Submitted:
04 January 2023
Posted:
05 January 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. MIT Function Is Essential in Arabidopsis
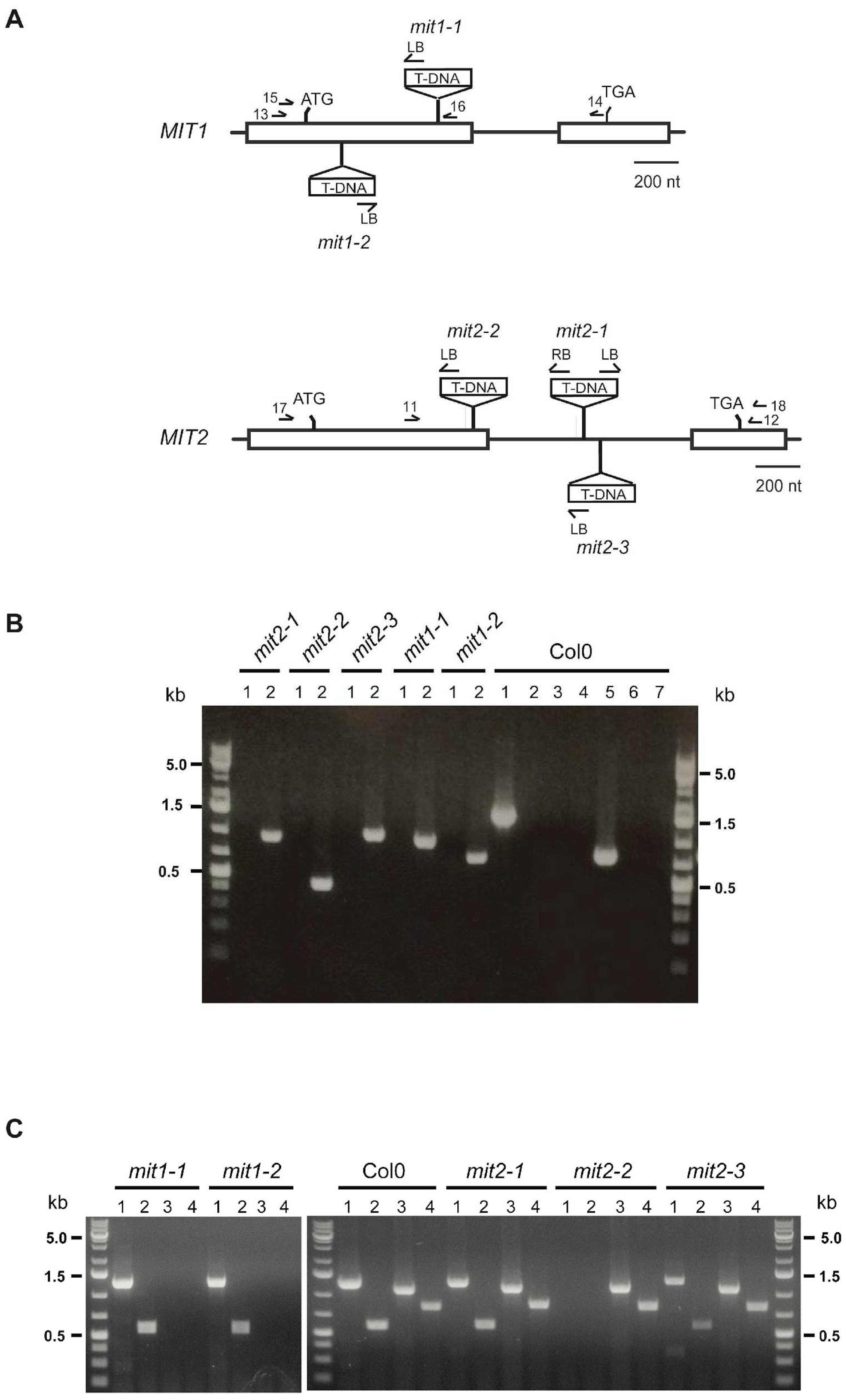
2.2. Isolation of mit1-1 x mit2-1, a MIT Knockdown Mutant
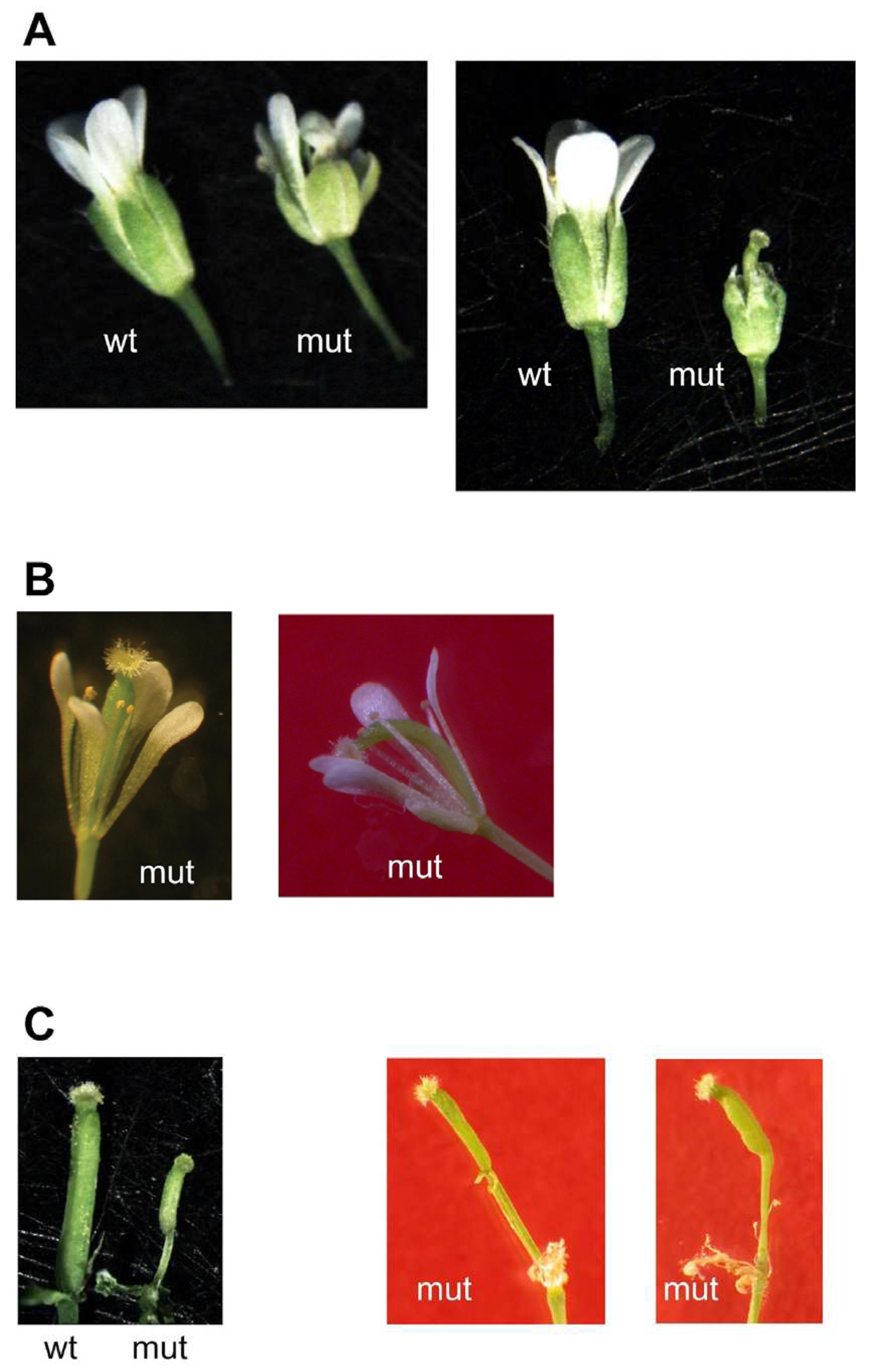
2.3. Growth of Double Homozygous Mutant Plants Is Severely Affected
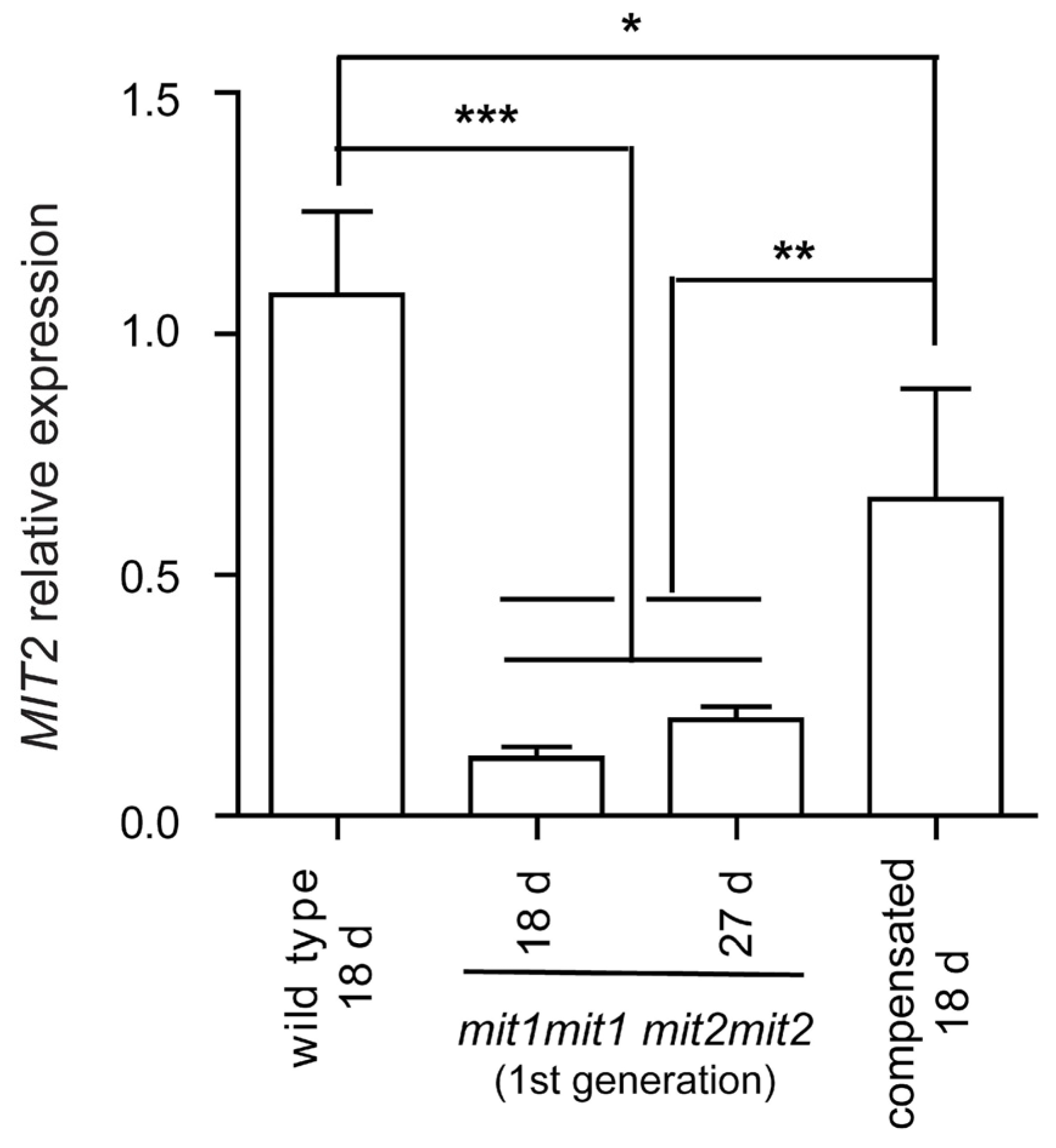
2.4. The Next Generation of Double Homozygous mit1-1 mit2-1 Mutant Plants Showed a Normal Phenotype
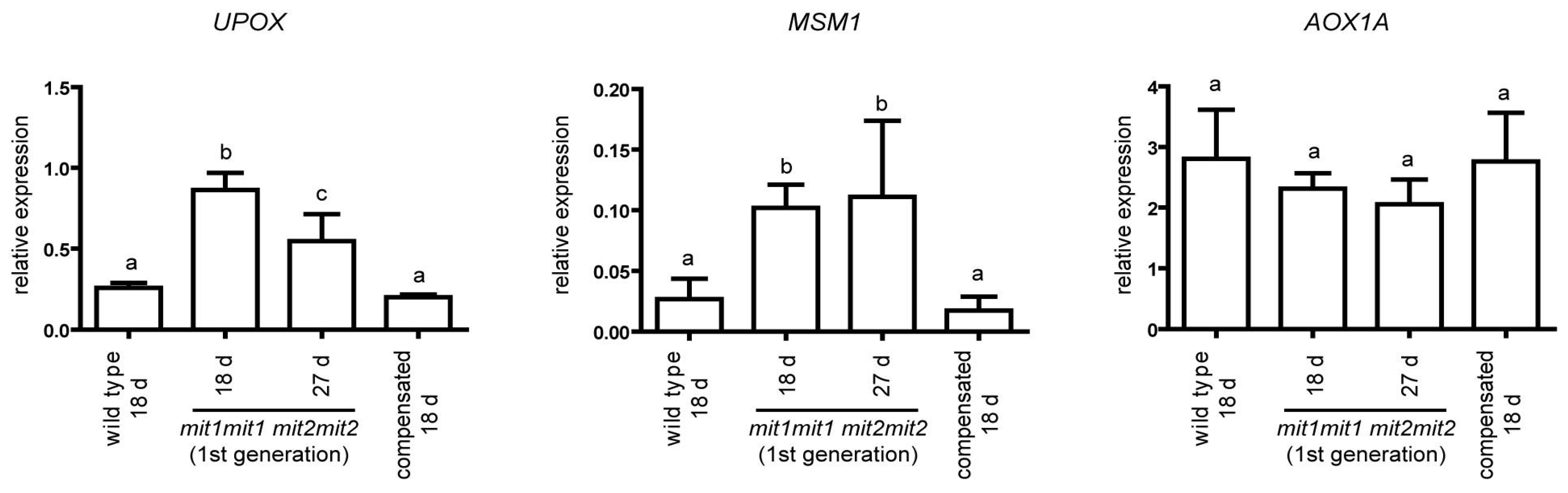
2.5. Analysis of Mitochondrial Function
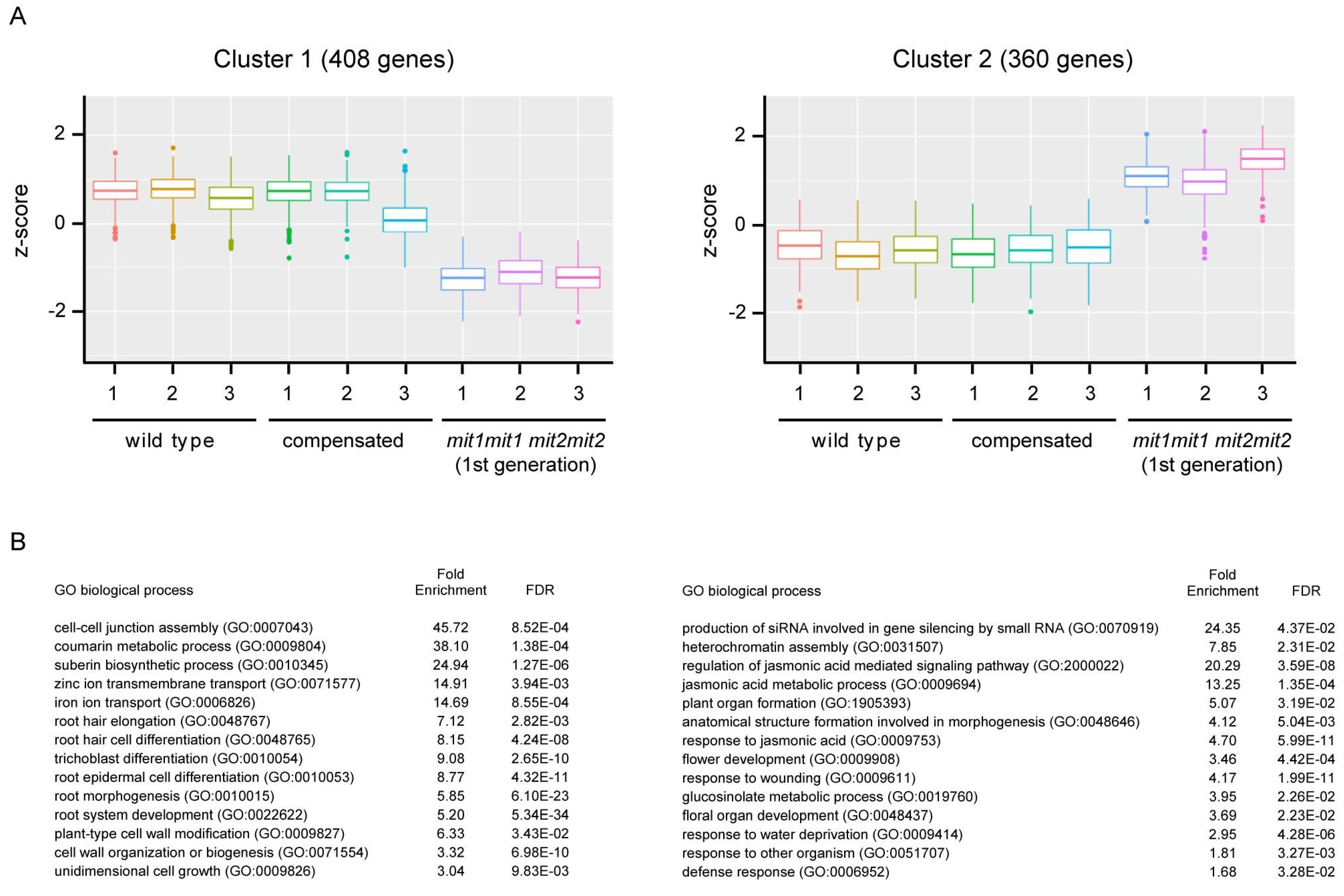
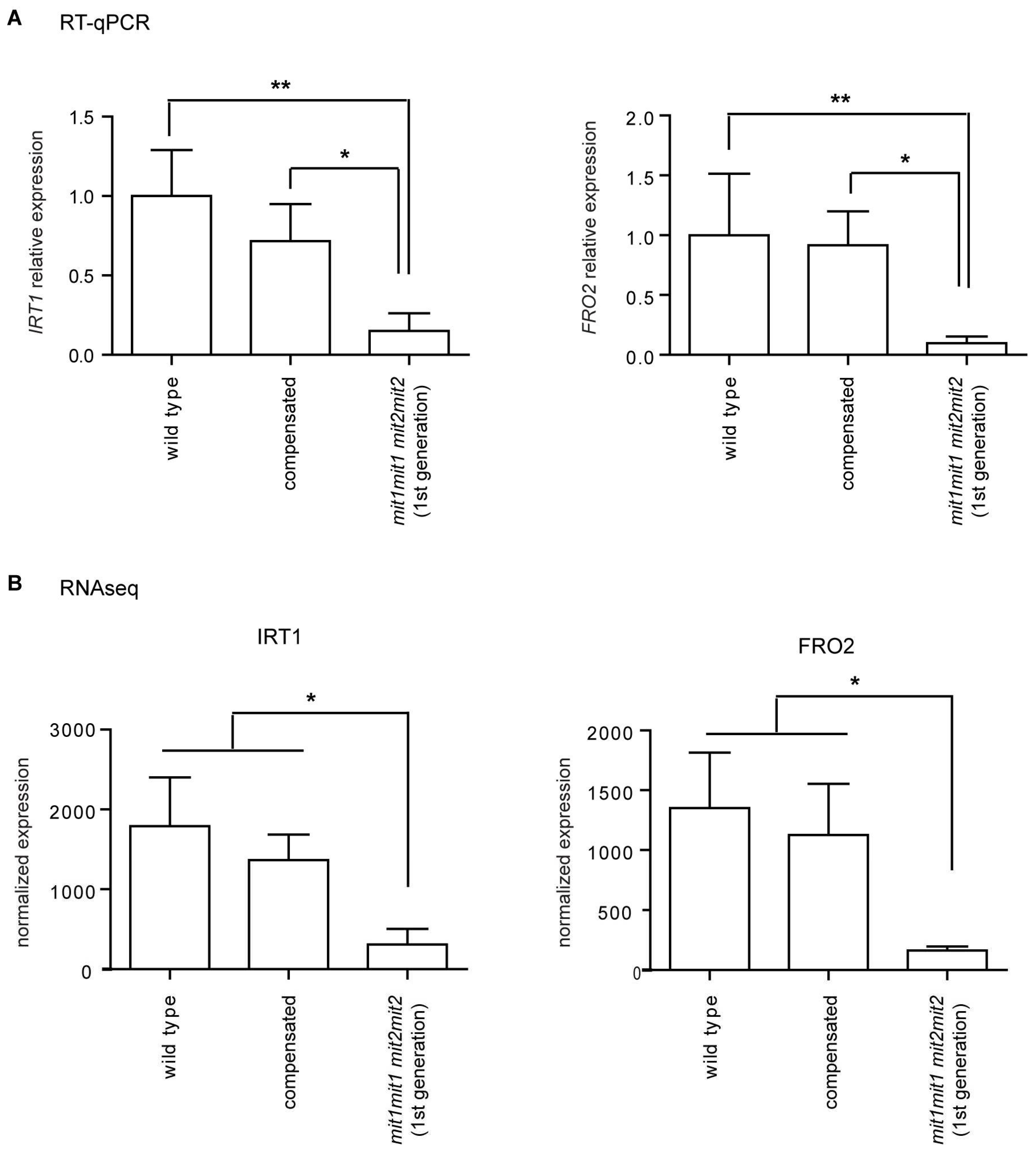
2.6. RNA-seq Analysis of Gene Expression in Double Homozygous Mutant Plants
3. Discussion
3.1. Phenotypic Alterations of the mit1-1 mit2-1 Double Homozygous Mutants
3.2. Phenotypic Recovery of Double Homozygous mit1-1 mit2-1 in Next Generations
3.3. Transcriptome of Double Homozygous mit1-1 mit2-1 Mutant Plants
4. Materials and Methods
4.1. Plant Material and Growth Conditions
4.2. Complementation of Mutant Δmrs3Δmrs4 Yeast Cells
4.3. Expression Analysis by RT-PCR and RT-qPCR
4.4. Transcriptome Analysis by RNA Sequencing
Supplementary Materials
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Aida, M. , Vernoux, X., Furutani, M., Traas, J. and Tsaka, M. Roles. of PIN-FORMED1 and MONOPTEROS in pattern formation of the apical region of the Arabidopsis embryo. Development 2002, 129, 3965–3974. [Google Scholar] [CrossRef]
- Anders, S. and Huber, W. Differential. expression analysis for sequence count data. Genome Biol. 2010, 11, R106. [Google Scholar] [CrossRef]
- Aung, M. S. and Masuda, H. (2020). How does rice defend against excess iron?: Physiological and molecular mechanisms. Front. Plant Sci. 2020, 11, 1102. [Google Scholar] [CrossRef]
- Bashir, K. , Ishimaru, Y., Shimo, H., Nagasaka, S., Fujimoto, M., Takanashi, H., Tsutsumi, N., An, G. and Nishizawa, N. K. The. rice mitochondrial iron transporter is essential for plant growth. Nature Commun 2011, 2, 322. [Google Scholar] [CrossRef] [PubMed]
- Benjamins, R. , Quint, A., Weijers, D., Hooykaas, P. and Offringa, R. The. PINOID protein kinase regulates organ development in Arabidopsis by enhancing polar auxin transport. Development 2001, 128, 4057–4067. [Google Scholar] [CrossRef]
- Benková, E. , Michniewicz, M., Sauer, M., Teichmann, T., Seifertová, D., Jürgens, G. and Friml, J. Local,. efflux-dependent auxin gradients as a common module for plant organ formation. Cell 2003, 115, 591–602. [Google Scholar] [CrossRef] [PubMed]
- Bennett, S. R. M. , Alvarez, J., Bossinger, G. and Smith, D.R. Morphogenesis. in pinoid mutants of Arabidopsis thaliana. Plant J. 1995, 8, 505–520. [Google Scholar] [CrossRef]
- Bolger, A. M. , Lohse, M. and Usadel, B. Trimmomatic:. a flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Boyes, D.C. , Zayed, A.M., Ascenzi, R., McCaskill, A.J., Hoffman, N.E., Davis, K.R., et al. Growth. stage-based phenotypic analysis of Arabidopsis: a model for hgh throughput functional genomics in plants. Plant Cell 2001, 13, 1499–1510. [Google Scholar]
- Briat, J.F. Briat, J.F., Dubos, C., Gaymard, F. Iron nutrition, biomass production, and plant product quality. TRENDS in Plant Sci. 2015, 20: 33-40.
- Chang, N. , Sun, Q., Hu, J., An, C. and Gao, H. Large. introns of 5 to 10 kilo base pairs can be spliced out in Arabidopsis. Genes 2017, 8, 200. [Google Scholar] [CrossRef]
- Cheng, C. Y. , Krishnakumar, V., Chan, A. P., Thibaud-Nissen, F., Schobel, S. and Town, C. D. Araport11:. a complete reannotation of the Arabidopsis thaliana reference genome. Plant J. 2017, 89, 789–804. [Google Scholar] [CrossRef] [PubMed]
- Christensen, S. K. , Dagenais, N., Chory, J. and Weigel, D. Regulation. of auxin response by the protein kinase PINOID. Cell 2000, 100, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Clifton, R. , Lister, R., Parker, K. L, Sappl, P. G., Elhafez, D., Millar, A. H., Day, D. A. and Whelan, J. Stress-induced. co-expression of alternative respiratory chain components in Arabidopsis thaliana. Plant. Mol. Biol. 2005, 58, 193–212. [Google Scholar] [CrossRef] [PubMed]
- Collins, J. , Anderson G. Physiology. of the Gastrointestinal Tract (Fifth Edition) 2012, Elsevier, New York.
- Curie, C. , Cassin, G., Couch, D., Divol, F., Higuchi, K., Le Jean, M., Misson, J., Schikora, A., Czernic, P., Mari, S. Metal movement within the plant: contribution of nicotianamine and yellow stripe 1-like transporters. Annals of botany 2009, 103, 1–11. [Google Scholar] [CrossRef] [PubMed]
- De Longevialle, A. F. , Meyer, E. H., Andrés, C., Taylor, N. L., Lurin, C., Millar, A. H. and Small, I. D. The. pentatricopeptide repeat gene OTP43 is required for trans-splicing of the mitochondrial nad1 intron 1 in Arabidopsis thaliana. Plant Cell 2007, 19, 3256–3265. [Google Scholar] [CrossRef] [PubMed]
- 18. Divol F, Couch D, Conéjéro G, Roschzttardtz H, Mari S, Curie C. The. Arabidopsis YELLOW STRIPE LIKE4 and 6 transporters control iron release from the chloroplast. Plant Cell. 25(3):1040-55.
- Duy, D. , Wanner, G., Meda, A.R., Wiren, N. von, Soll, J. Philippar, K. PIC1, an Ancient Permease in Arabidopsis Chloroplasts, Mediates Iron Transport. Plant Cell 2007, 19, 986–1006. [Google Scholar] [CrossRef] [PubMed]
- Eide, D. , Broderius, M., Fett, J., Guerinot, M. L. A novel iron-regulated metal transporter from plants identified by functional expression in yeast. Proceedings of the National Academy of Sciences of the United States of America 1996, 93, 5624–5628. [Google Scholar] [CrossRef]
- Eubel, H. , Braun, H. P. and Millar, A. H. Blue-native. PAGE in plants: a tool in analysis of protein-protein interactions. Plant Methods 2005, 1, 11. [Google Scholar]
- Eubel, H. , Jänsch, L. and Braun, H. P. New. insights into the respiratory chain of plant mitochondria. Supercomplexes and a unique composition of complex II. Plant Physiol. 2003, 133, 274–286. [Google Scholar]
- Foury, F. and Roganti, T. Deletion. of the mitochondrial carrier genes MRS3 and MRS4 suppresses mitochondrial iron accumulation in a yeast frataxin-deficient strain. J. Biol. Chem. 2002, 277, 24475–24483. [Google Scholar]
- Furutani, M. , Vernoux, T., Traas, J., Kato, T., Tasaka, M. and Aida, M. PIN-FORMED1. and PINOID regulate boundary formation and cotyledon development in Arabidopsis embryogenesis. Development 2004, 131, 5021–5030. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y. and Zhao, Y. Epigenetic suppression of T-DNA insertion mutants in arabidopsis. Mol. Plant 2013, 6, 539–545. [Google Scholar] [CrossRef]
- Guerinot, M.L. , Yi Y. Iron:. nutritious, noxious, and not readily available. Plant Physiol. 1994, 104, 815–820. [Google Scholar]
- Gibson, D. G. , Benders, G. A., Axelrod, K. C., Zaveri, J., Algire, M. A., Moodie, M., Montague, M. G., Craig Venter, J., Smith, H. O. and Hutchison III, C. A. One-step. assembly in yeast of 25 overlapping DNA fragments to form a complete synthetic Mycoplasma genitalium genome. Proc. Nat. Acad. Sci. USA 2008, 105, 20404–20409. [Google Scholar]
- Gilroy, S. and Jones, D. L. Through. form to function: root hair development and nutrient uptake. Tr. Plant Sci. 2000, 5, 56–60. [Google Scholar] [CrossRef]
- Grierson, C. , Nielsen, E., Ketelaarc, T. and Schiefelbein, J. Root. Hairs. The Arabidopsis Book 2014, 12, e0172. [Google Scholar] [CrossRef]
- Haferkamp I, Schmitz-Esser S. (2012) The plant mitochondrial carrier family: functional and evolutionary aspects. Front Plant Sci. 8;3:2.
- Ho 2012, L. H. , Giraud, E., Uggalla, V., Lister, R., Clifton, R., Glen, A., Thirkettle-Watts, D., Van Aken, O. and Whelan, J. Identification. of regulatory pathways controlling gene expression of stress-responsive mitochondrial proteins in Arabidopsis. Plant Physiol. 2008, 147, 1858–1873. [Google Scholar]
- Ivanova, A. , Law, S. R., Narsai, R., Duncan, O., Lee, J-H., Zhang, B., Van Aken, O., Radomiljac, J. D., van der Merwe, M., Yi, K. K. and Whelan, J. A. functional relationship between auxin and mitochondrial retrograde signaling regulates Alternative Oxidase1a expression in Arabidopsis. Plant Physiol. 2014, 165, 1233–1254. [Google Scholar]
- Jain, A. , Dashner, Z. S. and Connolly, E. L. Mitochondrial. iron transporters (MIT1 and MIT2) are essential for iron homeostasis and embryogenesis in Arabidopsis thaliana. Front. Plant Sci 2019, 10, 1449. [Google Scholar] [CrossRef]
- Jeong, J. , Cohu, C., Kerkeb, L., Pilon, M., Connolly, E.L. Guerinot, M. Lou Chloroplast. Fe(III) chelate reductase activity is essential for seedling viability under iron limiting conditions. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 10619–10624. [Google Scholar] [CrossRef]
- Kerchev, P. I. , De Clercq, I., Denecker, J., Mühlenbock, P., Kumpf, R., Nguyen, L., Audenaert, D., Dejonghe, W. and Van Breusegem, F. Mitochondrial. perturbation negatively affects auxin signaling. Mol. Plant 2014, 7, 1138–1150. [Google Scholar] [CrossRef] [PubMed]
- Kim, D. , Paggi, J. M., Park, C., Bennett, C. and Salzberg, S. L. Graph-based. genome alignment and genotyping with HISAT2 and HISAT-genotype. Nature Biotechnol. 2019, 37, 907–915. [Google Scholar] [CrossRef] [PubMed]
- 37. Kim LJ, Tsuyuki KM, Hu F, Park EY, Zhang J, Iraheta JG, Chia JC, Huang R, Tucker AE, Clyne M, Castellano C, Kim A, Chung DD, DaVeiga CT, Parsons EM, Vatamaniuk OK, Jeong J. Ferroportin. 3 is a dual-targeted mitochondrial/chloroplast iron exporter necessary for iron homeostasis in Arabidopsis. Plant J.
- Kobayashi, T. and Nishizawa, N. K. Iron uptake, translocation, and regulation in higher plants. Annu. Rev. Plant Biol. 2012, 63, 131–152. [Google Scholar] [CrossRef] [PubMed]
- Kolli, R. , Soll, J. and Carrie, C. OXA2b. is crucial for proper membrane insertion of COX2 during biogenesis of Complex IV in Plant Mitochondria. Plant Physiol. 2019, 179, 601–615. [Google Scholar] [CrossRef] [PubMed]
- Kühn, K. , Obata, T. , Feher, K., Bock, R., Fernie, A. R. and Meyer, E. H. Complete. mitochondrial complex I deficiency induces an up-regulation of respiratory fluxes that is abolished by traces of functional complex I. Plant Physiol. 2015, 168, 1537–1549. [Google Scholar] [PubMed]
- León, G. , Holuigue, L. and Jordana, X. Mitochondrial. complex II is essential for gametophyte development in Arabidopsis. Plant Physiol. 2007, 143, 1534–1546. [Google Scholar] [CrossRef] [PubMed]
- Li, L. and Kaplan, J. A. mitochondrial-vacuolar signaling pathway in yeast that affects iron and copper metabolism. J. Biol. Chem. 2004, 279, 33653–33661. [Google Scholar]
- Liao, Y. , Smyth G. , Shi W. The R package Rsubread is easier, faster, cheaper and better for alignment and quantification of RNA sequencing reads, Nucleic Acids Research 2019, 47, 8. [Google Scholar]
- Liu, C. , Xu, A. and Chua, N. Auxin. polar transport is essential for the establishment of bilateral simmetry during early plant embryogenesis. Plant Cell 1993, 5, 621–630. [Google Scholar] [CrossRef]
- Love, M.I. , Huber, W. & Anders, S. Moderated. estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 2014, 15, 550. [Google Scholar]
- Meyer, E. H. , Tomaz, T., Carroll, A. J., Estavillo, G., Delannoy, E., Tanz, S. K., Small, I. D., Pogson, B. J. and Millar, A. H. Remodeled. respiration in ndufs4 with low phosphorylation efficiency suppresses Arabidopsis germination and growth and alters control of metabolism at night. Plant Physiol. 2009, 151, 603–619. [Google Scholar] [PubMed]
- Mi, H. , Muruganujan, A., Casagrande, J. T. and Thomas, P. D. Large-scale. gene function analysis with the panther classification system. Nature Prot. 2013, 8, 1551–1566. [Google Scholar] [CrossRef]
- Mühlenhoff, U. , Stadler, J. A., Richhardt, N., Seubert, A., Eickhorst, T., Schweyen, R. J., Lill, R. and Wiesenberger, G. A. specific role of the yeast mitochondrial carriers Mrs3/4p in mitochondrial iron acquisition under iron-limiting conditions. J. Biol. Chem. 2003, 278, 40612–40620. [Google Scholar] [CrossRef] [PubMed]
- Müller, M. and Schmidt, W. Environmentally. induced plasticity of root hair development in Arabidopsis. Plant Physiol. 2004, 134, 409–419. [Google Scholar] [CrossRef]
- Ohbayashi, I. , Huang, S., Fukaki, H., Song, X., Sun, S., Morita, M. T., Tasaka, M., Millar, A. H. and Furutani, M. Mitochondrial. pyruvate dehydrogenase contributes to auxin-regulated organ development. Plant Physiol. 2019, 180, 896–909. [Google Scholar] [CrossRef] [PubMed]
- Okada, K. , Ueda, J., Komaki, M. K., Bell, C. J. and Shimura, Y. Requirement. of the auxin polar transport system in early stages of Arabidopsis floral bud formation. Plant Cell 1991, 3, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Oldenburg, K. R. , Vo, K. T., Michaelis, S. and Paddon, C. Recombination-mediated. PCR-directed plasmid construction in vivo in yeast. Nucleic Acids Res. 1997, 25, 451–452. [Google Scholar] [CrossRef] [PubMed]
- Osabe, K. , Harukawa, Y. , Miura, S. and Saze, H. Epigenetic Regulation of Intronic Transgenes in Arabidopsis. Sci. Rep. 2017, 7, 45166. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, G. , Pantopoulos K. Iron. metabolism and toxicity. Toxicol. Appl. Pharmacol. 2005, 202, 199–211. [Google Scholar] [CrossRef]
- Rajniak, J. , Giehl, R. F. H., Chang, E., Murgia, I., von Wirén, N. and Sattely, E. S. Biosynthesis. of redox-active metabolites in response to iron deficiency in plants. Nature Chem. Biol. 2018, 14, 442–450. [Google Scholar] [CrossRef]
- Restovic, F. , Espinoza-Corral, R., Gómez, I., Vicente-Carbajosa, J. and Jordana, X. An. active mitochondrial complex II present in mature seeds contains an embryo-specific iron–sulfur subunit regulated by ABA and bZIP53 and is involved in germination and seedling establishment. Front. Plant Sci. 2017, 8, 277. [Google Scholar] [CrossRef] [PubMed]
- Riaz, N. and Guerinot, M. L. All. together now: regulation of the iron deficiency response. J. Exp. Bot. 2021, 72, 2045–2055. [Google Scholar] [CrossRef] [PubMed]
- Robinson, N. J. , Procter, C. M., Connolly, E. L., Guerinot, M. L. A ferric-chelate reductase for iron uptake from soils. Nature 1999, 397, 694–697. [Google Scholar] [CrossRef] [PubMed]
- Roschzttardtz, H. , Fuentes, I., Vásquez, M., Corvalán, C., León, G., Gómez, I., Araya, A., Holuigue, L., Vicente-Carbajosa, J. and Jordana, X. A nuclear gene encoding the iron-sulfur subunit of mitochondrial complex II is regulated by B3 domain transcription factors during seed development in Arabidopsis. Plant Physiol. 2009, 150, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, K. S. , Koirala, P. S. and Neff, M. M. The. ben1-1 brassinosteroid-catabolism mutation is unstable due to epigenetic modifications of the intronic T-DNA insertion. G3 (Bethesda) 2013, 3, 1587–1595. [Google Scholar] [CrossRef] [PubMed]
- Santi, S. , Schmidt, W. Dissecting iron deficiency-induced proton extrusion in Arabidopsis roots. The New phytologist 2009, 183, 1072–1084. [Google Scholar] [PubMed]
- Schmidt SB, Eisenhut M, Schneider A. Chloroplast Transition Metal Regulation for Efficient Photosynthesis. Trends Plant Sci. 2020, 25, 817–828. [Google Scholar] [CrossRef]
- Tan YF, O’Toole N, Taylor NL, Millar AH Divalent. metal ions in plant mitochondria and their role in interactions with proteins and oxidative stress-induced damage to respiratory function. Plant Physiol. 2010, 152, 747–761.
- Tivendale ND, Belt K, Berkowitz O, Whelan J, Millar AH, Huang S. (2021) Knockdown of Succinate Dehydrogenase Assembly Factor 2 Induces Reactive Oxygen Species-Mediated Auxin Hypersensitivity Causing pH-dependent Root Elongation. Plant Cell Physiol. 62:1185-1198.
- Tivendale ND, Millar AH. (2022) How is auxin linked with cellular energy pathways to promote growth? New Phytol. 2022 233:2397-2404.
- Tsai, H. H. , Rodríguez-Celma, J. , Lan, P., Wu, Y. C., Vélez-Bermúdez, I. C. and Schmidt, W. Scopoletin. 8-hydroxylase-mediated fraxetin production is crucial for iron mobilization. Plant Physiol. 2018, 177, 194–207. [Google Scholar]
- Van Aken, O. , Zhang, B., Carrie, C., Uggalla, V., Paynter, E., Giraud, E. and Whelan, J. Defining. the mitochondrial stress response in Arabidopsis thaliana. Mol. Plant 2009, 2, 1310–1324. [Google Scholar] [CrossRef]
- Van Aken, O. and Whelan, J. Comparison. of transcriptional changes to chloroplast and mitochondrial perturbations reveals common and specific responses in Arabidopsis. Front. Plant Sci. 2012, 3, 281. [Google Scholar] [CrossRef] [PubMed]
- Vert, G. , Grotz, N., Dedaldechamp, F., Gaymard, F., Guerinot, M.L., Briat, J.F., Curie, C. IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell 2002, 14, 1223–1233. [Google Scholar] [CrossRef] [PubMed]
- Vigani, G. , Bashir, K., Ishimaru, Y., Lehmann, M., Casiraghi, F. M., Nakanishi, H., Seki, M., Geigenberger, P., Zocchi, G. and Nishizawa, N. K. Knocking. down mitochondrial iron transporter (MIT) reprogramsprimary and secondary metabolism in rice plants. J. Exp. Bot. 2016, 67, 1357–1368. [Google Scholar] [CrossRef] [PubMed]
- Vigani G, Maffi D, Zocchi G. Iron. availability affects the function of mitochondria in cucumber roots. New Phytol. 2009,182:127-136.
- Vigani G, Solti A, Thomine SB, Philippar K. Essential. and Detrimental - an Update on Intracellular Iron Trafficking and Homeostasis. Plant Cell Physiol. 2019, 60, 1420–1439. [Google Scholar] [CrossRef] [PubMed]
- Walter PB, Knutson MD, Paler-Martinez A, Lee S, Xu Y, Viteri FE, Ames BN. Iron. deficiency and iron excess damage mitochondria and mitochondrial DNA in rats. Proc Natl Acad Sci USA 2002, 99, 2264–2269. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y. , Lyu, W., Berkowitz, O., Radomiljac, J. D., Law, S. R., Murcha, M. W., Carrie, C., Teixera, P. F., Kmiec, B., Duncan, O., Van Aken, O., Narsai, R., Glaser, E., Huang, S., Roessner, U., Millar, A. H. and Whelan, J. Inactiavtion of complex I induces the expression of a twin cysteine protein that targets and affects cytosolic, chloroplastidic and mitochondrial function. Mol. Plant 2016, 9, 696–710. [Google Scholar] [PubMed]
- Xue, W. , Ruprecht, C., Street, N., Hematy, K., Chang, C., Frommer, W. B., Persson, S. and Niittylä, T. Paramutation-like. Interaction of T-DNA loci in Arabidopsis. PLoS ONE 2012, 7, e51651. [Google Scholar] [CrossRef]
- Yi, Y. , & Guerinot, M. L. Genetic evidence that induction of root Fe(III) chelate reductase activity is necessary foriron uptake under iron deficiency. The Plant journal: for cell and molecular biology 1996, 10, 835–844. [Google Scholar]
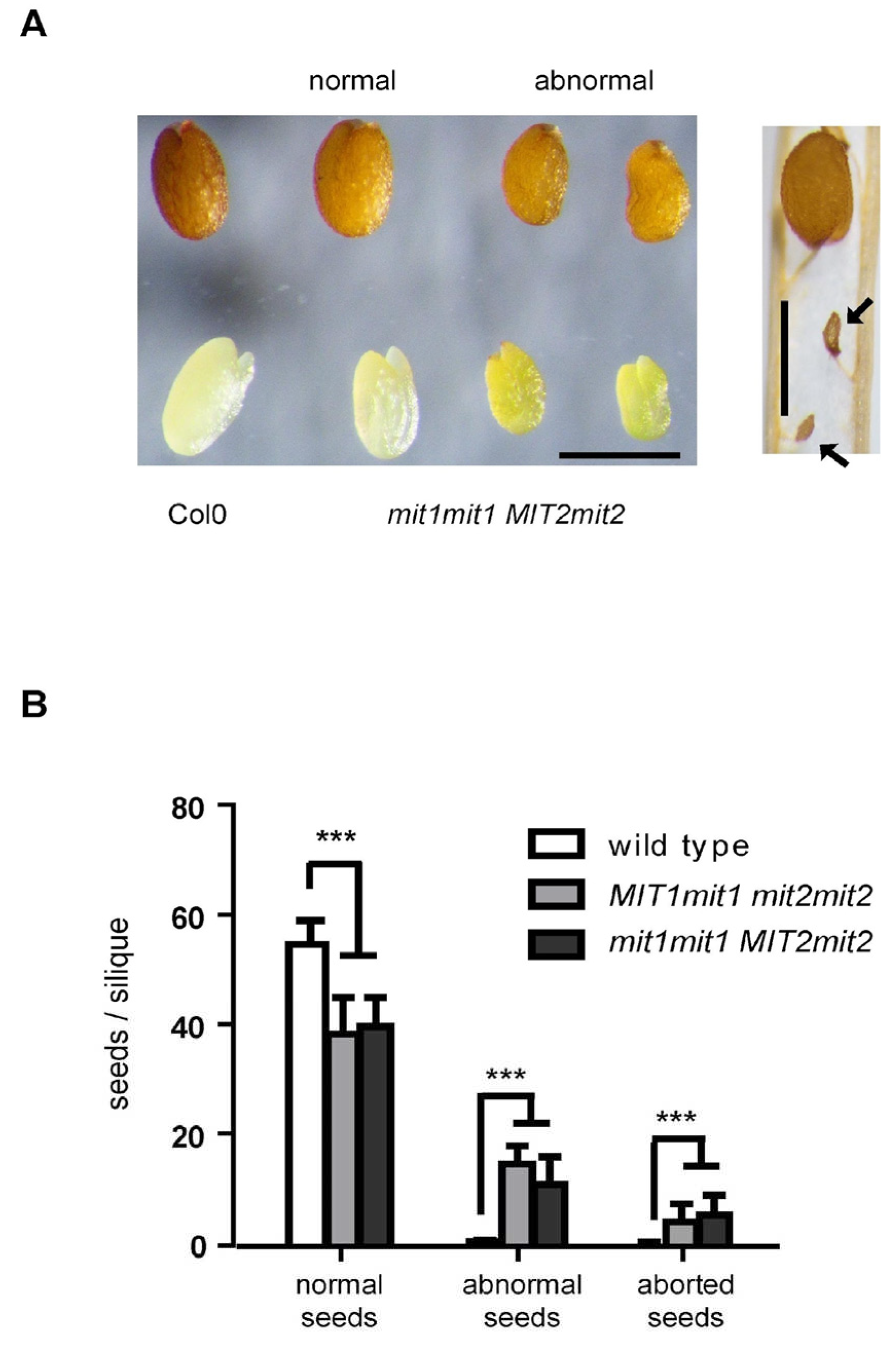

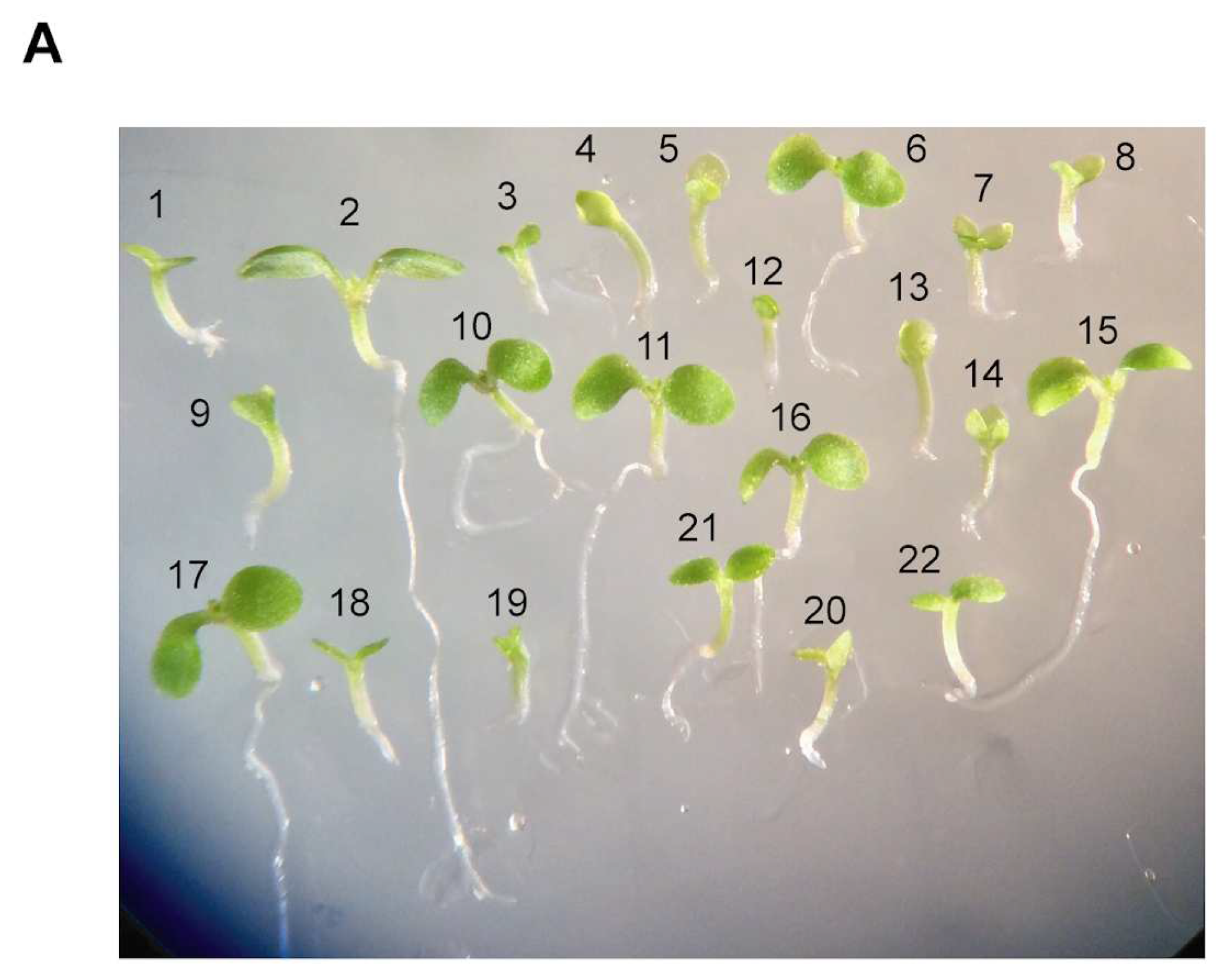
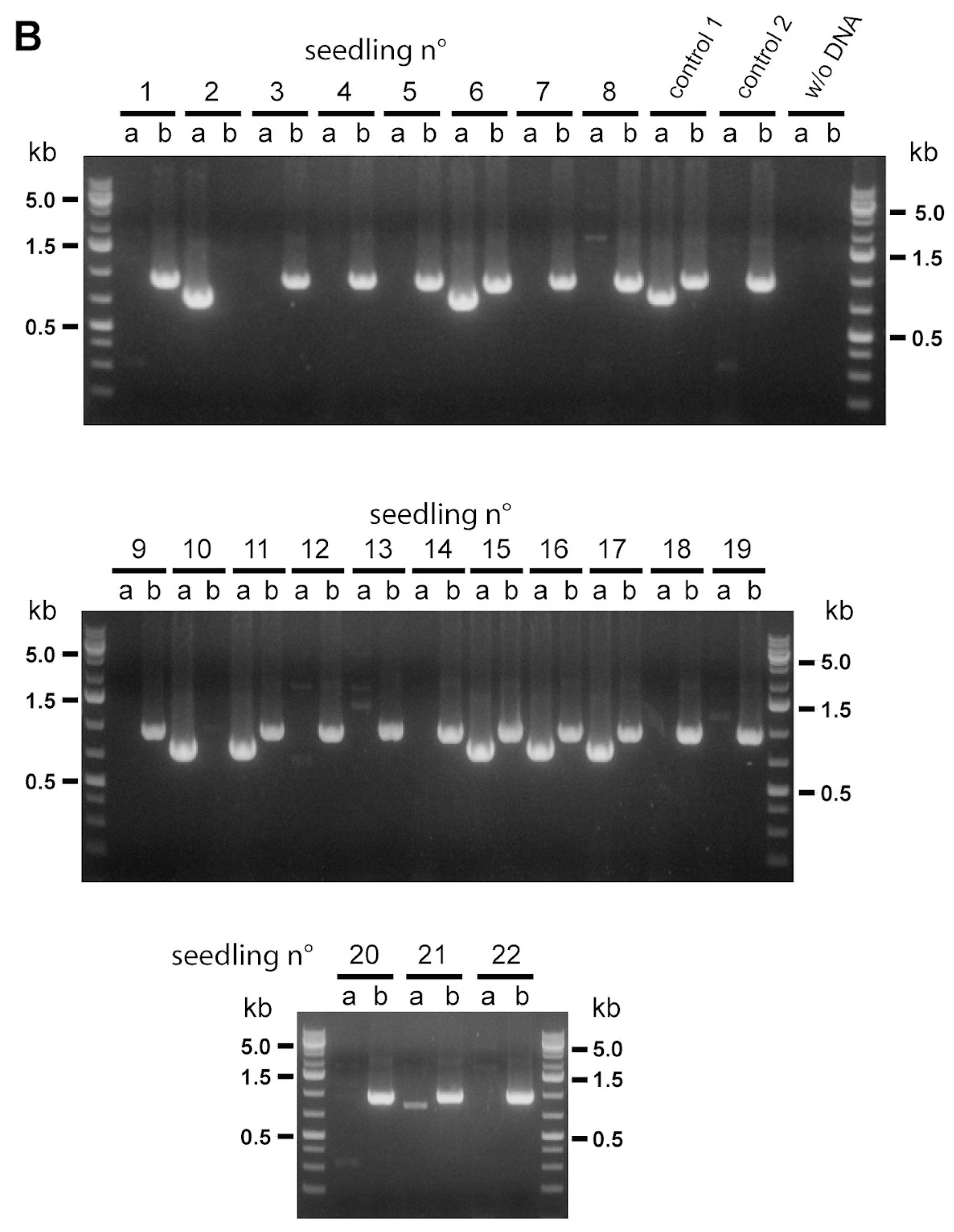


| “Abnormal” seeds from | ||
|---|---|---|
| MIT1mit1mit2mit2 | mit1mit1MIT2mit2 | |
| % germination | 94.4 ± 7.8 1 | 97.2 ± 2.6 2 |
| % establishment | 57.4 3 | 53.9 ± 11.9 4 |
| % 3-cotyledon embryos | 12.1 ± 9.3 5 | 14.8 ± 8.4 6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




