Submitted:
16 June 2023
Posted:
19 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
- Type of research
- Statistical analysis
- The methodology of physiotherapy sessions:
3. Results
3.1. Results of the comparative analysis of laboratory results
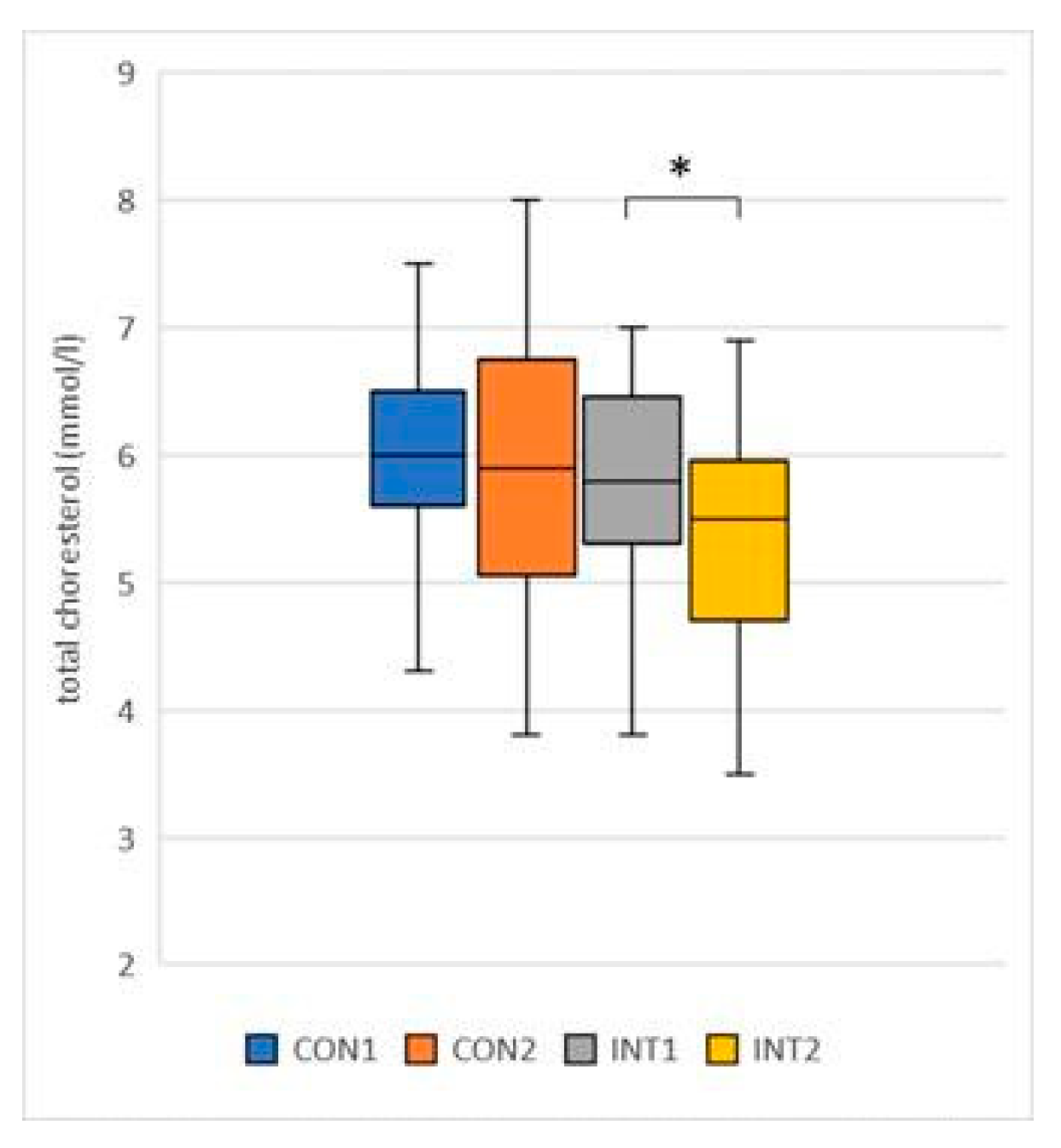
- total cholesterol (mmol/l): the mean decreased in both groups, with -0.14 in the control group from 6.02 to 5.88, but the change was not significant (p=0.636), while the change in the experimental group (-0.44) was significant (p=0.018), with a decrease from 5.81 to 5.37 (Figure 1);
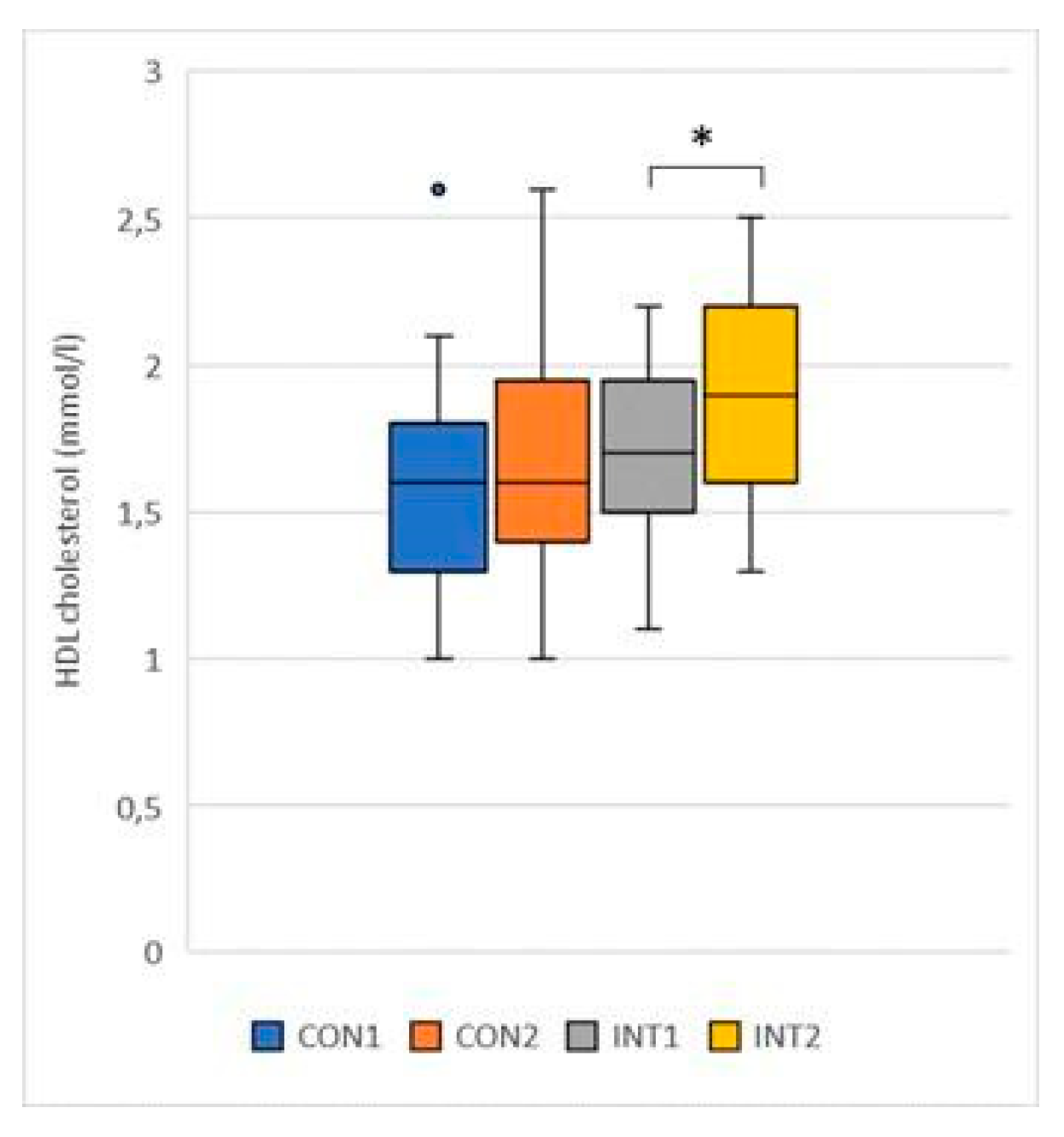
- HDL cholesterol (mmol/l): the mean of both groups showed an increase, in the control group by 0.07 from 1.59 to 1.66, the change was not significant (p=0.121), in the experimental group the change increased by 0.23 from 1.69 to 1.92, which was significant (p=0.004) (Figure 2);
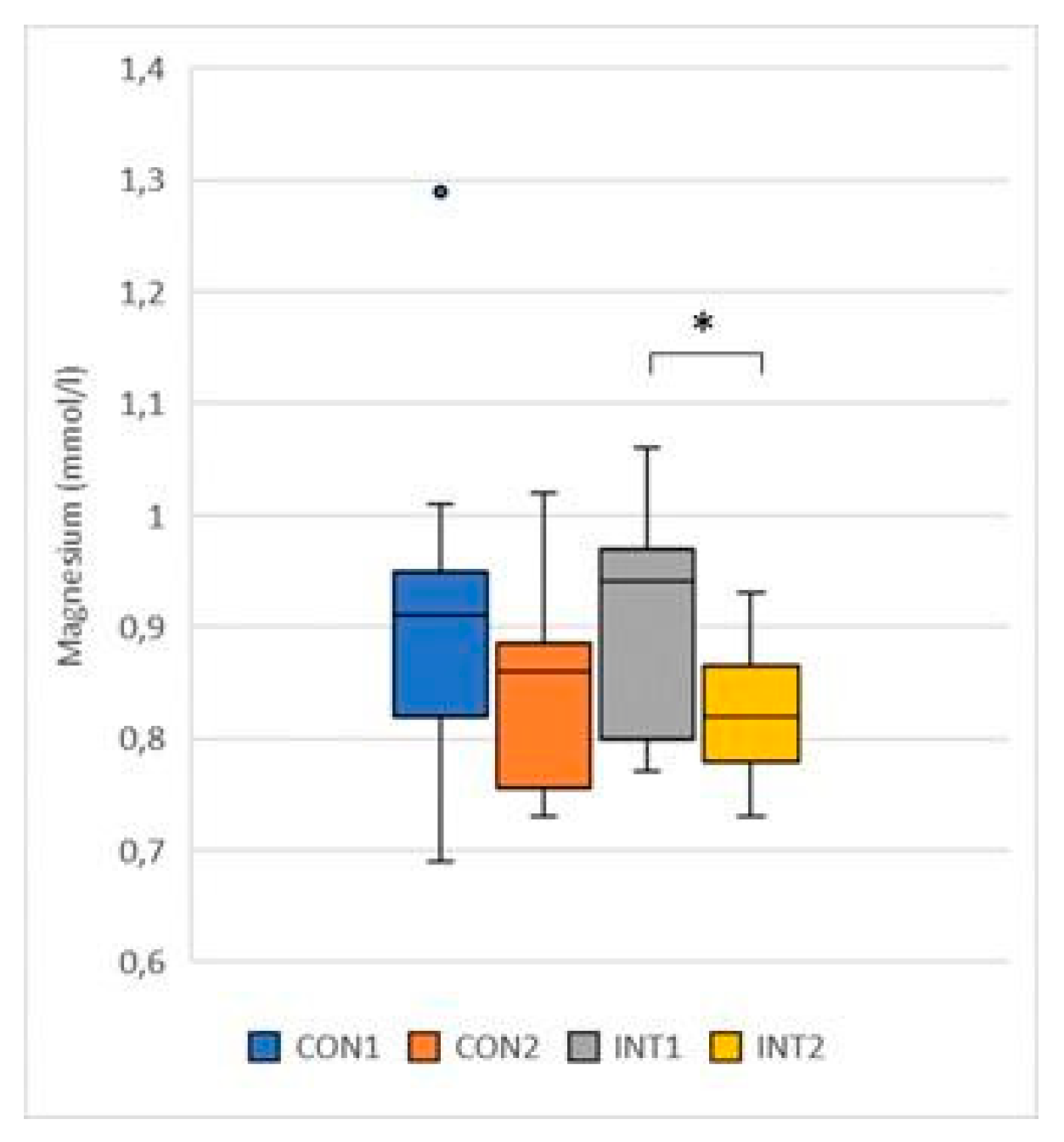
- Magnesium (mmol/l): the mean decreased in both groups, in the control group by -0.07, from 0.90 to 0.84, showing no significant change (p=0.096), in the experimental group the decrease was -0.07, from 0.90 to 0.83, reaching significance (p=0.002) (Figure 3);
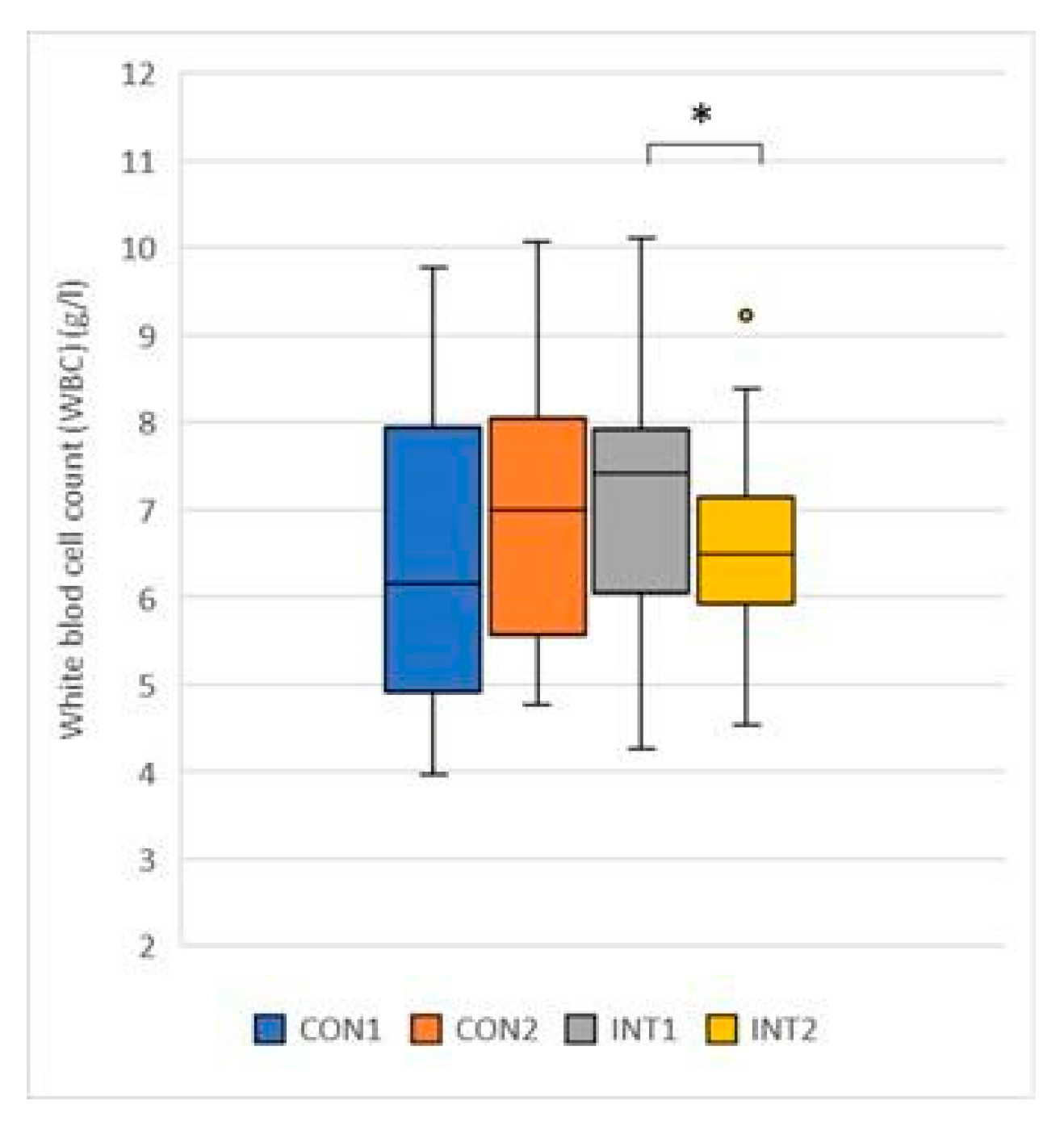
- White blood cell count (G/L): the mean showed an opposite trend, although neither group was outside the normal range at the beginning or at the end of the study. While it increased by 0.49 from 6.47 to 6.96 in the control group to a non-significant extent (p=0.232), it decreased significantly in the experimental group by -0.46 from 6.96 to 6.67 (p=0.030) (Figure 4).
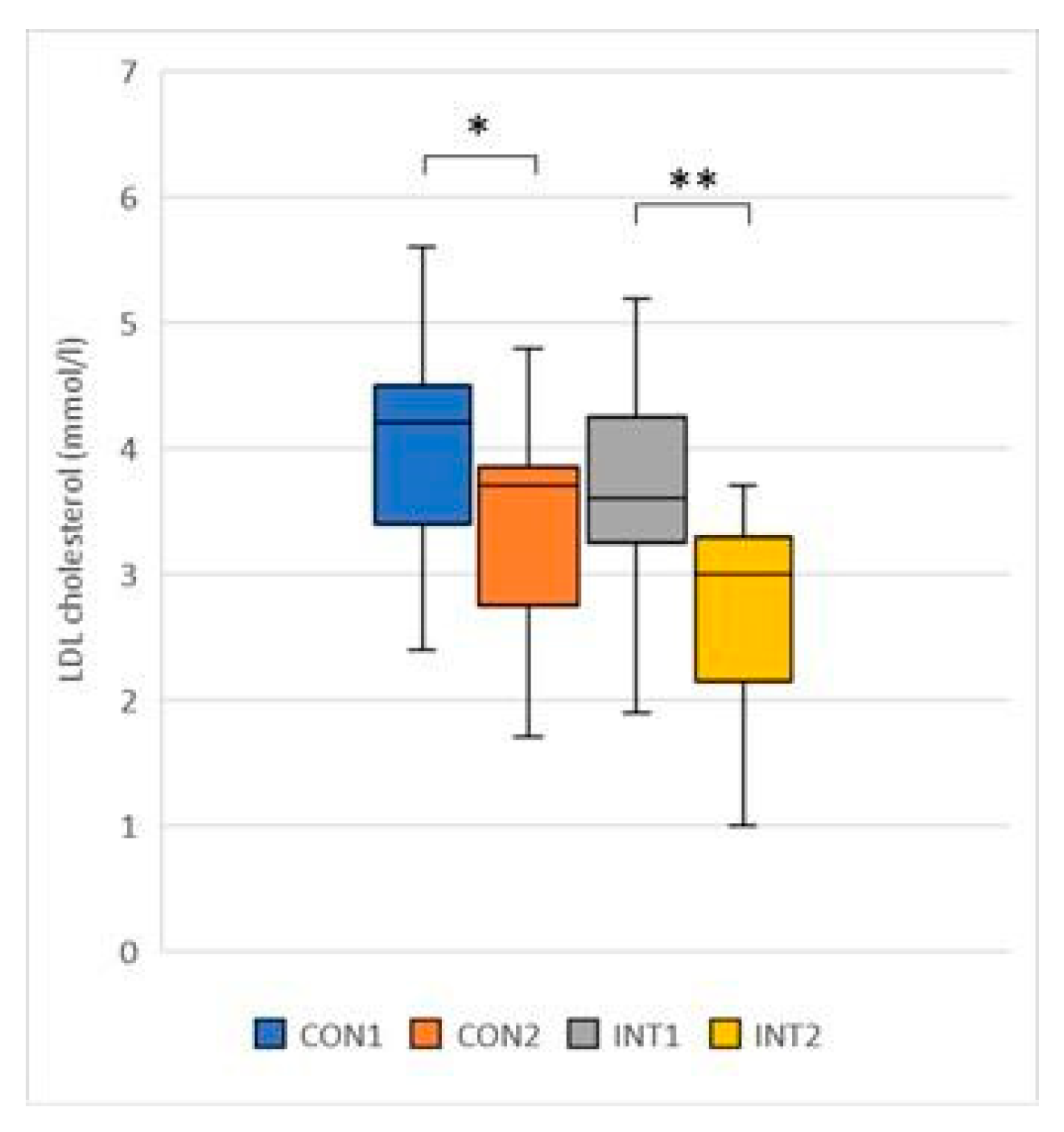
- LDL (mmol/l): the mean of both groups showed a significant decrease, with the control group decreasing by -0.74 from 4.08 to 3.35 (p=0.003) and the experimental group decreasing by -0.98 from 3.71 to 2.72 (p=0.000) (Figure 5);
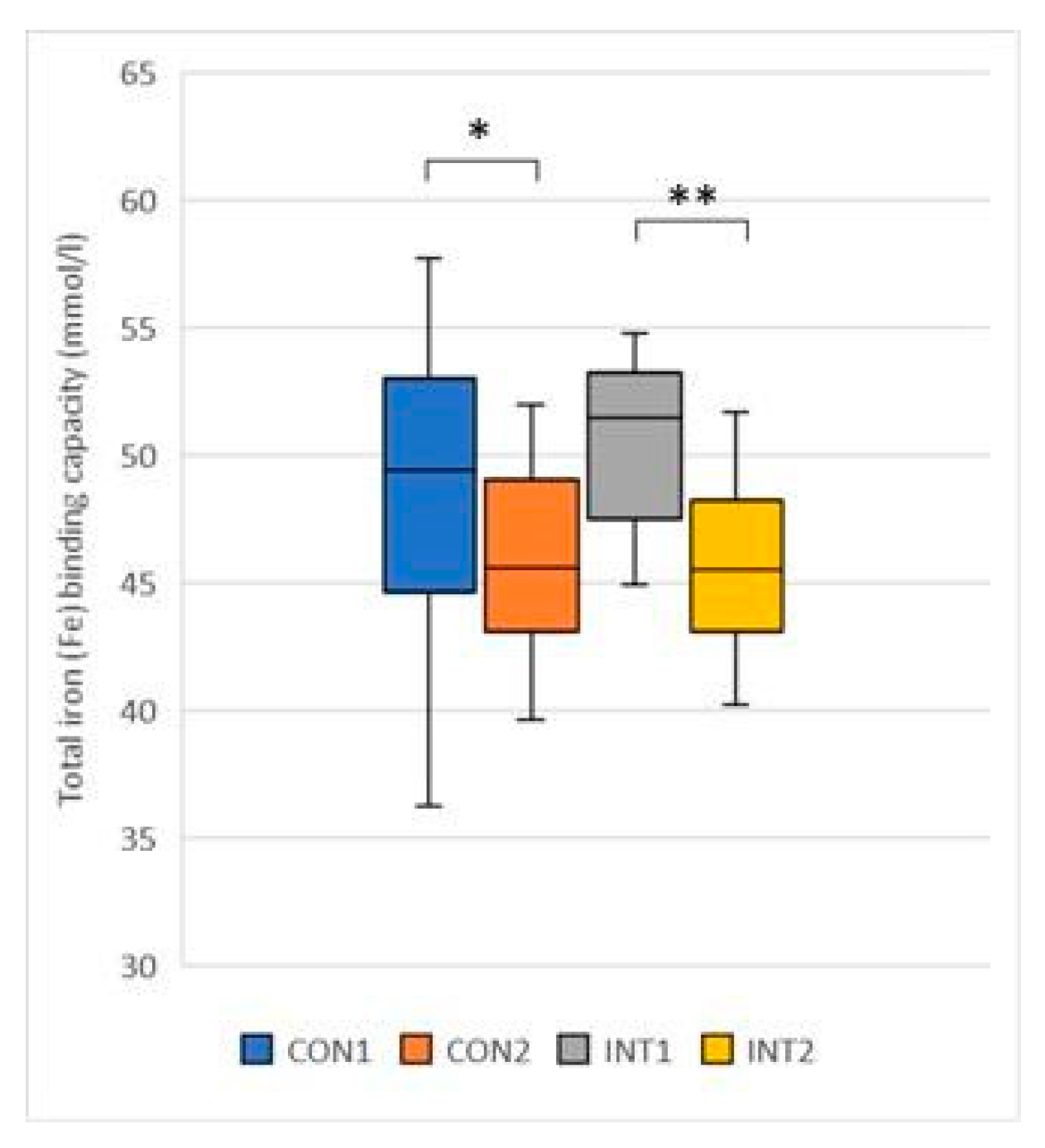
- Total iron-binding capacity: a significant decrease was observed in both groups, with a decrease of -3.67 from 49.38 to 45.71 in the control group (p=0.018) and -4.53 from 50.39 to 45.86 in the experimental group (p=0.000) (Figure 6).
- LDL/HDL ratio: significant reduction in both groups, but slightly greater reduction in the test group (control p=0.006, test p=0.000)
3.2. Results of a comparative study of Inbody results
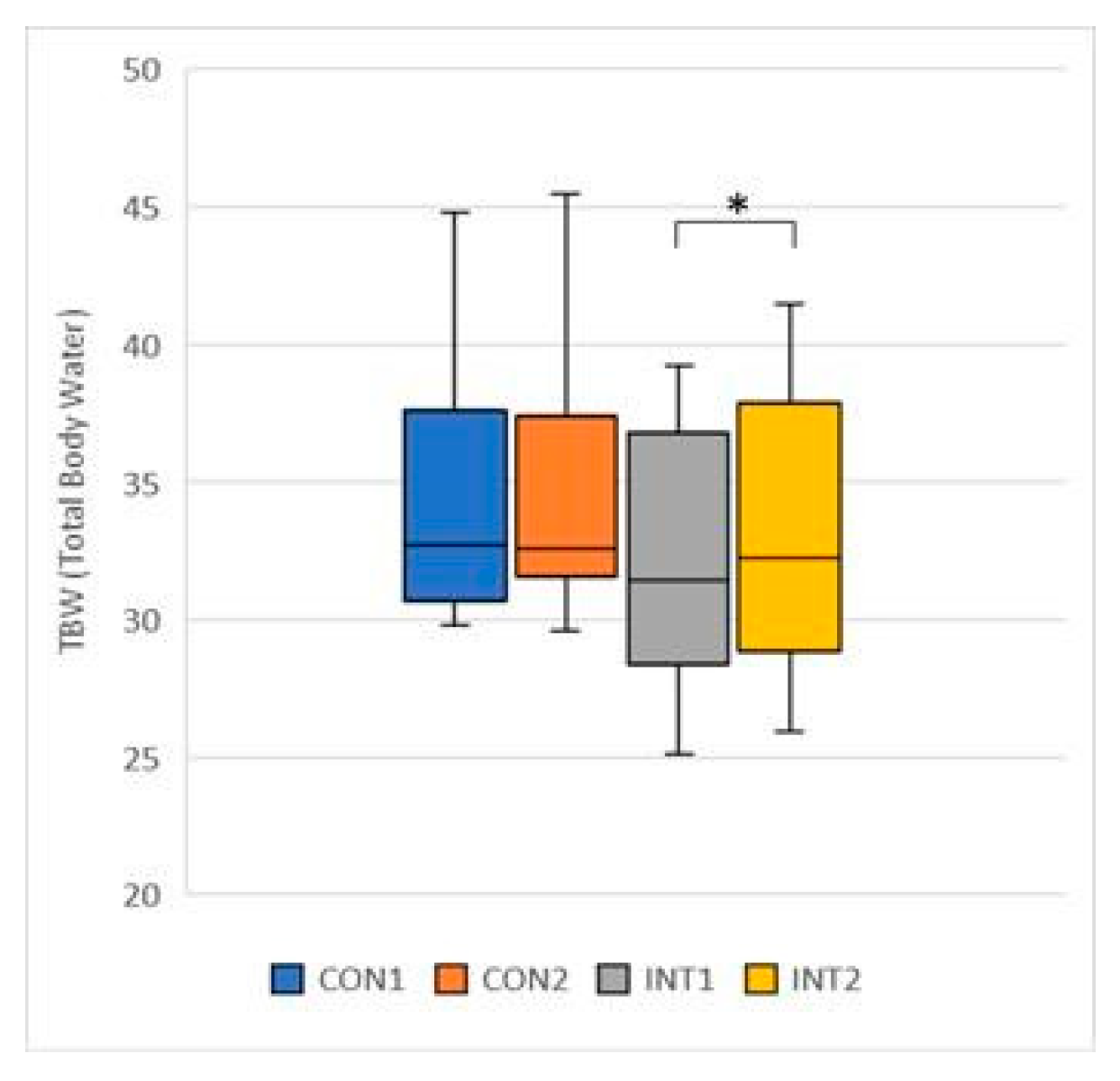
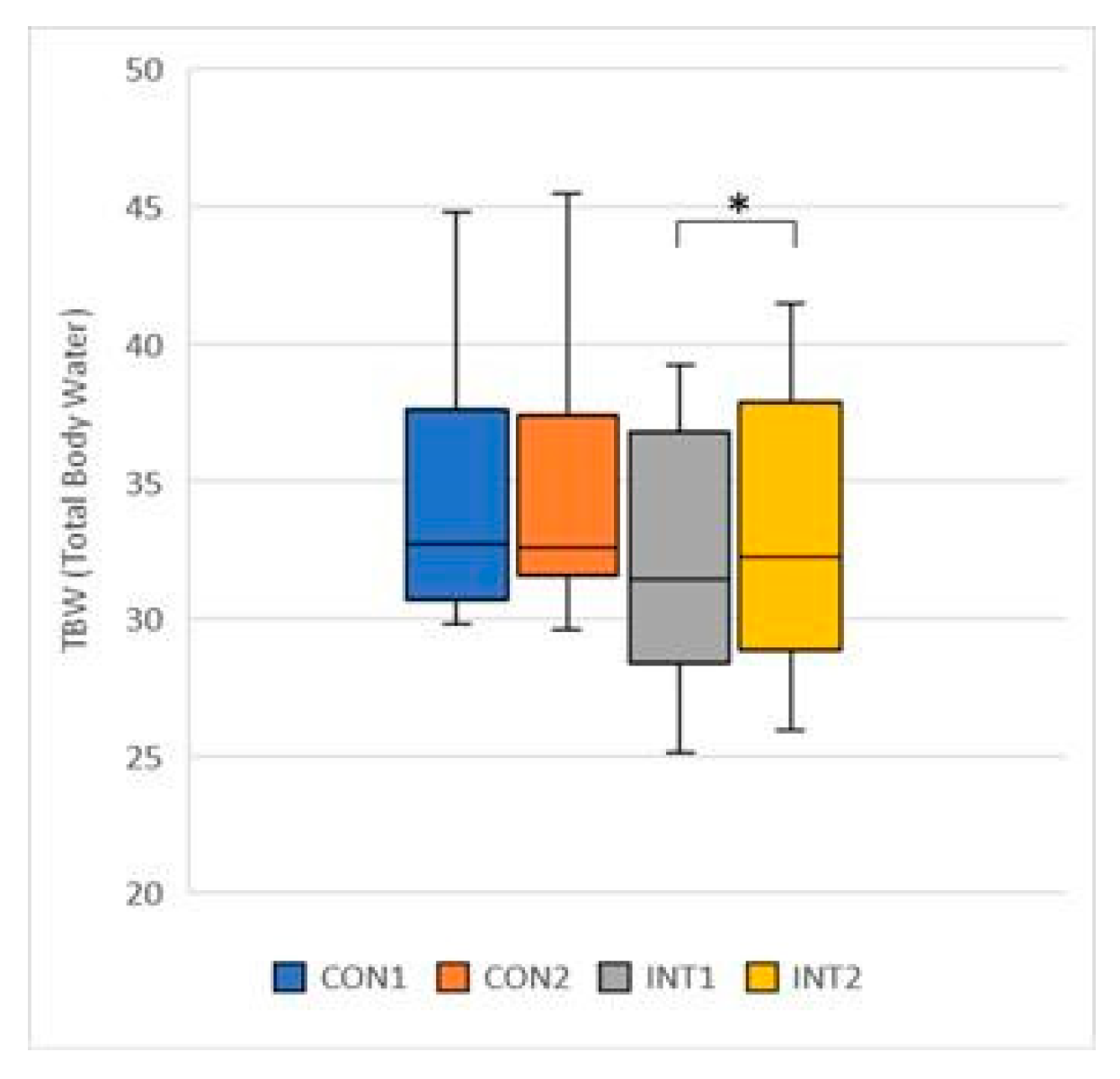
- TBW (Total Body Water): the mean increased for both groups, by 0.10 for the control group from 34.26 to 34.36, a change that was not significant (p=0.744), but by 0.72 for the experimental group from 32.26 to 32.98, a significant change (p=0.003) (Figure 7);
- ICW (Intracellular Water): the change of the mean for the control group was negligible, increasing by 0.06 from 20.88 to 20.95, (p=0.809), but for the experimental group the increase was 0.82, from 19.84 to 20.65, a significant increase (p=0.013) (Figure 8);
- Protein: the mean of the control group was 0.05, increasing from 9.04 to 9.08 by a non-significant amount (p=.0666), while for the experimental group the results increased by 0.48 from 8.58 to 9.06, a significant change (p=0.000) (Figure 9);
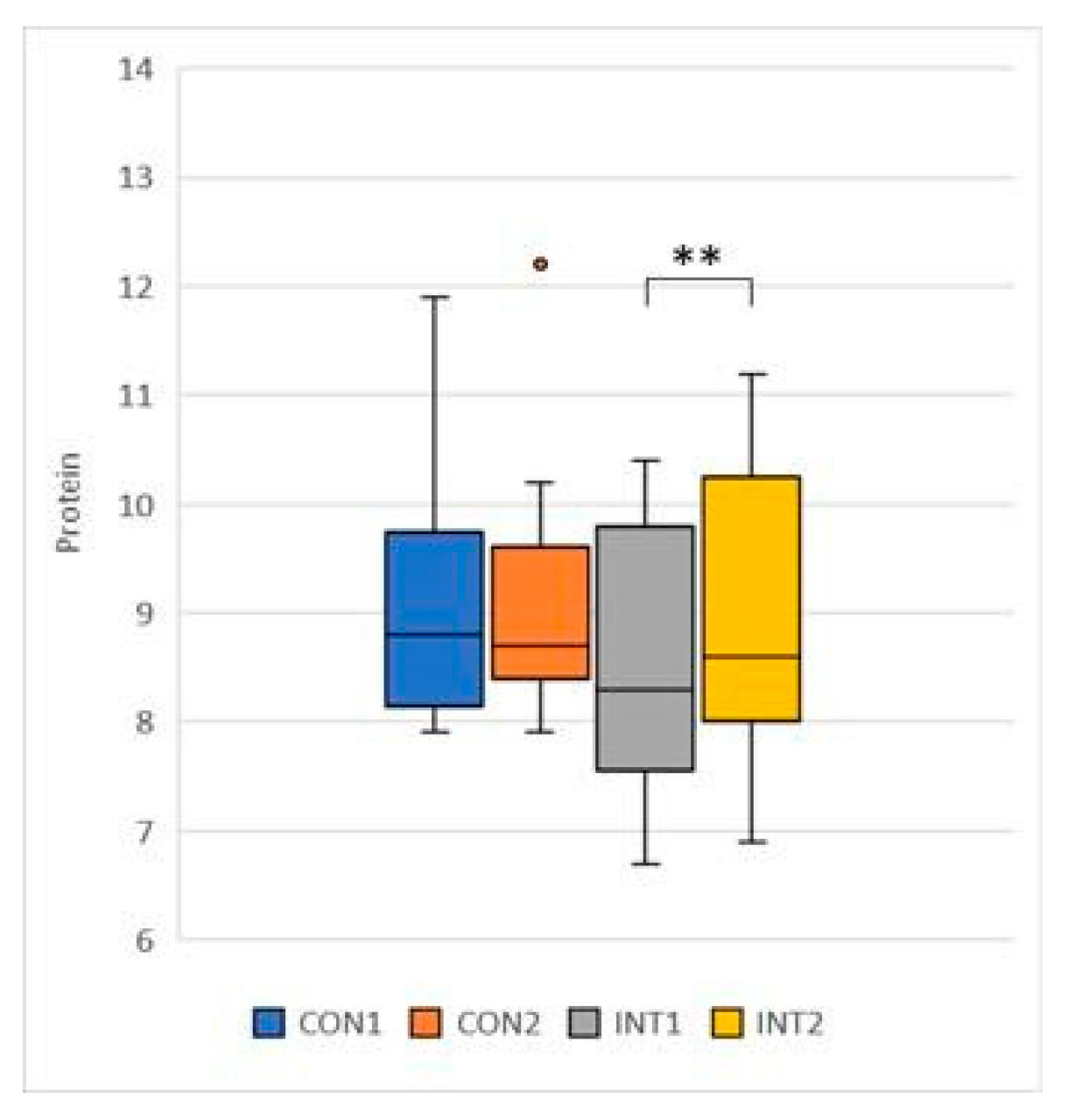
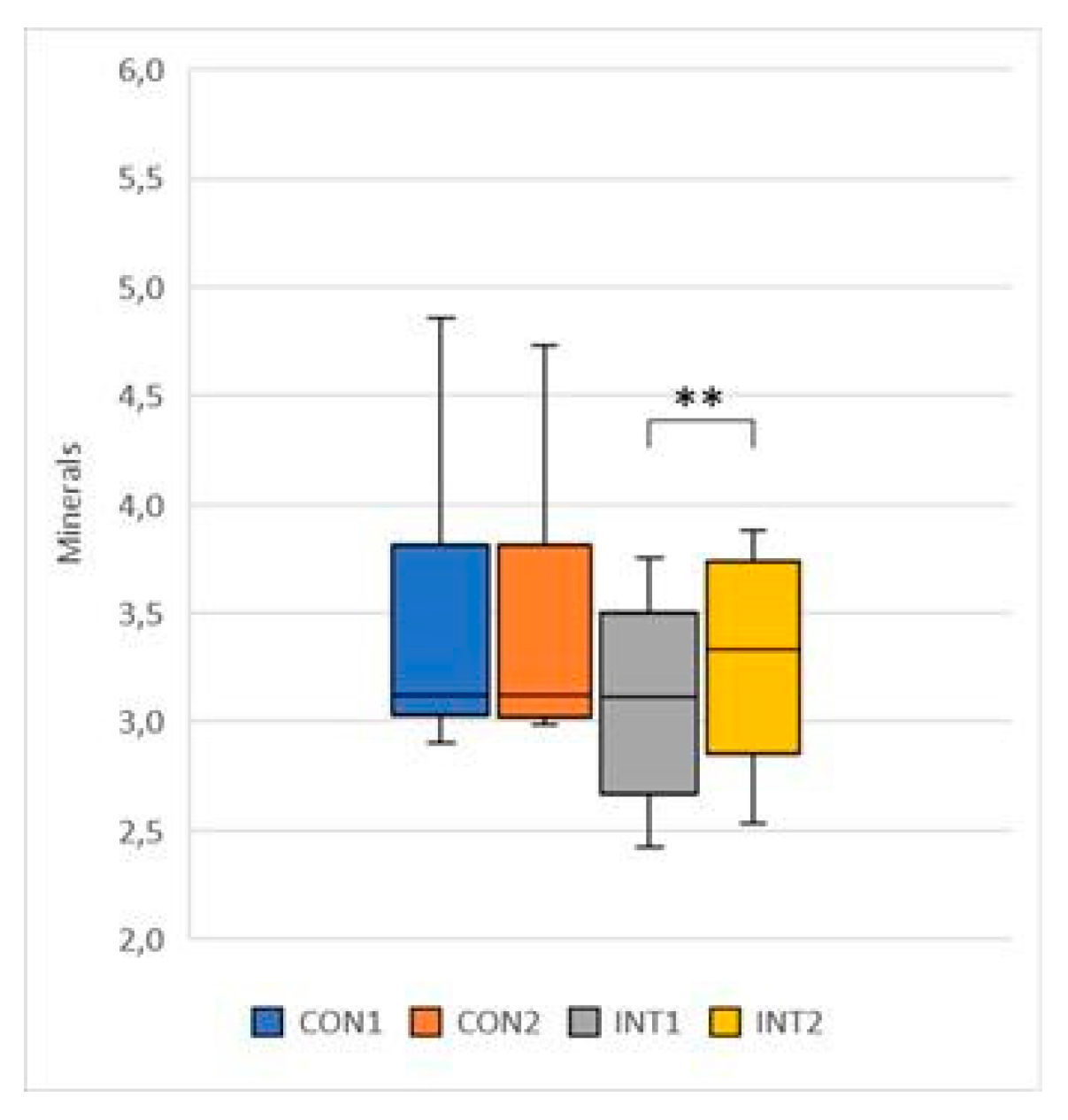
- Minerals: mean increased by 0.03 in the control group, from 3.39 to 3.42, with no significant change (p=0.286), and by 0.19 in the study group, from 3.10 to 3.30, with a significant change (p=0.000) (Figure 10);
- SMM (Skeletal Muscle Mass): the mean of both groups showed an increase. For the control group, the increase in muscle mass was 0.09 from 25.25 to 25.34, which was not significant (p=0.773), while for the experimental group, the increase in muscle mass was significant, with a significant result of 0.67 from 23.88 to 24.55, which was a significant change (p=0.002) (Figure 11);

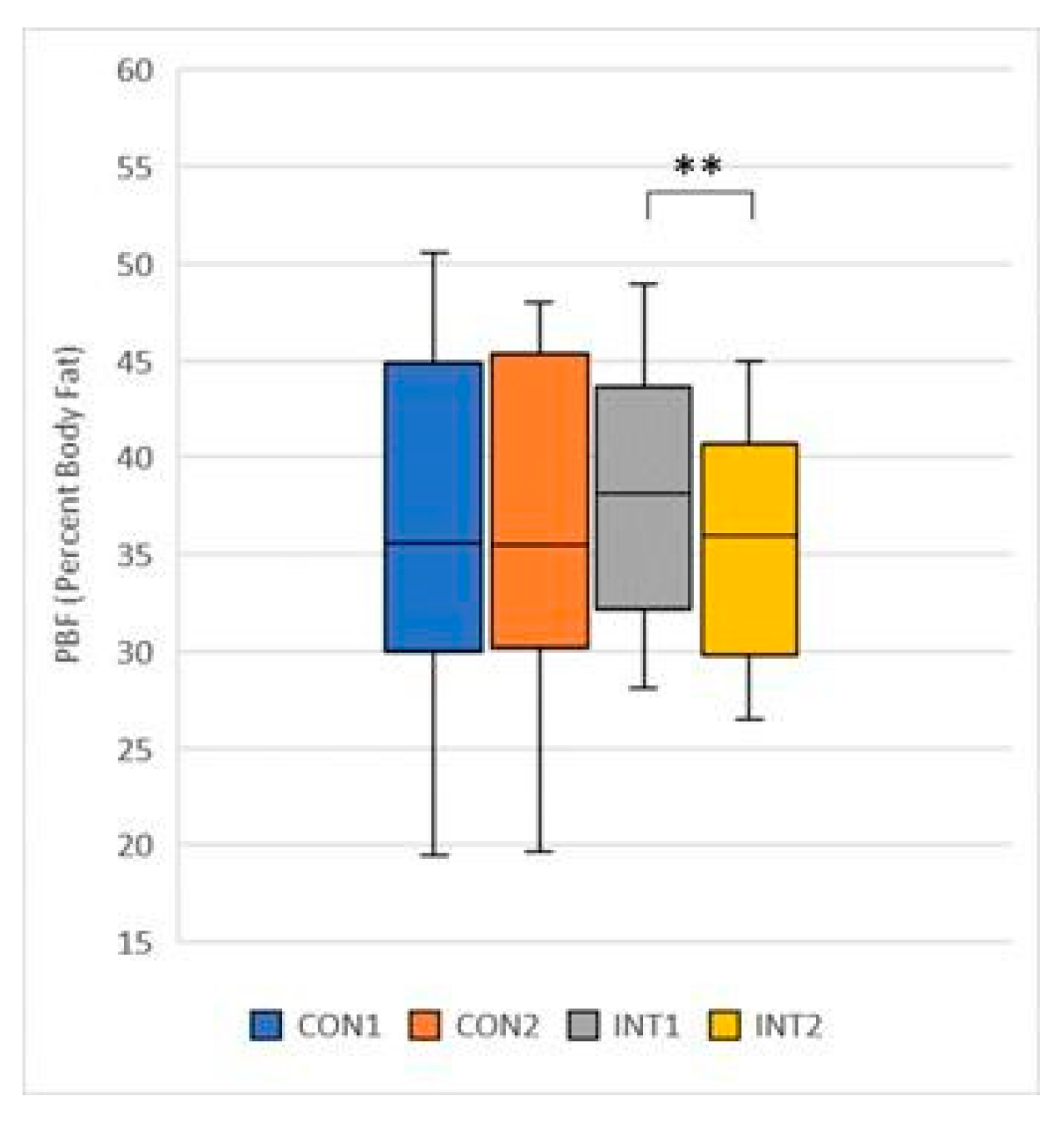
- PBF (Percent Body Fat): its mean decreased in both groups, in the control group by -0.98 from 37.55 to 36.57, which was not significant (p=0.068), in the experimental group the decrease was significant (p=0.000) by -2.57 from 38.30 to 35.73 (Figure 12);
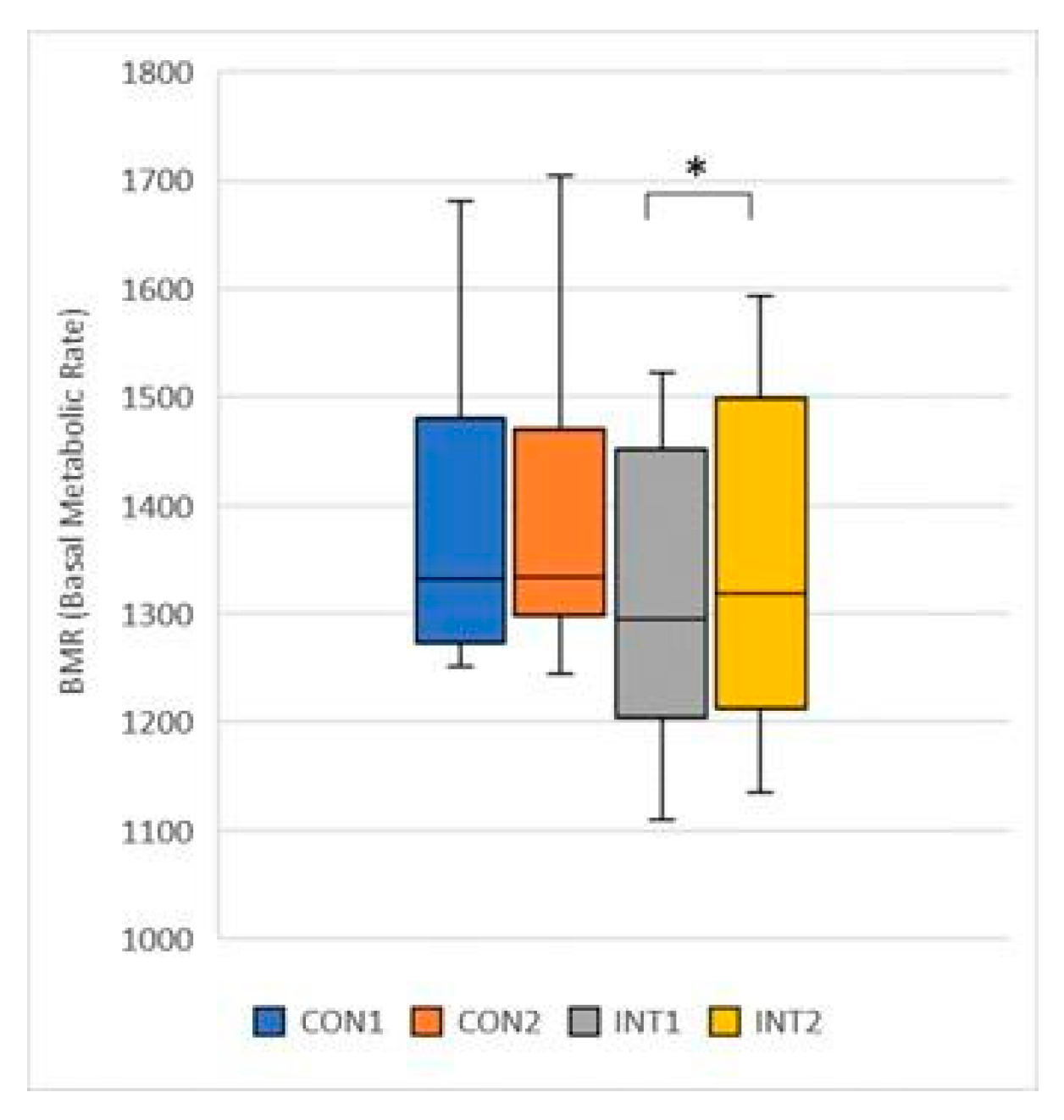
- BMR (Basal Metabolic Rate): the mean increased in both groups, in the control group by 3.38 from 1378.54 to 1381, with a non-significant increase (p=0.717), and in the experimental group by 26.08 from 1319.08 to 1345.15, with a significant change (p=0.022) (Figure 13);
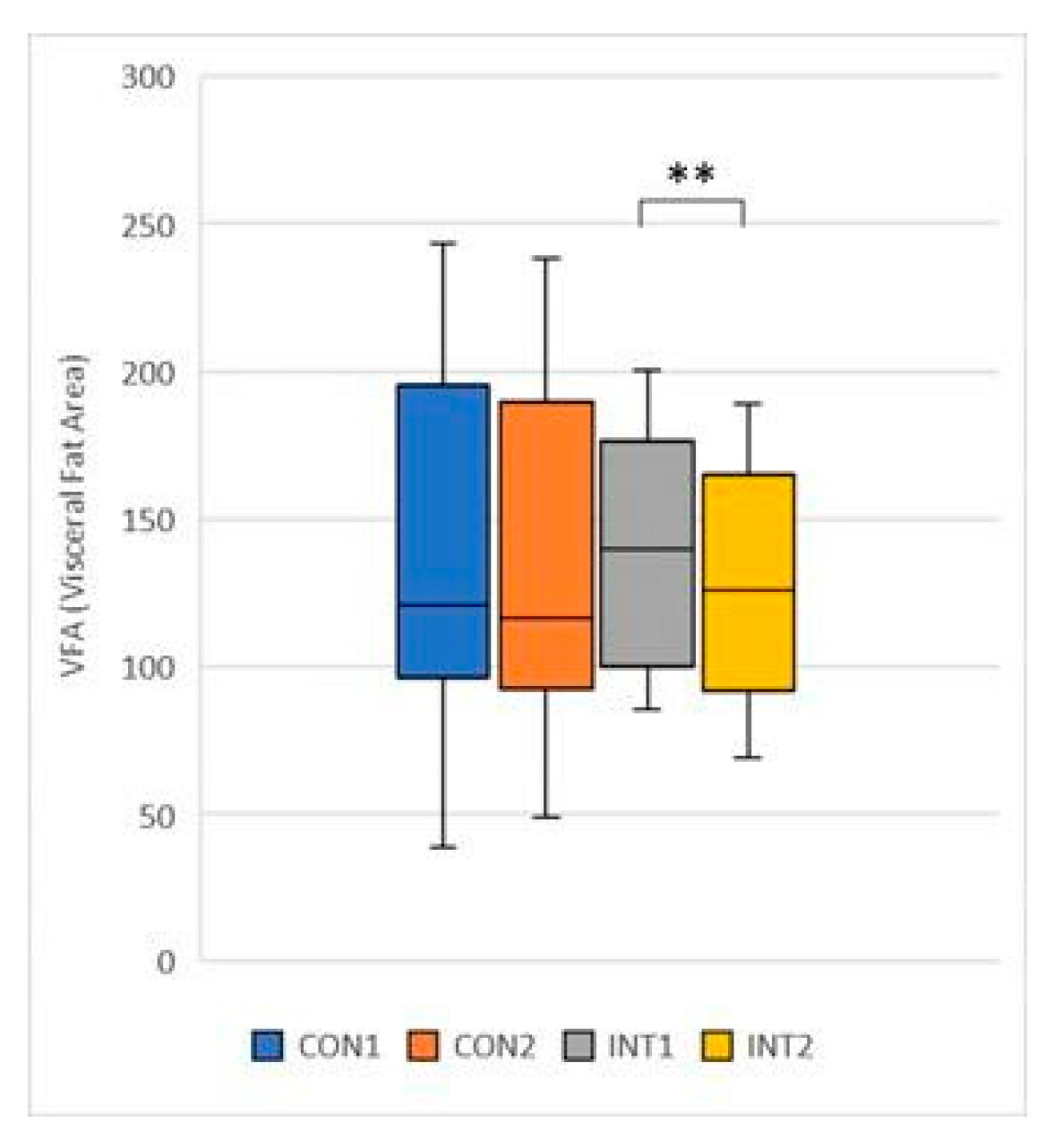
- VFA (Visceral Fat Area): its mean decreased in both groups, the control group by 2.65 from 140.89 to 138.25, non-significantly (p=0.353), while the experimental group showed a decrease of -10.33 from 138.12 to 127.79, significant (p=0.000) (Figure 14).
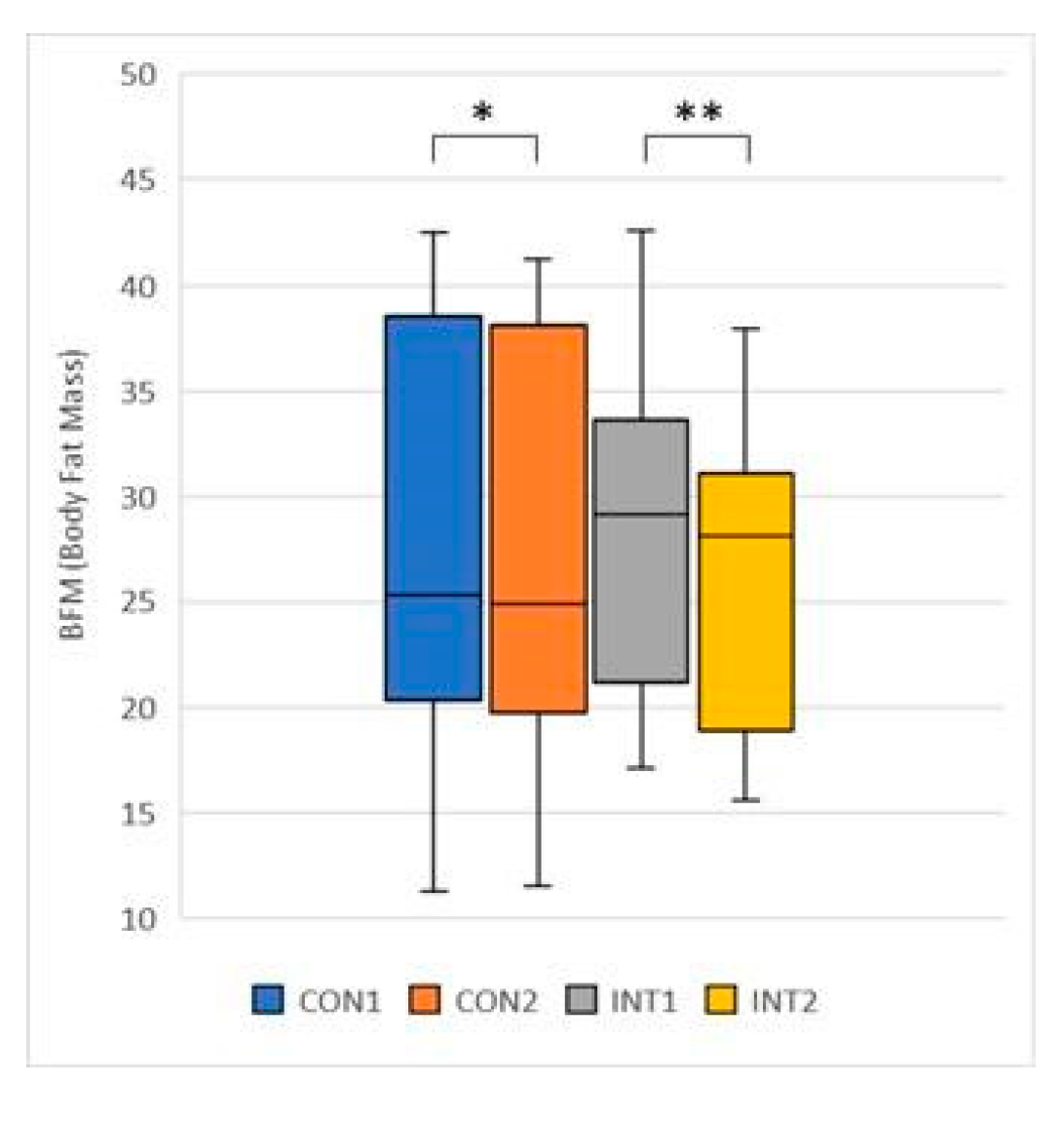
- BFM (Body Fat Mass): its mean decreased significantly in both groups, by -0.74 from 28.48 to 17.75 for the control group (p=0.002) and by -2.09 from 27.72 to 25.62 for the experimental group (p=0.000);
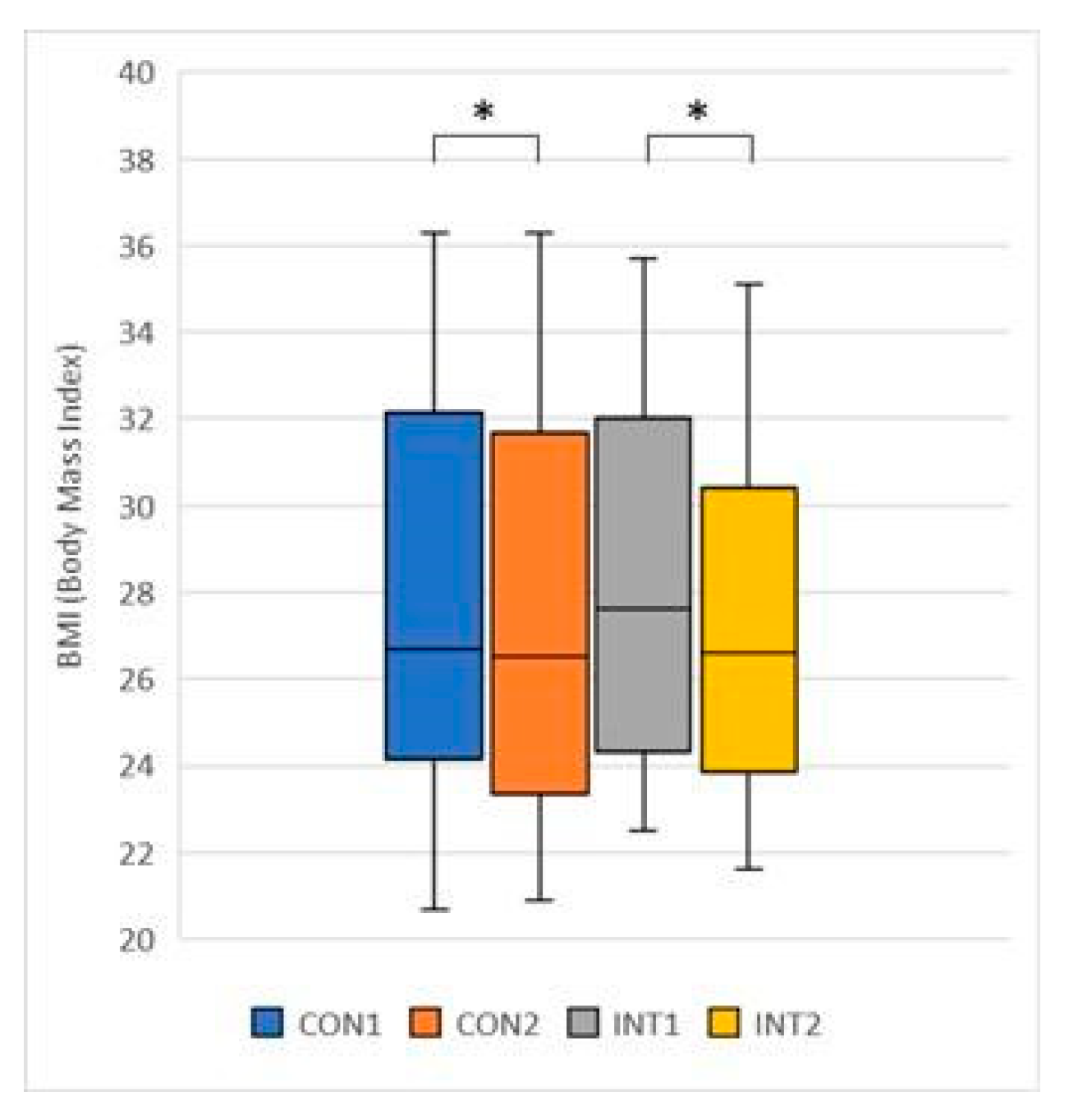
- BMI (Body Mass Index): its mean significantly decreased for both groups, -0.33 from 27.98 to 27.65 (p=0.014) for the control group and -0.86 from 28.09 to 27.23 (p=0.050) for the experimental group;
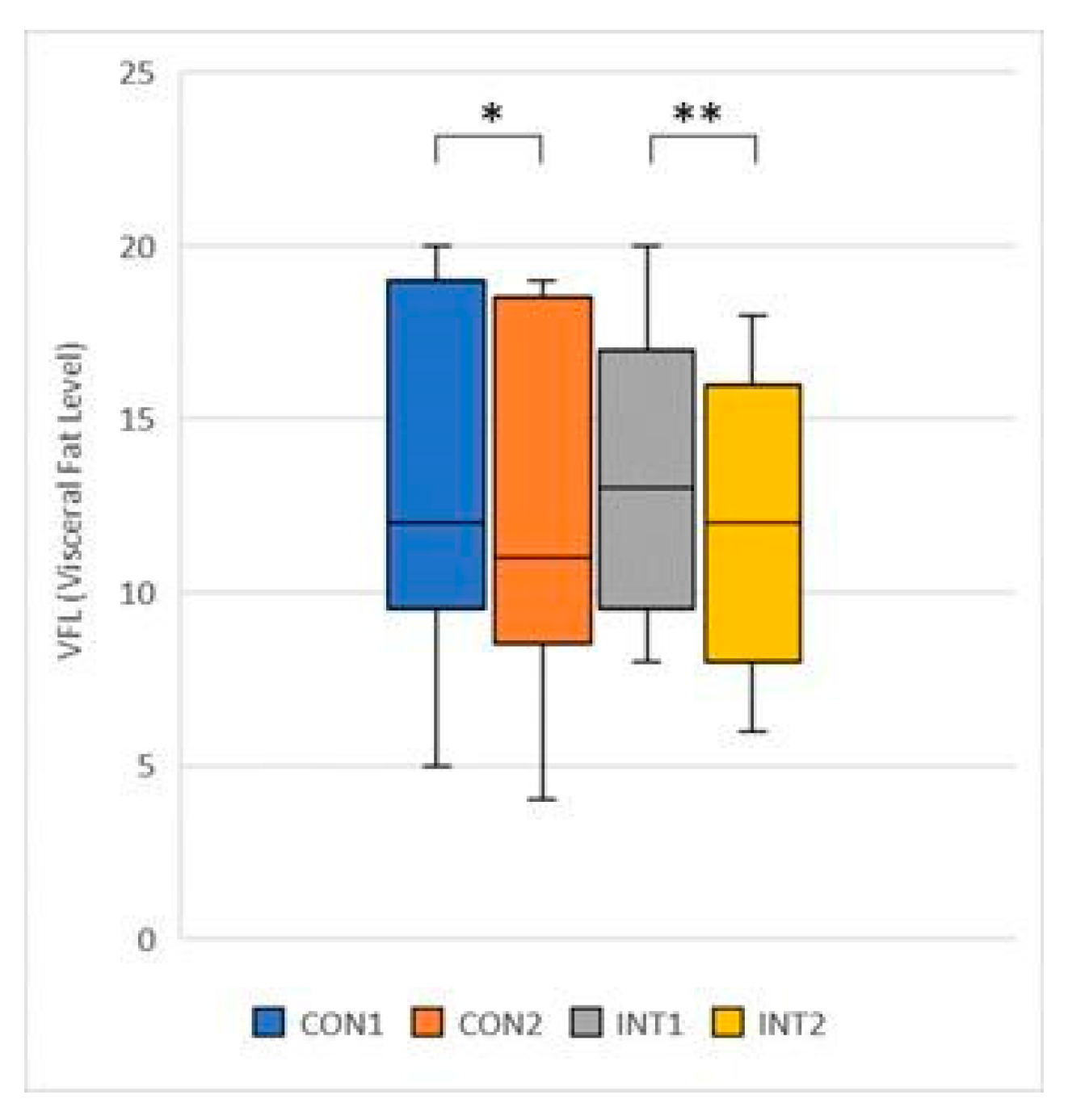
- VFL (Visceral Fat Level): mean significantly decreased in both groups, by -0.69 from 13.46 to 12.77 for the control group (p=0.013) and by -1.38 from 13.38 to 12 for the experimental group (p=0.000).
3.3. Range of motion testing of hip and knee joints (passive range of motion - PROM)
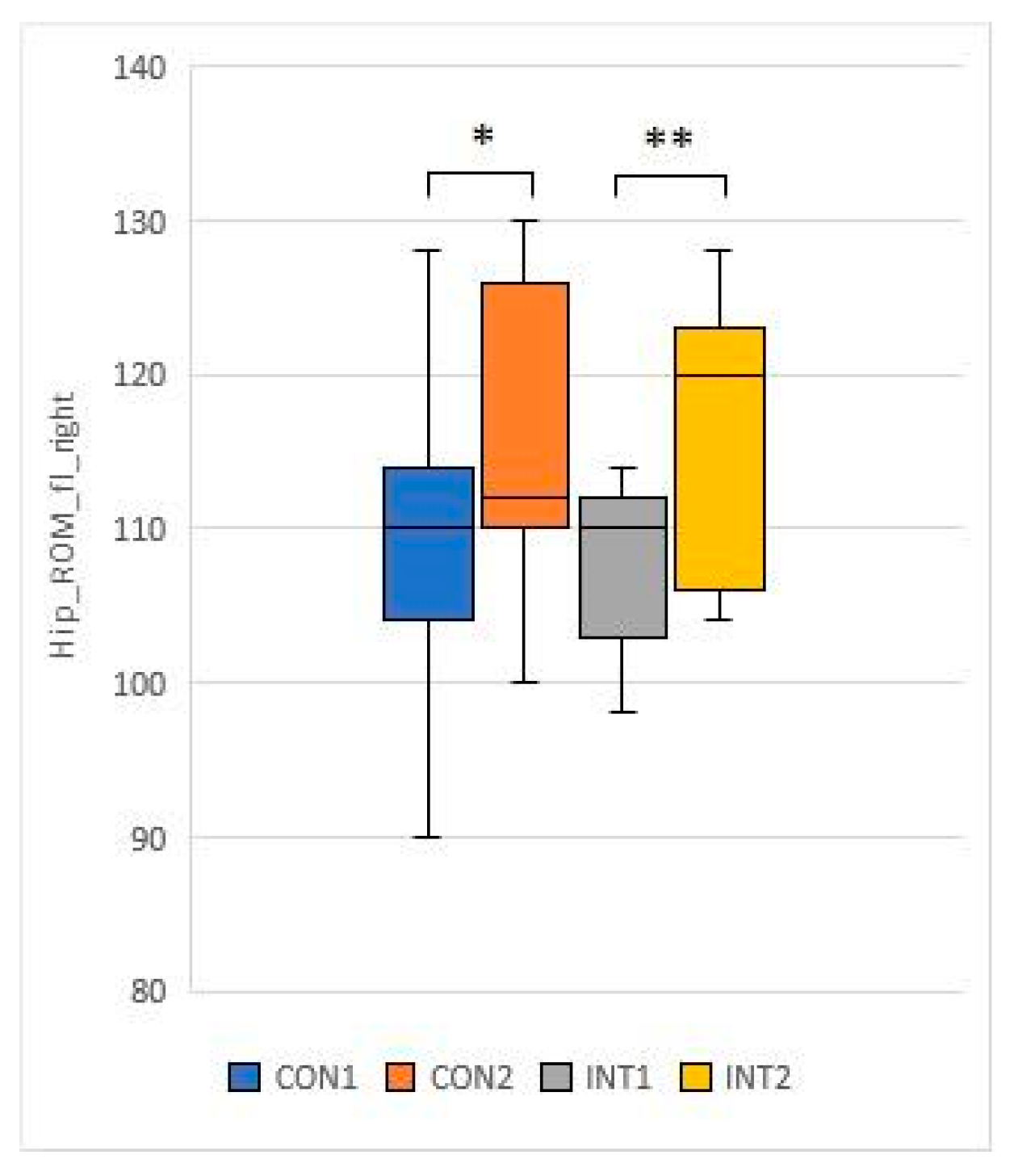
- Hip ROM fl L: its mean increased by 1.5 in the control group from 113.3 to 114.8, while in the experimental group it increased by 7.9 with significant magnitude (p=0.020) from 108.8 to 116.7
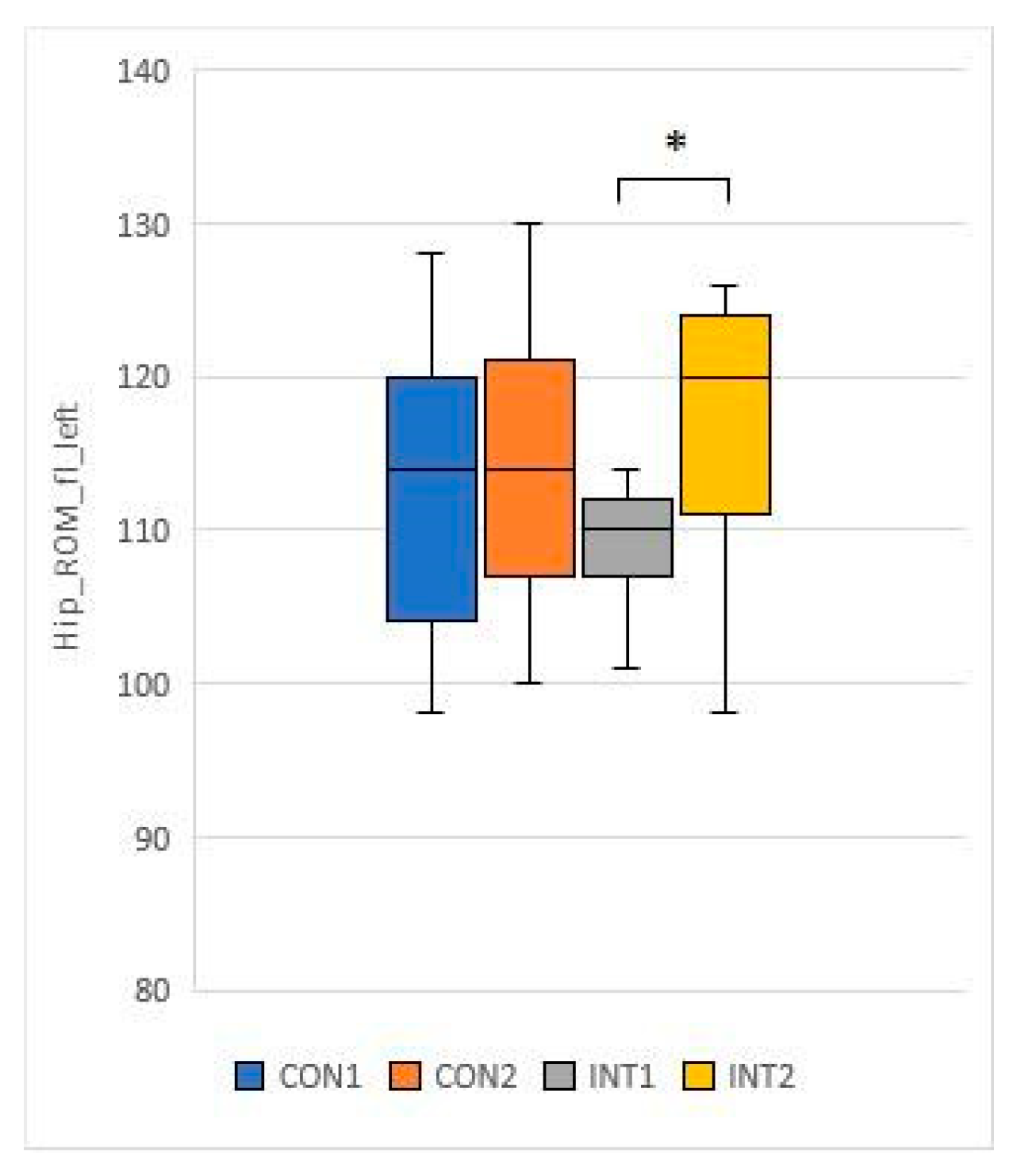
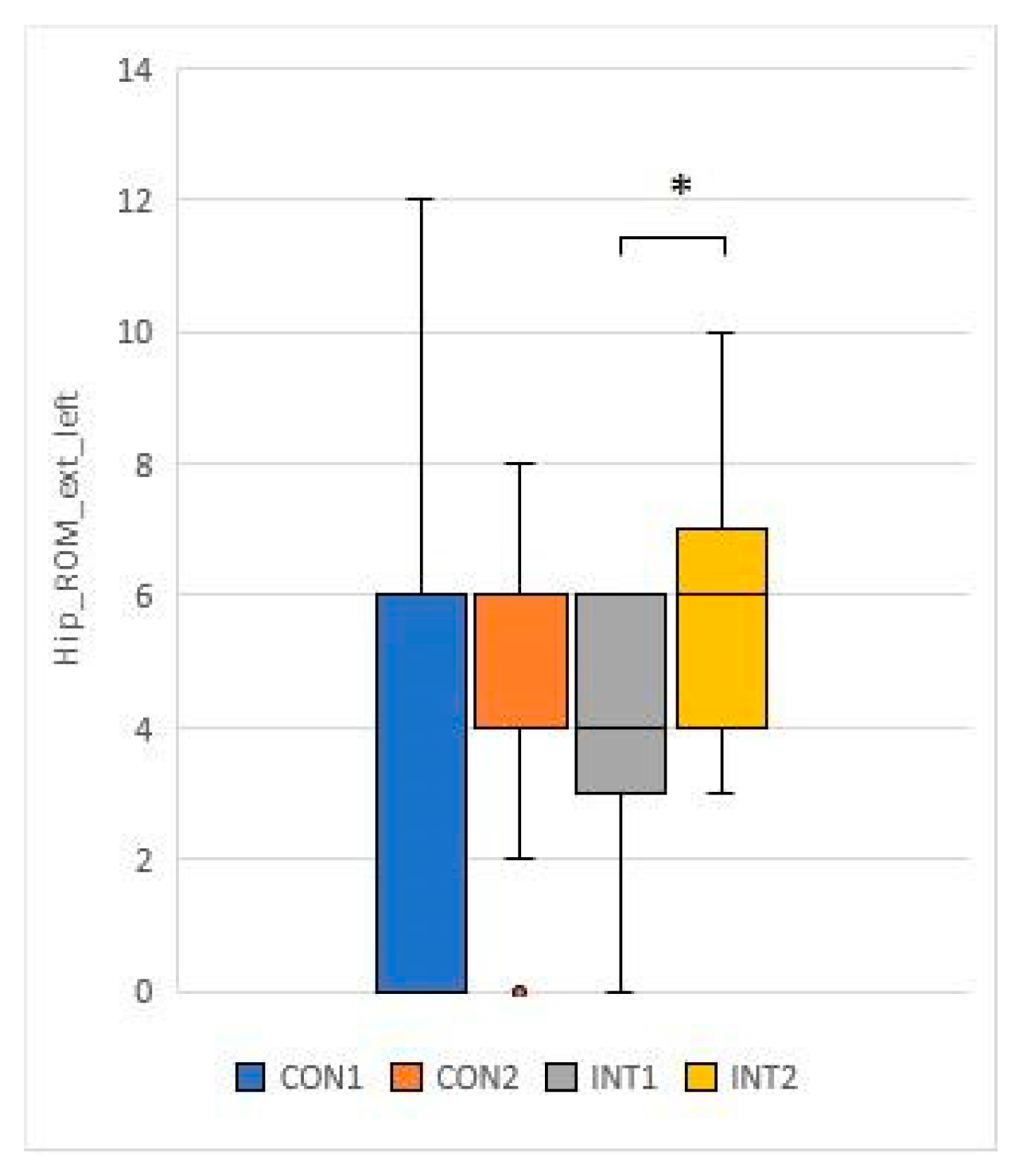
- Hip ROM ext L: mean increased by 0.1 from 4.7 to 4.8 in the control group and significantly (0.046) increased by 1.5 from 4.0 to 5.5 in the experimental group.
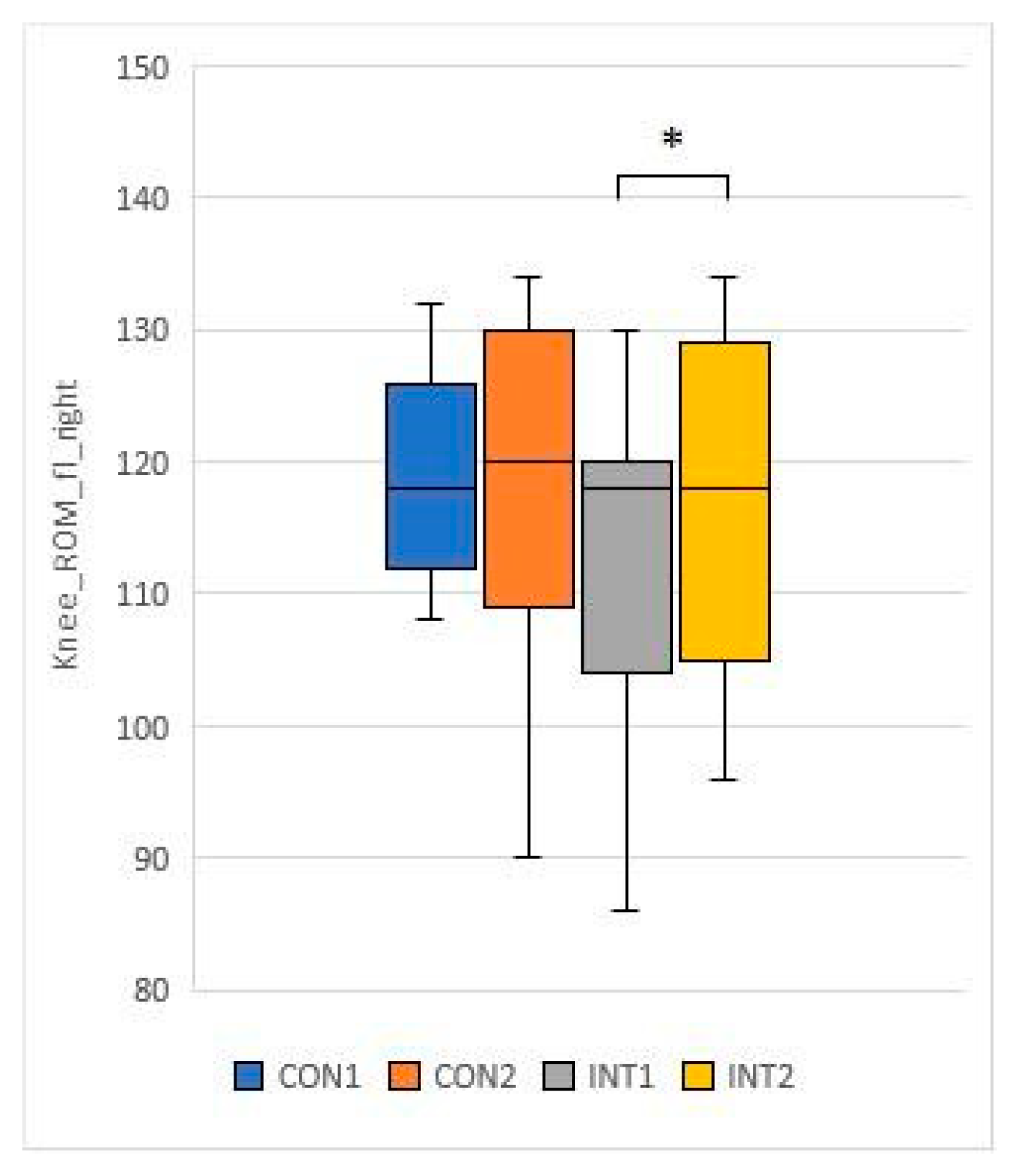
- Knee ROM fl R: mean decreased by 1.1 in the control group from 116.8 to 115.7, and decreased significantly by 6.3 in the experimental group (p=0.003) from 113.3 to 119.6.
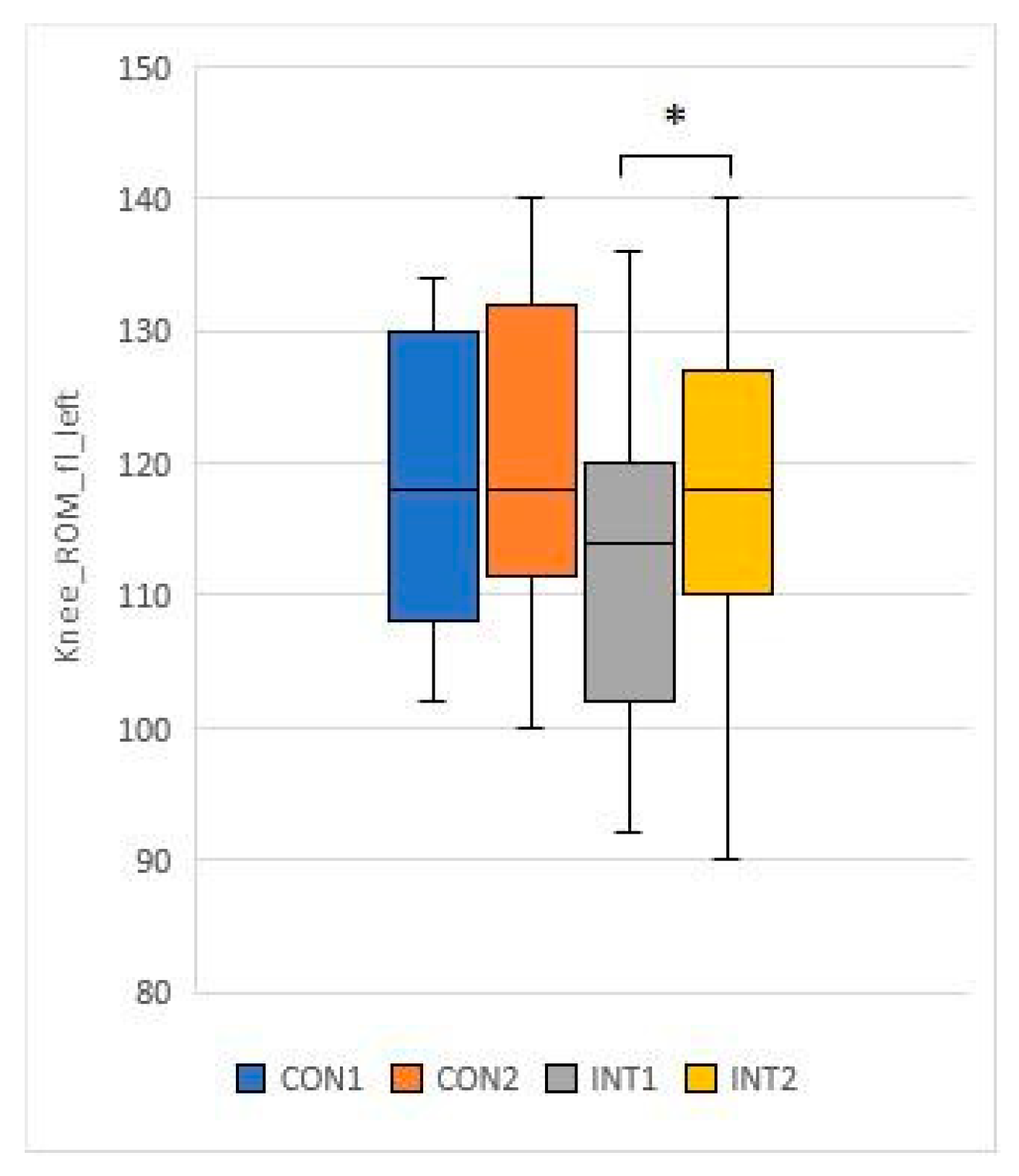
- Knee ROM fl L: mean increased by 1.9 in the control group from 115.5 to 117.4, in the experimental group it increased by 7.5, the change was significant (p=0.001) from 114 to 121.5
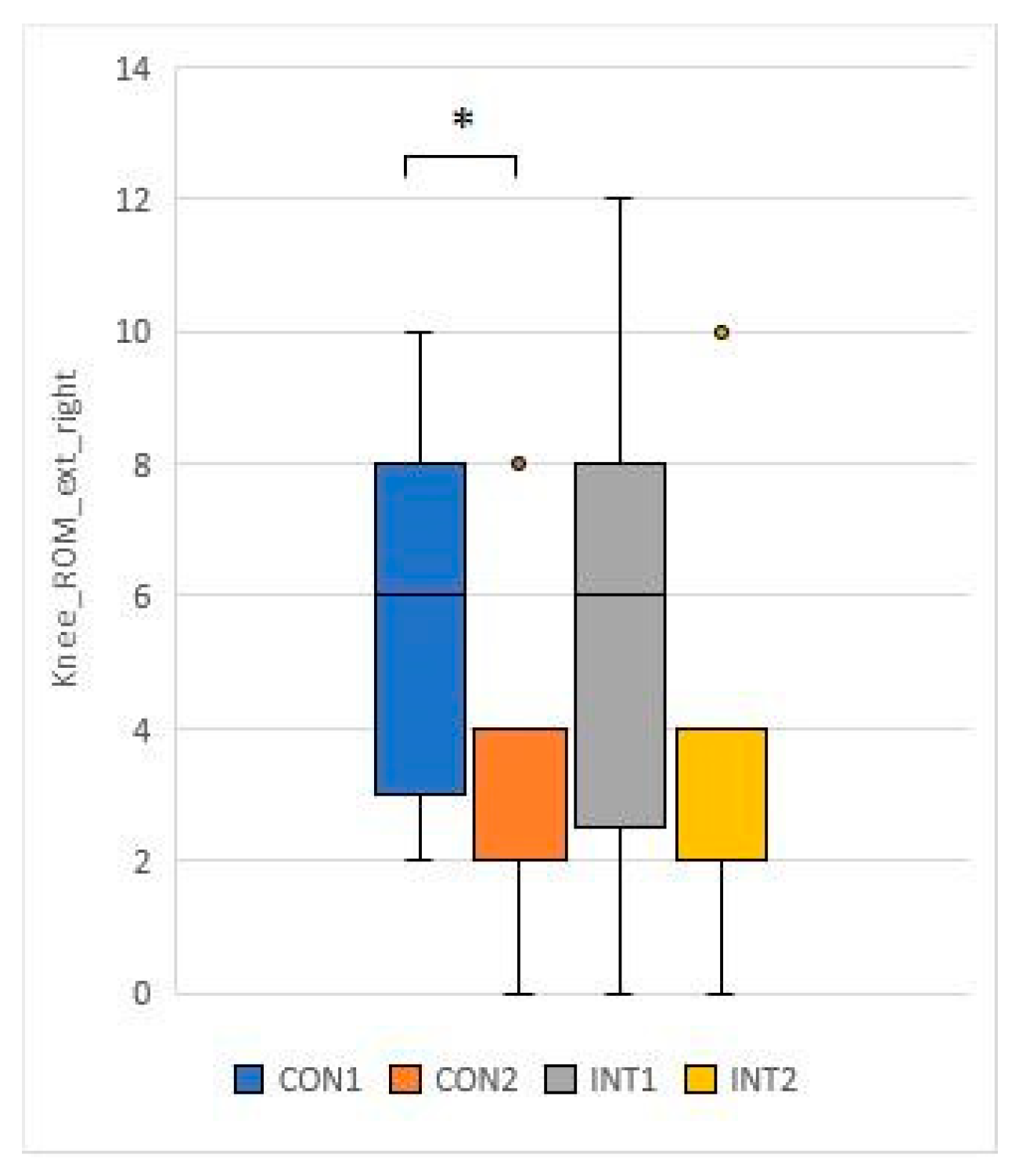
- Knee ROM ext R: mean decreased by 2.0 in the control group from 5.3 to 33.0, and decreased by 3.4 in the experimental group with significant magnitude (p=0.016) from 5.9 to 2.5.
- Mean hip ROM fl R: increased by 6.3 from 109.8 to 116.1 (p=0.018) in the control group and by 10.3 from 106.4 to 116.7 (p=0.000) in the experimental group.
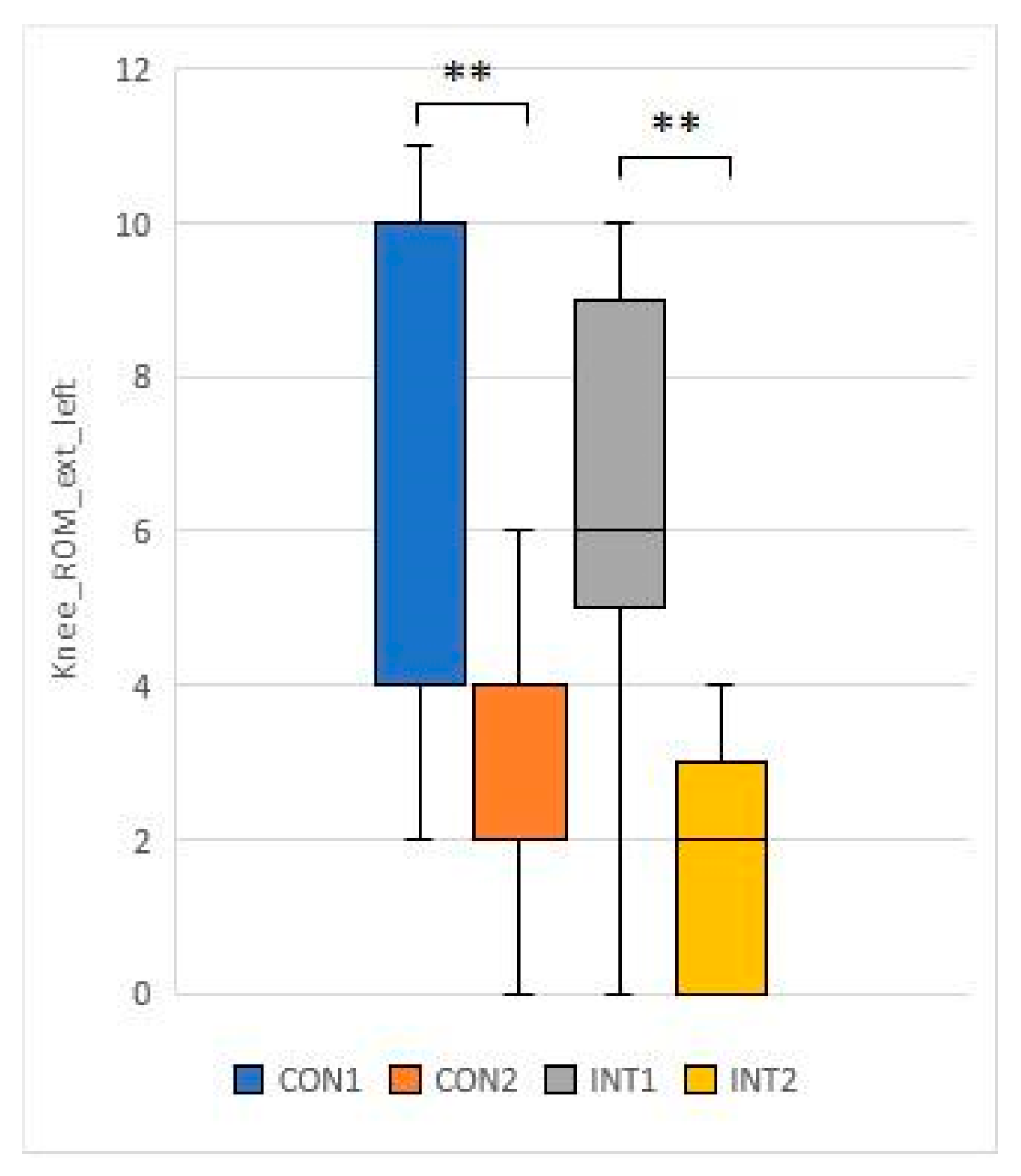
- Knee ROM ext L: mean decreased by 4.2 from 6.2 to 2.5 in the control group (p=0.000), while it decreased by 4.6 from 6.2 to 1.6 in the experimental group (p=0.000).
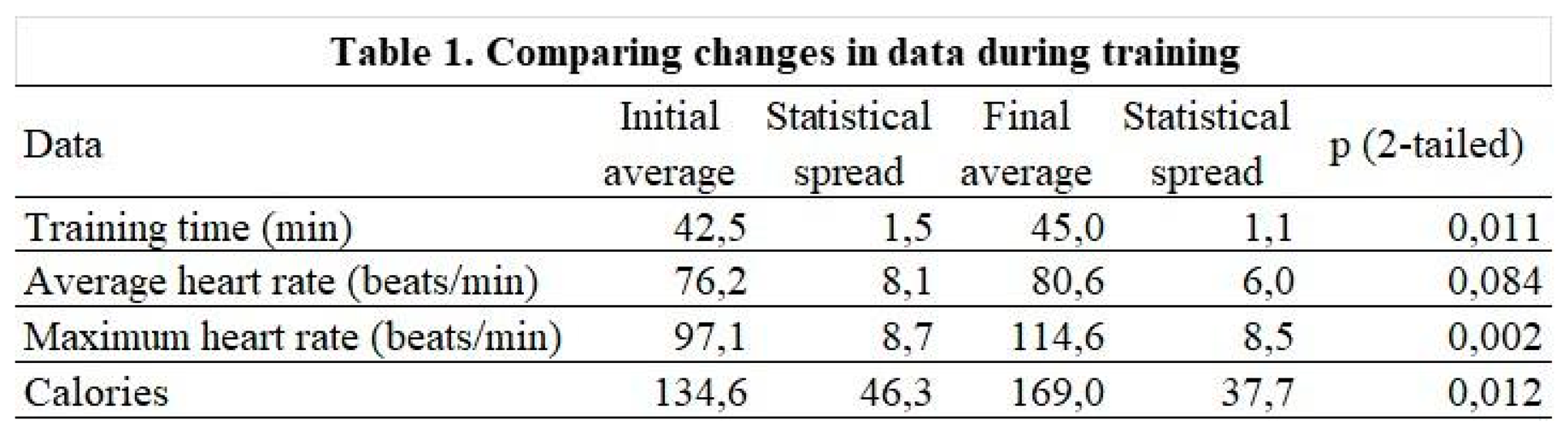
3.4. Results of the parameters measured during the physiotherapy session
4. Discussion
5. Conclusions
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Competing Interests
Consent for Publication
References
- Fritz, P.; Mayer, L.; Pető, A.; Németh, B. A kollagén szerepe és hatása az emberi szervezetre = The role and effect of collagen on the human body. Recreation 2020, 10, 8–11. ISSN 2064-4981. [CrossRef]
- Ricard-Blum, S. The collagen family. Cold Spring Harbor perspectives in biology 2011, 3. [Google Scholar] [CrossRef] [PubMed]
- León-López, A.; Morales-Peñaloza, A.; Martínez-Juárez, V. M.; Vargas-Torres, A.; Zeugolis, D. I.; Aguirre-Álvarez, G. Hydrolyzed Collagen-Sources and Applications. Molecules (Basel, Switzerland) 2019, 24, 4031. [Google Scholar] [CrossRef] [PubMed]
- Line, S.; Rhodes, C.; Yamada, Y. Molecular Biology of Cartilage Matrix. In Cellular and Molecular Biology of Bone.; Noda, M.; Academic press, Cambridge, Massachusetts, USA, 1993; pp. 539–555. ISBN: 9780080925004. 9, 9780. [Google Scholar]
- Farage, M. A.; Miller, K. W.; Elsner, P.; Maibach, H. I. Characteristics of the Aging Skin. Advances in wound care 2013, 2, 5–10. [Google Scholar] [CrossRef]
- Kirmse, M.; Oertzen-Hagemann, V.; de Marées, M.; Bloch, W.; Platen, P. Prolonged Collagen Peptide Supplementation and Resistance Exercise Training Affects Body Composition in Recreationally Active Men. Nutrients 2019, 11, 1154. [Google Scholar] [CrossRef]
- Zdzieblik, D.; Oesser, S.; Baumstark, M. W.; Gollhofer, A.; König, D. Collagen peptide supplementation in combination with resistance training improves body composition and increases muscle strength in elderly sarcopenic men: a randomised controlled trial. The British journal of nutrition 2015, 114, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Lodish, H.; Berk, A.; Matsudaira, P.; Kaiser, C.A.; Krieger, M.; Scott, M.P.; Zipursky, L.; Darnell, J. Molecular Cell Biology, 5th ed.; W. H. Freeman, New York, USA, 2005; 15. ISBN: 9780716743668.
- Lugo, J. P.; Saiyed, Z. M.; Lane, N. E. Efficacy and tolerability of an undenatured type II collagen supplement in modulating knee osteoarthritis symptoms: a multicenter randomized, double-blind, placebo-controlled study. Nutritional journal 2016, 15, 14. [Google Scholar] [CrossRef]
- Tariq, M.; Al Moutaery, A. R. Studies on the antisecretory, gastric anti-ulcer and cytoprotective properties of glycine. Research communications in molecular pathology and pharmacology, 1997; 97, 185–198. [Google Scholar] [PubMed]
- Graham, M. F.; Drucker, D. E.; Diegelmann, R. F.; Elson, C. O. Collagen synthesis by human intestinal smooth muscle cells in culture. Gastroenterology 1987, 92, 400–405. [Google Scholar] [CrossRef]
- Koutroubakis, I. E.; Petinaki, E.; Dimoulios, P.; Vardas, E.; Roussomoustakaki, M.; Maniatis, A. N.; Kouroumalis, E. A. Serum laminin and collagen IV in inflammatory bowel disease. Journal of clinical pathology 2003, 56, 817–820. [Google Scholar] [CrossRef]
- Tomosugi, N.; Yamamoto, S.; Takeuchi, M.; Yonekura, H.; Ishigaki, Y.; Numata, N.; Katsuda, S.; Sakai, Y. Effect of Collagen Tripeptide on Atherosclerosis in Healthy Humans. Journal of atherosclerosis and thrombosis 2017, 24, 530–538. [Google Scholar] [CrossRef]
- Saiga-Egusa, A.; Iwai, K.; Hayakawa, T.; Takahata, Y.; Morimatsu, F. Antihypertensive effects and endothelial progenitor cell activation by intake of chicken collagen hydrolysate in pre- and mild-hypertension. Bioscience, biotechnology, and biochemistry 2009, 73, 422–424. [Google Scholar] [CrossRef]
- Igase, M.; Kohara, K.; Okada, Y.; Ochi, M.; Igase, K.; Inoue, N.; Kutsuna, T.; Miura, H.; Ohyagi, Y. A double-blind, placebo-controlled, randomised clinical study of the effect of pork collagen peptide supplementation on atherosclerosis in healthy older individuals. Bioscience, biotechnology, and biochemistry 2018, 82, 893–895. [Google Scholar] [CrossRef]
- Choi, F.D.; Sung, C.T.; Juhasz, M.; Mesinkovsk, N.A. Oral Collagen Supplementation: A Systematic Review of Dermatological Applications. Journal of drugs in dermatology: JDD 2019, 18, 9–16. [Google Scholar] [PubMed]
- Collagen Market Size, Share & Trends Analysis Report By Source (Bovine, Porcine), By Product (Gelatin), By Application (Food & Beverages, Healthcare), By Region, And Segment Forecasts 2020-2027. Available online: https://www.grandviewresearch.com/industry-analysis/collagen-market (accessed on 24 October 2020).
- Range of Motion. Available online: https://www.physio-pedia.com/Range_of_Motion (accessed on 19 May 2023).
- Debasree Banerjee, Jane Kamuren, Grayson L. Baird, Amy Palmisciano, Ipsita Krishnan, Mary Whittenhall, James R. Klinger,and Corey E. Ventetuolo, The Modified Borg Dyspnea Scale does not predict hospitalization in pulmonary arterial hypertension, Published online 2017 Mar 16. [CrossRef]
- Dixon, A. The United Nations Decade of Healthy Ageing requires concerted global action. Nature Aging 2021, 1, 2. [Google Scholar] [CrossRef]
- Enomoto M, Adachi H, Hirai Y, Fukami A, Satoh A, Otsuka M, Kumagae S, Nanjo Y, Yoshikawa K, Esaki E, Kumagai E, Ogata K, Kasahara A, Tsukagawa E, Yokoi K, Ohbu-Murayama K, Imaizumi T: LDL-C/HDL-C ratio predicts carotid intima-media thickness progression better than HDL-C or LDL-C alone. J Lipids, 2012; 21: 153-159. 1.
- Haskell, W.L. The influence of exercise on the concentrations of triglyceride and cholesterol in human plasma. Exercise and Sport Sciences Reviews 1984, 12, 205–44. [Google Scholar] [CrossRef] [PubMed]
- Halverstadt, A.; Phares, D.A.; Wilund, K.R.; Goldberg, A.P.; Hagberg, J.M. Endurance exercise training raises high-density lipoprotein cholesterol and lowers small low-density lipoprotein and very low-density lipoprotein independent of body fat phenotypes in older men and women. Metabolism 2007, 56, 444–50. [Google Scholar] [CrossRef]
- Kokkinos, P.F.; Fernhall, B. Physical activity and high density lipoprotein cholesterol levels: what is the relationship? Sports Medicine 1999, 28, 307–14. [Google Scholar] [CrossRef]
- Fahlman, M.M.; Boardley, D.; Lambert, C.P.; Flynn, M.G. Effects of endurance training and resistance training on plasma lipoprotein profiles in elderly women. The Journals of Gerontology. Series A, Biological sciences and medical sciences 2002, 57, B54–B60. [Google Scholar] [CrossRef]
- Nielsen, F.H.; Lukaski, H.C. Update on the relationship between magnesium and exercise. Magnes Res 2006, 19, 180–189. [Google Scholar]
- Hadi, A.; Arab, A.; Moradi, S.; Pantovic, A.; Clark, C. C. T.; Ghaedi, E. The effect of l-arginine supplementation on lipid profile: a systematic review and meta-analysis of randomised controlled trials. The British journal of nutrition 2019, 122, 1021–1032. [Google Scholar] [CrossRef]
- Kujawska-Łuczak, M.; Suliburska, J.; Markuszewski, L.; Pupek-Musialik, D.; Jabłecka, A.; Bogdański, P. The effect of L-arginine and ascorbic acid on the visceral fat and the concentrations of metalloproteinases 2 and 9 in high-fat-diet rats. Endokrynologia Polska 2015, 66, 526–532. [Google Scholar] [CrossRef]
- Bogdański, P.; Suliburska, J.; Szulińska, M.; Sikora, M.; Walkowiak, J.; Jakubowski, H. L-Arginine and vitamin C attenuate pro-atherogenic effects of high-fat diet on biomarkers of endothelial dysfunction in rats. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie 2015, 76, 100–106. [Google Scholar] [CrossRef]
- Gad, M.Z. Anti-aging effects of l-arginine. Journal of Advanced Research 2010, 1, 169–177. [Google Scholar] [CrossRef]
- Park, B. G.; Park, Y. S.; Park, J. W.; Shin, E.; Shin, W. S. Anti-obesity potential of enzymatic fragments of hyaluronan on high-fat diet-induced obesity in C57BL/6 mice. Biochemical and biophysical research communications 2016, 473, 290–295. [Google Scholar] [CrossRef]
- Ross, R.; Neeland, I. J.; Yamashita, S.; Shai, I.; Seidell, J.; Magni, P.; Santos, R. D.; Arsenault, B.; Cuevas, A.; Hu, F. B.; Griffin, B. A.; Zambon, A.; Barter, P.; Fruchart, J. C.; Eckel, R. H.; Matsuzawa, Y.; Després, J. P. Waist circumference as a vital sign in clinical practice: a Consensus Statement from the IAS and ICCR Working Group on Visceral Obesity. Nature reviews. Endocrinology 2020, 16, 177–189. [Google Scholar] [CrossRef]
- Zdzieblik, D.; Oesser, S.; Gollhofer, A.; Koenig, D. Corrigendum: Improvement of activity-related knee joint discomfort following supplementation of specific collagen peptides. Applied physiology, nutrition, and metabolism = Physiologie appliquee, nutrition et metabolisme 2017, 42, 1237. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



