Submitted:
18 July 2023
Posted:
19 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2.1. Materials
2.1.1. Chemicals
2.1.2. Enzyme preparations
2.1.3. Plant materials
2.2. Enzyme-assisted extraction of rose petal by-product
2.3. Methods
2.3.1. Ultrafiltration of rose petal extract
2.3.1.1. Membranes
2.3.1.2. Experimental system
2.3.2. Phytochemical analyses
2.3.2.1. Polyphenolic content
2.3.2.2. Total antioxidant capacity
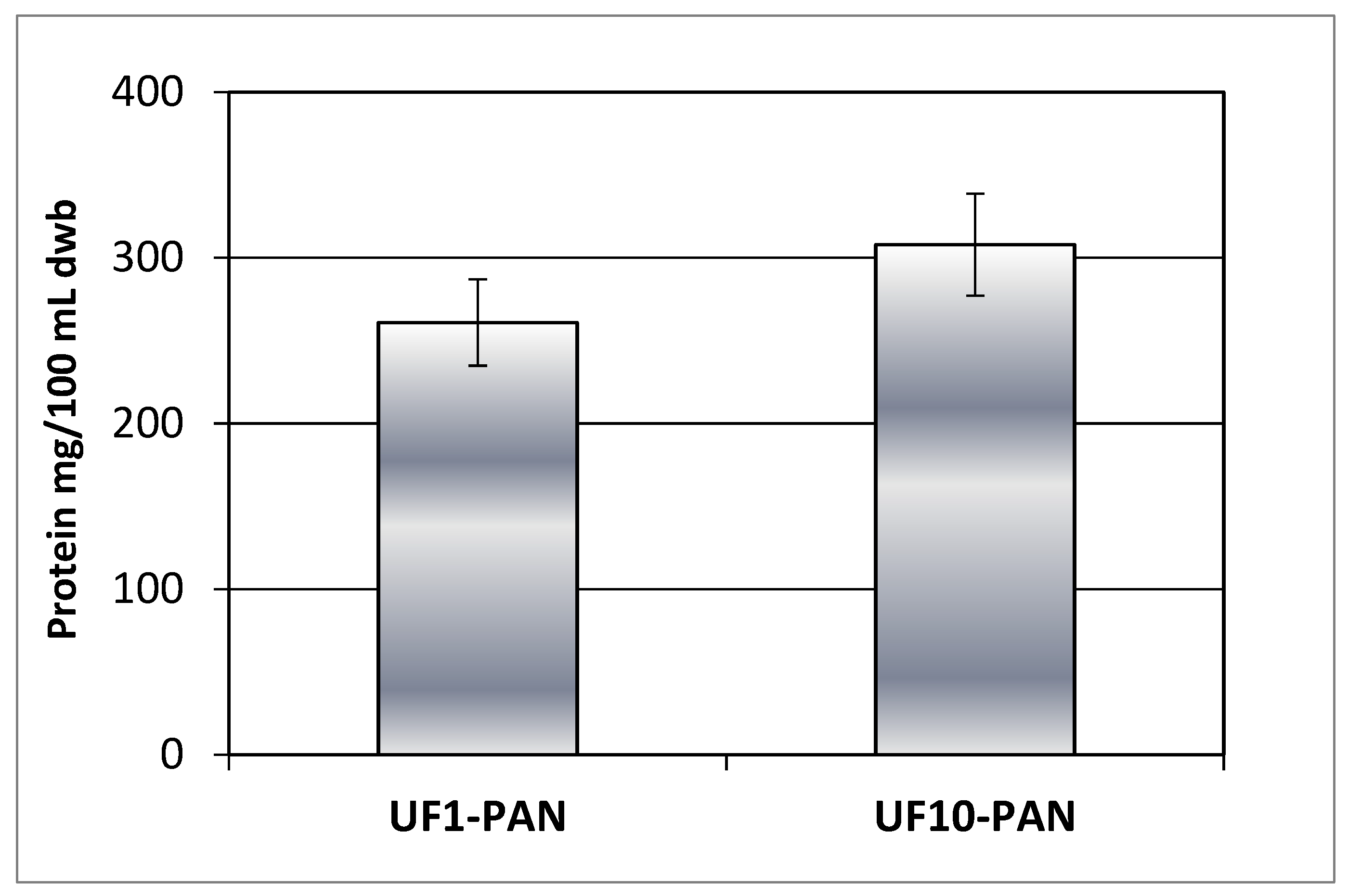
2.3.2.3. Protein content
2.4. Determination of dry matter content
2.5. Statistical analysis
3. Results and Discussion
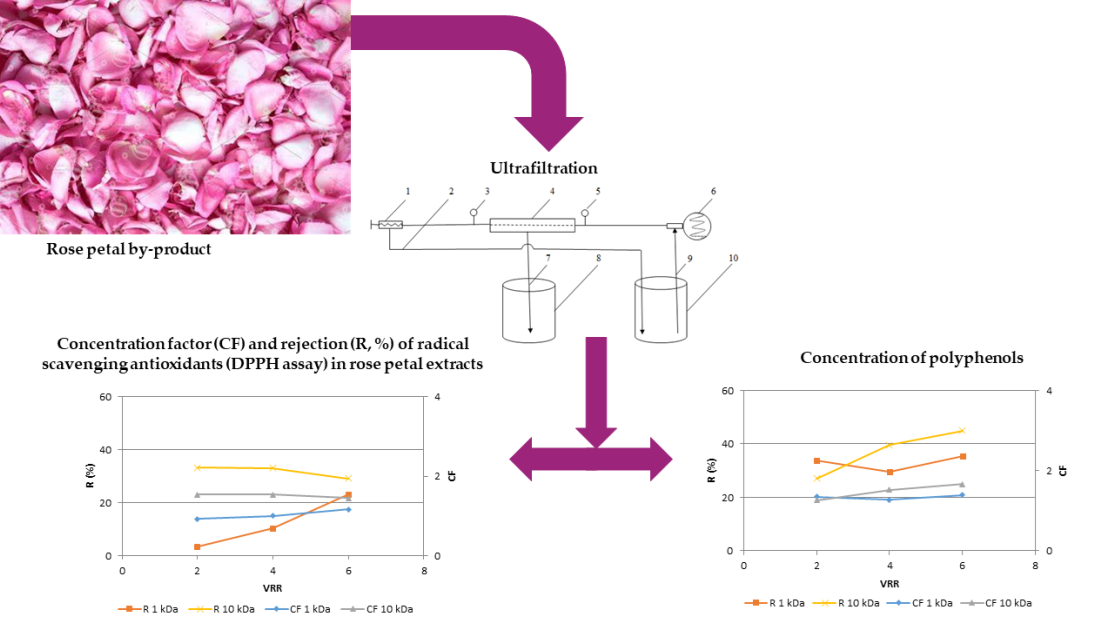
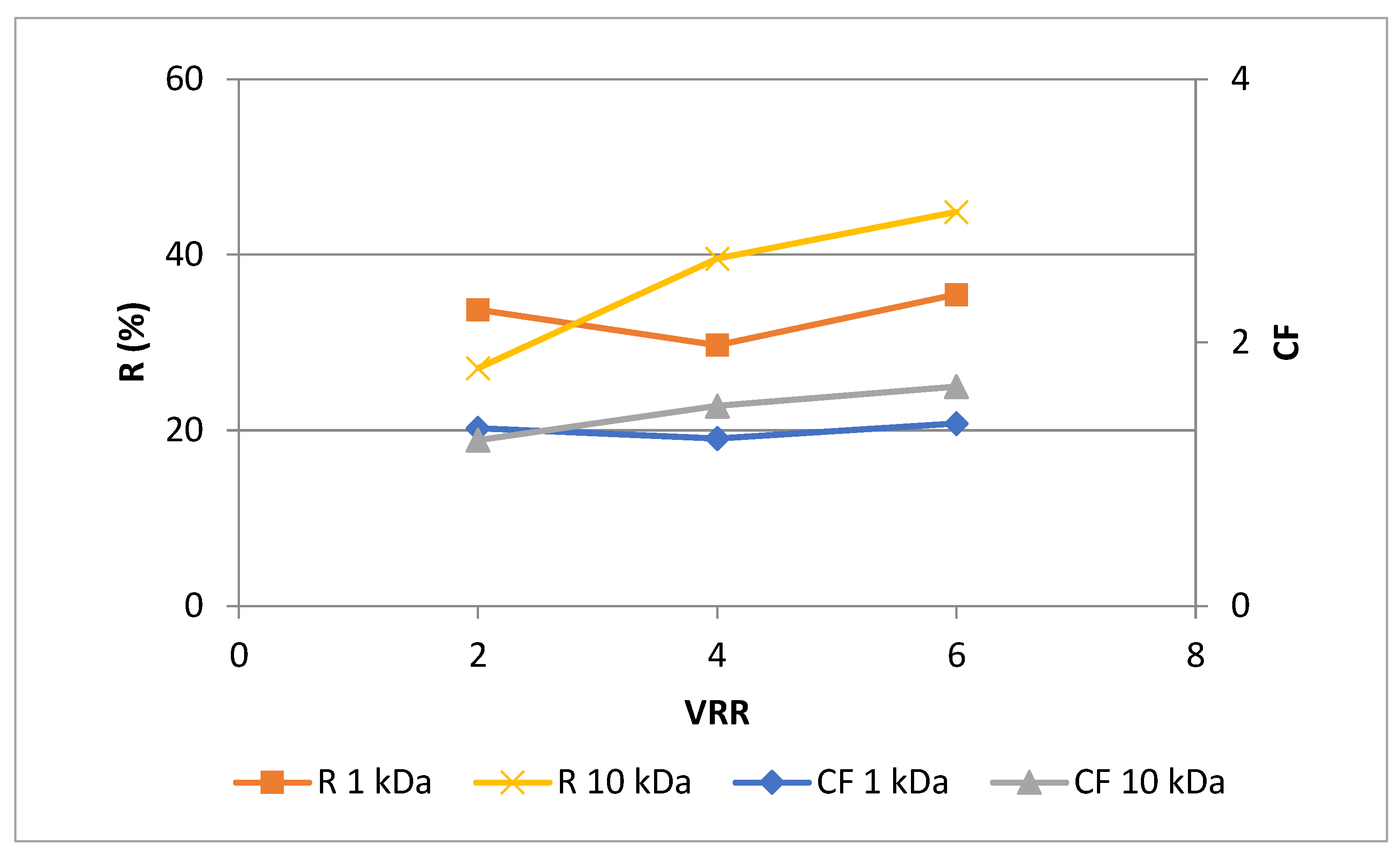
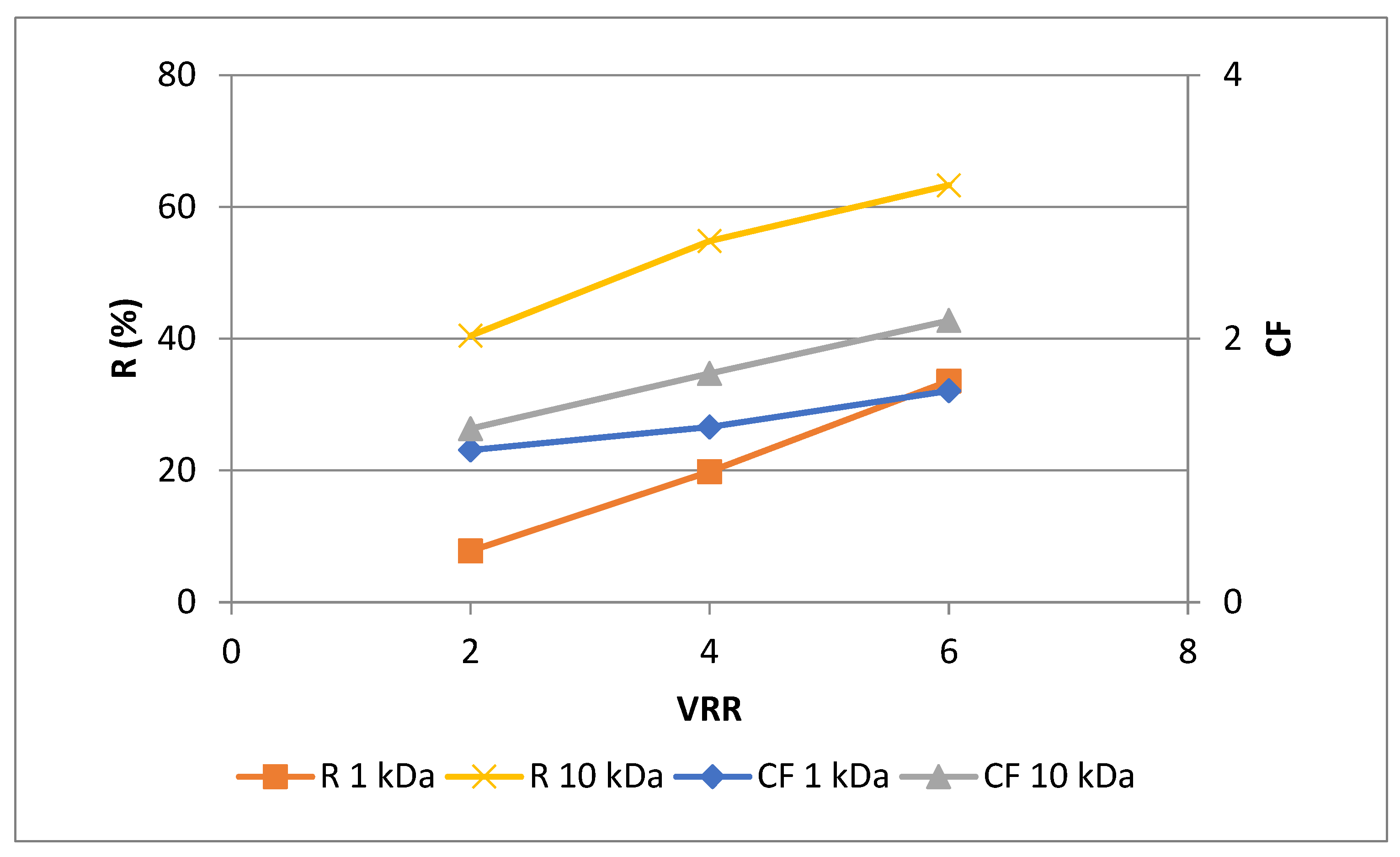
3.1. Effects of membrane’s molecular weight cut-ff and process parameters on the permeate flux during ultrafiltration for concentration of polyphenols in rose petal extract
3.2. Effects of membrane’s molecular weight cut-off and process parameters on the phytochemical characteristics of rose petal extract during ultrafiltration for concentration of polyphenols
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kroon, P. A.; Faulds, C. B.; Ryden, P.; Robertson, J. A.; Williamson, G. Release of covalently bound ferulic acid from fiber in the human colon. J. Agric. Food Chem. 1997, 45, 661–667. [Google Scholar] [CrossRef]
- Reinli, K.; Block, G. Phytoestrogen content of foods: a compendium of literature values. Nutr. Cancer Int. J. 1996, 26, 123–148. [Google Scholar] [CrossRef]
- Adlercreutz, H.; Mazur, W. Phyto-oestrogens and Western diseases. Ann. Med. 1997, 29, 95–120. [Google Scholar] [CrossRef]
- Rousseff, R. L.; Martin, S. F.; Youtsey, C. O. Quantitative survey of narirutin, naringin, heperidin, and neohesperidin in Citrus. J. Agric. Food Chem. 1987, 35, 1027–1030. [Google Scholar] [CrossRef]
- Vinokur,Y. ; Rodov, V.; Reznick, N.; Goldman, G.; Horev, B.; Umiei, N.; Friedman, H. Rose Petal Tea as an Antioxidant rich Beverage: Cultivar Effects. Food Sci. 2006, 71, 842–847. [Google Scholar] [CrossRef]
- Huang, S.W.; Frankel, E.N. Antioxidant activity of tea catechins in different lipid systems. J. Agric. Food Chem. 1997, 45, 3033–3038. [Google Scholar] [CrossRef]
- Cassano, A.; Jiao, B.; Drioli, E. Production of concentrated kiwifruit juice by integrated membrane processes. Food Res. Int. 2004, 37, 139–148. [Google Scholar] [CrossRef]
- Schieber, A.; Mihalev, K.; Berardini, N.; Mollov, P.; Carle, R. (2005). Flavonol glycosides from distilled petals of Rosa damascena Mill. Z Naturforsch C J Biosci. 2005, 60, 379–384. [Google Scholar] [CrossRef]
- Dushkova, M.; Mihalev, K.; Dinchev, A.; Vasilev, K.; Georgiev, D.; Terziyska, M. Concentration of Polyphenolic Antioxidants in Apple Juice and Extract Using Ultrafiltration. Membranes (Basel). 2022, 12, 1032. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Qaid, S.; Zait, M.; EL Kacemi, K.; ELMidaoui, A.; EL Hajji. H.; Taky, M. Ultrafiltration for clarification of Valencia orange juice: comparison of two flat sheet membranes on quality of juice production. J. Mater. Environ. Sci. 2017, 8, 1186–1194. [Google Scholar]
- Acosta, O.; Vaillant, F.; Pérez, A.M.; Dornier, M. Potential of ultrafiltration for separation and purification of ellagitannins in blackberry (Rubus adenotrichus Schltdl.) juice. Sep. Purif. Tecnol 2014, 125, 120–125. [Google Scholar] [CrossRef]
- Cassano, A.; Conidi, C.; Drioli, E. Clarification and concentration of pomegranate juice (Punica granatum L.) using membrane processes. J. Food Eng. 2011, 107, 366–373. [Google Scholar] [CrossRef]
- Cai, M.; Xie, C.; Lv, Y.; Yang, K.; Sun, P. Changes in physicochemical profiles and quality of apple juice treated by ultrafiltration and during its storage. Food Sci. Nutr. 2020, 8, 2913–2919. [Google Scholar] [CrossRef]
- Gaglianò, M.; Conidi, C.; De Luca, G.; Cassano, A. Partial removal of sugar from apple juice by nanofiltration and discontinuous diafiltration. Membranes 2022, 12, 712. [Google Scholar] [CrossRef]
- Lai, D.Q.; Tagashira, N.; Hagiwara, S.; Nakajima, M.; Kimura, T.; Nabetani, H. Influences of Technological Parameters on Cross-Flow Nanofiltration of Cranberry Juice. Membranes 2021, 11, 329. [Google Scholar] [CrossRef]
- Wei, D.S.; Hossain, M.; Saleh, Z.S. Separation of polyphenolics and sugar by ultrafiltration: Effects of operating conditions on fouling and diafiltration. Int. J. Nat. Sci. Eng. 2008, 1, 10–17. [Google Scholar]
- Toker, R; Karhan,M; Tetik,N; Turhan, I; Oziyci,H. R. Effect of Ultrafiltration and Concentration Processes on the Physical and Chemical Composition of Blood Orange Juice. J. Food Process. Preserv. 2014, 38, 1321–1329. [Google Scholar] [CrossRef]
- Galanakis, C.M.; Markouli, E.; Gekas, V. Recovery and fractionation of different phenolic classes from winery sludge using ultrafiltration. Sep. Purif. Technol. 2013, 107, 245–251. [Google Scholar] [CrossRef]
- Uyttebroek, M,; Vandezande, P. ; Miet Van Dael, M.V; Vloemans, S.; Noten, B.; Bongers, B,; Porto-Carrero, W.; Unamunzaga, M.M.; Bulut, M.; Lemmens, B. Concentration of phenolic compounds from apple pomace extracts by nanofiltration at lab and pilot scale with a techno-economic assessment. J. Food Process. Preserv. 2017, 41, e12629. [CrossRef]
- Mello, B.C.B.S.; Petrus, J.C.C.; Hubinger, M.D. Concentration of flavonoids and phenolic compounds in aqueous and ethanolic propolis extracts through nanofiltration. J. Food Eng. 2010, 96, 533–539. [Google Scholar] [CrossRef]
- Farnosova, E.N.; Rid, A.A.; Morozova, Y.A. Purification and concentration of potato juice by ultrafiltration and nanofiltration. IOP Conf. Ser.: Earth Environ. Sci. 2021, 723, 022066. [Google Scholar] [CrossRef]
- Magalhães, L.M.; Segundo, M.A.; Reis, S.; Lima, J.L.F.C. Methodological aspects about in vitro evaluation of antioxidant properties. Anal. Chim. Acta 2008, 613, 1–19. [Google Scholar] [CrossRef]
- Cissé, M.; Vaillant, F.; Pallet, D.; Dornier, M. Selecting ultrafiltration and nanofiltration membranes to concentrate anthocyanins from roselle extract (Hibiscus sabdariffa L. ). Food Res. Int. 2011, 44, 2607–2614. [Google Scholar] [CrossRef]
- Conidi, C.; Cassano, A.; Caiazzo, F.; Drioli, E. Separation and purification of phenolic compounds from pomegranate juice by ultrafiltration and nanofiltration membranes. J. Food Eng. 2017, 195, 1–13. [Google Scholar] [CrossRef]

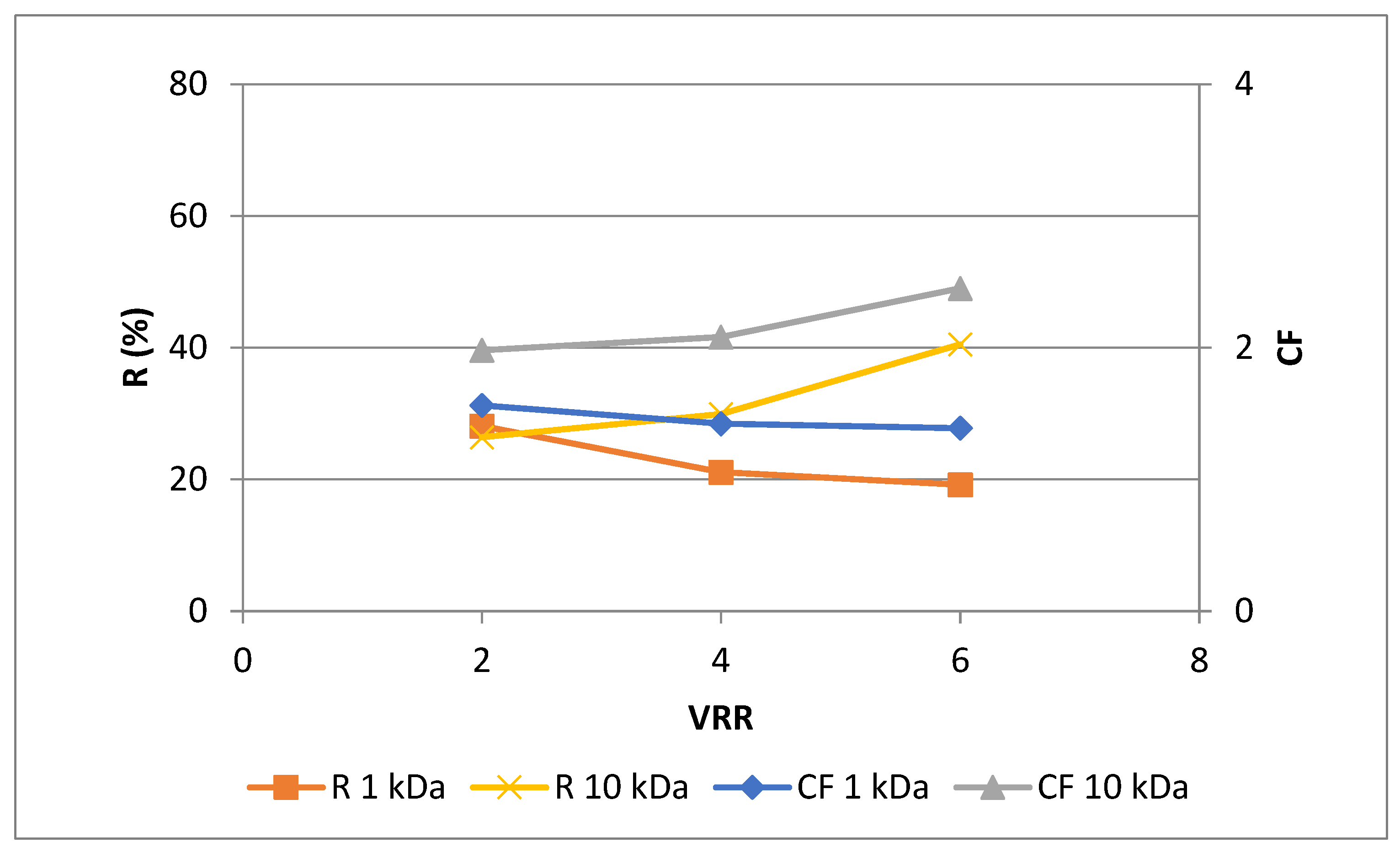
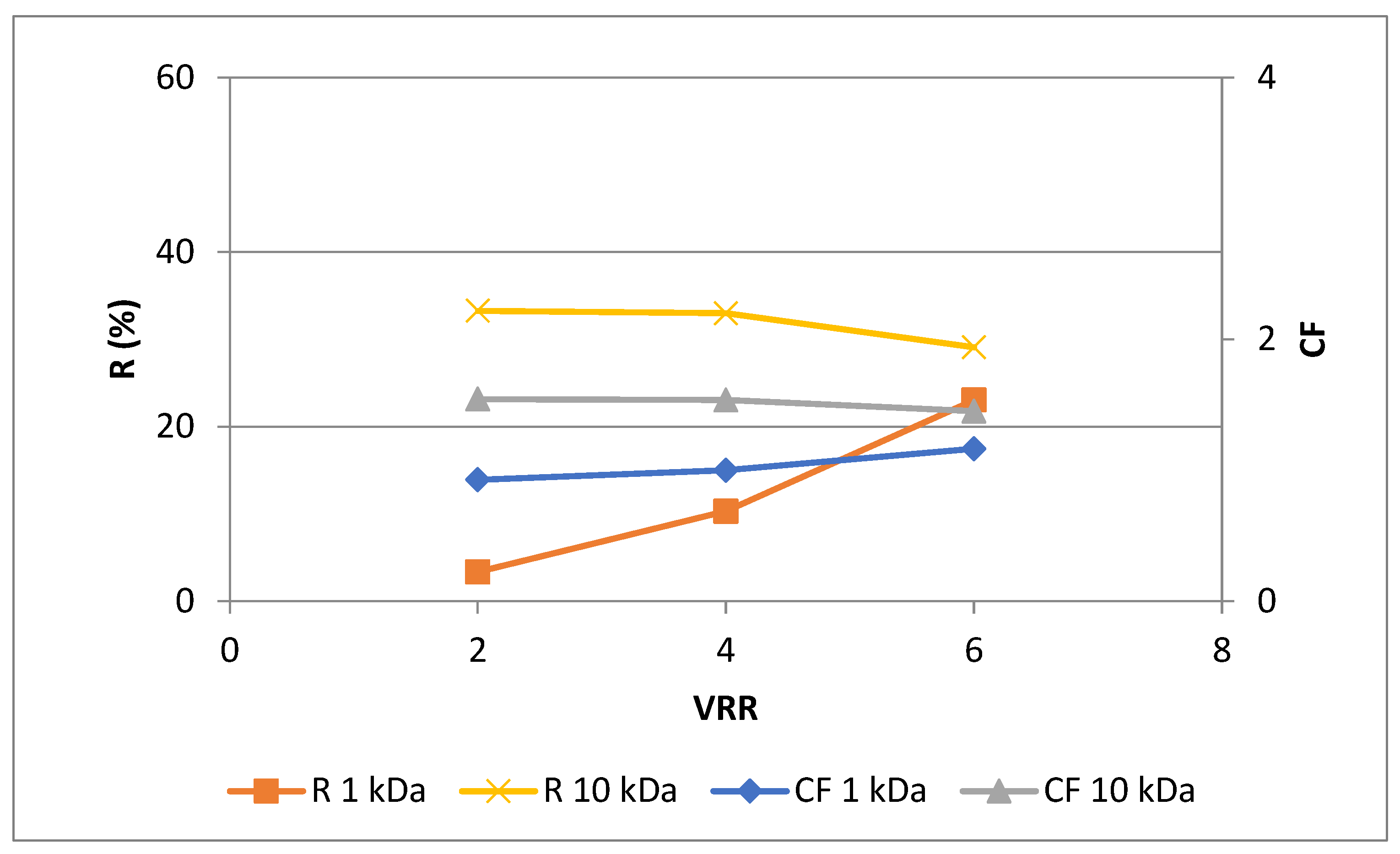
| Extract type | MWCO | |
|---|---|---|
| 1 kDa | 10 kDa | |
| Feed solution | 246 ± 11A | 246 ± 11A |
| Permeate | 220 ± 10aA | 226 ± 10aA |
| Retentate (VRRc = 2) | 332 ± 15aB | 310 ± 14aB |
| Retentate (VRR = 4) | 313 ± 14aB | 374 ± 17bC |
| Retentate (VRR = 6) | 341 ± 15aB | 410 ± 18bC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




