Submitted:
06 March 2023
Posted:
07 March 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental procedure
2.1. Sample preparation
2.2. Compressive strength
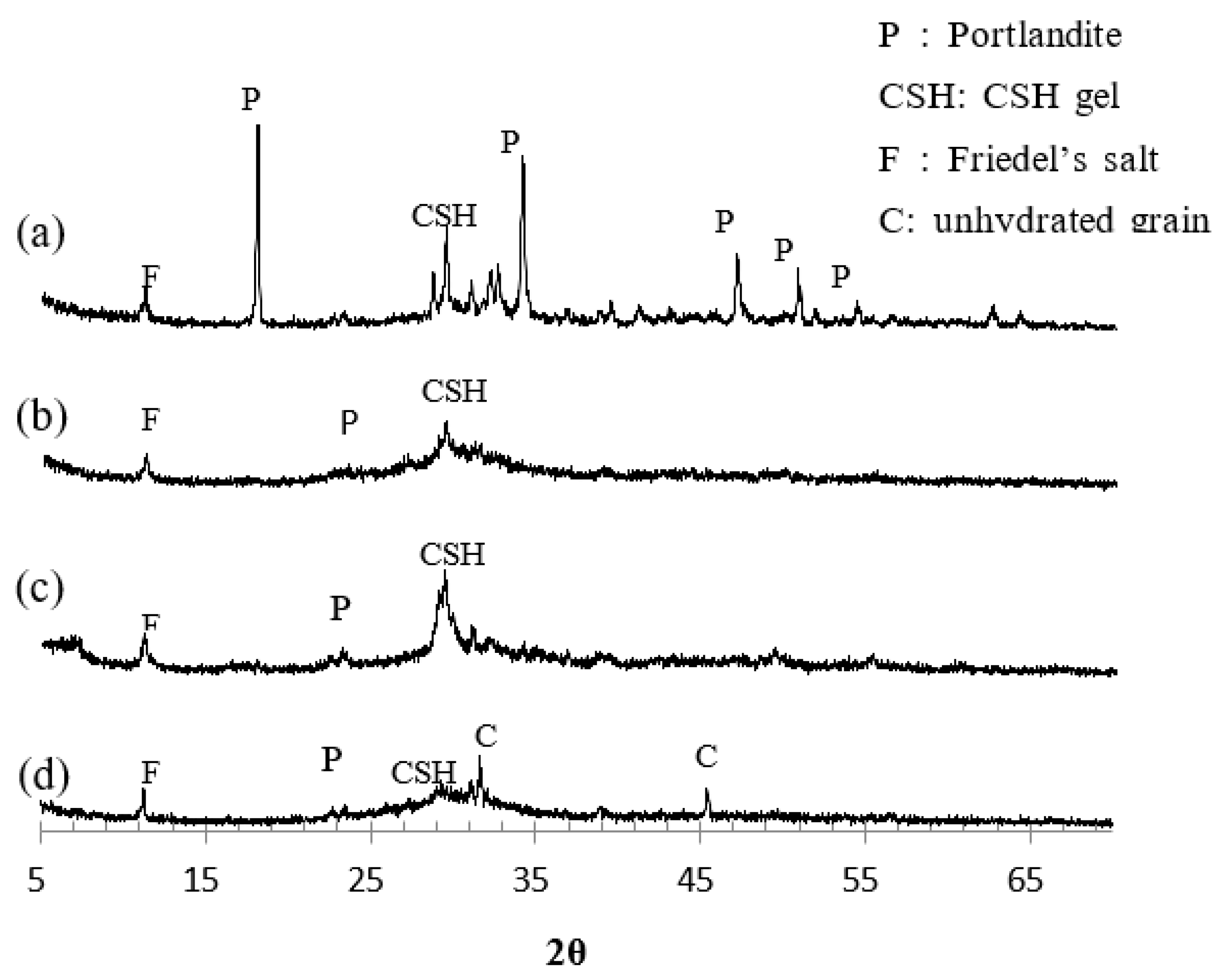
2.3. Hydration characteristic
2.4. Resistance to chloride penetration
2.5. Electrochemical chloride removal
3. Results
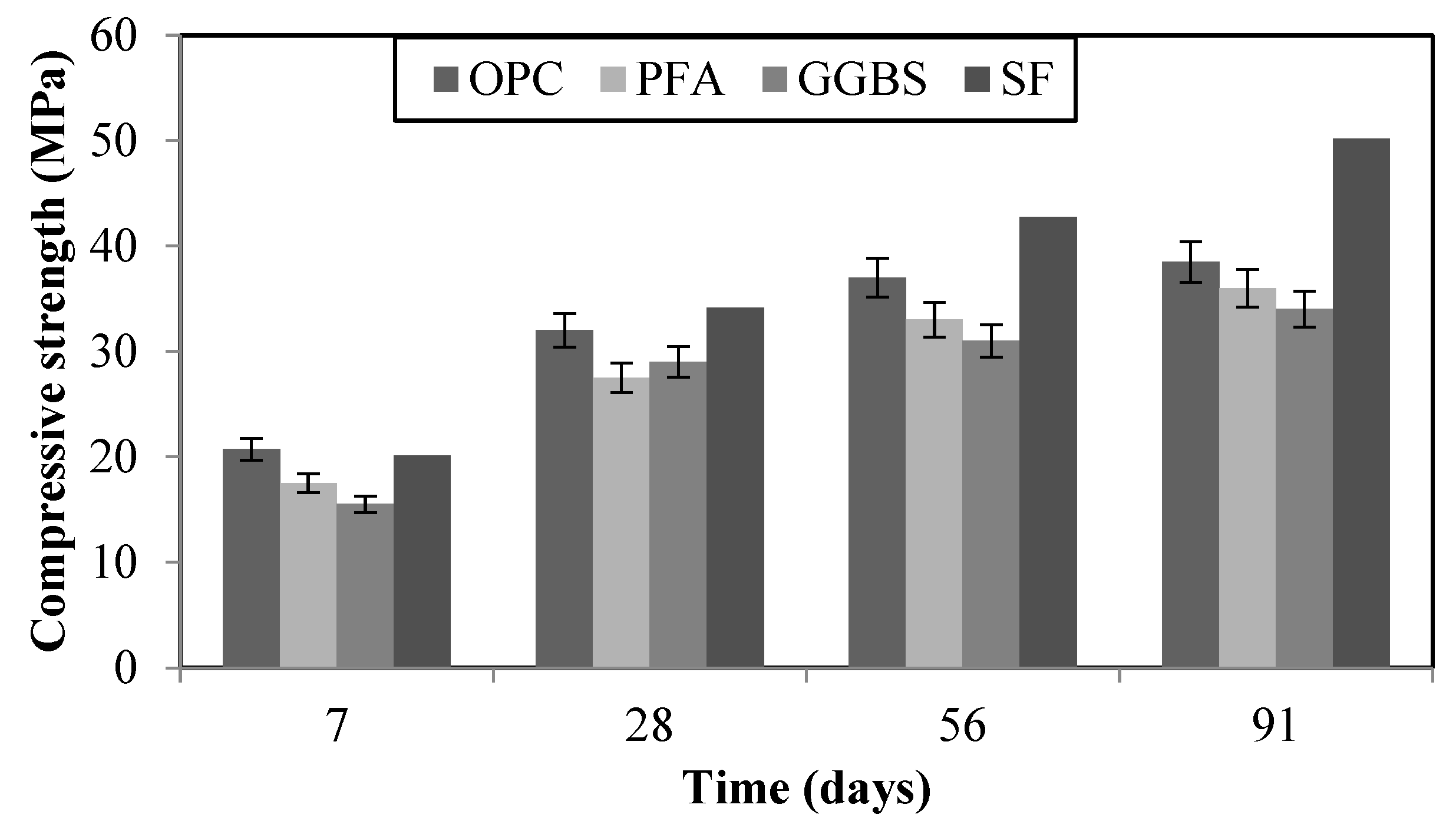
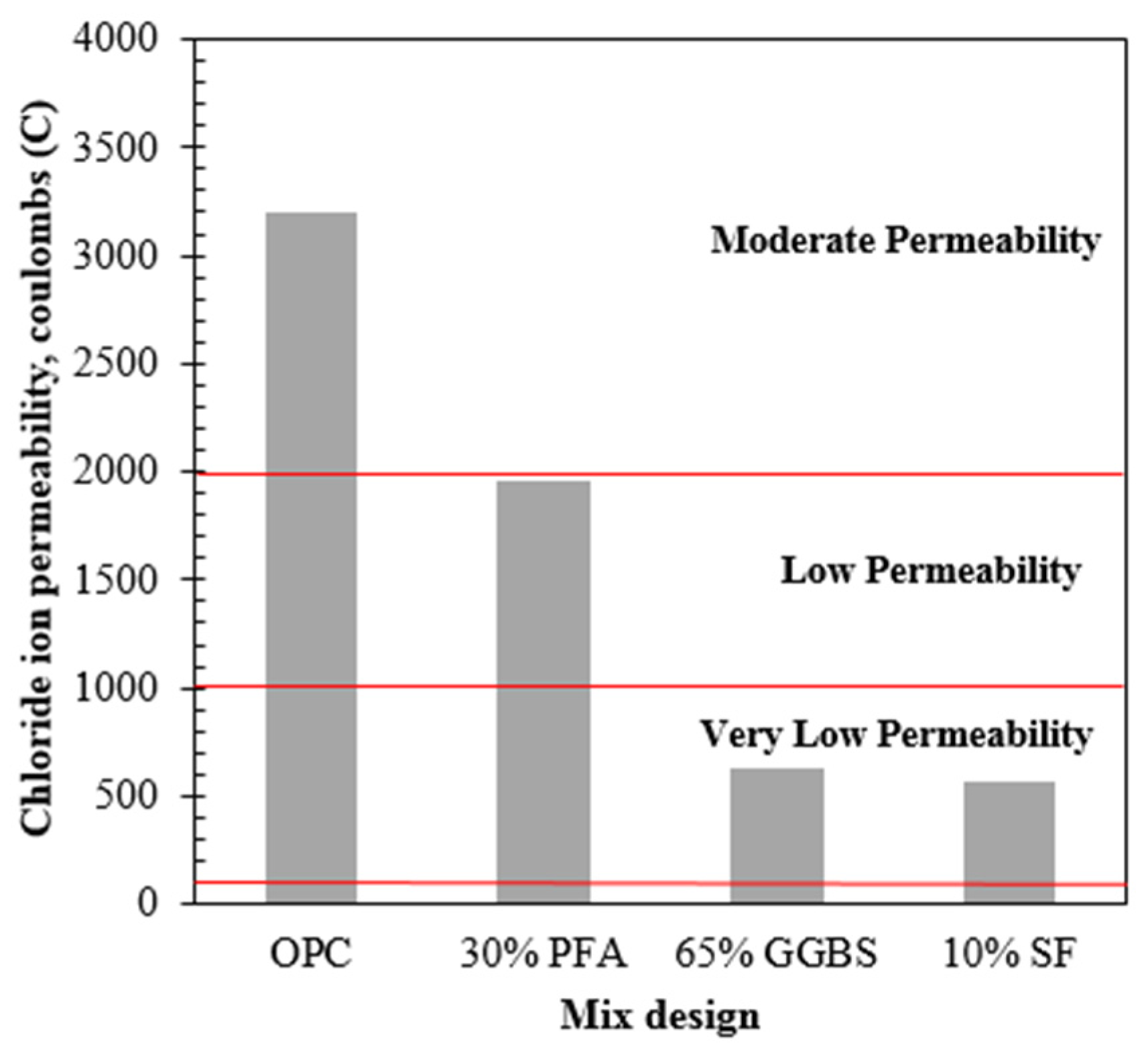
3.1. Development of strength and permeability
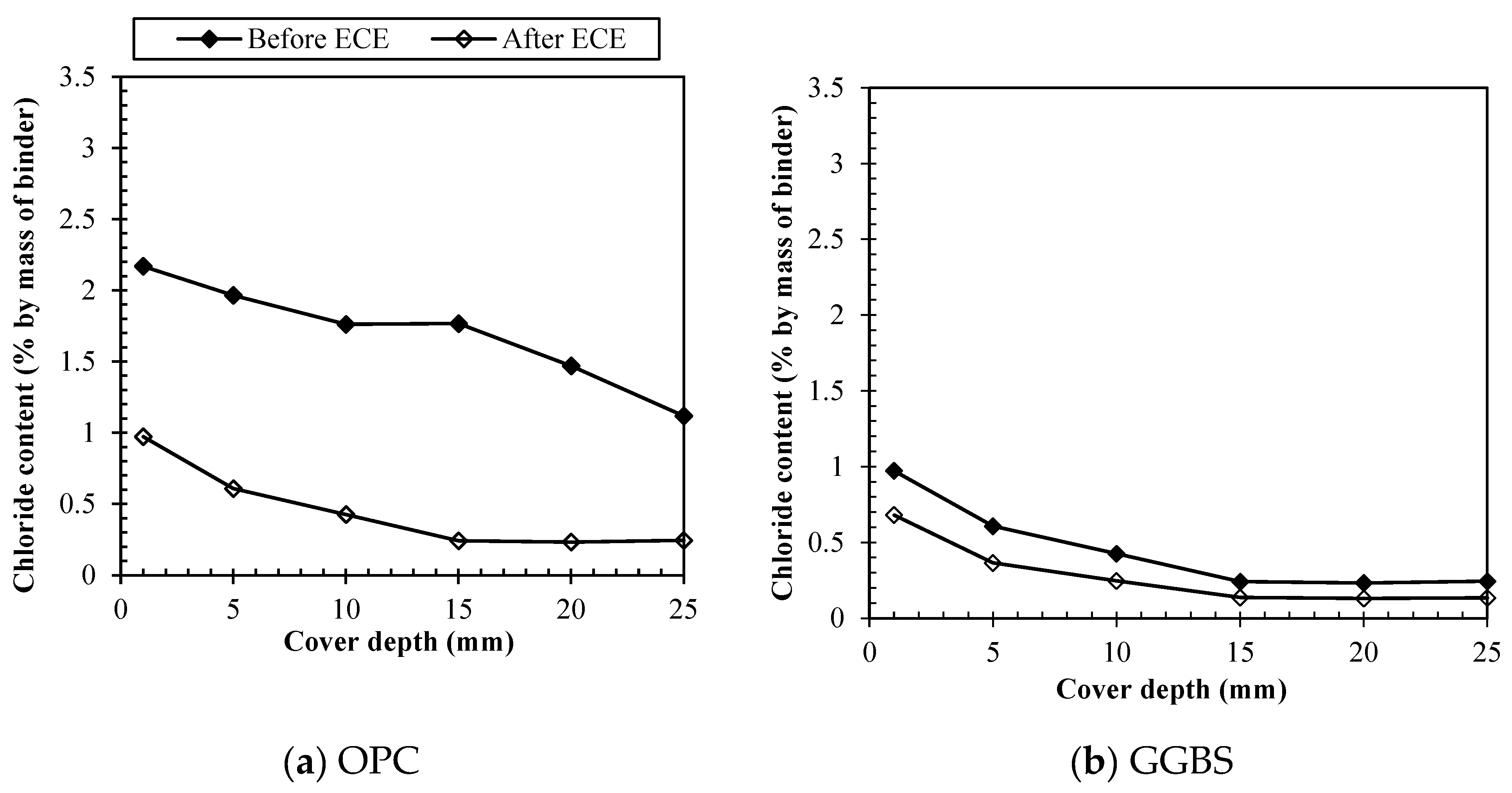
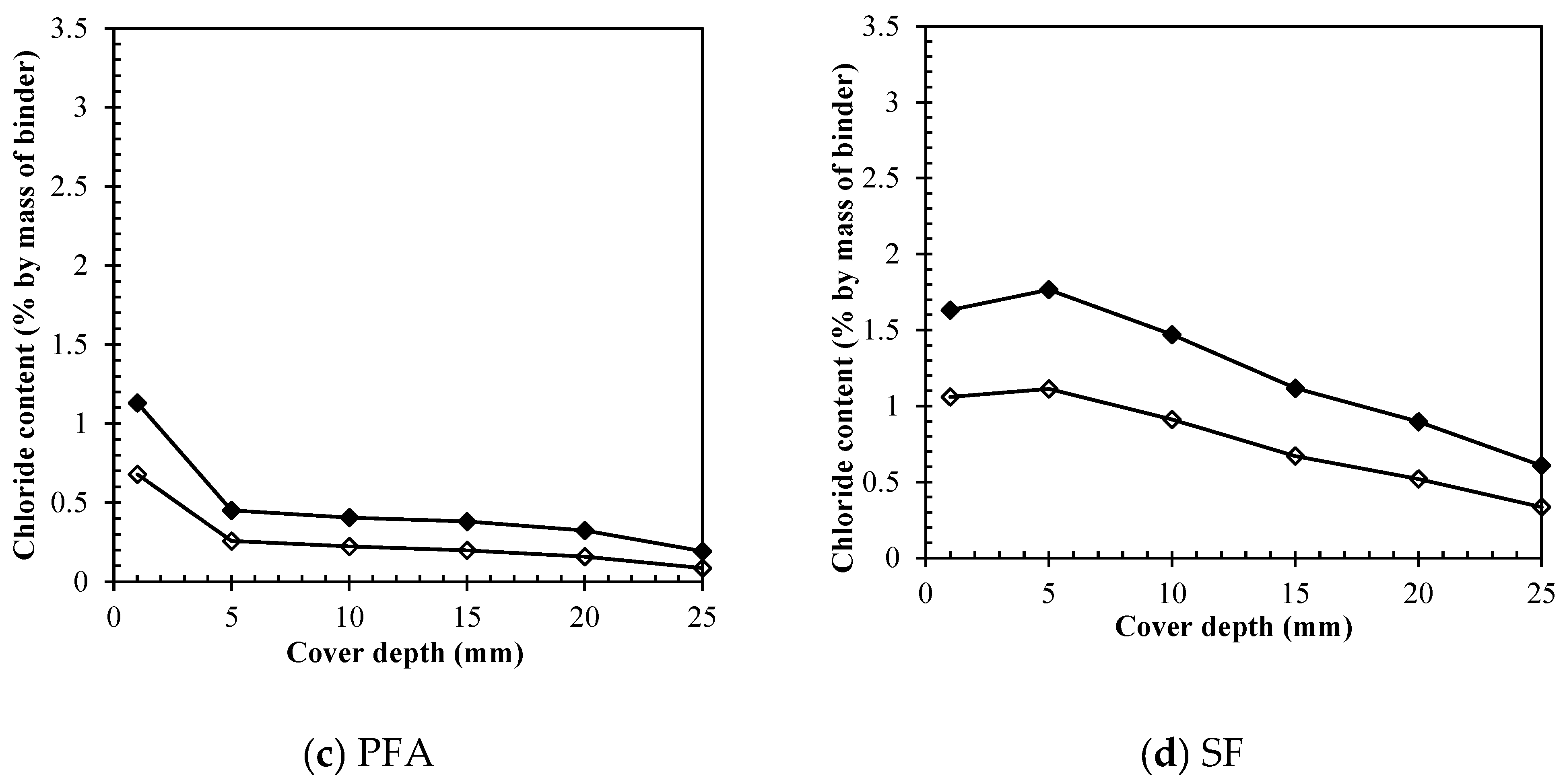
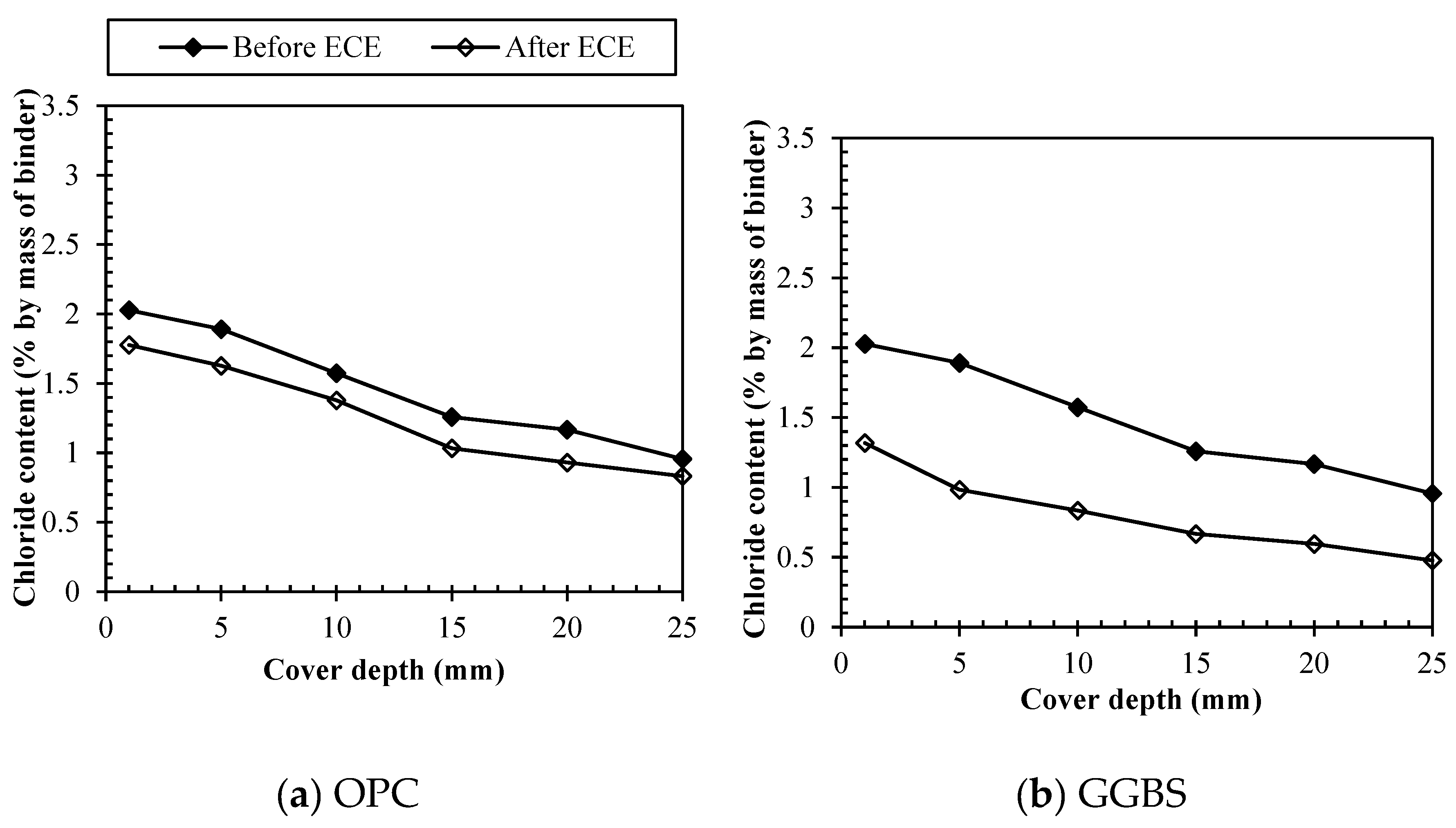
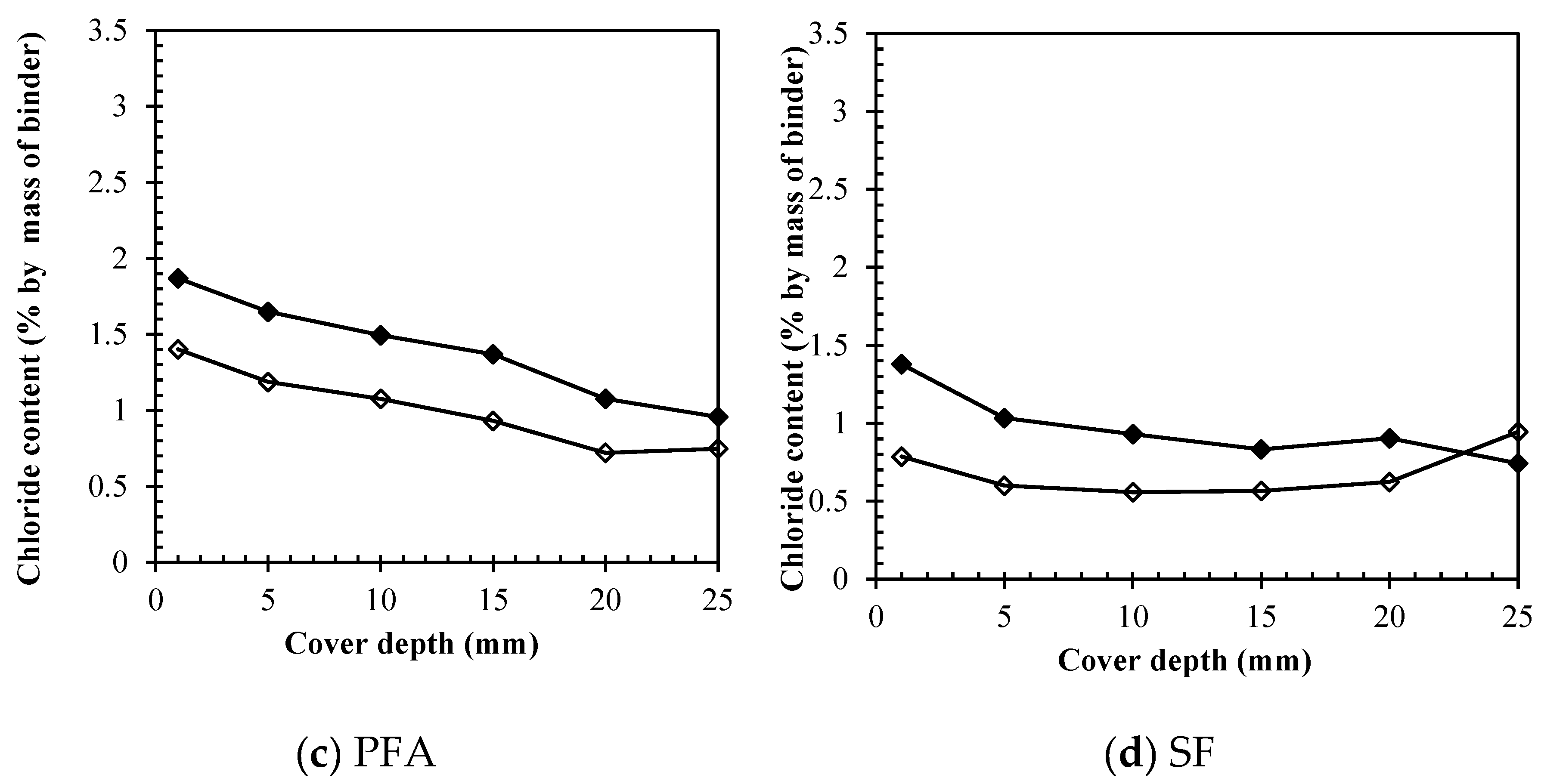
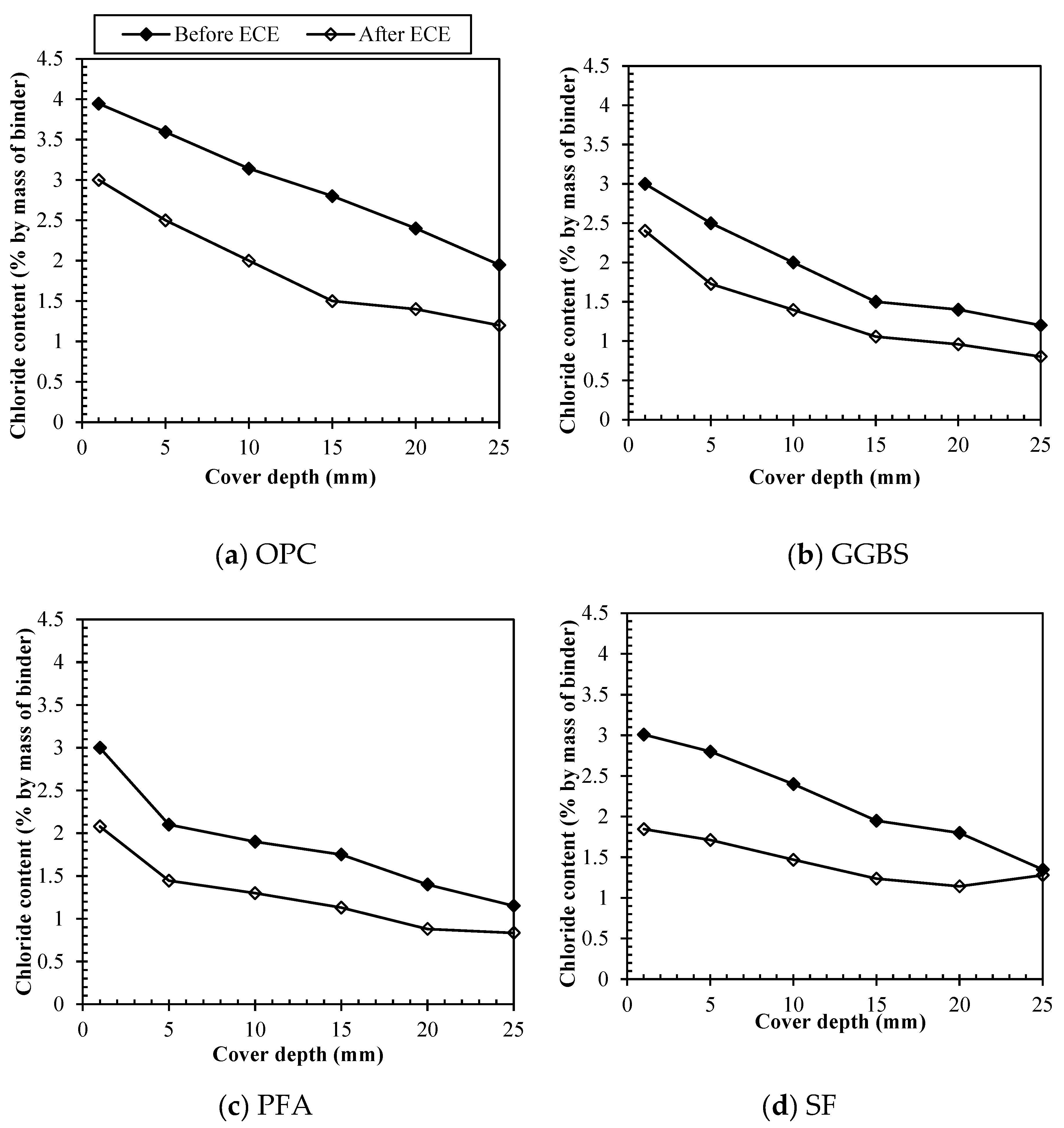
3.3. Removal of different chloride types by ECE
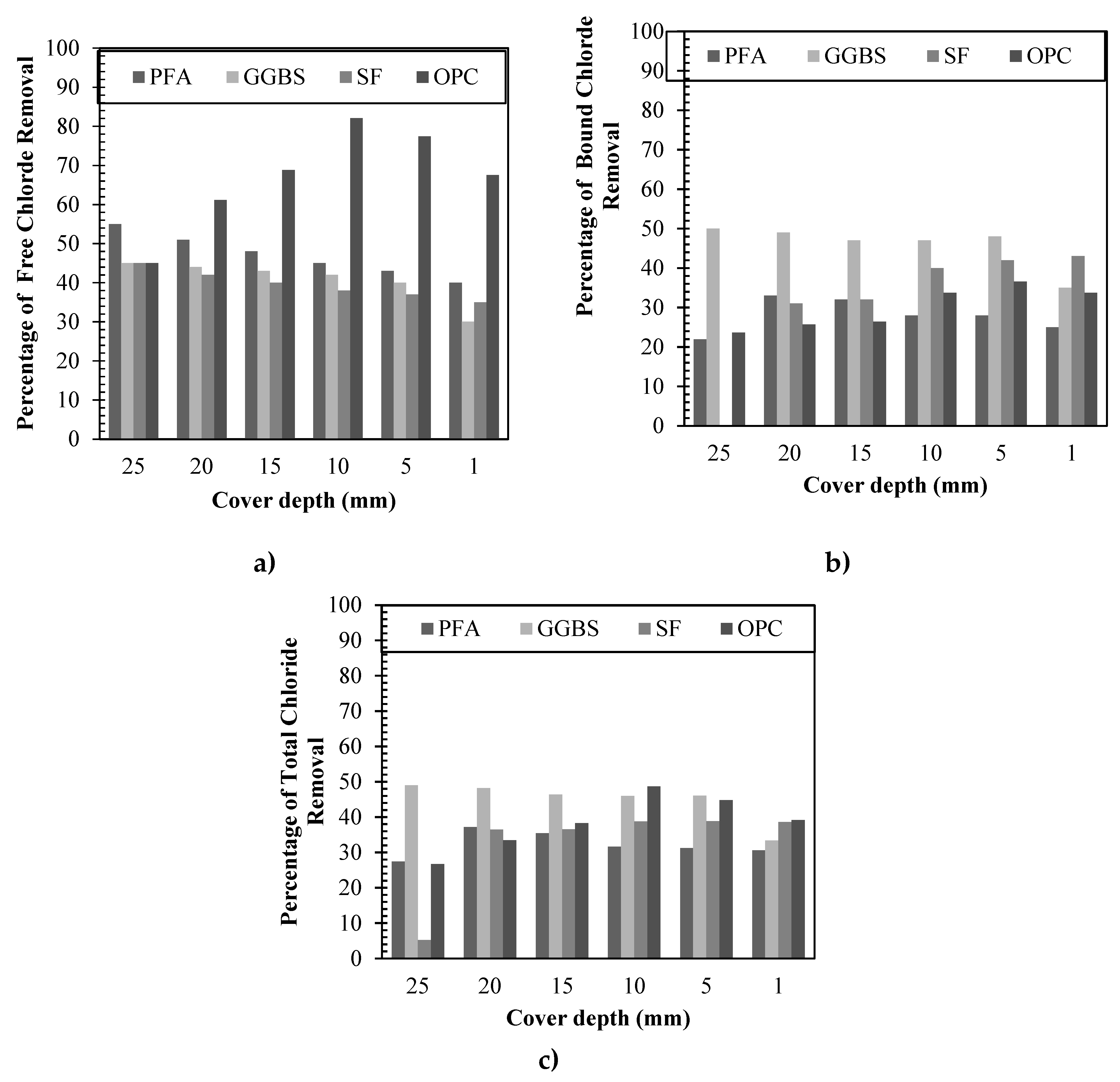
3.3.1. Free chlorides removal
3.3.2. Bound chlorides removal
3.3.3. Total chlorides removal
3.3.4. Direct comparison among binders
4. Discussions
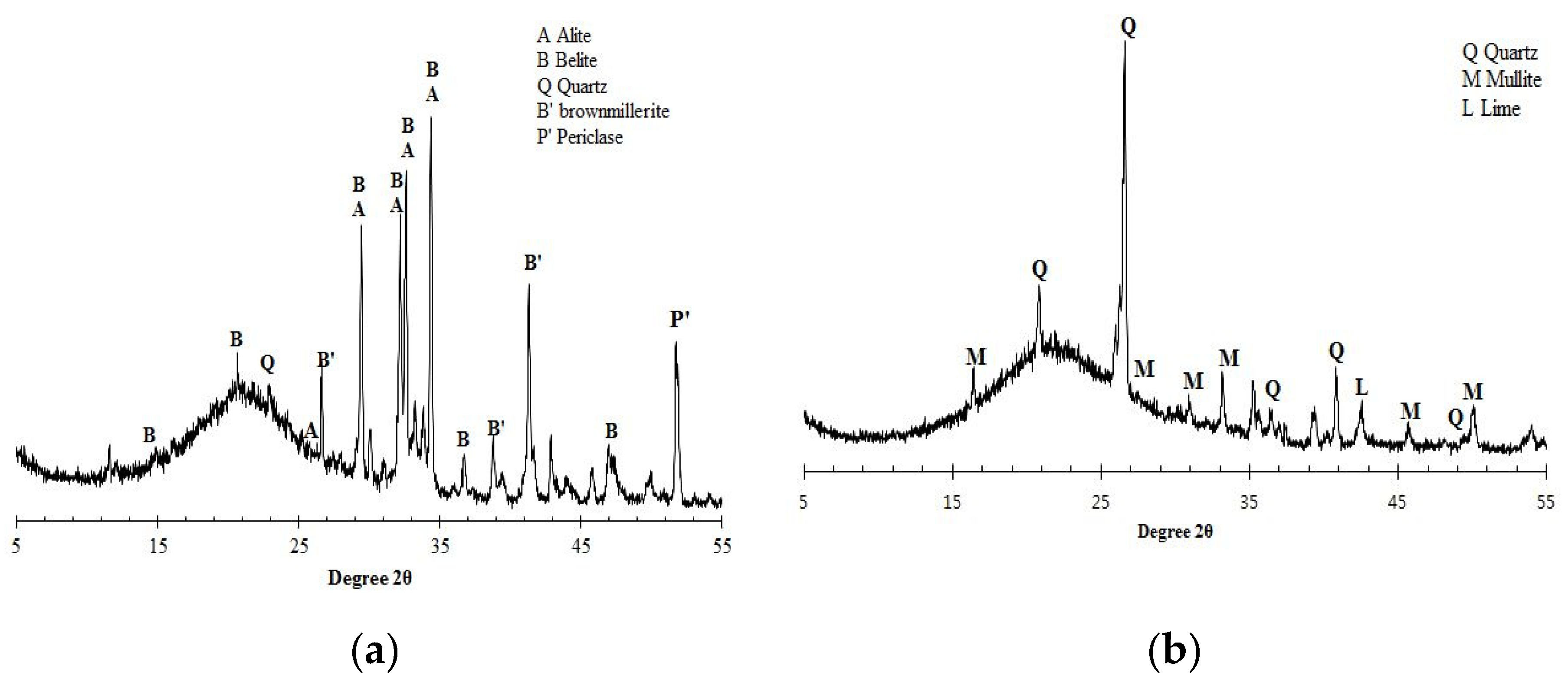
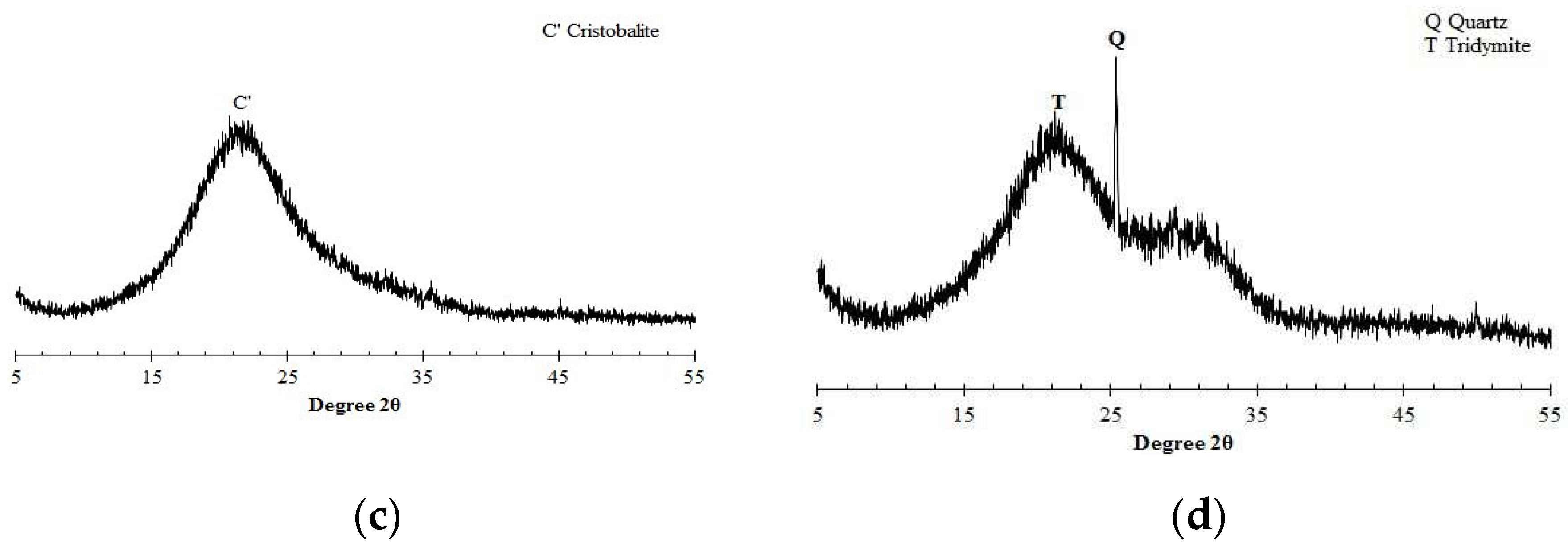
4.1. Influence of oxide composition of different binders
4.2. Influence of hydration products
5. Conclusion
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Puertas, F.; Martı́nez-Ramı́rez, S.; Alonso, S.; Vázquez, T. Alkali-activated fly ash/slag cements: strength behaviour and hydration products. Cement and concrete research 2000, 30, 1625–1632. [Google Scholar] [CrossRef]
- Arya, C.; Buenfeld, N.R.; Newman, J.B. Assessment of simple methods of determining the free chloride ion content of cement paste. Cement and Concrete Research 1987, 17, 907–918. [Google Scholar] [CrossRef]
- ASTM C 1202—Standard Test Method for Electrical Indication of Concrete's Ability to Resist Chloride Ion Penetration.; Annual Book of American Society for Testing Materials Standards, vol. C04.02. 2000.
- ASTM C39 / C39M-20; Standard Test Method for Compressive Strength of Cylindrical Concrete Specimens. ASTM International: West Conshohocken, PA, 2020. Available online: www.astm.
- Babu, K.G.; Kumar, V.S.R. Efficiency of GGBS in concrete. Cement and Concrete Research 2000, 30, 1031–1036. [Google Scholar] [CrossRef]
- Bonen, D.; Diamond, S. Occurrence of large silica fume-derived paticles in hydrated cement paste. Cement and Concrete Research 1992, 22, 1059–1066. [Google Scholar] [CrossRef]
- Alonzo, O.; Barringer, W.L.; Barton, S.G.; Bell, L.W.; Bennett, J.E.; Boyle, M.; Dixon, D.E. Guide for selecting proportions for high-strength concrete with portland cement and fly ash. ACI Mater J 1993, 90, 272–283. [Google Scholar]
- De Gutierrez, R.; Delvasto, S.; Talero, R. Permeability properties of cement mortars blended with silica fume, fly ash, and blast furnace slag. ASTM SPECIAL TECHNICAL PUBLICATION 2000, 1399, 190–196. [Google Scholar]
- Fauzi, A.; Nuruddin, M.F.; Malkawi, A.B.; Abdullah, M.M.A.B. Study of fly ash characterization as a cementitious material. Procedia Engineering 2016, 148, 487–493. [Google Scholar] [CrossRef]
- Glass, G.K.; Wang, Y.; Buenfeld, N.R. An investigation of experimental methods used to determine free and total chloride contents. Cement and concrete research 1996, 26, 1443–1449. [Google Scholar] [CrossRef]
- Ismail, M.; Muhammad, B. Electrochemical chloride extraction effect on blended cements. Advances in cement research 2011, 23, 241–248. [Google Scholar] [CrossRef]
- Kim, K.B.; Hwang, J.P.; Ann, K.Y. Influence of cementitious binder on chloride removal under electrochemical treatment in concrete. Construction and Building Materials 2016, 104, 191–197. [Google Scholar] [CrossRef]
- Lambert, P.; Page, C.L.; Short, N.R. Pore solution chemistry of the hydrated system tricalcium silicate/sodium chloride/water. Cement and Concrete Research 1985, 15, 675–680. [Google Scholar] [CrossRef]
- Lee, K.H.; Jung, Y.H.; Hwang, J.P.; Sim, J.S. Evaluation of Electrochemical Treatment of Chloride Contaminated Mortar Containing GGBS. Advances in Materials Science and Engineering. 2017. [CrossRef]
- Mehta, P.K.; Monteiro, P.J. Concrete: microstructure, properties, and materials. McGraw-Hill Education. 2014.
- Mohammed, T.U.; Hamada, H.; Hasnat, A.; Al Mamun, M.A. Corrosion of steel bars in concrete with the variation of microstructure of steel-concrete interface. Journal of Advanced Concrete Technology 2015, 13, 230–240. [Google Scholar] [CrossRef]
- Norton, B.; Bond, S.; Osmani, C.; Holmes, N. Influence of cement type on the efficiency of electrochemical chloride extraction. 2018. [Google Scholar]
- Pargar, F.; Koleva, D.A.; Van Breugel, K. Determination of chloride content in cementitious materials: from fundamental aspects to application of Ag/AgCl chloride sensors. Sensors 2017, 17, 2482. [Google Scholar] [CrossRef]
- Qureshi, L.A.; Ali, B.; Ali, A. Combined effects of supplementary cementitious materials (silica fume, GGBS, fly ash and rice husk ash) and steel fiber on the hardened properties of recycled aggregate concrete. Construction and Building Materials 2020, 263, 120636. [Google Scholar] [CrossRef]
- Ryou, J.S.; Ann, K.Y. Variation in the chloride threshold level for steel corrosion in concrete arising from different chloride sources. Magazine of Concrete Research 2008, 60, 177–187. [Google Scholar] [CrossRef]
- Siddique, R. Waste materials and by-products in concrete. Springer Science & Business Media. 2007.
- Song, S.; Jennings, H.M. Pore solution chemistry of alkali-activated ground granulated blast-furnace slag. Cement and concrete research 1999, 29, 159–170. [Google Scholar] [CrossRef]
- Song, S.; Sohn, D.; Jennings, H.M.; Mason, T.O. Hydration of alkali-activated ground granulated blast furnace slag. Journal of materials science 2000, 35, 249–257. [Google Scholar] [CrossRef]
- Stanish, K.D.; Hooton, R.D.; Thomas, M.D.A. Testing the chloride penetration resistance of concrete: a literature review. 1997.
- Stoop, B.T.; Polder, R.B. Redistribution of chloride after electrochemical chloride removal from reinforced concrete prisms. Heron 1999, 44. [Google Scholar]
- Sun, W.; Liu, J.; Yan, J.; Dai, Y. Study on the influence of chloride ions content on the sea sand concrete performance. American Journal of Civil Engineering 2016, 4, 50–54. [Google Scholar] [CrossRef]
- Wang, S.D.; Scrivener, K.L. Hydration products of alkali activated slag cement. Cement and Concrete Research 1995, 25, 561–571. [Google Scholar] [CrossRef]
- Yu, L.B.; Jiang, L.H.; Chu, H.Q.; Guo, M.Z.; Zhu, Z.Y.; Dong, H. Effect of electrochemical chloride removal and ground granulated blast furnace slag on the chloride binding of cement paste subjected to NaCl and Na2SO4 attack. Construction and Building Materials 2019, 220, 538–546. [Google Scholar] [CrossRef]
- Glass, G.K.; Buenfeld, N.R. The inhibitive effects of electrochemical treatment applied to steel in concrete. Corrosion Science 2000, 42, 923–927. [Google Scholar] [CrossRef]
- Tofeti Lima, T.; Ann, K.Y. Efficiency of Different Electrolytes on Electrochemical Chloride Extraction to Recover Concrete Structures under Chloride-Induced Corrosion. Advances in Materials Science and Engineering 2020. [CrossRef]
| Oxide Composition (%) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Binder | CaO | SiO2 | Al2O3 | Fe2O3 | MgO | SO3 | Ignition Loss (%) | Fineness (cm2/g) |
| OPC | 60.0 | 23.0 | 5.0 | 2.0 | 1.0 | 2.0 | 0.8 | 3120 |
| PFA | 4.22 | 55.0 | 21.1 | 10.9 | 1.2 | 0.06 | 5.0 | |
| GGBS | 33.5 | 44.2 | 14.0 | 0.8 | 4.9 | 1.4 | 0.1 | |
| SF | 0.54 | 94.9 | 0.27 | 0.8 | 0.9 | 0.8 | 1.0 | |
| Mix proportion | Binder (kg/m3) | Water(kg/m3) | Sand(kg/m3) | Gravel(kg/m3) | |
|---|---|---|---|---|---|
| 100% OPC | OPC | 400 | 180 | 1056 | 704 |
| GGBS 65% | OPC GGBS |
140 260 |
178.8 | 1049.1 | 699.4 |
| PFA 30% | OPC PFA |
280 120 |
178.9 | 1049.4 | 699.6 |
| SF 10% | OPC SF |
360 40 |
179 | 1050.2 | 700.1 |
| Mix type | Water | Binder | Sand | Gravel | Chloride source | Experiments applied | |||
|---|---|---|---|---|---|---|---|---|---|
| Paste | 0.45 | OPC | GGBS | SF | PFA | - | - | - | XRD |
| 1.0 | - | - | - | ||||||
| 0.35 | 0.65 | - | - | ||||||
| 0.7 | - | - | 0.3 | ||||||
| 0.9 | - | 0.1 | - | ||||||
| Mortar | 0.45 | Same for all | 2.64 | - | In cast 3%Immersion | ECE Chloride Profile Chloride Profile |
|||
| Concrete | 0.45 | Same for all | 2.64 | 3.12 | - | Compressive strength RCPT |
|||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




