Submitted:
20 April 2023
Posted:
21 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
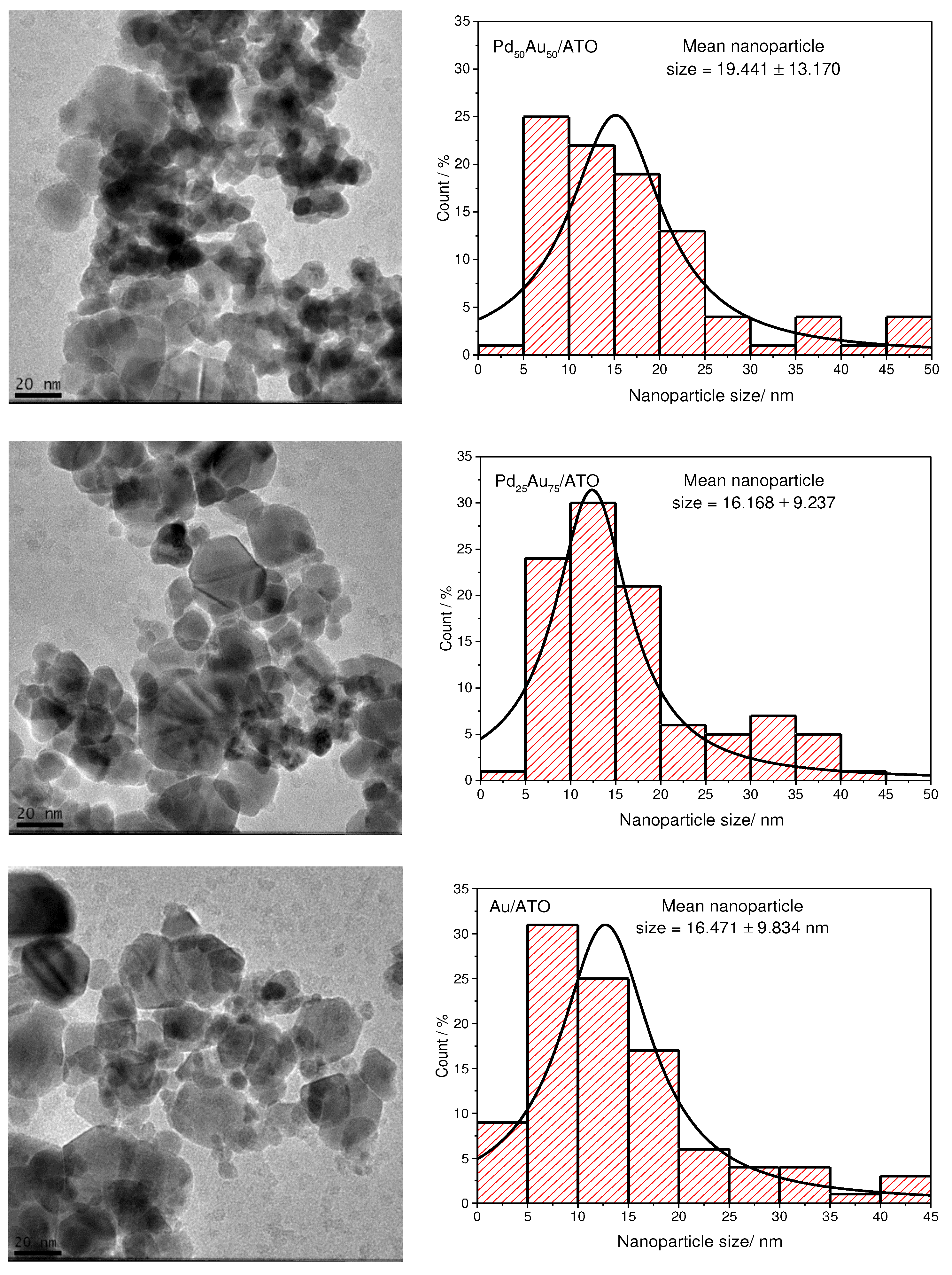
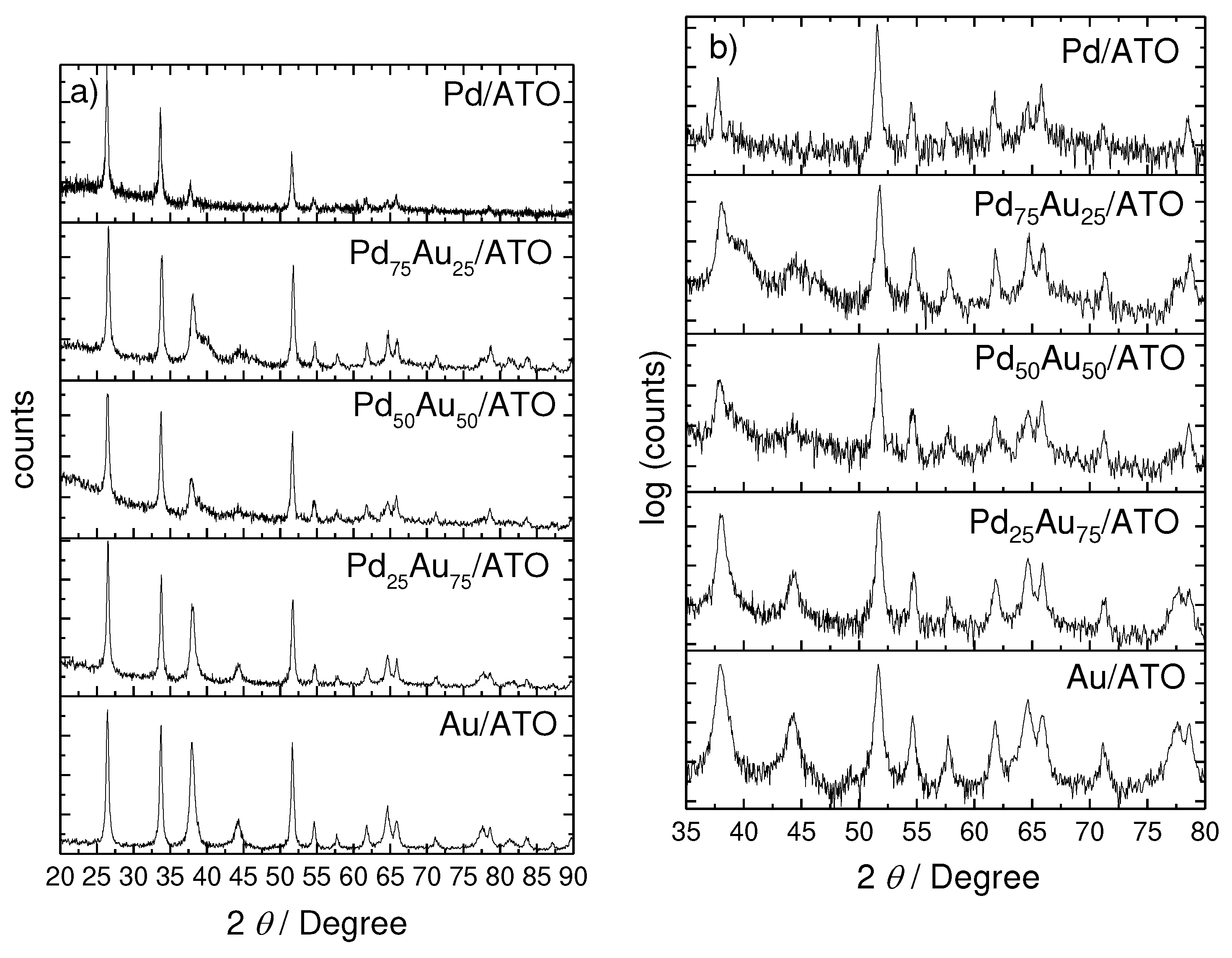
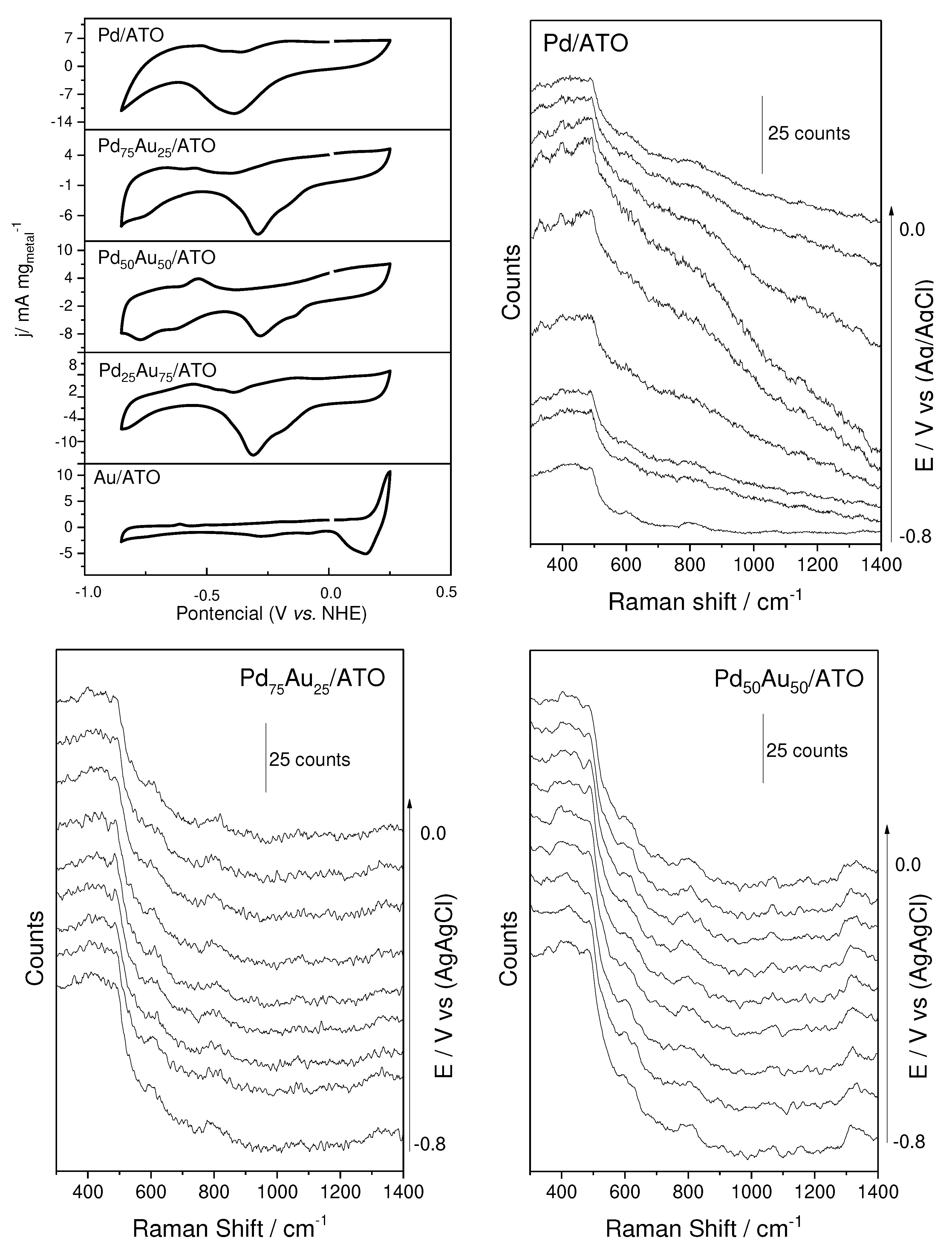
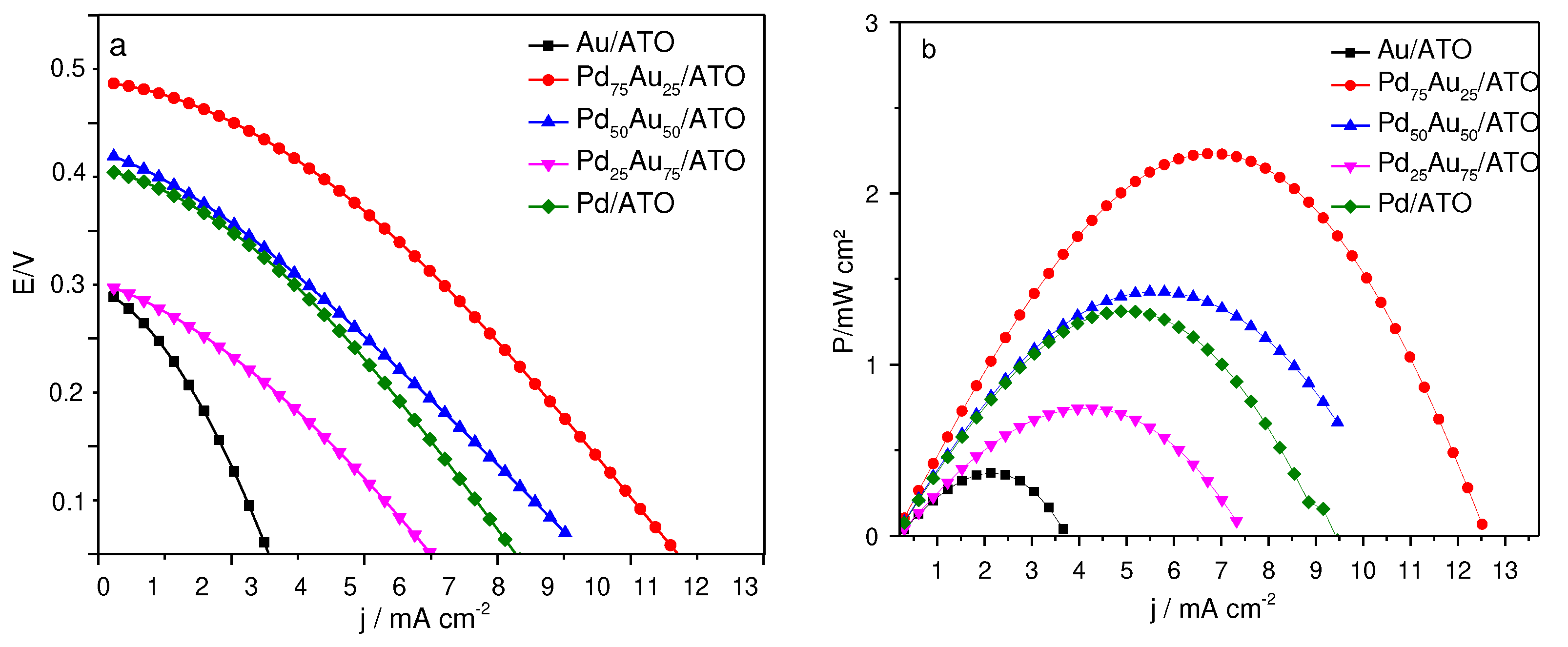
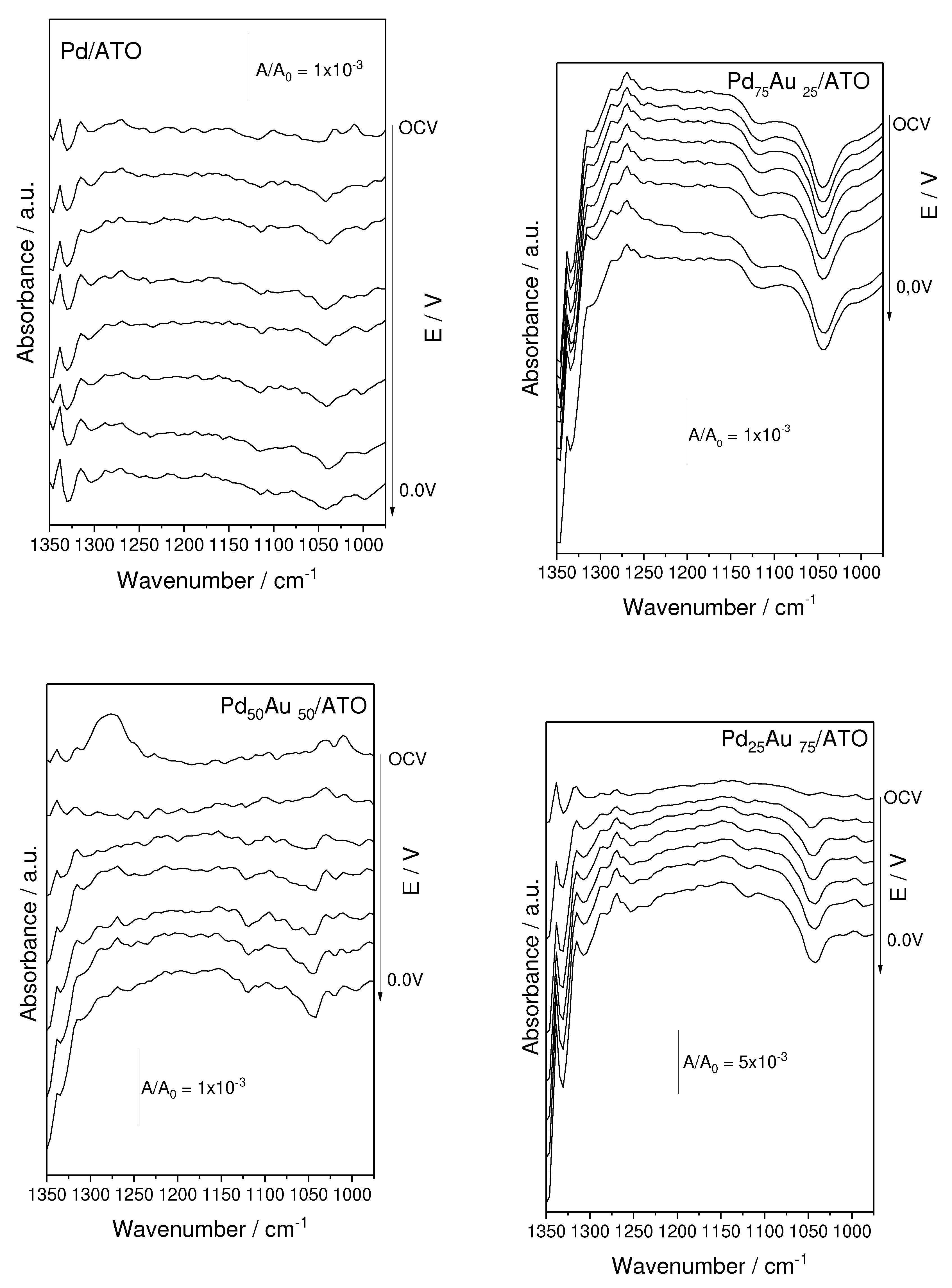
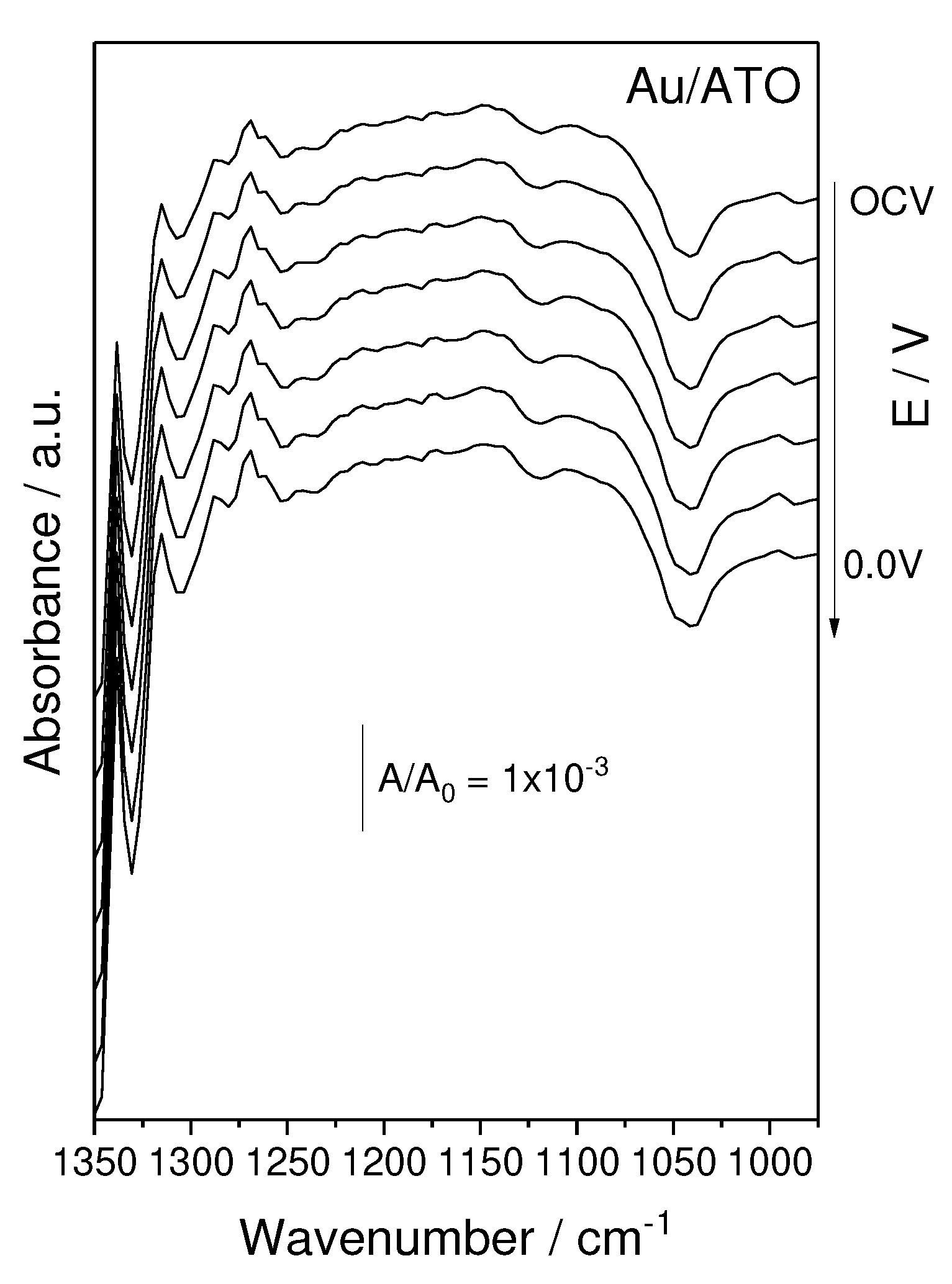
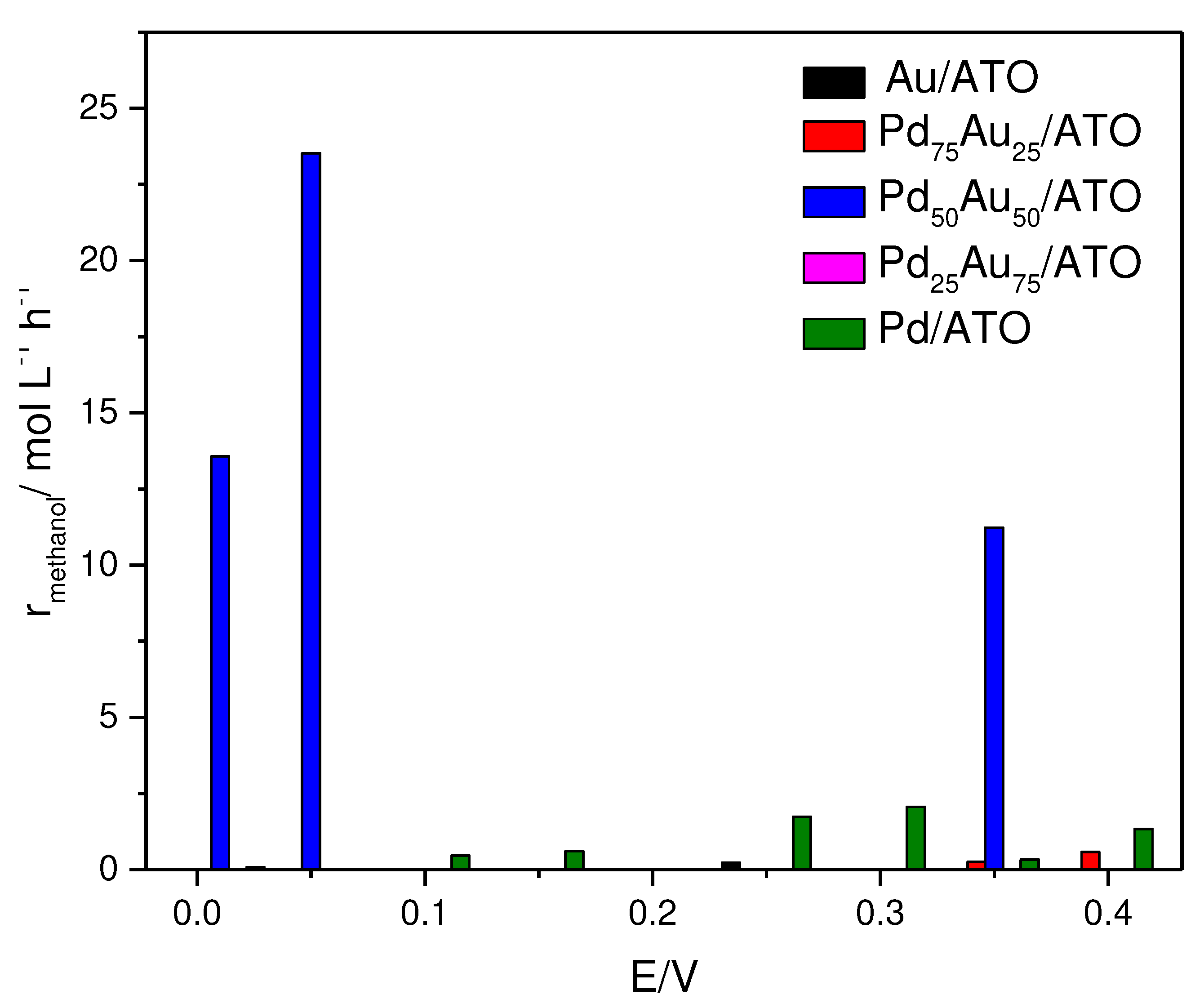
2. Results
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- H. Blanco, W. Nijs, J. Ruf, A. Faaij, Potential of Power-to-Methane in the EU energy transition to a low carbon system using cost optimization, Appl. Energy, 232 (2018) 323-340. [CrossRef]
- J. Jang, K. Shen, C.G. Morales-Guio, Electrochemical Direct Partial Oxidation of Methane to Methanol, Joule, 3 (2019) 2589-2593. [CrossRef]
- Premachandra, D.; Heagy, M.D. Morphology-Controlled WO3 for the Photocatalytic Oxidation of Methane to Methanol in Mild Conditions. Methane (2023) 2, 103-112. [CrossRef]
- R.F.B. de Souza, D.Z. Florio, E. Antolini, A.O. Neto, Partial Methane Oxidation in Fuel Cell-Type Reactors for Co-Generation of Energy and Chemicals: A Short Review, Catalysts, 12 (2022) 217. [CrossRef]
- T. Ikuno, J. Zheng, A. Vjunov, M. Sanchez-Sanchez, M.A. Ortuño, D.R. Pahls, J.L. Fulton, D.M. Camaioni, Z. Li, D. Ray, B.L. Mehdi, N.D. Browning, O.K. Farha, J.T. Hupp, C.J. Cramer, L. Gagliardi, J.A. Lercher, Methane Oxidation to Methanol Catalyzed by Cu-Oxo Clusters Stabilized in NU-1000 Metal–Organic Framework, J. Am. Chem. Soc., 139 (2017) 10294-10301. [CrossRef]
- V.L. Sushkevich, J.A. van Bokhoven, Kinetic study and effect of water on methane oxidation to methanol over copper-exchanged mordenite, Catal. Sci. Technol., 10 (2020) 382-390. [CrossRef]
- H. Dalton, The Leeuwenhoek Lecture 2000 The natural and unnatural history of methane-oxidizing bacteria, Philos. Trans. R. Soc. Lond., B, Biol. Sci. PHILOS T R SOC B, 360 (2005) 1207-1222. [CrossRef]
- K. Otsuka, I. Yamanaka, Electrochemical cells as reactors for selective oxygenation of hydrocarbons at low temperature, Catal. Today, 41 (1998) 311-325. [CrossRef]
- B. Lee, T. Hibino, Efficient and selective formation of methanol from methane in a fuel cell-type reactor, J. Catal., 279 (2011) 233-240. [CrossRef]
- A. Tomita, J. Nakajima, T. Hibino, Direct Oxidation of Methane to Methanol at Low Temperature and Pressure in an Electrochemical Fuel Cell, Angew. Chem., Int. Ed., 47 (2008) 1462-1464. [CrossRef]
- M.C.L. Santos, L.C. Nunes, L.M.G. Silva, A.S. Ramos, F.C. Fonseca, R.F.B. de Souza, A.O. Neto, Direct Alkaline Anion Exchange Membrane Fuel Cell to Converting Methane into Methanol, ChemistrySelect, 4 (2019) 11430-11434. [CrossRef]
- A.S. Ramos, M.C.L. Santos, C.M. Godoi, A. Oliveira Neto, R. Fernando B. De Souza, Obtaining C2 and C3 Products from Methane Using Pd/C as Anode in a Solid Fuel Cell-type Electrolyte Reactor, ChemCatChem, 12 (2020) 4517-4521. [CrossRef]
- H. Jiang, L. Zhang, Z. Han, Y. Tang, Y. Sun, P. Wan, Y. Chen, M.D. Argyle, M. Fan, Direct conversion of methane to methanol by electrochemical methods, GEE, (2021). [CrossRef]
- R.L. Cook, A.F. Sammells, Ambient Temperature Methane Activation to Condensed Species under Cathodic Conditions, J. Electrochem. Soc., 137 (1990) 2007-2008. [CrossRef]
- L.M.S. Garcia, S. Rajak, K. Chair, C.M. Godoy, A.J. Silva, P.V.R. Gomes, E.A. Sanches, A.S. Ramos, R.F.B. De Souza, A. Duong, A.O. Neto, Conversion of Methane into Methanol Using the [6,6’-(2,2’-Bipyridine-6,6’-Diyl)bis(1,3,5-Triazine-2,4-Diamine)](Nitrato-O)Coppe r(II) Complex in a Solid Electrolyte Reactor Fuel Cell Type, ACS Omega, 5 (2020) 16003-16009. [CrossRef]
- A.S. Ramos, M.C.L. Santos, C.M. Godoi, A. Oliveira Neto, R.F.B. De Souza, Obtaining C2 and C3 Products from Methane Using Pd/C as Anode in a Solid Fuel Cell-type Electrolyte Reactor, ChemCatChem, 12 (2020) 4517-4521. [CrossRef]
- A. Antzara, E. Heracleous, L. Silvester, D.B. Bukur, A.A. Lemonidou, Activity study of NiO-based oxygen carriers in chemical looping steam methane reforming, Catal. Today, 272 (2016) 32-41. [CrossRef]
- S. Wang, T. Itoh, T. Fujimori, M.M. de Castro, A. Silvestre-Albero, F. Rodríguez-Reinoso, T. Ohba, H. Kanoh, M. Endo, K. Kaneko, Formation of COx-Free H2 and Cup-Stacked Carbon Nanotubes over Nano-Ni Dispersed Single Wall Carbon Nanohorns, Langmuir, 28 (2012) 7564-7571. [CrossRef]
- M. Jafarian, M.G. Mahjani, H. Heli, F. Gobal, M. Heydarpoor, Electrocatalytic oxidation of methane at nickel hydroxide modified nickel electrode in alkaline solution, Electroc. Comm., 5 (2003) 184-188. [CrossRef]
- S. Hsieh, K. Chen, Anodic oxidation of methane, Journal of the Electrochemical Society, 124 (1977) 1171. [CrossRef]
- Y. He, C. Luan, Y. Fang, X. Feng, X. Peng, G. Yang, N. Tsubaki, Low-temperature direct conversion of methane to methanol over carbon materials supported Pd-Au nanoparticles, Catal. Today, 339 (2020) 48-53. [CrossRef]
- B. Wang, L. Tao, Y. Cheng, F. Yang, Y. Jin, C. Zhou, H. Yu, Y. Yang, Electrocatalytic Oxidation of Small Molecule Alcohols over Pt, Pd, and Au Catalysts: The Effect of Alcohol’s Hydrogen Bond Donation Ability and Molecular Structure Properties, Catalysts, 9 (2019). [CrossRef]
- R. Serra-Maia, F.M. Michel, Y. Kang, E.A. Stach, Decomposition of Hydrogen Peroxide Catalyzed by AuPd Nanocatalysts during Methane Oxidation to Methanol, ACS Catal, (2020) 5115-5123. [CrossRef]
- Y. He, J. Liang, Y. Imai, K. Ueda, H. Li, X. guo, G. Yang, Y. Yoneyama, N. Tsubaki, Highly selective synthesis of methanol from methane over carbon materials supported Pd-Au nanoparticles under mild conditions, Catal. Today, 352 (2020) 104-110. [CrossRef]
- R. McVicker, N. Agarwal, S.J. Freakley, Q. He, S. Althahban, S.H. Taylor, C.J. Kiely, G.J. Hutchings, Low temperature selective oxidation of methane using gold-palladium colloids, Catal. Today, 342 (2020) 32-38. [CrossRef]
- L. Sun, Z. Liu, Y. Bao, H. Li, W. Bao, High-activity of Pd catalyst supported on antimony tin oxide for hydrogen peroxide electroreduction, IJMR, 105 (2014) 584-587. [CrossRef]
- F. de Moura Souza, R.F.B. de Souza, B.L. Batista, M.C. dos Santos, F.C. Fonseca, A.O. Neto, J. Nandenha, Methane activation at low temperature in an acidic electrolyte using PdAu/C, PdCu/C, and PdTiO2/C electrocatalysts for PEMFC, Res. Chem. Intermed., 46 (2020) 2481-2496. [CrossRef]
- R.M. Piasentin, E.V. Spinacé, M.M. Tusi, A.O. Neto, Preparation of PdPtSn/C-Sb2O5. SnO2 electrocatalysts by borohydride reduction for ethanol electro-oxidation in alkaline medium, Int J Electrochem Sci, 6 (2011) 2255-2263.
- J.F. Coelho, N.G.P. Filho, I.M. Gutierrez, C.M. Godoi, P.V.R. Gomes, P.J. Zambiazi, R.F.B. de Souza, A.O. Neto, Methane-to-methanol conversion and power co-generation on palladium: nickel supported on antimony tin oxide catalysts in a polymeric electrolyte reactor-fuel cell (PER-FC), Res. Chem. Intermed., 48 (2022) 5155-5168. [CrossRef]
- C.M. Godoi, M.C.L. Santos, A.J. Silva, T.L. Tagomori, A.S. Ramos, R.F.B. de Souza, A.O. Neto, Methane conversion to higher value-added product and energy co-generation using anodes OF PdCu/C in a solid electrolyte reactor: alkaline fuel cell type monitored by differential mass spectroscopy, Res. Chem. Intermed., 47 (2021) 743-757. [CrossRef]
- R.M. Antoniassi, L. Otubo, J.M. Vaz, A. Oliveira Neto, E.V. Spinacé, Synthesis of Pt nanoparticles with preferential (100) orientation directly on the carbon support for Direct Ethanol Fuel Cell, J. Catal., 342 (2016) 67-74. [CrossRef]
- V.A. Ribeiro, O.V. Correa, A.O. Neto, M. Linardi, E.V. Spinacé, Preparation of PtRuNi/C electrocatalysts by an alcohol-reduction process for electro-oxidation of methanol, Appl. Catal., 372 (2010) 162-166. [CrossRef]
- G. Cognard, G. Ozouf, C. Beauger, G. Berthomé, D. Riassetto, L. Dubau, R. Chattot, M. Chatenet, F. Maillard, Benefits and limitations of Pt nanoparticles supported on highly porous antimony-doped tin dioxide aerogel as alternative cathode material for proton-exchange membrane fuel cells, Appl. Catal. B, 201 (2017) 381-390. [CrossRef]
- S. Bagheri, N. Muhd Julkapli, S. Bee Abd Hamid, Titanium Dioxide as a Catalyst Support in Heterogeneous Catalysis, Sci. World J., 2014 (2014) 727496. [CrossRef]
- W. Qu, Z. Wang, X. Sui, D. Gu, An efficient antimony doped tin oxide and carbon nanotubes hybrid support of Pd catalyst for formic acid electrooxidation, Int. J. Hydrog. Energy, 39 (2014) 5678-5688. [CrossRef]
- C.M. Godoi, I.M. Gutierrez, P.V.R. Gomes, J.F. Coelho, P.J. Zambiazi, L. Otubo, A.O. Neto, R.F.B. De Souza, Production of Methanol on PdCu A. T. O. in a Polymeric Electrolyte Reactor of the Fuel Cell Type from Methane, Methane, 2022, pp. 218-230. [CrossRef]
- J. Nandenha, R.F.B. De Souza, M.H.M.T. Assumpção, E.V. Spinacé, A.O. Neto, Preparation of PdAu/C-Sb2O5·SnO2 electrocatalysts by borohydride reduction process for direct formic acid fuel cell, Ionics, 19 (2013) 1207-1213. [CrossRef]
- F.H. Aragón, J.A.H. Coaquira, P. Hidalgo, S.W. da Silva, S.L.M. Brito, D. Gouvêa, P.C. Morais, Evidences of the evolution from solid solution to surface segregation in Ni-doped SnO2 nanoparticles using Raman spectroscopy, J Raman Spectrosc, 42 (2011) 1081-1086. [CrossRef]
- A. Leonardy, W.-Z. Hung, D.-S. Tsai, C.-C. Chou, Y.-S. Huang, Structural Features of SnO2 Nanowires and Raman Spectroscopy Analysis, Cryst. Growth Des., 9 (2009) 3958-3963. [CrossRef]
- M. Muniz-Miranda, A. Zoppi, F. Muniz-Miranda, N. Calisi, Palladium Oxide Nanoparticles: Preparation, Characterization and Catalytic Activity Evaluation, Coatings, 10 (2020) 207. [CrossRef]
- B.S. Yeo, S.L. Klaus, P.N. Ross, R.A. Mathies, A.T. Bell, Identification of Hydroperoxy Species as Reaction Intermediates in the Electrochemical Evolution of Oxygen on Gold, ChemPhysChem, 11 (2010) 1854-1857. [CrossRef]
- B.-x. Yan, Y.-y. Zhu, Y. Wei, H. Pei, Study on surface enhanced Raman scattering of Au and Au@Al2O3 spherical dimers based on 3D finite element method, Sci. Rep., 11 (2021) 8391. 10.1038/s41598-021-87997-z.
- R.F.B. De Souza, É.T. Neto, M.L. Calegaro, E.A. Santos, H.S. Martinho, M.C. dos Santos, Ethanol Electro-oxidation on Pt/C Electrocatalysts: An “In Situ” Raman Spectroelectrochemical Study, Electrocatalysis, 2 (2011) 28-34. 10.1007/s12678-010-0031-0.
- J. Nandenha, E.H. Fontes, R.M. Piasentin, F.C. Fonseca, A.O. Neto, Direct oxidation of methane at low temperature using Pt/C, Pd/C, Pt/C-ATO and Pd/C-ATO electrocatalysts prepared by sodium borohydride reduction process, J. Fuel Chem. Technol., 46 (2018) 1137-1145. [CrossRef]
- D. Scarano, S. Bertarione, G. Spoto, A. Zecchina, C. Otero Areán, FTIR spectroscopy of hydrogen, carbon monoxide, and methane adsorbed and co-adsorbed on zinc oxide, Thin Solid Films, 400 (2001) 50-55. [CrossRef]
- K. Hamada, H. Morishita, The Rotation-Vibrational Spectra and Structures of Methanol and Acetonitrile, Spectrosc. Lett., 13 (1980) 15-29. [CrossRef]
- P.A. Christensen, D. Linares-Moya, The Role of Adsorbed Formate and Oxygen in the Oxidation of Methanol at a Polycrystalline Pt Electrode in 0.1 M KOH: An In Situ Fourier Transform Infrared Study, J. Phys. Chem. C, 114 (2010) 1094-1101. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



