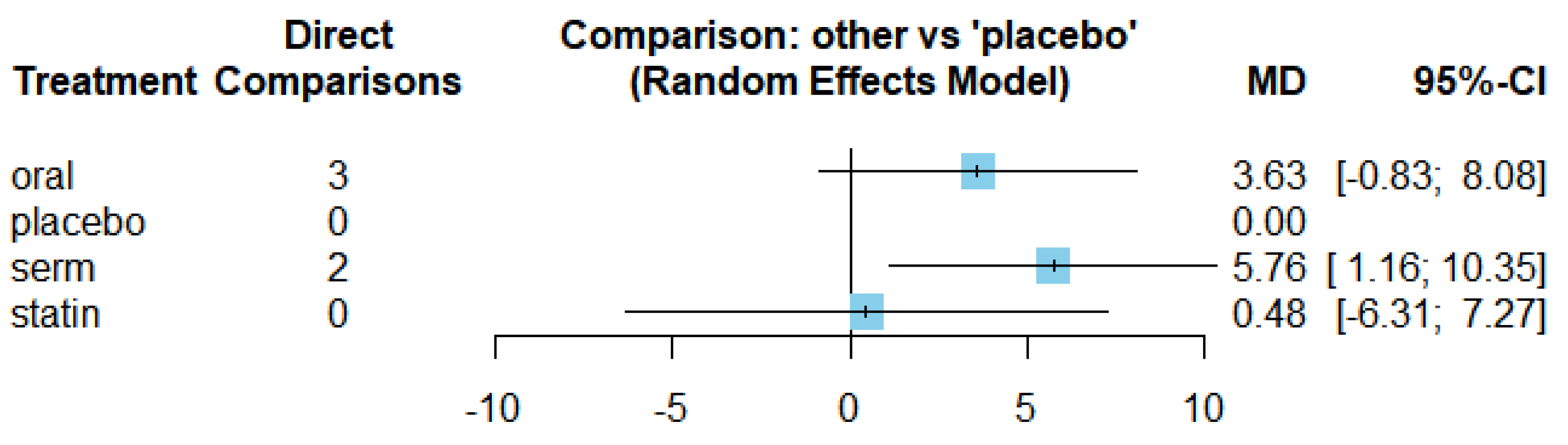
3.4. Efficacy of HRT on Increasing HDL cholesterol (HDL-C)
3.5. Efficacy of HRT on Increasing Estradiol
3.6. Summary
4. Discussion
4.1. Main findings
4.2. Strengths and Limitations
4.3. Implications and Recommendations
5. Conclusion
Funding
Author contributions
Availability of data and material
Code availability
Ethics approval
Consent to participate
Consent for publication
Acknowledgements

Conflicts of interest
References
- Wilson R.A. In: In Feminine Forever. Evans M., editor. Lippincott & Co.; Philadelphia, PA, USA: 1996.
- Ziel H.K., Finkle W.D. Increased risk on endometrial carcinoma among users of conjugated estrogens. N. Engl. J. Med. 1975;293:1167–1170. [CrossRef]
- Woodruff J.D., Pickar J.H. Incidence of endometrial hyperplasia in postmenopausal women taking conjugated estrogens (Premarin) with medroxyprogesterone acetate or conjugated estrogens alone. The Menopause Study Group. Am. J. Obstet. Gynecol. 1994;170:1213–1223. [CrossRef]
- Lobo R.A. Hormone-replacement therapy: Current thinking. Nat. Rev. Endocrinol. 2017;13:220–231. [CrossRef]
- Lobo R.A., Pickar J.H., Stevenson J.C., Mack W.J., Hodis H.N. Back to the future: Hormone replacement therapy as part of a prevention strategy for women at the onset of menopause. Atherosclerosis. 2016;254:296–304.
- Improving women’s health and Gender Justice since the 1995 Beijing Platform for Action [Internet]. World Health Organization; [cited 2023 Jul 7]. Available from: https://www.who.int/initiatives/beijing25.
- Hulley S., Grady D., Bush T., Furberg C., Herrington D., Riggs B., Vittinghoff E. Randomized trial of estrogen plus progestin for secondary prevention of coronary heart disease in postmenopausal women. Heart and Estrogen/progestin Replacement Study (HERS) Research Group. JAMA. 1998;280:605–613.
- Rossouw J.E., Anderson G.L., Prentice R.L., LaCroix A.Z., Kooperberg C., Stefanick M.L., Jackson R.D., Beresford S.A., Howard B.V., Johnson K.C., et al. Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: Principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321–333.
- Hormone-replacement therapy: Updated advice [Internet]. [cited 2023 Jul 7]. Available from: https://www.gov.uk/drug-safety-update/hormone-replacement-therapy-updated-advice.
- Manson J.E., Chlebowski R.T., Stefanick M.L., Aragaki A.K., Rossouw J.E., Prentice R.L., Anderson G., Howard B.V., Thomson C.A., LaCroix A.Z., et al. Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the Women’s Health Initiative randomized trials. JAMA. 2013;310:1353–1368.
- Salpeter S.R., Walsh J.M., Greyber E., Salpeter E.E. Brief report: Coronary heart disease events associated with hormone therapy in younger and older women. A meta-analysis. J. Gen. Intern. Med. 2006;21:363–366.
- Salpeter S.R., Walsh J.M., Greyber E., Ormiston T.M., Salpeter E.E. Mortality associated with hormone replacement therapy in younger and older women: A meta-analysis. J. Gen. Intern. Med. 2004;19:791–804.
- Schierbeck L.L., Rejnmark L., Tofteng C.L., Stilgren L., Eiken P., Mosekilde L., Køber L., Beck Jensen J.E. Effect of hormone replacement therapy on cardiovascular events in recently postmenopausal women: Randomized trial. BMJ. 2012;345:e6409.
- Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539-58.
- Schwarzer, G., Carpenter, J. R., & Rücker, Gerta. (2015). Meta-Analysis with R. Springer International Publishing.
- Egger M, Smith G D, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test BMJ 1997; 315 :629.
- Leal M, Díaz J, Serrano E, Abellán J, Carbonell LF. Hormone replacement therapy for oxidative stress in postmenopausal women with hot flushes. Obstetrics and Gynecology [Internet]. 2000 Jun 1 [cited 2021 Apr 20];95(6 Pt 1):804–9. Available from: https://pubmed.ncbi.nlm.nih.gov/10831971/.
- Nogawa N, Sumino H, Ichikawa S, Kumakura H, Takayama Y, Nakamura T, et al. Effect of long-term hormone replacement therapy on angiotensin-converting enzyme activity and bradykinin in postmenopausal women with essential hypertension and normotensive postmenopausal women. Menopause. 2001 May;8(3):210–5.
- Hall G, Ulla Pripp, Schenck-Gustafsson K, Landgren BM. Longterm effects of hormone replacement therapy on symptoms of angina pectoris, quality of life and compliance in women with coronary artery disease. 1998 Jan 1;28(3):235–42.
- Irahara M, Kuwahara A, Iwasa T, Ishikawa T, Ishihara O, Kugu K, et al. Assisted reproductive technology in Japan: a summary report of 1992-2014 by the Ethics Committee, Japan Society of Obstetrics and Gynecology. Reproductive Medicine and Biology. 2017 Apr;16(2):126–32.
- Carlsten H. A randomised controlled trial evaluating the effects of hormone replacement therapy (HRT) on bone mineral density (BMD) and disease course in postmenopausal women with rheumatoid arthritis (RA). http://isrctn.org/> 2012. [CrossRef]
- Hampton NRE, Rees MCP, Lowe DG, Rauramo I, Barlow D, Guillebaud J. Levonorgestrel intrauterine system (LNG-ius) with conjugated oral equine estrogen: A successful regimen for HRT in perimenopausal women. Human Reproduction. 2005;20(9):2653–60. [CrossRef]
- Harden CL, Herzog AG, Nikolov BG, Koppel BS, Christos PJ, Fowler K, et al. Hormone replacement therapy in women with epilepsy: A randomized, double-blind, placebo controlled study. Epilepsia. 2006;47(9):1447–51. [CrossRef]
- Hart DM, Farish E, Fletcher CD, Barnes JF, Hart H, Nolan D, et al. Long-term effects of continuous combined HRT on bone turnover and lipid metabolism in postmenopausal women. Osteoporosis International. 1998;8(4):326–32. [CrossRef]
- Hartmann BW, Huber JC, Kirchengast S, Söregi, M.D. G, Albrecht AE. Effect of hormone replacement therapy on growth hormone stimulation in women with premature ovarian failure. Fertility and Sterility. 1997;68(1):103–7. [CrossRef]
- Harvey J, Scheurer C, Kawakami F, Quebe-Fehling E, de Palacios PI, Ragavan V. Hormone replacement therapy and breast density changes. Climacteric. 2005;8(2):185–92. [CrossRef]
- Ki EY, Hur SY, Park JS, Do Han K, Park YG. Differences in the lipid profile and hormone replacement therapy use in Korean postmenopausal women: the Korea National Health and Nutrition Examination Survey (KNHANES) 2010–2012. Archives of Gynecology and Obstetrics. 2015 Dec 19;294(1):165–73.
- Kannel WB. Menopause and Risk of Cardiovascular Disease. Annals of Internal Medicine. 1976 Oct 1;85(4):447.
- Nie G, Yang X, Wang Y, Liang W, Li X, Luo Q, et al. The effects of menopause hormone therapy on lipid profile in Postmenopausal women: A systematic review and meta-analysis. Frontiers in Pharmacology. 2022;13. [CrossRef]
- Yang F, Li N, Gaman M-A, Wang N. Raloxifene has favorable effects on the lipid profile in women explaining its beneficial effect on cardiovascular risk: A meta-analysis of randomized controlled trials. Pharmacological Research. 2021;166:105512. [CrossRef]
- Alomar SA, Găman MA, Prabahar K, Arafah OA, Almarshood F, Baradwan S, et al. The effect of tamoxifen on the lipid profile in women: A systematic review and meta-analysis of randomized controlled trials. Experimental Gerontology. 2022 Mar;159:111680.
- Santen RJ, Allred DC, Ardoin SP, Archer DF, Boyd N, Braunstein GD, et al. Postmenopausal Hormone Therapy: An Endocrine Society Scientific Statement. The Journal of Clinical Endocrinology & Metabolism. 2010 Jul;95(7_supplement_1):s1–66.
- Stevenson JC, Arkadi Chines, Pan K, Ryan KA, Mirkin S. A Pooled Analysis of the Effects of Conjugated Estrogens/Bazedoxifene on Lipid Parameters in Postmenopausal Women From the Selective Estrogens, Menopause, and Response to Therapy (SMART) Trials. 2015 Jun 1;100(6):2329–38.
- Kilim SR. A Comparative Study of Lipid Profile and Oestradiol in Pre- and Post-Menopausal Women. Journal Of Clinical And Diagnostic Research. 2013.
- Mendelsohn ME. Protective effects of estrogen on the cardiovascular system. The American Journal of Cardiology. 2002 Jun;89(12):12–7.
- Naftolin F, Tan O. Faculty opinions recommendation of Postmenopausal hormone therapy: An endocrine society scientific statement. Faculty Opinions – Post-Publication Peer Review of the Biomedical Literature. 2011. [CrossRef]
- Oliver-Williams C, Glisic M, Shahzad S, Brown E, Pellegrino Baena C, Chadni M, et al. The route of administration, timing, duration and dose of postmenopausal hormone therapy and cardiovascular outcomes IN WOMEN: A systematic review. Human Reproduction Update. 2018;25(2):257–71. [CrossRef]
- Shufelt CL, Manson JE. Menopausal Hormone Therapy and Cardiovascular Disease: The Role of Formulation, Dose, and Route of Delivery. The Journal of Clinical Endocrinology and Metabolism [Internet]. 2021 Apr 23 [cited 2022 Jan 31];106(5):1245–54. Available from: https://www.uptodate.com/contents/menopausal-hormone-therapy-and-cardiovascular-risk/abstract-text/33506261/pubmed.
- Lin S, Sun L, Lin J, Yang XQ, Zhang L, Qiao JW, et al. Estradiol 1 mg and drospirenone 2 mg as hormone replacement therapy in postmenopausal Chinese women. 2011 Apr 6;14(4):472–81.
- Xu X, Jones M, Mishra GD. Age at natural menopause and development of chronic conditions and multimorbidity: results from an Australian prospective cohort. Human Reproduction. 2020 Jan 1;35(1):203–11.
- Assaf A, Bushmakin A, Joyce N, Louie M, Flores M, Moffatt M. The Relative Burden of Menopausal and Postmenopausal Symptoms versus Other Major Conditions: A Retrospective Analysis of the Medical Expenditure Panel Survey Data. 2017;10(6). Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5620512/pdf/ahdb-10-311.pdf.
- Unger CA. Hormone therapy for transgender patients. Transl Androl Urol. 2016 Dec;5(6):877-884. [CrossRef]
- Jaff N. Does one size fit all? the usefulness of menopause education across low and middle income countries. Maturitas. 2023;173:90. [CrossRef]
- Jalbuena JR. A32 menopausal medicine under difficult conditions: The Philippine experience. Maturitas. 1996;27:9–10. [CrossRef]
- Krishna S. Attitudes towards menopause and hormone replacement therapy in different cultures. International Congress Series. 2002;1229:207–14. [CrossRef]
- Depypere H, Pintiaux A, Desreux J, Hendrickx M, Neven P, Marchowicz E, et al. Coping with menopausal symptoms: An internet survey of Belgian postmenopausal women. Maturitas [Internet]. 2016 Aug 1 [cited 2020 Sep 2];90:24–30. Available from: https://pubmed.ncbi.nlm.nih.gov/27282790/.
- New Scheme for Cheaper Hormone Replacement Therapy Launches [Internet]. GOV.UK. 2023. Available from: https://www.gov.uk/government/news/new-scheme-for-cheaper-hormone-replacement-therapy-launches.
- Pharmaceutical Benefits Scheme - Services Australia [Internet]. www.servicesaustralia.gov.au.
- Available from: https://www.servicesaustralia.gov.au/pharmaceutical-benefits-scheme.
| Study ID | Authors | Year | Study Type | Sample Size | Standard deviation; present (P)/absent (A) | Country | Meta-analysis inclusion Y/N |
|---|---|---|---|---|---|---|---|
| 1 | Hall et al. | 1998 | RCT | 60 | P | Sweden | Y |
| 2 | Hall et al. | 1994 | RCT | 200 | P | UK | N |
| 3 | Hampton et al. | 2005 | Prospective | 82 | A | UK | N |
| 4 | Harden et al. | 2006 | Double blind RCT | 21 | A | USA& Israel | N |
| 5 | Hart et al. | 1998 | Single-centre trial | 52 | A | UK | N |
| 6 | Hartmann et al. | 1997 | Controlled clinical study | 48 | A | Austria | N |
| 7 | Harvey et al. | 2005 | RCT | 202 | A | N | |
| 8 | Hayashi et al. | 2011 | prospective | 32 | P | Japan | Y |
| 9 | Haywar et al. | 2001 | randomized cross-over study | 12 | P | Australia | N |
| 10 | Heald et al. | 2005 | Randomized triple crossover | 35 | A | UK | N |
| 11 | Heikkinen et al. | 2006 | RCT | 419 | P | Finland | N |
| 12 | Heikkinen et al. | 2000 | Double blind RCT | 419 | P | Finland | N |
| 13 | Hempling et al. | 1997 | RCT | 1232 | P | USA | N |
| 14 | Hill et al. | 1999 | RCT | 204 | A | USA | N |
| 15 | Hillard et al. | 1992 | Prospective | 79 | P | UK | N |
| 16 | Hillard et al. | 1994 | RCT | 96 | P | UK | N |
| 17 | Hirvonen et al. | 1997 | RCT | 173 | P | Finland | Y |
| 18 | Ho et al. | 2006 | RCT | 66 | P | China | Y |
| 19 | Hunt et al. | 1990 | Longitudinal cohort | 4544 | A | England & Wales | N |
| 20 | Ichikawa et al. | 2008 | RCT | 22 | P | Japan | Y |
| 21 | Johannisson et al. | 1997 | RCT | 774 | P | Sweden | N |
| 22 | Johnson et al. | 2002 | Double blind RCT | 438 | P | USA | N |
| 23 | Ke et al. | 2003 | Prospective | 27 | P | USA | N |
| 24 | Kernohan et al. | 2007 | Double-blind RCT | 30 | P | UK | N |
| 25 | Khastgir et al. | 2000 | questionnaire survey | 200 | A | UK | N |
| 26 | Kohrt et al. | 1995 | Non-random | 24 | P | USA | N |
| 27 | Komulainen et al. | 1999 | RCT | 458 | A | Finland | N |
| 28 | Kornhauser et al. | 1997 | Double blind RCT | 55 | P | Mexico | Y |
| 29 | Kristensen et al. | 1999 | Double blind RCT | 267 | P | Denmark | N |
| 30 | Bech et al. | 1998 | Double blind RCT | 105 | P | Denmark | N |
| 31 | Demetrio et al. | 2011 | Double blind RCT | 76 | A | Brazil | N |
| 32 | Kurabayashi et al. | 1997 | Non-random | 70 | P | Japan | N |
| 33 | Laivuori et al. | 2001 | RCT | 38 | A | Finland | N |
| 34 | Lambrinoudaki et al. | 2003 | Prospective | 84 | P | Greece | N |
| 35 | Lamon-Fava et al. | 2005 | Double blind RCT | 8 | P | USA | Y |
| 36 | Lazar et al. | 2003 | RCT | 76 | P | Brazil | N |
| 37 | Leal et al. | 2000 | Prospective | 49 | P | Spain | Y |
| 38 | Lemay et al. | 2001 | RCT | 30 | P | Canada | Y |
| 39 | Leonetti et al. | 1999 | RCT | 90 | P | USA | N |
| 40 | Lin et al. | 2011 | RCT | 244 | P | China | N |
| 41 | Liu et al. | 2004 | RCT | 123 | P | China | N |
| 42 | Nogawa et al. | 2001 | RCT | 35 | P | Japan | Y |
| 43 | Odabaşi et al. | 2007 | Prospective | 61 | P | Turkey | N |
| 44 | Odmark et al. | 1999 | RCT | 249 | A | Sweden | N |
| 45 | Onalan et al. | 2005 | Prospective | 286 | P | Turkey | N |
| Measurement | Comparison | Effect | 95% CI |
|---|---|---|---|
| TG | Oral + transdermal vs Control | -23.85 | [-43.31; -4.39] |
| FSH | Oral vs Control | -31.14 | [-37.28;-25] |
| Oral + transdermal vs Control | -39.12 | [-46.06; -32.18] | |
| Transdermal vs Control | -43.01 | [-50.86; -35.15] | |
| LDL-C | SERM vs Placebo | -11.61 | [-21.95; -1.27] |
| HDL-C | SERM vs Placebo | 5.76 | [1.16, 10.35] |
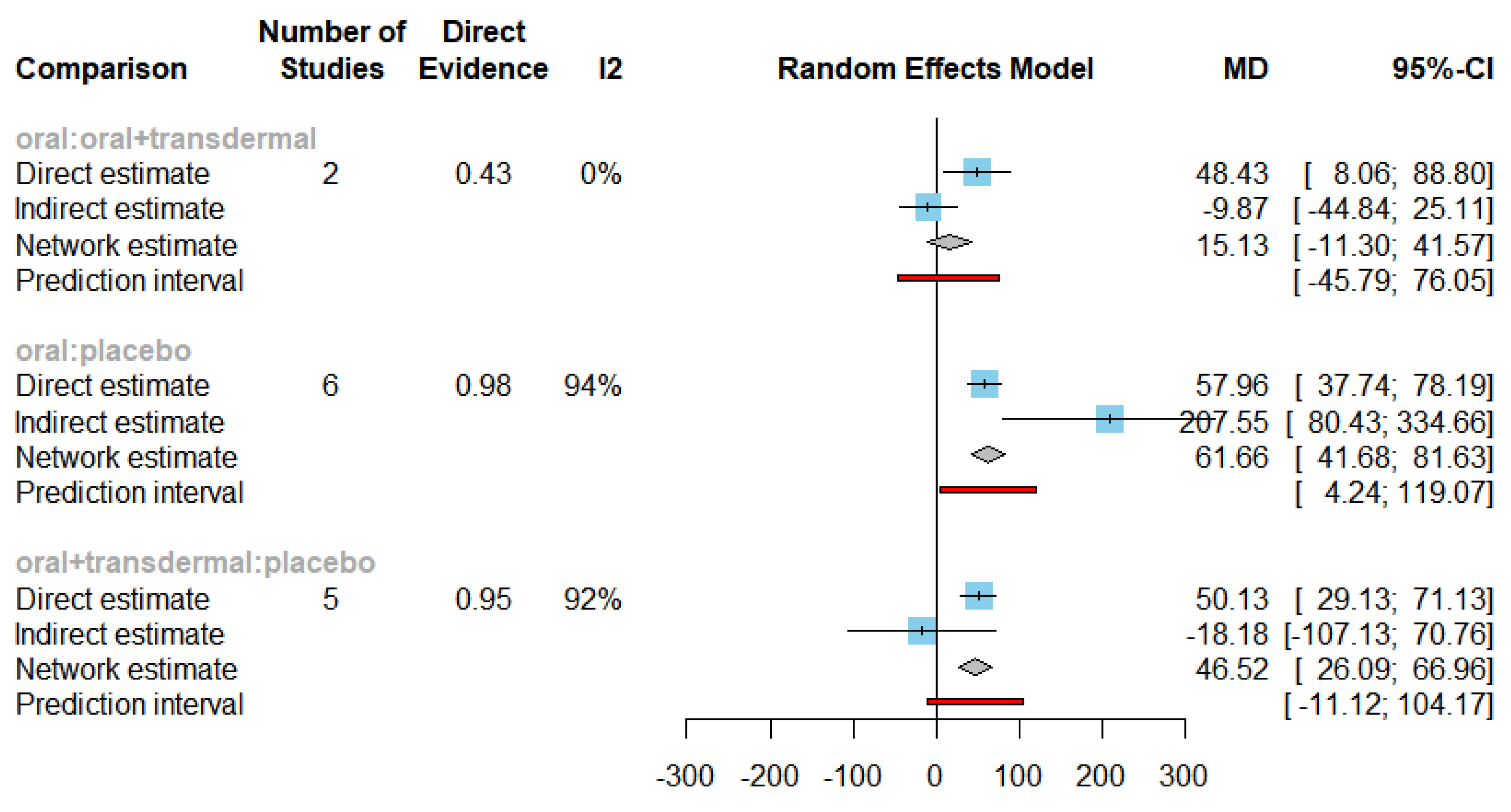
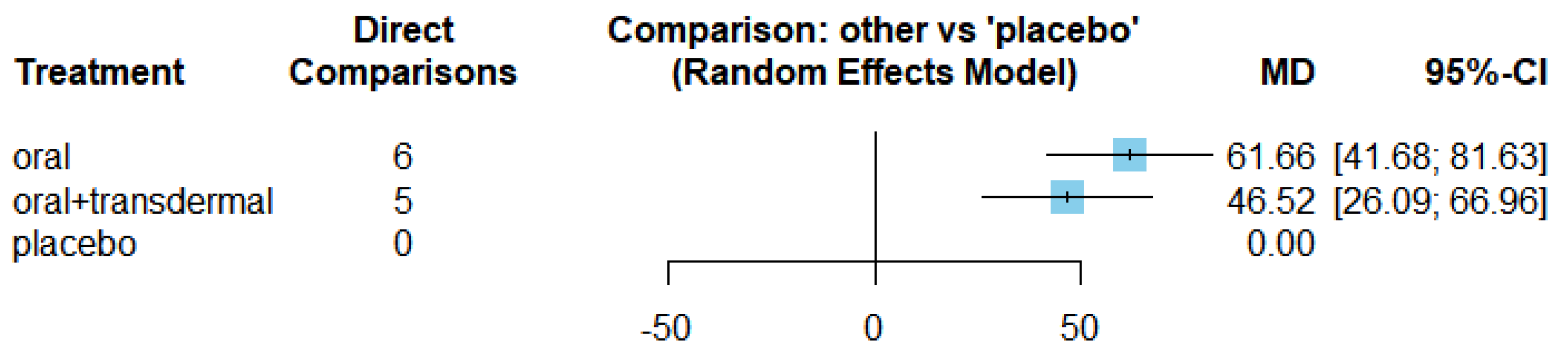
| Estradiol | Oral vs Placebo | 61.66 | [41.68, 81.63] |
| Oral + transdermal vs Placebo | 46.52 | [26.09, 66.96] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




